Safety and Efficacy of Nontuberculous Mycobacteria Treatment among Elderly Patients
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Study Population and Characteristics
3.2. Continuation of Treatment
3.3. Adverse Events Associated with Treatment
3.4. Efficacy
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Namkoong, H.; Kurashima, A.; Morimoto, K.; Hoshino, Y.; Hasegawa, N.; Ato, M.; Mitarai, S. Epidemiology of Pulmonary Nontuberculous Mycobacterial Disease, Japan1. Emerg. Infect. Dis. 2016, 22, 1116–1117. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; Iwai, K.; Uchimura, K.; Okumura, M.; Yoshiyama, T.; Yoshimori, K.; Ogata, H.; Kurashima, A.; Gemma, A.; Kudoh, S. A Steady Increase in Nontuberculous Mycobacteriosis Mortality and Estimated Prevalence in Japan. Ann. Am. Thorac. Soc. 2014, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Izumi, K.; Morimoto, K.; Hasegawa, N.; Uchimura, K.; Kawatsu, L.; Ato, M.; Mitarai, S. Epidemiology of Adults and Children Treated for Nontuberculous Mycobacterial Pulmonary Disease in Japan. Ann. Am. Thorac. Soc. 2018, 16, 341–347. [Google Scholar] [CrossRef] [PubMed]
- Morimoto, K.; Hasegawa, N.; Izumi, K.; Namkoong, H.; Uchimura, K.; Yoshiyama, T.; Hoshino, Y.; Kurashima, A.; Sokunaga, J.; Shibuya, S.; et al. A Laboratory-based Analysis of Nontuberculous Mycobacterial Lung Disease in Japan from 2012 to 2013. Ann. Am. Thorac. Soc. 2017, 14, 49–56. [Google Scholar] [CrossRef] [PubMed]
- Prevots, D.R.; Marras, T.K. Epidemiology of human pulmonary infection with nontuberculous mycobacteria: A review. Clin. Chest Med. 2014, 36, 13–34. [Google Scholar] [CrossRef] [PubMed]
- Gochi, M.; Takayanagi, N.; Kanauchi, T.; Ishiguro, T.; Yanagisawa, T.; Sugita, Y. Retrospective study of the predictors of mortality and radiographic deterioration in 782 patients with nodular/bronchiectatic Mycobacterium avium complex lung disease. BMJ Open 2015, 5, e008058. [Google Scholar] [CrossRef]
- Marras, T.K.; Vinnard, C.; Zhang, Q.; Hamilton, K.; Adjemian, J.; Eagle, G.; Zhang, R.; Chou, E.; Olivier, K.N. Relative risk of all-cause mortality in patients with nontuberculous mycobacterial lung disease in a US managed care population. Respir. Med. 2018, 145, 80–88. [Google Scholar] [CrossRef]
- Andréjak, C.; Thomsen, V.Ø.; Johansen, I.S.; Riis, A.H.; Benfield, T.; Duhaut, P.; Sørensen, H.T.; Lescure, F.-X.; Thomsen, R.W. Nontuberculous Pulmonary Mycobacteriosis in Denmark. Am. J. Respir. Crit. Care Med. 2010, 181, 514–521. [Google Scholar] [CrossRef]
- Haworth, C.S.; Banks, J.; Capstick, T.; Fisher, A.J.; Gorsuch, T.; Laurenson, I.F.; Leitch, A.; Loebinger, M.R.; Milburn, H.J.; Nightingale, M.; et al. British Thoracic Society guidelines for the management of non-tuberculous mycobacterial pulmonary disease (NTM-PD). Thorax 2017, 72, ii1–ii64. [Google Scholar] [CrossRef]
- Miwa, S.; Shirai, M.; Toyoshima, M.; Shirai, T.; Yasuda, K.; Yokomura, K.; Yamada, T.; Masuda, M.; Inui, N.; Chida, K.; et al. Efficacy of Clarithromycin and Ethambutol forMycobacterium aviumComplex Pulmonary Disease. A Preliminary Study. Ann. Am. Thorac. Soc. 2014, 11, 23–29. [Google Scholar] [CrossRef]
- Jeong, B.-H.; Jeon, K.; Park, H.Y.; Kim, S.-Y.; Lee, K.S.; Huh, H.J.; Ki, C.-S.; Lee, N.Y.; Shin, S.J.; Daley, C.L.; et al. Intermittent Antibiotic Therapy for Nodular BronchiectaticMycobacterium aviumComplex Lung Disease. Am. J. Respir. Crit. Care Med. 2015, 191, 96–103. [Google Scholar] [CrossRef] [PubMed]
- Evans, A.J.; Crisp, A.J.; Hubbard, R.B.; Colville, A.; Evans, S.A.; Johnston, I.D. Pulmonary Mycobacterium kansasii infection: Comparison of radiological appearances with pulmonary tuberculosis. Thorax 1996, 51, 1243–1247. [Google Scholar] [CrossRef] [PubMed]
- Santin, M.; Dorca, J.; Alcaide, F.; González, L.; Casas, S.; López, M.; Guerra, M.R. Long-term relapses after 12-month treatment for Mycobacterium kansasii lung disease. Eur. Respir. J. 2009, 33, 148–152. [Google Scholar] [CrossRef] [PubMed]
- Park, H.K.; Koh, W.-J.; Shim, T.S.; Kwon, O.J. Clinical Characteristics and Treatment Outcomes of Mycobacterium kansasiiLung Disease in Korea. Yonsei Med. J. 2010, 51, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Shitrit, D.; Baum, G.L.; Priess, R.; Lavy, A.; Shitrit, A.B.-G.; Raz, M.; Shlomi, D.; Daniele, B.; Kramer, M.R. Pulmonary Mycobacterium kansasii Infection in Israel, 1999–2004. Chest 2006, 129, 771–776. [Google Scholar] [CrossRef] [PubMed]
- Kobashi, Y.; Matsushima, T.; Oka, M. A double-blind randomized study of aminoglycoside infusion with combined therapy for pulmonary Mycobacterium avium complex disease. Respir. Med. 2007, 101, 130–138. [Google Scholar] [CrossRef]
- Griffith, D.E.; Brown-Elliott, B.A.; Wallace, J.R.J. Thrice-Weekly Clarithromycin-Containing Regimen for Treatment of Mycobacterium kansasii Lung Disease: Results of a Preliminary Study. Clin. Infect. Dis. 2003, 37, 1178–1182. [Google Scholar] [CrossRef]
- Campbell, A. Research First randomised trial of treatments for pulmonary disease caused by M avium intracellulare, M malmoense, and M xenopi in HIV negative patients: Rifampicin, ethambutol and isoniazid versus rifampicin and ethambutol. Thorax 2001, 56, 167–172. [Google Scholar] [CrossRef]
- Jenkins, A.; Campbell, P.A.; Banks, I.J.; Gelder, C.M.; Prescott, R.J.; Smith, A.P. Clarithromycin vs ciprofloxacin as adjuncts to rifampicin and ethambutol in treating opportunist mycobacterial lung diseases and an assessment of Mycobacterium vaccae immunotherapy. Thorax 2008, 63, 627–634. [Google Scholar] [CrossRef]
- Wallace, R.J.; Brown-Elliott, B.A.; McNulty, S.; Philley, J.V.; Killingley, J.; Wilson, R.W.; York, D.S.; Shepherd, S.; Griffith, D.E. Macrolide/Azalide therapy for nodular/bronchiectatic mycobacterium avium complex lung disease. Chest 2014, 146, 276–282. [Google Scholar] [CrossRef]
- Mycobacterium kansasii pulmonary infection: A prospective study of the results of nine months of treatment with rifampicin and ethambutol. Research Committee, British Thoracic Society. Thorax 1994, 49, 442–445. [CrossRef]
- Sauret, J.; Hernández-Flix, S.; Castro, E.; Hernández, L.; Ausina, V.; Coll, P. Treatment of pulmonary disease caused by Mycobacterium kansasii: Results of 18 vs 12 months’ chemotherapy. Tuber. Lung Dis. 1995, 76, 104–108. [Google Scholar] [CrossRef]
- Banks, J.; Hunter, A.M.; Campbell, I.A.; Jenkins, P.A.; Smith, A.P. Pulmonary infection with Mycobacterium kansasii in Wales, 1970–1979: Review of treatment and response. Thorax 1983, 38, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Brown-Elliott, B.A.; Langsjoen, B.; Zhang, Y.; Pan, X.; Girard, W.; Nelson, K.; Caccitolo, J.; Alvarez, J.; Shepherd, S.; et al. Clinical and Molecular Analysis of Macrolide Resistance inMycobacterium aviumComplex Lung Disease. Am. J. Respir. Crit. Care Med. 2006, 174, 928–934. [Google Scholar] [CrossRef]
- Griffith, D.E.; Aksamit, T.; Brown-Elliott, B.A.; Catanzaro, A.; Daley, C.; Gordin, F.; Holland, S.M.; Horsburgh, R.; Huitt, G.; Iademarco, M.F.; et al. An Official ATS/IDSA Statement: Diagnosis, Treatment, and Prevention of Nontuberculous Mycobacterial Diseases. Am. J. Respir. Crit. Care Med. 2007, 175, 367–416. [Google Scholar] [CrossRef] [PubMed]
- Daley, C.L.; Iaccarino, J.M.; Lange, C.; Cambau, E.; Wallace, R.J.; Andrejak, C.; Böttger, E.C.; Brozek, J.; Griffith, D.E.; Guglielmetti, L.; et al. Treatment of nontuberculous mycobacterial pulmonary disease: An official ATS/ERS/ESCMID/IDSA clinical practice guideline. Eur. Respir. J. 2020, 56, 2000535. [Google Scholar] [CrossRef]
- Jhun, B.W.; Moon, S.M.; Jeon, K.; Kwon, O.J.; Yoo, H.; Carriere, K.C.; Huh, H.J.; Lee, N.Y.; Shin, S.J.; Daley, C.L.; et al. Prognostic factors associated with long-term mortality in 1445 patients with nontuberculous mycobacterial pulmonary disease: A 15-year follow-up study. Eur. Respir. J. 2019, 55, 1900798. [Google Scholar] [CrossRef]
- Asakura, T.; Yamada, Y.; Suzuki, S.; Namkoong, H.; Okamori, S.; Kusumoto, T.; Niijima, Y.; Ozaki, A.; Hashimoto, M.; Yagi, K.; et al. Quantitative assessment of erector spinae muscles in patients with Mycobacterium avium complex lung disease. Respir. Med. 2018, 145, 66–72. [Google Scholar] [CrossRef]
- Horita, N.; Miyazawa, N.; Yoshiyama, T.; Kojima, R.; Omori, N.; Kaneko, T.; Ishigatsubo, Y. Poor performance status is a strong predictor for death in patients with smear-positive pulmonary TB admitted to two Japanese hospitals. Trans. R. Soc. Trop. Med. Hyg. 2013, 107, 451–456. [Google Scholar] [CrossRef]
- Horita, N.; Miyazawa, N.; Yoshiyama, T.; Tsukahara, T.; Takahashi, R.; Tsukiji, J.; Kato, H.; Kaneko, T.; Ishigatsubo, Y. Decreased activities of daily living is a strong risk factor for liver injury by anti-tuberculosis drugs. Respirology 2013, 18, 474–479. [Google Scholar] [CrossRef]
- Sinha, P.; Davis, J.; Saag, L.; Wanke, C.; Salgame, P.; Mesick, J.; Horsburgh, C.R.; Hochberg, N.S. Undernutrition and Tuberculosis: Public Health Implications. J. Infect. Dis. 2018, 219, 1356–1363. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.; Ito, Y.; Oguma, T.; Mio, T.; Niimi, A.; Hirai, T. Association between Mycobacterium avium complex lung disease and serum vitamin D status, antimicrobial peptide levels, and bone mineral density. Medicine 2018, 97, e12463. [Google Scholar] [CrossRef] [PubMed]
- Hirai, K.; Tanaka, A.; Homma, T.; Kaneko, K.; Uno, T.; Sato, H.; Manabe, R.; Ohta, S.; Kusumoto, S.; Yamaguchi, F.; et al. Comparison of three frailty models and a sarcopenia model in elderly patients with chronic obstructive pulmonary disease. Geriatr. Gerontol. Int. 2019, 19, 896–901. [Google Scholar] [CrossRef] [PubMed]
- Griffith, D.E.; Adjemian, J.; Brown-Elliott, B.A.; Philley, J.V.; Prevots, D.R.; Gaston, C.; Olivier, K.N.; Wallace, R.J. Semiquantitative Culture Analysis during Therapy forMycobacterium aviumComplex Lung Disease. Am. J. Respir. Crit. Care Med. 2015, 192, 754–760. [Google Scholar] [CrossRef] [PubMed]
- Diel, R.; Nienhaus, A.; Ringhausen, F.C.; Richter, E.; Welte, T.; Rabe, K.F.; Loddenkemper, R. Microbiologic Outcome of Interventions Against Mycobacterium avium Complex Pulmonary Disease. Chest 2018, 153, 888–921. [Google Scholar] [CrossRef]
- Baldwin, S.L.; Larsen, S.E.; Ordway, D.; Cassell, G.; Coler, R.N. The complexities and challenges of preventing and treating nontuberculous mycobacterial diseases. PLoS Negl. Trop. Dis. 2019, 13, e0007083. [Google Scholar] [CrossRef]
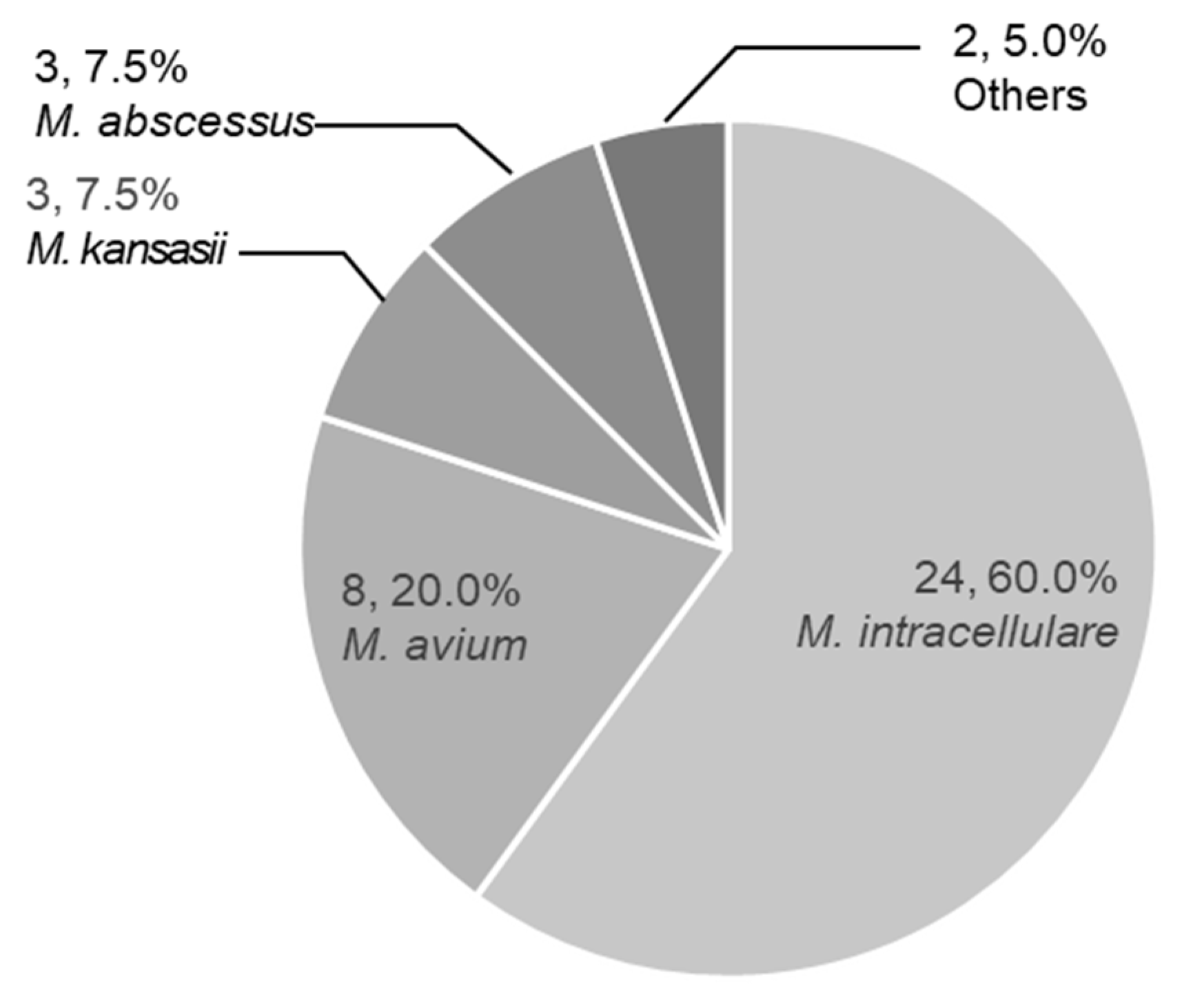
| Characteristics | Treated Elder n = 40 |
|---|---|
| Age (years old) | 79.1 (0.8) |
| Gender (female) | 26 (65.0) |
| BMI (kg/m2) | 20.0 (3.6) |
| Patient With Smoking History | 14 (41.2) |
| Patient With Habit of Alcohol | 8 (25.8) |
| Comorbidities | 24 (60.0) |
| Respiratory Disease | 18 (45.0) |
| Malignancy | 9 (22.5) |
| Heart Disease | 7 (17.5) |
| Patient with ADL Disability | 12 (30.0) |
| Symptoms | |
| Cough | 23 (57.5) |
| Sputum | 20 (50.0) |
| Hemoptysis | 10 (25.0) |
| Dyspnea | 2 (5.0) |
| Fever | 5 (12.5) |
| Weight Loss | 6 (15.0) |
| Radiological Type | |
| Fibrocavitary | 14 (35.0) |
| Nodular Bronchiectatic | 26 (65.0) |
| More Than Three Lobes Involved Based on CT | 22 (55.0) |
| Diagnosed by Bronchofiberscopy | 7 (17.5) |
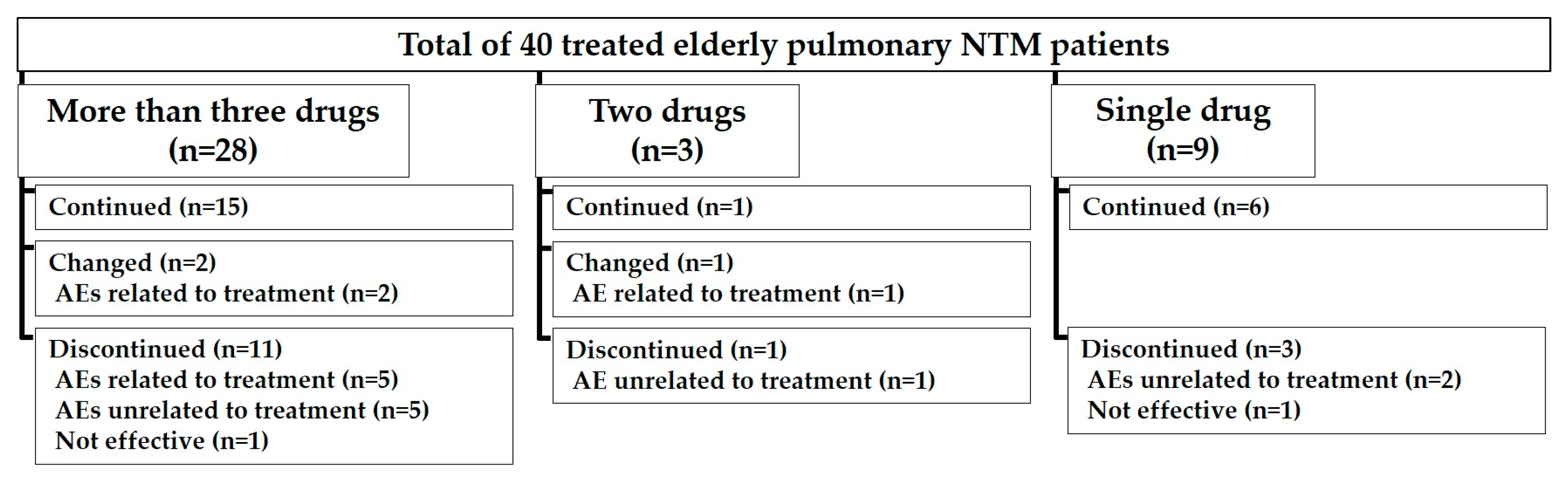
| Prescribed Number of Antimycobacterial Drugs | |
| Three or More Drugs | 28 (70.0) |
| Two Drugs | 3 (7.5) |
| Single Drug | 9 (22.5) |
| Use of Standard Regimens * | 24 (60.0) |
| Characteristics | Changed or Discontinued n = 18 (45.0%) | Continued n = 22 (55.0%) | p |
|---|---|---|---|
| Age (years old) | 80.2 (4.9) | 78.1 (4.7) | 0.169 |
| Gender (female) | 10 (55.6) | 16 (72.7) | 0.257 |
| BMI (kg/m2) | 18.8 (2.6) | 21.0 (4.1) | 0.097 |
| Patient With Smoking History | 7 (46.7) | 7 (36.8) | 0.563 |
| Patient With Habit of Alcohol | 4 (26.7) | 4 (18.2) | 0.916 |
| Comorbidities | |||
| Respiratory Disease | 10 (55.6) | 8 (36.4) | 0.225 |
| Malignancy | 5 (27.8) | 4 (18.2) | 0.705 |
| Impaired Immunity | 5 (27.8) | 2 (9.1) | 0.211 |
| Heart Disease | 6 (33.3) | 1 (4.6) | 0.033 |
| Patient With ADL Disability | 9 (50.0) | 3 (13.6) | 0.018 |
| Symptoms | |||
| Cough | 10 (55.6) | 13 (59.1) | 0.822 |
| Sputum | 9 (44.4) | 12 (54.6) | 0.525 |
| Hemoptysis | 3 (16.7) | 7 (31.8) | 0.465 |
| Dyspnea | 0 (0.0) | 2 (9.1) | 0.492 |
| Fever | 4 (22.2) | 1 (4.6) | 0.155 |
| Weight Loss | 3 (16.7) | 3 (13.) | 1.000 |
| Radiological Type | 0.641 | ||
| Fibrocavitary | 7 (38.9) | 7 (31.8) | |
| Nodular Bronchiectatic | 11 (61.1) | 15 (68.2) | |
| More Than Three Lobes Involved in CT | 11 (61.1) | 11 (50.0) | 0.482 |
| Diagnosed by Bronchofiberscopy | 4 (22.2) | 3 (13.6) | 0.680 |
| Prescribed Number of Antimycobacterial Drugs | 0.583 | ||
| Three or More Drugs | 13 (72.2) | 15 (68.2) | |
| Two Drugs | 2 (11.1) | 1 (4.6) | |
| Single Drug | 3 (16.7) | 6 (27.3) | |
| Standard Regimens * | 11 (61.1) | 13 (59.1) | 0.897 |
| Adverse Events Associated with Treatment | 11 (61.1) | 8 (36.4) | 0.203 |
| Characteristics | AE n = 19 (47.5%) | No AE n = 21 (52.5%) | p |
|---|---|---|---|
| Age (years old) | 77.7 (5.0) | 80.2 (4.5) | 0.116 |
| Gender (female) | 13 (68.4) | 13 (61.9) | 0.666 |
| BMI (kg/m2) | 19.5 (2.7) | 20.4 (4.3) | 0.341 |
| Patient With Smoking History | 5 (31.3) | 9 (50) | 0.315 |
| Patient With Habit of Alcohol | 3 (18.8) | 5 (33.3) | 0.433 |
| Comorbidities | |||
| Respiratory Disease | 8 (42.1) | 10 (47.6) | 0.761 |
| Malignancy | 2 (10.5) | 7 (33.3) | 0.133 |
| Impaired Immunity | 4 (21.1) | 3 (14.3) | 0.689 |
| Heart Disease | 4 (21.1) | 3 (14.3) | 0.574 |
| Patient With ADL Disability | 8 (42.1) | 4 (19.1) | 0.170 |
| Symptoms | |||
| Cough | 12 (63.2) | 11 (52.4) | 0.491 |
| Sputum | 10 (52.6) | 10 (47.6) | 0.752 |
| Hemoptysis | 6 (31.6) | 4 (19.1) | 0.473 |
| Dyspnea | 0 (0.0) | 2 (9.5) | 0.489 |
| Fever | 2 (10.5) | 3 (14.3) | 0.720 |
| Weight Loss | 3 (15.8) | 3 (14.3) | 1.000 |
| Radiological Type | 0.186 | ||
| Fibrocavitary | 9 (47.4) | 5 (23.8) | |
| Nodular Bronchiectatic | 10 (52.6) | 16 (76.2) | |
| More Than Three Lobes Involved in CT | 10 (52.6) | 12 (57.1) | 0.775 |
| Diagnosed by Bronchofiberscopy | 3 (15.8) | 4 (19.1) | 1.000 |
| Prescribed Number of Antimycobacterial Drugs | 0.258 | ||
| Three or More Drugs | 15 (79.0) | 13 (61.9) | |
| Two Drugs | 2 (10.5) | 1 (4.8) | |
| Single Drug | 2 (10.5) | 7 (33.3) | |
| Standard Regimens * | 13 (68.4) | 11 (52.4) | 0.349 |
| Characteristics | Sputum Culture Conversion Failed n = 12 (30.0%) | Sputum Culture Conversion Achieved n = 28 (70.0%) | p |
|---|---|---|---|
| Age (years old) | 79.4 (5.5) | 78.9 (4.7) | 0.756 |
| Gender (female) | 9 (75.0) | 17 (60.7) | 0.385 |
| BMI (kg/m2) | 18.3 (3.7) | 20.7 (3.4) | 0.212 |
| Patient With Smoking History | 4 (40.0) | 10 (41.7) | 1.000 |
| Patient With Habit of Alcohol | 1 (11.1) | 7 (31.8) | 0.379 |
| Comorbidities | |||
| Respiratory Disease | 6 (50.0) | 12 (31.8) | 0.173 |
| Malignancy | 2 (16.7) | 7 (25.0) | 0.697 |
| Impaired Immunity | 1 (8.3) | 6 (21.4) | 0.652 |
| Heart Disease | 3 (25.0) | 4 (14.3) | 0.410 |
| Patient With ADL Disability | 4 (33.3) | 8 (28.6) | 1.000 |
| Symptoms | |||
| Cough | 7 (58.3) | 16 (57.1) | 0.944 |
| Sputum | 6 (50.0) | 14 (50.0) | 1.000 |
| Hemoptysis | 6 (50.0) | 4 (14.3) | 0.041 |
| Dyspnea | 1 (8.3) | 1 (3.6) | 0.515 |
| Fever | 1 (8.3) | 4 (14.3) | 1.000 |
| Weight Loss | 2 (16.7) | 4 (14.3) | 1.000 |
| Radiological Type | 0.484 | ||
| Fibrocavitary | 3 (25.0) | 11 (39.3) | |
| Nodular Bronchiectatic | 9 (75.0) | 17 (60.7) | |
| More Than Three Lobes Involved in CT | 7 (58.3) | 15 (53.6) | 0.077 |
| Diagnosed by Bronchofiberscopy | 0 (0.0) | 7 (25.0) | 0.081 |
| Prescribed Number of Antimycobacterial Drugs | 0.116 | ||
| Three or More Drugs | 6 (50.0) | 22 (78.6) | |
| Two Drugs | 2 (16.7) | 1 (3.6) | |
| Single Drug | 4 (33.3) | 5 (17.9) | |
| Standard Regimens * | 3 (25.0) | 21 (75.0) | 0.005 |
| Characteristics | Unchanged or Deteriorated n = 17 (42.5%) | Improved n = 23 (57.5%) | p |
|---|---|---|---|
| Age (years old) | 78.5 (4.8) | 79.5 (5.0) | 0.556 |
| Gender (Female) | 14 (82.4) | 12 (52.2) | 0.092 |
| BMI (kg/m2) | 19.9 (4.7) | 20.1 (2.8) | 0.622 |
| Patient With Smoking History | 3 (17.6) | 11 (47.8) | 0.038 |
| Patient With Habit of Alcohol | 1 (5.9) | 7 (30.4) | 0.095 |
| Comorbidities | |||
| Respiratory Disease | 7 (41.2) | 11 (47.8) | 0.676 |
| Malignancy | 4 (23.5) | 5 (21.7) | 0.893 |
| Impaired Immunity | 3 (17.6) | 4 (17.4) | 1.000 |
| Heart Disease | 4 (23.5) | 3 (13.0) | 0.432 |
| Patient with ADL Disability | 7 (41.2) | 5 (21.7) | 0.296 |
| Symptoms | |||
| Cough | 12 (70.6) | 11 (47.8) | 0.202 |
| Sputum | 11 (64.7) | 9 (39.1) | 0.200 |
| Hemoptysis | 3 (17.6) | 7 (30.4) | 0.471 |
| Dyspnea | 1 (5.9) | 1 (4.4) | 1.000 |
| Fever | 2 (11.8) | 3 (13.0) | 1.000 |
| Weight Loss | 1 (5.9) | 5 (21.7) | 0.216 |
| Radiological Type | 0.041 | ||
| Fibrocavitary | 9 (52.9) | 5 (21.7) | |
| Nodular Bronchiectatic | 8 (47.1) | 18 (78.3) | |
| More Than Three Lobes Involved in CT | 11 (64.7) | 11 (47.8) | 0.289 |
| Diagnosed by Bronchofiberscopy | 2 (11.8) | 5 (21.7) | 0.677 |
| Prescribed Number of Antimycobacterial Drugs | 0.495 | ||
| Three or More Drugs | 10 (58.8) | 18 (78.3) | |
| Two Drugs | 2 (11.8) | 1 (4.4) | |
| Single Drug | 5 (29.4) | 4 (17.4) | |
| Standard Regimens * | 7 (41.2) | 17 (73.9) | 0.037 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Uchida, Y.; Terada, J.; Homma, T.; Mikuni, H.; Hirai, K.; Saito, H.; Honda, R.; Sagara, H. Safety and Efficacy of Nontuberculous Mycobacteria Treatment among Elderly Patients. Medicina 2020, 56, 517. https://doi.org/10.3390/medicina56100517
Uchida Y, Terada J, Homma T, Mikuni H, Hirai K, Saito H, Honda R, Sagara H. Safety and Efficacy of Nontuberculous Mycobacteria Treatment among Elderly Patients. Medicina. 2020; 56(10):517. https://doi.org/10.3390/medicina56100517
Chicago/Turabian StyleUchida, Yoshitaka, Jiro Terada, Tetsuya Homma, Hatsuko Mikuni, Kuniaki Hirai, Haruhisa Saito, Ryoichi Honda, and Hironori Sagara. 2020. "Safety and Efficacy of Nontuberculous Mycobacteria Treatment among Elderly Patients" Medicina 56, no. 10: 517. https://doi.org/10.3390/medicina56100517
APA StyleUchida, Y., Terada, J., Homma, T., Mikuni, H., Hirai, K., Saito, H., Honda, R., & Sagara, H. (2020). Safety and Efficacy of Nontuberculous Mycobacteria Treatment among Elderly Patients. Medicina, 56(10), 517. https://doi.org/10.3390/medicina56100517






