An Investigation of Oxidative Stress and Thiol/Disulphide Homeostasis in Graves’ Disease
Abstract
1. Introduction
2. Materials and Methods
2.1. Thiol/Disulphide Homeostasis Parameters
2.2. Statistical Analyses
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Menconi, F.; Marcocci, C.; Marinò, M. Diagnosis and classification of Graves’ disease. Autoimmun. Rev. 2014, 13, 398–402. [Google Scholar] [CrossRef] [PubMed]
- Alves, C.; Eidson, M.; Zakarija, M.; McKenzie, J. Graves disease presenting as painful thyroiditis. Eur. J. Pediatr. 1989, 148, 603–604. [Google Scholar] [CrossRef] [PubMed]
- Hubel, C.A. Oxidative stress in the pathogenesis of preeclampsia. Proc. Soc. Exp. Biol. Med. 1999, 222, 222–235. [Google Scholar] [CrossRef] [PubMed]
- Venditti, P.; Balestrieri, M.; Di Meo, S.; De Leo, T. Effect of thyroid state on lipid peroxidation, antioxidant defences, and susceptibility to oxidative stress in rat tissues. J. Endocrinol. 1997, 155, 151–157. [Google Scholar] [CrossRef] [PubMed]
- Finkel, T. Redox-dependent signal transduction. FEBS Lett. 2000, 476, 52–54. [Google Scholar] [CrossRef]
- Jones, D.P.; Liang, Y. Measuring the poise of thiol/disulfide couples in vivo. Free Radic. Biol. Med. 2009, 47, 1329–1338. [Google Scholar] [CrossRef] [PubMed]
- Turell, L.; Radi, R.; Alvarez, B. The thiol pool in human plasma: The central contribution of albumin to redox processes. Free Radic. Biol. Med. 2013, 65, 244–253. [Google Scholar] [CrossRef] [PubMed]
- Biswas, S.; Chida, A.S.; Rahman, I. Redox modifications of protein–thiols: Emerging roles in cell signaling. Biochem. Pharmacol. 2006, 71, 551–564. [Google Scholar] [CrossRef] [PubMed]
- Go, Y.-M.; Jones, D.P. Cysteine/cystine redox signaling in cardiovascular disease. Free Radic. Biol. Med. 2011, 50, 495–509. [Google Scholar] [CrossRef] [PubMed]
- Matteucci, E.; Giampietro, O. Thiol signalling network with an eye to diabetes. Molecules 2010, 15, 8890–8903. [Google Scholar] [CrossRef] [PubMed]
- Erel, O.; Neselioglu, S. A novel and automated assay for thiol/disulphide homeostasis. Clin. Biochem. 2014, 47, 326–332. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.-L.; Louie, S.; Cross, C.E.; Motchnik, P.; Halliwell, B. Antioxidant protection against hypochlorous acid in human plasma. J. Lab. Clin. Med. 1993, 121, 257–262. [Google Scholar]
- Pang, S.; Riddick, L. Hirsutism. In Pediatric Endocrinology, A Clinical Guide; Lifshitz, F., Ed.; Marcel Dekker, Inc.: New York, NY, USA, 1990. [Google Scholar]
- Costantini, F.; Pierdomenico, S.D.; Cesare, D.D.; De Remigis, P.; Bucciarelli, T.; Bittolo-Bon, G.; Cazzolato, G.; Nubile, G.; Guagnano, M.T.; Sensi, S. Effect of thyroid function on LDL oxidation. Arterioscler. Thromb. Vasc. Biol. 1998, 18, 732–737. [Google Scholar] [CrossRef]
- Finaud, J.; Lac, G.; Filaire, E. Oxidative stress. Sports Med. 2006, 36, 327–358. [Google Scholar] [CrossRef] [PubMed]
- Randerath, K.; Zhou, G.-D.; Monk, S.A.; Randerath, E. Enhanced levels in neonatal rat liver of 7, 8-dihydro-8-oxo-2′-deoxyguanosine (8-hydroxydeoxyguanosine), a major mutagenic oxidative DNA lesion. Carcinogenesis 1997, 18, 1419–1421. [Google Scholar] [CrossRef] [PubMed]
- Mill, C.P.; Chester, J.A.; Riese, D.J. EGFR may couple moderate alcohol consumption to increased breast cancer risk. Breast Cancer Targ. Ther. 2009, 1, 31. [Google Scholar]
- Jackson, A.L.; Loeb, L.A. The contribution of endogenous sources of DNA damage to the multiple mutations in cancer. Mutat. Res./Fundam. Mol. Mech. Mutagenes. 2001, 477, 7–21. [Google Scholar] [CrossRef]
- Ajjan, R.; Watson, P.; Weetman, A. Cytokines and thyroid function. Adv. Neuroimmunol. 1996, 6, 359–386. [Google Scholar] [CrossRef]
- Song, Y.; Driessens, N.; Costa, M.; De Deken, X.; Detours, V.; Corvilain, B.; Maenhaut, C.; Miot, F.; Van Sande, J.; Many, M.-C. Roles of hydrogen peroxide in thyroid physiology and disease. J. Clin. Endocrinol. Metab. 2007, 92, 3764–3773. [Google Scholar] [CrossRef]
- Marcocci, C.; Leo, M.; Altea, M.A. Oxidative stress in Graves’ disease. Eur. Thyr. J. 2012, 1, 80–87. [Google Scholar] [CrossRef]
- Aslan, M.; Cosar, N.; Celik, H.; Aksoy, N.; Dulger, A.C.; Begenik, H.; Soyoral, Y.U.; Kucukoglu, M.E.; Selek, S. Evaluation of oxidative status in patients with hyperthyroidism. Endocrine 2011, 40, 285–289. [Google Scholar] [CrossRef] [PubMed]
- Erdamar, H.; Demirci, H.; Yaman, H.; Erbil, M.K.; Yakar, T.; Sancak, B.; Elbeg, S.; Biberoğlu, G.; Yetkin, I. The effect of hypothyroidism, hyperthyroidism, and their treatment on parameters of oxidative stress and antioxidant status. Clin. Chem. Lab. Med. 2008, 46, 1004–1010. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, R.; Cristani, M.; Vicchio, T.; Alibrandi, A.; Giovinazzo, S.; Saija, A.; Campennì, A.; Trimarchi, F.; Gangemi, S. Increased serum interleukin-37 (IL-37) levels correlate with oxidative stress parameters in Hashimoto’s thyroiditis. J. Endocrinol. Investig. 2019, 42, 199–205. [Google Scholar] [CrossRef] [PubMed]
- Ruggeri, R.M.; Vicchio, T.M.; Cristani, M.; Certo, R.; Caccamo, D.; Alibrandi, A.; Giovinazzo, S.; Saija, A.; Campennì, A.; Trimarchi, F. Oxidative stress and advanced glycation end products in Hashimoto’s thyroiditis. Thyroid 2016, 26, 504–511. [Google Scholar] [CrossRef] [PubMed]
- Elmas, B.; Karacan, M.; Dervişoğlu, P.; Kösecik, M.; İşgüven, Ş.P.; Bal, C. Dynamic thiol/disulphide homeostasis as a novel indicator of oxidative stress in obese children and its relationship with inflammatory-cardiovascular markers. Anatol. J. Cardiol. 2017, 18, 361. [Google Scholar] [CrossRef] [PubMed]
- Turkyilmaz, E.; Yildirim, M.; Cendek, B.D.; Baran, P.; Alisik, M.; Dalgaci, F.; Yavuz, A.F. Evaluation of oxidative stress markers and intra-extracellular antioxidant activities in patients with endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2016, 199, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Koken, T.; Kahraman, A.; Serteser, M. Hemodiyalizin protein karbonil içeriği ve sülfidril grupları üzerine etkisi. Türk Nefroloji Diyaliz ve Transplantasyon Dergisi 2001, 10, 64–66. [Google Scholar]
- Sen, C.K.; Packer, L. Thiol homeostasis and supplements in physical exercise. Am. J. Clin. Nutr. 2000, 72, 653S–669S. [Google Scholar] [CrossRef]
- Cremers, C.M.; Jakob, U. Oxidant sensing by reversible disulfide bond formation. J. Biol. Chem. 2013, 288, 26489–26496. [Google Scholar] [CrossRef]
- Çakatay, U. HİPERTİROİDİ HASTALARINDA PLAZMA OKSİDATİF PROTEİN HASARI. J. Istanbul Fac. Med. 2000, 63. Available online: http://dergipark.org.tr/iuitfd/issue/9301/116258 (accessed on 6 February 2019).
- Venditti, P.; De Rosa, R.; Caldarone, G.; Di Meo, S. Effect of prolonged exercise on oxidative damage and susceptibility to oxidants of rat tissues in severe hyperthyroidism. Arch. Biochem. Biophys. 2005, 442, 229–237. [Google Scholar] [CrossRef] [PubMed]
| Data | Control (n = 35) | Graves (n = 33) | p-Values |
|---|---|---|---|
| Age (Years) | 33.09 ± 10.65 | 31.94 ± 10.28 | =0.653 |
| Gender (male), n (%) | 12 (34.3) | 15 (45.5) | =0.885 |
| BMI (kg/m2) | 24.16 ± 1.65 | 24.76 ± 2.39 | =0.232 |
| Albumin (g/L) | 4.93 ± 0.51 | 4.79 ± 0.36 | =0.192 |
| TSH (µU/mL) | 1.94 ± 1.04 | 0.03 ± 0.07 | <0.001 |
| FT3 (pg/dL) | 3.06 ± 0.33 | 8.51 ± 4.79 | <0.001 |
| FT4 (ng/dL) | 1.01 ± 0.13 | 2.61 ± 1.33 | <0.001 |
| Data | Control (n = 35) | Graves (n = 33) | p-Values |
|---|---|---|---|
| Native thiol (mmol/L) | 454.95 ± 57.89 | 397.42 ± 51.26 | <0.001 |
| Total thiol (mmol/L) | 489.79 ± 58.05 | 439.47 ± 42.02 | <0.001 |
| Disulphide (mmol/L) | 17.42 ± 6.67 | 21.03 ± 9.10 | 0.066 |
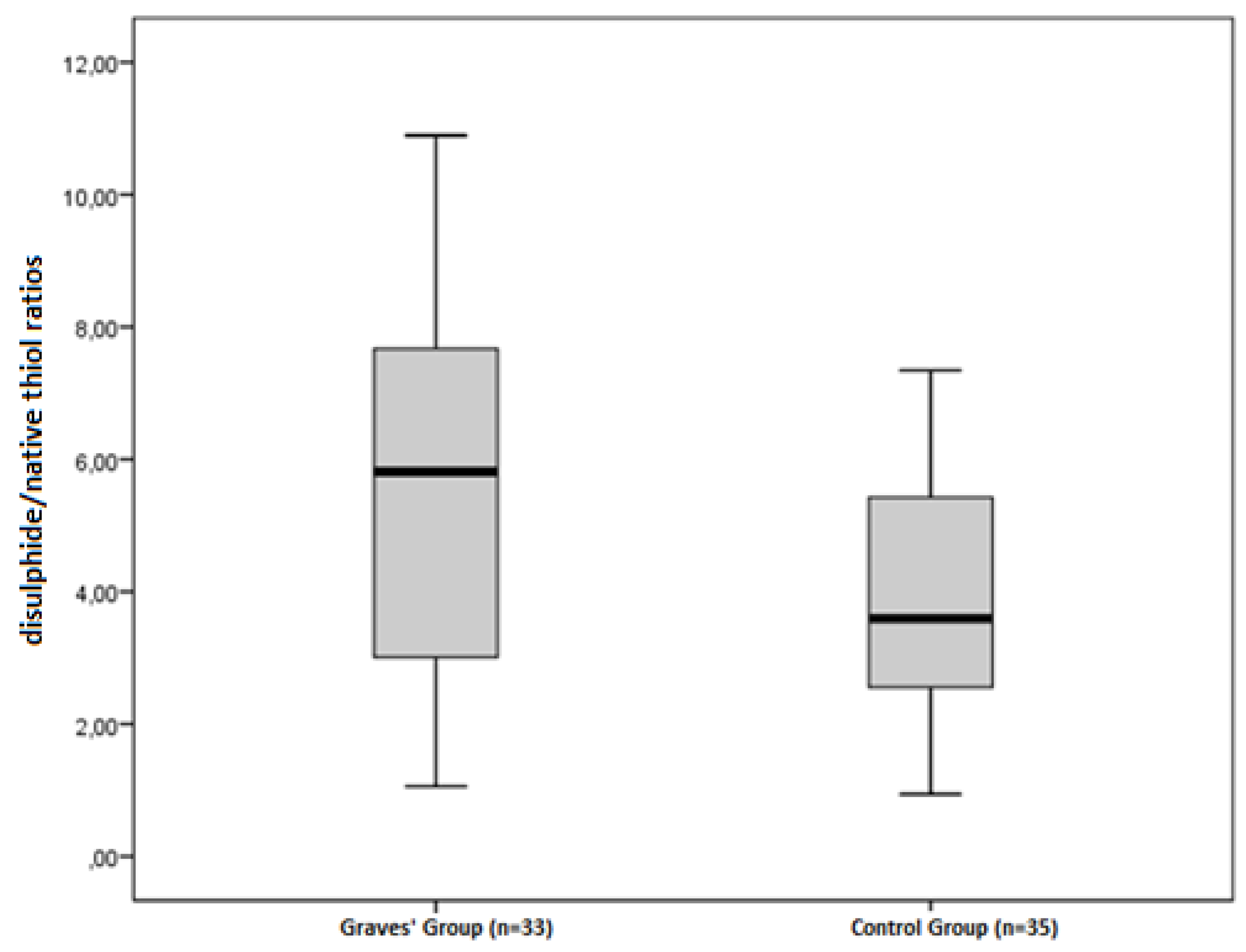
| Disulphide/Native thiol (%) | 3.92 ± 1.62 | 5.56 ± 2.81 | 0.004 |
| Disulphide/Total thiol (%) | 3.59 ± 1.38 | 4.89 ± 2.28 | 0.006 |
| Native thiol/Total thiol (%) | 92.82 ± 2.77 | 90.22 ± 4.56 | 0.006 |
| TAS (mmol TroloxEqv/L) | 0.60 ± 0.15 | 0.55 ± 0.15 | 0,198 |
| TOS (µmol H2O2 Eqv/L) | 1.95 ± 0.44 | 2.40 ± 0.34 | <0.001 |
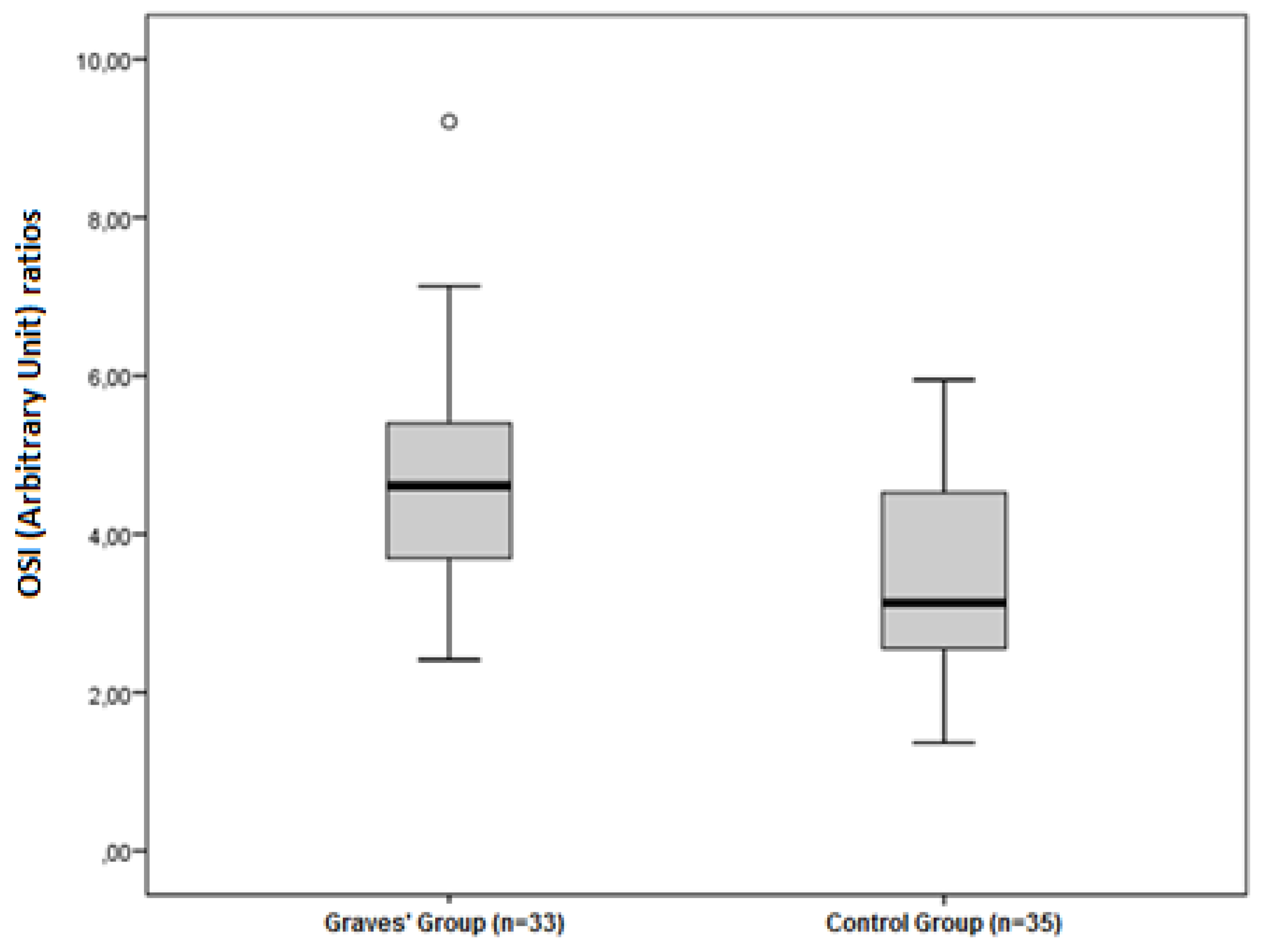
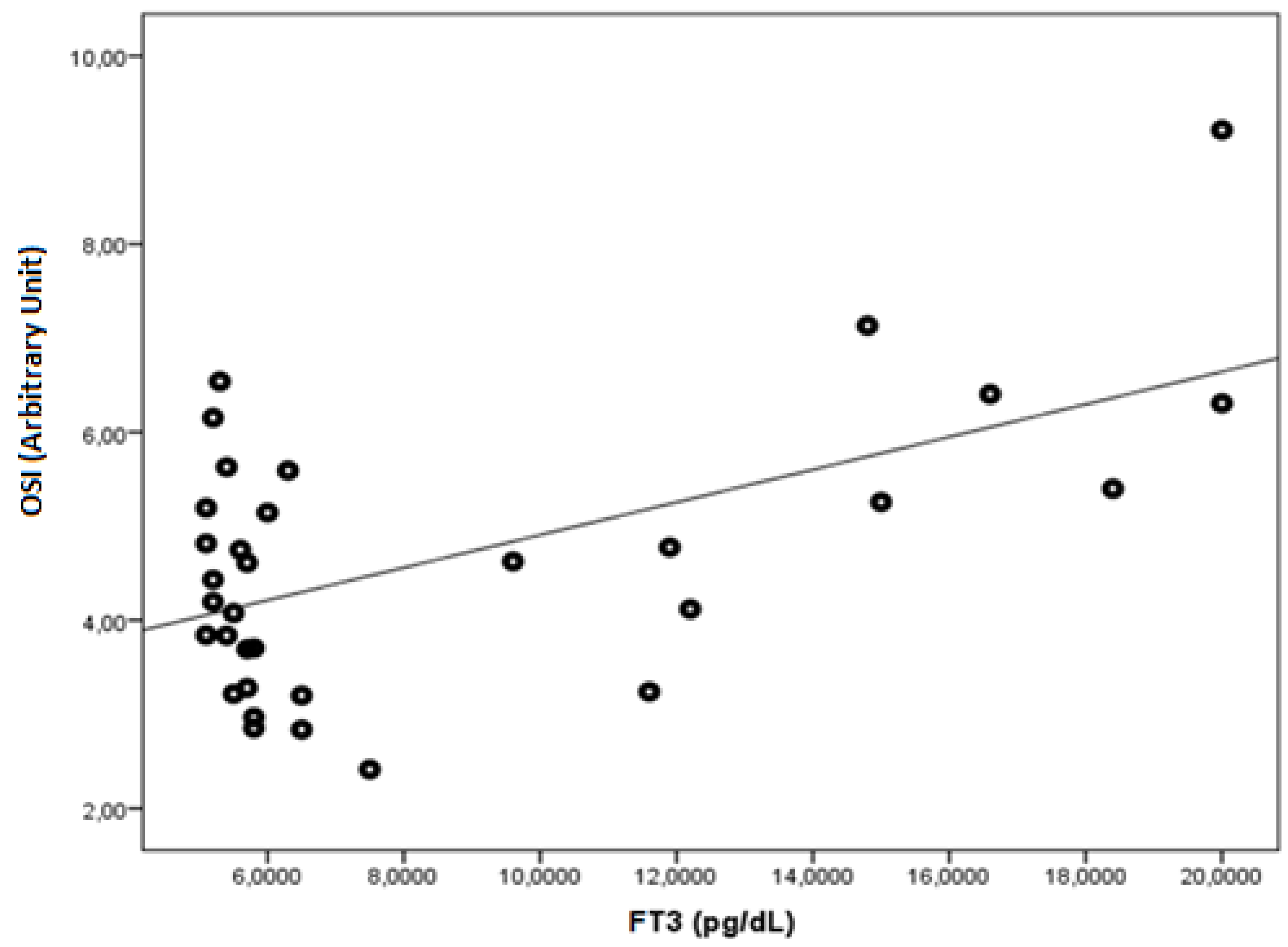
| OSI (Arbitrary Unite) | 3.47 ± 1.27 | 4.65 ± 1.46 | 0.001 |
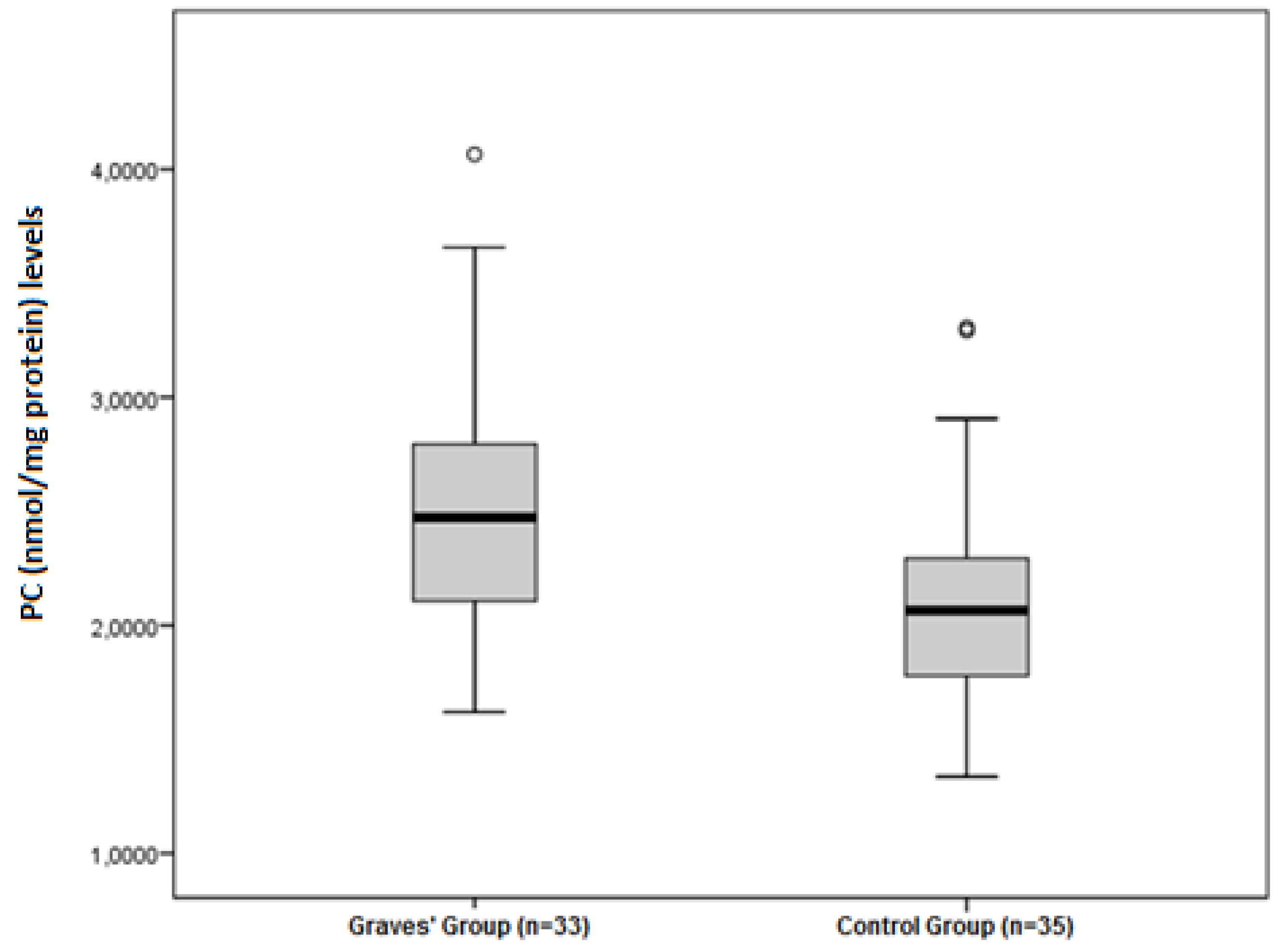
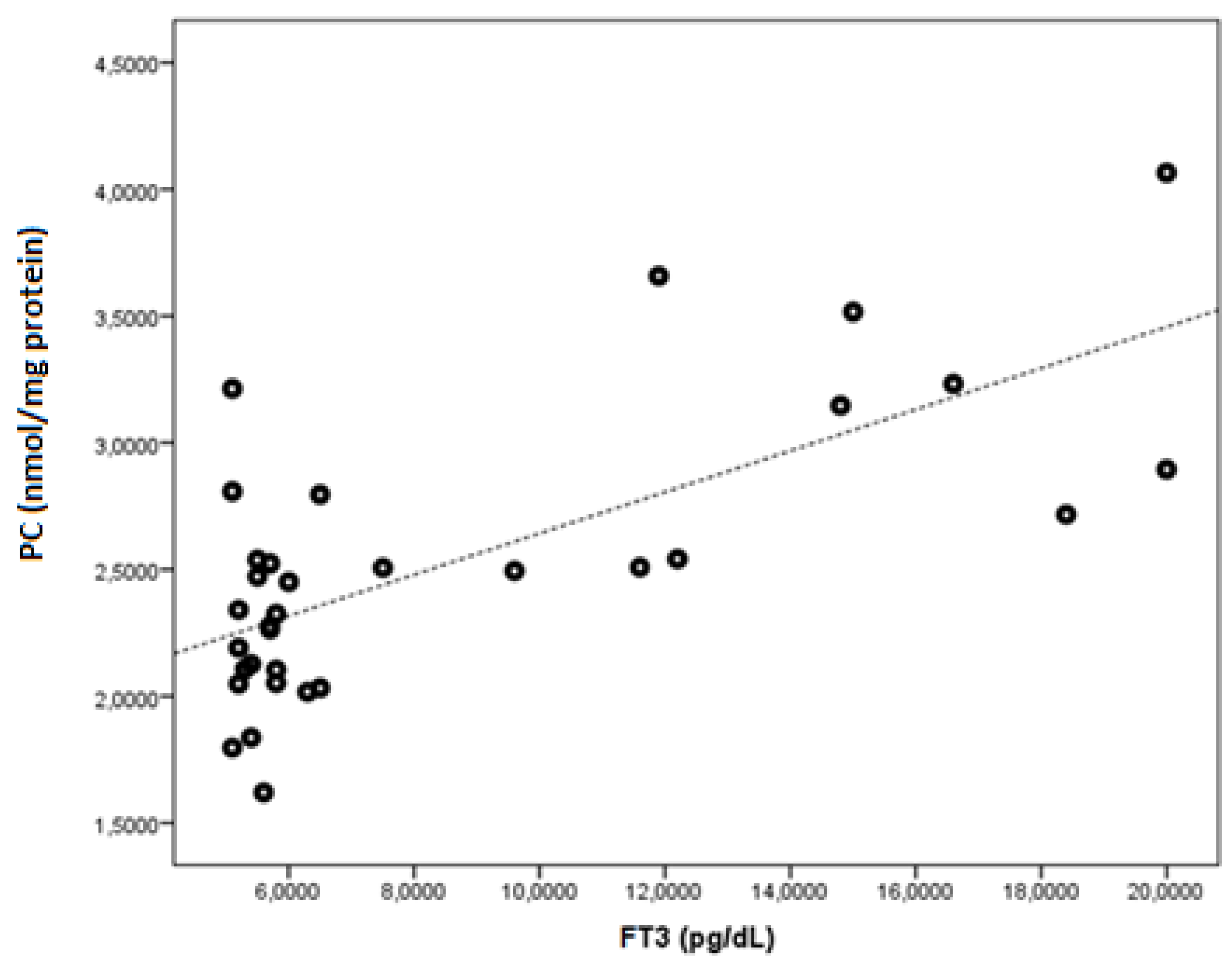
| PC (nmol/mg protein) | 2.09 ± 0.49 | 2.52 ± 0.56 | 0.001 |
| 8-OHdG (ng/mL) | 24.52 ± 3.35 | 23.66 ± 3.21 | 0.282 |
| Native Thiol | Total Thiol | Disulphide | Disulphide/Native Thiol | Disulphide/Total Thiol | Native Thiol/Total Thiol | PC | 8OHdG | TAS | TOS | OSI | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
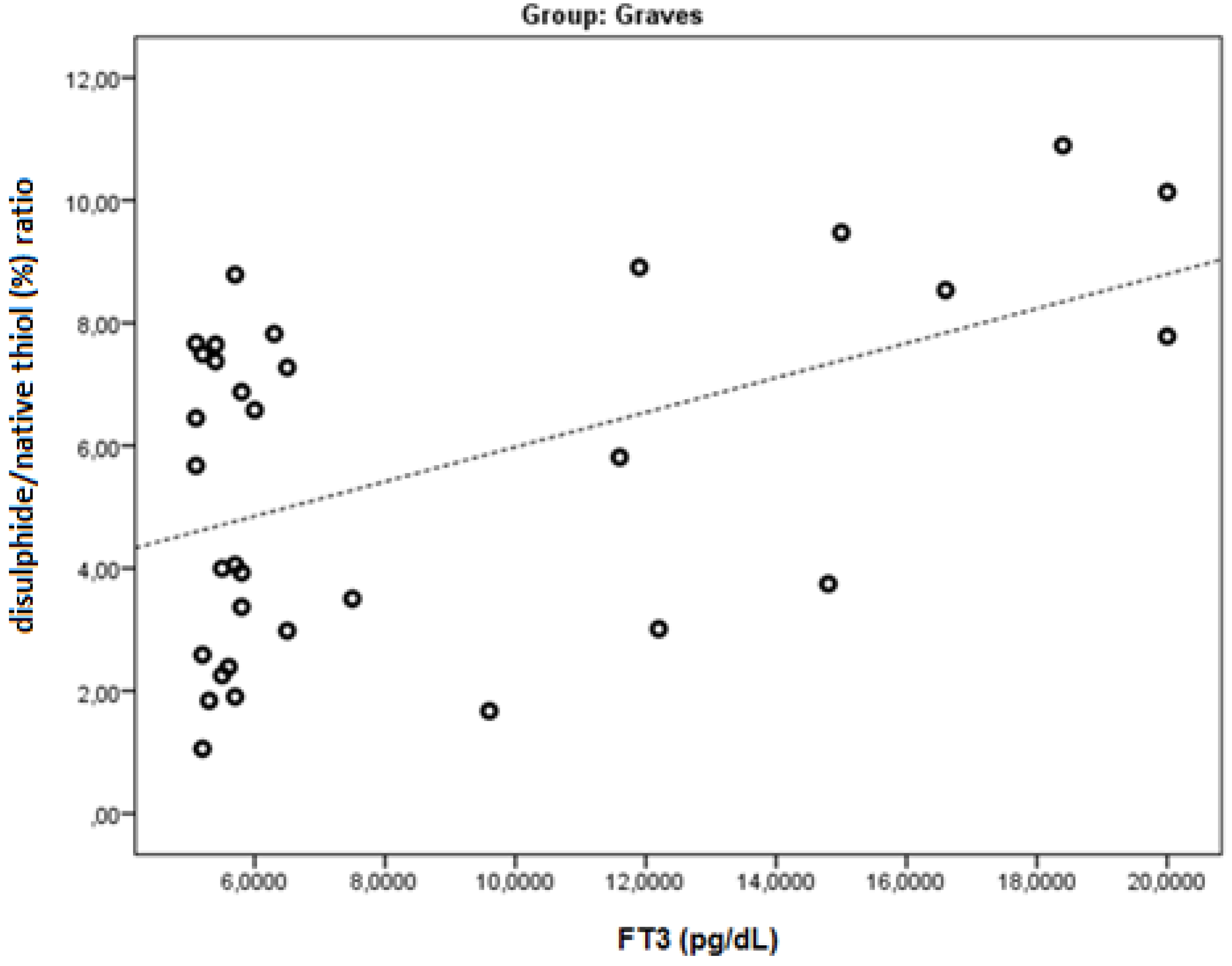
| FT3 | r | −0.543 | −0.505 | 0.364 | 0.481 | 0.464 | −0.464 | 0.700 | 0.267 | −0.286 | 0.512 | 0.569 |
| p | 0.001 | 0.003 | 0.037 | 0.005 | 0.006 | 0.006 | <0.001 | 0.134 | 0.106 | 0.002 | 0.001 | |
| FT4 | r | −0.562 | −0.493 | 0.445 | 0.525 | 0.518 | −0.518 | 0.553 | 0.217 | −0.201 | 0.376 | 0.441 |
| p | 0.001 | 0.004 | 0.010 | 0.002 | 0.002 | 0.002 | 0.001 | 0.225 | 0.262 | 0.031 | 0.010 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agan, V.; Celik, H.; Eren, M.A.; Agan, F.Z.; Erel, O.; Neselioglu, S.; Koyuncu, I.; Gonel, A. An Investigation of Oxidative Stress and Thiol/Disulphide Homeostasis in Graves’ Disease. Medicina 2019, 55, 275. https://doi.org/10.3390/medicina55060275
Agan V, Celik H, Eren MA, Agan FZ, Erel O, Neselioglu S, Koyuncu I, Gonel A. An Investigation of Oxidative Stress and Thiol/Disulphide Homeostasis in Graves’ Disease. Medicina. 2019; 55(6):275. https://doi.org/10.3390/medicina55060275
Chicago/Turabian StyleAgan, Veysel, Hakim Celik, Mehmet Ali Eren, Fatma Zehra Agan, Ozcan Erel, Salim Neselioglu, Ismail Koyuncu, and Ataman Gonel. 2019. "An Investigation of Oxidative Stress and Thiol/Disulphide Homeostasis in Graves’ Disease" Medicina 55, no. 6: 275. https://doi.org/10.3390/medicina55060275
APA StyleAgan, V., Celik, H., Eren, M. A., Agan, F. Z., Erel, O., Neselioglu, S., Koyuncu, I., & Gonel, A. (2019). An Investigation of Oxidative Stress and Thiol/Disulphide Homeostasis in Graves’ Disease. Medicina, 55(6), 275. https://doi.org/10.3390/medicina55060275





