1. Introduction
Liver disease arises from diverse etiologies—including drug-induced injury [
1], viral hepatitis [
2], alcoholic liver disease [
3], and non-alcoholic fatty liver disease [
4]—and, if left untreated, chronic inflammation progresses to hepatic fibrosis and ultimately cirrhosis. Hepatic fibrosis is characterized by repeated injury-repair cycles leading to activation of hepatic stellate cells (HSCs) and excessive accumulation of extracellular matrix (ECM) proteins such as type I/III collagen and fibronectin (Fn) [
5]. Under physiological conditions, ECM synthesis and degradation remain in equilibrium; when ECM deposition outpaces degradation, irreversible fibrosis ensues. Current anti-fibrotic strategies target inhibition of ECM accumulation and induction of HSC apoptosis [
6].
Fungal-derived medicines have played an important role in both traditional and modern healthcare, offering a wide range of bioactive compounds with anti-inflammatory, antioxidant, immunomodulatory, and hepatoprotective effects. In particular, basidiomycete fungi such as
Ganoderma,
Cordyceps, and
Antrodia have been extensively studied for their therapeutic potential in chronic liver diseases and other metabolic disorders. Among these,
Ganoderma species are especially valued for their triterpenoids and polysaccharides, which have been shown to modulate key fibrogenic pathways and protect against liver injury [
7]. The carbon tetrachloride (CCl
4)–induced mouse model is one of the most widely used and well-characterized models for evaluating hepatoprotective agents and was appropriate for the present fibrosis-induction design. In hepatocytes, CCl
4 is bioactivated by cytochrome P450 (CYP) enzymes into trichloromethyl (•CCl
3) and trichloromethyl peroxy (•CCl
3OO) radicals, which initiate lipid peroxidation and oxidative stress, trigger pro-inflammatory cytokine release, and drive HSC activation and ECM overproduction [
8].
Ganoderma species have long been employed in traditional Asian medicine to promote liver health, enhance immunity, and delay aging [
9]. Research on the genus
Ganoderma has overwhelmingly focused on
Ganoderma lucidum, whose triterpenoid constituents—particularly ganoderic acids—have been extensively characterized for hepatoprotective and anti-inflammatory activities [
10]. By contrast,
Ganoderma amboinense remains relatively understudied despite its unique biology and chemical profile. Of over 2000 known
Ganoderma species,
G. amboinense is notable for its antler-shaped fruiting bodies that develop under elevated CO
2 conditions, where they accumulate exceptionally high levels of lanostane-type triterpenoids—most prominently ganoderic acid A—and β-1,3-glucan polysaccharides at the apex [
11,
12,
13]. These bioactive compounds scavenge reactive oxygen species (ROS), suppress inflammation, and inhibit fibrogenic signaling pathways [
14,
15]. Although
G. lucidum remains the most widely studied
Ganoderma species,
G. amboinense has drawn increasing attention due to its distinct phytochemical profile and rare morphology. In particular, it produces unique lanostane-type triterpenoids such as ganoderiol F and ganoderic acid X, which exhibit hepatoprotective and anti-proliferative activities not commonly found in
G. lucidum [
11,
16,
17]. Previous studies have shown that
G. amboinense extracts can protect against acetaminophen-induced liver injury in mice [
18], suggesting its therapeutic potential in hepatic diseases. Furthermore, its antler-shaped fruiting body is highly valued in East Asian traditional medicine, yet remains scientifically underexplored. Therefore, this study aims to not only optimize the bioactive extraction and delivery format but also to scientifically validate the pharmacological properties of this culturally important and underutilized species.
Ganoderma amboinense, a lesser-known species within the
Ganoderma genus, has garnered interest in recent years due to its unique morphology and cultural appeal, particularly among East Asian populations. Unlike the extensively studied
Ganoderma lucidum,
G. amboinense remains underexplored, and its phytochemical composition, extraction protocols, and functional dosage forms are still largely undefined. The distinct appearance of
G. amboinense has long been admired in traditional medicine contexts; however, scientific investigations into its bioactivity and medicinal potential have lagged behind. Moreover, emerging evidence suggests that the bioavailability and pharmacological efficacy of
Ganoderma species can be significantly influenced by formulation type and processing techniques. In light of this, the present study focuses on the extraction and functional characterization of
G. amboinense, aiming to explore its bioactive lanostane-type triterpenoids and to assess their potential biological effects. This investigation represents a novel contribution to the field, offering valuable insights into a species that has been historically appreciated but scientifically neglected. When comparing
Ganoderma amboinense and
Ganoderma lucidum, several key differences in phytochemistry and pharmacology emerge.
G. lucidum is well-known for its triterpenoid-rich profile—particularly ganoderic acids—which have been extensively studied for their hepatoprotective, anti-inflammatory, and antifibrotic properties [
19,
20]. In contrast,
G. amboinense has recently been shown to contain novel lanostane-type triterpenoids with distinct structural features [
11], yet their functional bioactivity remains poorly understood. Given the highly lipophilic nature and poor oral bioavailability of ganoderic acids, particularly GA-A, there is strong scientific rationale for developing delivery systems that enhance their systemic exposure. Sublingual administration can bypass first-pass metabolism and gastrointestinal degradation, leading to improved absorption and bioavailability [
10,
16,
17]. Moreover, formulation optimization allows for the protection of oxidation-prone triterpenoids and better control over release kinetics, which are critical for maintaining pharmacological activity during storage and administration [
10,
21,
22]. These considerations support the exploration of innovative dosage forms for
G. amboinense triterpenoids to maximize their therapeutic potential.
Sublingual dripping pill formulations harness the dense sublingual vasculature to bypass gastrointestinal degradation and achieve rapid, precise delivery of lipophilic actives [
23,
24]. By incorporating PEG 4000 at optimized ratios, release kinetics can be tuned from immediate to sustained profiles, while co-formulating red quinoa (RQ) bran extract (
Chenopodium formosanum) provides potent antioxidant protection [
25], preventing oxidative degradation of sensitive lanostane triterpenoids during processing and storage [
26]. Together, these excipients enable a stable, controllable dripping pill system for reliable sublingual delivery of
Ganoderma amboinense triterpenoids.
In this study, we optimized a one-pot extraction of GA to maximize yields of GA-A and β-1,3-glucan and scaled the process to a 300 L pilot plant. This one-pot extraction approach was specifically optimized to enable the simultaneous recovery of ganoderic acid A (GA-A) and β-1,3-glucan from G. amboinense. Such co-extraction streamlines processing, reduces solvent use, and minimizes degradation of thermolabile or oxidation-sensitive compounds. Importantly, ganoderic acid A (IUPAC name: (3β,7β,15α)-3,7,15-trihydroxy-11,23-epoxy-lanost-8,16-dien-26-oic acid) is a lanostane-type triterpenoid with well-documented hepatoprotective and anti-fibrotic activities, while β-1,3-glucan is a bioactive polysaccharide known for its immunomodulatory, antioxidant, and tissue-repair–supporting properties. The co-presence of these two complementary bioactives may provide additive or synergistic benefits, thereby enhancing the therapeutic potential of the final formulation. We then prepared a granulated extract powder and an innovative sublingual dripping pill (GDP) using PEG 4000/RQ phenolics as composite excipients. Finally, we conducted a head-to-head comparison of GDP and purified GA-A in a CCl4-induced mouse fibrosis model. Endpoints included body and liver weight ratios, serum AST/ALT, histopathology (H&E, Sirius Red), inflammatory cytokines (IL-1β, IL-6, COX-2, TNF-α), and fibrotic markers (TGF-β1, CTGF, α-SMA). We hypothesize that the novel GDP formulation will demonstrate superior GA-A bioavailability and anti-fibrotic efficacy compared to conventional powder and pure compound forms, thereby informing preclinical pharmacokinetic and safety evaluations. While the current study provides functional evidence supporting improved performance of the GDP formulation, direct pharmacokinetic or safety studies were not conducted, and confirmation of bioavailability advantages will require future dedicated investigations.
2. Materials and Methods
2.1. Chemicals
All solvents and standards were of analytical or HPLC grade. Ethanol (95%) for co-extraction was purchased from ECHO CHEMICAL Co., Ltd. (Miaoli, Taiwan); methanol and acetonitrile for HPLC mobile phases were obtained from Avantor Inc. (Radnor, PA, USA); and formic acid (≥98%) was supplied by Honeywell Taiwan Ltd. (New Taipei, Taiwan). Reference standards of ganoderic acid A (>98% purity) were sourced from Chengdu Must Bio-Technology Co. Ltd. (Chengdu, Sichuan, China), while rutin and β-1,3-glucan standards were acquired from Sigma-Aldrich Co. (St. Louis, MO, USA). Carbon tetrachloride (≥99%) for induction of liver fibrosis was obtained from ALPS GHEM Co., Ltd. (New Taipei, Taiwan) and diluted in olive oil (1:4, v/v) immediately before use. Total protein concentrations were determined using the BCA Protein Assay Kit (Cat. No. 23225; Merck KGaA, Millipore, Darmstadt, Germany). Serum levels of TNF-α and IL-6 were measured with ELISA kits (Cat. Nos. 50349-MNAE and 50136-MNAE; Sino Biological Inc., North Wales, PA, USA), and CTGF concentrations with the CTGF ELISA Kit (LS-G26584; Lifespan Biosciences Inc., Seattle, WA, USA). For immunoblotting and immunohistochemistry, we used rabbit anti-mouse TNF-α polyclonal antibody (AB2148P; EMD Millipore, Temecula, CA, USA), mouse anti-rat IL-6 monoclonal antibody (sc-57315; Santa Cruz Biotechnology, Dallas, TX, USA), anti-TGF-β1 polyclonal antibody (39303; Signalway Antibody, College Park, MD, USA), anti-α-smooth muscle actin (ACTA2) monoclonal antibody (04-1094; EMD Millipore, Temecula, CA, USA), and anti-CTGF antibody (FNab02054; Wuhan Fine Biotech, Wuhan, Hubei, China), each used at the manufacturer’s recommended dilution.
2.2. Preparation of Test Substances
2.2.1. Extraction of G. amboinense Fruiting Body
The Ganoderma amboinense fruiting bodies (antler-type reishi) used in this study were provided by GE Health R&D Co., Ltd. (Taitung, Taiwan). The strain is a proprietary culture maintained in the company collection and used to produce the characteristic antler-shaped fruiting bodies. This extraction protocol was developed based on preliminary pilot-scale extractions (1–3 kg scale) performed in-house, which demonstrated consistent yields of ganoderic acid A and good product stability, supporting the decision to proceed with 300-L scale-up.
Ganoderma amboinense fruiting bodies contain both water-soluble constituents (e.g., polysaccharides) and lipid-soluble constituents (e.g., triterpenoids). To ensure comprehensive recovery of bioactive compounds, water and ethanol extractions were performed separately and then combined. A 300-L extractor (YC-MEC-300, YENCHEN MACHINERY CO., LTD., Taoyuan, Taiwan) was charged with 23 kg of G. amboinense, followed by an initial 2-h aqueous extraction with 200 L of water at 80 °C and a second 1-h aqueous extraction with 100 L of water at 80 °C. The two aqueous extracts were combined and vacuum-concentrated (VC-200, YENCHEN MACHINERY CO., LTD., Taoyuan, Taiwan). This was followed by two ethanolic extractions (120 L and 80 L of 95% ethanol, at 70 °C for 2 h and 1 h, respectively), with filtrates pooled and concentrated to yield the final extract.
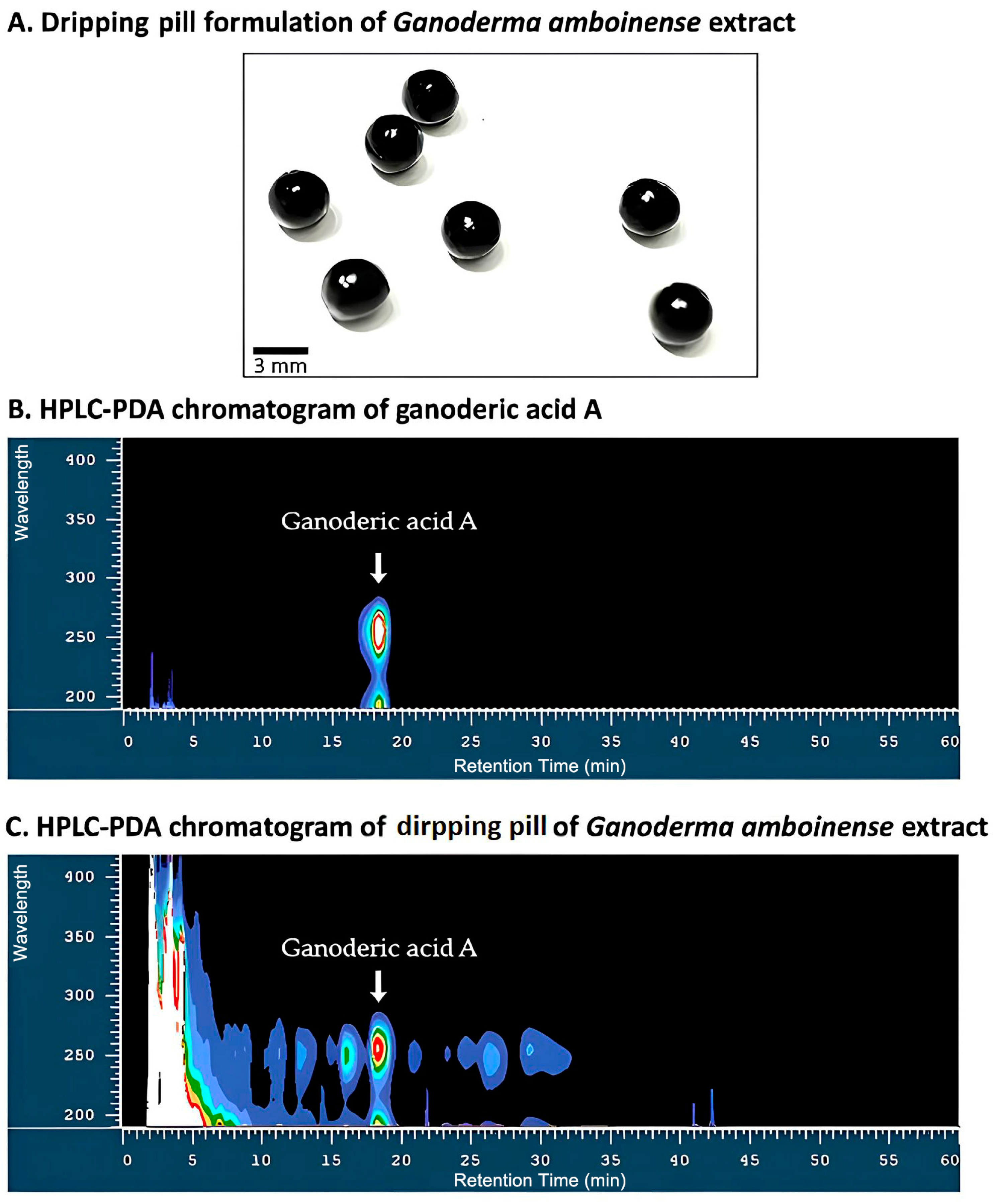
2.2.2. Preparation of Dripping Pill Formulations
The concentrated G. amboinense extract was lyophilized to yield a dry powder. For the sublingual dripping pill (GDP), the extract powder was blended with PEG 4000 (50% w/w) and red quinoa phenolic extract (20% w/w) at 70 °C until homogeneous, then passed through a 20-mesh sieve. The molten mixture was formed into uniform spherical droplets using a drip-casting machine (LUNG SHENG Precision Industries Co., Ltd., Kaohsiung, Taiwan) equipped with a 3.0 mm nozzle, operating at a constant feed rate (5 mL/min) and temperature (70 °C). Droplets solidified within approximately 2 min at room temperature due to PEG 4000 crystallization. Residual surface oil was removed by centrifugation and blotting. Weight uniformity testing of 30 randomly selected units yielded a mean weight of 30 mg, meeting pharmacopeial requirements for solid dosage forms. The inclusion of red quinoa phenolics was specifically intended to inhibit oxidative degradation of ganoderic acid A during storage and thereby prolong product shelf life.
2.2.3. Quantitative Analysis of Active Constituents
GDP samples were dissolved in methanol (1:4,
w/
v), diluted, and filtered (0.45 μm). Ganoderic acid A was quantified by HPLC (Model L-2130, Hitachi Co., Tokyo, Japan) on a YMC-Triart C
18 column (250 × 4.6 mm, 5 μm) with a gradient of acetonitrile and 0.1% formic acid in water (30–40.7% A over 25 min, to 100% A by 40 min; 1 mL/min; 253 nm; 40 °C), using a previously described method with slight modifications [
27]. Rutin from the red quinoa phenolic extract was analyzed using a modified HPLC method based on Lin et al. [
15] with an Ascentis
® C18 column (250 × 4.6 mm, 5 μm). The mobile phase consisted of acetonitrile (solvent A) and 0.1% formic acid in water (solvent B) with the following gradient: 0–20 min, 17% A; 20–25 min, 17–65% A; 25–35 min, 65% A; 35–40 min, 65–100% A; 40–45 min, 100% A; 45–50 min, 100–17% A; 50–55 min, 17% A. The flow rate was 1 mL/min, the detection wavelength was 250 nm, the injection volume was 20 μL, and the column temperature was maintained at 40 °C.
2.3. In Vivo Evaluation in a CCl4-Induced Mouse Fibrosis Model
2.3.1. Animal Housing and Group Allocation
Male BALB/c mice (8 weeks old, weighing approximately 20–25 g) were obtained from the National Laboratory Animal Center in Taipei, Taiwan. Animals were maintained under controlled conditions (24 ± 1 °C, 60% relative humidity) on a 12 h light–dark cycle (lights on from 08:00 to 20:00), with free access to standard chow and water throughout the study. Mice were randomized into six experimental groups (
n = 7 per group). The group size was selected to ensure sufficient statistical power to detect meaningful differences in key endpoints while adhering to the 3Rs principle (Replacement, Reduction, and Refinement) for ethical animal use. The normal control group received intraperitoneal injections of olive oil vehicle only. The fibrosis control group was administered carbon tetrachloride (CCl
4) dissolved in olive oil at a dose of 0.4 mL per kilogram of body weight, without any test article. Treatment groups received CCl
4 in the same manner and, in addition, one of the following by oral gavage once daily: a
Ganoderma amboinense powder (GP) formulation at 410 mg/kg/day;
Ganoderma amboinense dripping pills (GDP) at either 92.25 mg/kg/day or 461.25 mg/kg/day; or purified ganoderic acid A (GAA) at 1.07 mg/kg/day. Dosages for mice were calculated according to the U.S. Food and Drug Administration (FDA) body surface area conversion guidance [
28], using the formula: Mouse dose (mg/kg) = [Human daily dose (mg) ÷ 60] × 12.3, where 60 kg represents the reference adult human body weight and 12.3 is the body surface area scaling factor for mice. Animals were observed daily for appearance, activity, and food intake to monitor potential adverse effects.
2.3.2. Fibrosis Induction Protocol
The CCl
4-induced hepatic fibrosis mouse model was established based on a previously reported method with modifications [
29]. The entire experiment lasted 6 weeks. From Day 1, treatment groups received daily oral administration of GDP, GP, or GAA. Starting in Week 3, all groups except the normal control (NOR) received intraperitoneal injections of CCl
4 (0.4 μL/g body weight in olive oil) three times per week for six consecutive weeks, for a total of 18 injections. This prospective design allowed GDP, GP, or GAA to be administered as a prophylactic intervention, enabling evaluation of their ability to prevent or attenuate fibrosis development. At the end of Week 6, all animals were sacrificed for histopathological, biochemical, and molecular analyses.
2.3.3. Sacrifice and Sample Collection
At the end of the sixth week, mice were fasted overnight (14 h), weighed, and euthanized by CO2 asphyxiation. Blood was collected by cardiac puncture, allowed to clot at room temperature for two hours, then centrifuged at 3000× g for 10 min (Z326K, Hermle Labortechnik, Wehingen, Germany) to obtain serum, which was stored at –20 °C for later biochemical assays. Livers were excised, blotted dry, and weighed. Representative portions were fixed in 10% neutral-buffered formalin for histological processing, while the remaining tissue was rinsed with 0.9% saline and frozen at –80 °C for biochemical and molecular analyses.
2.3.4. Serum Biochemistry
Serum activities of aspartate aminotransferase (AST) and alanine aminotransferase (ALT) were measured by a certified clinical laboratory using an automated biochemical analyzer (Beckman Coulter AU-700, Beckman Coulter Inc., Brea, CA, USA).
2.3.5. Histopathological Assessment
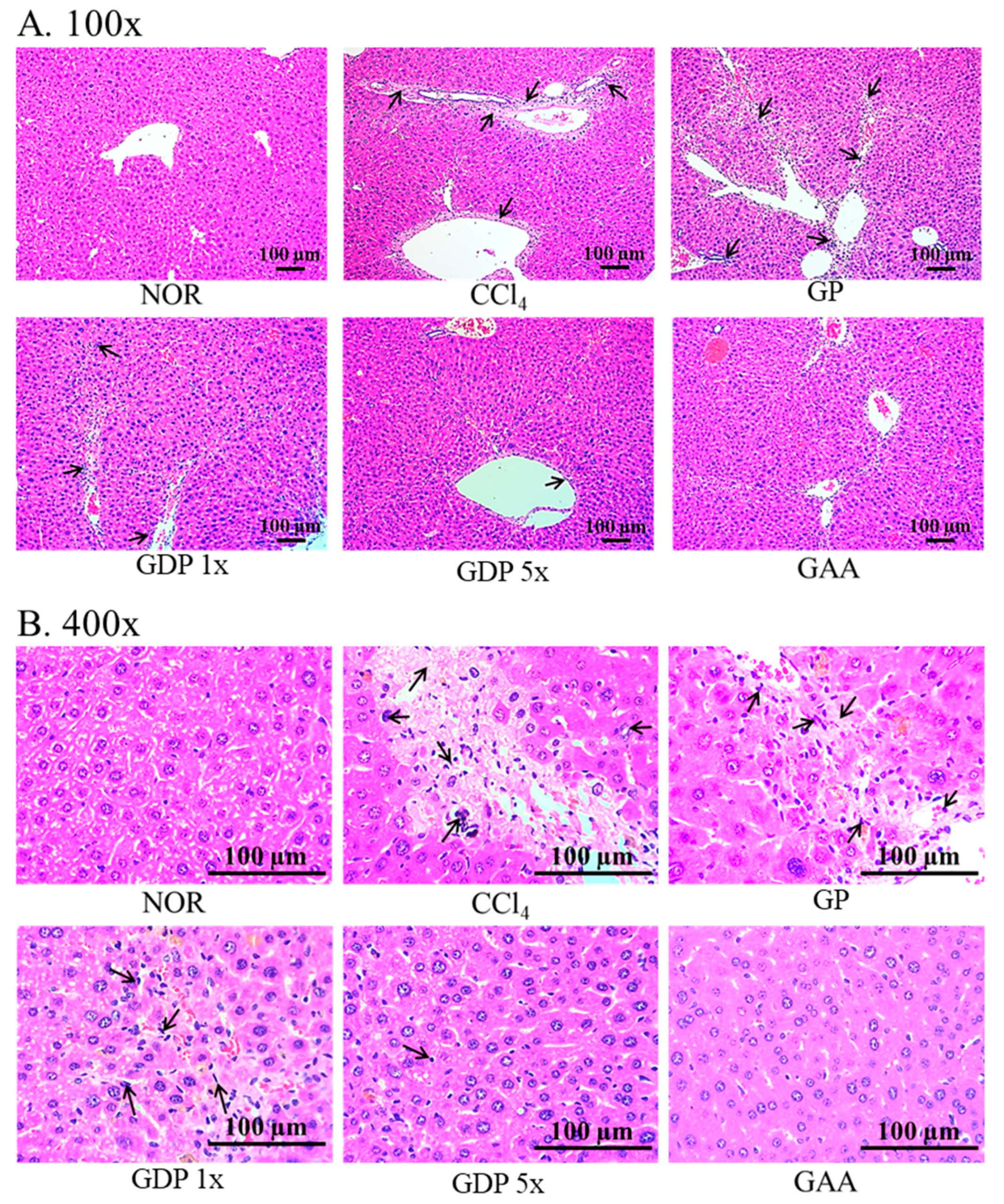
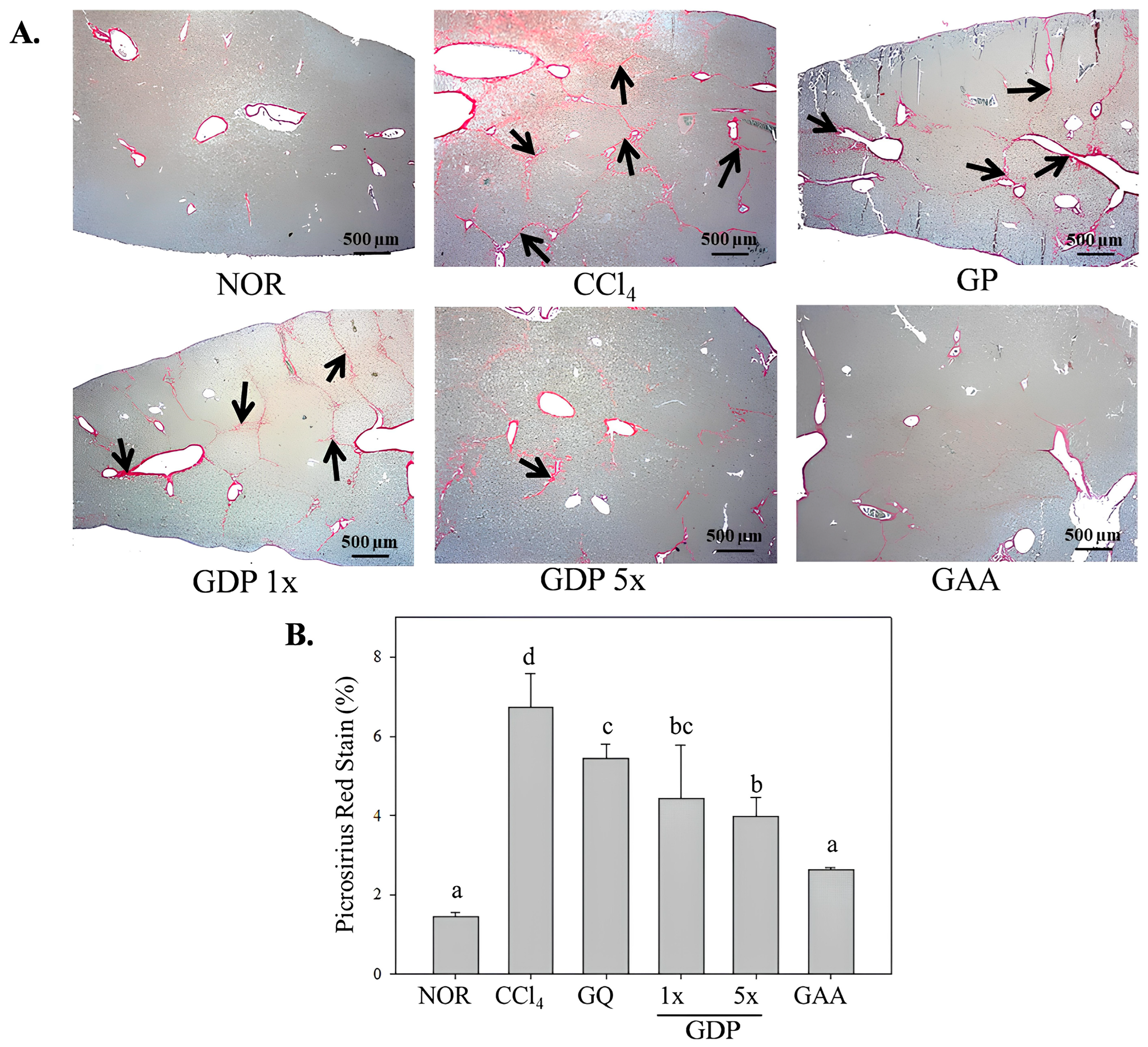
Formalin-fixed liver specimens were paraffin-embedded and sectioned at 3 μm thickness. Sections were stained with hematoxylin and eosin to evaluate general morphology and with picrosirius red to visualize collagen deposition. Stained sections were examined under light microscopy (EX 20, Sunny Optical Technology Co. Ltd., Yuyao, Zhejiang Province, China) at magnifications of 40×, 100×, and 400×, and representative images were captured for semi-quantitative assessment of fibrosis.
2.3.6. Liver Homogenate Preparation and Protein Quantification
Frozen liver tissue (~0.1 g) was homogenized in 1 mL ice-cold phosphate-buffered saline (0.1 M, pH 7.4) and centrifuged at 10,000× g for 10 min at 4 °C. The supernatant was collected, and the procedure was repeated until the homogenate was free of particulate debris. Total protein concentration in the final supernatant was determined by the bicinchoninic acid (BCA) assay, using bovine serum albumin standards and measuring absorbance at 562 nm after a 30 min reaction at 37 °C.
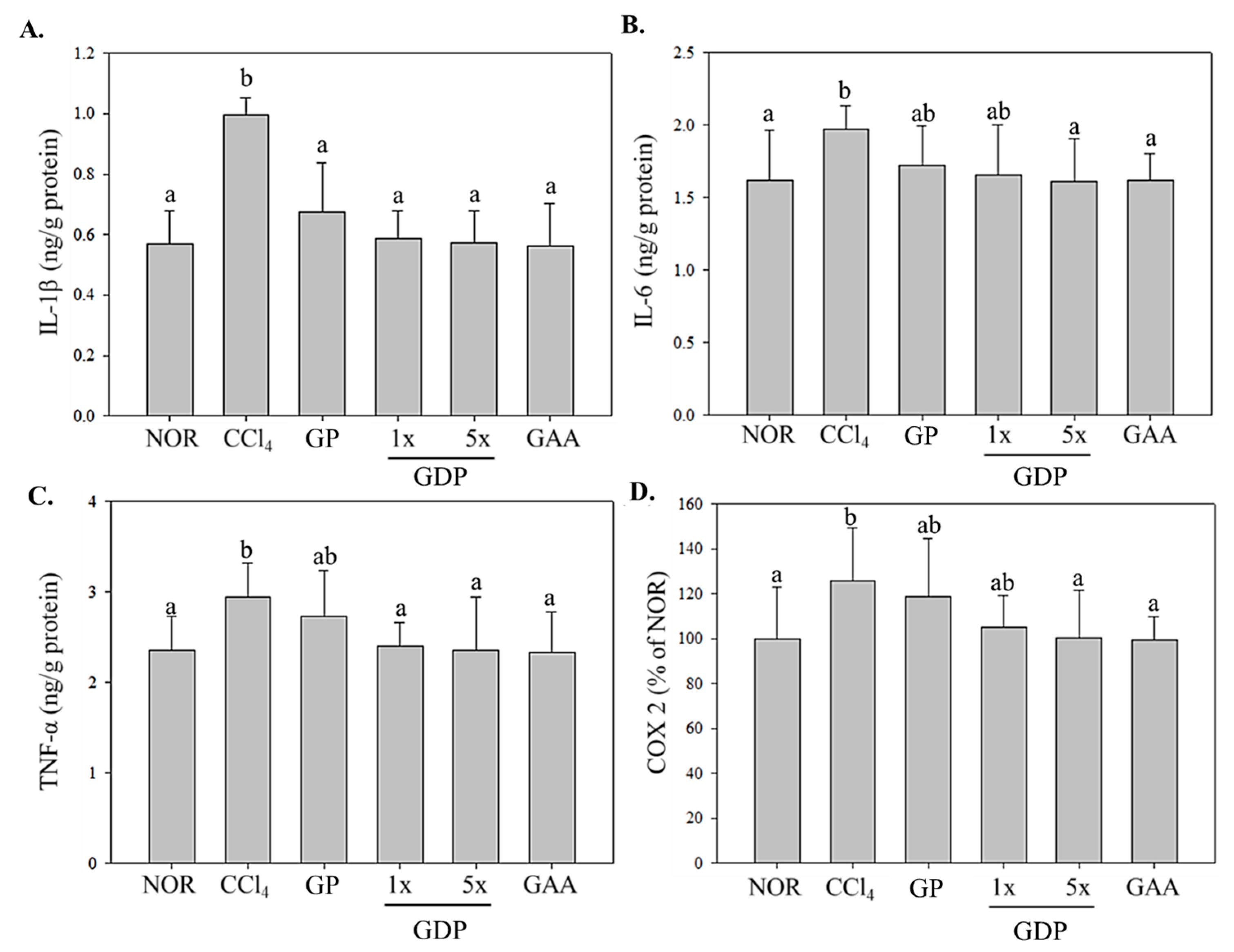
2.3.7. Quantification of Inflammatory and Fibrotic Biomarkers
Levels of tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6), connective tissue growth factor (CTGF), transforming growth factor-β1 (TGF-β1), and α-smooth muscle actin (α-SMA) in liver homogenates were measured by enzyme-linked immunosorbent assay (ELISA). Homogenate samples (100 μL) were incubated in antibody-coated 96-well plates at 37 °C for 1 h, washed three times with PBS-Tween 20, then incubated with primary antibody (100 μL) for an additional hour. After washing, wells were incubated with horseradish peroxidase-conjugated secondary antibody (100 μL) for 1 h, followed by incubation with tetramethylbenzidine substrate for 15 min. The reaction was terminated with 2 N sulfuric acid, and absorbance was read at 450 nm. Biomarker concentrations were interpolated from standard curves.
2.4. Statistical Analysis
All quantitative data are expressed as mean ± standard deviation. Group comparisons were performed by one-way analysis of variance followed by Duncan’s multiple range test using SPSS version 22.0. Differences were considered statistically significant at p < 0.05.
4. Discussion
Notably, Hsu et al. (2008) demonstrated that extracts of
G. amboinense could protect mice from acetaminophen-induced acute liver injury, suggesting hepatoprotective potential [
18]. Additional studies revealed that specific triterpenes isolated from
G. amboinense, such as ganoderiol F and ganoderic acid X, exhibit antiproliferative effects and promote apoptosis in cancer cell lines [
16,
17]. These compounds act through mechanisms involving topoisomerase inhibition and cellular senescence, thereby indicating that
G. amboinense harbors functionally potent metabolites distinct from those of
G. lucidum. While both species produce lanostane triterpenoids,
G. lucidum compounds have been more thoroughly investigated and commercialized. In contrast,
G. amboinense possesses unique chemical scaffolds and therapeutic properties that are just beginning to be elucidated. Given the known influence of formulation on bioactivity, further investigation into optimal extraction conditions and delivery formats for
G. amboinense is warranted. In this context, our one-pot ethanol extraction was intentionally optimized to co-recover ganoderic acid A (GA-A) and β-1,3-glucan, thereby enabling simultaneous enrichment of a potent antifibrotic triterpenoid and an immunomodulatory polysaccharide known to enhance antioxidant defense and tissue repair [
30,
31]. This dual-component recovery not only streamlines processing but also preserves potential synergistic effects between triterpenoid and polysaccharide fractions in liver protection [
32]. This study contributes to bridging that knowledge gap by applying systematic extraction and characterization methodologies, highlighting the untapped pharmacological potential of this culturally significant yet scientifically overlooked species.
Dripping pills are an advanced dosage form widely applied in traditional Chinese medicine modernization due to their advantages in enhancing bioavailability, precise dosing, and patient compliance. Particularly in cardiovascular therapeutics, formulations such as Compound Danshen Dripping Pills (CDDP) have been shown to improve outcomes in patients with stable angina and myocardial ischemia by delivering multi-component agents with consistent pharmacokinetics and enhanced systemic exposure [
21,
22]. These benefits suggest that the dripping pill system could serve as a valuable delivery vehicle for other traditional medicinal materials, especially those rich in lipophilic compounds like triterpenoids.
Ganoderma amboinense, a rare and visually distinctive species of
Ganoderma highly appreciated in East Asian cultures, contains a range of lanostane-type triterpenoids with potential bioactivity [
11]. Despite its phytochemical richness, its pharmacological functions and delivery technologies remain underexplored. Unlike
Ganoderma lucidum, which has been studied in powders, capsules, and aqueous extracts, there is no current research on the encapsulation of
Ganoderma extracts—particularly from
G. amboinense—into dripping pills for functional evaluation. To the best of our knowledge, this is the first study to formulate
Ganoderma extract into a dripping pill dosage form and systematically evaluate its functional effects in vivo. By applying ethanol extraction and scale-up encapsulation, we were able to deliver a stable form of
G. amboinense that demonstrates hepatoprotective efficacy in a CCl
4-induced liver fibrosis mouse model. Our findings provide new evidence that the combination of traditional fungal medicine and modern drug delivery systems could yield promising functional health products, potentially expanding the clinical applications of medicinal mushrooms.
The present study demonstrates that the ethanol-extracted dripping pill formulation of G. amboinense fruiting body (GDP) confers superior protective effects against CCl4-induced hepatic fibrosis in mice compared to the unextracted powder form GP. Across all measured endpoints—body weight preservation, liver function markers (AST, ALT), histopathological structure, inflammatory cytokine expression, and pro-fibrotic marker suppression—the GDP groups (particularly the 5× dose) exhibited significantly greater efficacy than GP, which showed only marginal or non-significant trends. Notably, GDP mitigated CCl4-induced weight loss and hepatomegaly, significantly reducing liver weight and the liver-to-body weight ratio, while GP did not yield significant improvement. The hepatoprotective superiority of GDP was further evidenced by the normalization of serum AST and ALT activities, where the 5× formulation reduced both markers to levels comparable to those achieved by purified ganoderic acid A. In contrast, GP failed to significantly alter these enzymes, highlighting the limited bioactivity of the crude powder form.
The GDP formulation represents a pharmacologically optimized delivery system for
Ganoderma amboinense, providing enhanced antifibrotic and anti-inflammatory effects over the crude powder and matching the efficacy of isolated ganoderic acid A. These results support the broader utility of dripping pills as a novel dosage form for lipophilic fungal constituents and validate the translational potential of
G. amboinense in liver protection strategies. A key innovation in this formulation is the inclusion of red quinoa (
Chenopodium formosanum) extract, rich in the bioflavonoid rutin, as an excipient during the dripping pill manufacturing process. Rutin has been demonstrated to exert anti-inflammatory effects via NF-κB and MAPK pathway inhibition in macrophage models (e.g., DH82 cells), and other studies have shown its hepatoprotective and anti-fibrotic roles through suppression of TLR4/P2X7/NF-κB signaling in hepatic models [
33]. Additionally, rutin has been reported to alleviate liver injury by restoring redox balance and mitochondrial function in hepatocytes [
34], further supporting its role as a functional stabilizer and synergistic component.
In this study, we demonstrated that Ganoderma amboinense extract formulated into a dripping pill dosage form (GDP) significantly attenuates CCl4-induced hepatic injury and fibrosis in mice. While previous studies have reported the hepatoprotective effects of ganoderic acids, especially ganoderic acid A (GAA), our results further establish the superior therapeutic potential of GDP compared to the raw powder form GP, both in serum biomarkers and histopathological features. Notably, the GDP 5× group outperformed the unprocessed powder group in preserving body weight, reducing liver-to-body weight ratio, and normalizing serum AST and ALT activities, indicating better systemic and hepatic protection. Histological analysis also revealed significant improvements in liver architecture and a marked reduction in collagen deposition in the GDP groups, comparable to purified GAA.
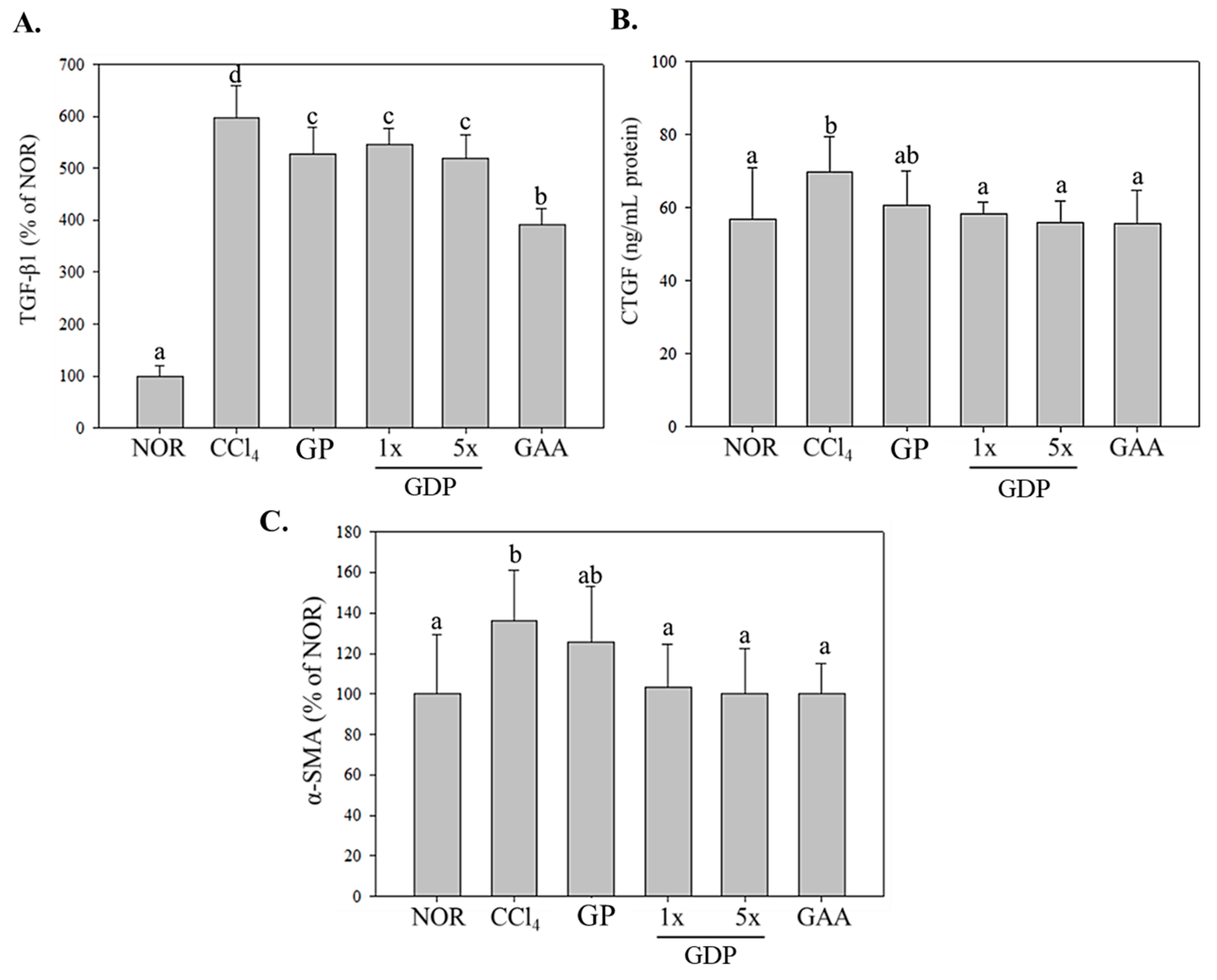
Histological examination supported these biochemical findings: GDP-treated livers maintained clearer hepatocyte architecture and exhibited markedly reduced necrosis and collagen deposition, with fibrotic areas diminished by ~60%. These improvements were absent or minimal in the GP group. At the molecular level, the dripping pill significantly suppressed hepatic expression of inflammatory mediators (IL-1β, IL-6, TNF-α, and COX-2) and fibrogenic markers (TGF-β1, CTGF, and α-SMA), while GP displayed only minor, non-significant reductions. The enhanced efficacy of the GDP formulation likely results from multiple factors: (1) improved solubility and absorption of bioactive lanostane triterpenoids due to ethanol extraction; and (2) sustained and controlled release enabled by the dripping pill matrix. Collectively, these features maximize the systemic bioavailability and pharmacological performance of
Ganoderma compounds. In contrast, the unextracted powder lacks both bioactive enrichment and delivery optimization, resulting in subtherapeutic exposure. Moreover, GDP significantly suppressed the expression of hepatic pro-inflammatory cytokines, including IL-1β, TNF-α, IL-6, and COX-2, highlighting its anti-inflammatory effects. These outcomes are consistent with previous studies showing that triterpenoids from
Ganoderma species downregulate NF-κB signaling and inflammatory mediators, contributing to reduced hepatic inflammation [
35]. In particular, GDP 5× provided significant reductions in pro-fibrotic markers such as TGF-β1, CTGF, and α-SMA, which are critical mediators in hepatic stellate cell activation and extracellular matrix accumulation, underscoring its potential anti-fibrotic efficacy. To further contextualize these findings, the inhibition of TGF-β1 signaling observed with GDP parallels the mechanism targeted by clinical antifibrotic agents such as pirfenidone, which is currently approved for idiopathic pulmonary fibrosis and under investigation for hepatic fibrosis. While pirfenidone primarily attenuates TGF-β1–mediated fibroblast activation and collagen synthesis, our results show that GDP (particularly at the 5× dose) significantly reduced hepatic TGF-β1 expression together with downstream fibrogenic markers (CTGF, α-SMA). This suggests that GDP exerts comparable pathway-level modulation. Although direct head-to-head efficacy studies are required, the present findings highlight GDP as a natural multi-component alternative that may synergize with, or provide complementary benefits to, existing antifibrotic pharmacotherapies.
To our knowledge, this is the first study to evaluate the functional efficacy of
Ganoderma amboinense ethanol extract delivered via a dripping pill system, an innovative dosage form that enhances active compound stability, improves bioavailability, and ensures consistent dosing. While dripping pills have been extensively used in traditional Chinese medicine for cardiovascular and metabolic disorders [
36], their application in fungal medicine remains unexplored until now. The current findings suggest that GDP, supported by the bioactivity of red quinoa-derived rutin, represents a promising delivery platform for fungal triterpenoids in hepatic health applications. Future studies should directly evaluate the pharmacokinetics and safety profile of the GDP formulation in preclinical settings to substantiate its potential for further development into functional or therapeutic products.