Research Progress on the Biological Activities and Clinical Applications of Pseudoprotodioscin
Abstract
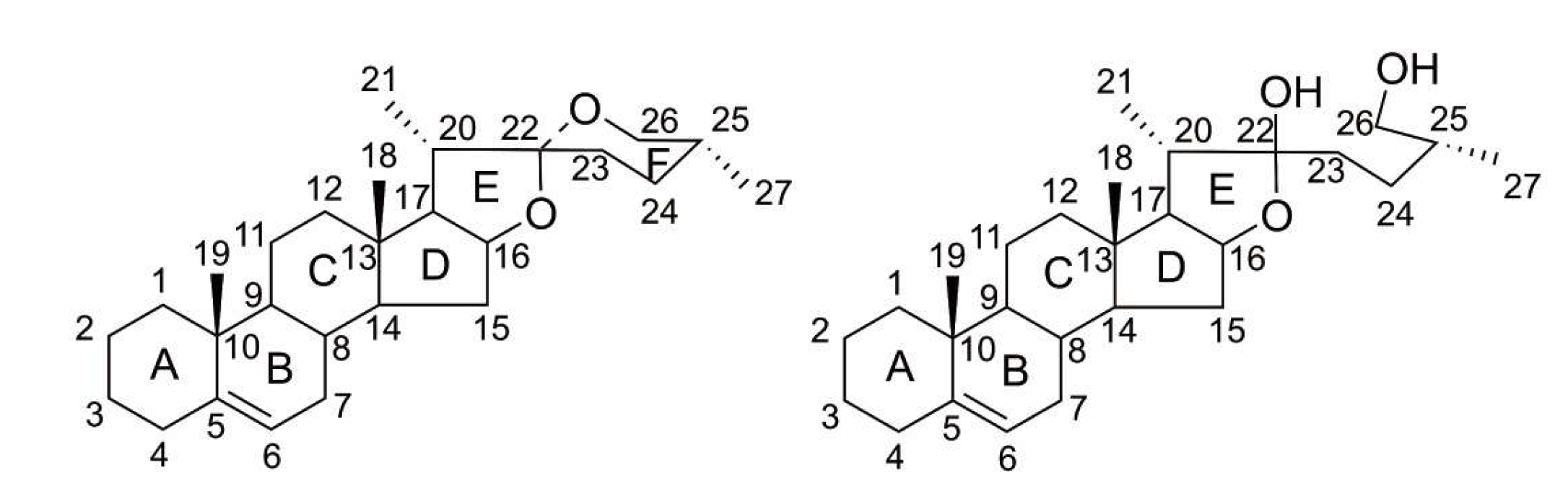
1. Introduction
2. The Source and Content of PPD
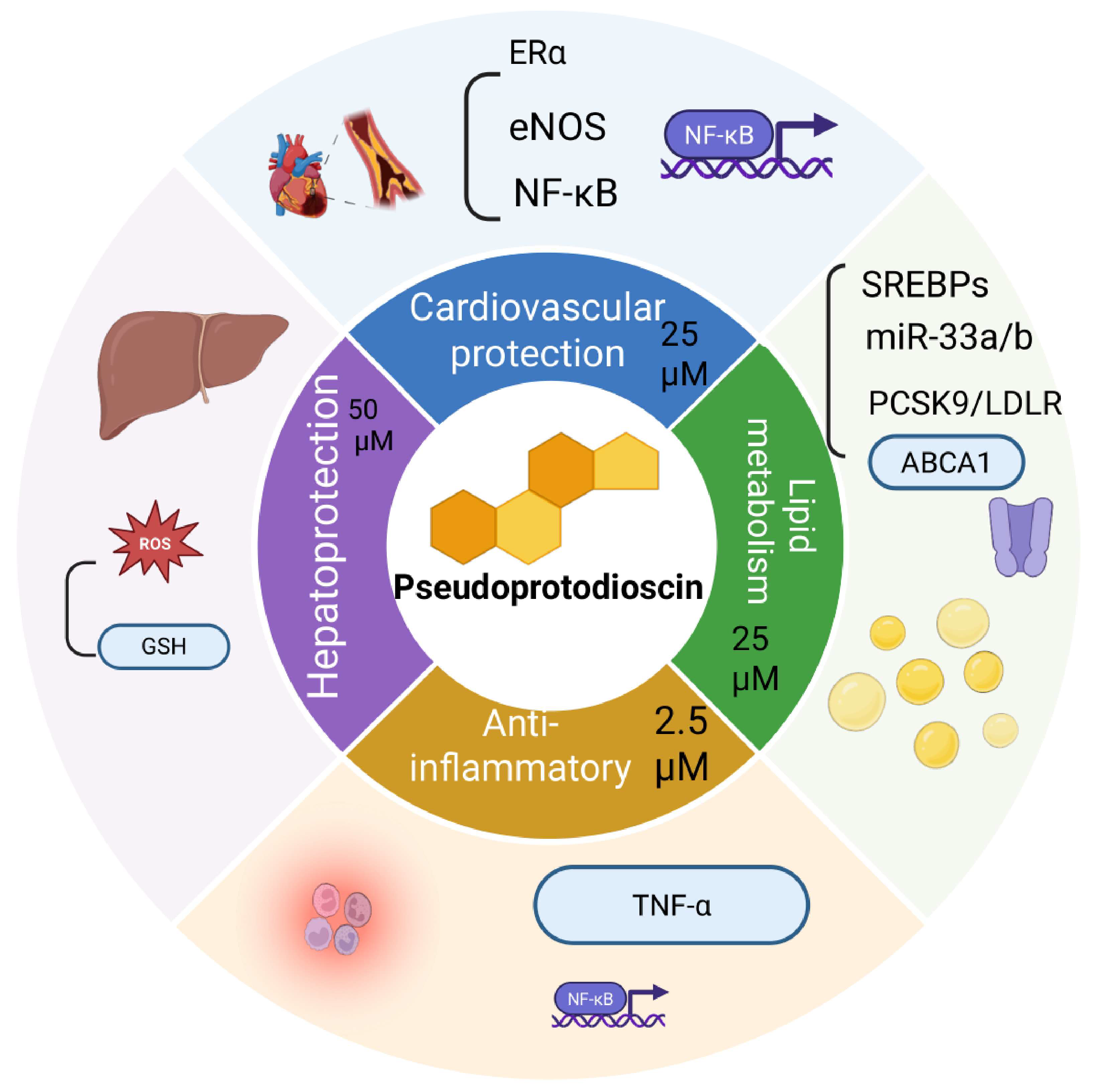
3. Pharmacology Activities and Mechanisms
3.1. Anticancer Effects by PPD
3.2. Against Cardiovascular Disease by PPD
3.3. Anti-Inflammatory Effects by PPD
3.4. Hepatoprotection Effects by PPD and Relevant Extracts
3.5. Other Bioactivity Effects
| Detection Method | Sample Preparation and Extraction Procedure | Content | Source | Production Area | Reference |
|---|---|---|---|---|---|
| Ultra-high-performance liquid chromatography (UPLC) | Powder mixed with methanol and heated under reflux for 30 min. | 0~0.874 mg/g | Paris polyphylla var. yunnanensis | Yunnan | [29] |
| High-performance liquid chromatography with evaporative light scattering detection (HPLC-ELSD) | Powder mixed with 70% ethanol and sonicated for 30 min (360 W power, 40 kHz frequency). | 7.78~19.36 mg/g | Dioscorea composita Hemsl. | Huize Country, Yunnan Province | [30] |
| HPLC | Powder mixed with 50% methanol and sonicated for 20 min. | 1.47~16.24 mg/g | Dioscorea nipponica Makino | Lingshou Country, Shijiazhuang City, Zhangjiakou City, Hebei Province | [35] |
| High-performance liquid chromatography coupled with quadrupole/time-of-flight mass spectrometry (HPLC-Q/TOF-MS) | Powder extracted with methanol and sonicated for 20 min. | 0.02~0.27 mg/g | The rhizome of Dioscorea nipponica Makino (RDN) | [78] | |
| HPLC-ELSD | Powder mixed with ethanol, allowed to stand overnight, and then sonicated for 30 min. | 5.443~6.670 mg/g | Dioscorea zingiberensis C. H. Wright | Zhechuan Country, Xishan Country, and Neixiang Country, Henan Province | [31] |
| Ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry (UPLC-QTOF/MS) | Powder mixed with ethanol and subjected to reflux extraction. | 13.821 ± 0.037 mg/mL | Dioscorea nipponica | Korean peninsula, Japan | [48] |
| HPLC | Powder mixed with ethanol overnight and subjected to ultrasound for 30 min. | Dioscorea pathaica | Cichuan Province, Guizhou Province, Yunan Province, Hunan Province | [79] | |
| HPLC | Powder mixed with methanol and sonicated for 20 min. | 0.037~0.187% | Discorea nipponica Makino | Jilin Province, Heilongjiang Province, Hebei Province | [80] |
| HPLC | Powder mixed with ethanol and ultrasonically treated for 40 min. | 0.7348%~1.0023% | Dioscorea zingiberensis C. H. Wright | Yunxi City, Hubei Province, Ankang City, Shanxi Province, Anhua City, Hunan Province | [81] |
| Ultra-high-performance liquid chromatography coupled with tandem mass spectrometry (UHPLC-MS/MS) | Powder mixed with ethanol and ultrasonically treated (250 W, 40 kHz) for 30 min. | 0.73 μg/g | Tribulus terrestris | - | [32] |
| HPLC | Slices crushed, mixed with 50% methanol, and ultrasonically treated for 30 min. | 421.04~1248.6 μg/g | Dioscoreae nipponicae Rhizoma | Haerbin City, Heilongjiang Province | [35] |
| HPLC | Powder mixed with ethanol and subjected to extraction. | 1.740~1.928 mg/g | Dioscorea nipponica L | Heilongjiang | [82] |
| HPLC | Powder mixed with ethanol and ultrasonically processed for 40 min (55 khz). | Dioscorea nipponica Makino | Heilongjiang | [83] | |
| HPLC | Sample extracted with 95% ethanol. | Dioscorea nipponica Makino | [84] | ||
| UHPLC-MS/MS | Powder mixed with ethanol and subjected to ultrasound for 30 min (250 W, 40 kHz). | 13.1~65.17 μg/g | P. kingianum | Guizhou Province, Yunnan Province | [33] |
| HPLC | Powder mixed with sulfuric acid, ultrasonically treated for 20 min, and then hydrolyzed for 4 h at 100 °C. | 0.4% | Dioscorea zingiberensis C. H. Wright | Henan | [85] |
| UPLC | Powder mixed with ethanol solution, heated, and subjected to reflux for 40 min at 85 °C. | Dioscorea zingiberensis C. H. Wright | Hunan, Hubei, Shaanxi, Gansu, Sichuan, and so on. | [86] | |
| Near-infrared reflectance spectroscopy (NIRS) | Powder mixed with ethanol and subjected to ultrasound for 30 min. | Dioscorea zingiberensis C. H. Wright | Henan, Shanxi, Hunan, Hubei | [87] | |
| Liquid chromatography–electrospray ionization–mass spectrometry (LC–ESI-MS) | Powder mixed with methanol. | - | Dioscorea nipponica Makino | [88] | |
| Liquid chromatography–mass spectrometry (LC-MS), HPLC | Powder mixed with ethanol and subjected to ultrasound at 60 °C and 53 kHz for 60 min. | - | Paris polyphylla var. yunnanensis | Cultured in vitro | [34] |
| Liquid chromatography with electrospray ionization tandem mass spectrometry (LC-ESI/MS/MS) | - | Tribulus terrestris | Bulgaria, Turkey, Greece, Serbia, Macedonia, Georgia, and Iran | [89] | |
| Guge Fengtong preparation (GGFT) | Matrix solid-phase dispersion extraction. Heat–reflux extraction. Ultrasonic extraction. | 0.0017~0.0462% | Guge Fengtong preparation (GGFT) | - | [90] |
| High-performance liquid chromatography with diode-array detection (HPLC-DVD) | Powder mixed with 80% methanol solution and subjected to ultrasound for 30 min. | 0.197~0.219 mg/g | Dieda Zhitong San | Anshan, China, Anshan Phsrmarceueical Co., Ltd. | [91] |
| HPLC | Powder mixed with 75% methanol and subjected to ultrasound (power: 300 W; frequency: 40 kHz) for 45 min. | 0.431~0.740 | GuGeFengTongPian | Tonghua, China, Tongyao Pharmaceutical Group Stock Co., Ltd. | [92] |
| High-performance liquid chromatography with quantitative analysis of multi-components by a single marker (HPLC-QAMS) | Powder mixed with 50% methanol and subjected to ultrasound (power: 500 W; frequency: 40 kHz) for 30 min. | 0.2580~0.3210 | ChuanLongGuCiPian (TaiJi) | Ziyang, China, Sichuan Hetai Pharmaceutical Company Limited | [93] |
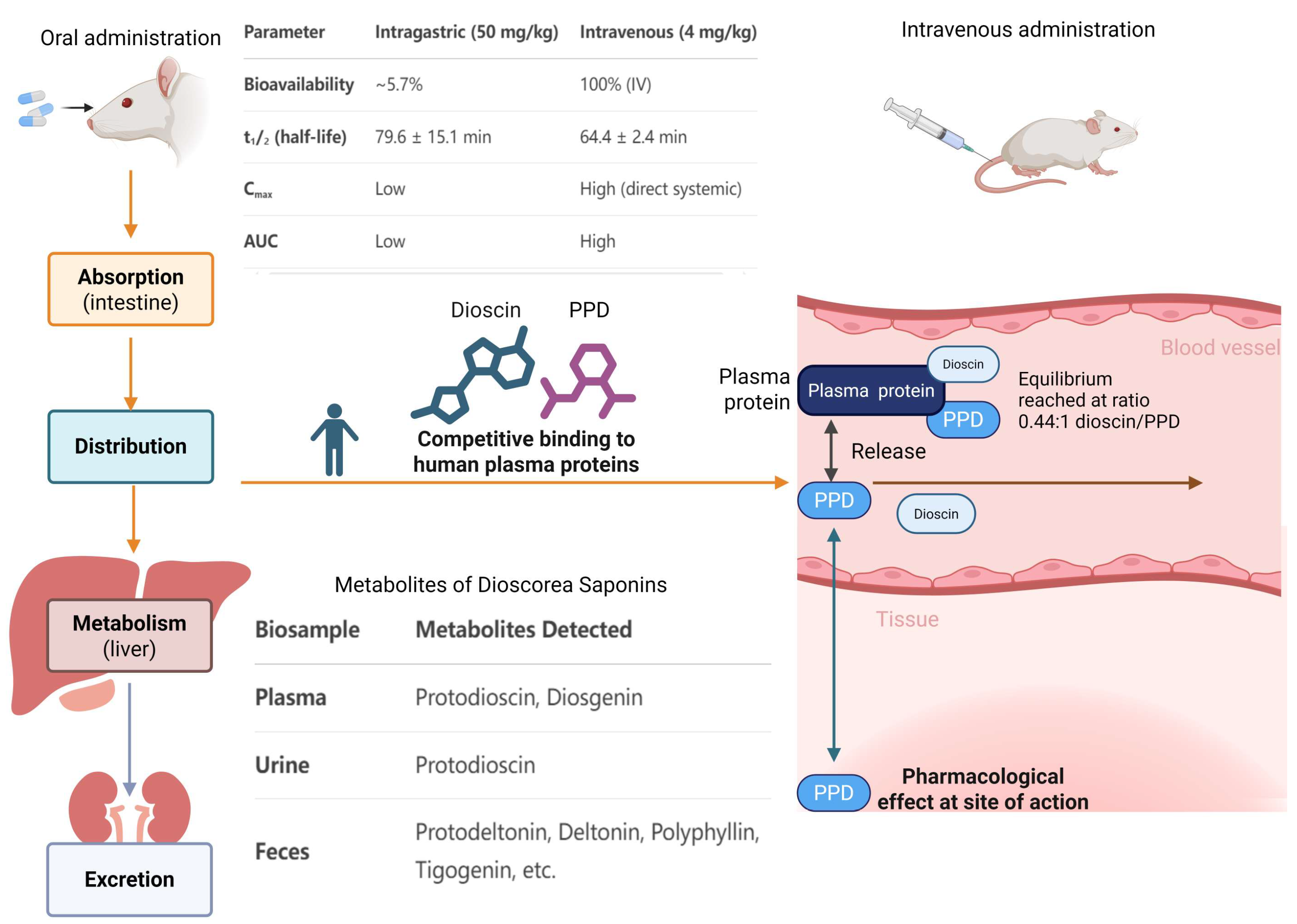
4. Pharmacokinetics
4.1. Pharmacokinetic Parameters
4.2. Drug–Drug Interaction
4.3. Metabolites
5. Clinical Applications of PPD in TCM
5.1. DXXK in the Treatment of Diabetic Kidney Injury
5.2. DXXK in the Treatment of Cardiotoxic Protection
| Clinical Prescription | Molecular Mechanisms | Biological Effects | Reference |
|---|---|---|---|
| Guge fengtong tablets | - | Rheumatic pain and numbness. | [92] |
| Guge fengtong tablets | - | Tonify the kidney, strengthen bones, promote blood circulation, and alleviate pain. | [93] |
| Chuanlongguci capsule | - | Hyperosteogeny. | [141] |
| Fengshi Bikang Jiaonang capsule | - | Rheumatoid arthritis belongs to the syndrome of cold-dampness obstructing the collaterals. | [159] |
| DXXK | Alleviates hyperlipidemia, fat accumulation, and the formation of atherosclerosis in ApoE−/− mice, reverses the expression of PCSK9 mRNA in liver tissues and circulating PCSK9 levels in ApoE−/− mice, and upregulates the expression of liver LDLR. | Alleviates lipid disorder and ameliorates atherosclerosis. | [160] |
| DXXK | Inhibits inflammatory reaction by regulating TLR4/MyD88/NF-κB signal transduction. | Inhibits inflammatory reaction. | [161] |
| DXXK | Reduces the blood viscosity in rats with blood stasis (p < 0.01); significantly decreases the Fibrinogen (FIB) content in rats with blood stasis, and prolongs prothrombin time (PT) and thrombin time (TT) (p < 0.05). | Prolongs the prothrombin time and reduces the fibrinogen content. | [162] |
| DXXK | Downregulates the expression of Cyclooxygenase-2 (COX-2) to inhibit LPS-induced PGE2 production without affecting the expression of COX-1. | Inflammatory mediator. | [163] |
| DXXK | Increases Superoxide Dismutase (SOD) activity and the expression of Mn-SOD mRNA in myocardial tissue; significantly reduces Malondialdehyde (MDA) levels. | Antioxidant. | [164] |
6. Conclusions and Future Prospects
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PPD | pseudoprotodioscin |
| TCM | Traditional Chinese Medicine |
| DXXK | Di’ ao Xin Xue Kang |
| HPLC | high-performance liquid chromatography |
| A375 | human malignant melanoma cell line |
| L929 | murine connective tissue fibroblast cell line |
| Hela | human cervical adenocarcinoma cell line |
| IC50 | half-maximal inhibitory concentration |
| TG | triglyceride |
| SREBP | sterol regulatory element-binding protein |
| ABCA1 | ATP-binding cassette transporter A1 |
| THP-1 | human acute monocytic leukemia cell line |
| HMGCR | 3-hydroxy-3-methylglutaryl-CoA reductase |
| FAS | fatty acid synthase |
| ACC | acetyl-CoA carboxylase |
| LDL | low-density lipoprotein |
| PCSK9 | proprotein convertase subtilisin/kexin type 9 |
| B16F1 cells | mouse melanoma cell line |
| HepG2 cells | human hepatocellular carcinoma cell line |
| GSH | glutathione |
| ROS | reactive oxygen species |
| ADME | absorption, distribution, metabolism, and excretion |
| t1/2 | half-lives |
| Cmax | maximum serum concentration |
| AUC | area under the curve |
| DM | diabetes mellitus |
| T1DM/T2DM | type 1 or type 2 diabetes mellitus |
| TGFβ1 | Transforming Growth Factor-βeta 1 |
| Smad2 | Sma and Mad-related protein 2 |
| Smad3 | Sma and Mad-related protein 3 |
| UPLC | ultra-high-performance liquid chromatography |
| HPLC-Q/TOF-MS | high-performance liquid chromatography coupled with quadrupole/time-of-flight mass spectrometry |
| HPLC-ELSD | high-performance liquid chromatography with evaporative light scattering detection |
| UPLC-QTOF/MS | ultra-performance liquid chromatography coupled with quadrupole time-of-flight mass spectrometry |
| UHPLC-MS/MS | ultra-high-performance liquid chromatography coupled with tandem mass spectrometry |
| NIRS | near-infrared reflectance spectroscopy |
| LC–ESI-MS | liquid chromatography–electrospray ionization–mass spectrometry |
| LC-MS | liquid chromatography–mass spectrometry |
| LC-ESI/MS/MS | liquid chromatography with electrospray ionization tandem mass spectrometry |
| GGTF | Guge Fengtong preparation |
| HPLC-DVD | high-performance liquid chromatography with diode-array detection |
| HPLC-QAMS | high-performance liquid chromatography with quantitative analysis of multi-components by a single marker |
| NF-κB | Nuclear Factor kappa-light-chain-enhancer of activated B cells |
| COX-2 | Cyclooxygenase-2 |
| SOD | Superoxide Dismutase |
| MDA | Malondialdehyde |
| Mn-SOD mRNA | Manganese-Superoxide Dismutase messenger Ribonucleic Acid |
| HIF-1α | Hypoxia-Inducible Factor 1-αlpha |
| FIB | fibrinogen |
| DP | Dioscorea Panthaica Prain et Burkill |
| PT | prothrombin time |
| TT | thrombin time |
References
- Liu, T. Proterties of Protodioscin-Glycosidase-1 and Molecular Cloning, Expression of Protodioscin-glycosidase-1Gene. Ph.D. Thesis, Yanbian University, Yanji, China, 2013. [Google Scholar]
- Wang, Q.; Yu, J.; Yang, W.; Niu, T.; Jeong, H.; Wang, X. Advances in chemical constituents and biological activities of Dioscorea rhizome. J. Chin. Med. Mater. 2023, 46, 1570–1578. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, L.; Liu, J.; Wang, W.; Yu, H.; Li, J.; Chen, Q.; Wang, T. Effect and mechanism of dioscin from Dioscorea spongiosa on uric acid excretion in animal model of hyperuricemia. J. Ethnopharmacol. 2018, 214, 29–36. [Google Scholar] [CrossRef] [PubMed]
- Kawabata, T.; Cui, M.Y.; Hasegawa, T.; Takano, F.; Ohta, T. Anti-inflammatory and anti-melanogenic steroidal saponin glycosides from Fenugreek (Trigonella foenum-graecum L.) seeds. Planta Med. 2011, 77, 705–710. [Google Scholar] [CrossRef] [PubMed]
- Han, J.; Shi, G.; Li, W.; Xie, Y.; Li, F.; Jiang, D. Preventive effect of dioscin against monosodium urate-mediated gouty arthritis through inhibiting inflammasome NLRP3 and TLR4/NF-κappaB signaling pathway activation: An in vivo and in vitro study. J. Nat. Med. 2021, 75, 37–47. [Google Scholar] [CrossRef]
- Yin, J.; Tezuka, Y.; Kouda, K.; Tran, Q.L.; Miyahara, T.; Chen, Y.; Kadota, S. Antiosteoporotic Activity of the Water Extract of Dioscorea spongiosa. Biol. Pharm. Bull. 2004, 27, 583–586. [Google Scholar] [CrossRef]
- Katz, L.; Baltz, R.H. Natural product discovery: Past, present, and future. J. Ind. Microbiol. Biotechnol. 2016, 43, 155–176. [Google Scholar] [CrossRef]
- Song, Y.; Zhang, M.; Chang, S.; Chu, G.; Ji, H. DerivaPredict: A User-Friendly Tool for Predicting and Evaluating Active Derivatives of Natural Products. Molecules 2025, 30, 1683. [Google Scholar] [CrossRef]
- Dasari, S.; Njiki, S.; Mbemi, A.; Yedjou, C.G.; Tchounwou, P.B. Pharmacological Effects of Cisplatin Combination with Natural Products in Cancer Chemotherapy. Int. J. Mol. Sci. 2022, 23, 1532. [Google Scholar] [CrossRef]
- Fan, G.; Li, F.; Wang, P.; Jin, X.; Liu, R. Natural-Product-Mediated Autophagy in the Treatment of Various Liver Diseases. Int. J. Mol. Sci. 2022, 23, 15109. [Google Scholar] [CrossRef]
- Gao, C.L.; Song, J.Q.; Yang, Z.N.; Wang, H.; Wu, X.Y.; Shao, C.; Dai, H.X.; Chen, K.; Guo, Y.W.; Pang, T.; et al. Chemoproteomics of Marine Natural Product Naamidine J Unveils CSE1L as a Therapeutic Target in Acute Lung Injury. J. Am. Chem. Soc. 2024, 146, 28384–28397. [Google Scholar] [CrossRef]
- Zhao, H.; Yang, Y.; Wang, S.; Yang, X.; Zhou, K.; Xu, C.; Zhang, X.; Fan, J.; Hou, D.; Li, X.; et al. NPASS database update 2023: Quantitative natural product activity and species source database for biomedical research. Nucleic Acids Res. 2023, 51, D621–D628. [Google Scholar] [CrossRef]
- Adegboye, O.; Field, M.A.; Kupz, A.; Pai, S.; Sharma, D.; Smout, M.J.; Wangchuk, P.; Wong, Y.; Loiseau, C. Natural-Product-Based Solutions for Tropical Infectious Diseases. Clin. Microbiol. Rev. 2021, 34, e0034820. [Google Scholar] [CrossRef] [PubMed]
- Chopra, B.; Dhingra, A.K. Natural products: A lead for drug discovery and development. Phytother. Res. 2021, 35, 4660–4702. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Song, J.; Kong, L.; Yuan, T.; Li, W.; Zhang, W.; Hou, B.; Lu, Y.; Du, G. The strategies and techniques of drug discovery from natural products. Pharmacol. Ther. 2020, 216, 107686. [Google Scholar] [CrossRef] [PubMed]
- Specchio, N.; Pietrafusa, N.; Cross, H.J. Source of cannabinoids: What is available, what is used, and where does it come from? Epileptic Disord. 2020, 22, 1–9. [Google Scholar] [CrossRef]
- Thomas, E.; Stewart, L.E.; Darley, B.A.; Pham, A.M.; Esteban, I.; Panda, S.S. Plant-Based Natural Products and Extracts: Potential Source to Develop New Antiviral Drug Candidates. Molecules 2021, 26, 6197. [Google Scholar] [CrossRef]
- Thibodeaux, C.J.; Melançon, C.E., 3rd; Liu, H.W. Natural-product sugar biosynthesis and enzymatic glycodiversification. Angew. Chem. Int. Ed. Engl. 2008, 47, 9814–9859. [Google Scholar] [CrossRef]
- Courdavault, V.; O’Connor, S.E.; Jensen, M.K.; Papon, N. Metabolic engineering for plant natural products biosynthesis: New procedures, concrete achievements and remaining limits. Nat. Prod. Rep. 2021, 38, 2145–2153. [Google Scholar] [CrossRef]
- Nelson, S.; Parkinson, E.I. Synthetic-bioinformatic natural product-inspired peptides. Nat. Prod. Rep. 2025, 42, 50–66. [Google Scholar] [CrossRef]
- Sharma, A.N.; Verma, R. Source, Synthesis, and Biological Evaluation of Natural Occurring 2,2′-Bipyridines. Chem. Biodivers. 2023, 20, e202300764. [Google Scholar] [CrossRef]
- Alqathama, A.A.; Ahmad, R.; Alsaedi, R.B.; Alghamdi, R.A.; Abkar, E.H.; Alrehaly, R.H.; Abdalla, A.N. The vital role of animal, marine, and microbial natural products against COVID-19. Pharm. Biol. 2022, 60, 509–524. [Google Scholar] [CrossRef] [PubMed]
- Yeshi, K.; Morisco, P.; Wangchuk, P. Animal-derived natural products of Sowa Rigpa medicine: Their pharmacopoeial description, current utilization and zoological identification. J. Ethnopharmacol. 2017, 207, 192–202. [Google Scholar] [CrossRef] [PubMed]
- Li, F.J.; Hu, J.H.; Ren, X.; Zhou, C.M.; Liu, Q.; Zhang, Y.Q. Toad venom: A comprehensive review of chemical constituents, anticancer activities, and mechanisms. Arch. Pharm. 2021, 354, e2100060. [Google Scholar] [CrossRef] [PubMed]
- Sahayasheela, V.J.; Lankadasari, M.B.; Dan, V.M.; Dastager, S.G.; Pandian, G.N.; Sugiyama, H. Artificial intelligence in microbial natural product drug discovery: Current and emerging role. Nat. Prod. Rep. 2022, 39, 2215–2230. [Google Scholar] [CrossRef]
- Put, H.; Gerstmans, H.; Vande Capelle, H.; Fauvart, M.; Michiels, J.; Masschelein, J. Bacillus subtilis as a host for natural product discovery and engineering of biosynthetic gene clusters. Nat. Prod. Rep. 2024, 41, 1113–1151. [Google Scholar] [CrossRef]
- Ruan, J.; Liu, Y.; Chao, L.; Wang, T.; Liu, E.; Zhang, Y. Isolation and identification of steroidal saponins from Dioscorea spongiosa II. J. Shenyang Pharm. Univ. 2016, 33, 438–443. [Google Scholar] [CrossRef]
- Wang, J.; Gao, H.; Guan, Y.; Meng, X. Overview of Research and Application of Dioscorea Research and Practice on Chinese Medicines. Chin. Med. J. Res. Prac. 2019, 33, 83–86. [Google Scholar] [CrossRef]
- Zan, K.; Gao, Y.; Zheng, J.; Cui, G.; Liu, J.; Guo, L.; Ma, S. Simultaneous determination of eleven steroidal saponins in cultivated Paris polyphylla Smith var. yunnanensis (Franch.) Hand.-Mazz. by HPLC. Chin. J. Pharm. Anal. 2017, 37, 1572–1577. [Google Scholar] [CrossRef]
- Ma, J.; Cui, N.; Yu, H.; Li, Z.; Liu, E.; Bie, S. Effect of drying methods on the active components in Dioscorea composita Hemsl. based on simultaneous determination of 4 saponins. Cent. South Pharm. 2021, 19, 413–418. [Google Scholar]
- Bai, Y.; Zuo, C.; Xie, C.; Lei, J.; Li, L. HPLC-ELSD Determination of Pseudoprodioscin in Dioscorea zingiberensis. Chin. J. Exp. Tradit. Med. Formulae 2012, 18, 84–86. [Google Scholar] [CrossRef]
- Liu, K.; Wang, L.; Han, B.; Lyu, X.; Shi, L.; Zhang, C. Effects of Different Stir-Frying Temperature and Time on the Contents of Four Dioscin Components of Tribuli Fructus. J. Chin. Med. Mater. 2022, 45, 67–72. [Google Scholar] [CrossRef]
- Wang, S.; Wang, L.; Fang, J.; Liu, K.; Wang, Y.; Zhang, C. Correlation Analysis Between Color and Content Changes of Five Components of Wine-processed Polygonatum kingianum Rhizoma During Processing. Chin. J. Exp. Tradit. Med. Formulae 2022, 28, 156–162. [Google Scholar] [CrossRef]
- Zhang, Y.; Jin, L.; Wu, Y.; Li, R.; Wang, K.; Li, H. Chemical constituents of steroidal saponins in rhizome of Paris polyphylla var. yunnanensis cultured in vitro. China J. Chin. Mater. Med. 2021, 46, 4936–4944. [Google Scholar] [CrossRef]
- Yang, G.; Chang, Y.; Xue, Z.; Song, J.; Zhang, D.; Zheng, Y.; Guo, L. Quality control of Dioscoreae nipponicae Rhizoma based on fingerprint and quantitative analysis. China J. Chin. Mater. Med. 2020, 45, 4949–4956. [Google Scholar] [CrossRef]
- Bray, F.; Laversanne, M.; Weiderpass, E.; Soerjomataram, I. The ever-increasing importance of cancer as a leading cause of premature death worldwide. Cancer 2021, 127, 3029–3030. [Google Scholar] [CrossRef] [PubMed]
- Filho, A.M.; Laversanne, M.; Ferlay, J.; Colombet, M.; Piñeros, M.; Znaor, A.; Parkin, D.M.; Soerjomataram, I.; Bray, F. The GLOBOCAN 2022 cancer estimates: Data sources, methods, and a snapshot of the cancer burden worldwide. Int. J. Cancer 2025, 156, 1336–1346. [Google Scholar] [CrossRef] [PubMed]
- Biller, L.H.; Schrag, D. Diagnosis and Treatment of Metastatic Colorectal Cancer: A Review. JAMA 2021, 325, 669–685. [Google Scholar] [CrossRef]
- Cao, W.; Chen, H.; Yu, Y.; Li, N.; Chen, W. Changing profiles of cancer burden worldwide and in China: A secondary analysis of the global cancer statistics 2020. Chin. Med. J. 2021, 134, 783–791. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Sun, Y.; Sha, Y.; Cui, G.; Meng, F.; Zhong, Z. Lysosomal-mediated drug release and activation for cancer therapy and immunotherapy. Adv. Drug Deliv. Rev. 2022, 192, 114624. [Google Scholar] [CrossRef]
- Jiang, Y.; Jiang, Z.; Wang, M.; Ma, L. Current understandings and clinical translation of nanomedicines for breast cancer therapy. Adv. Drug Deliv. Rev. 2022, 180, 114034. [Google Scholar] [CrossRef]
- Guo, W.; Wang, H.; Li, C. Signal pathways of melanoma and targeted therapy. Signal Transduct. Target. Ther. 2016, 6, 424. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Cao, J.; Topatana, W.; Juengpanich, S.; Li, S.; Zhang, B.; Shen, J.; Cai, L.; Cai, X.; Chen, M. Targeting mutant p53 for cancer therapy: Direct and indirect strategies. J. Hematol. Oncol. 2021, 14, 157. [Google Scholar] [CrossRef] [PubMed]
- Punekar, S.R.; Velcheti, V.; Neel, B.G.; Wong, K. The current state of the art and future trends in RAS- targeted cancer therapies. Clin. Oncol. 2022, 19, 637–655. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Shi, M.; Ren, Y.; Xu, H.; Weng, S.; Ning, W.; Ge, X.; Liu, L.; Guo, C.; Duo, M.; et al. Recent advances and applications of CRISPR-Cas9 in cancer immunotherapy. Mol. Cancer 2023, 22, 35. [Google Scholar] [CrossRef]
- Dong, M.; Feng, X.; Wu, L.; Wang, B.; Ikejima, T. Two new steroidal saponins from the rhizomes of Dioscorea panthaica and their cytotoxic activity. Planta Med. 2001, 67, 853–857. [Google Scholar] [CrossRef]
- Hwang, J.; Park, K.; Ryuk, J.; Kim, H.; Ko, B. Development of an Oriental Medicine Discrimination Method through Analysis of Steroidal Saponins in Dioscorea nipponica Makino and Their Anti-Osteosarcoma Effects. Molecules 2019, 24, 4022. [Google Scholar] [CrossRef]
- Tang, Q. Inhibitory Effects of Pseudoprodioscin on Endometrial Carcinoma by Regulating miR-182-5P-FoxO1. Master’s Thesis, Tianjin Medical University, Tianjin, China, 2020. [Google Scholar]
- Shufelt, C.L.; Pacheco, C.; Tweet, M.S.; Miller, V.M. Sex-Specific Physiology and Cardiovascular Disease. Adv. Exp. Med. Biol. 2018, 1065, 433–454. [Google Scholar] [CrossRef]
- Nadruz, W., Jr.; Kitzman, D.; Windham, B.G.; Kucharska-Newton, A.; Butler, K.; Palta, P.; Griswold, M.E.; Wagenknecht, L.E.; Heiss, G.; Solomon, S.D.; et al. Cardiovascular Dysfunction and Frailty Among Older Adults in the Community: The ARIC Study. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 72, 958–964. [Google Scholar] [CrossRef]
- Totoń-Żurańska, J.; Mikolajczyk, T.P.; Saju, B.; Guzik, T.J. Vascular remodelling in cardiovascular diseases: Hypertension, oxidation, and inflammation. Clin. Sci. 2024, 138, 817–850. [Google Scholar] [CrossRef]
- Bourdrel, T.; Bind, M.-A.; Béjot, Y.; Morel, O.; Argach, J.-F. Cardiovascular effects of air pollution. Annu. Rev. Med. 2022, 73, 393–406. [Google Scholar] [CrossRef]
- Mallet, R.T.; Burtscher, J.; Richalet, J.P.; Millet, G.P.; Burtscher, M. Impact of High Altitude on Cardiovascular Health: Current Perspectives. Vasc. Health Risk Manag. 2021, 17, 317–335. [Google Scholar] [CrossRef]
- Thomas, M. Up in the air: Links between the environment and cardiovascular disease. Cardiovasc. Res. 2019, 115, e144–e146. [Google Scholar] [CrossRef] [PubMed]
- Kondo, T.; Nakano, Y.; Adachi, S.; Murohara, T. Effects of Tobacco Smoking on Cardiovascular Disease. Circ. J. 2019, 83, 1980–1985. [Google Scholar] [CrossRef] [PubMed]
- Roerecke, M. Alcohol’s Impact on the Cardiovascular System. Nutrients 2021, 13, 3419. [Google Scholar] [CrossRef] [PubMed]
- Osborne, M.T.; Shin, L.M.; Mehta, N.N.; Pitman, R.K.; Fayad, Z.; Tawakol, A. Disentangling the Links between Psychosocial Stress and Cardiovascular Disease. Circ. Cardiovasc. Imaging 2020, 13, e010931. [Google Scholar] [CrossRef]
- Merz, A.; Cheng, S. Sex differences in cardiovascular ageing. Heart 2016, 102, 825–831. [Google Scholar] [CrossRef]
- Karki, P.K.; Chalise, S.; Kayastha, R.; Khakurel, G. Atherogenic Index of Plasma in Postmenopausal Women. J. Nepal Health Res. Counc. 2018, 16, 175–177. [Google Scholar] [CrossRef]
- Xiao, J.; Yin, Y.-z.; Sun, B. An In Vivo Estrogen Deficiency Mouse Model for Screening Exogenous Estrogen Treatments of Cardiovascular Dysfunction After Menopause. J. Vis. Exp. 2019, 150, e59536. [Google Scholar] [CrossRef]
- Gai, Y.; Li, Y.; Xu, Z.; Chen, J. Pseudoprotodioscin inhibits SREBPs and microRNA 33a/b levels and reduces the gene expression regarding the synthesis of cholesterol and triglycerides. Fitoterapia 2019, 139, 104393. [Google Scholar] [CrossRef]
- Zarrin, A.A.; Bao, K.; Lupardus, P.; Vucic, D. Kinase inhibition in autoimmunity and inflammation. Nat. Rev. Drug Discov. 2021, 20, 39–63. [Google Scholar] [CrossRef]
- Soehnlein, O.; Libby, P. Targeting inflammation in atherosclerosis—From experimental insights to the clinic. Nat. Rev. Drug Discov. 2021, 20, 589–610. [Google Scholar] [CrossRef]
- Leńska-Mieciek, M.; Madetko-Alster, N.; Alster, P.; Królicki, L.; Fiszer, U.; Koziorowski, D. Inflammation in multiple system atrophy. Front. Immunol. 2023, 14, 1214677. [Google Scholar] [CrossRef]
- Fullerton, J.N.; Gilroy, D.W. Resolution of inflammation: A new therapeutic frontier. Nat. Rev. Drug Discov. 2016, 15, 551–567. [Google Scholar] [CrossRef]
- Lin, Y.-J.; Anzaghe, M.; Schülke, S. Update on the Pathomechanism, Diagnosis, and Treatment Options for Rheumatoid Arthritis. Cells 2020, 9, 880. [Google Scholar] [CrossRef] [PubMed]
- Lontchi-Yimagou, E.; Sobngwi, E.; Matsha, T.E.; Kengne, A.P. Diabetes Mellitus and Inflammation. Curr. Diabetes Rep. 2013, 13, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Kong, P.; Cui, Z.; Huang, X.F.; Zhang, D.D.; Guo, R.J.; Han, M. Inflammation and atherosclerosis: Signaling pathways and therapeutic intervention. Signal Transduct. Target. Ther. 2022, 7, 131. [Google Scholar] [CrossRef] [PubMed]
- Feng, D.; Hwang, S.; Guillot, A.; Wang, Y.; Guan, Y.; Chen, C.; Maccioni, L.; Gao, B. Inflammation in Alcohol-Associated Hepatitis: Pathogenesis and Therapeutic Targets. Cell. Mol. Gastroenterol. Hepatol. 2024, 18, 101352. [Google Scholar] [CrossRef]
- Sun, B.; Yang, D.; Yin, Y.; Xiao, J. Estrogenic and anti-inflammatory effects of pseudoprotodioscin in atherosclerosis-prone mice: Insights into endothelial cells and perivascular adipose tissues. Eur. J. Pharmacol. 2020, 869, 172887. [Google Scholar] [CrossRef]
- Jiménez-Arellanes, M.A.; Gutiérrez-Rebolledo, G.A.; Meckes-Fischer, M.; León-Díaz, R. Medical plant extracts and natural compounds with a hepatoprotective effect against damage caused by antitubercular drugs: A review. Asian Pac. J. Trop. Med. 2016, 9, 1141–1149. [Google Scholar] [CrossRef]
- Pareek, A.; Pant, M.; Gupta, M.M.; Kashania, P.; Ratan, Y.; Jain, V.; Pareek, A.; Chuturgoon, A.A. Moringa oleifera: An Updated Comprehensive Review of Its Pharmacological Activities, Ethnomedicinal, Phytopharmaceutical Formulation, Clinical, Phytochemical, and Toxicological Aspects. Int. J. Mol. Sci. 2023, 24, 2098. [Google Scholar] [CrossRef]
- Prajapati, J.; Bhatt, N.; Rawal, R. Hepatoprotective effects of phytochemicals and plant extracts against chemotherapy-induced liver damage in animal models: A systematic review. Arch. Toxicol. 2025, 99, 887–914. [Google Scholar] [CrossRef] [PubMed]
- Pereira, C.; Barros, L.; Ferreira, I.C.F.R. Extraction, identification, fractionation and isolation of phenolic compounds in plants with hepatoprotective effects. J. Sci. Food Agric. 2015, 96, 1068–1084. [Google Scholar] [CrossRef] [PubMed]
- Siddiqui, M.A.; Ali, Z.; Chittiboyina, A.G.; Khan, I.A. Hepatoprotective Effect of Steroidal Glycosides from Dioscorea villosa on Hydrogen Peroxide-Induced Hepatotoxicity in HepG2 Cells. Front. Pharmacol. 2018, 9, 797. [Google Scholar] [CrossRef] [PubMed]
- Aquino, R.; Conti, C.; De Simone, F.; Orsi, N.; Pizza, C.; Stein, M.L. Antiviral Activity of Constituents of Tamus communis. J. Chemother. 2016, 3, 305–309. [Google Scholar] [CrossRef]
- Yang, G.; Liu, P.; Shi, H.; Fan, W.; Feng, X.; Chen, J.; Jing, S.; Wang, L.; Zheng, Y.; Zhang, D.; et al. Identification of anti-inflammatory components in Dioscorea nipponica Makino based on HPLC-MS/MS, quantitative analysis of multiple components by single marker and chemometric methods. J. Chromatogr. B 2022, 1213, 123531. [Google Scholar] [CrossRef]
- Jin, W.; Zhang, Q.; Liu, A. Determination of pseudoprotodiosgenin in Huangshan medicinal materials by HPLC. In Proceedings of the 2009 National Symposium on Traditional Chinese Medicine, Guangzhou, China, 8–11 November 2009; pp. 175–179. [Google Scholar]
- Shang, H.; Zheng, Y.; Wang, M.; Bai, R.; Zhang, B.; Liao, M. Determination of protodioscin, protogracillin, methyl protodioscin, pseudoprotodioscin, dioscin and gracillin in Dioscoreae Nipponicae Rhizoma from different habitats by RP-HPLC. Drugs Clin. 2018, 33, 1879–1883. [Google Scholar]
- Zuo, C.; Lv, Y.; Zhang, X. Comprehensive evaluation and comparison on the quality of Dioscorea zingiberensis C.H. Wright from different growing areas. Prac. J. Med. Pharm. 2016, 12, 1108–1111. [Google Scholar] [CrossRef]
- Yu, S.; Liu, S.; Shao, J. Fingerprint Analysis of the Methanol Extract of Dioscorea nipponica. Herald Med. 2010, 29, 664–666. [Google Scholar]
- Meng, X.; Yu, D.; Yang, G.; Sun, H.; Wang, X. The Effect of Genders and Flower Removal on the Dioscin, Protodioscin and Total Saponin Contents in Dioscorea nipponica. World Sci. Technol./Mod. Tradit. Chin. Med. Mater. Med. 2010, 12, 250–253. [Google Scholar] [CrossRef]
- Zhang, Y.; Pan, J.; Mo, C.; Feng, K.; Chen, X.; Bi, K. Isolation and identification of steroidal saponins from Dioscorea zingiberensis. Cent. South Pharm. 2012, 10, 443–445. [Google Scholar]
- Xie, C.; Hu, Y.; Zuo, C.; Yan, B.; Jingwei, L.; Wang, X. Quality Standard of Dioscorea zingiberensis. Chin. J. Exp. Tradit. Med. Formulae 2013, 19, 109–113. [Google Scholar]
- Tao, X.; Gong, H.; Xie, C.; Zhang, J.; Li, Y.; Geng, X.; Liu, Q.; Lei, J. Quality evaluation of Dioscorea zingiberensis from different origins based on UPLC fingerprint and chemometrics. Chin. Tradit. Herb. Drugs 2021, 52, 227–233. [Google Scholar] [CrossRef]
- Xie, C.; Zuo, C.; Bai, Y.; Lei, J. Rapid Determination of Pseudoprodioscin in Dioscorea zingiberensis by Near Infrared Reflectance Spectroscopy. China Pharm. 2013, 24, 1396–1399. [Google Scholar] [CrossRef]
- Lin, S.; Wang, D.; Yang, D.; Yao, J.; Tong, Y.; Chen, J. Characterization of steroidal saponins in crude extract from Dioscorea nipponica Makino by liquid chromatography tandem multi-stage mass spectrometry. Anal. Chim. Acta 2007, 599, 98–106. [Google Scholar] [CrossRef]
- Dinchev, D.; Janda, B.; Evstatieva, L.; Oleszek, W.; Aslani, M.R.; Kostova, I. Distribution of steroidal saponins in Tribulus terrestris from different geographical regions. Phytochemistry 2008, 69, 176–186. [Google Scholar] [CrossRef]
- Cheng, X.; Qi, L.; Wang, Q.; Liu, X.; Boubertakh, B.; Wan, J.; Liu, E.-H.; Li, P. Highly efficient sample preparation and quantification of constituents from traditional Chinese herbal medicines using matrix solid-phase dispersion extraction and UPLC-MS/MS. Analyst 2013, 138, 2279–2288. [Google Scholar] [CrossRef]
- Gao, S.; Bai, X.; Wen, L.; Li, Z.; Fang, Z. Determination of six kinds of index components in Dieda Zhitong San by HPLC-DVD variable wavelength method. Chin. Tradit. Herb. Drugs 2016, 47, 2863–2867. [Google Scholar] [CrossRef]
- Ji, S.; Zhou, H.; Wang, Y.; Yin, X. Comprehensive quality evaluation of Guge fengtong tablets. China Pharm. 2024, 35, 948–954. [Google Scholar] [CrossRef]
- Zhong, L.; Gao, J.; Chen, H.; Li, C. Comprehensive quality evaluation of Chuanlong bone spur slices based on HPLC-QAMS multi-index component quantitative control and chemometrics. Chin. Med. 2022, 25, 607–613. [Google Scholar]
- Hochhaus, G.; Barrett, J.S.; Derendorf, H. Evolution of pharmacokinetics and pharmacokinetic/dynamic correlations during the 20th century. J. Clin. Pharmacol. 2000, 40, 908–917. [Google Scholar] [CrossRef]
- Batchelor, H.K.; Marriott, J.F. Paediatric pharmacokinetics: Key considerations. Br. J. Clin. Pharmacol. 2015, 79, 395–404. [Google Scholar] [CrossRef] [PubMed]
- Guidi, M.; Csajka, C.; Buclin, T. Parametric Approaches in Population Pharmacokinetics. J. Clin. Pharmacol. 2022, 62, 125–141. [Google Scholar] [CrossRef] [PubMed]
- Liao, M.; Chen, X.; Chen, J.; Liu, M.; Wang, J.; Chen, Z.; Xie, Z.; Yao, M. Determination of pseudoprotodioscin in rat plasma by UPLC–MS/MS: Assay development and application to pharmacokinetic study. J. Chromatogr. B 2016, 1026, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Wang, Y.; Gu, J.; Chen, X.; Zhong, D. Determination of dioscin in rat plasma by liquid chromatography-tandem mass spectrometry. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2005, 817, 271–275. [Google Scholar] [CrossRef]
- Du, D.; Gao, B.; Xin, G.; Sun, A.; Huang, B.; Zhang, R.; Xing, Z.; Chen, Q.; He, Y.; Huang, W. Determination of deltonin in rat plasma by using HPLC-MS/MS and the application of this method in pharmacokinetic studies. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2013, 931, 1–5. [Google Scholar] [CrossRef]
- Shah, S.; Dhawan, V.; Holm, R.; Nagarsenker, M.S.; Perrie, Y. Liposomes: Advancements and innovation in the manufacturing process. Adv. Drug Deliv. Rev. 2020, 154–155, 102–122. [Google Scholar] [CrossRef]
- Perumal, S.; Atchudan, R.; Lee, W. A Review of Polymeric Micelles and Their Applications. Polymers 2022, 14, 2510. [Google Scholar] [CrossRef]
- Santonocito, D.; Puglia, C. Applications of Lipid-based Nanocarriers for Parenteral Drug Delivery. Curr. Med. Chem. 2022, 29, 4152–4169. [Google Scholar] [CrossRef]
- Garcia-Fuentes, M.; Gonzalez-Díaz, H.; Csaba, N. Nanocarriers & drug delivery: Rational design and applications. Curr. Top. Med. Chem. 2014, 14, 1095–1096. [Google Scholar] [CrossRef]
- Liao, M.; Dai, C.; Liu, M.; Chen, J.; Chen, Z.; Xie, Z.; Yao, M. Simultaneous determination of four furostanol glycosides in rat plasma by UPLC–MS/MS and its application to PK study after oral administration of Dioscorea nipponica extracts. J. Pharm. Biomed. Anal. 2016, 117, 372–379. [Google Scholar] [CrossRef]
- Di, L. An update on the importance of plasma protein binding in drug discovery and development. Expert Opin. Drug Discov. 2021, 16, 1453–1465. [Google Scholar] [CrossRef]
- Schulz, J.A.; Stresser, D.M.; Kalvass, J.C. Plasma protein-mediated uptake and contradictions to the free drug hypothesis: A critical review. Drug Metab. Rev. 2023, 55, 205–238. [Google Scholar] [CrossRef]
- Momper, J.D.; Nikanjam, M.; Best, B.M.; Mirochnick, M.; Capparelli, E.V.; Cressey, T.R. Brief Report: Dolutegravir Plasma Protein Binding and Unbound Concentrations During Pregnancy and Postpartum. J. Acquir. Immune Defic. Syndr. 2023, 94, 332–336. [Google Scholar] [CrossRef]
- Ulldemolins, M.; Roberts, J.A.; Rello, J.; Paterson, D.L.; Lipman, J. The effects of hypoalbuminaemia on optimizing antibacterial dosing in critically ill patients. Clin. Pharmacokinet. 2011, 50, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Darandale, S.S.; Shevalkar, G.B.; Vavia, P.R. Effect of Lipid Composition in Propofol Formulations: Decisive Component in Reducing the Free Propofol Content and Improving Pharmacodynamic Profiles. AAPS PharmSciTech 2017, 18, 441–450. [Google Scholar] [CrossRef]
- Sjögren, E.; Andersson, S.; Sundgren-Andersson, A.K.; Halldin, M.M.; Stålberg, O. Assessment of Free Drug Concentration in Cyclodextrin Formulations Is Essential to Determine Drug Potency in Functional In Vitro Assays. J. Pharm. Sci. 2016, 105, 2913–2920. [Google Scholar] [CrossRef] [PubMed]
- Xu, Q.; Sang, Y.; Gao, A.; Li, L. The effects of drug-drug interaction on linezolid pharmacokinetics: A systematic review. Eur. J. Clin. Pharmacol. 2024, 80, 785–795. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Leng, J.; Liu, Y. Deep learning for drug-drug interaction extraction from the literature: A review. Brief. Bioinform. 2020, 21, 1609–1627. [Google Scholar] [CrossRef]
- Li, Y.; Talebi, Z.; Chen, X.; Sparreboom, A.; Hu, S. Endogenous Biomarkers for SLC Transporter-Mediated Drug-Drug Interaction Evaluation. Molecules 2021, 26, 5500. [Google Scholar] [CrossRef]
- Alemayehu, T.; Geremew, G.; Tegegne, A.; Tadesse, G.; Getachew, D.; Ayele, H.; Yazie, A.; Fentahun, S.; Abebe, T.; Minwagaw, T.; et al. Drug-drug interaction among elderly patients in Africa: A systematic review and meta-analysis. BMC Pharmacol. Toxicol. 2025, 26, 92. [Google Scholar] [CrossRef]
- Fravel, M.A.; Ernst, M. Drug Interactions with Antihypertensives. Curr. Hypertens. Rep. 2021, 23, 14. [Google Scholar] [CrossRef]
- Humphreys, S.; Davis, J.; Iqbal, S.; Kamel, A.; Kulmatycki, K.; Lao, Y.; Liu, X.; Rodgers, J.; Snoeys, J.; Vigil, A.; et al. Considerations and recommendations for assessment of plasma protein binding and drug-drug interactions for siRNA therapeutics. Nucleic Acids Res. 2022, 50, 6020–6037. [Google Scholar] [CrossRef] [PubMed]
- Ali, M.; Al-Lohedan, H. Experimental and Computational Investigation on the Interaction of Anticancer Drug Gemcitabine with Human Plasma Protein: Effect of Copresence of Ibuprofen on the Binding. Molecules 2022, 27, 1635. [Google Scholar] [CrossRef] [PubMed]
- Lv, X.; Wang, Z.; Wang, Z.; Yin, H.; Xia, Y.; Jiang, L.; Liu, Y. Drug-drug interaction potentials of tucatinib inhibition of human UDP-glucuronosyltransferases. Chem.-Biol. Interact. 2023, 381, 110574. [Google Scholar] [CrossRef] [PubMed]
- Goodchild, J.; Donaldson, M. A clinically significant drug interaction between warfarin and amoxicillin resulting in persistent postoperative bleeding in a dental patient. Gen. Dent. 2013, 61, 50–54. [Google Scholar]
- Kumar, D.; Trivedi, N. Disease-drug and drug-drug interaction in COVID-19: Risk and assessment. Biomed. Pharmacother. 2021, 139, 111642. [Google Scholar] [CrossRef]
- Krishnan, S.; Ramsden, D.; Ferguson, D.; Stahl, S.H.; Wang, J.; McGinnity, D.F.; Hariparsad, N. Challenges and Opportunities for Improved Drug-Drug Interaction Predictions for Renal OCT2 and MATE1/2-K Transporters. Clin. Pharmacol. Ther. 2022, 112, 562–572. [Google Scholar] [CrossRef]
- Mozaffari, S.; Mousavi, T.; Nikfar, S.; Abdollahi, M. Common gastrointestinal drug-drug interactions in geriatrics and the importance of careful planning. Expert Opin. Drug Metab. Toxicol. 2023, 19, 807–828. [Google Scholar] [CrossRef]
- Zhao, Y.; Yin, J.; Zhang, L.; Zhang, Y.; Chen, X. Drug-drug interaction prediction: Databases, web servers and computational models. Brief. Bioinform. 2023, 25, bbad445. [Google Scholar] [CrossRef]
- Nyamabo, A.; Yu, H.; Liu, Z.; Shi, J. Drug-drug interaction prediction with learnable size-adaptive molecular substructures. Brief. Bioinform. 2022, 23, bbab441. [Google Scholar] [CrossRef]
- Qiu, Y.; Zhang, Y.; Deng, Y.; Liu, S.; Zhang, W. A Comprehensive Review of Computational Methods for Drug-Drug Interaction Detection. IEEE/ACM Trans. Comput. Biol. Bioinform. 2022, 19, 1968–1985. [Google Scholar] [CrossRef]
- Xiong, G.; Yang, Z.; Yi, J.; Wang, N.; Wang, L.; Zhu, H.; Wu, C.; Lu, A.; Chen, X.; Liu, S.; et al. DDInter: An online drug-drug interaction database towards improving clinical decision-making and patient safety. Nucleic Acids Res. 2022, 50, D1200–D1207. [Google Scholar] [CrossRef] [PubMed]
- Qing, L.; Xue, Y.; Ding, L.; Liu, Y.; Liang, J.; Liao, X. A Rapid Study of Botanical Drug-Drug Interaction with Protein by Re-ligand Fishing using Human Serum Albumin-Functionalized Magnetic Nanoparticles. Nat. Prod. Commun. 2015, 10, 2161–2163. [Google Scholar] [CrossRef] [PubMed]
- Meyer, U.A.; Zanger, U.M. Molecular mechanisms of genetic polymorphisms of drug metabolism. Annu. Rev. Pharmacol. Toxicol. 1997, 37, 269–296. [Google Scholar] [CrossRef] [PubMed]
- Gibson, T.P. Renal disease and drug metabolism: An overview. Am. J. Kidney Dis. 1986, 8, 7–17. [Google Scholar] [CrossRef]
- Schwartz, M.A. Metabolism of bumetanide. J. Clin. Pharmacol. 1981, 21, 555–563. [Google Scholar] [CrossRef]
- Azam, S.; Park, J.Y.; Kim, I.S.; Choi, D.K. Piperine and Its Metabolite’s Pharmacology in Neurodegenerative and Neurological Diseases. Biomedicines 2022, 10, 154. [Google Scholar] [CrossRef]
- Wu, Y.; Pan, L.; Chen, Z.; Zheng, Y.; Diao, X.; Zhong, D. Metabolite Identification in the Preclinical and Clinical Phase of Drug Development. Curr. Drug Metab. 2021, 22, 838–857. [Google Scholar] [CrossRef]
- Kamble, S.H.; León, F.; King, T.I.; Berthold, E.C.; Lopera-Londoño, C.; Siva Rama Raju, K.; Hampson, A.J.; Sharma, A.; Avery, B.A.; McMahon, L.R.; et al. Metabolism of a Kratom Alkaloid Metabolite in Human Plasma Increases Its Opioid Potency and Efficacy. ACS Pharmacol. Transl. Sci. 2020, 3, 1063–1068. [Google Scholar] [CrossRef]
- Li, A.P. Overview: Evaluation of metabolism-based drug toxicity in drug development. Chem. Biol. Interact. 2009, 179, 1–3. [Google Scholar] [CrossRef]
- He, C.; Wan, H. Drug metabolism and metabolite safety assessment in drug discovery and development. Expert. Opin. Drug Metab. Toxicol. 2018, 14, 1071–1085. [Google Scholar] [CrossRef] [PubMed]
- Ni, Y.; Gao, H.; Ouyang, W.; Yang, G.; Cheng, M.; Ding, L. Pharmacokinetics, metabolite profiling, safety and tolerability of YZJ-4729 tartrate, a novel G protein-biased μ-opioid receptor agonist, in healthy Chinese subjects. Front. Pharmacol. 2023, 14, 1295319. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Guo, J.L.; Xue, J.C.; Bai, C.L.; Guo, Y. Phthalate metabolites: Characterization, toxicities, global distribution, and exposure assessment. Environ. Pollut. 2021, 291, 118106. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.-N.; Pang, Y.-X.; He, X.-C.; Zhang, Y.-Z.; Zhang, J.-Y.; Zhao, Z.-Z.; Yi, T.; Chen, H.-B. UPLC-QTOF-MS identification of metabolites in rat biosamples after oral administration of Dioscorea saponins: A comparative study. J. Ethnopharmacol. 2015, 165, 127–140. [Google Scholar] [CrossRef]
- Sun, F.; Fang, S.B.; Li, B. Clinical Research on the Treatment of Osteoarthritis with Chuanlonggushu Capsules. J. Pharm. Res. 2009, 28, 49–50. [Google Scholar]
- Che, X.; Qu, G.; Fang, S.; Li, B. Observation on the Clinical Efficacy of 120 Cases of Osteophyte Treated with Dragon Bone Thorn Capsules. J. Pharm. Res. 2008, 27, 750–752. [Google Scholar]
- Jinyu, Y. The effect observation of chuanlongguci capsule on the treatment of hyperosteogeny. Chin. J. Clin. Ration. Drug Use 2015, 8, 25–26. [Google Scholar] [CrossRef]
- Xiaohong, G. One case of allergic reaction caused by Ruixiu Bi Kang Capsules. West. J. Tradit. Chin. Med. 2006, 6, 26. [Google Scholar]
- Liao, Z.; Liu, X.; Li, L.; Li, S.; Xing, X.; Zheng, X.; Song, W.; Gui, P.; Liu, Q.; Rong, G.; et al. Mechanism of the Proprietary Chinese Medicine “JiuLiWan” to Treat Ulcerative Colitis Revealed by Network Pharmacology, Molecular Docking, and Experimental Verification In Vitro. ACS Omega 2025, 10, 19598–19613. [Google Scholar] [CrossRef]
- Guan, H.R.; Li, B.; Zhang, Z.H.; Wu, H.S.; He, X.L.; Dong, Y.J.; Su, J.; Lv, G.Y.; Chen, S.H. Integrated bioinformatics and network pharmacology to explore the therapeutic target and molecular mechanisms of Bailing capsule on polycystic ovary syndrome. BMC Complement. Med. Ther. 2023, 23, 458. [Google Scholar] [CrossRef]
- Gao, J.; Xu, E.; Wang, H.; Wang, L.; Chen, S.; Wang, C.; Meng, F. Integrated serum pharmacochemistry, network pharmacology, and pharmacokinetics to clarify the effective components and pharmacological mechanisms of the proprietary Chinese medicine Jinkui Shenqi Pill in treating kidney yang deficiency syndrome. J. Pharm. Biomed. Anal. 2024, 247, 116251. [Google Scholar] [CrossRef]
- Li, K.; Chen, Y.; Zhang, Y. Isolation and identification of Diosgenin and pseudoprotodiosgenin from Diaoxinxinkang. Chin. Pharm. 2007, 30, 2339–2341. [Google Scholar]
- Zhang, W.; Chen, W.; Chen, G. Research progress of Diao Xuexikang. Chin. Tradit. Pat. Med. 2022, 44, 183–186. [Google Scholar] [CrossRef]
- Echouffo-Tcheugui, J.B.; Perreault, L.; Ji, L.; Dagogo-Jack, S. Diagnosis and Management of Prediabetes: A Review. JAMA 2023, 329, 1206–1216. [Google Scholar] [CrossRef] [PubMed]
- Peng, Y.; Yao, S.Y.; Chen, Q.; Jin, H.; Du, M.Q.; Xue, Y.H.; Liu, S. True or false? Alzheimer’s disease is type 3 diabetes: Evidences from bench to bedside. Ageing Res. Rev. 2024, 99, 102383. [Google Scholar] [CrossRef]
- Ansari, S.; Khoo, B.; Tan, T. Targeting the incretin system in obesity and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 2024, 20, 447–459. [Google Scholar] [CrossRef]
- Sun, H.; Saeedi, P.; Karuranga, S.; Pinkepank, M.; Ogurtsova, K.; Duncan, B.B.; Stein, C.; Basit, A.; Chan, J.C.N.; Mbanya, J.C.; et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res. Clin. Pract. 2022, 183, 109119. [Google Scholar] [CrossRef]
- Persson, F.; Rossing, P. Diagnosis of diabetic kidney disease: State of the art and future perspective. Kidney Int. Suppl. 2018, 8, 2–7. [Google Scholar] [CrossRef]
- Zhang, C.; Ji, Z.; Xu, N.; Yuan, J.; Zeng, W.; Wang, Y.; He, Q.; Dong, J.; Zhang, X.; Yang, D.; et al. Integrating network pharmacology and experimental validation to decipher the pharmacological mechanism of DXXK in treating diabetic kidney injury. Sci. Rep. 2024, 14, 22319. [Google Scholar] [CrossRef]
- Jain, D.; Aronow, W. Cardiotoxicity of cancer chemotherapy in clinical practice. Hosp. Pract. 2019, 47, 6–15. [Google Scholar] [CrossRef] [PubMed]
- Dempke, W.C.M.; Zielinski, R.; Winkler, C.; Silberman, S.; Reuther, S.; Priebe, W. Anthracycline-induced cardiotoxicity—Are we about to clear this hurdle? Eur. J. Cancer 2023, 185, 94–104. [Google Scholar] [CrossRef] [PubMed]
- Kong, C.Y.; Guo, Z.; Song, P.; Zhang, X.; Yuan, Y.P.; Teng, T.; Yan, L.; Tang, Q.Z. Underlying the Mechanisms of Doxorubicin-Induced Acute Cardiotoxicity: Oxidative Stress and Cell Death. Int. J. Biol. Sci. 2022, 18, 760–770. [Google Scholar] [CrossRef] [PubMed]
- Brell, J.M. 5-Fluorouracil Cardiotoxicity: Known but Unknown. JACC CardioOncol. 2021, 3, 110–112. [Google Scholar] [CrossRef]
- Li, X.; Liang, J.; Qin, A.; Wang, T.; Liu, S.; Li, W.; Yuan, C.; Qu, L.; Zou, W. Protective effect of Di’ao Xinxuekang capsule against doxorubicin-induced chronic cardiotoxicity. J. Ethnopharmacol. 2022, 287, 114943. [Google Scholar] [CrossRef]
- Lijun, Z. Rheumatism bi kang capsule pharmacodynamics experiment research. Guide China Med. 2014, 12, 52–53. [Google Scholar] [CrossRef]
- Qu, L.; Li, D.; Gao, X.; Li, Y.; Wu, J.; Zou, W. Di’ao Xinxuekang Capsule, a Chinese Medicinal Product, Decreases Serum Lipids Levels in High-Fat Diet-Fed ApoE(-/-) Mice by Downregulating PCSK9. Front. Pharmacol. 2018, 9, 1170. [Google Scholar] [CrossRef]
- Zhang, W.; Li, G.; Qi, Q.; Na, S.; Lyu, L.; Chen, G. Regulatory effect of Di’ao Xinxuekang on TLR4/MyD88 /NF-κB signaling pathway in atherosclerotic rats. China J. Chin. Mater. Med. 2020, 45, 602–608. [Google Scholar]
- Bai, Z.; Zhang, J.; Wei, Y. Effect of Di’aoxingxuekang on the hemorhelogy and blood system in blood stasis rats. J. Lanzhou Univ. (Med. Sci.) 2013, 39, 38–40. [Google Scholar] [CrossRef]
- Yu, Y.; Li, X.; Qu, L.; Chen, Y.; Dai, Y.; Wang, M.; Zou, W. DXXK exerts anti-inflammatory effects by inhibiting the lipopolysaccharide-induced NF-kappaB/COX-2 signalling pathway and the expression of inflammatory mediators. J. Ethnopharmacol. 2016, 178, 199–208. [Google Scholar] [CrossRef]
- Chen, H.; Zhu, K.; Zhang, Z.; Yu, L.; Kang, Y.; Liu, X. Protection and mechanism of D′i ao Xinxue Kang against myocardial ischemia-reperfusion injury in rats. Chin. Tradit. Herb. Drugs 2010, 41, 2018–2023. [Google Scholar]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, J.; Feng, S.; Du, Z.; Wu, Z.; Mo, W.; Chen, X.; Wu, J.; Lin, Y.; Cheng, C.; Sui, X.; et al. Research Progress on the Biological Activities and Clinical Applications of Pseudoprotodioscin. Curr. Issues Mol. Biol. 2025, 47, 927. https://doi.org/10.3390/cimb47110927
Li J, Feng S, Du Z, Wu Z, Mo W, Chen X, Wu J, Lin Y, Cheng C, Sui X, et al. Research Progress on the Biological Activities and Clinical Applications of Pseudoprotodioscin. Current Issues in Molecular Biology. 2025; 47(11):927. https://doi.org/10.3390/cimb47110927
Chicago/Turabian StyleLi, Jie, Senling Feng, Zhenya Du, Zhuzhu Wu, Wei Mo, Xiaoming Chen, Jiancong Wu, Yanming Lin, Chunsong Cheng, Xinbing Sui, and et al. 2025. "Research Progress on the Biological Activities and Clinical Applications of Pseudoprotodioscin" Current Issues in Molecular Biology 47, no. 11: 927. https://doi.org/10.3390/cimb47110927
APA StyleLi, J., Feng, S., Du, Z., Wu, Z., Mo, W., Chen, X., Wu, J., Lin, Y., Cheng, C., Sui, X., & Wu, Q. (2025). Research Progress on the Biological Activities and Clinical Applications of Pseudoprotodioscin. Current Issues in Molecular Biology, 47(11), 927. https://doi.org/10.3390/cimb47110927







