Effect of High-Intensity Interval Training on Cardiac Apoptosis Markers in Methamphetamine-Dependent Rats
Abstract
:1. Introduction
2. Materials and Methods
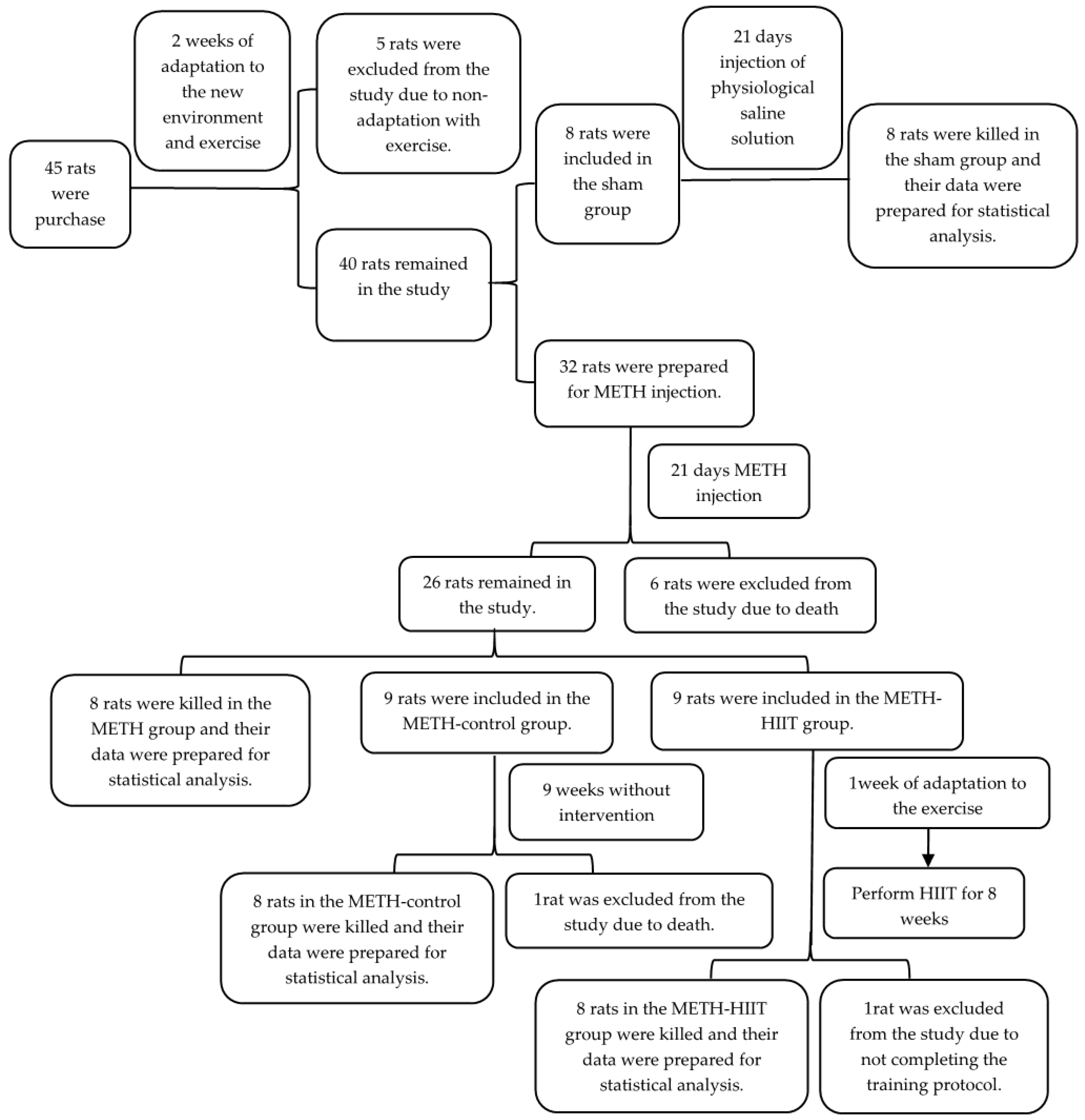
2.1. Study Design and Animals
2.2. METH Injection
2.3. Exercise Training Protocol
2.4. Tissue Extraction
2.5. Primer Design and Synthesis
2.6. RNA Extraction and cDNA Synthesis
2.7. Real Time RT-PCR
2.8. Statistical Analysis
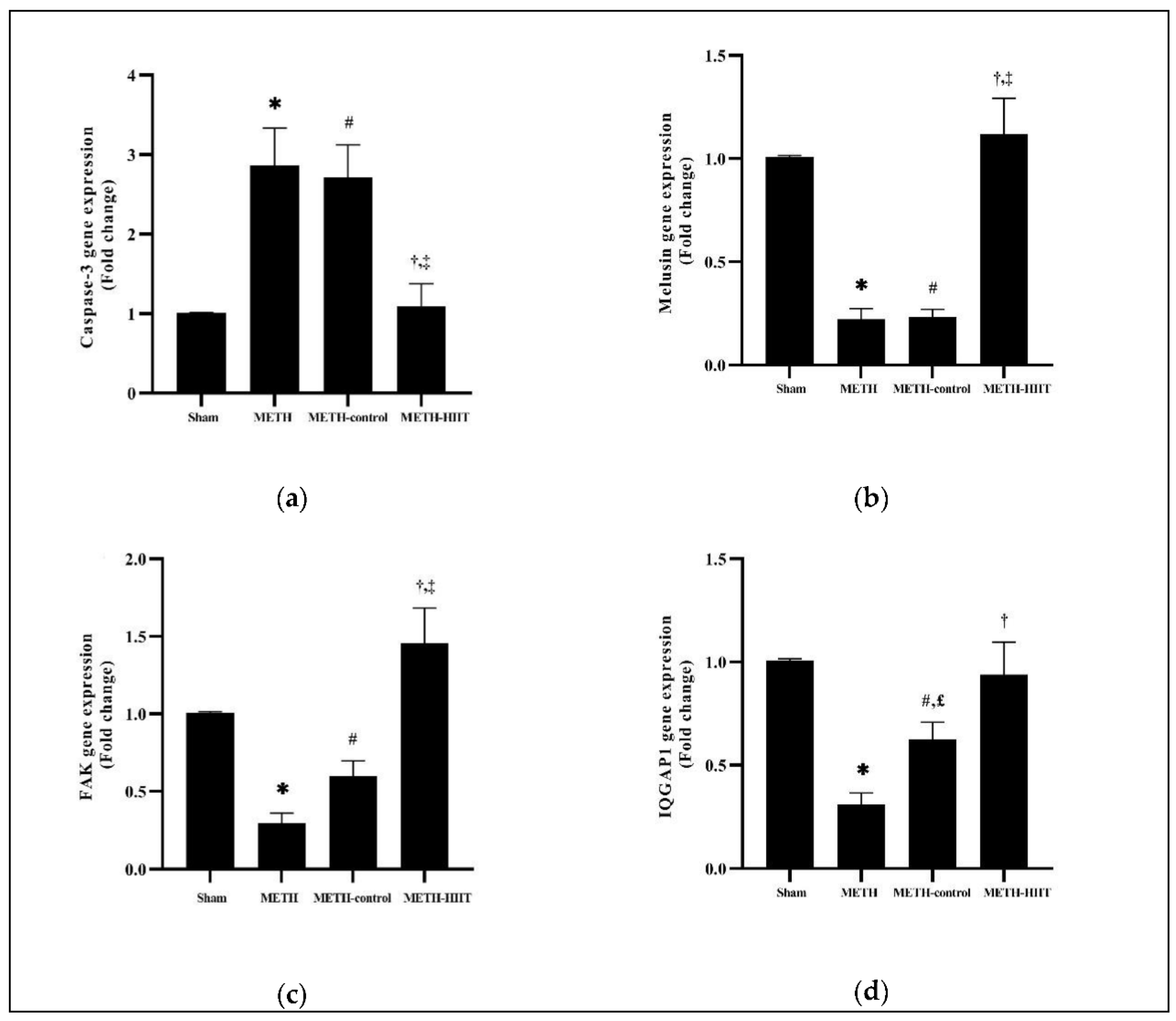
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Paratz, E.D.; Cunningham, N.J.; MacIsaac, A.I. The Cardiac Complications of Methamphetamines. Heart Lung Circ. 2016, 25, 325–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yu, Q.; Larson, D.F.; Watson, R.R. Heart disease, methamphetamine and AIDS. Life Sci. 2003, 73, 129–140. [Google Scholar] [CrossRef]
- Liou, C.M.; Tsai, S.C.; Kuo, C.H.; Williams, T.; Ting, H.; Lee, S. Da Chronic methamphetamine exposure induces cardiac Fas-dependent and mitochondria-dependent apoptosis. Cardiovasc. Toxicol. 2014, 14, 134–144. [Google Scholar] [CrossRef]
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Creagh, E.M.; Martin, S.J. Caspases: Cellular demolition experts. Biochem. Soc. Trans. 2001, 29, 696–702. [Google Scholar] [CrossRef] [PubMed]
- Condorelli, G.; Roncarati, R.; Ross, J.; Pisani, A.; Stassi, G.; Todaro, M.; Trocha, S.; Drusco, A.; Gu, Y.; Russo, M.A.; et al. Heart-targeted overexpression of caspase3 in mice increases infarct size and depresses cardiac function. Proc. Natl. Acad. Sci. USA 2001, 98, 9977–9982. [Google Scholar] [CrossRef] [Green Version]
- Brancaccio, M.; Guazzone, S.; Menini, N.; Sibona, E.; Hirsch, E.; De Andrea, M.; Rocchi, M.; Altruda, F.; Tarone, G.; Silengo, L. Melusin is a new muscle-specific interactor for β1 integrin cytoplasmic domain. J. Biol. Chem. 1999, 274, 29282–29288. [Google Scholar] [CrossRef] [Green Version]
- Sorge, M.; Brancaccio, M. Melusin promotes a protective signal transduction cascade in stressed hearts. Front. Mol. Biosci. 2016, 3, 53. [Google Scholar] [CrossRef] [Green Version]
- Palumbo, V.; Segat, L.; Padovan, L.; Amoroso, A.; Trimarco, B.; Izzo, R.; Lembo, G.; Zagrosek, V.R.; Knoll, R.; Brancaccio, M.; et al. Melusin gene (ITGB1BP2) nucleotide variations study in hypertensive and cardiopathic patients. BMC Med. Genet. 2009, 10, 140. [Google Scholar] [CrossRef] [Green Version]
- Sbroggiò, M.; Bertero, A.; Velasco, S.; Fusella, F.; De Blasio, E.; Bahou, W.F.; Silengo, L.; Turco, E.; Brancaccio, M.; Tarone, G. ERK1/2 activation in heart is controlled by melusin, focal adhesion kinase and the scaffold protein IQGAP1. J. Cell Sci. 2011, 124, 3515–3524. [Google Scholar] [CrossRef] [Green Version]
- Waardenberg, A.J.; Bernardo, B.C.; Ng, D.C.H.; Shepherd, P.R.; Cemerlang, N.; Sbroggio, M.; Wells, C.A.; Dalrymple, B.P.; Brancaccio, M.; Lin, R.C.Y.; et al. Phosphoinositide 3-kinase (PI3K(p110α)) directly regulates key components of the Z-disc and cardiac structure. J. Biol. Chem. 2011, 286, 30837–30846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ryu, S.J.; Cho, K.A.; Oh, Y.S.; Park, S.C. Role of Src-specific phosphorylation site on focal adhesion kinase for senescence-associated apoptosis resistance. Apoptosis 2006, 11, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Sato, A.; Naito, T.; Hiramoto, A.; Goda, K.; Omi, T.; Kitade, Y.; Sasaki, T.; Matsuda, A.; Fukushima, M.; Wataya, Y.; et al. Association of RNase L with a Ras GTPase-activating-like protein IQGAP1 in mediating the apoptosis of a human cancer cell-line. FEBS J. 2010, 277, 4464–4473. [Google Scholar] [CrossRef]
- Takemura, G.; Fujiwara, H. Role of apoptosis in remodeling after myocardial infarction. Pharmacol. Ther. 2004, 104, 1–16. [Google Scholar] [CrossRef]
- Sun, X.; Wang, Y.; Xia, B.; Li, Z.; Dai, J.; Qiu, P.; Ma, A.; Lin, Z.; Huang, J.; Wang, J.; et al. Methamphetamine produces cardiac damage and apoptosis by decreasing melusin. Toxicol. Appl. Pharmacol. 2019, 378, 114543. [Google Scholar] [CrossRef] [PubMed]
- Bowles, D.K.; Laughlin, M.H. Mechanism of beneficial effects of physical activity on atherosclerosis and coronary heart disease. J. Appl. Physiol. 2011, 111, 308–310. [Google Scholar] [CrossRef] [Green Version]
- Marques-Aleixo, I.; Santos-Alves, E.; Oliveira, P.J.; Moreira, P.I.; Magalhães, J.; Ascensão, A. The beneficial role of exercise in mitigating doxorubicin-induced Mitochondrionopathy. Biochim. Biophys. Acta Rev. Cancer 2018, 1869, 189–199. [Google Scholar] [CrossRef]
- Kwak, H.-B. Effects of aging and exercise training on apoptosis in the heart. J. Exerc. Rehabil. 2013, 9, 212–219. [Google Scholar] [CrossRef]
- Timm, K.N.; Tyler, D.J. The Role of AMPK Activation for Cardioprotection in Doxorubicin-Induced Cardiotoxicity. Cardiovasc. Drugs Ther. 2020, 34, 255–269. [Google Scholar] [CrossRef] [Green Version]
- Cassidy, S.; Thoma, C.; Houghton, D.; Trenell, M.I. High-intensity interval training: A review of its impact on glucose control and cardiometabolic health. Diabetologia 2017, 60, 7–23. [Google Scholar] [CrossRef] [Green Version]
- Viana, R.B.; de Lira, C.A.B.; Naves, J.P.A.; Coswig, V.S.; Del Vecchio, F.B.; Ramirez-Campillo, R.; Vieira, C.A.; Gentil, P. Can We Draw General Conclusions from Interval Training Studies? Sports Med. 2018, 48, 2001–2009. [Google Scholar] [CrossRef]
- Gibala, M.J.; Little, J.P.; Macdonald, M.J.; Hawley, J.A. Physiological adaptations to low-volume, high-intensity interval training in health and disease. J. Physiol. 2012, 5905, 1077–1084. [Google Scholar] [CrossRef]
- Peterson, J.M.; Bryner, R.W.; Sindler, A.; Frisbee, J.C.; Alway, S.E. Mitochondrial apoptotic signaling is elevated in cardiac but not skeletal muscle in the obese Zucker rat and is reduced with aerobic exercise. J. Appl. Physiol. 2008, 105, 1934–1943. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, K.; Wang, L.; Wang, C.; Yang, Y.; Hu, D.; Ding, R. Effects of high-intensity interval versus continuous moderate-intensity aerobic exercise on apoptosis, oxidative stress and metabolism of the infarcted myocardium in a rat model. Mol. Med. Rep. 2015, 12, 2374–2382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banaei, P.; Nazem, F.; Nazari, A.; Arjomand, A. Preconditioning Effect of High-Intensity Interval Training (HIIT) and Berberine Supplementation on the Gene Expression of Angiogenesis Regulators and Caspase-3 Protein in the Rats with Myocardial Ischemia-Reperfusion (IR) Injury. Biomed Res. Int. 2020, 2020, 4104965. [Google Scholar] [CrossRef] [PubMed]
- Wolff, A.M.; Rasmussen, T.P.; Wichern, C.R.; Peterson, M.R.; Stayton, M.M.; Thomas, D.P. Effects of pericardiectomy on training- and myocardial infarction-induced left ventricular hypertrophy, chamber dimensions and gene expression. Int. J. Sports Med. 2017, 38, 27–34. [Google Scholar] [CrossRef] [PubMed]
- Ballester, J.; Valentine, G.; Sofuoglu, M. Pharmacological treatments for methamphetamine addiction: Current status and future directions. Expert Rev. Clin. Pharmacol. 2017, 10, 305–314. [Google Scholar] [CrossRef]
- Abdullah, C.S.; Aishwarya, R.; Alam, S.; Morshed, M.; Remex, N.S.; Nitu, S.; Kolluru, G.K.; Traylor, J.; Miriyala, S.; Panchatcharam, M.; et al. Methamphetamine induces cardiomyopathy by Sigmar1 inhibition-dependent impairment of mitochondrial dynamics and function. Commun. Biol. 2020, 3, 682. [Google Scholar] [CrossRef]
- Fashi, M.; Agha-alinejad, H.; Mahabadi, H.A.; Rezaei, B.; Pakrad, B. The Effects of Aerobic Exercise on NF-κB and TNF-α in Lung Tissue of Male Rat. Nov. Biomed. 2015, 3, 131–134. [Google Scholar] [CrossRef]
- Chen, R.; Wang, B.; Chen, L.; Cai, D.; Li, B.; Chen, C.; Huang, E.; Liu, C.; Lin, Z.; Xie, W.B.; et al. DNA damage-inducible transcript 4 (DDIT4) mediates methamphetamine-induced autophagy and apoptosis through mTOR signaling pathway in cardiomyocytes. Toxicol. Appl. Pharmacol. 2016, 295, 1–11. [Google Scholar] [CrossRef]
- Lord, K.C.; Shenouda, S.K.; McIlwain, E.; Charalampidis, D.; Lucchesi, P.A.; Varner, K.J. Oxidative stress contributes to methamphetamine-induced left ventricular dysfunction. Cardiovasc. Res. 2010, 87, 111–118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vona, R.; Ascione, B.; Malorni, W.; Straface, E. Mitochondria and sex-specific cardiac function. Adv. Exp. Med. Biol. 2018, 1065, 241–256. [Google Scholar] [CrossRef] [PubMed]
- Delfan, M.; Delphan, M.; Kordi, M.R.; Ravasi, A.A.; Safa, M.; Gorgani-Firuzjaee, S.; Fatemi, A.; Bandarian, F.; Nasli-Esfahani, E. High intensity interval training improves diabetic cardiomyopathy via miR-1 dependent suppression of cardiomyocyte apoptosis in diabetic rats. J. Diabetes Metab. Disord. 2020, 19, 145–152. [Google Scholar] [CrossRef] [PubMed]
- Qian, W.; Wang, Z.; Xu, T.; Li, D. Anti-apoptotic effects and mechanisms of salvianolic acid a on cardiomyocytes in ischemia-reperfusion injury. Histol. Histopathol. 2019, 34, 223–231. [Google Scholar] [CrossRef]
- Bafghi, A.F.; Homaee, H.M.; Azarbayjani, M.A. Effects of High Intensity Interval Training and Curcumin Supplement on Antioxidant Enzyme in Heart Tissue of Diabetic Rats. Iran. J. Diabetes Obes. 2017, 8, 135–141. [Google Scholar]
- Silva, L.R.B.; Gentil, P.; Seguro, C.S.; de Oliveira, J.C.M.; Silva, M.S.; Marques, V.A.; Beltrame, T.; Rebelo, A.C.S. High-Intensity Interval Training Improves Cardiac Autonomic Function in Patients with Type 2 Diabetes: A Randomized Controlled Trial. Biology 2022, 11, 66. [Google Scholar] [CrossRef]
| Weeks | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Number of intervals | 4 | 4 | 4 | 4 | 4 | 4 | 4 | 4 |
| Effort duration (min) | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 |
| Effort velocity (m/min) | 22 | 24 | 26 | 28 | 30 | 32 | 34 | 36 |
| Rest duration | 2 | 2 | 3 | 3 | 4 | 4 | 4 | 4 |
| Rest velocity (m/min) | 10 | 10 | 11 | 11 | 12 | 12 | 13 | 13 |
| Gene Name | Forward Primer | Reverse Primer |
|---|---|---|
| Caspase-3 | 5′-GCAGCAGCCTCAAATTGTTGACTA-3′ | 5′-TGCTCCGGCTCAAACCATC-3′ |
| Melusin | 5′-GGGTGAAGGCCAGTCAAACT-3′ | 5′-TGCTCCACGTTTATGACCCC-3′ |
| FAK | 5′-CTTAATCTGGCCAGGACGGT-3′ | 5′-GAAGCACGGTTTGAGAGGTG-3′ |
| IQGAP1 | 5′-ACAATCTGGAGACGCAAGCA-3′ | 5′-AGCTGCTCTCGGTTATACGC-3′ |
| GAPDH | 5′-CAACTCCCTCAAGATTGTCAGCAA-3′ | 5′-GGCATGGACTGTGGTCATGA-3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shahrabadi, H.; Haghighi, A.H.; Askari, R.; Asadi-Shekaari, M.; Souza, D.C.; Gentil, P. Effect of High-Intensity Interval Training on Cardiac Apoptosis Markers in Methamphetamine-Dependent Rats. Curr. Issues Mol. Biol. 2022, 44, 3030-3038. https://doi.org/10.3390/cimb44070209
Shahrabadi H, Haghighi AH, Askari R, Asadi-Shekaari M, Souza DC, Gentil P. Effect of High-Intensity Interval Training on Cardiac Apoptosis Markers in Methamphetamine-Dependent Rats. Current Issues in Molecular Biology. 2022; 44(7):3030-3038. https://doi.org/10.3390/cimb44070209
Chicago/Turabian StyleShahrabadi, Hadi, Amir Hossein Haghighi, Roya Askari, Majid Asadi-Shekaari, Daniel Costa Souza, and Paulo Gentil. 2022. "Effect of High-Intensity Interval Training on Cardiac Apoptosis Markers in Methamphetamine-Dependent Rats" Current Issues in Molecular Biology 44, no. 7: 3030-3038. https://doi.org/10.3390/cimb44070209
APA StyleShahrabadi, H., Haghighi, A. H., Askari, R., Asadi-Shekaari, M., Souza, D. C., & Gentil, P. (2022). Effect of High-Intensity Interval Training on Cardiac Apoptosis Markers in Methamphetamine-Dependent Rats. Current Issues in Molecular Biology, 44(7), 3030-3038. https://doi.org/10.3390/cimb44070209








