Interactome Analysis of KIN (Kin17) Shows New Functions of This Protein
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture for BioID
2.2. Generation of BirA* Fusion Protein Expression Cell Lines
2.3. Induction and Harvest
2.4. Synchronization
2.5. Irradiation
2.6. BioID Purification
2.7. Digestion and LC-MS/MS
2.8. Computational Analyses
2.9. Interactome Design
2.10. Immunolocalization
2.11. In Vitro Methylation Assay
3. Results
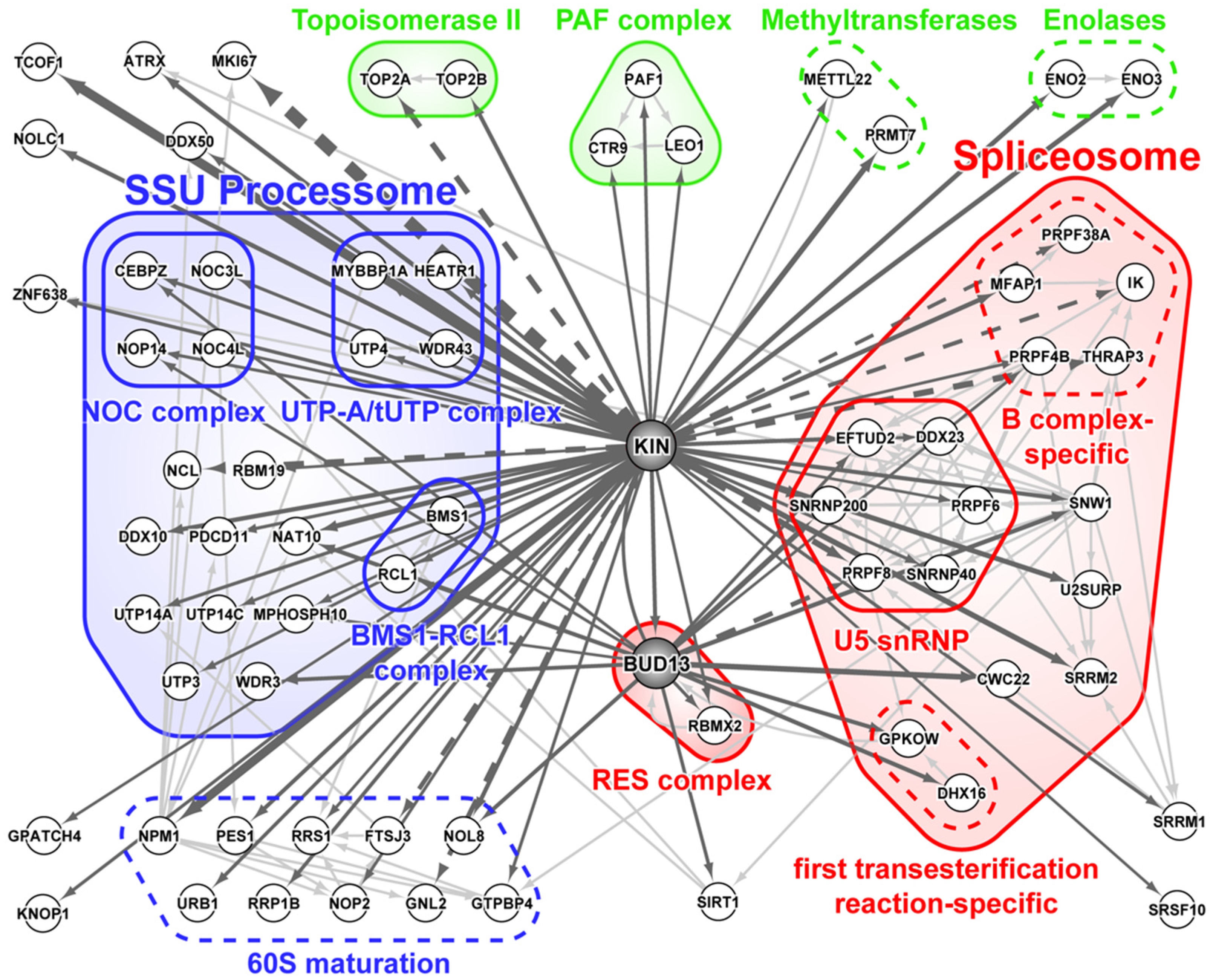
3.1. KIN Mostly Associates with Proteins Involved in Ribosome Biogenesis and Splicing
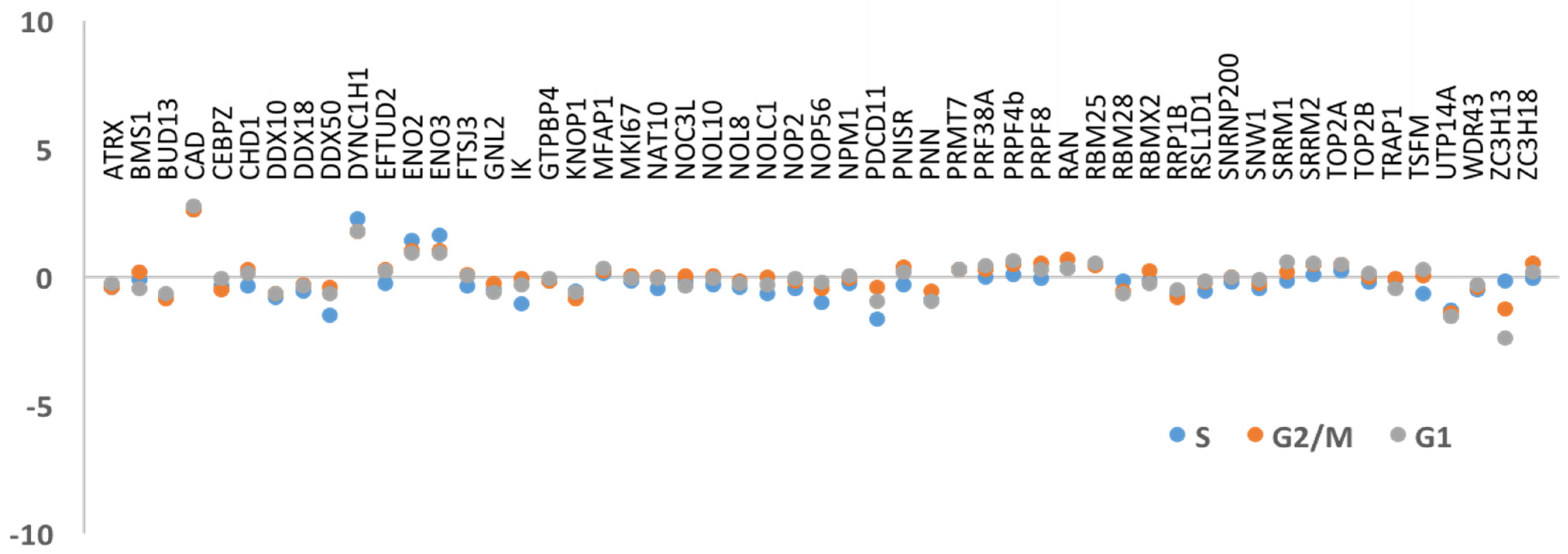
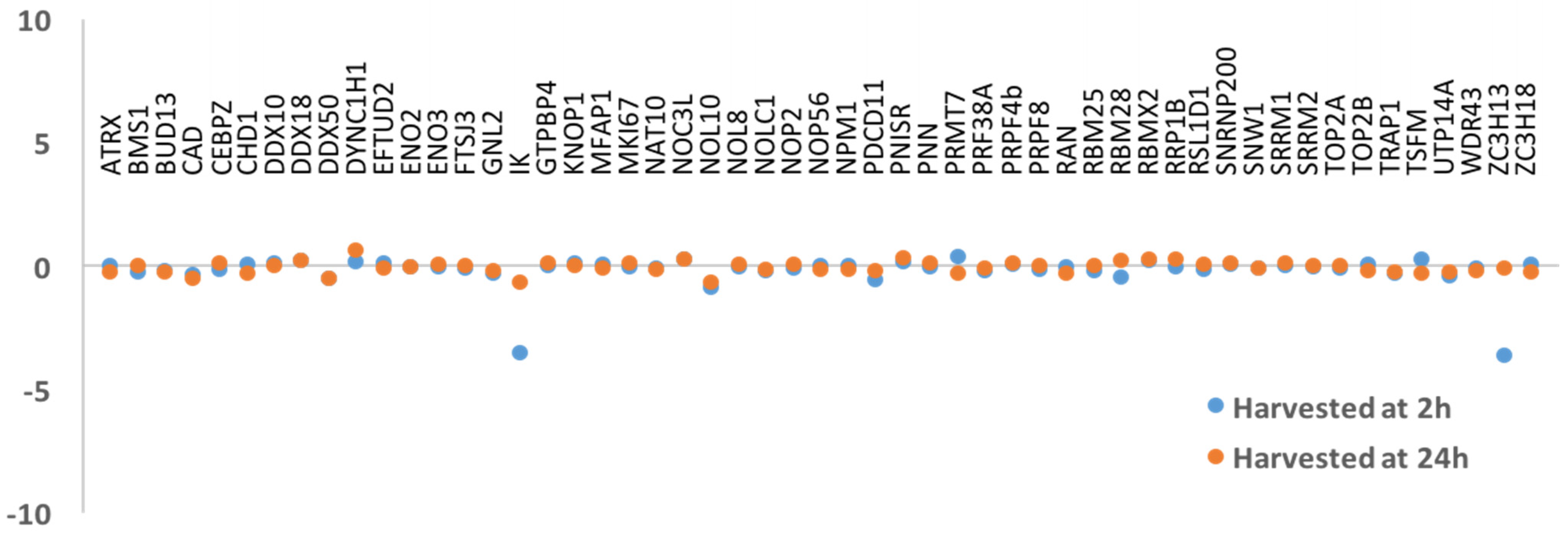
3.2. DNA Replication and/or Repair Proteins Were Not Obvious Interactors in Synchronized and/or Irradiated Cells
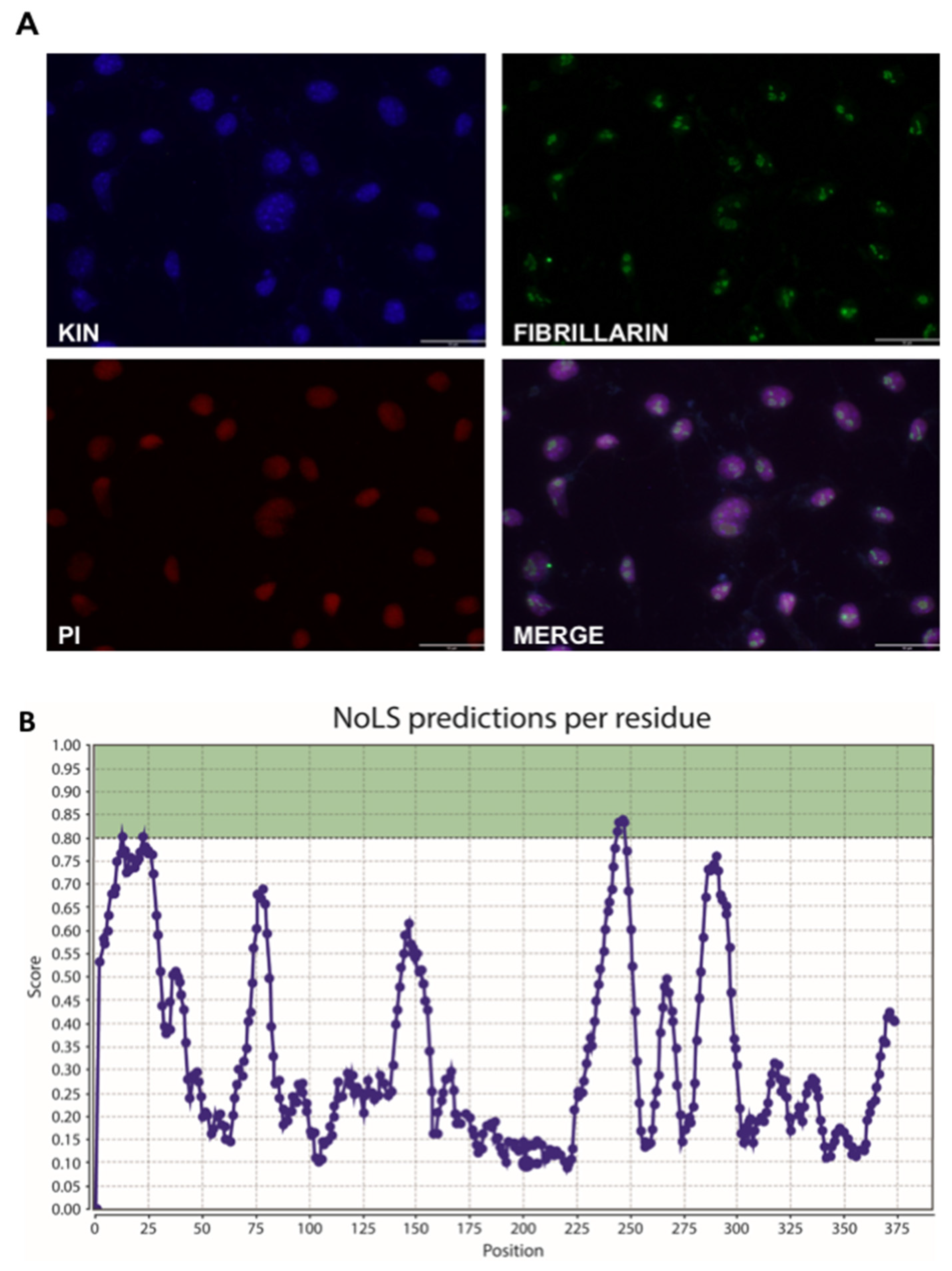
3.3. KIN Is Present in the Nucleolus as Well as Other Parts of the Nucleus
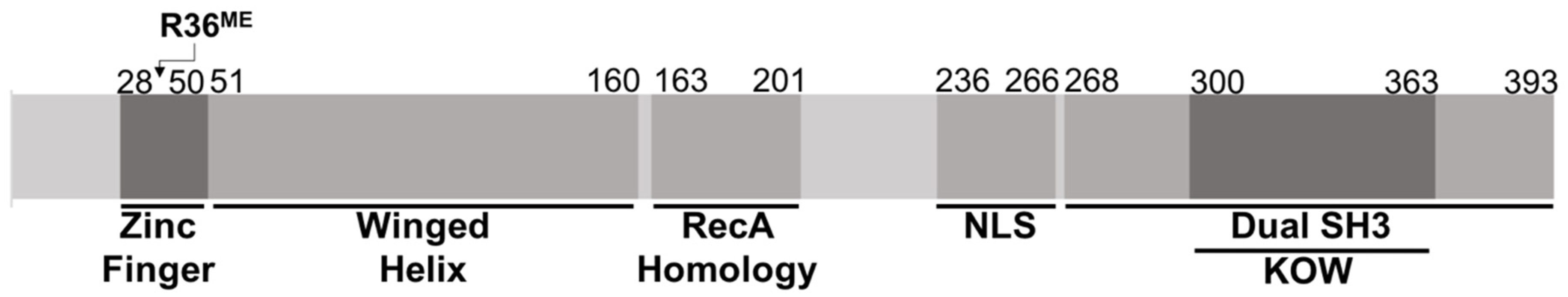
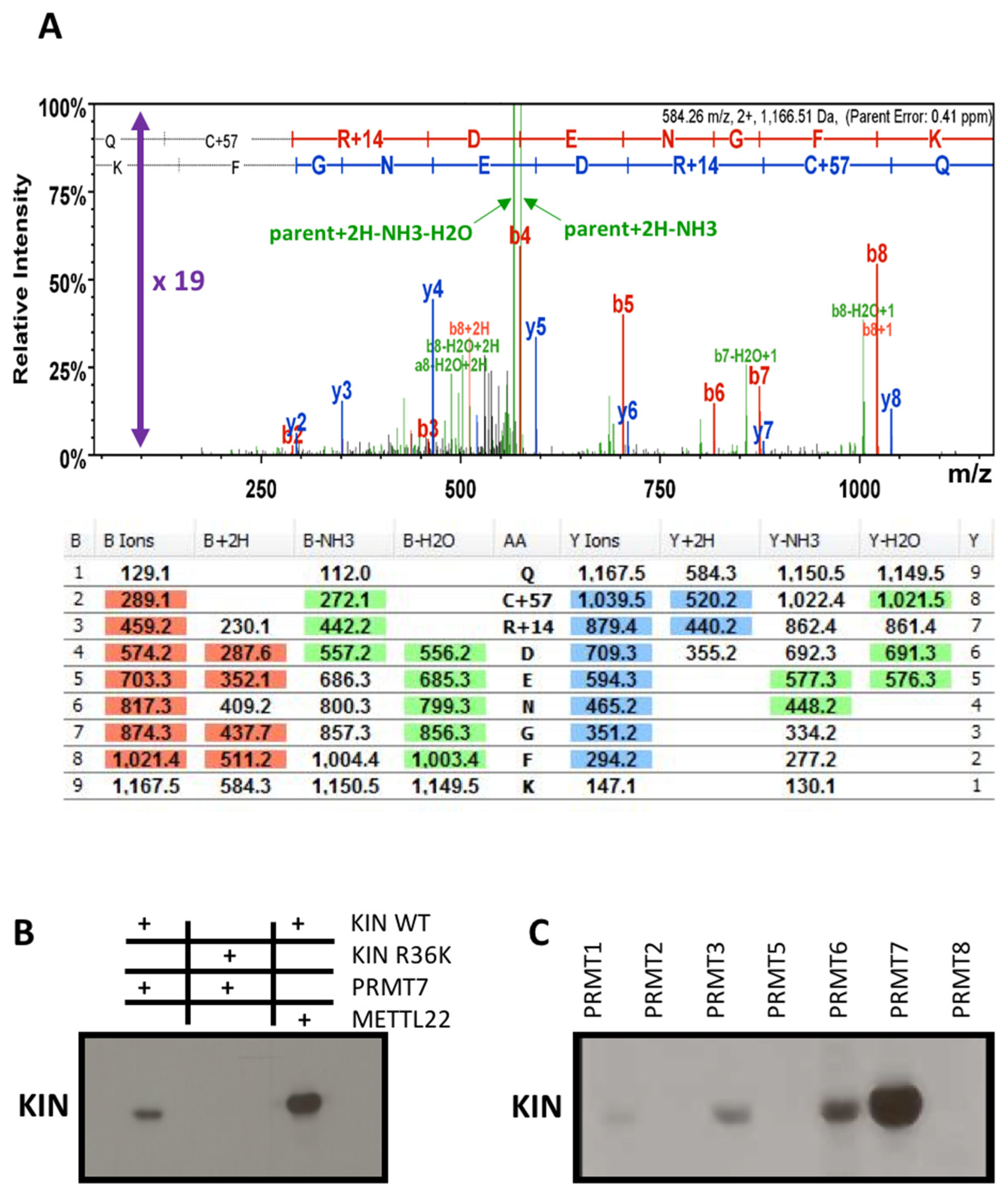
3.4. Methyltransferase PRMT7 Methylates KIN at Arginine 36
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zeng, T.; Gao, H.; Yu, P.; He, H.; Ouyang, X.; Deng, L.; Zhang, Y. Up-regulation of kin17 is essential for proliferation of breast cancer. PLoS ONE 2011, 6, e25343. [Google Scholar] [CrossRef]
- Gao, X.; Liu, Z.; Zhong, M.; Wu, K.; Zhang, Y.; Wang, H.; Zeng, T. Knockdown of DNA/RNA-binding protein KIN17 promotes apoptosis of triple-negative breast cancer cells. Oncol. Lett. 2019, 17, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Yu, M.; Zhang, Z.; Yu, H.; Xue, C.; Yuan, K.; Miao, M.; Shi, H. KIN enhances stem cell-like properties to promote chemoresistance in colorectal carcinoma. Biochem. Biophys. Res. Commun. 2014, 448, 63–69. [Google Scholar] [CrossRef] [PubMed]
- Ramos, A.C.; Gaspar, V.P.; Kelmer, S.M.; Sellani, T.A.; Batista, A.G.; De Lima Neto, Q.A.; Rodrigues, E.G.; Fernandez, M.A. The kin17 Protein in Murine Melanoma Cells. Int. J. Mol. Sci. 2015, 16, 27912–27920. [Google Scholar] [CrossRef] [PubMed]
- Biard, D.S.; Miccoli, L.; Despras, E.; Frobert, Y.; Creminon, C.; Angulo, J.F. Ionizing radiation triggers chromatin-bound kin17 complex formation in human cells. J. Biol. Chem. 2002, 277, 19156–19165. [Google Scholar] [CrossRef]
- Kou, W.Z.; Xu, S.L.; Wang, Y.; Wang, L.W.; Wang, L.; Chai, X.Y.; Hua, Q.L. Expression of Kin17 promotes the proliferation of hepatocellular carcinoma cells in vitro and in vivo. Oncol. Lett. 2014, 8, 1190–1194. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, S.; Gao, H.; Wu, K.; Ouyang, X.; Zhu, Z.; Yu, X.; Zeng, T. Upregulation of KIN17 is associated with non-small cell lung cancer invasiveness. Oncol. Lett. 2017, 13, 2274–2280. [Google Scholar] [CrossRef] [PubMed]
- Despras, E.; Miccoli, L.; Creminon, C.; Rouillard, D.; Angulo, J.F.; Biard, D.S. Depletion of KIN17, a human DNA replication protein, increases the radiosensitivity of RKO cells. Radiat. Res. 2003, 159, 748–758. [Google Scholar] [CrossRef]
- Pattaro Júnior, J.R.; Caruso, Í.P.; de Lima Neto, Q.A.; Duarte Junior, F.F.; dos Santos Rando, F.; Gerhardt, E.C.M.; Fernandez, M.A.; Seixas, F.A.V. Biophysical characterization and molecular phylogeny of human KIN protein. Eur. Biophys. J. 2019, 48, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Angulo, J.F.; Moreau, P.L.; Maunoury, R.; Laporte, J.; Hill, A.M.; Bertolotti, R.; Devoret, R. KIN, a mammalian nuclear protein immunologically related to E. coli RecA protein. Mutat. Res. 1989, 217, 123–134. [Google Scholar] [CrossRef]
- Kannouche, P.; Mauffrey, P.; Pinon-Lataillade, G.; Mattei, M.G.; Sarasin, A.; Daya-Grosjean, L.; Angulo, J.F. Molecular cloning and characterization of the human KIN17 cDNA encoding a component of the UVC response that is conserved among metazoans. Carcinogenesis 2000, 21, 1701–1710. [Google Scholar] [CrossRef] [PubMed]
- Le, M.X.; Haddad, D.; Ling, A.K.; Li, C.; So, C.C.; Chopra, A.; Hu, R.; Angulo, J.F.; Moffat, J.; Martin, A. Kin17 facilitates multiple double-strand break repair pathways that govern B cell class switching. Sci. Rep. 2016, 6, 37215. [Google Scholar] [CrossRef] [PubMed]
- Miccoli, L.; Biard, D.S.; Creminon, C.; Angulo, J.F. Human kin17 protein directly interacts with the simian virus 40 large T antigen and inhibits DNA replication. Cancer Res. 2002, 62, 5425–5435. [Google Scholar] [PubMed]
- Miccoli, L.; Frouin, I.; Novac, O.; Di Paola, D.; Harper, F.; Zannis-Hadjopoulos, M.; Maga, G.; Biard, D.S.; Angulo, J.F. The human stress-activated protein kin17 belongs to the multiprotein DNA replication complex and associates in vivo with mammalian replication origins. Mol. Cell Biol. 2005, 25, 3814–3830. [Google Scholar] [CrossRef] [PubMed]
- Rappsilber, J.; Ryder, U.; Lamond, A.I.; Mann, M. Large-scale proteomic analysis of the human spliceosome. Genome Res. 2002, 12, 1231–1245. [Google Scholar] [CrossRef]
- Angulo, J.F.; Rouer, E.; Mazin, A.; Mattei, M.G.; Tissier, A.; Horellou, P.; Benarous, R.; Devoret, R. Identification and expression of the cDNA of KIN17, a zinc-finger gene located on mouse chromosome 2, encoding a new DNA-binding protein. Nucleic Acids Res. 1991, 19, 5117–5123. [Google Scholar] [CrossRef]
- Carlier, L.; Couprie, J.; le Maire, A.; Guilhaudis, L.; Milazzo-Segalas, I.; Courcon, M.; Moutiez, M.; Gondry, M.; Davoust, D.; Gilquin, B.; et al. Solution structure of the region 51-160 of human KIN17 reveals an atypical winged helix domain. Protein Sci. 2007, 16, 2750–2755. [Google Scholar] [CrossRef]
- Le Maire, A.; Schiltz, M.; Stura, E.A.; Pinon-Lataillade, G.; Couprie, J.; Moutiez, M.; Gondry, M.; Angulo, J.F.; Zinn-Justin, S. A tandem of SH3-like domains participates in RNA binding in KIN17, a human protein activated in response to genotoxics. J. Mol. Biol. 2006, 364, 764–776. [Google Scholar] [CrossRef]
- Kyrpides, N.C.; Woese, C.R.; Ouzounis, C.A. KOW: A novel motif linking a bacterial transcription factor with ribosomal proteins. Trends Biochem. Sci. 1996, 21, 425–426. [Google Scholar] [CrossRef]
- Le Maire, A.; Schiltz, M.; Braud, S.; Gondry, M.; Charbonnier, J.B.; Zinn-Justin, S.; Stura, E. Crystallization and halide phasing of the C-terminal domain of human KIN17. Acta Crystallogr. Sect. F Struct. Biol. Cryst. Commun. 2006, 62 Pt 3, 245–248. [Google Scholar] [CrossRef]
- Pinon-Lataillade, G.; Masson, C.; Bernardino-Sgherri, J.; Henriot, V.; Mauffrey, P.; Frobert, Y.; Araneda, S.; Angulo, J.F. KIN17 encodes an RNA-binding protein and is expressed during mouse spermatogenesis. J. Cell Sci. 2004, 117 Pt 16, 3691–3702. [Google Scholar] [CrossRef]
- Treiber, T.; Treiber, N.; Plessmann, U.; Harlander, S.; Daiß, J.L.; Eichner, N.; Lehmann, G.; Schall, K.; Urlaub, H.; Meister, G. A Compendium of RNA-Binding Proteins that Regulate MicroRNA Biogenesis. Mol. Cell 2017, 66, 270.e13–284.e13. [Google Scholar] [CrossRef] [PubMed]
- Roux, K.J.; Kim, D.I.; Burke, B. BioID: A screen for protein-protein interactions. Curr. Protoc. Protein Sci. 2013, 74, 19–23. [Google Scholar] [CrossRef]
- Miranda, T.B.; Miranda, M.; Frankel, A.; Clarke, S. PRMT7 is a member of the protein arginine methyltransferase family with a distinct substrate specificity. J. Biol. Chem. 2004, 279, 22902–22907. [Google Scholar] [CrossRef]
- Couzens, A.L.; Knight, J.D.; Kean, M.J.; Teo, G.; Weiss, A.; Dunham, W.H.; Lin, Z.Y.; Bagshaw, R.D.; Sicheri, F.; Pawson, T.; et al. Protein interaction network of the mammalian Hippo pathway reveals mechanisms of kinase-phosphatase interactions. Sci. Signal. 2013, 6, rs15. [Google Scholar] [CrossRef]
- Rodier, G.; Coulombe, P.; Tanguay, P.L.; Boutonnet, C.; Meloche, S. Phosphorylation of Skp2 regulated by CDK2 and Cdc14B protects it from degradation by APC(Cdh1) in G1 phase. EMBO J. 2008, 27, 679–691. [Google Scholar] [CrossRef]
- Tanguay, P.L.; Rodier, G.; Meloche, S. C-terminal domain phosphorylation of ERK3 controlled by Cdk1 and Cdc14 regulates its stability in mitosis. Biochem. J. 2010, 428, 103–111. [Google Scholar] [CrossRef]
- Dressler, L.G.; Seamer, L.C.; Owens, M.A.; Clark, G.M.; McGuire, W.L. DNA flow cytometry and prognostic factors in 1331 frozen breast cancer specimens. Cancer 1988, 61, 420–427. [Google Scholar] [CrossRef]
- Coyaud, E.; Mis, M.; Laurent, E.M.; Dunham, W.H.; Couzens, A.L.; Robitaille, M.; Gingras, A.C.; Angers, S.; Raught, B. BioID-based Identification of Skp Cullin F-box (SCF)beta-TrCP1/2 E3 Ligase Substrates. Mol. Cell Proteom. 2015, 14, 1781–1795. [Google Scholar] [CrossRef] [PubMed]
- Lavallee-Adam, M.; Rousseau, J.; Domecq, C.; Bouchard, A.; Forget, D.; Faubert, D.; Blanchette, M.; Coulombe, B. Discovery of cell compartment specific protein-protein interactions using affinity purification combined with tandem mass spectrometry. J. Proteome Res. 2013, 12, 272–281. [Google Scholar] [CrossRef] [PubMed]
- Firat-Karalar, E.N.; Rauniyar, N.; Yates, J.R., 3rd; Stearns, T. Proximity interactions among centrosome components identify regulators of centriole duplication. Curr. Biol. 2014, 24, 664–670. [Google Scholar] [CrossRef]
- Cloutier, P.; Poitras, C.; Durand, M.; Hekmat, O.; Fiola-Masson, E.; Bouchard, A.; Faubert, D.; Chabot, B.; Coulombe, B. R2TP/Prefoldin-like component RUVBL1/RUVBL2 directly interacts with ZNHIT2 to regulate assembly of U5 small nuclear ribonucleoprotein. Nat. Commun. 2017, 8, 15615. [Google Scholar] [CrossRef]
- Teo, G.; Liu, G.; Zhang, J.; Nesvizhskii, A.I.; Gingras, A.C.; Choi, H. SAINTexpress: Improvements and additional features in Significance Analysis of INTeractome software. J. Proteom. 2014, 100, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Choi, H.; Larsen, B.; Lin, Z.Y.; Breitkreutz, A.; Mellacheruvu, D.; Fermin, D.; Qin, Z.S.; Tyers, M.; Gingras, A.C.; Nesvizhskii, A.I. SAINT: Probabilistic scoring of affinity purification-mass spectrometry data. Nat. Methods 2011, 8, 70–73. [Google Scholar] [CrossRef]
- Prieto, C.; De Las Rivas, J. APID: Agile Protein Interaction DataAnalyzer. Nucleic Acids Res. 2006, 34, W298–W302. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, P.; Lavallee-Adam, M.; Faubert, D.; Blanchette, M.; Coulombe, B. Methylation of the DNA/RNA-binding protein Kin17 by METTL22 affects its association with chromatin. J. Proteomics 2014, 100, 115–124. [Google Scholar] [CrossRef]
- Zhu, B.; Mandal, S.S.; Pham, A.D.; Zheng, Y.; Erdjument-Bromage, H.; Batra, S.K.; Tempst, P.; Reinberg, D. The human PAF complex coordinates transcription with events downstream of RNA synthesis. Genes Dev. 2005, 19, 1668–1673. [Google Scholar] [CrossRef] [PubMed]
- Cloutier, P.; Lavallee-Adam, M.; Faubert, D.; Blanchette, M.; Coulombe, B. A newly uncovered group of distantly related lysine methyltransferases preferentially interact with molecular chaperones to regulate their activity. PLoS Genet. 2013, 9, e1003210. [Google Scholar] [CrossRef]
- Phipps, K.R.; Charette, J.; Baserga, S.J. The small subunit processome in ribosome biogenesis-progress and prospects. Wiley Interdiscip Rev. RNA 2011, 2, 1–21. [Google Scholar] [CrossRef]
- Kornprobst, M.; Turk, M.; Kellner, N.; Cheng, J.; Flemming, D.; Koš-Braun, I.; Koš, M.; Thoms, M.; Berninghausen, O.; Beckmann, R.; et al. Architecture of the 90S Pre-ribosome: A Structural View on the Birth of the Eukaryotic Ribosome. Cell 2016, 166, 380–393. [Google Scholar] [CrossRef]
- Hegele, A.; Kamburov, A.; Grossmann, A.; Sourlis, C.; Wowro, S.; Weimann, M.; Will, C.L.; Pena, V.; Luhrmann, R.; Stelzl, U. Dynamic protein-protein interaction wiring of the human spliceosome. Mol. Cell 2012, 45, 567–580. [Google Scholar] [CrossRef] [PubMed]
- Trowitzsch, S.; Weber, G.; Luhrmann, R.; Wahl, M.C. An unusual RNA recognition motif acts as a scaffold for multiple proteins in the pre-mRNA retention and splicing complex. J. Biol. Chem. 2008, 283, 32317–32327. [Google Scholar] [CrossRef] [PubMed]
- Wysoczanski, P.; Zweckstetter, M. Retention and splicing complex (RES)—The importance of cooperativity. RNA Biol. 2016, 13, 128–133. [Google Scholar] [CrossRef][Green Version]
- Hutchins, J.R.; Toyoda, Y.; Hegemann, B.; Poser, I.; Heriche, J.K.; Sykora, M.M.; Augsburg, M.; Hudecz, O.; Buschhorn, B.A.; Bulkescher, J.; et al. Systematic analysis of human protein complexes identifies chromosome segregation proteins. Science 2010, 328, 593–599. [Google Scholar] [CrossRef]
- Deaconescu, A.M.; Chambers, A.L.; Smith, A.J.; Nickels, B.E.; Hochschild, A.; Savery, N.J.; Darst, S.A. Structural basis for bacterial transcription-coupled DNA repair. Cell 2006, 124, 507–520. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.S.; Lam, Y.W.; Leung, A.K.; Ong, S.E.; Lyon, C.E.; Lamond, A.I.; Mann, M. Nucleolar proteome dynamics. Nature 2005, 433, 77–83. [Google Scholar] [CrossRef]
- Garcia-Molina, A.; Xing, S.; Huijser, P. Functional characterisation of Arabidopsis SPL7 conserved protein domains suggests novel regulatory mechanisms in the Cu deficiency response. BMC Plant. Biol. 2014, 14, 231. [Google Scholar] [CrossRef]
- Scott, M.S.; Troshin, P.V.; Barton, G.J. NoD: A Nucleolar localization sequence detector for eukaryotic and viral proteins. BMC Bioinform. 2011, 12, 317. [Google Scholar] [CrossRef]
- Zurita-Lopez, C.I.; Sandberg, T.; Kelly, R.; Clarke, S.G. Human protein arginine methyltransferase 7 (PRMT7) is a type III enzyme forming omega-NG-monomethylated arginine residues. J. Biol. Chem. 2012, 287, 7859–7870. [Google Scholar] [CrossRef]
- Kim, M.S.; Pinto, S.M.; Getnet, D.; Nirujogi, R.S.; Manda, S.S.; Chaerkady, R.; Madugundu, A.K.; Kelkar, D.S.; Isserlin, R.; Jain, S.; et al. A draft map of the human proteome. Nature 2014, 509, 575–581. [Google Scholar] [CrossRef]
- Lambert, M.J.; Cochran, W.O.; Wilde, B.M.; Olsen, K.G.; Cooper, C.D. Evidence for widespread subfunctionalization of splice forms in vertebrate genomes. Genome Res. 2015, 25, 624–632. [Google Scholar] [CrossRef]
- Thoms, M.; Thomson, E.; Bassler, J.; Gnadig, M.; Griesel, S.; Hurt, E. The Exosome Is Recruited to RNA Substrates through Specific Adaptor Proteins. Cell 2015, 162, 1029–1038. [Google Scholar] [CrossRef] [PubMed]
- Pelletier, J.; Thomas, G.; Volarevic, S. Ribosome biogenesis in cancer: New players and therapeutic avenues. Nat. Rev. Cancer 2018, 18, 51–63. [Google Scholar] [CrossRef] [PubMed]
- Quin, J.E.; Devlin, J.R.; Cameron, D.; Hannan, K.M.; Pearson, R.B.; Hannan, R.D. Targeting the nucleolus for cancer intervention. Biochim. Biophys. Acta 2014, 1842, 802–816. [Google Scholar] [CrossRef]
- Nag, A.; Steitz, J.A. Tri-snRNP-associated proteins interact with subunits of the TRAMP and nuclear exosome complexes, linking RNA decay and pre-mRNA splicing. RNA Biol. 2012, 9, 334–342. [Google Scholar] [CrossRef]
- Shkreta, L.; Chabot, B. The RNA Splicing Response to DNA Damage. Biomolecules 2015, 5, 2935–2977. [Google Scholar] [CrossRef]
- Scott, D.D.; Trahan, C.; Zindy, P.J.; Aguilar, L.C.; Delubac, M.Y.; Van Nostrand, E.L.; Adivarahan, S.; Wei, K.E.; Yeo, G.W.; Zenklusen, D.; et al. Nol12 is a multifunctional RNA binding protein at the nexus of RNA and DNA metabolism. Nucleic Acids Res. 2017, 45, 12509–12528. [Google Scholar] [CrossRef] [PubMed]
- Berger, C.M.; Gaume, X.; Bouvet, P. The roles of nucleolin subcellular localization in cancer. Biochimie 2015, 113, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Antoniali, G.; Lirussi, L.; Poletto, M.; Tell, G. Emerging roles of the nucleolus in regulating the DNA damage response: The noncanonical DNA repair enzyme APE1/Ref-1 as a paradigmatical example. Antioxid. Redox Signal. 2014, 20, 621–639. [Google Scholar] [CrossRef]
- Black, C.S.; Garside, E.L.; MacMillan, A.M.; Rader, S.D. Conserved structure of Snu13 from the highly reduced spliceosome of Cyanidioschyzon merolae. Protein Sci. 2016, 25, 911–916. [Google Scholar] [CrossRef] [PubMed]
- Rothe, B.; Manival, X.; Rolland, N.; Charron, C.; Senty-Segault, V.; Branlant, C.; Charpentier, B. Implication of the box C/D snoRNP assembly factor Rsa1p in U3 snoRNP assembly. Nucleic Acids Res. 2017, 45, 7455–7473. [Google Scholar] [CrossRef]
- Yoshikawa, H.; Komatsu, W.; Hayano, T.; Miura, Y.; Homma, K.; Izumikawa, K.; Ishikawa, H.; Miyazawa, N.; Tachikawa, H.; Yamauchi, Y.; et al. Splicing factor 2-associated protein p32 participates in ribosome biogenesis by regulating the binding of Nop52 and fibrillarin to preribosome particles. Mol. Cell Proteom. 2011, 10, M110.006148. [Google Scholar] [CrossRef] [PubMed]
- Lebaron, S.; Froment, C.; Fromont-Racine, M.; Rain, J.C.; Monsarrat, B.; Caizergues-Ferrer, M.; Henry, Y. The splicing ATPase prp43p is a component of multiple preribosomal particles. Mol. Cell Biol. 2005, 25, 9269–9282. [Google Scholar] [CrossRef][Green Version]
- Combs, D.J.; Nagel, R.J.; Ares, M., Jr.; Stevens, S.W. Prp43p is a DEAH-box spliceosome disassembly factor essential for ribosome biogenesis. Mol. Cell Biol. 2006, 26, 523–534. [Google Scholar] [CrossRef]
- Laity, J.H.; Lee, B.M.; Wright, P.E. Zinc finger proteins: New insights into structural and functional diversity. Curr. Opin Struct. Biol. 2001, 11, 39–46. [Google Scholar] [CrossRef]
- Bedford, M.T.; Clarke, S.G. Protein arginine methylation in mammals: Who, what, and why. Mol. Cell 2009, 33, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Bedford, M.T.; Richard, S. Arginine methylation an emerging regulator of protein function. Mol. Cell 2005, 18, 263–272. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaspar, V.P.; Ramos, A.C.; Cloutier, P.; Pattaro Junior, J.R.; Duarte Junior, F.F.; Bouchard, A.; Seixas, F.A.V.; Coulombe, B.; Fernandez, M.A. Interactome Analysis of KIN (Kin17) Shows New Functions of This Protein. Curr. Issues Mol. Biol. 2021, 43, 767-781. https://doi.org/10.3390/cimb43020056
Gaspar VP, Ramos AC, Cloutier P, Pattaro Junior JR, Duarte Junior FF, Bouchard A, Seixas FAV, Coulombe B, Fernandez MA. Interactome Analysis of KIN (Kin17) Shows New Functions of This Protein. Current Issues in Molecular Biology. 2021; 43(2):767-781. https://doi.org/10.3390/cimb43020056
Chicago/Turabian StyleGaspar, Vanessa Pinatto, Anelise Cardoso Ramos, Philippe Cloutier, José Renato Pattaro Junior, Francisco Ferreira Duarte Junior, Annie Bouchard, Flavio Augusto Vicente Seixas, Benoit Coulombe, and Maria Aparecida Fernandez. 2021. "Interactome Analysis of KIN (Kin17) Shows New Functions of This Protein" Current Issues in Molecular Biology 43, no. 2: 767-781. https://doi.org/10.3390/cimb43020056
APA StyleGaspar, V. P., Ramos, A. C., Cloutier, P., Pattaro Junior, J. R., Duarte Junior, F. F., Bouchard, A., Seixas, F. A. V., Coulombe, B., & Fernandez, M. A. (2021). Interactome Analysis of KIN (Kin17) Shows New Functions of This Protein. Current Issues in Molecular Biology, 43(2), 767-781. https://doi.org/10.3390/cimb43020056







