Rhenium Perrhenate (188ReO4) Induced Apoptosis and Reduced Cancerous Phenotype in Liver Cancer Cells
Abstract
1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Generation of HCC-LEM
2.3. Cell Proliferation Assay
2.4. Viability Assay
2.5. Cell Cycle Analysis
2.6. Apoptosis Assessment by Flow Cytometric Analysis
2.7. Quantitative Reverse Transcriptase–Polymerase Chain Reaction (qRT-PCR)
2.8. Histological Assessment and Immunofluorescence Assay
2.9. Immunoblotting Analysis
2.10. Hepatocellular Carcinoma (HCC) Mouse Model
2.11. Tumor Formation Assay
2.12. Statistical Analysis
3. Results
3.1. Assessment of 2D Construction
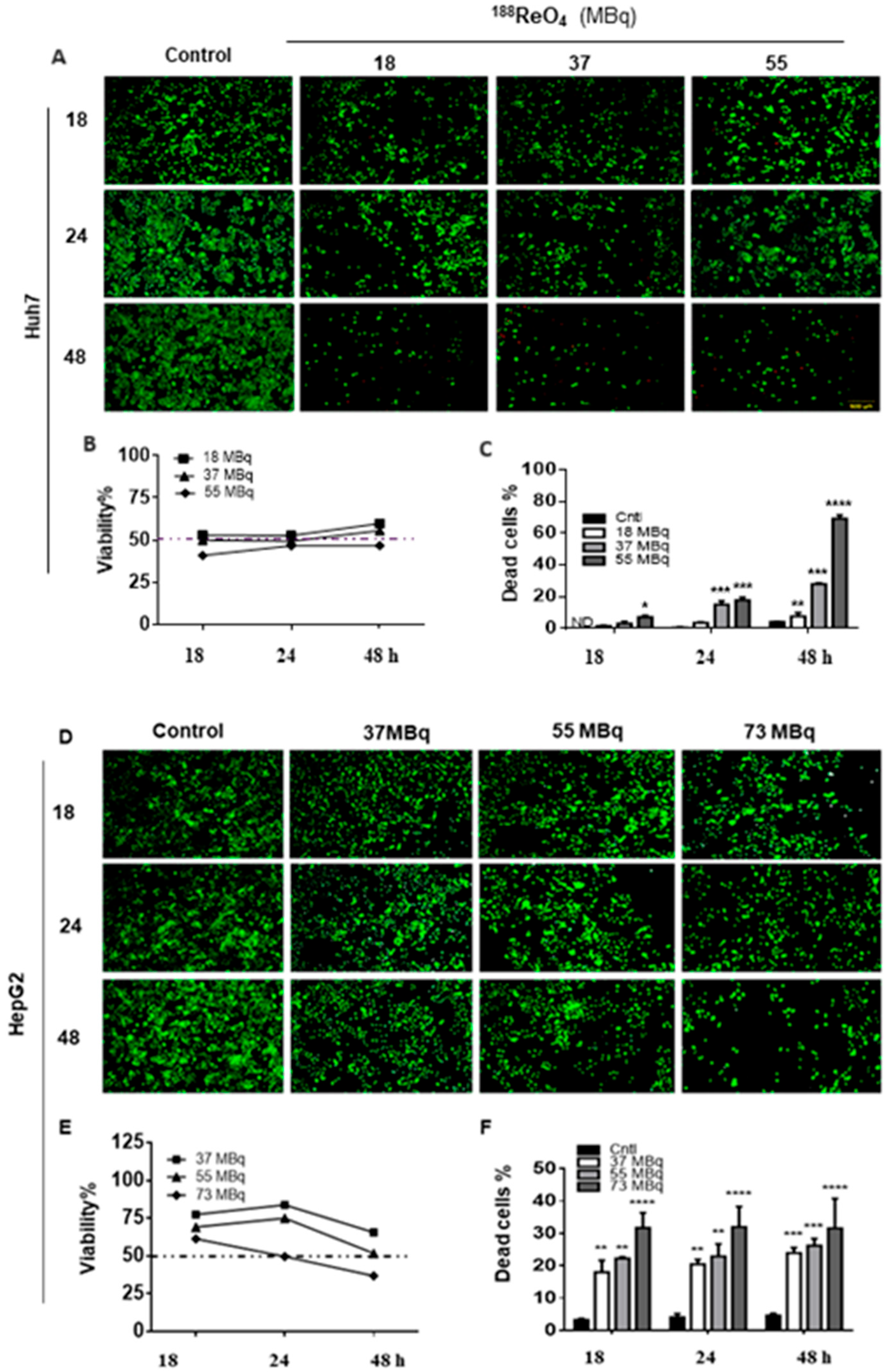
3.1.1. 188ReO4 Treatment Reduced Viability in 2D Cultured Liver Cancer Cells
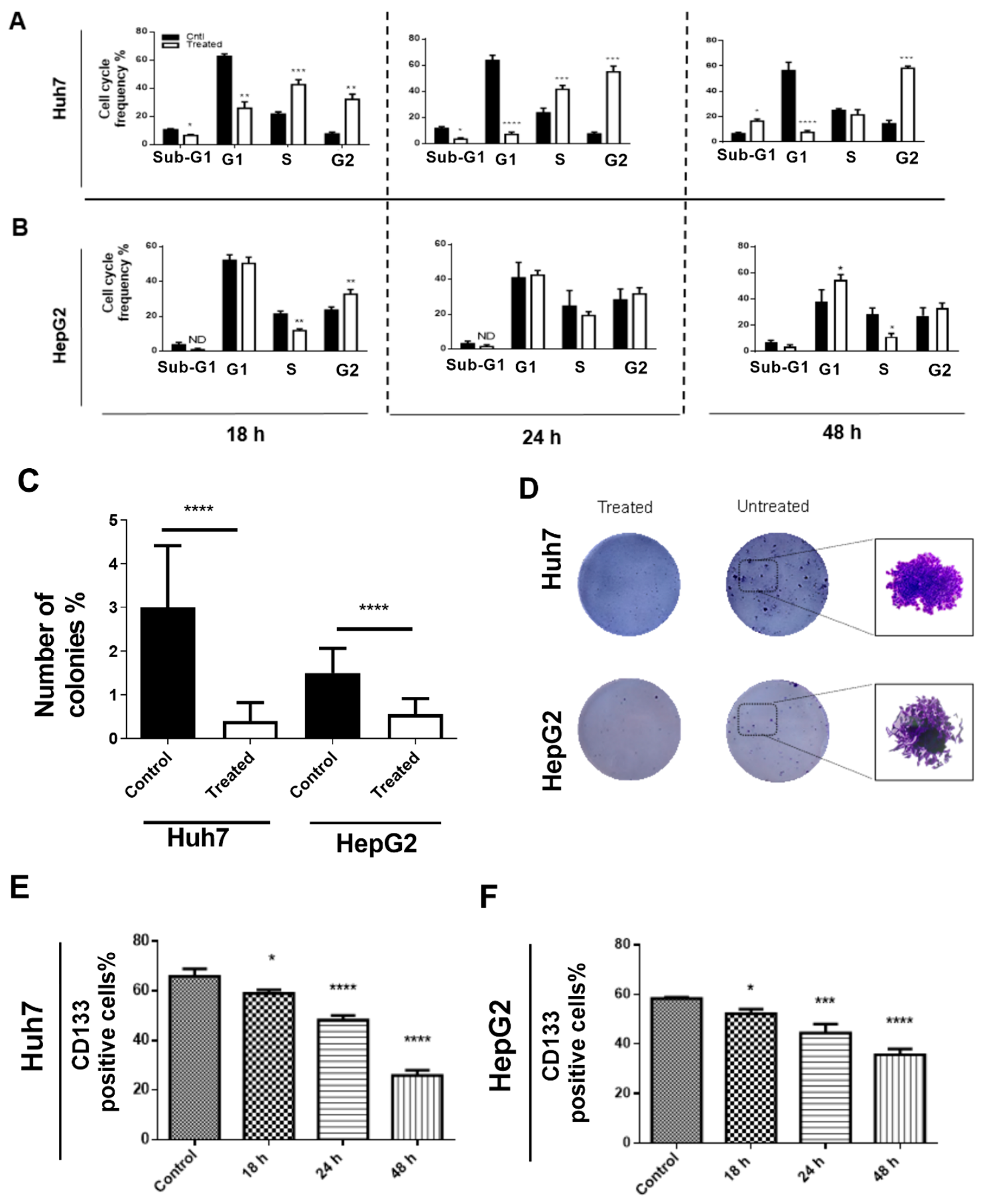
3.1.2. 188ReO4 Induced G2/M Arrest in Huh7 Cancer Cells but Not HepG2
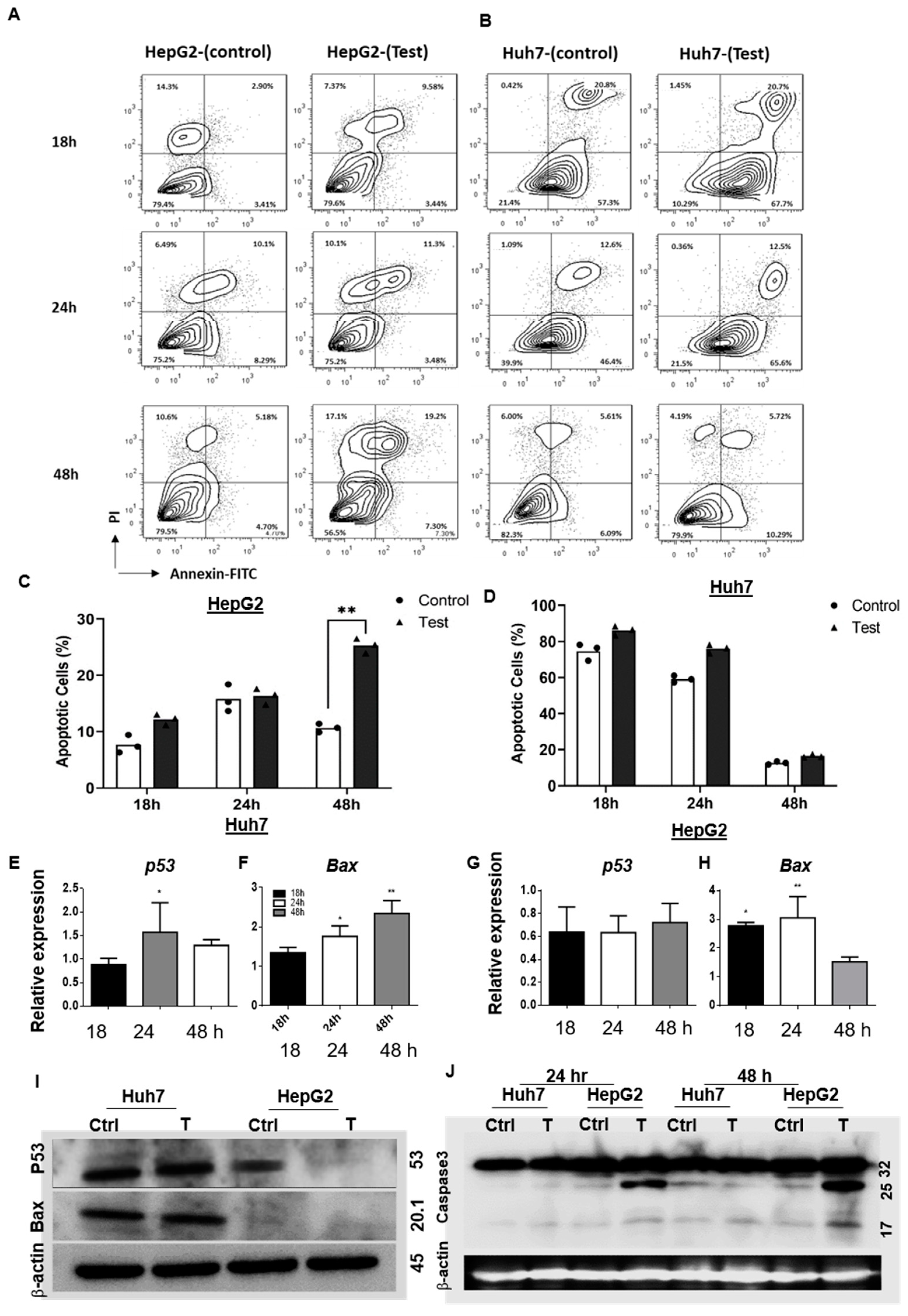
3.1.3. 188ReO4 Induced Apoptosis in Hepatic Cancer Cells
3.2. Assessment of 3D Constructs
3.2.1. 188ReO4 Exposure Reduced Viability in Both Cell Lines in 3D LEM Constructs
3.2.2. 188ReO4 Induced Apoptosis in Cells Cultured on 3D LEM Constructs
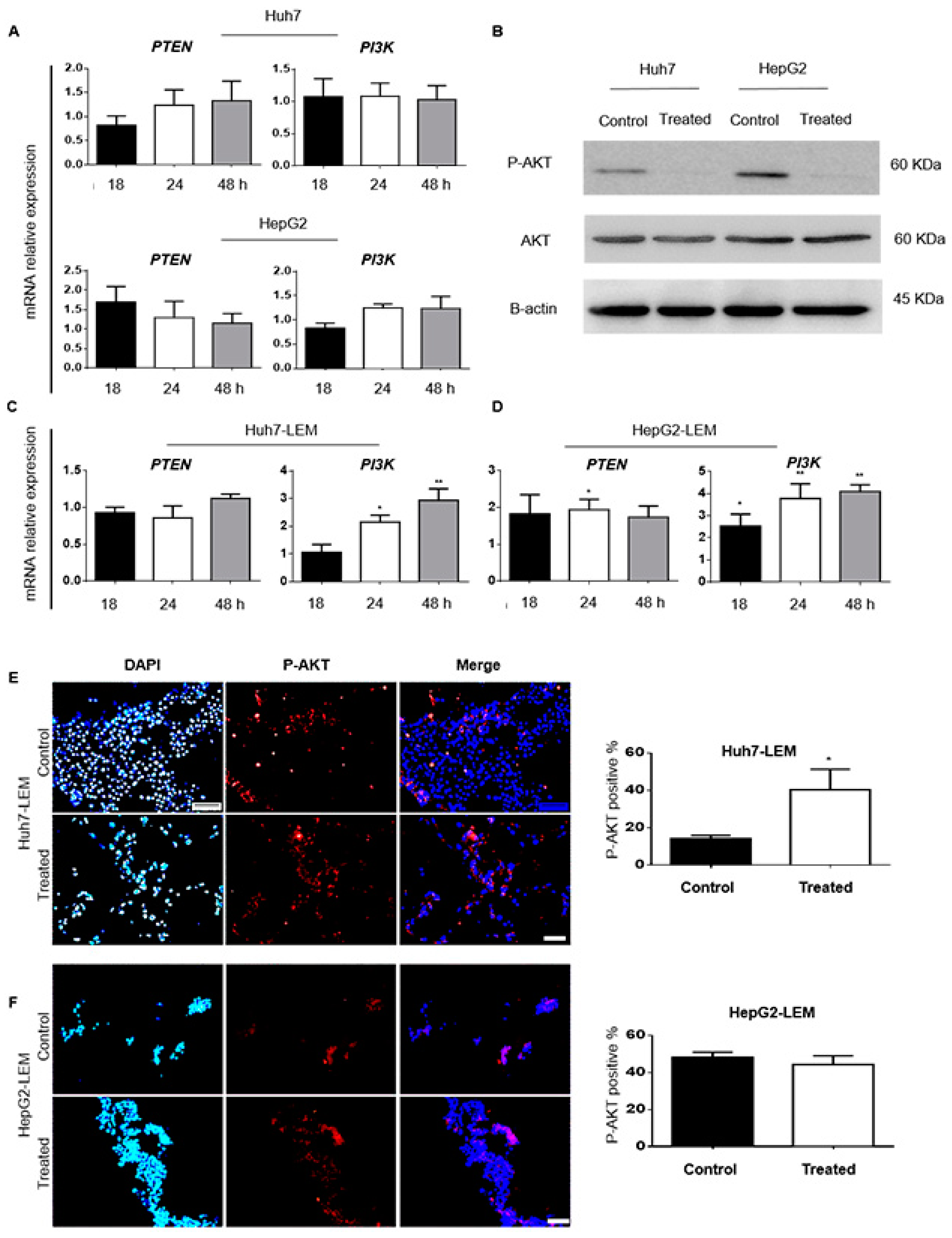
3.2.3. 188ReO4 Overcame Radio-Resistance in 2D but Not in 3D Condition
3.2.4. 188ReO4-Treated Cells Did Not Initiate or Develop Tumor in Nude Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Board Review Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Sun, H.-C.; Wang, Z.; Cong, W.-M.; Wang, J.-H.; Zeng, M.-S.; Yang, J.-M.; Bie, P.; Liu, L.-X.; Wen, T.-F.; et al. Guidelines for Diagnosis and Treatment of Primary Liver Cancer in China (2017 Edition). Liver Cancer 2018, 7, 235–260. [Google Scholar] [CrossRef]
- Lasfar, A. Introductory Chapter: Liver Cancer, Risk Factors and Current Therapies; IntechOpen: London, UK, 2018. [Google Scholar]
- Hernaez, R.; El-Serag, H.B. How we approach it: Treatment options for hepatocellular carcinoma. Am. J. Gastroenterol. 2018, 113, 791–794. [Google Scholar] [CrossRef] [PubMed]
- Asadian, S.; Mirzaei, H.; Kalantari, B.A.; Davarpanah, M.R.; Mohamadi, M.; Shpichka, A.; Nasehi, L.; Es, H.A.; Timashev, P.; Najimi, M.; et al. β-radiating radionuclides in cancer treatment, novel insight into promising approach. Pharmacol. Res. 2020, 160, 105070. [Google Scholar] [CrossRef] [PubMed]
- Lovitt, C.J.; Shelper, T.B.; Avery, V.M. Advanced Cell Culture Techniques for Cancer Drug Discovery. Biology 2014, 3, 345–367. [Google Scholar] [CrossRef]
- Xia, L.; Sakban, R.B.; Qu, Y.; Hong, X.; Zhang, W.; Nugraha, B.; Tong, W.H.; Ananthanarayanan, A.; Zheng, B.; Chau, I.Y.-Y.; et al. Tethered spheroids as an in vitro hepatocyte model for drug safety screening. Biomaterials 2012, 33, 2165–2176. [Google Scholar] [CrossRef]
- Yamashita, T.; Ji, J.; Budhu, A.; Forgues, M.; Yang, W.; Wang, H.Y.; Jia, H.; Ye, Q.; Qin, L.; Wauthier, E.; et al. EpCAM-positive hepatocellular carcinoma cells are tumor-initiating cells with stem/progenitor cell features. Gastroenterology 2009, 136, 1012–1024.e4. [Google Scholar] [CrossRef]
- Kumar, C.; Sharma, R.; Vats, K.; Mallia, M.B.; Das, T.; Sarma, H.D.; Dash, A. Comparison of the efficacy of 177Lu-EDTMP, 177Lu-DOTMP and 188Re-HEDP towards bone osteosarcoma: An in vitro study. J. Radioanal. Nucl. Chem. 2018, 319, 51–59. [Google Scholar] [CrossRef]
- Liu, R.; Zhao, T.; Swat, M.H.; Reynoso, F.J.; Higley, K.A. Development of computational model for cell dose and DNA damage quantification of multicellular system. Int. J. Radiat. Biol. 2019, 95, 1484–1497. [Google Scholar] [CrossRef]
- Roobol, S.J.; Hartjes, T.A.; Slotman, J.A.; de Kruijff, R.; Torrelo, G.; Abraham, T.E.; Bruchertseifer, F.; Morgenstern, A.; Kanaar, R.; Van Gent, D.C.; et al. Uptake and subcellular distribution of radiolabeled polymersomes for radiotherapy. Nanotheranostics 2020, 4, 14–25. [Google Scholar] [CrossRef]
- Falzone, N.; Ackerman, N.L.; Rosales, L.D.L.F.; Bernal, M.A.; Liu, X.; Peeters, S.G.; Soto, M.S.; Corroyer-Dulmont, A.; Bernaudin, M.; Grimoin, E.; et al. Dosimetric evaluation of radionuclides for VCAM-1-targeted radionuclide therapy of early brain metastases. Theranostics 2018, 8, 292–303. [Google Scholar] [CrossRef]
- Terry, S.Y.A.; Nonnekens, J.; Aerts, A.; Baatout, S.; De Jong, M.; Cornelissen, B.; Pouget, J.-P. Call to arms: Need for radiobiology in molecular radionuclide therapy. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1588–1590. [Google Scholar] [CrossRef]
- Vinci, M.; Gowan, S.; Boxall, F.; Patterson, L.; Zimmermann, M.; Court, W.; Lomas, C.; Mendiola, M.; Hardisson, D.; Eccles, S.A. Advances in establishment and analysis of three-dimensional tumor spheroid-based functional assays for target validation and drug evaluation. BMC Biol. 2012, 10, 29. [Google Scholar] [CrossRef] [PubMed]
- Leong, D.T.; Ng, K.W. Probing the relevance of 3D cancer models in nanomedicine research. Adv. Drug Deliv. Rev. 2014, 79, 95–106. [Google Scholar] [CrossRef]
- Saldin, L.T.; Cramer, M.C.; Velankar, S.S.; White, L.J.; Badylak, S.F. Extracellular matrix hydrogels from decellularized tissues: Structure and function. Acta Biomater. 2017, 49, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Nietzer, S.; Baur, F.; Sieber, S.; Hansmann, J.; Schwarz, T.; Stoffer, C.; Häfner, H.; Gasser, M.; Waaga-Gasser, A.M.; Walles, H.; et al. Mimicking Metastases Including Tumor Stroma: A New Technique to Generate a Three-Dimensional Colorectal Cancer Model Based on a Biological Decellularized Intestinal Scaffold. Tissue Eng. Part C Methods 2016, 22, 621–635. [Google Scholar] [CrossRef]
- Guo, L.; Dial, S.; Shi, L.; Branham, W.; Liu, J.; Fang, J.-L.; Green, B.; Deng, H.; Kaput, J.; Ning, B. Similarities and Differences in the Expression of Drug-Metabolizing Enzymes between Human Hepatic Cell Lines and Primary Human Hepatocytes. Drug Metab. Dispos. 2010, 39, 528–538. [Google Scholar] [CrossRef] [PubMed]
- Chao, H.-R.; Tsou, T.-C.; Chen, H.-T.; Chang, E.E.; Tsai, F.-Y.; Lin, D.-Y.; Chen, F.-A.; Wang, Y.-F. The inhibition effect of 2,3,7,8-tetrachlorinated dibenzo-p-dioxin-induced aryl hydrocarbon receptor activation in human hepatoma cells with the treatment of cadmium chloride. J. Hazard. Mater. 2009, 170, 351–356. [Google Scholar] [CrossRef] [PubMed]
- Wee, X.K.; Yeo, W.K.; Zhang, B.; Tan, V.; Lim, K.M.; Tay, T.-E.; Go, M.-L. Synthesis and evaluation of functionalized isoindigos as antiproliferative agents. Bioorganic Med. Chem. 2009, 17, 7562–7571. [Google Scholar] [CrossRef] [PubMed]
- Lahrood, F.N.; Saheli, M.; Farzaneh, Z.; Taheri, P.; Dorraj, M.; Baharvand, H.; Vosough, M.; Piryaei, A. Generation of Transplantable Three-Dimensional Hepatic-Patch to Improve the Functionality of Hepatic Cells In Vitro and In Vivo. Stem Cells Dev. 2020, 29, 301–313. [Google Scholar] [CrossRef]
- Asadian-Samieh, N.G.; Mehdi, A.; Massoud, V. 188 Rhenium Treatment Increases DACT2 Expression In Hepatocellular Carcinoma Cell Line. Cell J. 2021, 24, 3. [Google Scholar]
- Lepareur, N.; Lacœuille, F.; Bouvry, C.; Hindré, F.; Garcion, E.; Chérel, M.; Noiret, N.; Garin, E.; Knapp, F.F.R.J. Rhenium-188 Labeled Radiopharmaceuticals: Current Clinical Applications in Oncology and Promising Perspectives. Front. Med. 2019, 6, 132. [Google Scholar] [CrossRef] [PubMed]
- Seelam, S.R.; Banka, V.K.; Lee, Y.-S.; Jeong, J.M. 188 Re Labeled liver therapeutic drugs for hepatic carcinoma (HCC). J. Radiopharm. Mol. Probes 2019, 5, 26–35. [Google Scholar]
- Sharma, S.V.; Haber, D.A.; Settleman, J. Cell line-based platforms to evaluate the therapeutic efficacy of candidate anticancer agents. Nat. Cancer 2010, 10, 241–253. [Google Scholar] [CrossRef]
- Masters, J.R.W. Human cancer cell lines: Fact and fantasy. Nat. Rev. Mol. Cell Biol. 2000, 1, 233–236. [Google Scholar] [CrossRef]
- Debnath, J.; Brugge, J.S. Modelling glandular epithelial cancers in three-dimensional cultures. Nat. Cancer 2005, 5, 675–688. [Google Scholar] [CrossRef] [PubMed]
- Ridky, T.; Chow, J.M.; Wong, D.J.; Khavari, P.A. Invasive three-dimensional organotypic neoplasia from multiple normal human epithelia. Nat. Med. 2010, 16, 1450–1455. [Google Scholar] [CrossRef]
- Simpson, P.V.; Casari, I.; Paternoster, S.; Skelton, B.; Falasca, M.; Massi, M. Defining the Anti-Cancer Activity of Tricarbonyl Rhenium Complexes: Induction of G2/M Cell Cycle Arrest and Blockade of Aurora-A Kinase Phosphorylation. Chem. A Eur. J. 2017, 23, 6518–6521. [Google Scholar] [CrossRef]
- Simpson, P.V.; Falasca, M.; Massi, M. Properties and prospects for rhenium(i) tricarbonyl N-heterocyclic carbene complexes. Chem. Commun. 2018, 54, 12429–12438. [Google Scholar] [CrossRef]
- Gilbertz, D.; Van Beuningen, A.P.; Rhein, K. Early changes in cell cycle kinetics after ionizing irradiation below 1 Gy. Int. J. Radiat. Biol. 1998, 73, 187–195. [Google Scholar] [CrossRef]
- Meredith, R.; Wessels, B.; Knox, S. Risks to Normal Tissues From Radionuclide Therapy. Semin. Nucl. Med. 2008, 38, 347–357. [Google Scholar] [CrossRef]
- Gheibi, N.; Azad, M.; Vosough, M.; Asadian, S.; Piryaei, A.; Farzaneh, Z.; Kalantari, B.; Timashev, P.; Shipichka, A.; Davarpanah, M.; et al. 188Rhenium Treatment Increases DACT2 Expression In Hepatocellular Carcinoma Cell Line. Cell J. 2022; in press. [Google Scholar]
- Neti, P.V.; Howell, R.W. Log normal distribution of cellular uptake of radioactivity: Implications for biologic responses to radiopharmaceuticals. J. Nucl. Med. 2006, 47, 1049–1058. [Google Scholar]
- Kim, J.B. (Ed.) Three-Dimensional Tissue Culture Models in Cancer Biology. Seminars in Cancer Biology; Elsevier: Amsterdam, The Netherlands, 2005. [Google Scholar]
- Minchinton, A.I.; Tannock, I.F. Drug penetration in solid tumours. Nat. Cancer 2006, 6, 583–592. [Google Scholar] [CrossRef]
- Lee, J.S.; Shin, J.; Park, H.-M.; Kim, Y.-G.; Kim, B.-G.; Oh, J.-W.; Cho, S.-W. Liver Extracellular Matrix Providing Dual Functions of Two-Dimensional Substrate Coating and Three-Dimensional Injectable Hydrogel Platform for Liver Tissue Engineering. Biomacromolecules 2014, 15, 206–218. [Google Scholar] [CrossRef]
- Begg, A.C.; Stewart, F.A.; Vens, C. Strategies to improve radiotherapy with targeted drugs. Nat. Cancer 2011, 11, 239–253. [Google Scholar] [CrossRef] [PubMed]
- Levine, A.J.; Oren, M. The first 30 years of p53: Growing ever more complex. Nat. Cancer 2009, 9, 749–758. [Google Scholar] [CrossRef] [PubMed]
- He, M.; Zhao, M.; Shen, B.; Prise, K.; Shao, C. Radiation-induced intercellular signaling mediated by cytochrome-c via a p53-dependent pathway in hepatoma cells. Oncogene 2010, 30, 1947–1955. [Google Scholar] [CrossRef] [PubMed]
- Bressac, B.; Galvin, K.M.; Liang, T.J.; Isselbacher, K.J.; Wands, J.R.; Ozturk, M. Abnormal structure and expression of p53 gene in human hepatocellular carcinoma. Proc. Natl. Acad. Sci. USA 1990, 87, 1973–1977. [Google Scholar] [CrossRef]
- Zhang, Y.; Fujita, N.; Tsuruo, T.J.O. Caspase-mediated cleavage of p21 Waf1/Cip1 converts cancer cells from growth arrest to undergoing apoptosis. Oncogene 1999, 18, 1131–1138. [Google Scholar] [CrossRef]
- Maier, P.; Hartmann, L.; Wenz, F.; Herskind, C. Cellular Pathways in Response to Ionizing Radiation and Their Targetability for Tumor Radiosensitization. Int. J. Mol. Sci. 2016, 17, 102. [Google Scholar] [CrossRef]
- Ogura, A.; Oowada, S.; Kon, Y.; Hirayama, A.; Yasui, H.; Meike, S.; Kobayashi, S.; Kuwabara, M.; Inanami, O. Redox regulation in radiation-induced cytochrome c release from mitochondria of human lung carcinoma A549 cells. Cancer Lett. 2009, 277, 64–71. [Google Scholar] [CrossRef] [PubMed]
- Richter, C.; Frei, B.J. Medicine. Ca2+ release from mitochondria induced by prooxidants. Free. Radic. Biol. Med. 1988, 4, 365–375. [Google Scholar] [CrossRef]
- Meads, M.B.; Gatenby, R.A.; Dalton, W.S. Environment-mediated drug resistance: A major contributor to minimal residual disease. Nat. Cancer 2009, 9, 665–674. [Google Scholar] [CrossRef]
- Sermeus, A.; Cosse, J.-P.; Crespin, M.; Mainfroid, V.; de Longueville, F.; Ninane, N.; Raes, M.; Remacle, J.; Michiels, C. Hypoxia induces protection against etoposide-induced apoptosis: Molecular profiling of changes in gene expression and transcription factor activity. Mol. Cancer 2008, 7, 27. [Google Scholar] [CrossRef]
- Chang, L.; Graham, P.H.; Ni, J.; Hao, J.; Bucci, J.; Cozzi, P.J.; Li, Y. Targeting PI3K/Akt/mTOR signaling pathway in the treatment of prostate cancer radioresistance. Crit. Rev. Oncol. 2015, 96, 507–517. [Google Scholar] [CrossRef] [PubMed]
- Bamodu, O.A.; Chang, H.-L.; Ong, J.-R.; Lee, W.-H.; Yeh, C.-T.; Tsai, J.-T. Elevated PDK1 Expression Drives PI3K/AKT/MTOR Signaling Promotes Radiation-Resistant and Dedifferentiated Phenotype of Hepatocellular Carcinoma. Cells 2020, 9, 746. [Google Scholar] [CrossRef] [PubMed]
- Burma, S.; Chen, D.J. Role of DNA–PK in the cellular response to DNA double-strand breaks. DNA Repair 2004, 3, 909–918. [Google Scholar] [CrossRef]
- Toulany, M.; Kasten-Pisula, U.; Brammer, I.; Wang, S.; Chen, J.; Dittmann, K.; Baumann, M.; Dikomey, E.; Rodemann, H.P. Blockage of Epidermal Growth Factor Receptor-Phosphatidylinositol 3-Kinase-AKT Signaling Increases Radiosensitivity of K-RAS Mutated Human Tumor Cells In vitro by Affecting DNA Repair. Clin. Cancer Res. 2006, 12, 4119–4126. [Google Scholar] [CrossRef]
- Leung, M.; Kievit, F.; Florczyk, S.; Veiseh, O.; Wu, J.; Park, J.O.; Zhang, M. Chitosan-Alginate Scaffold Culture System for Hepatocellular Carcinoma Increases Malignancy and Drug Resistance. Pharm. Res. 2010, 27, 1939–1948. [Google Scholar] [CrossRef] [PubMed]
- Hussein, K.H.; Park, K.M.; Ghim, J.H.; Yang, S.R.; Woo, H.M. Three dimensional culture of HepG2 liver cells on a rat decellularized liver matrix for pharmacological studies. J. Biomed. Mater. Res. Part B Appl. Biomater. 2016, 104, 263–273. [Google Scholar] [CrossRef]
- Mazza, G.; Rombouts, K.; Hall, A.R.; Urbani, L.; Luong, T.V.; Al-Akkad, W.; Longato, L.; Brown, D.; Maghsoudlou, P.; Dhillon, A.P.; et al. Decellularized human liver as a natural 3D-scaffold for liver bioengineering and transplantation. Sci. Rep. 2015, 5, 13079. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Asadian, S.; Piryaei, A.; Gheibi, N.; Aziz Kalantari, B.; Reza Davarpanah, M.; Azad, M.; Kapustina, V.; Alikhani, M.; Moghbeli Nejad, S.; Keshavarz Alikhani, H.; et al. Rhenium Perrhenate (188ReO4) Induced Apoptosis and Reduced Cancerous Phenotype in Liver Cancer Cells. Cells 2022, 11, 305. https://doi.org/10.3390/cells11020305
Asadian S, Piryaei A, Gheibi N, Aziz Kalantari B, Reza Davarpanah M, Azad M, Kapustina V, Alikhani M, Moghbeli Nejad S, Keshavarz Alikhani H, et al. Rhenium Perrhenate (188ReO4) Induced Apoptosis and Reduced Cancerous Phenotype in Liver Cancer Cells. Cells. 2022; 11(2):305. https://doi.org/10.3390/cells11020305
Chicago/Turabian StyleAsadian, Samieh, Abbas Piryaei, Nematollah Gheibi, Bagher Aziz Kalantari, Mohamad Reza Davarpanah, Mehdi Azad, Valentina Kapustina, Mehdi Alikhani, Sahar Moghbeli Nejad, Hani Keshavarz Alikhani, and et al. 2022. "Rhenium Perrhenate (188ReO4) Induced Apoptosis and Reduced Cancerous Phenotype in Liver Cancer Cells" Cells 11, no. 2: 305. https://doi.org/10.3390/cells11020305
APA StyleAsadian, S., Piryaei, A., Gheibi, N., Aziz Kalantari, B., Reza Davarpanah, M., Azad, M., Kapustina, V., Alikhani, M., Moghbeli Nejad, S., Keshavarz Alikhani, H., Mohamadi, M., Shpichka, A., Timashev, P., Hassan, M., & Vosough, M. (2022). Rhenium Perrhenate (188ReO4) Induced Apoptosis and Reduced Cancerous Phenotype in Liver Cancer Cells. Cells, 11(2), 305. https://doi.org/10.3390/cells11020305







