pH Dependent Antimicrobial Peptides and Proteins, Their Mechanisms of Action and Potential as Therapeutic Agents
Abstract
:1. Introduction
2. An Overview of pH Dependent Peptides and Proteins with Antimicrobial Activity
2.1. Fish
2.2. Amphibians
2.3. Humans and Other Mammals
2.4. Marine Invertebrates
2.5. Terrestrial Invertebrates
3. Potential Applications of pH Dependent Antimicrobial Peptides and Proteins
4. Discussion
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Prestinaci, F.; Pezzotti, P.; Pantosti, A. Antimicrobial resistance: A global multifaceted phenomenon. Pathog. Glob. Health 2015, 109, 309–318. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Antimicrobial Resistance: Global Report on Surveillance 2014; World Health Organization: Geneva, Switzerland, 2014. [Google Scholar]
- O’Neill, J. Tackling Drug-Resistant Infections Globally: Final Report and Recommendations. Available online: https://amr-review.org/sites/default/files/160518_Final%20paper_with%20cover.pdf (accessed on 10 June 2016).
- Phoenix, D.A.; Harris, F.; Dennison, S.R. Novel Antimicrobial Agents and Strategies; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2014. [Google Scholar]
- Nobrega, F.L.; Costa, A.R.; Kluskens, L.D.; Azeredo, J. Revisiting phage therapy: New applications for old resources. Trends Microbiol. 2015, 23, 185–191. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Pierpoint, L. Photodynamic therapy based on 5-aminolevulinic acid and its use as an antimicrobial agent. Med. Res. Rev. 2012, 32, 1292–1327. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Using sound for microbial eradication-light at the end of the tunnel? FEMS Microbiol. Lett. 2014, 356, 20–22. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Sounding the death knell for microbes? Trends Mol. Med. 2014, 20, 363–367. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Anionic antimicrobial peptides from eukaryotic organisms. Curr. Protein Pept. Sci. 2009, 10, 585–606. [Google Scholar] [CrossRef] [PubMed]
- Dutta, P.; Das, S. Mammalian antimicrobial peptides: Promising therapeutic targets against infection and chronic inflammation. Curr. Top. Med. Chem. 2016, 16, 99–129. [Google Scholar] [CrossRef] [PubMed]
- Steinbuch, K.B.; Fridman, M. Mechanisms of resistance to membrane-disrupting antibiotics in gram-positive and gram-negative bacteria. MedChemComm 2016, 7, 86–102. [Google Scholar] [CrossRef]
- Fox, J.L. Antimicrobial peptides stage a comeback. Nat. Biotechnol. 2013, 31, 379–382. [Google Scholar] [CrossRef] [PubMed]
- Midura-Nowaczek, K.; Markowska, A. Antimicrobial peptides and their analogs: Searching for new potential therapeutics. Perspect. Med. Chem. 2014, 6, 73–80. [Google Scholar] [CrossRef] [PubMed]
- Zasloff, M. Antimicrobial Peptides: Do They Have a Future as Therapeutics? Birkhauser Verlag AG: Basel, Switzerland, 2016; pp. 147–154. [Google Scholar]
- Andersson, D.I.; Hughes, D.; Kubicek-Sutherland, J.Z. Mechanisms and consequences of bacterial resistance to antimicrobial peptides. Drug Resist. Updat. 2016, 26, 43–57. [Google Scholar] [CrossRef] [PubMed]
- Lee, T.-H.; Hall, K.N.; Aguilar, M.-I. Antimicrobial peptide structure and mechanism of action: A focus on the role of membrane structure. Curr. Top. Med. Chem. 2016, 16, 25–39. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, D.G. Antimicrobial peptides (amps) with dual mechanisms: Membrane: Disruption and apoptosis. J. Microbiol. Biotechnol. 2015, 25, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Cytrynska, M.; Zdybicka-Barabas, A. Defense peptides: Recent developments. Biomol. Concepts 2015, 6, 237–251. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Dennison, S.R.; Harris, F. Cationic antimicrobial peptides. In Antimicrobial Peptides; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 39–81. [Google Scholar]
- Cruz, J.; Ortiz, C.; Guzman, F.; Fernandez-Lafuente, R.; Torres, R. Antimicrobial peptides: Promising compounds against pathogenic microorganisms. Curr. Med. Chem. 2014, 21, 2299–2321. [Google Scholar] [CrossRef] [PubMed]
- Thaker, H.D.; Cankaya, A.; Scott, R.W.; Tew, G.N. Role of amphiphilicity in the design of synthetic mimics of antimicrobial peptides with gram-negative activity. ACS Med. Chem. Lett. 2013, 4, 481–485. [Google Scholar] [CrossRef] [PubMed]
- Xiong, M.; Lee, M.W.; Mansbach, R.A.; Song, Z.; Bao, Y.; Peek, R.M.; Yao, C.; Chen, L.-F.; Ferguson, A.L.; Wong, G.C.L.; et al. Helical antimicrobial polypeptides with radial amphiphilicity. Proc. Natl. Acad. Sci. USA 2015, 112, 13155–13160. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Dennison, S.R.; Harris, F. Models for the membrane interactions of antimicrobial peptides. In Antimicrobial Peptides; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 145–180. [Google Scholar]
- Hirsch, J.G. Phagocytin: A bactericidal substance from polymorphonuclear leucocytes. J. Exp. Med. 1956, 103, 589–611. [Google Scholar] [CrossRef] [PubMed]
- Hirsch, J.G. Further studies on preparation and properties of phagocytin. J. Exp. Med. 1960, 111, 323–337. [Google Scholar] [CrossRef] [PubMed]
- Kenward, M.A.; Brown, M.R.W.; Fryer, J.J. Influence of calcium or manganese on the resistance to edta, polymyxin-b or cold shock, and the composition of pseudomonas-aeruginosa grown in glucose-depleted or magnesium-depleted batch cultures. J. Appl. Bacteriol. 1979, 47, 489–503. [Google Scholar] [CrossRef] [PubMed]
- Selsted, M.E.; Szklarek, D.; Lehrer, R.I. Purification and antibacterial activity of antimicrobial peptides of rabbit granulocytes. Infect. Immun. 1984, 45, 150–154. [Google Scholar] [PubMed]
- Daher, K.A.; Selsted, M.E.; Lehrer, R.I. Direct inactivation of viruses by human granulocyte defensins. J. Virol. 1986, 60, 1068–1074. [Google Scholar] [PubMed]
- McDonald, M.; Mannion, M.; Pike, D.; Lewis, K.; Flynn, A.; Brannan, A.M.; Browne, M.J.; Jackman, D.; Madera, L.; Coombs, M.R.P.; et al. Structure-function relationships in histidine-rich antimicrobial peptides from atlantic cod. Biochim. Biophys. Acta 2015, 1848, 1451–1461. [Google Scholar] [CrossRef] [PubMed]
- Khatami, M.H.; Bromberek, M.; Saika-Voivod, I.; Booth, V. Molecular dynamics simulations of histidine-containing cod antimicrobial peptide paralogs in self-assembled bilayers. Biochim. Biophys. Acta 2014, 1838, 2778–2787. [Google Scholar] [CrossRef] [PubMed]
- Shang, D.; Sun, Y.; Wang, C.; Ma, L.; Li, J.; Wang, X. Rational design of anti-microbial peptides with enhanced activity and low cytotoxicity based on the structure of the arginine/histidine-rich peptide, chensinin-1. J. Appl. Microbiol. 2012, 113, 677–685. [Google Scholar] [CrossRef] [PubMed]
- Shang, D.J.; Sun, Y.; Wang, C.; Wei, S.; Ma, L.J.; Sun, L. Membrane interaction and antibacterial properties of chensinin-1, an antimicrobial peptide with atypical structural features from the skin of rana chensinensis. Appl. Microbiol. Biotechnol. 2012, 96, 1551–1560. [Google Scholar] [CrossRef] [PubMed]
- Goessler-Schoefberger, R.; Hesser, G.; Muik, M.; Wechselberger, C.; Jilek, A. An orphan dermaseptin from frog skin reversibly assembles to amyloid-like aggregates in a pH-dependent fashion. FEBS J. 2009, 276, 5849–5859. [Google Scholar] [CrossRef] [PubMed]
- Glaser, R.; Harder, J.; Lange, H.; Bartels, J.; Christophers, E.; Schroder, J.M. Antimicrobial psoriasin (s100a7) protects human skin from escherichia coli infection. Nat. Immunol. 2005, 6, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Michalek, M.; Gelhaus, C.; Hecht, O.; Podschun, R.; Schroeder, J.M.; Leippe, M.; Groetzinger, J. The human antimicrobial protein psoriasin acts by permeabilization of bacterial membranes. Dev. Comp. Immunol. 2009, 33, 740–746. [Google Scholar] [CrossRef] [PubMed]
- Mildner, M.; Stichenwirth, M.; Abtin, A.; Eckhart, L.; Sam, C.; Glaser, R.; Schroder, J.M.; Gmeiner, R.; Mlitz, V.; Pammer, J.; et al. Psoriasin (s100a7) is a major escherichia coli-cidal factor of the female genital tract. Mucosal Immunol. 2010, 3, 602–609. [Google Scholar] [CrossRef] [PubMed]
- Edstrom Hagerwall, A.M.; Rydengard, V.; Fernlund, P.; Morgelin, M.; Baumgarten, M.; Cole, A.M.; Malmsten, M.; Kragelund, B.B.; Sorensen, O.E. β-microseminoprotein endows post coital seminal plasma with potent candidacidal activity by a calcium- and pH-dependent mechanism. PLoS Pathog. 2012, 8, e1002625. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Garcia, B.; Lee, P.H.A.; Yamasaki, K.; Gallo, R.L. Anti-fungal activity of cathelicidins and their potential role in candida albicans skin infection. J. Investig. Dermatol. 2005, 125, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Lombardi, L.; Maisetta, G.; Batoni, G.; Tavanti, A. Insights into the antimicrobial properties of hepcidins: Advantages and drawbacks as potential therapeutic agents. Molecules 2015, 20, 6319–6341. [Google Scholar] [CrossRef] [PubMed]
- Tavanti, A.; Maisetta, G.; Del Gaudio, G.; Petruzzelli, R.; Sanguinetti, M.; Batoni, G.; Senesi, S. Fungicidal activity of the human peptide hepcidin 20 alone or in combination with other antifungals against candida glabrata isolates. Peptides 2011, 32, 2484–2487. [Google Scholar] [CrossRef] [PubMed]
- Maisetta, G.; Petruzzelli, R.; Brancatisano, F.L.; Esin, S.; Vitali, A.; Campa, M.; Batoni, G. Antimicrobial activity of human hepcidin 20 and 25 against clinically relevant bacterial strains: Effect of copper and acidic pH. Peptides 2010, 31, 1995–2002. [Google Scholar] [CrossRef] [PubMed]
- Mak, P.; Siwek, M.; Pohl, J.; Dubin, A. Menstrual hemocidin hbb115–146 is an acidophilic antibacterial peptide potentiating the activity of human defensins, cathelicidin and lysozyme. Am. J. Reprod. Immunol. 2007, 57, 81–91. [Google Scholar] [CrossRef] [PubMed]
- Maisetta, G.; Vitali, A.; Scorciapino, M.A.; Rinaldi, A.C.; Petruzzelli, R.; Brancatisano, F.L.; Esin, S.; Stringaro, A.; Colone, M.; Luzi, C.; et al. pH-dependent disruption of escherichiacoli atcc 25922 and model membranes by the human antimicrobial peptides hepcidin 20 and 25. FEBS J. 2013, 280, 2842–2854. [Google Scholar] [CrossRef] [PubMed]
- Del Gaudio, G.; Lombardi, L.; Maisetta, G.; Esin, S.; Batoni, G.; Sanguinetti, M.; Senesi, S.; Tavanti, A. Antifungal activity of the non cytotoxic human peptide hepcidin 20 against fluconazole resistant candida glabrata in human vaginal fluid. Antimicrob. Agents Chemother. 2013. [Google Scholar] [CrossRef] [PubMed]
- Mochon, A.B.; Liu, H. The antimicrobial peptide histatin-5 causes a spatially restricted disruption on the candida albicans surface, allowing rapid entry of the peptide into the cytoplasm. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- Kacprzyk, L.; Rydengard, V.; Morgelin, M.; Davoudi, M.; Pasupuleti, M.; Malmsten, M.; Schmidtchen, A. Antimicrobial activity of histidine-rich peptides is dependent on acidic conditions. Biochim. Biophys. Acta 2007, 1768, 2667–2680. [Google Scholar] [CrossRef] [PubMed]
- Viejo-Diaz, M.; Andres, M.T.; Fierro, J.F. Modulation of in vitro fungicidal activity of human lactoferrin against candida albicans by extracellular cation concentration and target cell metabolic activity. Antimicrob. Agents Chemother. 2004, 48, 1242–1248. [Google Scholar] [CrossRef] [PubMed]
- Song, C.; Weichbrodt, C.; Salnikov, E.S.; Dynowski, M.; Forsberg, B.O.; Bechinger, B.; Steinem, C.; de Groot, B.L.; Zachariae, U.; Zeth, K. Crystal structure and functional mechanism of a human antimicrobial membrane channel. Proc. Natl. Acad. Sci. USA 2013, 110, 4586–4591. [Google Scholar] [CrossRef] [PubMed]
- Paulmann, M.; Arnold, T.; Linke, D.; Özdirekcan, S.; Kopp, A.; Gutsmann, T.; Kalbacher, H.; Wanke, I.; Schuenemann, V.J.; Habeck, M.; et al. Structure-activity analysis of the dermcidin-derived peptide dcd-1l, an anionic antimicrobial peptide present in human sweat. J. Biol. Chem. 2012, 287, 8434–8443. [Google Scholar] [CrossRef] [PubMed]
- Becucci, L.; Valensin, D.; Innocenti, M.; Guidelli, R. Dermcidin, an anionic antimicrobial peptide: Influence of lipid charge, pH and Zn2+ on its interaction with a biomimetic membrane. Soft Matter 2014, 10, 616–626. [Google Scholar] [CrossRef] [PubMed]
- Dashper, S.G.; Liu, S.W.; Reynolds, E.C. Antimicrobial peptides and their potential as oral therapeutic agents. Int. J. Pept. Res. Ther. 2007, 13, 505–516. [Google Scholar] [CrossRef]
- Malkoski, M.; Dashper, S.G.; O’Brien-Simpson, N.M.; Talbo, G.H.; Macris, M.; Cross, K.J.; Reynolds, E.C. Kappacin, a novel antibacterial peptide from bovine milk. Antimicrob. Agents Chemother. 2001, 45, 2309–2315. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, M.R.; Tang, Y.Q.; Shen, A.J.; Bayer, A.S.; Selsted, M.E. Purification and in vitro activities of rabbit platelet microbicidal proteins. Infect. Immun. 1997, 65, 1023–1031. [Google Scholar] [PubMed]
- Sawyer, J.G.; Martin, N.L.; Hancock, R.E. Interaction of macrophage cationic proteins with the outer membrane of pseudomonas aeruginosa. Infect. Immun. 1988, 56, 693–698. [Google Scholar] [PubMed]
- Lehrer, R.I.; Lichtenstein, A.K.; Ganz, T. Defensins: Antimicrobial and cytotoxic peptides of mammalian cells. Annu. Rev. Immunol. 1993, 11, 105–128. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, W.; Wakabayashi, H.; Takase, M.; Kawase, K.; Shimamura, S.; Tomita, M. Killing of candida-albicans by lactoferricin-b, a potent antimicrobial peptide derived from the n-terminal region of bovine lactoferrin. Med. Microbiol. Immunol. 1993, 182, 97–105. [Google Scholar] [CrossRef] [PubMed]
- Bellamy, W.R.; Wakabayashi, H.; Takase, M.; Kawase, K.; Shimamura, S.; Tomita, M. Role of cell-binding in the antibacterial mechanism of lactoferricin B. J. Appl. Bacteriol. 1993, 75, 478–484. [Google Scholar] [PubMed]
- Domeneghetti, S.; Franzoi, M.; Damiano, N.; Norante, R.; El Haifawy, N.M.; Mammi, S.; Marin, O.; Bellanda, M.; Venier, P. Structural and antimicrobial features of peptides related to myticin c, a special defense molecule from the mediterranean mussel mytilus galloprovincialis. J. Agric. Food Chem. 2015, 63, 9251–9259. [Google Scholar] [CrossRef] [PubMed]
- Martinez-Lopez, A.; Antonio Encinar, J.; Maria Medina-Gali, R.; Balseiro, P.; Garcia-Valtanen, P.; Figueras, A.; Novoa, B.; Estepa, A. pH-dependent solution structure and activity of a reduced form of the host-defense peptide myticin c (myt c) from the mussel mytilus galloprovincialis. Mar. Drugs 2013, 11, 2328–2346. [Google Scholar] [CrossRef] [PubMed]
- Yoo, S.; Kim, J.-Y.; Park, S.-C.; Choi, D.Y.; Seo, C.H.; Hahm, K.-S.; Park, Y. Effect of acidic pH on antibacterial action of peptide isolated from korean pen shell (atrina pectinata). J. Pept. Sci. 2011, 17, 353–357. [Google Scholar] [CrossRef] [PubMed]
- Fedders, H.; Leippe, M. A reverse search for antimicrobial peptides in ciona intestinalis: Identification of a gene family expressed in hemocytes and evaluation of activity. Dev. Comp. Immunol. 2008, 32, 286–298. [Google Scholar] [CrossRef] [PubMed]
- Fedders, H.; Michalek, M.; Groetzinger, J.; Leippe, M. An exceptional salt-tolerant antimicrobial peptide derived from a novel gene family of haemocytes of the marine invertebrate ciona intestinalis. Biochem. J. 2008, 416, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Schlusselhuber, M.; Humblot, V.; Casale, S.; Methivier, C.; Verdon, J.; Leippe, M.; Berjeaud, J.-M. Potent antimicrobial peptides against legionella pneumophila and its environmental host, acanthamoeba castellanii. Appl. Microbiol. Biotechnol. 2015, 99, 4879–4891. [Google Scholar] [CrossRef] [PubMed]
- Fedders, H.; Podschun, R.; Leippe, M. The antimicrobial peptide ci-mam-a24 is highly active against multidrug-resistant and anaerobic bacteria pathogenic for humans. Int. J. Antimicrob. Agents 2010, 36, 264–266. [Google Scholar] [CrossRef] [PubMed]
- Mulder, K.C.; de Lima, L.A.; Aguiar, P.S.; Carneiro, F.C.; Franco, O.L.; Dias, S.C.; Parachin, N.S. Production of a modified peptide clavanin in pichia pastoris: Cloning, expression, purification and in vitro activities. AMB Express 2015, 5, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Tincu, J.A.; Taylor, S.W. Antimicrobial peptides from marine invertebrates. Antimicrob. Agents Chemother. 2004, 48, 3645–3654. [Google Scholar] [CrossRef] [PubMed]
- In, I.H.; Zhao, C.; Nguyen, T.; Menzel, L.; Waring, A.J.; Lehrer, R.I.; Sherman, M.A. Clavaspirin, an antibacterial and haemolytic peptide from styela clava. J. Peptid. Res. 2001, 58, 445–456. [Google Scholar] [CrossRef]
- Lehrer, R.I.; Andrew Tincu, J.; Taylor, S.W.; Menzel, L.P.; Waring, A.J. Natural peptide antibiotics from tunicates: Structures, functions and potential uses. Integr. Comp. Biol. 2003, 43, 313–322. [Google Scholar] [CrossRef] [PubMed]
- Lehrer, R.I.; Lee, I.H.; Menzel, L.; Waring, A.; Zhao, C. Clavanins and styelins, α-helical antimicrobial peptides from the hemocytes of styela clava. Adv. Exp. Med. Biol. 2001, 484, 71–76. [Google Scholar] [PubMed]
- Lee, I.H.; Cho, Y.; Lehrer, R.I. Effects of pH and salinity on the antimicrobial properties of clavanins. Infect. Immun. 1997, 65, 2898–2903. [Google Scholar] [PubMed]
- Lee, I.H.; Zhao, C.Q.; Cho, Y.; Harwig, S.S.L.; Cooper, E.L.; Lehrer, R.I. Clavanins, α-helical antimicrobial peptides from tunicate hemocytes. FEBS Lett. 1997, 400, 158–162. [Google Scholar] [CrossRef]
- Van Kan, E.J.M.; Demel, R.A.; Breukink, E.; van der Bent, A.; de Kruijff, B. Clavanin permeabilizes target membranes via two distinctly different pH-dependent mechanisms. Biochemistry 2002, 41, 7529–7539. [Google Scholar] [CrossRef] [PubMed]
- Van Kan, E.J.M.; Demel, R.A.; van der Bent, A.; de Kruijff, B. The role of the abundant phenylalanines in the mode of action of the antimicrobial peptide clavanin. Biochim. Biophys. Acta 2003, 1615, 84–92. [Google Scholar] [CrossRef]
- Van Kan, E.J.M.; Ganchev, D.N.; Snel, M.M.E.; Chupin, V.; van der Bent, A.; de Kruijff, B. The peptide antibiotic clavanin a interacts strongly and specifically with lipid bilayers. Biochemistry 2003, 42, 11366–11372. [Google Scholar] [CrossRef] [PubMed]
- Van Kan, E.J.M.; van der Bent, A.; Demel, R.A.; de Kruijff, B. Membrane activity of the peptide antibiotic clavanin and the importance of its glycine residues. Biochemistry 2001, 40, 6398–6405. [Google Scholar] [CrossRef] [PubMed]
- Saude, A.C.M.; Ombredane, A.S.; Silva, O.N.; Barbosa, J.A.R.G.; Moreno, S.E.; Guerra Araujo, A.C.; Falcao, R.; Silva, L.P.; Dias, S.C.; Franco, O.L. Clavanin bacterial sepsis control using a novel methacrylate nanocarrier. Int. J. Nanomed. 2014, 9, 5055–5069. [Google Scholar]
- Taylor, S.W.; Craig, A.G.; Fischer, W.H.; Park, M.; Lehrer, R.I. Styelin d, an extensively modified antimicrobial peptide from ascidian hemocytes. J. Biol. Chem. 2000, 275, 38417–38426. [Google Scholar] [CrossRef] [PubMed]
- Lai, R.; Takeuchi, H.; Lomas, L.O.; Jonczy, J.; Rigden, D.J.; Rees, H.H.; Turner, P.C. A new type of antimicrobial protein with multiple histidines from the hard tick, amblyomma hebraeum. FASEB J. 2004, 18, 1447–1449. [Google Scholar] [CrossRef] [PubMed]
- Andra, J.; Herbst, R.; Leippe, M. Amoebapores, archaic effector peptides of protozoan origin, are discharged into phagosomes and kill bacteria by permeabilizing their membranes. Dev. Comp. Immunol. 2003, 27, 291–304. [Google Scholar] [CrossRef]
- Bruhn, H.; Riekens, B.; Berninghausen, O.; Leippe, M. Amoebapores and nk-lysin, members of a class of structurally distinct antimicrobial and cytolytic peptides from protozoa and mammals: A comparative functional analysis. Biochem. J. 2003, 375, 737–744. [Google Scholar] [CrossRef] [PubMed]
- Leippe, M. Pore-forming toxins from pathogenic amoebae. Appl. Microbiol. Biotechnol. 2014, 98, 4347–4353. [Google Scholar] [CrossRef] [PubMed]
- Leippe, M.; Bruhn, H.; Hecht, O.; Grotzinger, J. Ancient weapons: The three-dimensional structure of amoebapore a. Trends Parasitol. 2005, 21, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Mann, B.J.; Loftus, B.J. The molecular biology and pathogenicity of entamoeba histolytica. In Pathogen Genomics: Impact on Human Health; Shaw, K.J., Ed.; Humana Press: Totowa, NJ, USA, 2002; pp. 281–302. [Google Scholar]
- Michalek, M.; Sonnichsen, F.D.; Wechselberger, R.; Dingley, A.J.; Hung, C.W.; Kopp, A.; Wienk, H.; Simanski, M.; Herbst, R.; Lorenzen, I.; et al. Structure and function of a unique pore-forming protein from a pathogenic acanthamoeba. Nat. Chem. Biol. 2013, 9, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Banyai, L.; Patthy, L. Amoebapore homologs of caenorhabditis elegans. Biochim. Biophys. Acta 1998, 1429, 259–264. [Google Scholar] [CrossRef]
- Hoeckendorf, A.; Stanisak, M.; Leippe, M. The saposin-like protein spp-12 is an antimicrobial polypeptide in the pharyngeal neurons of caenorhabditis elegans and participates in defence against a natural bacterial pathogen. Biochem. J. 2012, 445, 205–212. [Google Scholar] [CrossRef] [PubMed]
- Hoeckendorf, A.; Leippe, M. Spp-3, a saposin-like protein of caenorhabditis elegans, displays antimicrobial and pore-forming activity and is located in the intestine and in one head neuron. Dev. Comp. Immunol. 2012, 38, 181–186. [Google Scholar] [CrossRef] [PubMed]
- Roeder, T.; Stanisak, M.; Gelhaus, C.; Bruchhaus, I.; Groetzinger, J.; Leippe, M. Caenopores are antimicrobial peptides in the nematode caenorhabditis elegans instrumental in nutrition and immunity. Dev. Comp. Immunol. 2010, 34, 203–209. [Google Scholar] [CrossRef] [PubMed]
- Dierking, K.; Yang, W.; Schulenburg, H. Antimicrobial effectors in the nematode Caenorhabditis elegans: An outgroup to the Arthropoda. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2016, 371. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Dennison, S.R.; Harris, F. Antimicrobial peptides: Their history, evolution, and functional promiscuity. In Antimicrobial Peptides; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 1–37. [Google Scholar]
- Bruhn, O.; Groetzinger, J.; Cascorbi, I.; Jung, S. Antimicrobial peptides and proteins of the horse-insights into a well-armed organism. Vet. Res. 2011, 42. [Google Scholar] [CrossRef] [PubMed]
- Fan, L.; Sun, J.; Zhou, M.; Zhou, J.; Lao, X.; Zheng, H.; Xu, H. Dramp: A comprehensive data repository of antimicrobial peptides. Sci. Rep. 2016, 6, 24482. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Mishra, B.; Lau, K.; Lushnikova, T.; Golla, R.; Wang, X. Antimicrobial peptides in 2014. Pharmaceuticals 2015, 8, 123–150. [Google Scholar] [CrossRef] [PubMed]
- Browne, M.J.; Feng, C.Y.; Booth, V.; Rise, M.L. Characterization and expression studies of gaduscidin-1 and gaduscidin-2; paralogous antimicrobial peptide-like transcripts from Atlantic cod (gadus morhua). Dev. Comp. Immunol. 2011, 35, 399–408. [Google Scholar] [CrossRef] [PubMed]
- Rise, M.L.; Hall, J.R.; Alcock, B.P.; Hori, T.S. Dynamic expression profiles of virus-responsive and putative antimicrobial peptide-encoding transcripts during Atlantic cod (gadus morhua) embryonic and early larval development. Gene 2012, 509, 232–246. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; He, L.; Li, G.; Zhai, N.; Jiang, H.; Chen, Y. Role of helicity of α-helical antimicrobial peptides to improve specificity. Protein Cell 2014, 5, 631–642. [Google Scholar] [CrossRef] [PubMed]
- Burton, M.F.; Steel, P.G. The chemistry and biology of LL-37. Natl. Prod. Rep. 2009, 26, 1572–1584. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Singh, J.; Phoenix, D.A. On the selectivity and efficacy of defense peptides with respect to cancer cells. Med. Res. Rev. 2013, 33, 190–234. [Google Scholar] [CrossRef] [PubMed]
- Sengupta, D.; Leontiadou, H.; Mark, A.E.; Marrink, S.-J. Toroidal pores formed by antimicrobial peptides show significant disorder. Biochim. Biophys. Acta Biomembr. 2008, 1778, 2308–2317. [Google Scholar] [CrossRef] [PubMed]
- Burkhard, B.; Christopher, A. The polymorphic nature of membrane-active peptides from biophysical and structural investigations. Curr. Protein Pept. Sci. 2012, 13, 602–610. [Google Scholar]
- Shagaghi, N.; Palombo, E.A.; Clayton, A.H.A.; Bhave, M. Archetypal tryptophan-rich antimicrobial peptides: Properties and applications. World J. Microbiol. Biotechnol. 2016, 32, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.I.; Prenner, E.J.; Vogel, H.J. Tryptophan- and arginine-rich antimicrobial peptides: Structures and mechanisms of action. Biochim. Biophys. Acta (BBA)-Biomembr. 2006, 1758, 1184–1202. [Google Scholar] [CrossRef] [PubMed]
- Dong, W.B.; Sun, Y.; Shang, D.J. Interactions between chensinin-1, a natural antimicrobial peptide derived from rana chensinensis, and lipopolysaccharide. Biopolymers 2015, 103, 719–726. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Harris, F.; Mura, M.; Dennison, S.R. The increasing role of phosphatidylethanolamine as a lipid receptor in the action of host defence peptides. Prog. Lipid Res. 2015, 59, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Dennison, S.R.; Harris, F.; Mura, M.; Morton, L.H.G.; Zvelindovsky, A.; Phoenix, D.A. A novel form of bacterial resistance to the action of eukaryotic host defense peptides, the use of a lipid receptor. Biochemistry 2013, 52, 6021–6029. [Google Scholar] [CrossRef] [PubMed]
- Mansour, S.C.; Pena, O.M.; Hancock, R.E.W. Host defense peptides: Front-line immunomodulators. Trends Immunol. 2016, 35, 443–450. [Google Scholar] [CrossRef] [PubMed]
- Kuroda, K.; Okumura, K.; Isogai, H.; Isogai, E. The human cathelicidin antimicrobial peptide LL-37 and mimics are potential anticancer drugs. Front. Oncol. 2015, 5, 144. [Google Scholar] [CrossRef] [PubMed]
- Hancock, R.E.W.; Haney, E.F.; Gill, E.E. The immunology of host defence peptides: Beyond antimicrobial activity. Nat. Rev. Immunol. 2016, 16, 321–334. [Google Scholar] [CrossRef] [PubMed]
- Conlon, J.M. Reflections on a systematic nomenclature for antimicrobial peptides from the skins of frogs of the family ranidae. Peptides 2008, 29, 1815–1819. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Lee, B.J.; Lee, M.H.; Hong, S.G.; Ryu, P.D. Mechanisms of selective antimicrobial activity of gaegurin 4. Korean J. Physiol. Pharmacol. 2009, 13, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.T.V.; Holthausen, D.; Jacob, J.; George, S. Host defense peptides from Asian frogs as potential clinical therapies. Antibiot. Basel 2015, 4, 136–159. [Google Scholar] [CrossRef] [PubMed]
- Haney, E.F.; Hunter, H.N.; Matsuzaki, K.; Vogel, H.J. Solution NMR studies of amphibian antimicrobial peptides: Linking structure to function? Biochim. Biophys. Acta Biomembr. 2009, 1788, 1639–1655. [Google Scholar] [CrossRef] [PubMed]
- Kozić, M.; Vukičević, D.; Simunić, J.; Rončević, T.; Antcheva, N.; Tossi, A.; Juretić, D. Predicting the minimal inhibitory concentration for antimicrobial peptides with rana-box domain. J. Chem. Inf. Model. 2015, 55, 2275–2287. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.J.; Kim, S.S.; Lee, M.H.; Lee, B.J.; Ryu, P.D. Role of c-terminal heptapeptide in pore-forming activity of antimicrobial agent, gaegurin 4. J. Pept. Res. 2004, 64, 151–158. [Google Scholar] [CrossRef] [PubMed]
- Won, H.-S.; Kang, S.-J.; Lee, B.-J. Action mechanism and structural requirements of the antimicrobial peptides, gaegurins. Biochim. Biophys. Acta Biomembr. 2009, 1788, 1620–1629. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, B.O.; Stange, E.F.; Wehkamp, J. Waking the wimp: Redox-modulation activates human β-defensin 1. Gut Microbes 2011, 2, 262–266. [Google Scholar] [CrossRef] [PubMed]
- Schroeder, B.O.; Wu, Z.; Nuding, S.; Groscurth, S.; Marcinowski, M.; Beisner, J.; Buchner, J.; Schaller, M.; Stange, E.F.; Wehkamp, J. Reduction of disulphide bonds unmasks potent antimicrobial activity of human beta-defensin 1. Nature 2011, 469, 419–423. [Google Scholar] [CrossRef] [PubMed]
- Lillywhite, H.B. Water relations of tetrapod integument. J. Exp. Biol. 2006, 209, 202–226. [Google Scholar] [CrossRef] [PubMed]
- Dennison, S.R.; Harris, F.; Phoenix, D.A. Chapter three-langmuir–blodgett approach to investigate antimicrobial peptide–membrane interactions. In Advances in Planar Lipid Bilayers and Liposomes; Aleš, I., Chandrashekhar, V.K., Eds.; Academic Press: Cambridge, MA, USA, 2014; Volume 20, pp. 83–110. [Google Scholar]
- Eun, S.Y.; Jang, H.K.; Han, S.K.; Ryu, P.D.; Lee, B.J.; Han, K.H.; Kim, S.J. A helix-induced oligomeric transition of gaegurin 4, an antimicrobial peptide isolated from a Korean frog. Mol. Cells 2006, 21, 229–236. [Google Scholar] [PubMed]
- Wechselberger, C. Cloning of cdnas encoding new peptides of the dermaseptin-family. Biochim. Biophys. Acta 1998, 1388, 279–283. [Google Scholar] [CrossRef]
- Maji, S.K.; Perrin, M.H.; Sawaya, M.R.; Jessberger, S.; Vadodaria, K.; Rissman, R.A.; Singru, P.S.; Nilsson, K.P.R.; Simon, R.; Schubert, D.; et al. Functional amyloids as natural storage of peptide hormones in pituitary secretory granules. Science 2009, 325, 328–332. [Google Scholar] [CrossRef] [PubMed]
- Franco, O.L. Peptide promiscuity: An evolutionary concept for plant defense. FEBS Lett. 2011, 585, 995–1000. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Aberrant action of amyloidogenic host defense peptides: A new paradigm to investigate neurodegenerative disorders? FASEB J. 2012, 26, 1776–1781. [Google Scholar] [CrossRef] [PubMed]
- Gossler-Schofberger, R.; Hesser, G.; Reif, M.M.; Friedmann, J.; Duscher, B.; Toca-Herrera, J.L.; Oostenbrink, C.; Jilek, A. A stereochemical switch in the adrs model system, a candidate for a functional amyloid. Arch. Biochem. Biophys. 2012, 522, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Phoenix, D.A.; Dennison, S.R.; Harris, F. Graphical techniques to visualize the amphiphilic structures of antimicrobial peptides. In Antimicrobial Peptides; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2013; pp. 115–144. [Google Scholar]
- Fritz, G.; Heizmann, C.W. 3D structures of the calcium and zinc binding s100 proteins. In Handbook of Metalloproteins; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2006. [Google Scholar]
- Rezvanpour, A.; Shaw, G.S. Unique s100 target protein interactions. Gen. Physiol. Biophys. 2009, 28, F39–F46. [Google Scholar] [PubMed]
- Santamaria-Kisiel, L.; Rintala-Dempsey, A.C.; Shaw, G.S. Calcium-dependent and -independent interactions of the s100 protein family. Biochem. J. 2006, 396, 201–214. [Google Scholar] [CrossRef] [PubMed]
- Zimmer, D.B.; Sadosky, P.W.; Weber, D.J. Molecular mechanisms of s100-target protein interactions. Microsc. Res. Tech. 2003, 60, 552–559. [Google Scholar] [CrossRef] [PubMed]
- Brogden, N.K.; Mehalick, L.; Fischer, C.L.; Wertz, P.W.; Brogden, K.A. The emerging role of peptides and lipids as antimicrobial epidermal barriers and modulators of local inflammation. Skin Pharmacol. Physiol. 2012, 25, 167–181. [Google Scholar] [CrossRef] [PubMed]
- Harder, J.; Schroder, J.M.; Glaser, R. The skin surface as antimicrobial barrier: Present concepts and future outlooks. Exp. Dermatol. 2013, 22, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wiesner, J.; Vilcinskas, A. Antimicrobial peptides: The ancient arm of the human immune system. Virulence 2010, 1, 440–464. [Google Scholar] [CrossRef] [PubMed]
- Shukeir, N.; Garde, S.; Wu, J.Z.J.; Panchal, C.; Rabbani, S.A. Prostate secretory protein of 94 amino acids (psp-94) and its peptide (pck3145) as potential therapeutic modalities for prostate cancer. Anti-Cancer Drugs 2005, 16, 1045–1051. [Google Scholar] [CrossRef] [PubMed]
- Sutcliffe, S.; De Marzo, A.M.; Sfanos, K.S.; Laurence, M. Msmb variation and prostate cancer risk: Clues towards a possible fungal etiology. Prostate 2014, 74, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Kosciuczuk, E.M.; Lisowski, P.; Jarczak, J.; Strzalkowska, N.; Jozwik, A.; Horbanczuk, J.; Krzyzewski, J.; Zwierzchowski, L.; Bagnicka, E. Cathelicidins: Family of antimicrobial peptides. A review. Mol. Biol. Rep. 2012, 39, 10957–10970. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, M.; Majewski, S. The role of antimicrobial peptides in chronic inflammatory skin diseases. Postepy Dermatol. Alergol. 2016, 33, 6–12. [Google Scholar] [CrossRef] [PubMed]
- Reinholz, M.; Ruzicka, T.; Schauber, J. Cathelicidin LL-37: An antimicrobial peptide with a role in inflammatory skin disease. Ann. Dermatol. 2012, 24, 126–135. [Google Scholar] [CrossRef] [PubMed]
- Linde, A.; Lushington, G.H.; Abello, J.; Melgarejo, T. Clinical relevance of cathelicidin in infectious disease. J. Clin. Cell. Immunol. 2013, S13. [Google Scholar] [CrossRef]
- Ganz, T.; Nemeth, E. Hepcidin and iron homeostasis. Biochim. Biophys. Acta 2012, 1823, 1434–1443. [Google Scholar] [CrossRef] [PubMed]
- Melino, S.; Santone, C.; Di Nardo, P.; Sarkar, B. Histatins: Salivary peptides with Copper(II)- and Zinc(II)-binding motifs perspectives for biomedical applications. FEBS J. 2014, 281, 657–672. [Google Scholar] [CrossRef] [PubMed]
- De Sousa-Pereira, P.; Amado, F.; Abrantes, J.; Ferreira, R.; Esteues, P.J.; Vitorino, R. An evolutionary perspective of mammal salivary peptide families: Cystatins, histatins, statherin and prps. Arch. Oral Biol. 2013, 58, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Calderón-Santiago, M.; Luque de Castro, M.D. The dual trend in histatins research. Trends Anal. Chem. 2009, 28, 1011–1018. [Google Scholar] [CrossRef]
- Han, J.; Jyoti, M.A.; Song, H.-Y.; Jang, W.S. Antifungal activity and action mechanism of histatin 5-halocidin hybrid peptides against Candida ssp. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- White, M.R.; Helmerhorst, E.J.; Ligtenberg, A.; Karpel, M.; Tecle, T.; Siqueira, W.L.; Oppenheim, F.G.; Hartshorn, K.L. Multiple components contribute to ability of saliva to inhibit influenza viruses. Oral Microbiol. Immunol. 2009, 24, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Vukosavljevic, D.; Custodio, W.; Del Bel Cury, A.A.; Siqueira, W.L. The effect of histatin 5, adsorbed on pmma and hydroxyapatite, on candida albicans colonization. Yeast 2012, 29, 459–466. [Google Scholar] [CrossRef] [PubMed]
- Jang, W.S.; Edgerton, M. Salivary histatins: Structure, function, and mechanisms of antifungal activity. In Candida and Candidiasis, 2nd ed.; American Society of Microbiology: Washington, DC, USA, 2012. [Google Scholar]
- Fabian, T.K.; Hermann, P.; Beck, A.; Fejerdy, P.; Fabian, G. Salivary defense proteins: Their network and role in innate and acquired oral immunity. Int. J. Mol. Sci. 2012, 13, 4295–4320. [Google Scholar] [CrossRef] [PubMed]
- Galgut, P.N. The relevance of pH to gingivitis and periodontitis. J. Int. Acad. Periodontol. 2001, 3, 61–67. [Google Scholar] [PubMed]
- Hold, K.M.; de Boer, B.S.; Zuidema, J.; Maes, R.A.A. Saliva as an analytical tool in toxicology. Int. J. Drug Test. 1999, 1, 1–36. [Google Scholar]
- Forssten, S.D.; Björklund, M.; Ouwehand, A.C. Streptococcus mutans, caries and simulation models. Nutrients 2010, 2, 290–298. [Google Scholar] [CrossRef] [PubMed]
- Davis, D.A. How human pathogenic fungi sense and adapt to pH: The link to virulence. Curr. Opin. Microbiol. 2009, 12, 365–370. [Google Scholar] [CrossRef] [PubMed]
- Metwalli, K.H.; Khan, S.A.; Krom, B.P.; Jabra-Rizk, M.A. Streptococcus mutans, candida albicans, and the human mouth: A sticky situation. PLoS Pathog. 2013, 9, e1003616. [Google Scholar] [CrossRef] [PubMed]
- Xu, T.; Levitz, S.M.; Diamond, R.D.; Oppenheim, F.G. Anticandidal activity of major human salivary histatins. Infect. Immun. 1991, 59, 2549–2554. [Google Scholar] [PubMed]
- Puri, S.; Li, R.; Ruszaj, D.; Tati, S.; Edgerton, M. Iron binding modulates candidacidal properties of salivary histatin 5. J. Dent. Res. 2015, 94, 201–208. [Google Scholar] [CrossRef] [PubMed]
- Kanwar, J.R.; Roy, K.; Patel, Y.; Zhou, S.-F.; Singh, M.R.; Singh, D.; Nasir, M.; Sehgal, R.; Sehgal, A.; Singh, R.S.; et al. Multifunctional iron bound lactoferrin and nanomedicinal approaches to enhance its bioactive functions. Molecules 2015, 20, 9703–9731. [Google Scholar] [CrossRef] [PubMed]
- Singh, P.K.; Parsek, M.R.; Greenberg, E.P.; Welsh, M.J. A component of innate immunity prevents bacterial biofilm development. Nature 2002, 417, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Ammons, M.C.; Ward, L.S.; Fisher, S.T.; Wolcott, R.D.; James, G.A. In vitro susceptibility of established biofilms composed of a clinical wound isolate of pseudomonas aeruginosa treated with lactoferrin and xylitol. Int. J. Antimicrob. Agents 2009, 33, 230–236. [Google Scholar] [CrossRef] [PubMed]
- Ammons, M.C.; Ward, L.S.; James, G.A. Anti-biofilm efficacy of a lactoferrin/xylitol wound hydrogel used in combination with silver wound dressings. Int. Wound J. 2011, 8, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Ammons, M.C.; Copié, V. Lactoferrin: A bioinspired, anti-biofilm therapeutic. Biofouling 2013, 29, 443–455. [Google Scholar] [CrossRef] [PubMed]
- Hurdle, J.G.; O’Neill, A.J.; Chopra, I.; Lee, R.E. Targeting bacterial membrane function: An underexploited mechanism for treating persistent infections. Nat. Rev. Microbiol. 2011, 9, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Sinha, M.; Kaushik, S.; Kaur, P.; Sharma, S.; Singh, T.P. Antimicrobial lactoferrin peptides: The hidden players in the protective function of a multifunctional protein. Int. J. Pept. 2013, 2013, 390230. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, M.R. Platelets: At the nexus of antimicrobial defence. Nat. Rev. Microbiol. 2014, 12, 426–437. [Google Scholar] [CrossRef] [PubMed]
- Yeaman, M.R. The role of platelets in antimicrobial host defense. Clin. Infect. Dis. 1997, 25, 951–968. [Google Scholar] [CrossRef] [PubMed]
- Berkebile, A.R.; McCray, P.B. Effects of airway surface liquid pH on host defense in cystic fibrosis. Int. J. Biochem. Cell Biol. 2014, 52, 124–129. [Google Scholar] [CrossRef] [PubMed]
- Lecaille, F.; Lalmanach, G.; Andrault, P.M. Antimicrobial proteins and peptides in human lung diseases: A friend and foe partnership with host proteases. Biochimie 2016, 122, 151–168. [Google Scholar] [CrossRef] [PubMed]
- Cutting, G.R. Cystic fibrosis genetics: From molecular understanding to clinical application. Nat. Rev. Genet. 2015, 16, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Laubel, D.M.; Yiml, S.; Ryan, L.K.; Kisich, K.O.; Diamond, G. Antimicrobial peptides in the airway. Curr. Top. Microbiol. Immunol. 2006, 306, 153–182. [Google Scholar]
- Waterer, G.W. Airway defense mechanisms. Clin. Chest Med. 2012, 33, 199–209. [Google Scholar] [CrossRef] [PubMed]
- Gray, R.D.; McCullagh, B.N.; McCray, P.B. Nets and cf lung disease: Current status and future prospects. Antibiot. Basel 2015, 4, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Rahman, S.; Gadjeva, M. Does netosis contribute to the bacterial pathoadaptation in cystic fibrosis? Front. Immunol. 2014, 5, 378. [Google Scholar] [CrossRef] [PubMed]
- Nel, J.G.; Theron, A.J.; Pool, R.; Durandt, C.; Tintinger, G.R.; Anderson, R. Neutrophil extracellular traps and their role in health and disease. S. Afr. J. Sci. 2016, 112, 36–44. [Google Scholar] [CrossRef]
- Walton, W.G.; Ahmad, S.; Little, M.R.; Kim, C.S.K.; Tyrrell, J.; Lin, Q.; Di, Y.P.; Tarran, R.; Redinbo, M.R. Structural features essential to the antimicrobial functions of human splunc1. Biochemistry 2016, 55, 2979–2991. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Bartlett, J.A.; Di, M.E.; Bomberger, J.M.; Chan, Y.R.; Gakhar, L.; Mallampalli, R.K.; McCray, P.B.; Di, Y.P. SPLUNC1/BPIFA1 contributes to pulmonary host defense against klebsiella pneumoniae respiratory infection. Am. J. Pathol. 2013, 182, 1519–1531. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zhang, X.; Wu, J.; French, S.W.; He, Z. New insights on the palate, lung, and nasal epithelium clone (plunc) proteins: Based on molecular and functional analysis of its homolog of YH1/SPLUNC1. Exp. Mol. Pathol. 2016, 100, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, S.; Tyrrell, J.; Walton, W.G.; Tripathy, A.; Redinbo, M.R.; Tarran, R. SPLUNC1 has antimicrobial and antibiofilm activity against burkholderia cepacia complex. Antimicrob. Agents Chemother. 2016, 60, 6003–6012. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.H.; Stoltz, D.A.; Karp, P.H.; Ernst, S.E.; Pezzulo, A.A.; Moninger, T.O.; Rector, M.V.; Reznikov, L.R.; Launspach, J.L.; Chaloner, K.; et al. Loss of anion transport without increased sodium absorption characterizes newborn porcine cystic fibrosis airway epithelia. Cell 2010, 143, 911–923. [Google Scholar] [CrossRef] [PubMed]
- Abou Alaiwa, M.H.; Beer, A.M.; Pezzulo, A.A.; Launspach, J.L.; Horan, R.A.; Stoltz, D.A.; Starner, T.D.; Welsh, M.J.; Zabner, J. Neonates with cystic fibrosis have a reduced nasal liquid pH; a small pilot study. J. Cyst. Fibros. 2014, 13, 373–377. [Google Scholar] [CrossRef] [PubMed]
- Garland, A.L.; Walton, W.G.; Coakley, R.D.; Tan, C.D.; Gilmore, R.C.; Hobbs, C.A.; Tripathy, A.; Clunes, L.A.; Bencharit, S.; Stutts, M.J.; et al. Molecular basis for pH-dependent mucosal dehydration in cystic fibrosis airways. Proc. Natl. Acad. Sci. USA 2013, 110, 15973–15978. [Google Scholar] [CrossRef] [PubMed]
- Pezzulo, A.A.; Tang, X.X.; Hoegger, M.J.; Abou Alaiwa, M.H.; Ramachandran, S.; Moninger, T.O.; Karp, P.H.; Wohlford-Lenane, C.L.; Haagsman, H.P.; van Eijk, M.; et al. Reduced airway surface pH impairs bacterial killing in the porcine cystic fibrosis lung. Nature 2012, 487, 109–113. [Google Scholar] [CrossRef] [PubMed]
- Abou Alaiwa, M.H.; Reznikov, L.R.; Gansemer, N.D.; Sheets, K.A.; Horswill, A.R.; Stoltz, D.A.; Zabner, J.; Welsh, M.J. pH modulates the activity and synergism of the airway surface liquid antimicrobials β-defensin-3 and LL-37. Proc. Natl. Acad. Sci. USA 2014, 111, 18703–18708. [Google Scholar] [CrossRef] [PubMed]
- Parkins, M.D.; Floto, R.A. Emerging bacterial pathogens and changing concepts of bacterial pathogenesis in cystic fibrosis. J. Cyst. Fibros. 2015, 14, 293–304. [Google Scholar] [CrossRef] [PubMed]
- Ciofu, O.; Hansen, C.R.; Hoiby, N. Respiratory bacterial infections in cystic fibrosis. Curr. Opin. Pulm. Med. 2013, 19, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Garnett, J.P. Splunc1: Link between acidity and dehydration of the airway surface liquid in CF. Thorax 2014, 69, 1004. [Google Scholar] [CrossRef]
- Tang, X.X.; Ostedgaard, L.S.; Hoegger, M.J.; Moninger, T.O.; Karp, P.H.; McMenimen, J.D.; Choudhury, B.; Varki, A.; Stoltz, D.A.; Welsh, M.J. Acidic pH increases airway surface liquid viscosity in cystic fibrosis. J. Clin. Investig. 2016, 126, 879–891. [Google Scholar] [CrossRef] [PubMed]
- Zeth, K. Dermcidin: What is its antibiotic potential? Future Microbiol. 2013, 8, 817–819. [Google Scholar] [CrossRef] [PubMed]
- Schittek, B. The multiple facets of dermcidin in cell survival and host defense. J. Innat Immun. 2012, 4, 349–360. [Google Scholar] [CrossRef] [PubMed]
- Burian, M.; Schittek, B. The secrets of dermcidin action. Int. J. Med. Microbiol. 2015, 305, 283–286. [Google Scholar] [CrossRef] [PubMed]
- Čipáková, I.; Gašperík, J.; Hostinová, E. Expression and purification of human antimicrobial peptide, dermcidin, in escherichia coli. Protein Expres. Purif. 2006, 45, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Lai, Y.P.; Peng, Y.F.; Zuo, Y.; Li, J.; Huang, J.; Wang, L.F.; Wu, Z.R. Functional and structural characterization of recombinant dermcidin-1l, a human antimicrobial peptide. Biochem. Biophys. Res. Commun. 2005, 328, 243–250. [Google Scholar] [CrossRef] [PubMed]
- Schittek, B.; Hipfel, R.; Sauer, B.; Bauer, J.; Kalbacher, H.; Stevanovic, S.; Schirle, M.; Schroeder, K.; Blin, N.; Meier, F.; et al. Dermcidin: A novel human antibiotic peptide secreted by sweat glands. Nat. Immunol. 2001, 2, 1133–1137. [Google Scholar] [CrossRef] [PubMed]
- Steffen, H.; Rieg, S.; Wiedemann, I.; Kalbacher, H.; Deeg, M.; Sahl, H.G.; Peschel, A.; Götz, F.; Garbe, C.; Schittek, B. Naturally processed dermcidin-derived peptides do not permeabilize bacterial membranes and kill microorganisms irrespective of their charge. Antimicrob. Agents Chemother. 2006, 50, 2608–2620. [Google Scholar] [CrossRef] [PubMed]
- Vuong, C.; Voyich, J.M.; Fischer, E.R.; Braughton, K.R.; Whitney, A.R.; DeLeo, F.R.; Otto, M. Polysaccharide intercellular adhesin (pia) protects staphylococcus epidermidis against major components of the human innate immune system. Cell. Microbiol. 2004, 6, 269–275. [Google Scholar] [CrossRef] [PubMed]
- Benkerroum, N. Antimicrobial peptides generated from milk proteins: A survey and prospects for application in the food industry. A review. Int. J. Dairy Technol. 2010, 63, 320–338. [Google Scholar] [CrossRef]
- Huang, R.; Li, M.; Gregory, R.L. Bacterial interactions in dental biofilm. Virulence 2011, 2, 435–444. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; He, J.; Xue, J.; Wang, Y.; Li, K.; Zhang, K.; Guo, Q.; Liu, X.; Zhou, Y.; Cheng, L.; et al. Oral cavity contains distinct niches with dynamic microbial communities. Environ. Microbiol. 2015, 17, 699–710. [Google Scholar] [CrossRef] [PubMed]
- Dashper, S.G.; O’Brien-Simpson, N.M.; Cross, K.J.; Paolini, R.A.; Hoffmann, B.; Catmull, D.V.; Malkoski, M.; Reynolds, E.C. Divalent metal cations increase the activity of the antimicrobial peptide kappacin. Antimicrob. Agents Chemother. 2005, 49, 2322–2328. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, R.; Mills, S.; Ross, R.P.; Fitzgerald, G.F.; Stanton, C. Bioactive peptides from casein and whey proteins. In Milk and Diary Products; Kanekanian, A., Ed.; Wiley Blackwell: West Sussex, UK, 2014; pp. 23–54. [Google Scholar]
- Costa, M.M.; Dios, S.; Alonso-Gutierrez, J.; Romero, A.; Novoa, B.; Figueras, A. Evidence of high individual diversity on myticin c in mussel (mytilus galloprovincialis). Dev. Comp. Immunol. 2009, 33, 162–170. [Google Scholar] [CrossRef] [PubMed]
- Vera, M.; Martinez, P.; Poisa-Beiro, L.; Figueras, A.; Novoa, B. Genomic organization, molecular diversification, and evolution of antimicrobial peptide myticin-c genes in the mussel (mytilus galloprovincialis). PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Pallavicini, A.; del Mar Costa, M.; Gestal, C.; Dreos, R.; Figueras, A.; Venier, P.; Novoa, B. High sequence variability of myticin transcripts in hemocytes of immune-stimulated mussels suggests ancient host-pathogen interactions. Dev. Comp. Immunol. 2008, 32, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Balseiro, P.; Falco, A.; Romero, A.; Dios, S.; Martinez-Lopez, A.; Figueras, A.; Estepa, A.; Novoa, B. Mytilus galloprovincialis Myticin C: A chemotactic molecule with antiviral activity and immunoregulatory properties. PLoS ONE 2011, 6. [Google Scholar] [CrossRef] [PubMed]
- Brogden, K.A.; Bates, A.M.; Fischer, C.L. Antimicrobial Peptides. In Antimicrobial Peptides in Host Defense: Functions beyond Antimicrobial Activity; Harder, J., Schroder, J.M., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 129–146. [Google Scholar]
- Valdivia-Silva, J.; Medina-Tamayo, J.; Garcia-Zepeda, E.A. Chemokine-derived peptides: Novel antimicrobial and antineoplasic agents. Int. J. Mol. Sci. 2015, 16, 12958–12985. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.; Moser, B. Antimicrobial activities of chemokines: Not just a side-effect? Front. Immunol. 2012, 3, 213. [Google Scholar] [CrossRef] [PubMed]
- Yount, N.Y.; Waring, A.J.; Gank, K.D.; Welch, W.H.; Kupferwasser, D.; Yeaman, M.R. Structural correlates of antimicrobial efficacy in il-8 and related human kinocidins. Biochim. Biophys. Acta Biomembr. 2007, 1768, 598–608. [Google Scholar] [CrossRef] [PubMed]
- Bjorstad, A.; Fu, H.M.; Karlsson, A.; Dahlgren, C.; Bylund, J. Interleukin-8-derived peptide has antibacterial activity. Antimicrob. Agents Chemother. 2005, 49, 3889–3895. [Google Scholar] [CrossRef] [PubMed]
- Di Bella, M.A.; Fedders, H.; De Leo, G.; Leippe, M. Localization of antimicrobial peptides in the tunic of ciona intestinalis (ascidiacea, tunicata) and their involvement in local inflammatory-like reactions. Results Immunol. 2011, 1, 70–75. [Google Scholar] [CrossRef] [PubMed]
- Di Bella, M.A.; Fedders, H.; Leippe, M.; De Leo, G. Antimicrobial peptides in the tunic of ciona intestinalis. In Worldwide Research Efforts in the Fighting against Microbial Pathogensfrom Basic Research to Technological Developments; Mendez-Vilas, A., Ed.; Brown Walker Press: Boca Raton, FL, USA, 2013; pp. 63–67. [Google Scholar]
- Jena, P.; Mishra, B.; Leippe, M.; Hasilik, A.; Griffiths, G.; Sonawane, A. Membrane-active antimicrobial peptides and human placental lysosomal extracts are highly active against mycobacteria. Peptides 2011, 32, 881–887. [Google Scholar] [CrossRef] [PubMed]
- Carratala, J.; Garcia-Vidal, C. An update on legionella. Curr. Opin. Infect. Dis. 2010, 23, 152–157. [Google Scholar] [CrossRef] [PubMed]
- Fields, B.S.; Benson, R.F.; Besser, R.E. Legionella and Legionnaires’ disease: 25 years of investigation. Clin. Microbiol. Rev. 2002, 15, 506–526. [Google Scholar] [CrossRef] [PubMed]
- Declerck, P. Biofilms: The environmental playground of legionella pneumophila. Environ. Microbiol. 2010, 12, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Sturgill-Koszycki, S.; Swanson, M.S. Legionella pneumophila replication vacuoles mature into acidic, endocytic organelles. J. Exp. Med. 2000, 192, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Isaac, D.T.; Isberg, R. Master Manipulators: An update on legionella pneumophila icm/Dot translocated substrates and their host targets. Future Microbiol. 2014, 9, 343–359. [Google Scholar] [CrossRef] [PubMed]
- Vandal, O.H.; Nathan, C.F.; Ehrt, S. Acid resistance in mycobacterium tuberculosis. J. Bacteriol. 2009, 191, 4714–4721. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.H.; Cho, Y.; Lehrer, R.I. Styelins, broad-spectrum antimicrobial peptides from the solitary tunicate, styela clava. Comp. Biochem. Physiol. B 1997, 118, 515–521. [Google Scholar] [CrossRef]
- Tasiemski, A.; Schikorski, D.; Le Marrec-Croq, F.; Pontoire-Van Camp, C.; Boidin-Wichlacz, C.; Sautiere, P.E. Hedistin: A novel antimicrobial peptide containing bromotryptophan constitutively expressed in the nk cells-like of the marine annelid, nereis diversicolor. Dev. Comp. Immunol. 2007, 31, 749–762. [Google Scholar] [CrossRef] [PubMed]
- Shinnar, A.E.; Butler, K.L.; Park, H.J. Cathelicidin family of antimicrobial peptides: Proteolytic processing and protease resistance. Bioorg. Chem. 2003, 31, 425–436. [Google Scholar] [CrossRef]
- Nguyen, B.; Le Caer, J.-P.; Mourier, G.; Thai, R.; Lamthanh, H.; Servent, D.; Benoit, E.; Molgó, J. Characterization of a novel conus bandanus conopeptide belonging to the m-superfamily containing bromotryptophan. Mar. Drugs 2014, 12, 3449–3465. [Google Scholar] [CrossRef] [PubMed]
- Buczek, O.; Bulaj, G.; Olivera, B.M. Conotoxins and the posttranslational modification of secreted gene products. Cell. Mol. Life Sci. CMLS 2005, 62, 3067–3079. [Google Scholar] [CrossRef] [PubMed]
- Gerwig, G.J.; Hocking, H.G.; Stöcklin, R.; Kamerling, J.P.; Boelens, R. Glycosylation of conotoxins. Mar. Drugs 2013, 11, 623–642. [Google Scholar] [CrossRef] [PubMed]
- Bittner, S.; Scherzer, R.; Harlev, E. The five bromotryptophans. Amino Acids 2007, 33, 19–42. [Google Scholar] [CrossRef] [PubMed]
- Hajdušek, O.; Šíma, R.; Ayllón, N.; Jalovecká, M.; Perner, J.; de la Fuente, J.; Kopáček, P. Interaction of the tick immune system with transmitted pathogens. Front. Cell. Infect. Microbiol. 2013, 3, 26. [Google Scholar] [CrossRef] [PubMed]
- Nyirjesy, P.; Sobel, J.D. Genital mycotic infections in patients with diabetes. Postgrad. Med. 2013, 125, 33–46. [Google Scholar] [CrossRef] [PubMed]
- Fogaça, A.C.; Lorenzini, D.M.; Kaku, L.M.; Esteves, E.; Bulet, P.; Daffre, S. Cysteine-rich antimicrobial peptides of the cattle tick boophilus microplus: Isolation, structural characterization and tissue expression profile. Dev. Comp. Immunol. 2004, 28, 191–200. [Google Scholar] [CrossRef] [PubMed]
- Esteves, E.; Fogaca, A.C.; Maldonado, R.; Silva, F.D.; Manso, P.P.; Pelajo-Machado, M.; Valle, D.; Daffre, S. Antimicrobial activity in the tick rhipicephalus (boophilus) microplus eggs: Cellular localization and temporal expression of microplusin during oogenesis and embryogenesis. Dev. Comp. Immunol. 2009, 33, 913–919. [Google Scholar] [CrossRef] [PubMed]
- Joazeiro, A.C.; Coutinho, M.L.; Martins, J.R.; Masuda, A.; Seixas, A.; Vaz, I.D. Antimicrobial peptides in rhipicephalus (boophilus) microplus. Acta Sci. Vet. 2012, 40, 14. [Google Scholar]
- Silva, F.D.; Rezende, C.A.; Rossi, D.C.; Esteves, E.; Dyszy, F.H.; Schreier, S.; Gueiros-Filho, F.; Campos, C.B.; Pires, J.R.; Daffre, S. Structure and mode of action of microplusin, a copper ii-chelating antimicrobial peptide from the cattle tick rhipicephalus (boophilus) microplus. J. Biol. Chem. 2009, 284, 34735–34746. [Google Scholar] [CrossRef] [PubMed]
- Silva, F.D.; Rossi, D.C.P.; Martinez, L.R.; Frases, S.; Fonseca, F.L.; Campos, C.B.L.; Rodrigues, M.L.; Nosanchuk, J.D.; Daffre, S. Effects of microplusin, a copper-chelating antimicrobial peptide, against cryptococcus neoformans. FEMS Microbiol. Lett. 2011, 324, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Leippe, M.; Herbst, R. Ancient weapons for attack and defense: The pore-forming polypeptides of pathogenic enteric and free-living amoeboid protozoa. J. Eukaryot. Microbiol. 2004, 51, 516–521. [Google Scholar] [CrossRef] [PubMed]
- Bogaerts, A.; Beets, I.; Schoofs, L.; Verleyen, P. Antimicrobial peptides in caenorhabditis elegans. ISJ Invertebr. Surviv. J. 2010, 7, 45–52. [Google Scholar]
- Ewbank, J.J.; Zugasti, O.C. Elegans: Model host and tool for antimicrobial drug discovery. Dis. Mod. Mech. 2011, 4, 300–304. [Google Scholar] [CrossRef] [PubMed]
- Squiban, B.; Kurz, C.L.C. Elegans: An all in one model for antimicrobial drug discovery. Curr. Drug Targets 2011, 12, 967–977. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Hou, A. Host-microbe interactions in caenorhabditis elegans. ISRN Microbiol. 2013, 2013, 356451–356451. [Google Scholar] [CrossRef] [PubMed]
- Tarr, D.E.K. Distribution and characteristics of abfs, cecropins, nemapores, and lysozymes in nematodes. Dev. Comp. Immunol. 2012, 36, 502–520. [Google Scholar] [CrossRef] [PubMed]
- Tarr, D.E.K. Nematode antimicrobial peptides. ISJ Invertebr. Surviv. J. 2012, 9, 122–133. [Google Scholar]
- Reynolds, E.C.; Dashper, S.G.; O’Brien-Simpson, N.M.; Talbo, G.H.; Malkosi, M. Derived from Milk Protein Casein; for Use in Dentistry. U.S. Patent 7588752 B2, 15 September 2009. [Google Scholar]
- Kent, R.M.; Fitzgerald, G.F.; Hill, C.; Stanton, C.; Ross, R.P. Novel approaches to improve the intrinsic microbiological safety of powdered infant milk formula. Nutrients 2015, 7, 1217–1244. [Google Scholar] [CrossRef] [PubMed]
- Mankar, S.; Anoop, A.; Sen, S.; Maji, S.K. Nanomaterials: Amyloids reflect their brighter side. Nano Rev. 2011, 2. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Kim, J.H.; Lee, J.S.; Park, C.B. β-sheet-forming, self-assembled peptide nanomaterials towards optical, energy, and healthcare applications. Small 2015, 11, 3623–3640. [Google Scholar] [CrossRef] [PubMed]
- Pinkse, M.; Evaristo, G.; Pieterse, M.; Yu, Y.; Verhaert, P. Ms approaches to select peptides with post-translational modifications from amphibian defense secretions prior to full sequence elucidation. EuPA Open Proteom. 2014, 5, 32–40. [Google Scholar] [CrossRef]
- Xhindoli, D.; Pacor, S.; Benincasa, M.; Scocchi, M.; Gennaro, R.; Tossi, A. The human cathelicidin LL-37—A pore-forming antibacterial peptide and host-cell modulator. Biochim. Biophys. Acta 2016, 1858, 546–566. [Google Scholar] [CrossRef] [PubMed]
- Fabisiak, A.; Murawska, N.; Fichna, J. LL-37: Cathelicidin-related antimicrobial peptide with pleiotropic activity. Pharmacol. Rep. 2016, 68, 802–808. [Google Scholar] [CrossRef] [PubMed]
- Duplantier, A.J.; van Hoek, M.L. The human cathelicidin antimicrobial peptide LL-37 as a potential treatment for polymicrobial infected wounds. Front. Immunol. 2013, 4, 143. [Google Scholar] [CrossRef] [PubMed]
- Gronberg, A.; Dieterich, C.; Mahlapuu, M. New Treatment of Chronic Ulcers. Patent WO 2015075406 A1, 28 May 2015. [Google Scholar]
- Gronberg, A.; Mahlapuu, M.; Stahle, M.; Whately-Smith, C.; Rollman, O. Treatment with LL-37 is safe and effective in enhancing healing of hard-to-heal venous leg ulcers: A randomized, placebo-controlled clinical trial. Wound Repair Regen. 2014, 22, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Yazdanpanah, L.; Nasiri, M.; Adarvishi, S. Literature review on the management of diabetic foot ulcer. World J. Diabetes 2015, 6, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Fumakia, M.; Ho, E.A. Nanoparticles encapsulated with LL37 and serpin a1 promotes wound healing and synergistically enhances antibacterial activity. Mol. Pharm. 2016, 13, 2318–2331. [Google Scholar] [CrossRef] [PubMed]
- Chereddy, K.K.; Her, C.H.; Comune, M.; Moia, C.; Lopes, A.; Porporato, P.E.; Vanacker, J.; Lam, M.C.; Steinstraesser, L.; Sonveaux, P.; et al. Plga nanoparticles loaded with host defense peptide LL37 promote wound healing. J. Control. Release 2014, 194, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Mishra, B.; Epand, R.F.; Epand, R.M. High-quality 3d structures shine light on antibacterial, anti-biofilm and antiviral activities of human cathelicidin LL-37 and its fragments. Biochim. Biophys. Acta Biomembr. 2014, 1838, 2160–2172. [Google Scholar] [CrossRef] [PubMed]
- Goblyos, A.; Schimmel, K.J.; Valentijn, A.R.; Fathers, L.M.; Cordfunke, R.A.; Chan, H.L.; Oostendorp, J.; Nibbering, P.H.; Drijfhout, J.W.; Hiemstra, P.S.; et al. Development of a nose cream containing the synthetic antimicrobial peptide p60.4ac for eradication of methicillin-resistant staphylococcus aureus carriage. J. Pharm. Sci. 2013, 102, 3539–3544. [Google Scholar] [CrossRef] [PubMed]
- Abad, C.L.; Pulia, M.S.; Safdar, N. Does the nose know? An update on mrsa decolonization strategies. Curr. Infect. Dis. Rep. 2013, 15, 455–464. [Google Scholar] [CrossRef] [PubMed]
- Peek, F.A.W.; Nell, M.J.; Brand, R.; Jansen-Werkhoven, T.M.; van Hoogdalem, E.J.; Frijns, J.H.M. Double-blind placebo-controlled study of the novel peptide drug p60.4ac in chronic middle ear infection. In Proceedings of the 49th Interscience Conference on Antimicrobial Agents, San Francisco, CA, USA, 2009; pp. L1–L337.
- Hall-Stoodley, L.; Hu, F.Z.; Gieseke, A.; Nistico, L.; Nguyen, D.; Hayes, J.; Forbes, M.; Greenberg, D.P.; Dice, B.; Burrows, A.; et al. Direct detection of bacterial biofilms on the middle-ear mucosa of children with chronic otitis media. JAMA 2006, 296, 202–211. [Google Scholar] [CrossRef] [PubMed]
- Haisma, E.M.; de Breij, A.; Chan, H.; van Dissel, J.T.; Drijfhout, J.W.; Hiemstra, P.S.; El Ghalbzouri, A.; Nibbering, P.H. LL-37-derived peptides eradicate multidrug-resistant staphylococcus aureus from thermally wounded human skin equivalents. Antimicrob. Agents Chemother. 2014, 58, 4411–4419. [Google Scholar] [CrossRef] [PubMed]
- Church, D.; Elsayed, S.; Reid, O.; Winston, B.; Lindsay, R. Burn wound infections. Clin. Microbiol. Rev. 2006, 19, 403–434. [Google Scholar] [CrossRef] [PubMed]
- Khurshid, Z.; Najeeb, S.; Mali, M.; Moin, S.F.; Raza, S.Q.; Zohaib, S.; Sefat, F.; Zafar, M.S. Histatin peptides: Pharmacological functions and their applications in dentistry. Saudi Pharm. J. 2016. [Google Scholar] [CrossRef]
- Liu, Z.; Ma, S.; Duan, S.; Xuliang, D.; Sun, Y.; Zhang, X.; Xu, X.; Guan, B.; Wang, C.; Hu, M.; et al. Modification of titanium substrates with chimeric peptides comprising antimicrobial and titanium-binding motifs connected by linkers to inhibit biofilm formation. ACS Appl. Mater. Interfaces 2016, 8, 5124–5136. [Google Scholar] [CrossRef] [PubMed]
- Makihira, S.; Nikawa, H.; Shuto, T.; Nishimura, M.; Mine, Y.; Tsuji, K.; Okamoto, K.; Sakai, Y.; Sakai, M.; Imari, N.; et al. Evaluation of trabecular bone formation in a canine model surrounding a dental implant fixture immobilized with an antimicrobial peptide derived from histatin. J. Mater. Sci.Mater. Med. 2011, 22, 2765–2772. [Google Scholar] [CrossRef] [PubMed]
- Kong, E.F.; Tsui, C.; Boyce, H.; Ibrahim, A.; Hoag, S.W.; Karlsson, A.J.; Meiller, T.F.; Jabra-Rizk, M.A. Development and in vivo evaluation of a novel histatin-5 bioadhesive hydrogel formulation against oral candidiasis. Antimicrob. Agents Chemother. 2016, 60, 881–889. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cuesta, C.; Sarrion-Pérez, M.-G.; Bagán, J.V. Current treatment of oral candidiasis: A literature review. J. Clin. Exp. Dent. 2014, 6, e576–e582. [Google Scholar] [CrossRef] [PubMed]
- Tati, S.; Li, R.; Puri, S.; Kumar, R.; Davidow, P.; Edgerton, M. Histatin 5-spermidine conjugates have enhanced fungicidal activity and efficacy as a topical therapeutic for oral candidiasis. Antimicrob. Agents Chemother. 2014, 58, 756–766. [Google Scholar] [CrossRef] [PubMed]
- How, K.Y.; Song, K.P.; Chan, K.G. Porphyromonas gingivalis: An overview of periodontopathic pathogen below the gum line. Front. Microbiol. 2016, 7, 14. [Google Scholar] [CrossRef] [PubMed]
- Cheng, D.J.; Oppenheim, F.G.; Helmerhorst, E.J. Antifungal Formulation and Method of Preparation. Patent WO2009005798A3, 13 August 2009. [Google Scholar]
- Pal, S.; Tak, Y.K.; Han, E.; Rangasamy, S.; Song, J.M. A multifunctional composite of an antibacterial higher-valent silver metallopharmaceutical and a potent wound healing polypeptide: A combined killing and healing approach to wound care. New J. Chem. 2014, 38, 3889–3898. [Google Scholar] [CrossRef]
- Silva, O.N.; Fensterseifer, I.C.M.; Rodrigues, E.A.; Holanda, H.H.S.; Novaes, N.R.F.; Cunha, J.P.A.; Rezende, T.M.B.; Magalhães, K.G.; Moreno, S.E.; Jerônimo, M.S.; et al. Clavanin a improves outcome of complications from different bacterial infections. Antimicrob. Agents Chemother. 2015, 59, 1620–1626. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; He, J.; Eckert, R.; Yarbrough, D.; Lux, R.; Anderson, M.; Shi, W. Design and characterization of an acid-activated antimicrobial peptide. Chem. Biol. Drug Des. 2010, 75, 127–132. [Google Scholar] [CrossRef] [PubMed]
- Yadav, K.; Prakash, S. Dental caries: A review. Asian J. Biomed. Pharm. Sci. 2016, 6, 1–7. [Google Scholar]
- Legrand, D.; Pierce, A.; Mazurier, J. Secreted lactoferrin and lactoferrin-related peptides: Insight into structure and biological functions. In Bioactive Proteins and Peptides as Functional Foods and Nutraceuticals; Wiley-Blackwell: Oxford, UK, 2010; pp. 179–202. [Google Scholar]
- Mayeur, S.; Spahis, S.; Pouliot, Y.; Levy, E. Lactoferrin, a pleiotropic protein in health and disease. Antioxid. Redox Signal. 2016, 24, 813–835. [Google Scholar] [CrossRef] [PubMed]
- Bruni, N.; Capucchio, M.T.; Biasibetti, E.; Pessione, E.; Cirrincione, S.; Giraudo, L.; Corona, A.; Dosio, F. Antimicrobial activity of lactoferrin-related peptides and applications in human and veterinary medicine. Molecules 2016, 21. [Google Scholar] [CrossRef] [PubMed]
- Brouwer, C.P.; Rahman, M.; Welling, M.M. Discovery and development of a synthetic peptide derived from lactoferrin for clinical use. Peptides 2011, 32, 1953–1963. [Google Scholar] [CrossRef] [PubMed]
- Theolier, J.; Fliss, I.; Jean, J.; Hammami, R. Milkamp: A comprehensive database of antimicrobial peptides of dairy origin. Dairy Sci. Technol. 2014, 94, 181–193. [Google Scholar] [CrossRef]
- Wakabayashi, H.; Oda, H.; Yamauchi, K.; Abe, F. Lactoferrin for prevention of common viral infections. J. Infect. Chemother. 2014, 20, 666–671. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Lima, C.F.; Rodrigues, L.R. Anticancer effects of lactoferrin: Underlying mechanisms and future trends in cancer therapy. Nutr. Rev. 2014, 72, 763–773. [Google Scholar] [CrossRef] [PubMed]
- Freire, J.M.; Gaspar, D.; Veiga, A.S.; Castanho, M. Shifting gear in antimicrobial and anticancer peptides biophysical studies: From vesicles to cells. J. Peptid. Sci. 2015, 21, 178–185. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, Y.; Li, Z.; Lan, X.; Leung, P.H.M.; Li, J.; Yang, M.; Ko, F.; Qin, L. Mechanism of anticancer effects of antimicrobial peptides. J. Fiber Bioeng. Inform. 2015, 8, 25–36. [Google Scholar] [CrossRef]
- Burns, K.E.; McCleerey, T.P.; Thévenin, D. pH-selective cytotoxicity of phlip-antimicrobial peptide conjugates. Sci. Rep. 2016, 6, 28465. [Google Scholar] [CrossRef] [PubMed]
- Boohaker, R.J.; Lee, M.W.; Vishnubhotla, P.; Perez, J.M.; Khaled, A.R. The use of therapeutic peptides to target and to kill cancer cells. Curr. Med. Chem. 2012, 19, 3794–3804. [Google Scholar] [CrossRef] [PubMed]
- Nappi, C.; Tommaselli, G.A.; Morra, I.; Massaro, M.; Formisano, C.; Di Carlo, C. Efficacy and tolerability of oral bovine lactoferrin compared to ferrous sulfate in pregnant women with iron deficiency anemia: A prospective controlled randomized study. Acta Obstet. Gynecol. Scand. 2009, 88, 1031–1035. [Google Scholar] [CrossRef] [PubMed]
- Manzoni, P.; Rinaldi, M.; Cattani, S.; Pugni, L.; Romeo, M.G.; Messner, H.; Stolfi, I.; Decembrino, L.; Laforgia, N.; Vagnarelli, F.; et al. Bovine lactoferrin supplementation for prevention of late-onset sepsis in very low-birth-weight neonates: A randomized trial. JAMA 2009, 302, 1421–1428. [Google Scholar] [CrossRef] [PubMed]
- De Bortoli, N.; Leonardi, G.; Ciancia, E.; Merlo, A.; Bellini, M.; Costa, F.; Mumolo, M.G.; Ricchiuti, A.; Cristiani, F.; Santi, S.; et al. Helicobacter pylori eradication: A randomized prospective study of triple therapy versus triple therapy plus lactoferrin and probiotics. Am. J. Gastroenterol. 2007, 102, 951–956. [Google Scholar] [CrossRef] [PubMed]
- Moreau-Marquis, S.; Coutermarsh, B.; Stanton, B.A. Combination of hypothiocyanite and lactoferrin (alx-109) enhances the ability of tobramycin and aztreonam to eliminate pseudomonas aeruginosa biofilms growing on cystic fibrosis airway epithelial cells. J. Antimicrob. Chem. 2015, 70, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Boxer, L.A. How to approach neutropenia. ASH Educ. Program. 2012, 2012, 174–182. [Google Scholar]
- Cooper, C.A.; Maga, E.A.; Murray, J.D. Production of human lactoferrin and lysozyme in the milk of transgenic dairy animals: Past, present, and future. Transgenic Res. 2015, 24, 605–614. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.S.; Li, X.; Wang, Z. Apd3: The antimicrobial peptide database as a tool for research and education. Nucleic Acids Res. 2016, 44, D1087–D1093. [Google Scholar] [CrossRef] [PubMed]
- Harris, F.; Dennison, S.R.; Phoenix, D.A. Anionic antimicrobial peptides from eukaryotic organisms and their mechanisms of action. Curr. Chem. Biol. 2011, 5, 142–153. [Google Scholar] [CrossRef]
- Rieg, S.; Seeber, S.; Steffen, H.; Humeny, A.; Kalbacher, H.; Stevanovic, S.; Kimura, A.; Garbe, C.; Schittek, B. Generation of multiple stable dermcidin-derived antimicrobial peptides in sweat of different body sites. J. Investig. Dermatol. 2006, 126, 354–365. [Google Scholar] [CrossRef] [PubMed]
- Schmid-Wendtner, M.H.; Korting, H.C. The pH of the skin surface and its impact on the barrier function. Skin Pharmacol. Physiol. 2006, 19, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Mao, Y.; Niu, S.; Xu, X.; Wang, J.; Su, Y.; Wu, Y.; Zhong, S. The effect of an adding histidine on biological activity and stability of pc-pis from pseudosciaena crocea. PLoS ONE 2013, 8. [Google Scholar] [CrossRef] [PubMed]
- Pace, C.N.; Scholtz, J.M. A helix propensity scale based on experimental studies of peptides and proteins. Biophys. J. 1998, 75, 422–427. [Google Scholar] [CrossRef]
- Embleton, N.D.; Berrington, J.E.; McGuire, W.; Stewart, C.J.; Cummings, S.P. Lactoferrin: Antimicrobial activity and therapeutic potential. Semin. Fetal Neonatal Med. 2013, 18, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Krulwich, T. Bacterial Energetics: A Treatise on Structure and Function; Elsevier Science: Amsterdam, The Netherlands, 2012. [Google Scholar]
- Krulwich, T.A.; Sachs, G.; Padan, E. Molecular aspects of bacterial pH sensing and homeostasis. Nat. Rev. Microbiol. 2011, 9, 330–343. [Google Scholar] [CrossRef] [PubMed]
- Kaneti, G.; Meir, O.; Mor, A. Controlling bacterial infections by inhibiting proton-dependent processes. Biochim. Biophys. Acta 2016, 1858, 995–1003. [Google Scholar] [CrossRef] [PubMed]
- Peters, B.M.; Shirtliff, M.E.; Jabra-Rizk, M.A. Antimicrobial peptides: Primeval molecules or future drugs? PLoS Pathog. 2010, 6, e1001067. [Google Scholar] [CrossRef] [PubMed]
- Wilmes, M.; Cammue, B.P.A.; Sahl, H.G.; Thevissen, K. Antibiotic activities of host defense peptides: More to it than lipid bilayer perturbation. Nat. Prod. Rep. 2011, 28, 1350–1358. [Google Scholar] [CrossRef] [PubMed]
- Farha, M.A.; Verschoor, C.P.; Bowdish, D.; Brown, E.D. Collapsing the proton motive force to identify synergistic combinations against staphylococcus aureus. Chem. Boil. 2013, 20, 1168–1178. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.; Lee, D.; Choi, H.; Kim, H.H.; Kim, H.; Hwang, J.S.; Lee, D.G.; Kim, J.I. Synthesis and antimicrobial activity of cysteine-free coprisin nonapeptides. Biochem. Biophys. Res. Commun. 2014, 443, 483–488. [Google Scholar] [CrossRef] [PubMed]
- Nagoba, B.S.; Suryawanshi, N.M.; Wadher, B.; Selkar, S. Acidic environment and wound healing: A review. Wounds Compend. Clin. Res. Pract. 2015, 27, 5–11. [Google Scholar]
- Walkenhorst, W.F.; Klein, J.W.; Vo, P.; Wimley, W.C. pH dependence of microbe sterilization by cationic antimicrobial peptides. Antimicrob. Agents Chemother. 2013, 57, 3312–3320. [Google Scholar] [CrossRef] [PubMed]
- Lienkamp, K.; Madkour, A.E.; Tew, G.N. Antibacterial peptidomimetics: Polymeric synthetic mimics of antimicrobial peptides. In Polymer Composites-Polyolefin Fractionation-Polymeric Peptidomimetics-Collagens; Abe, A., Kausch, H.H., Moller, M., Pasch, H., Eds.; Springer: Heidelberg, Germany, 2013; Volume 251, pp. 141–172. [Google Scholar]
- Kharidia, R.; Tu, Z.; Chen, L.; Liang, J.F. Activity and selectivity of histidine-containing lytic peptides to antibiotic-resistant bacteria. Arch. Microbiol. 2012, 194, 769–778. [Google Scholar] [CrossRef] [PubMed]
- Arnusch, C.J.; Albada, H.B.; Liskamp, R.M.J.; Shai, Y. Nanostructure determines antifungal activity of de novo designed pH dependent histidine containing ultra-short lipopeptides. Biophys. J. 2010, 98, 278A–279A. [Google Scholar] [CrossRef]
- Arnusch, C.J.; Albada, H.B.; van Vaardegem, M.; Liskamp, R.M.J.; Sahl, H.-G.; Shadkchan, Y.; Osherov, N.; Shai, Y. Trivalent ultrashort lipopeptides are potent pH dependent antifungal agents. J. Med. Chem. 2012, 55, 1296–1302. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Bennett, W.F.D.; Ding, Y.; Zhang, L.; Fan, H.Y.; Zhao, D.; Zheng, T.; Ouyang, P.-K.; Li, J.; Wu, Y.; et al. Design and characterization of a multifunctional pH-triggered peptide c8 for selective anticancer activity. Adv. Healthc. Mater. 2015, 4, 2709–2718. [Google Scholar] [CrossRef] [PubMed]
- Callahan, D.J.; Liu, W.; Li, X.; Dreher, M.R.; Hassouneh, W.; Kim, M.; Marszalek, P.; Chilkoti, A. Triple stimulus-responsive polypeptide nanoparticles that enhance intratumoral spatial distribution. Nano Lett. 2012, 12, 2165–2170. [Google Scholar] [CrossRef] [PubMed]
- Han, S.-S.; Li, Z.-Y.; Zhu, J.-Y.; Han, K.; Zeng, Z.-Y.; Hong, W.; Li, W.-X.; Jia, H.-Z.; Liu, Y.; Zhuo, R.-X.; et al. Dual-pH sensitive charge-reversal polypeptide micelles for tumor-triggered targeting uptake and nuclear drug delivery. Small 2015, 11, 2543–2554. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.-H.; Choi, C.W.; Kim, H.-W.; Kim, D.H.; Kwak, T.W.; Lee, H.M.; Kim, C.H.; Chung, C.W.; Jeong, Y.-I.; Kang, D.H. Dextran-b-poly(l-histidine) copolymer nanoparticles for pH-responsive drug delivery to tumor cells. Int. J. Nanomed. 2013, 8, 3197–3207. [Google Scholar]
- Ferrer-Miralles, N.; Luis Corchero, J.; Kumar, P.; Cedano, J.A.; Gupta, K.C.; Villaverde, A.; Vazquez, E. Biological activities of histidine-rich peptides; merging biotechnology and nanomedicine. Microb. Cell Fact. 2011, 10, 101. [Google Scholar] [CrossRef] [PubMed]
- Majdoul, S.; Seye, A.K.; Kichler, A.; Holic, N.; Galy, A.; Bechinger, B.; Fenard, D. Molecular determinants of vectofusin-1 and its derivatives for the enhancement of lentivirally mediated gene transfer into hematopoietic stem/progenitor cells. J. Biol. Chem. 2016, 291, 2161–2169. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, N.; Yano, Y.; Kawano, K.; Matsuzaki, K. A pH-dependent charge reversal peptide for cancer targeting. Eur. Biophys. J. 2016. [Google Scholar] [CrossRef] [PubMed]
- Sousa, A.M.; Pereira, M.O. Pseudomonas aeruginosa diversification during infection development in cystic fibrosis lungs—A review. Pathogens 2014, 3, 680–703. [Google Scholar] [CrossRef] [PubMed]
- Folkesson, A.; Jelsbak, L.; Yang, L.; Johansen, H.K.; Ciofu, O.; Hoiby, N.; Molin, S. Adaptation of pseudomonas aeruginosa to the cystic fibrosis airway: An evolutionary perspective. Nat. Rev. Microbiol. 2012, 10, 841–851. [Google Scholar] [CrossRef] [PubMed]
- Lashua, L.P.; Melvin, J.A.; Deslouches, B.; Pilewski, J.M.; Montelaro, R.C.; Bomberger, J.M. Engineered cationic antimicrobial peptide (eCAP) prevents pseudomonas aeruginosa biofilm growth on airway epithelial cells. J. Antimicrob. Chemother. 2016, 71, 2200–2207. [Google Scholar] [CrossRef] [PubMed]
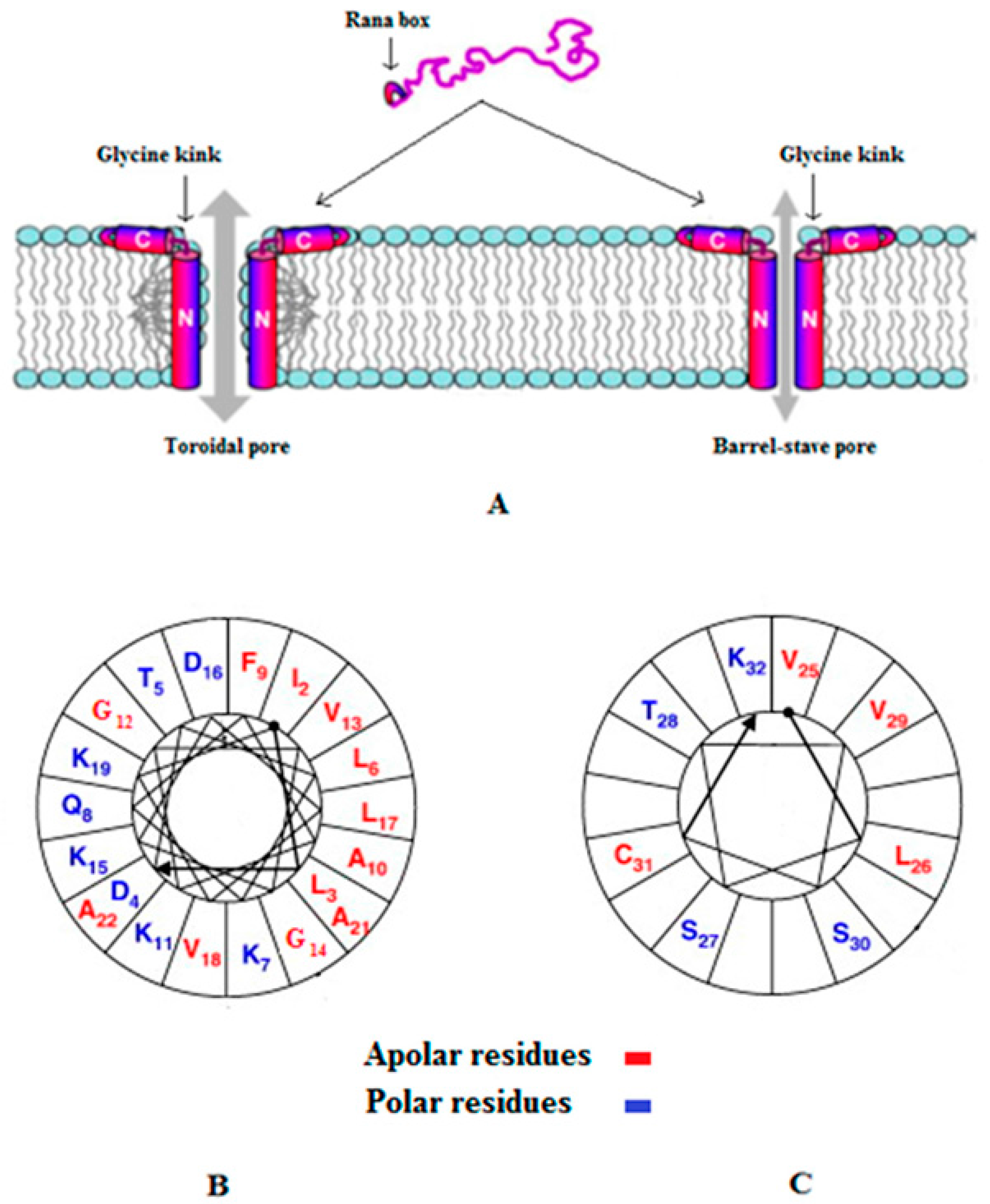
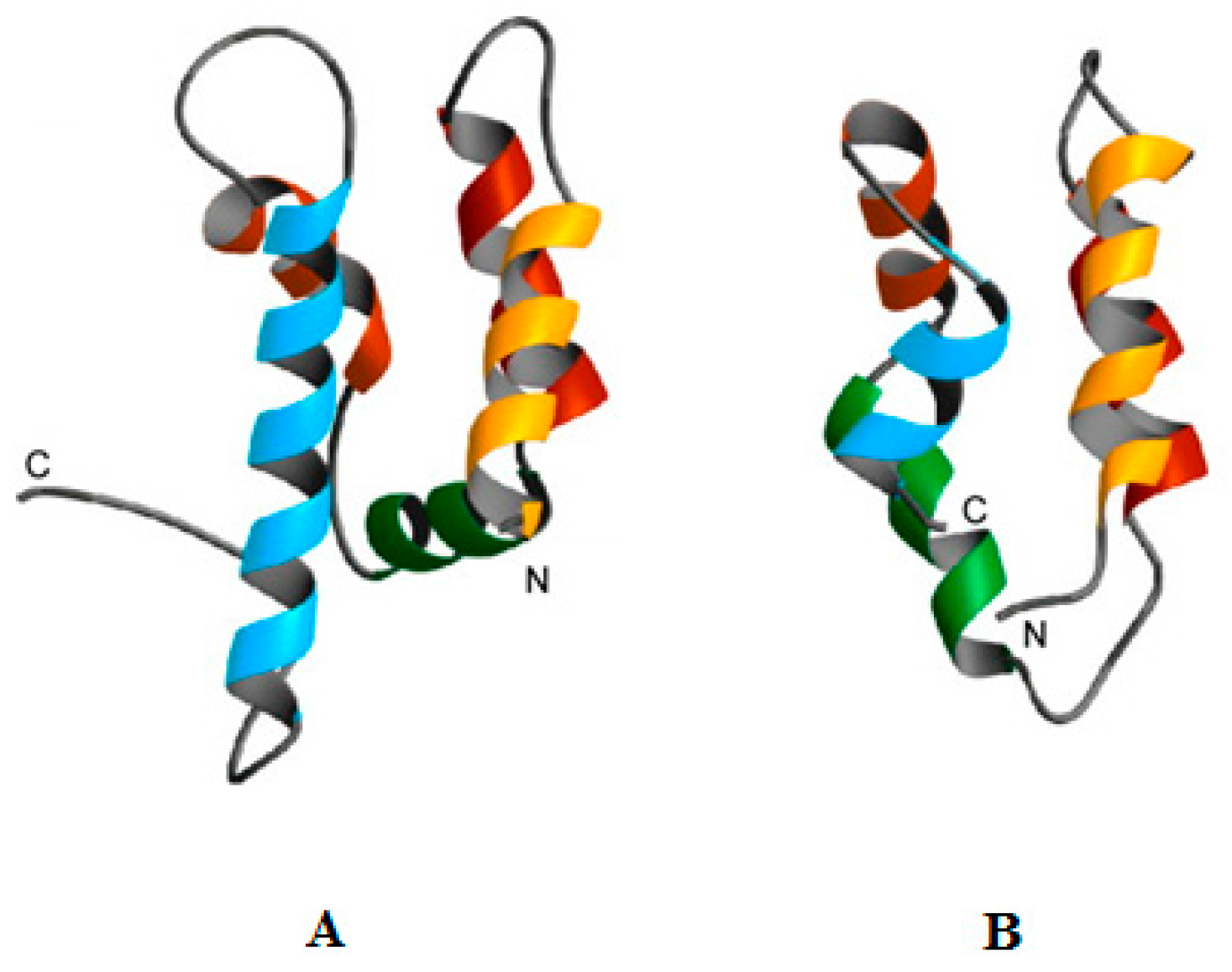
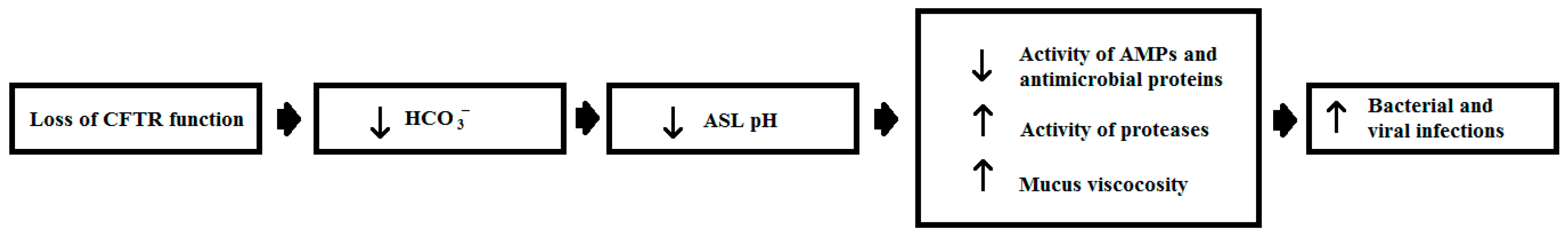
| 1. | A global public awareness campaign. |
| 2. | Improve sanitation and hygiene to prevent the spread of infection. |
| 3. | Reduce the unnecessary use of antimicrobials in agriculture and their dissemination in the environment. |
| 4. | Improve the global surveillance of drug resistance and antimicrobial consumption in humans and animals. |
| 5. | Promote new rapid diagnostics to reduce use of unnecessary antimicrobials. |
| 6. | Promote the development and use of vaccines and alternatives. |
| 7. | Improve the number, pay and recognition of people working in the field of infectious diseases. |
| 8. | A global innovation fund for early stage and non-commercial research and development. |
| 9. | Better incentives to promote investment for new drugs. |
| Antimicrobial Peptides | Indication | Phase | Company |
|---|---|---|---|
| Pexiganan (MSI-78), an analogue of magainin. | Topical cream for the treatment of diabetic foot infections and ulcers. | 3 | Dipexium Pharma /MacroChem/Genaera |
| Iseganan (IB-367), a derivative of protegrin 1. | Mouthwash for the treatment of chemotherapy induced oral mucositis. | 3 | Ardea Biosciences/national Cancer Institute. |
| Mouthwash for the treatment of ventilator-associated pneumonia. | 3 | IntraBiotics Pharmaceuticals. | |
| PAC-113 (P-113) a synthetic derivative of histatin 3 and histatin 5. | Oral gel for the treatment of candidiasis | Pacgen Biopharmaceuticals | |
| Omiganan (MBI 226, MX-226, CSL-001), an analogue of indolicidin. | Topical cream for the treatment of skin antisepsis, prevention of catheter infections/Rosacea. | 3 | Mallinckrodt/Cutanea Life Sciences, Inc. |
| Topical cream for the treatment of usual type vulvar intraepithelial neoplasia/moderate to severe inflammatory acne vulgaris/mild to moderate atopic dermatitis. | 3 | Cutanea Life Sciences, Inc. | |
| OP-145, a derivative of LL-37. | Ear drops for the treatment of chronic bacterial middle-ear infection. | 2 | OctoPlus |
| hLF1–11, a derivative of lactoferrin. | Intravenous administration for the treatment of neutropenic stem cell transplantation patients. Prevention of bacteraemia and fungal infections. | 1/2 | AM Pharma. |
| Brilacidin, (PMX-30063), a defensin mimetic. | Intravenous administration for the treatment of acute bacterial skin and skin structure Infection caused by Gram-positive bacteria, including methicillin-resistant Staphylococcus aureus (MRSA). | 3 | Cellceutix. |
| Oral rinse for the treatment of ulcerative mucositis associated with chemo/radiation therapy of cancer. | 2 | Cellceutix. | |
| Arenicins, naturally occurring AMPs. | For the treatment of infections due to MDR Gram-positive bacteria. | Preclinical | Adenium Biotech |
| Novexatin (NP213), a synthetic AMP. | Brush on treatment for fungal infections of the toenail. | 1/2 | NovaBiotics |
| C16G2, a synthetic specifically targeted AMP. | Mouthwash for the treatment of tooth decay caused by Streptococcus mutans | 2 | C3 Jian, Inc. |
| Lytixar (LTX-109), a peptidomimetic. | Topical antibiotic for the treatment of nasal carriers of MRSA. | 1/2 | Lytix Biopharma. |
| Topical cream for the treatment of infections due to Gram-positive bacteria. | 2 | Lytix Biopharma. |
| Vertebrates | AMPs | Host Organism | Key References |
|---|---|---|---|
| Fish | Gaduscidin-1 and gaduscidin-2 | Gadus morhua | [29,30] |
| Amphibians | Chensinin-1 | Rana chensinensis | [31,32] |
| Esculentin-2EM | Glandirana emeljanovi | This work | |
| Dermaseptin PD-3-7 | Pachymedusa dacnicolor | [33] | |
| Humans | Phagocytin | [24,25] | |
| Psoriasin | [34,35,36] | ||
| β-microseminoprotein | [37] | ||
| LL-37 | [38] | ||
| Hep-25 and hep-20 | [39,40,41,42,43,44] | ||
| Histatins | [45,46] | ||
| Lactoferrin | [47] | ||
| DCD-1(L) | [48,49,50] | ||
| Kappacin A and kappacin B | [51,52] | ||
| Rabbits | Phagocytin | [24,25] | |
| Platelet microbiocidal proteins | [53] | ||
| NP1 and NP2 | [54,55] | ||
| Horses | Phagocytin | [24,25] | |
| Guinea pigs | Phagocytin | [24,25] | |
| Mice | CRAMP | [38] | |
| Cattle | Lactoferricin B | [56,57]. | |
| Invertebrates | AMPs | Host Organism | Key References |
| Marine | Myticin C | Mytilus galloprovincialis | [58,59] |
| KPS-1 | Atrina pectinate | [60] | |
| Ci-PAP-A22 and Ci-MAM-A24 | Ciona intestinalis | [61,62,63,64] | |
| Clavaspirin and clavanins | Styela clava | [65,66,67,68,69,70,71,72,73,74,75,76] | |
| Styelins | Styela clava | [68,69,77] | |
| Terrestrial | Hebraein | Amblyomma hebraeum | [78] |
| Amoebapores | Entamoeba histolytica | [79,80,81,82,83] | |
| Acanthaporin | Acanthamoeba culbertsoni | [84] | |
| Caenopores | Caenorhabditis elegans | [85,86,87,88,89] |
| Lipid | pH | Lysis (%) | α-Helicity (%) |
|---|---|---|---|
| Dimyristoyl-phosphatidylserine (DMPS) | 6 | 17 | 30 |
| 8 | 63 | 49 | |
| Dimyristoyl- phosphatidylglycerol (DMPG) | 6 | 23 | 51 |
| 8 | 94 | 73 | |
| Dimyristoyl-phosphatidylcholine (DMPC) | 6 | 52 | 45 |
| 8 | 73 | 15 | |
| Dimyristoyl-phosphatidylethanolamine (DMPE) | 6 | 60 | 49 |
| 8 | 83 | 31 |
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malik, E.; Dennison, S.R.; Harris, F.; Phoenix, D.A. pH Dependent Antimicrobial Peptides and Proteins, Their Mechanisms of Action and Potential as Therapeutic Agents. Pharmaceuticals 2016, 9, 67. https://doi.org/10.3390/ph9040067
Malik E, Dennison SR, Harris F, Phoenix DA. pH Dependent Antimicrobial Peptides and Proteins, Their Mechanisms of Action and Potential as Therapeutic Agents. Pharmaceuticals. 2016; 9(4):67. https://doi.org/10.3390/ph9040067
Chicago/Turabian StyleMalik, Erum, Sarah R. Dennison, Frederick Harris, and David A. Phoenix. 2016. "pH Dependent Antimicrobial Peptides and Proteins, Their Mechanisms of Action and Potential as Therapeutic Agents" Pharmaceuticals 9, no. 4: 67. https://doi.org/10.3390/ph9040067
APA StyleMalik, E., Dennison, S. R., Harris, F., & Phoenix, D. A. (2016). pH Dependent Antimicrobial Peptides and Proteins, Their Mechanisms of Action and Potential as Therapeutic Agents. Pharmaceuticals, 9(4), 67. https://doi.org/10.3390/ph9040067





