Design and Prototype of an Automated Column-Switching HPLC System for Radiometabolite Analysis
Abstract
:1. Introduction
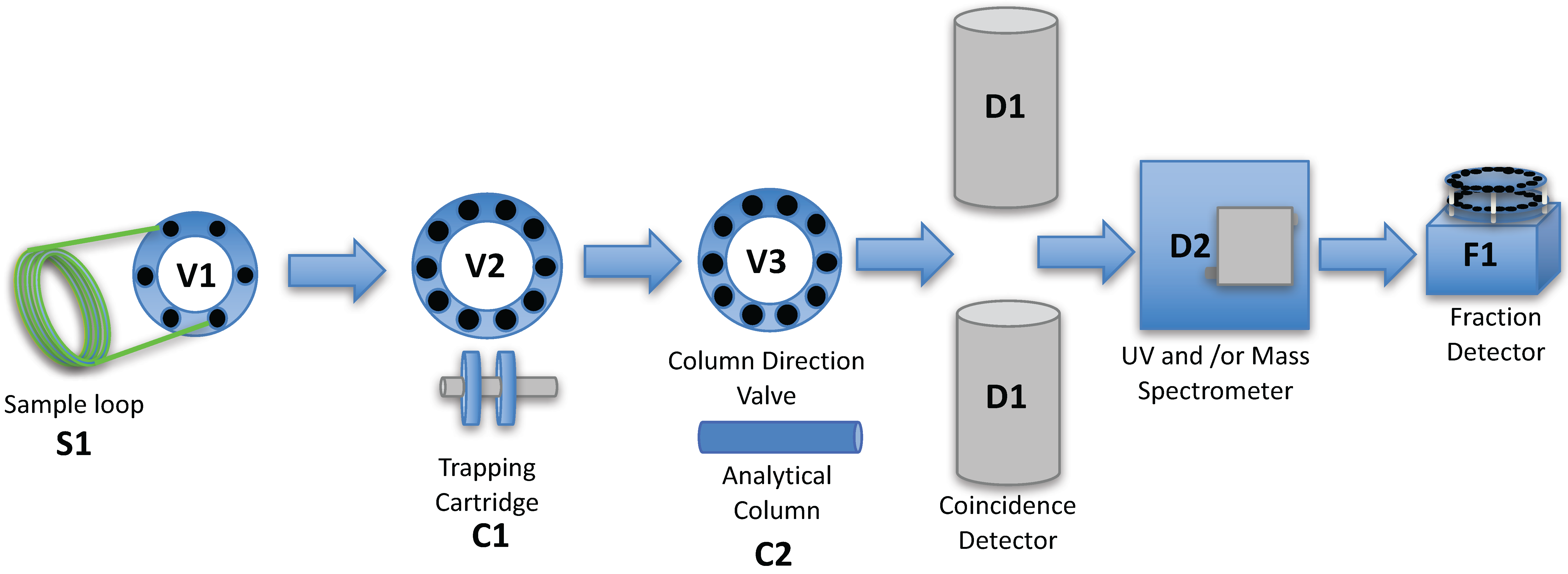
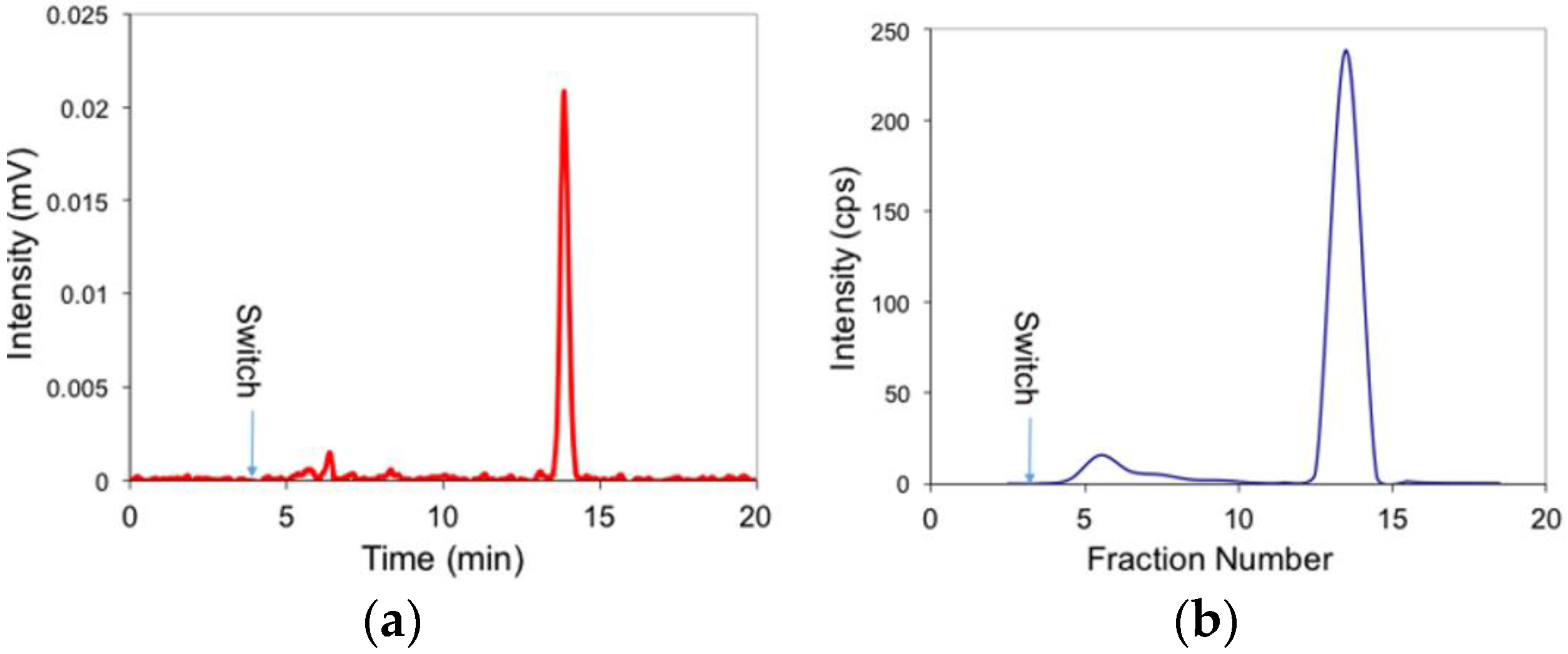
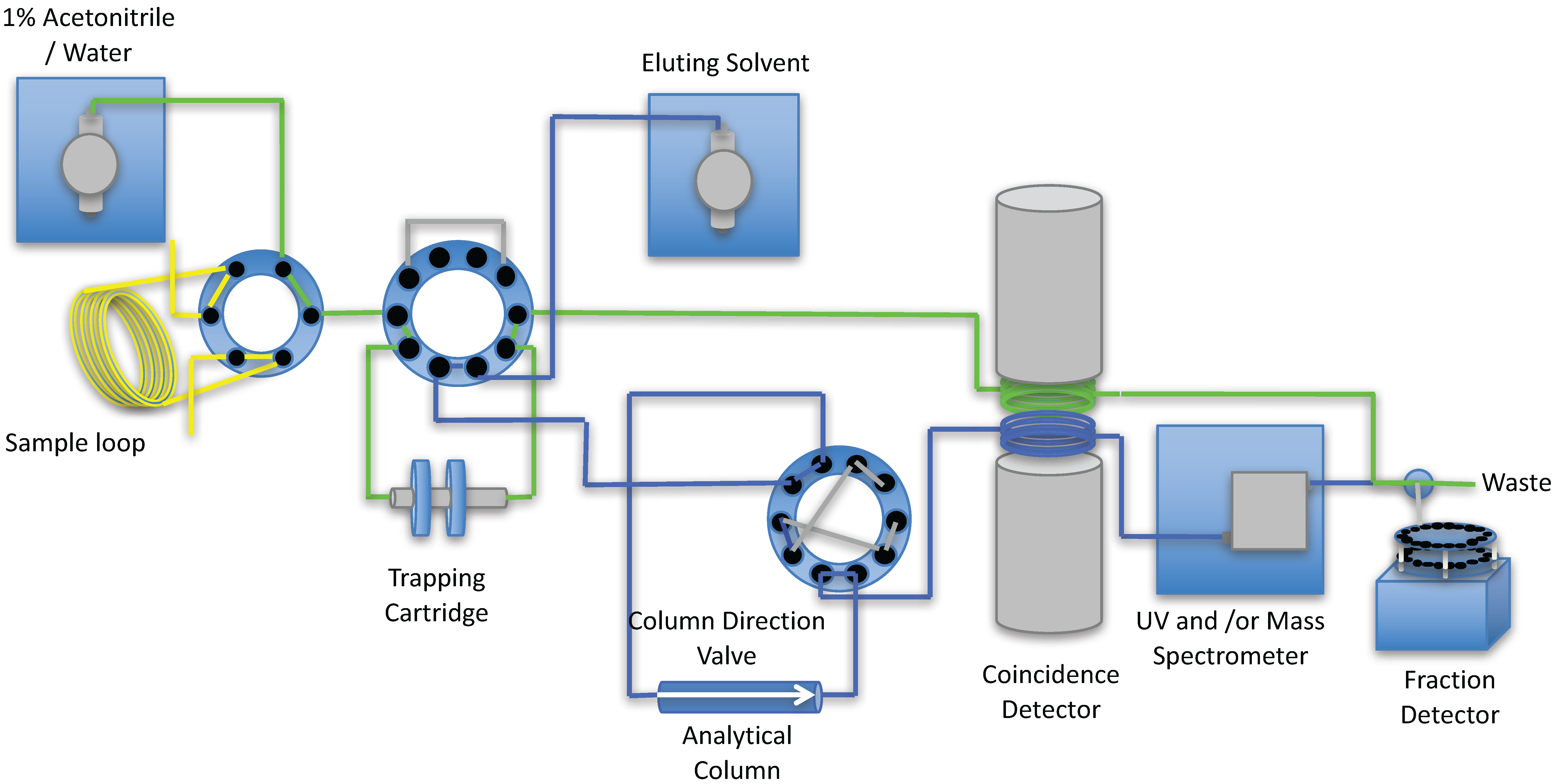
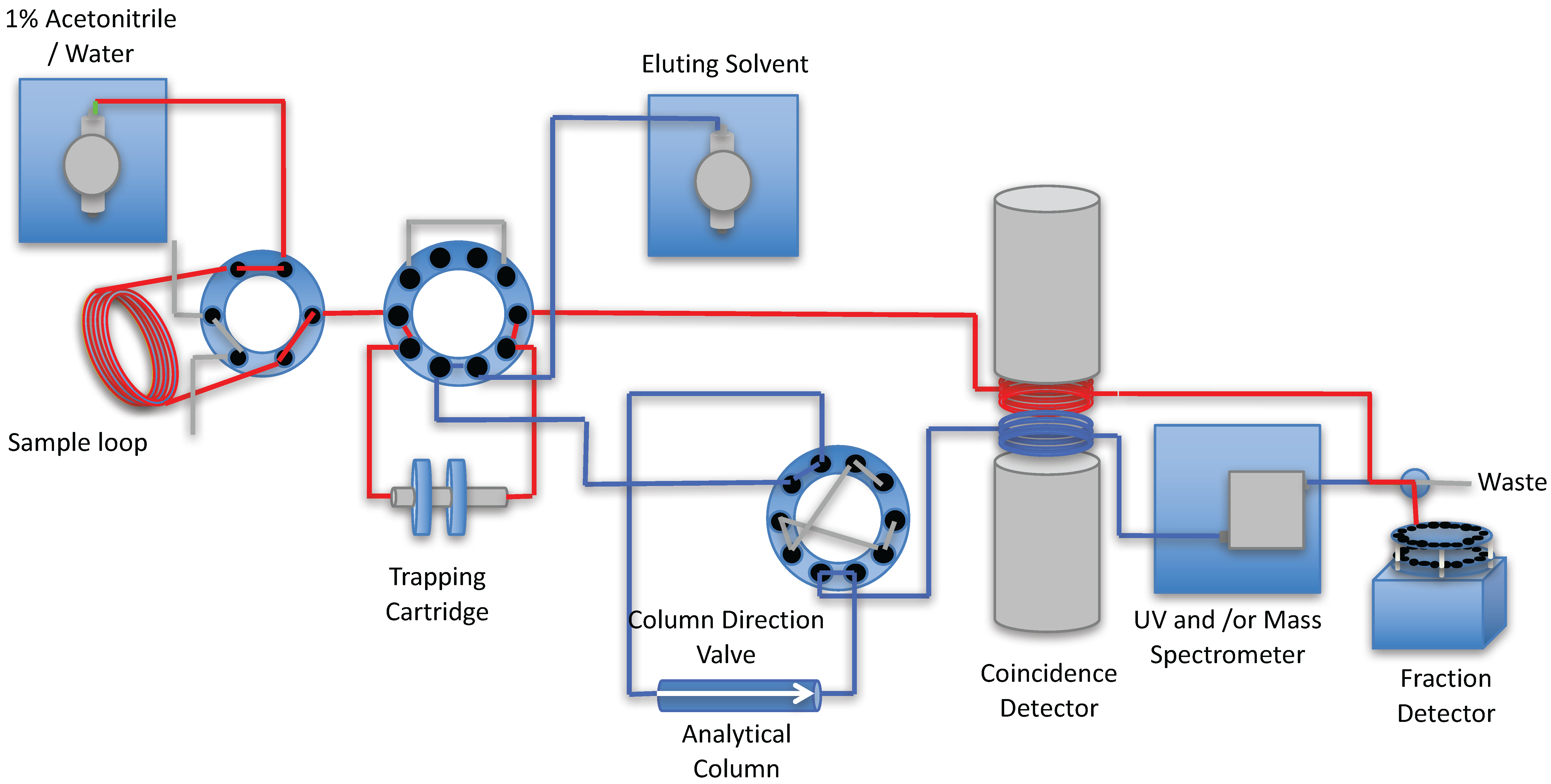
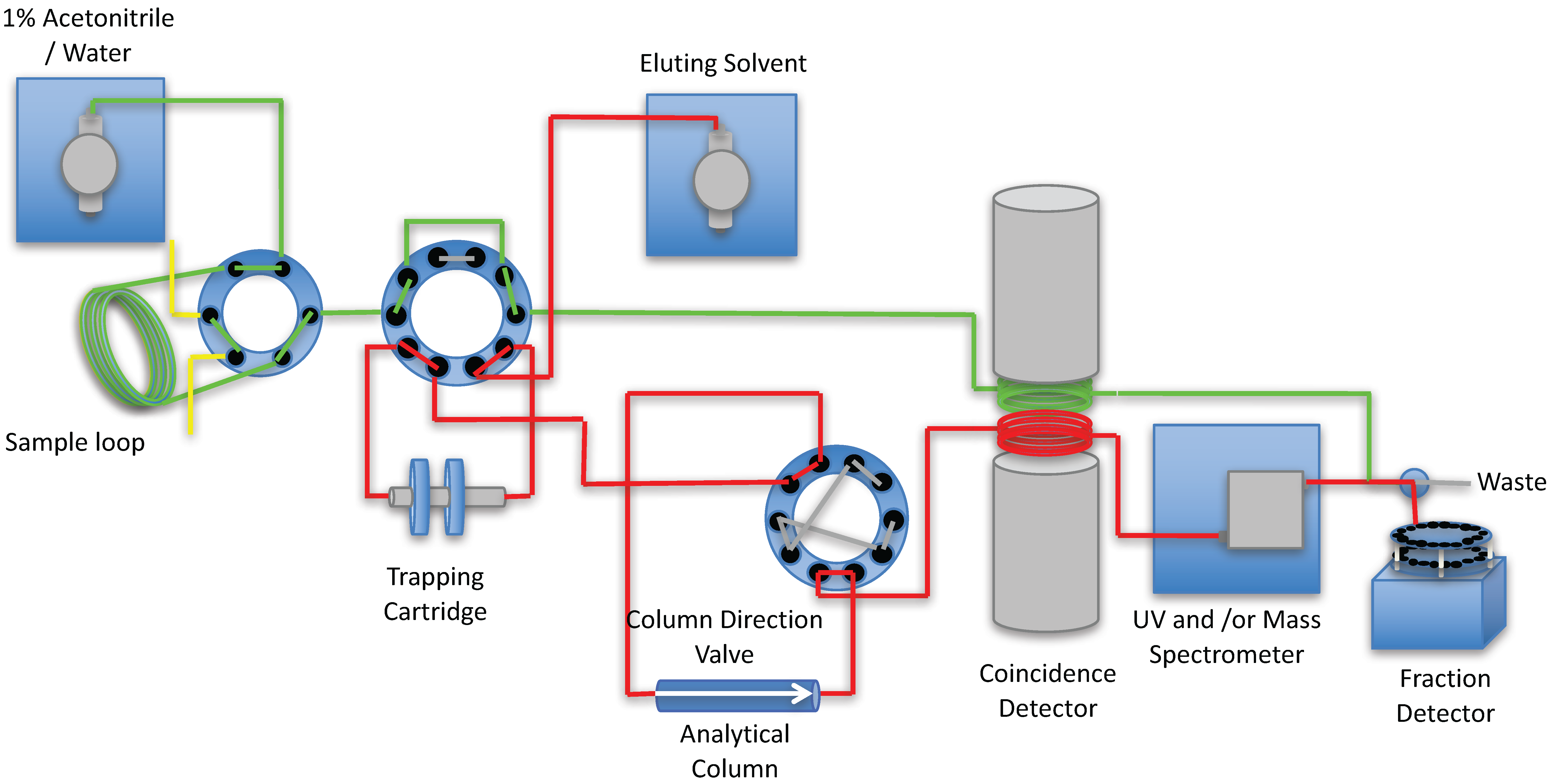
2. Results and Discussion
3. Experimental Section
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| CNS | Central nervous system |
| PET | Positron emission tomography |
| HPLC | High performance liquid chromatography |
| UV | Ultraviolet |
References
- Pike, V.W. Considerations in the Development of Reversibly Binding PET Radioligands for Brain Imaging. Curr. Med. Chem. 2016, 23, 1818–1869. [Google Scholar] [CrossRef] [PubMed]
- Pike, V.W. PET radiotracers: Crossing the blood-brain barrier and surviving metabolism. Trends Pharmacol. Sci. 2009, 30, 431–440. [Google Scholar] [CrossRef] [PubMed]
- Hilton, J.; Yokoi, F.; Dannals, R.F.; Ravert, H.T.; Szabo, Z.; Wong, D.F. Column-switching HPLC for the analysis of plasma in PET imaging studies. Nucl. Med. Biol. 2000, 27, 627–630. [Google Scholar] [CrossRef]
- Wilson, A.A.; Garcia, A.; Parkes, J.; McCormick, P.; Stephenson, K.A.; Houle, S.; Vasdev, N. Radiosynthesis and initial evaluation of [18F]-FEPPA for PET imaging of peripheral benzodiazepine receptors. Nucl. Med. Biol. 2008, 35, 305–314. [Google Scholar] [CrossRef] [PubMed]
- Optimized, highly efficient membrane for one-step plasma separation from whole blood without the use of centrifugation. Available online: http://www.pall.com/pdfs/OEM-Materials-and-Devices/09.2730_VividPlasma_DS_6pg.pdf (accessed on 25 July 2016).
- Vasdev, N.; Collier, T.L. Automated analysis systems. U.S. Patent 20,160,061,787, 3 March 2016. [Google Scholar]

© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vasdev, N.; Collier, T.L. Design and Prototype of an Automated Column-Switching HPLC System for Radiometabolite Analysis. Pharmaceuticals 2016, 9, 51. https://doi.org/10.3390/ph9030051
Vasdev N, Collier TL. Design and Prototype of an Automated Column-Switching HPLC System for Radiometabolite Analysis. Pharmaceuticals. 2016; 9(3):51. https://doi.org/10.3390/ph9030051
Chicago/Turabian StyleVasdev, Neil, and Thomas Lee Collier. 2016. "Design and Prototype of an Automated Column-Switching HPLC System for Radiometabolite Analysis" Pharmaceuticals 9, no. 3: 51. https://doi.org/10.3390/ph9030051
APA StyleVasdev, N., & Collier, T. L. (2016). Design and Prototype of an Automated Column-Switching HPLC System for Radiometabolite Analysis. Pharmaceuticals, 9(3), 51. https://doi.org/10.3390/ph9030051





