Pre-Clinical Assessment of 177Lu-Labeled Trastuzumab Targeting HER2 for Treatment and Management of Cancer Patients with Disseminated Intraperitoneal Disease
Abstract
:1. Introduction
2. Experimental
2.1. mAb Conjugation and Radiolabeling
2.2. Cell Lines
2.3. In Vitro Studies
Radioimmunoassays
2.4. In Vivo Studies
2.4.1. Biodistributions
2.4.2. Therapy Studies
2.4.3. Statistical Analyses
2.4.4. γ-Scintigraphy
3. Results and Discussion
3.1. In Vitro Analysis of 177Lu-Trastuzumab
3.2. Direct Quantitation of 177Lu-Trastuzumab Tumor Targeting and Normal Organ Distribution
| Time (h) | ||||||
|---|---|---|---|---|---|---|
| RIC | Tissue | 24 | 48 | 72 | 96 | 168 |
| Trastuzumab | Blood | 12.68 ± 3.08 | 13.72 ± 0.79 | 9.74 ± 2.67 | 10.06 ± 2.75 | 3.85 ± 1.50 |
| Tumor | 16.24 ± 4.18 | 19.08 ± 3.23 | 21.75 ± 11.66 | 24.70 ± 10.29 | 19.59 ± 9.84 | |
| Liver | 7.80 ± 2.52 | 5.72 ± 1.57 | 6.39 ± 1.40 | 6.70 ± 1.71 | 5.18 ± 2.87 | |
| Spleen | 5.63 ± 1.45 | 3.75 ± 2.33 | 6.38 ± 2.64 | 5.69 ± 2.12 | 4.11 ± 2.12 | |
| Kidney | 5.33 ± 0.46 | 3.87 ± 2.38 | 4.47 ± 1.48 | 4.24 ± 0.62 | 2.54 ± 0.48 | |
| Lung | 6.51 ± 1.66 | 6.57 ± 0.34 | 5.42 ± 1.28 | 3.98 ± 1.75 | 2.10 ± 0.81 | |
| Heart | 5.06 ± 1.50 | 4.87 ± 0.53 | 3.49 ± 1.05 | 2.83 ± 0.81 | 1.33 ± 0.49 | |
| Femur | 1.67 ± 0.61 | 1.81 ± 0.44 | 1.58 ± 0.28 | 1.55 ± 0.49 | 1.05 ± 0.36 | |
| HuIgG | Blood | 15.99 ± 1.99 | 14.81 ± 1.14 | 14.68 ± 2.59 | 11.20 ± 2.02 | 11.30 ± 1.59 |
| Tumor | 7.51 ± 1.98 | 7.41 ± 0.73 | 5.70 ± 0.96 | 5.06 ± 1.89 | 7.68 ± 2.16 | |
| Liver | 6.33 ± 1.61 | 6.21 ± 0.77 | 7.35 ± 0.74 | 6.14 ± 2.43 | 5.32 ± 0.82 | |
| Spleen | 5.63 ± 0.86 | 5.74 ± 1.39 | 5.83 ± 1.08 | 9.88 ± 8.79 | 6.56 ± 1.56 | |
| Kidney | 6.00 ± 0.87 | 6.04 ± 0.21 | 5.86 ± 0.83 | 3.84 ± 1.72 | 4.75 ± 0.78 | |
| Lung | 7.59 ± 0.56 | 7.09 ± 0.22 | 6.22 ± 2.19 | 4.88 ± 1.23 | 5.58 ± 1.24 | |
| Heart | 6.40 ± 1.09 | 5.06 ± 0.78 | 4.69 ± 0.87 | 3.73 ± 0.53 | 3.56 ± 0.58 | |
| Femur | 2.09 ± 0.16 | 1.98 ± 0.25 | 1.41 ± 0.69 | 2.19 ± 1.16 | 1.59 ± 0.35 | |
3.3. Evaluation of 177Lu-Trastuzumab for Targeting Intraperitoneal Tumor Burden
| 177Lu-Trastuzumab | 177Lu-HuIgG | |||||
|---|---|---|---|---|---|---|
| Tissue | 24 h | 72 h | 24 h | 72 h | ||
| Blood | 10.47 ± 9.19 | 10.15 ± 6.27 | 16.8 ± 2.60 | 5.13 ± 3.17 | ||
| Tumor | 13.44 ± 11.52 | 31.70 ± 16.20 | 5.84 ± 1.27 | 5.52 ± 4.92 | ||
| Liver | 4.83 ± 3.63 | 6.11 ± 3.33 | 6.76 ± 1.01 | 2.95 ± 2.06 | ||
| Spleen | 6.81 ± 5.66 | 10.63 ± 6.37 | 6.67 ± 1.17 | 4.22 ± 2.42 | ||
| Kidney | 4.46 ± 3.45 | 5.05 ± 1.69 | 7.07 ± 1.25 | 2.46 ± 1.29 | ||
| Lungs | 4.81 ± 3.84 | 4.30 ± 2.17 | 7.48 ± 1.70 | 2.28 ± 1.24 | ||
| Heart | 3.60 ± 2.97 | 3.59 ± 1.80 | 6.01 ± 1.23 | 1.70 ± 1.03 | ||
| Femur | 1.96 ± 1.58 | 2.54 ± 1.05 | 2.72 ± 0.68 | 1.19 ± 0.62 | ||
3.4. Validation of 177Lu-Trastuzumab as a Radioimmunotherapeutic and Determination of an Effective Therapeutic Dose
| Dose (μCi) | Treatment | ||||
|---|---|---|---|---|---|
| None | Trastuzumab | HuIgG | |||
| Median Survival(d) | Median Survival(d) | Therapeutic Index | Median Survival(d) | Therapeutic Index | |
| 0 | 10 | ||||
| 125 | 46 | 4.6 | 14 | 1.4 | |
| 250 | 91 | 9.1 | --- | --- | |
| 375 | 124.5 | 12.45 | 31 | 3.1 | |
| 500 | 69.5 | 6.95 | --- | --- | |
| 750 | 17 | 1.7 | 16 | 1.6 | |
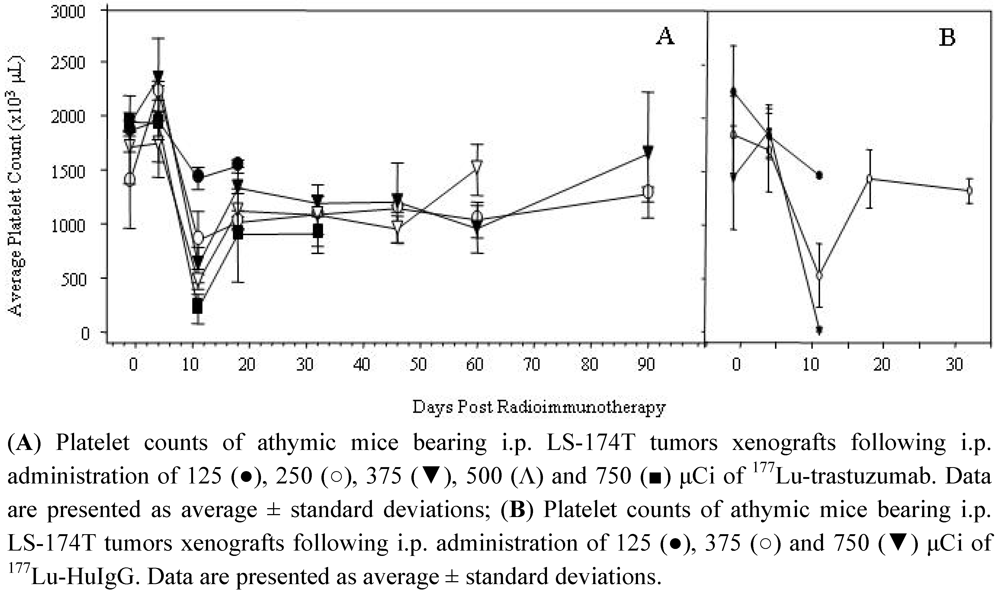
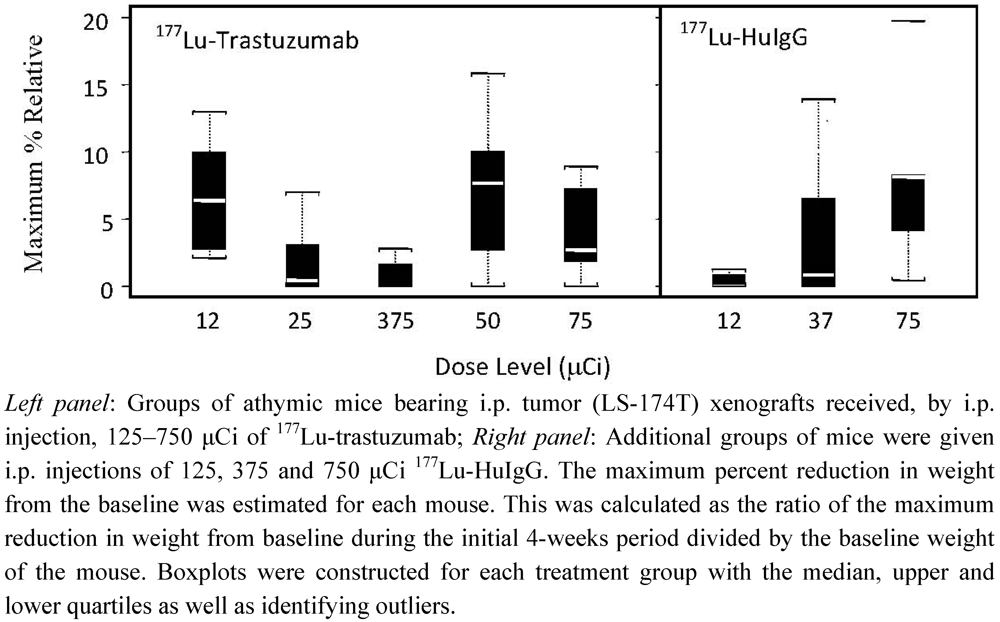
3.5. Validation of Tumor Targeting with 177Lu-Trastuzumab by Planar γ-Scintigraphy
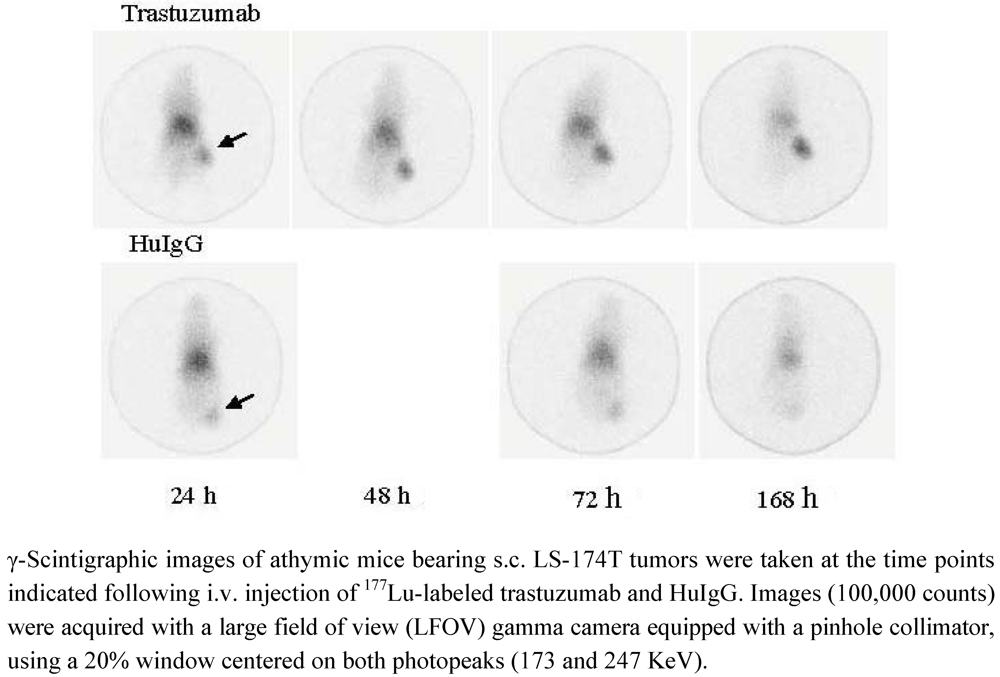
4. Conclusions
Conflict of Interest
Acknowledgements
References
- Agus, D.B.; Bunn, P.A., Jr.; Franklin, W.; Garcia, M.; Ozols, R.F. HER-2/neu as a therapeutic target in non-small cell lung cancer, prostate cancer, and ovarian canc. Semin. Oncol. 2000, 27, 53–63. [Google Scholar]
- Natali, P.G.; Nicotra, M.R.; Bigotti, A.; Venturo, I.; Slamon, D.J.; Fendly, B.M.; Ullrich, A. Expression of the p185 encoded by HER2 oncogene in normal and transformed human tissues. Int. J. Cancer 1990, 45, 457–461. [Google Scholar]
- Ross, J.S.; McKenna, B.J. The HER-2/neu oncogene in tumors of the gastrointestinal tract. Cancer Invest. 2001, 19, 554–568. [Google Scholar]
- Metro, G.; Mottolese, M.; Fabi, A. HER-2-positive metastatic breast cancer: Trastuzumab and beyond. Expert Opin. Pharmacother. 2008, 9, 2583–2601. [Google Scholar]
- Bookman, M.A.; Darcy, K.M.; Clarke-Pearson, D.; Boothby, R.A.; Horowitz, I.R. Evaluation of monoclonal humanized anti-HER2 antibody, trastuzumab, in patients with recurrent or refractory ovarian or primary peritoneal carcinoma with overexpression of HER2: A phase II trial of the gynecologic oncology group. J. Clin. Oncol. 2003, 21, 283–290. [Google Scholar] [CrossRef]
- Hofmann, M.; Stoss, O.; Shi, D.; Buttner, R.; van de Vijver, M.; Kim, W.; Ochiai, A.; Ruschoff, J.; Henkel, T. Assessment of a HER2 scoring system for gastric cancer: Results from a validation study. Histopathology 2008, 52, 797–805. [Google Scholar]
- Brady, E.D.; Milenic, D.E.; Brechbiel, M.W. Antibody guided precision radiation therapy. Discov. Med. 2004, 4, 213–219. [Google Scholar]
- Milenic, D.; Garmestani, K.; Brady, E.D.; Albert, P.S.; Ma, D.; Abdulla, A.; Brechbiel, M.W. Targeting of HER2 antigen for the treatment of disseminated peritoneal disease. Clin. Cancer Res. 2004, 23, 7834–7841. [Google Scholar]
- Rasaneh, S.; Rajabi, H.; Babaei, M.H.; Daha, F.J. 177Lu labeling of herceptin and preclinical validation as a new radiopharmaceutical for radioimmunotherapy of breast cancer. Nucl. Med. Biol. 2010, 37, 949–955. [Google Scholar]
- Rasaneh, S.; Rajabi, H.; Babaei, M.H.; Daha, F.J.; Salouti, M. Radiolabeling of trastuzumab with 177Lu via DOTA, a new radiopharmaceutical for radioimmunotherapy of breast cancer. Nucl. Med. Biol. 2009, 36, 363–369. [Google Scholar]
- Milenic, D.E.; Garmestani, K.; Brady, E.D.; Albert, P.S.; Abdulla, A.; Flynn, J.; Brechbiel, M.W. Potentiation of high-let radiation by gemcitabine: Targeting HER2 with trastuzumab to treat disseminated peritoneal disease. Clin. Cancer Res. 2007, 13, 1926–1935. [Google Scholar]
- Milenic, D.E.; Garmestani, K.; Brady, E.D.; Albert, P.S.; Ma, D.; Abdulla, A.; Brechbiel, M.W. Alpha-particle radioimmunotherapy of disseminated peritoneal disease using a 212Pb-labeled radioimmunoconjugate targeting HER2. Cancer Biother. Radiopharm. 2005, 20, 557–568. [Google Scholar]
- Milenic, D.E.; Garmestani, K.; Brady, E.D.; Baidoo, K.E.; Albert, P.S.; Wong, K.J.; Flynn, J.; Brechbiel, M.W. Multimodality therapy: Potentiation of high linear energy transfer radiation with paclitaxel for the treatment of disseminated peritoneal disease. Clin. Cancer Res. 2008, 14, 5108–5115. [Google Scholar]
- Milenic, D.E.; Brady, E.D.; Garmestani, K.; Albert, P.S.; Abdulla, A.; Brechbiel, M.W. Improved efficacy of α-particle-targeted radiation therapy: Dual targeting of human epidermal growth factor receptor-2 and tumor-associated glycoprotein 72. Cancer 2010, 116, 1059–1066. [Google Scholar]
- Palm, S.; Enmon, R.M., Jr.; Matei, C.; Kolbert, K.S.; Xu, S.; Zanzonico, P.B.; Finn, R.L.; Koutcher, J.A.; Larson, S.M.; Sgouros, G. Pharmacokinetics and biodistribution of 86Y-trastuzumab for 90Y dosimetry in an ovarian carcinoma model: Correlative MicroPET and MRI. J. Nucl. Med. 2003, 44, 1148–1155. [Google Scholar]
- Costantini, D.L.; Chan, C.; Cai, Z.; Vallis, K.A.; Reilly, R.M. 111In-labeled trastuzumab (Herceptin) modified with nuclear localization sequences (NLS): An auger electron-emitting radiotherapeutic agent for HER2/neu-amplified breast cancer. J. Nucl. Med. 2007, 48, 1357–1368. [Google Scholar]
- Chen, K.T.; Lee, T.W.; Lo, J.M. In vivo examination of 188Re(I)-tricarbonyl-labeled trastuzumab to target HER2-overexpressing breast cancer. Nucl. Med. Biol. 2009, 36, 355–361. [Google Scholar] [CrossRef]
- Schlom, J.; Siler, K.; Milenic, D.E.; Eggensperger, D.; Colcher, D.; Miller, L.S.; Houchens, D.; Cheng, R.; Kaplan, D.; Goeckeler, W. Monoclonal antibody-based therapy of a human tumor xenograft with a 177lutetium-labeled immunoconjugate. Cancer Res. 1991, 51, 2889–2896. [Google Scholar]
- Mulligan, T.; Carrasquillo, J.A.; Chung, Y.; Milenic, D.E.; Schlom, J.; Feuerstein, I.; Paik, C.; Perentesis, P.; Reynolds, J.; Curt, G.; et al. Phase I study of intravenous Lu-labeled CC49 murine monoclonal antibody in patients with advanced adenocarcinoma. Clin. Cancer Res. 1995, 1, 1447–1454. [Google Scholar]
- Meredith, R.F.; Partridge, E.E.; Alvarez, R.D.; Khazaeli, M.B.; Plott, G.; Russell, C.D.; Wheeler, R.H.; Liu, T.; Grizzle, W.E.; Schlom, J.; LoBuglio, A.F. Intraperitoneal radioimmunotherapy of ovarian cancer with lutetium-177-CC49. J. Nucl. Med. 1996, 37, 1491–1496. [Google Scholar]
- Howell, R.W.; Rao, D.V.; Sastry, K.S. Macroscopic dosimetry for radioimmunotherapy: Nonuniform activity distributions in solid tumors. Med. Phys. 1989, 16, 66–74. [Google Scholar]
- Croxtall, J.D.; McKeage, K. Trastuzumab: In HER2-positive metastatic gastric cancer. Drugs 2010, 70, 2259–2267. [Google Scholar]
- Mascelli, M.A.; Zhou, H.; Sweet, R.; Getsy, J.; Davis, H.M.; Graham, M.; Abernethy, D. Molecular, biologic, and pharmacokinetic properties of monoclonal antibodies: Impact of these parameters on early clinical development. J. Clin. Pharmacol. 2007, 47, 553–565. [Google Scholar] [CrossRef]
- Nayak, T.K.; Garmestani, K.; Milenic, D.E.; Brechbiel, M.W. Pet and MRI of metastatic peritoneal and pulmonary colorectal cancer in mice with HER1-targeted 89Zr labeled panitumumab. J. Nucl. Med. 2011. In press.. [Google Scholar]
- Milenic, D.E.; Wong, K.J.; Baidoo, K.E.; Nayak, T.K.; Regino, C.A.; Garmestani, K.; Brechbiel, M.W. Targeting HER2: A report on the in vitro and in vivo pre-clinical data supporting trastuzumab as a radioimmunoconjugate for clinical trials. MAbs 2010, 2, 550–564. [Google Scholar] [CrossRef]
- Brechbiel, M.W.; Gansow, O.A. Synthesis of C-functionalized trans-cyclohexyldiethylene-triamine-pentaacetic acids for labelling of monoclonal antibodies with the bismuth-212 α-particle emitter. J. Chem. Soc. Perkin. Trans.1 1992, 1173–1178. [Google Scholar] [CrossRef]
- Pippin, C.G.; Parker, T.A.; McMurry, T.J.; Brechbiel, M.W. Spectrophotometric method for the determination of a bifunctional DTPA ligand in DTPA-monoclonal antibody conjugates. Bioconjug. Chem. 1992, 3, 342–345. [Google Scholar]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Xu, F.; Yu, Y.; Le, X.F.; Boyer, C.; Mills, G.B.; Bast, R.C., Jr. The outcome of heregulin-induced activation of ovarian cancer cells depends on the relative levels of HER-2 and HER-3 expression. Clin. Cancer Res. 1999, 5, 3653–3660. [Google Scholar]
- Tom, B.H.; Rutzky, L.H.; Jakstys, M.H. Human colonic adenocarcinoma cells. Establishment and description of a new cell line. In Vitro 1976, 12, 180–191. [Google Scholar] [CrossRef]
- Buchsbaum, D.J.; Rogers, B.E.; Khazaeli, M.B.; Mayo, M.S.; Milenic, D.E.; Kashmiri, S.V.; Anderson, C.J.; Chappell, L.L.; Brechbiel, M.W.; Curiel, D.T. Targeting strategies for cancer radiotherapy. Clin. Cancer Res. 1999, 5, 3048s–3055s. [Google Scholar]
- Milenic, D.E.; Garmestani, K.; Chappell, L.L.; Dadachova, E.; Yordanov, A.; Ma, D.; Schlom, J.; Brechbiel, M.W. In vivo comparison of macrocyclic and acyclic ligands for radiolabeling of monoclonal antibodies with 177Lu for radioimmunotherapeutic applications. Nucl. Med. Biol. 2002, 29, 431–442. [Google Scholar] [CrossRef]
- Kelly, M.P.; Lee, S.T.; Lee, F.T.; Smyth, F.E.; Davis, I.D.; Brechbiel, M.W.; Scott, A.M. Therapeutic efficacy of 177Lu-CHX-A''-DTPA-hu3S193 radioimmunotherapy in prostate cancer is enhanced by EGFR inhibition or docetaxel chemotherapy. Prostate 2009, 69, 92–104. [Google Scholar]
- Persson, M.; Gedda, L.; Lundqvist, H.; Tolmachev, V.; Nordgren, H.; Malmstrom, P.U.; Carlsson, J. [177Lu]Pertuzumab: Experimental therapy of HER-2-expressing xenografts. Cancer Res. 2007, 67, 326–331. [Google Scholar]
- Persson, M.; Tolmachev, V.; Andersson, K.; Gedda, L.; Sandstrom, M.; Carlsson, J. [177Lu]pertuzumab: Experimental studies on targeting of HER-2 positive tumour cells. Eur. J. Nucl. Med. Mol. Imaging 2005, 32, 1457–1462. [Google Scholar] [CrossRef]
- Salouti, M.; Babaei, M.H.; Rajabi, H.; Rasaee, M.J. Preparation and biological evaluation of 177Lu conjugated PR81 for radioimmunotherapy of breast cancer. Nucl. Med Biol. 2011, 38, 849–855. [Google Scholar]
- Rasaneh, S.; Rajabi, H.; Hossein Babaei, M.; Johari Daha, F. Toxicity of trastuzumab labeled 177Lu on MCF7 and SKBr3 cell lines. Appl. Radiat. Isot. 2010, 68, 1964–1966. [Google Scholar]
- de Korte, M.A.; de Vries, E.G.; Lub-de Hooge, M.N.; Jager, P.L.; Gietema, J.A.; van der Graaf, W.T.; Sluiter, W.J.; van Veldhuisen, D.J.; Suter, T.M.; Sleijfer, D.T.; et al. 111Indium-trastuzumab visualises myocardial human epidermal growth factor receptor 2 expression shortly after anthracycline treatment but not during heart failure: A clue to uncover the mechanisms of trastuzumab-related cardiotoxicity. Eur. J. Cancer 2007, 43, 2046–2051. [Google Scholar]
- Dijkers, E.C.; Kosterink, J.G.; Rademaker, A.P.; Perk, L.R.; van Dongen, G.A.; Bart, J.; de Jong, J.R.; de Vries, E.G.; Lub-de Hooge, M.N. Development and characterization of clinical-grade 89Zr-trastuzumab for HER2/neu immunopet imaging. J. Nucl. Med. 2009, 50, 974–981. [Google Scholar]
- Tsai, S.W.; Sun, Y.; Williams, L.E.; Raubitschek, A.A.; Wu, A.M.; Shively, J.E. Biodistribution and radioimmunotherapy of human breast cancer xenografts with radiometal-labeled DOTA conjugated anti-HER2/neu antibody 4D5. Bioconjug. Chem. 2000, 11, 327–334. [Google Scholar]
- Milenic, D.E.; Brady, E.D.; Brechbiel, M.W. Antibody-targeted radiation cancer therapy. Nat. Rev. Drug Discov. 2004, 3, 488–499. [Google Scholar]
© 2012 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Ray, G.L.; Baidoo, K.E.; Keller, L.M.M.; Albert, P.S.; Brechbiel, M.W.; Milenic, D.E. Pre-Clinical Assessment of 177Lu-Labeled Trastuzumab Targeting HER2 for Treatment and Management of Cancer Patients with Disseminated Intraperitoneal Disease. Pharmaceuticals 2012, 5, 1-15. https://doi.org/10.3390/ph5010001
Ray GL, Baidoo KE, Keller LMM, Albert PS, Brechbiel MW, Milenic DE. Pre-Clinical Assessment of 177Lu-Labeled Trastuzumab Targeting HER2 for Treatment and Management of Cancer Patients with Disseminated Intraperitoneal Disease. Pharmaceuticals. 2012; 5(1):1-15. https://doi.org/10.3390/ph5010001
Chicago/Turabian StyleRay, Geoffrey L., Kwamena E. Baidoo, Lanea M. M. Keller, Paul S. Albert, Martin W. Brechbiel, and Diane E. Milenic. 2012. "Pre-Clinical Assessment of 177Lu-Labeled Trastuzumab Targeting HER2 for Treatment and Management of Cancer Patients with Disseminated Intraperitoneal Disease" Pharmaceuticals 5, no. 1: 1-15. https://doi.org/10.3390/ph5010001
APA StyleRay, G. L., Baidoo, K. E., Keller, L. M. M., Albert, P. S., Brechbiel, M. W., & Milenic, D. E. (2012). Pre-Clinical Assessment of 177Lu-Labeled Trastuzumab Targeting HER2 for Treatment and Management of Cancer Patients with Disseminated Intraperitoneal Disease. Pharmaceuticals, 5(1), 1-15. https://doi.org/10.3390/ph5010001




