Synthesis and Antimicrobial Activity of Some New Quinoxaline Derivatives
Abstract
:1. Introduction
2. Result and Discussion
2.1. Synthesis
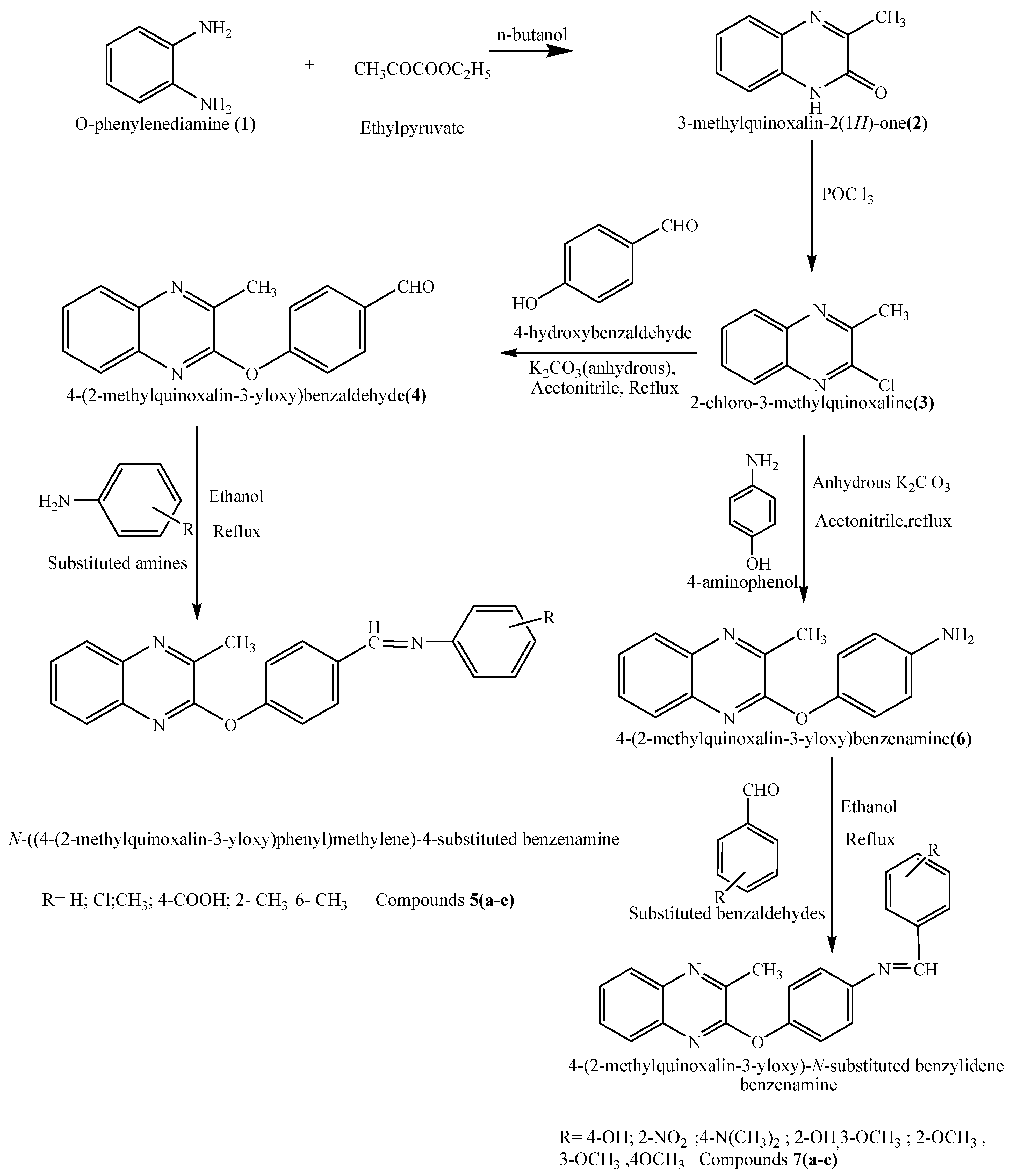
2.2. Antibacterial Activity
2.3. Antifungal Activity
| Compounds | Zone of Inhibition | |||||
|---|---|---|---|---|---|---|
| S. aureus (NCIM 2079) | B. subtilis (NCIM 2439) | E. coli (NCIM 2831) | P. aerug. (NCIM 2863) | A. niger (NCIM 618) | C. alb. (NCIM 3557) | |
| 4 | + + + | + + | + + | + + | + + | + + |
| 5a | + + | - | + + + | + + | - | - |
| 5b | + + | + + | + + | - | - | - |
| 5c | + + + | + + | + + + | - | - | + + |
| 5d | + + + | + + + | + + + | + + | - | - |
| 5e | + + + | + + | + + | + + | - | + + |
| 6 | + + | + + | + + | + + | + + | + + |
| 7a | + + + | + + + | +++ | ++ | + + | + + |
| 7b | + + | + + | + + | + + | + + | - |
| 7c | + + + | + + | + + + | + + | + + | - |
| 7d | + + | + + | - | - | + + | - |
| 7e | + + | + + | + + + | + + | + + | - |
| Ciprofloxacin | + + + | + + + | + + + | + + + | ||
| Fluconazole | + + + | + + + | ||||
3. Experimental
3.1. General
3.1.1. Synthesis of 3-methylquinoxalin-2-(1H)-one (2)
3.1.2. Synthesis of 2-chloro-3-methylquinoxaline (3)
3.1.3. Synthesis of 4-(2-methylquinoxalinyloxy) benzaldehyde (4)
3.2. General procedure for the synthesis of 2-[4-(substituted benziminomethyl) phenoxy]-3-methyl-quinoxalines 5a–e
3.2.1. N-[(4-(2-Methylquinoxalin-3-yloxy)-phenyl)-methylenebenzamine (5a)
3.2.2. [(4-(2-Methylquinoxalin-3-yloxy)-phenyl)-methylene)-2-chlorobenzamine (5b)
3.2.3. N-[{4(2-Methylquinoxalin-3-yloxy)-phenyl}-methylyne]-4-methylbenzeneamine (5c)
3.2.4. 4-((4-(2-Methylquinoxalin-3yloxy)-phenyl) - methylene-aminobenzoicacid (5d)
3.2.5. N-[(2-methylquinoxalin-3-yloxy)-phenyl)-methylene)-2, 6-dimethylbenzamine (5e)
3.3. Synthesis of 4-(2-methylquinoxalin-3-yloxy)-benzamine (6)
3.4. General procedure for the synthesis of 4-(2-methylquinoxalin-3-yloxy)-N-substituted benzylidine benzamines 7a–e
3.4.1. 4-[-{4-(2-Methylquinoxalin-3-yloxy)-phenyl}-iminomethyl]-phenol (7a)
3.4.2. 4-(2-methylquinoxalin-3-yloxy)-N-(2-nitrobenzylidene)-benzamine (7b)
3.4.3. 4-[-{4-(2-Methylquinoxalin-3-yloxy)-phenyleneimino}-methyl]-N-dimethylbenzamine (7c)
3.4.4. [{4-(2-Methylquinoxaline-3-yloxy)-phenyleneimino}-methyl]-6-methoxyphenol (7d)
3.4.5. 4-(2-Methylquinoxalin-3-yloxy)-n-(3, 4, 5-trimethoxybenzylidene)-benzamine (7e)
3.5. Antibacterial Activity
| Compounds | R | Reaction Time | Crystallization Solvents | M.P. (°C) | Mobile phase | Rf value |
|---|---|---|---|---|---|---|
| 4 | - | 30 hours | Ethanol | 116–117 | Ethyl acetate: n-Hexane (1:1) | 0.72 |
| 5a | H | 10 hours | Ethanol | 140 | Ethyl acetate: n-Hexane (1:1) | 0.80 |
| 5b | 2-Cl | 17 hours | Ethanol | 142 | Ethyl acetate: n-Hexane (1:1) | 0.78 |
| 5c | 4-CH3 | 9 hours | Ethanol | 159–160 | Ethyl acetate | 0.75 |
| 5d | 4-COOH | 16 hours | Ethanol | 221–222 | Ethyl acetate: n-Hexane (1:1) | 0.50 |
| 5e | 2-CH3,6-CH3 | 5 hours | Ethanol | 131–132 | Ethyl acetate | 0.87 |
| 6 | - | 30 hours | Ethanol | 178 | Ethyl acetate: n-Hexane (1:1) | 0.70 |
| 7a | 4-OH | 6 hours | Ethanol | 220 | Ethyl acetate: n-Hexane (1:1) | 0.60 |
| 7b | 2-NO2 | 1 hours | Ethanol | 171–172 | Ethyl acetate: n-Hexane (1:1) | 0.77 |
| 7c | 4-N(CH3)2 | 15 hours | Ethanol | 220 | Ethyl acetate: n-Hexane (1:1) | 0.70 |
| 7d | 2-OH,3-OCH3 | 1 hours | Ethanol | 190 | Ethyl acetate: n-Hexane (1:1) | 0.85 |
| 7e | 3,4,5(OCH3)3 | 21 hours | Ethanol | 135 | Ethyl acetate: n-Hexane (1:1) | 0.65 |
3.6. Antifungal Activity
Acknowledgements
References
- Badran, M.M.; Abonzid, K.A.; Hussein, M.H. Synthesis of certain substituted quinoxalines as antimicrobials agents. Part II. Arch. Pharm. Res. 2003, 26, 107–113. [Google Scholar]
- Griffith, R.K.; Chittur, S.V.; Chen, Y.C. Inhibition of glucosamine-6-Phosphate synthase from candida albicans by quinoxaline-2, 3-diones. Med. Chem. Res. 1992, 2, 467–473. [Google Scholar]
- E-lGendy, A.A.; El-Meligie, S.; El-Ansry, A.; Ahmedy, A.M. Synthesis of some quinoxaline derivatives containing Indoline-2, 3-dione or, thiazolidine residue as potential antimicrobials agents. Arch. Pharm. Res. 1995, 18, 44–47. [Google Scholar]
- Reddy-Sastry, C.V.; Shrinivas-Rao, K.; Krishanan, V.S.H.; Rastogi, K.; Jain, M.L.; Narayanan, G. Synthesis and biological activity of some new tetrazolobenzoxazines as bis-tetrazoloquinoxalines. Indian J. Chem. 1990, 29, 396–403. [Google Scholar]
- El-Hawash, S.A.; Habib, N.S.; Franki, N.H. Synthesis and antimicrobial testing of 1,2,4-triazolo[4,3-a] quinoxalines,1,2,4-triazino[4,3-a] quinoxalines and 2-pyrazolylquinoxalines. Pharmazie 1999, 54, 808–815. [Google Scholar]
- Westphal, G.; Wasiki, H.; Zielinski, U.; Weberr, F.G.; Tonew, M.; Tonew, E. Potentielle virostatica. Pharmazie 1977, 35, 570–571. [Google Scholar]
- Monge, A.; Martinez-Crespo, F.J.; Cerai, A.L.; Palop, J.A.; Narro, S.; Senador, V.; Marin, A.; Sainz, Y.; Gonzalez, M.; Hamilton, E.; Barker, A.J. Hypoxia selective agents derived from 2-quinoxalinecarbonitrile 1, 2-di-N-oxides. J. Med. Chem. 1995, 38, 4488–4495. [Google Scholar]
- Michael, J.W.; Ben-Hadda, T.; Kotchevan, A.T.; Ramdani, A.; Touzani, R.; Elkadiri, S.; Hakkou, A.; Boukka, M.; Elli, T. 2, 3-bifunctionalized quinoxalines: Synthesis, DNA Interactions and Evaluation of Anticancer, Anti-tuberculosis and Antifungal Activity. Molecules 2002, 7, 641–656. [Google Scholar]
- Rangisetty, J.B.; Gupta, C.N.; Prasad, A.L.; Srinavas, P.; Sridhar, N.; Perimoo, P.; Veeranjaneyulu, A. Synthesis of new arylaminoquinoxalines and their antimalarial activity in mice. J. Pharm. Pharmacol. 2001, 53, 1409–1413. [Google Scholar] [PubMed]
- Wagle, S.; Adhikari, A.V.; Kumari, N.S. Synthesis of some new 2-(3-methyl-7-substituted-2-oxoquinoxalinyl)-5-(aryl)-1,3,4-oxadiazoles as potential non-steroidal anti-inflammatory and analgesic agents. Ind. J. Chem. 2008, 47, 439–448. [Google Scholar]
- Hong, Y.S.; Kim, H.M.; Park, Y.T.; Kirn, H.S. Synthesis of 1-arenesulphonyl-2-quinoxalinones. Bull. Korean Chem. Soc. 2000, 21, 133. [Google Scholar]
- Ali, M.M.; Ismail, M.M.F.; Elgamy, M.S.A.; Zahran, M.A.; Ammar, Y.A. Synthesis and antimicrobial activity of some novel quinoxalinones derivatives. Molecules 2000, 5, 864–873. [Google Scholar] [CrossRef]
- Dubey, P.K.; Naidu, A.; Vayas, S.; Vineel, B.G. Facile ring opening of 2-aryl[1,2,4]oxadiazino[5,6-b]quinoxalines with sodium alkoxides. Ind. J. Chem. 2005, 44B, 573–576. [Google Scholar]
- Vayas, D.A.; Chauhan, N.A.; Parikh, A.R. Synthesis and antimicrobial activity of quinoxaline based thiazolidinones and azetidinones. Ind. J. Chem. 2007, 46, 1699–1702. [Google Scholar]
- L’Italien, J.; Banks, C.K. 2-Hydroxy-3-alkylquinoxalines. J. Am. Chem. Soc. 1951, 73, 3246. [Google Scholar]
- Leese, C.L.; Rydon, H.N. Polyazanaphthalenes. Part I. Some derivatives of 1:4:5-Triazanaphthalenes and Quinoxaline. J. Chem. Soc. 1955, 303–308. [Google Scholar]
- Platt, B.C.; Sharp, T.M. N1-sulphanilamides derived from aminoquinoxalines and aminomethylquinoxalines. J. Chem. Soc. 1948, 2129. [Google Scholar]
- Collin, C.H. Microbiological Methods; Butterworths: London, UK, 1964; p. 92. [Google Scholar]
- Gravestock, M.B.; Ryley, J.F. Antifungal Chemotherapy. Ann. Rep. Med. Chem. 1984, 19, 127. [Google Scholar]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an Open Access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Singh, D.P.; Deivedi, S.K.; Hashim, S.R.; Singhal, R.G. Synthesis and Antimicrobial Activity of Some New Quinoxaline Derivatives. Pharmaceuticals 2010, 3, 2416-2425. https://doi.org/10.3390/ph3082416
Singh DP, Deivedi SK, Hashim SR, Singhal RG. Synthesis and Antimicrobial Activity of Some New Quinoxaline Derivatives. Pharmaceuticals. 2010; 3(8):2416-2425. https://doi.org/10.3390/ph3082416
Chicago/Turabian StyleSingh, Dharmchand Prasad, Sanjay Kumar Deivedi, Syed Riaz Hashim, and Ram Gopal Singhal. 2010. "Synthesis and Antimicrobial Activity of Some New Quinoxaline Derivatives" Pharmaceuticals 3, no. 8: 2416-2425. https://doi.org/10.3390/ph3082416
APA StyleSingh, D. P., Deivedi, S. K., Hashim, S. R., & Singhal, R. G. (2010). Synthesis and Antimicrobial Activity of Some New Quinoxaline Derivatives. Pharmaceuticals, 3(8), 2416-2425. https://doi.org/10.3390/ph3082416




