Acid-Sensing Ion Channels and Pain
Abstract
:1. Introduction
2. ASICs: An Overview
2.1. Structure
2.2. Property
2.3. Distribution
3. Role of ASICs in Pain Sensation
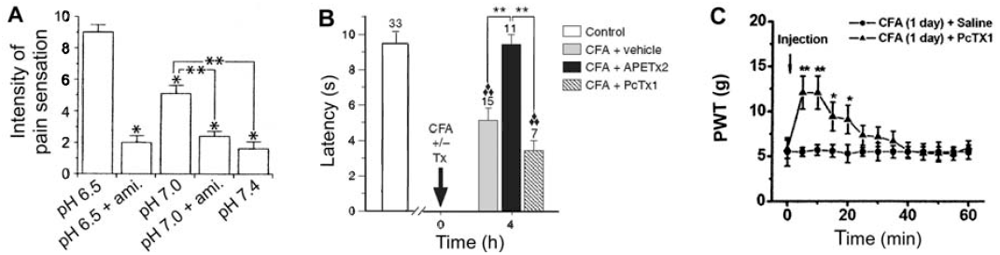
3.1. Primary inflammatory pain
3.2. Cardiac pain
3.3. GI pain and respiratory sensation
3.4. Chronic pathological pain
4. ASICs as a Potential Therapeutic Target for Pain Treatment
5. Conclusions
Acknowledgements
References
- Reeh, P.W.; Steen, K.H. Tissue acidosis in nociception and pain. Prog. Brain Res. 1996, 113, 143–151. [Google Scholar]
- Hunt, J.F.; Fang, K.; Malik, R.; Snyder, A.; Malhotra, N.; Platts-Mills, T.A.; Gaston, B. Endogenous airway acidification: Implications for asthma pathophysiology. Am. J. Respir. Crit. Care Med. 2000, 161, 694–699. [Google Scholar]
- Benson, C.J.; Sutherland, S.P. Toward an understanding of the molecules that sense myocardial ischemia. Ann. N. Y. Acad. Sci. 2001, 940, 96–109. [Google Scholar]
- Mantyh, P.W.; Clohisy, D.R.; Koltzenburg, M.; Hunt, S.P. Molecular mechanisms of cancer pain. Nat. Rev. Cancer 2002, 2, 201–209. [Google Scholar]
- Steen, K.H.; Reeh, P.W. Sustained graded pain and hyperalgesia from harmless experimental tissue acidosis in human skin. Neurosci. Lett. 1993, 154, 113–116. [Google Scholar]
- Bevan, S.; Yeats, J. Protons activate a cation conductance in a sub-population of rat dorsal root ganglion neurones. J. Physiol. 1991, 433, 145–161. [Google Scholar]
- Waldmann, R.; Lazdunski, M. H(+)-gated cation channels: neuronal acid sensors in the NaC/DEG family of ion channels. Curr. Opin. Neurobiol. 1998, 8, 418–424. [Google Scholar]
- Caterina, M.J.; Julius, D. The vanilloid receptor: a molecular gateway to the pain pathway. Annu. Rev. Neurosci. 2001, 24, 487–517. [Google Scholar]
- Kellenberger, S.; Schild, L. Epithelial sodium channel/degenerin family of ion channels: A variety of functions for a shared structure. Physiol. Rev. 2002, 82, 735–767. [Google Scholar]
- Krishtal, O. The ASICs: signaling molecules? Modulators? Trends Neurosci. 2003, 26, 477–483. [Google Scholar] [CrossRef] [PubMed]
- Wemmie, J.A.; Price, M.P.; Welsh, M.J. Acid-sensing ion channels: Advances, questions and therapeutic opportunities. Trends Neurosci. 2006, 29, 578–586. [Google Scholar]
- Akopian, A.N.; Chen, C.C.; Ding, Y.; Cesare, P.; Wood, J.N. A new member of the acid-sensing ion channel family. Neuroreport 2000, 11, 2217–2222. [Google Scholar]
- Holzer, P. Acid-sensitive ion channels and receptors. Handb. Exp. Pharmacol. 2009, 194, 283–332. [Google Scholar]
- Jasti, J.; Furukawa, H.; Gonzales, E.B.; Gouaux, E. Structure of acid-sensing ion channel 1 at 1.9 Ǻ resolution and low pH. Nature 2007, 449, 316–323. [Google Scholar] [PubMed]
- Waldmann, R. Proton-gated cation channels—Neuronal acid sensors in the central and peripheral nervous system. Adv. Exp. Med. Biol. 2001, 502, 293–304. [Google Scholar]
- Diochot, S.; Salinas, M.; Baron, A.; Escoubas, P.; Lazdunski, M. Peptides inhibitors of acid-sensing ion channels. Toxicon 2007, 49, 271–284. [Google Scholar]
- Benson, C.J.; Xie, J.; Wemmie, J.A.; Price, M.P.; Henss, J.M.; Welsh, M.J.; Snyder, P.M. Heteromultimers of DEG/ENaC subunits form H+-gated channels in mouse sensory neurons. Proc. Natl. Acad. Sci. USA 2002, 99, 2338–2343. [Google Scholar]
- Waldmann, R.; Champigny, G.; Lingueglia, E.; De Weille, J.R.; Heurteaux, C.; Lazdunski, M. H(+)-gated cation channels. Ann. N. Y. Acad. Sci. 1999, 868, 67–76. [Google Scholar]
- Alvarez de la Rosa, D.; Zhang, P.; Shao, D.; White, F.; Canessa, C.M. Functional implications of the localization and activity of acid-sensitive channels in rat peripheral nervous system. Proc. Natl. Acad. Sci. USA. 2002, 99, 2326–2331. [Google Scholar]
- Hesselager, M.; Timmermann, D.B.; Ahring, P.K. pH dependency and desensitization kinetics of heterologously expressed combinations of acid-sensing ion channel subunits. J. Biol. Chem. 2004, 279, 11006–11015. [Google Scholar]
- Waldmann, R.; Champigny, G.; Bassilana, F.; Heurteaux, C.; Lazdunski, M. A proton-gated cation channel involved in acid-sensing. Nature 1997, 386, 173–177. [Google Scholar]
- Yermolaieva, O.; Leonard, A.S.; Schnizler, M.K.; Abboud, F.M.; Welsh, M.J. Extracellular acidosis increases neuronal cell calcium by activating acid-sensing ion channel 1a. Proc. Natl. Acad. Sci. USA 2004, 101, 6752–6757. [Google Scholar]
- Dubé, G.R.; Elagoz, A.; Mangat, H. Acid sensing ion channels and acid nociception. Curr. Pharm. Des. 2009, 15, 1750–1766. [Google Scholar]
- Price, M.P.; Snyder, P.M.; Welsh, M.J. Cloning and expression of a novel human brain Na+ channel. J. Biol. Chem. 1996, 271, 7879–7882. [Google Scholar]
- Chen, C.C.; England, S.; Akopian, A.N.; Wood, J.N. A sensory neuron-specific, proton-gated ion channel. Proc. Natl. Acad. Sci. USA. 1998, 95, 10240–10245. [Google Scholar]
- Sutherland, S.P.; Benson, C.J.; Adelman, J.P.; McCleskey, E.W. Acid-sensing ion channel 3 matches the acid-gated current in cardiac ischemia-sensing neurons. Proc. Natl. Acad. Sci. USA. 2001, 98, 711–716. [Google Scholar]
- Page, A.J.; Brierley, S.M.; Martin, C.M.; Price, M.P.; Symonds, E.; Butler, R.; Wemmie, J.A.; Blackshaw, L.A. Different contributions of ASIC channels 1a, 2, and 3 in gastrointestinal mechanosensory function. Gut 2005, 54, 1408–1415. [Google Scholar] [CrossRef] [PubMed]
- Hughes, P.A.; Brierley, S.M.; Young, R.L.; Blackshaw, L.A. Localization and comparative analysis of acid-sensing ion channel (ASIC1, 2, and 3) mRNA expression in mouse colonic sensory neurons within thoracolumbar dorsal root ganglia. J. Comp. Neurol. 2007, 500, 863–875. [Google Scholar] [CrossRef] [PubMed]
- Olson, T.H.; Riedl, M.S.; Vulchanova, L.; Ortiz-Gonzalez, X.R.; Elde, R. An acid sensing ion channel (ASIC) localizes to small primary afferent neurons in rats. Neuroreport 1998, 9, 1109–1113. [Google Scholar]
- Ugawa, S.; Ueda, T.; Yamamura, H.; Shimada, S. In situ hybridization evidence for the coexistence of ASIC and TRPV1 within rat single sensory neurons. Brain Res. Mol. Brain Res. 2005, 136, 125–133. [Google Scholar] [PubMed]
- Price, M.P.; McIlwrath, S.L.; Xie, J.; Cheng, C.; Qiao, J.; Tarr, D.E.; Sluka, K.A.; Brennan, T.J.; Lewin, G.R.; Welsh, M.J. The DRASIC cation channel contributes to the detection of cutaneous touch and acid stimuli in mice. Neuron 2001, 32, 1071–1083. [Google Scholar] [CrossRef] [PubMed]
- Xie, J.; Price, M.P.; Berger, A.L.; Welsh, M.J. DRASIC contributes to pH-gated currents in large dorsal root ganglion sensory neurons by forming heteromultimeric channels. J. Neurophysiol. 2002, 87, 2835–2843. [Google Scholar]
- Wemmie, J.A.; Chen, J.; Askwith, C.C.; Hruska-Hageman, A.M.; Price, M.P.; Nolan, B.C.; Yoder, P.G.; Lamani, E.; Hoshi, T.; Freeman, J.H., Jr.; Welsh, M.J. The acid-activated ion channel ASIC contributes to synaptic plasticity, learning, and memory. Neuron 2002, 34, 463–477. [Google Scholar] [CrossRef] [PubMed]
- Alvarez de la Rosa, D.; Krueger, S.R.; Kolar, A.; Shao, D.; Fitzsimonds, R.M.; Canessa, C.M. Distribution, subcellular localization and ontogeny of ASIC1 in the mammalian central nervous system. J. Physiol. 2003, 546, 77–87. [Google Scholar]
- Jovov, B.; Tousson, A.; McMahon, L.L.; Benos, D.J. Immunolocalization of the acid-sensing ion channel 2a in the rat cerebellum. Histochem. Cell Biol. 2003, 119, 437–446. [Google Scholar]
- Askwith, C.C.; Wemmie, J.A.; Price, M.P.; Rokhlina, T.; Welsh, M.J. Acid-sensing ion channel 2 (ASIC2) modulates ASIC1 H+-activated currents in hippocampal neurons. J. Biol. Chem. 2004, 279, 18296–18305. [Google Scholar]
- Wu, L.J.; Duan, B.; Mei, Y.D.; Gao, J.; Chen, J.G.; Zhuo, M.; Xu, L.; Wu, M.; Xu, T.L. Characterization of acid-sensing ion channels in dorsal horn neurons of rat spinal cord. J. Biol. Chem. 2004, 279, 43716–43724. [Google Scholar]
- Duan, B.; Wu, L.J.; Yu, Y.Q.; Ding, Y.; Jing, L.; Xu, L.; Chen, J.; Xu, T.L. Upregulation of acid-sensing ion channel ASIC1a in spinal dorsal horn neurons contributes to inflammatory pain hypersensitivity. J. Neurosci. 2007, 27, 11139–11148. [Google Scholar]
- Xu, T.L.; Duan, B. Calcium-permeable acid-sensing ion channel in nociceptive plasticity: A new target for pain control. Prog. Neurobiol. 2009, 87, 171–180. [Google Scholar]
- Gründer, S.; Geissler, H.S.; Bässler, E.L.; Ruppersberg, J.P. New member of acid-sensing ion channels from pituitary gland. Neuroreport 2000, 11, 1607–1611. [Google Scholar]
- Brockway, L.M.; Zhou, Z.H.; Bubien, J.K.; Jovov, B.; Benos, D.J.; Keyser, K.T. Rabbit retinal neurons and glia express a variety of ENaC/DEG subunits. Am. J. Physiol. Cell Physiol. 2002, 283, C126–C134. [Google Scholar]
- Lingueglia, E. Acid-sensing ion channels in sensory perception. J. Biol. Chem. 2007, 282, 17325–17329. [Google Scholar]
- Basbaum, A.I.; Bautista, D.M.; Scherrer, G.; Julius, D. Cellular and molecular mechanisms of pain. Cell 2009, 139, 267–284. [Google Scholar]
- Costigan, M.; Scholz, J.; Woolf, C.J. Neuropathic pain: a maladaptive response of the nervous system to damage. Annu. Rev. Neurosci. 2009, 32, 1–32. [Google Scholar]
- McCleskey, E.W.; Gold, M.S. Ion channels of nociception. Annu. Rev. Physiol. 1999, 61, 835–856. [Google Scholar]
- Steen, K.H.; Issberner, U.; Reeh, P.W. Pain due to experimental acidosis in human skin: Evidence for non-adapting nociceptor excitation. Neurosci. Lett. 1995, 199, 29–32. [Google Scholar]
- Ugawa, S.; Ueda, T.; Ishida, Y.; Nishigaki, M.; Shibata, Y.; Shimada, S. Amiloride-blockable acid-sensing ion channels are leading acid sensors expressed in human nociceptors. J. Clin. Invest. 2002, 110, 1185–1190. [Google Scholar]
- Jones, N.G.; Slater, R.; Cadiou, H.; McNaughton, P.; McMahon, S.B. Acid-induced pain and its modulation in humans. J. Neurosci. 2004, 24, 10974–10979. [Google Scholar]
- Voilley, N.; de Weille, J.; Mamet, J.; Lazdunski, M. Nonsteroid anti-inflammatory drugs inhibit both the activity and the inflammation-induced expression of acid-sensing ion channels in nociceptors. J. Neurosci. 2001, 21, 8026–8033. [Google Scholar]
- Deval, E.; Noël, J.; Lay, N.; Alloui, A.; Diochot, S.; Friend, V.; Jodar, M.; Lazdunski, M.; Lingueglia, E. ASIC3, a sensor of acidic and primary inflammatory pain. EMBO J. 2008, 27, 3047–3055. [Google Scholar]
- Escoubas, P.; De Weille, J.R.; Lecoq, A.; Diochot, S.; Waldmann, R.; Champigny, G.; Moinier, D.; Ménez, A.; Lazdunski, M. Isolation of a tarantula toxin specific for a class of proton-gated Na+ channels. J. Biol. Chem. 2000, 275, 25116–25121. [Google Scholar]
- Mamet, J.; Baron, A.; Lazdunski, M.; Voilley, N. Proinflammatory mediators, stimulators of sensory neuron excitability via the expression of acid-sensing ion channels. J. Neurosci. 2002, 22, 10662–10670. [Google Scholar]
- Sluka, K.A.; Price, M.P.; Breese, N.M.; Stucky, C.L.; Wemmie, J.A.; Welsh, M.J. Chronic hyperalgesia induced by repeated acid injections in muscle is abolished by the loss of ASIC3, but not ASIC1. Pain 2003, 106, 229–239. [Google Scholar]
- Sluka, K.A.; Radhakrishnan, R.; Benson, C.J.; Eshcol, J.O.; Price, M.P.; Babinski, K.; Audette, K.M.; Yeomans, D.C.; Wilson, S.P. ASIC3 in muscle mediates mechanical, but not heat, hyperalgesia associated with muscle inflammation. Pain 2007, 129, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Fujii, Y.; Ozaki, N.; Taguchi, T.; Mizumura, K.; Furukawa, K.; Sugiura, Y. TRP channels and ASICs mediate mechanical hyperalgesia in models of inflammatory muscle pain and delayed onset muscle soreness. Pain 2008, 140, 292–304. [Google Scholar]
- Ikeuchi, M.; Kolker, S.J.; Burnes, L.A.; Walder, R.Y.; Sluka, K.A. Role of ASIC3 in the primary and secondary hyperalgesia produced by joint inflammation in mice. Pain 2008, 137, 662–669. [Google Scholar]
- Walder, R.Y.; Rasmussen, L.A.; Rainier, J.D.; Light, A.R.; Wemmie, J.A.; Sluka, K.A. ASIC1 and ASIC3 play different roles in the development of Hyperalgesia after inflammatory muscle injury. J. Pain 2010, 11, 210–218. [Google Scholar]
- Radhakrishnan, R.; Moore, S.A.; Sluka, K.A. Unilateral carrageenan injection into muscle or joint induces chronic bilateral hyperalgesia in rats. Pain 2003, 104, 567–577. [Google Scholar]
- Ikeuchi, M.; Kolker, S.J.; Sluka, K.A. Acid-sensing ion channel 3 expression in mouse knee joint afferents and effects of carrageenan-induced arthritis. J. Pain 2009, 10, 336–342. [Google Scholar]
- Drew, L.J.; Rohrer, D.K.; Price, M.P.; Blaver, K.E.; Cockayne, D.A.; Cesare, P.; Wood, J.N. Acid-sensing ion channels ASIC2 and ASIC3 do not contribute to mechanically activated currents in mammalian sensory neurones. J. Physiol. 2004, 556, 691–710. [Google Scholar]
- Mogil, J.S.; Breese, N.M.; Witty, M.F.; Ritchie, J.; Rainville, M.L.; Ase, A.; Abbadi, N.; Stucky, C.L.; Seguela, P. Transgenic expression of a dominant-negative ASIC3 subunit leads to increased sensitivity to mechanical and inflammatory stimuli. J. Neurosci. 2005, 25, 9893–9901. [Google Scholar]
- Staniland, A.A.; McMahon, S.B. Mice lacking acid-sensing ion channels (ASIC) 1 or 2, but not ASIC3, show increased pain behaviour in the formalin test. Eur. J. Pain 2009, 13, 554–563. [Google Scholar] [CrossRef] [PubMed]
- Sluka, K.A.; Winter, O.C.; Wemmie, J.A. Acid-sensing ion channels: A new target for pain and CNS diseases. Curr. Opin. Drug Discov. Devel. 2009, 12, 693–704. [Google Scholar]
- Cervero, F.; Laird, J.M. Understanding the signaling and transmission of visceral nociceptive events. J. Neurobiol. 2004, 61, 45–54. [Google Scholar]
- Yagi, J.; Wenk, H.N.; Naves, L.A.; McCleskey, E.W. Sustained currents through ASIC3 ion channels at the modest pH changes that occur during myocardial ischemia. Circ. Res. 2006, 99, 501–509. [Google Scholar]
- Benson, C.J.; Eckert, S.P.; McCleskey, E.W. Acid-evoked currents in cardiac sensory neurons: A possible mediator of myocardial ischemic sensation. Circ. Res. 1999, 84, 921–928. [Google Scholar]
- Naves, L.A.; McCleskey, E.W. An acid-sensing ion channel that detects ischemic pain. Braz. J. Med. Biol. Res. 2005, 38, 1561–1569. [Google Scholar]
- Immke, D.C.; McCleskey, E.W. Lactate enhances the acid-sensing Na+ channel on ischemia-sensing neurons. Nat. Neurosci. 2001, 4, 869–870. [Google Scholar]
- Immke, D.C.; McCleskey, E.W. Protons open acid-sensing ion channels by catalyzing relief of Ca2+ blockade. Neuron 2003, 37, 75–84. [Google Scholar]
- Hattori, T.; Chen, J.; Harding, A.M.; Price, M.P.; Lu, Y.; Abboud, F.M.; Benson, C.J. ASIC2a and ASIC3 heteromultimerize to form pH-sensitive channels in mouse cardiac dorsal root ganglia neurons. Circ. Res. 2009, 105, 279–286. [Google Scholar]
- Holzer, P. Taste receptors in the gastrointestinal tract. V. Acid sensing in the gastrointestinal tract. Am. J. Physiol. Gastrointest. Liver Physiol. 2007, 292, G699–G705. [Google Scholar] [CrossRef]
- Schicho, R.; Florian, W.; Liebmann, I.; Holzer, P.; Lippe, I.T. Increased expression of TRPV1 receptor in dorsal root ganglia by acid insult of the rat gastric mucosa. Eur. J. Neurosci. 2004, 19, 1811–1818. [Google Scholar]
- Yiangou, Y.; Facer, P.; Smith, J.A.; Sangameswaran, L.; Eglen, R.; Birch, R.; Knowles, C.; Williams, N.; Anand, P. Increased acid-sensing ion channel ASIC-3 in inflamed human intestine. Eur. J. Gastroenterol. Hepatol. 2001, 13, 891–896. [Google Scholar]
- Wultsch, T.; Painsipp, E.; Shahbazian, A.; Mitrovic, M.; Edelsbrunner, M.; Lazdunski, M.; Waldmann, R.; Holzer, P. Deletion of the acid-sensing ion channel ASIC3 prevents gastritis-induced acid hyperresponsiveness of the stomach-brainstem axis. Pain 2008, 134, 245–253. [Google Scholar]
- Page, A.J.; Brierley, S.M.; Martin, C.M.; Hughes, P.A.; Blackshaw, L.A. Acid sensing ion channels 2 and 3 are required for inhibition of visceral nociceptors by benzamil. Pain 2007, 133, 150–160. [Google Scholar]
- Hobson, A.R.; Aziz, Q. Modulation of visceral nociceptive pathways. Curr. Opin. Pharmacol. 2007, 7, 593–597. [Google Scholar]
- Coleridge, H.M.; Coleridge, J.C. Pulmonary reflexes: neural mechanisms of pulmonary defense. Annu. Rev. Physiol. 1994, 56, 69–91. [Google Scholar]
- Lee, L.Y.; Pisarri, T.E. Afferent properties and reflex functions of bronchopulmonary C-fibers. Respir. Physiol. 2001, 125, 47–65. [Google Scholar]
- Fisher, J.T. The TRPV1 ion channel: implications for respiratory sensation and dyspnea. Respir. Physiol. Neurobiol. 2009, 167, 45–52. [Google Scholar]
- Lee, L.Y. Respiratory sensations evoked by activation of bronchopulmonary C-fibers. Respir. Physiol. Neurobiol. 2009, 167, 26–35. [Google Scholar]
- Ricciardolo, F.L.; Steinhoff, M.; Amadesi, S.; Guerrini, R.; Tognetto, M.; Trevisani, M.; Creminon, C.; Bertrand, C.; Bunnett, N.W.; Fabbri, L.M.; Salvadori, S.; Geppetti, P. Presence and bronchomotor activity of protease-activated receptor-2 in guinea pig airways. Am. J. Respir. Crit. Care Med. 2000, 161, 1672–1680. [Google Scholar] [PubMed]
- Kodric, M.; Shah, A.N.; Fabbri, L.M.; Confalonieri, M. An investigation of airway acidification in asthma using induced sputum: A study of feasibility and correlation. Am. J. Respir. Crit. Care Med. 2007, 175, 905–910. [Google Scholar]
- Ricciardolo, F.L.; Gaston, B.; Hunt, J. Acid stress in the pathology of asthma. J. Allergy Clin. Immunol. 2004, 113, 610–619. [Google Scholar]
- Kollarik, M.; Ru, F.; Undem, B.J. Acid-sensitive vagal sensory pathways and cough. Pulm. Pharmacol. Ther. 2007, 20, 402–411. [Google Scholar]
- Gu, Q.; Lee, L.Y. Characterization of acid-signaling in rat vagal pulmonary sensory neurons. Am. J. Physiol. Lung Cell. Mol. Physiol. 2006, 291, L58–L65. [Google Scholar]
- Gu, Q.; Lee, L.Y. Regulation of acid signaling in rat pulmonary sensory neurons by protease-activated receptor-2. Am. J. Physiol. Lung. Cell Mol. Physiol. 2010, 298, L454–L461. [Google Scholar]
- Woolf, C.J.; Salter, M.W. Neuronal plasticity: increasing the gain in pain. Science 2000, 288, 1765–1769. [Google Scholar]
- Treede, R.D.; Jensen, T.S.; Campbell, J.N.; Cruccu, G.; Dostrovsky, J.O.; Griffin, J.W.; Hansson, P.; Hughes, R.; Nurmikko, T.; Serra, J. Neuropathic pain: redefinition and a grading system for clinical and research purposes. Neurology 2008, 70, 1630–1635. [Google Scholar]
- Jensen, T.S.; Madsen, C.S.; Finnerup, N.B. Pharmacology and treatment of neuropathic pains. Curr. Opin. Neurol. 2009, 22, 467–474. [Google Scholar]
- O'Connor, A.B.; Dworkin, R.H. Treatment of neuropathic pain: an overview of recent guidelines. Am. J. Med. 2009, 122, S22–S32. [Google Scholar]
- Harvey, V.L.; Dickenson, A.H. Mechanisms of pain in nonmalignant disease. Curr. Opin. Support Palliat Care. 2008, 2, 133–139. [Google Scholar]
- Jarvis, M.F.; Boyce-Rustay, J.M. Neuropathic pain: models and mechanisms. Curr. Pharm. Des. 2009, 15, 1711–1716. [Google Scholar]
- Sommer, C. Painful neuropathies. Curr. Opin. Neurol. 2003, 16, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Schaible, H.G.; Schmelz, M.; Tegeder, I. Pathophysiology and treatment of pain in joint disease. Adv. Drug Deliv. Rev. 2006, 58, 323–342. [Google Scholar]
- Ohtori, S.; Inoue, G.; Koshi, T.; Ito, T.; Doya, H.; Saito, T.; Moriya, H.; Takahashi, K. Up-regulation of acid-sensing ion channel 3 in dorsal root ganglion neurons following application of nucleus pulposus on nerve root in rats. Spine 2006, 31, 2048–2052. [Google Scholar]
- Mazzuca, M.; Heurteaux, C.; Alloui, A.; Diochot, S.; Baron, A.; Voilley, N.; Blondeau, N.; Escoubas, P.; Gélot, A.; Cupo, A.; Zimmer, A.; Zimmer, A.M.; Eschalier, A.; Lazdunski, M. A tarantula peptide against pain via ASIC1a channels and opioid mechanisms. Nat. Neurosci. 2007, 10, 943–945. [Google Scholar]
- Xiong, Z.G.; Zhu, X.M.; Chu, X.P.; Minami, M.; Hey, J.; Wei, W.L.; MacDonald, J.F.; Wemmie, J.A.; Price, M.P.; Welsh, M.J.; Simon, R.P. Neuroprotection in ischemia: Blocking calcium-permeable acid-sensing ion channels. Cell 2004, 118, 687–698. [Google Scholar]
- Xiong, Z.G.; Chu, X.P.; Simon, R.P. Ca2+-permeable acid-sensing ion channels and ischemic brain injury. J. Membr. Biol. 2006, 209, 59–68. [Google Scholar]
- Friese, M.A.; Craner, M.J.; Etzensperger, R.; Vergo, S.; Wemmie, J.A.; Welsh, M.J.; Vincent, A.; Fugger, L. Acid-sensing ion channel-1 contributes to axonal degeneration in autoimmune inflammation of the central nervous system. Nat. Med. 2007, 13, 1483–1489. [Google Scholar]
- Askwith, C.C.; Cheng, C.; Ikuma, M.; Benson, C.; Price, M.P.; Welsh, M.J. Neuropeptide FF and FMRFamide potentiate acid-evoked currents from sensory neurons and proton-gated DEG/ENaC channels. Neuron 2000, 26, 133–141. [Google Scholar]
- Catarsi, S.; Babinski, K.; Séguéla, P. Selective modulation of heteromeric ASIC proton-gated channels by neuropeptide FF. Neuropharmacology 2001, 41, 592–600. [Google Scholar]
- Deval, E.; Baron, A.; Lingueglia, E.; Mazarguil, H.; Zajac, J.M.; Lazdunski, M. Effects of neuropeptide SF and related peptides on acid sensing ion channel 3 and sensory neuron excitability. Neuropharmacology 2003, 44, 662–671. [Google Scholar]
- Xie, J.; Price, M.P.; Wemmie, J.A.; Askwith, C.C.; Welsh, M.J. ASIC3 and ASIC1 mediate FMRFamide-related peptide enhancement of H+-gated currents in cultured dorsal root ganglion neurons. J. Neurophysiol. 2003, 89, 2459–2465. [Google Scholar]
- Lingueglia, E.; Deval, E.; Lazdunski, M. FMRFamide-gated sodium channel and ASIC channels: A new class of ionotropic receptors for FMRFamide and related peptides. Peptides 2006, 27, 1138–1152. [Google Scholar]
- Yang, H.Y.T.; Tao, T.; Iadarola, M.J. Modulatory role of neuropeptide FF system in nociception and opiate analgesia. Neuropeptides 2008, 42, 1–18. [Google Scholar]
- Allen, N.J.; Attwell, D. Modulation of ASIC channels in rat cerebellar Purkinje neurons by ischaemia-related signals. J. Physiol. 2002, 543, 521–529. [Google Scholar]
- Poirot, O.; Vukicevic, M.; Boesch, A.; Kellenberger, S. Selective regulation of acid-sensing ion channel 1 by serine proteases. J. Biol. Chem. 2004, 279, 38448–38457. [Google Scholar]
- Vukicevic, M.; Weder, G.; Boillat, A.; Boesch, A.; Kellenberger, S. Trypsin cleaves acid-sensing ion channel 1a in a domain that is critical for channel gating. J. Biol. Chem. 2006, 281, 714–722. [Google Scholar]
- Andrey, F.; Tsintsadze, T.; Volkova, T.; Lozovaya, N.; Krishtal, O. Acid sensing ionic channels: modulation by redox reagents. Biochim. Biophys. Acta 2005, 1745, 1–6. [Google Scholar]
- Chu, X.P.; Close, N.; Saugstad, J.A.; Xiong, Z.G. ASIC1a-specific modulation of acid-sensing ion channels in mouse cortical neurons by redox reagents. J. Neurosci. 2006, 26, 5329–5339. [Google Scholar]
- Cadiou, H.; Studer, M.; Jones, N.G.; Smith, E.S.; Ballard, A.; McMahon, S.B.; McNaughton, P.A. Modulation of acid-sensing ion channel activity by nitric oxide. J. Neurosci. 2007, 27, 13251–13260. [Google Scholar]
- Smith, E.S.; Cadiou, H.; McNaughton, P.A. Arachidonic acid potentiates acid-sensing ion channels in rat sensory neurons by a direct action. Neuroscience 2007, 145, 686–698. [Google Scholar]
- Xu, T.L.; Xiong, Z.G. Dynamic regulation of acid-sensing ion channels by extracellular and intracellular modulators. Curr. Med. Chem. 2007, 14, 1753–1763. [Google Scholar]
- Rocha-González, H.I.; Herrejon-Abreu, E.B.; López-Santillán, F.J.; García-López, B.E.; Murbartián, J.; Granados-Soto, V. Acid increases inflammatory pain in rats: effect of local peripheral ASICs inhibitors. Eur. J. Pharmacol. 2009, 603, 56–61. [Google Scholar]
- Voilley, N. Acid-sensing ion channels (ASICs): New targets for the analgesic effects of non-steroid anti-inflammatory drugs (NSAIDs). Curr. Drug Targets Inflamm. Allergy 2004, 3, 71–79. [Google Scholar]
- Escoubas, P.; Bernard, C.; Lambeau, G.; Lazdunski, M.; Darbon, H. Recombinant production and solution structure of PcTx1, the specific peptide inhibitor of ASIC1a proton-gated cation channels. Protein Sci. 2003, 12, 1332–1343. [Google Scholar]
- Diochot, S.; Baron, A.; Rash, L.D.; Deval, E.; Escoubas, P.; Scarzello, S.; Salinas, M.; Lazdunski, M. A new sea anemone peptide, APETx2, inhibits ASIC3, a major acid-sensitive channel in sensory neurons. EMBO J. 2004, 23, 1516–1525. [Google Scholar] [CrossRef] [PubMed]
© 2010 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Gu, Q.; Lee, L.-Y. Acid-Sensing Ion Channels and Pain. Pharmaceuticals 2010, 3, 1411-1425. https://doi.org/10.3390/ph3051411
Gu Q, Lee L-Y. Acid-Sensing Ion Channels and Pain. Pharmaceuticals. 2010; 3(5):1411-1425. https://doi.org/10.3390/ph3051411
Chicago/Turabian StyleGu, Qihai, and Lu-Yuan Lee. 2010. "Acid-Sensing Ion Channels and Pain" Pharmaceuticals 3, no. 5: 1411-1425. https://doi.org/10.3390/ph3051411
APA StyleGu, Q., & Lee, L.-Y. (2010). Acid-Sensing Ion Channels and Pain. Pharmaceuticals, 3(5), 1411-1425. https://doi.org/10.3390/ph3051411



