Herbacetin Alleviates Influenza Virus-Induced Lung Injury and Fibrosis by Targeting the Neuraminidase Protein
Abstract
1. Introduction
2. Results
2.1. IAV Infection Induces Expression of Fibrotic Proteins to Promote PF In Vivo
2.2. IAV Infection Activates the TGF-β/Smad3 Pathway Both In Vivo and In Vitro
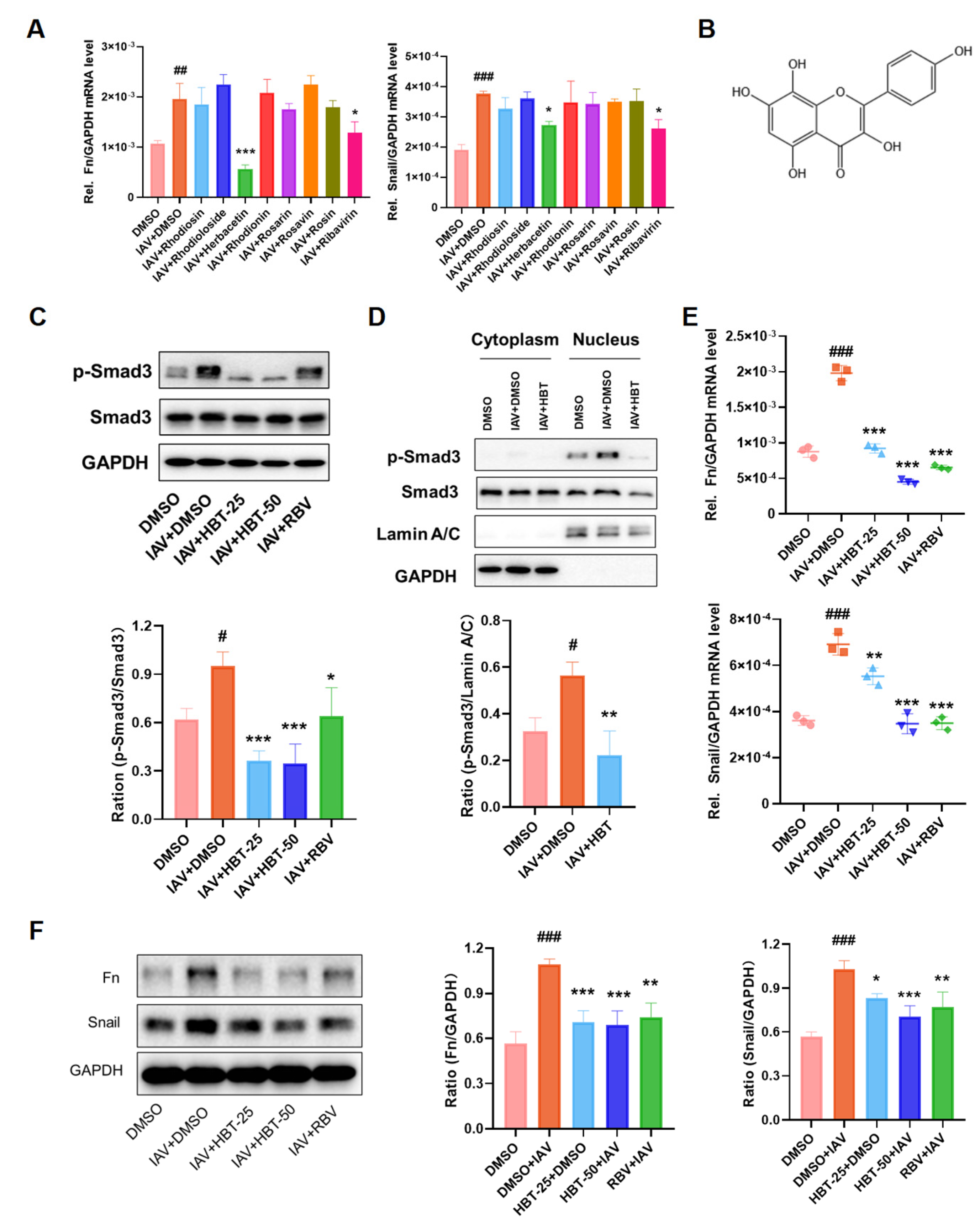
2.3. HBT Anti-Fibrosis Induced by IAV In Vitro
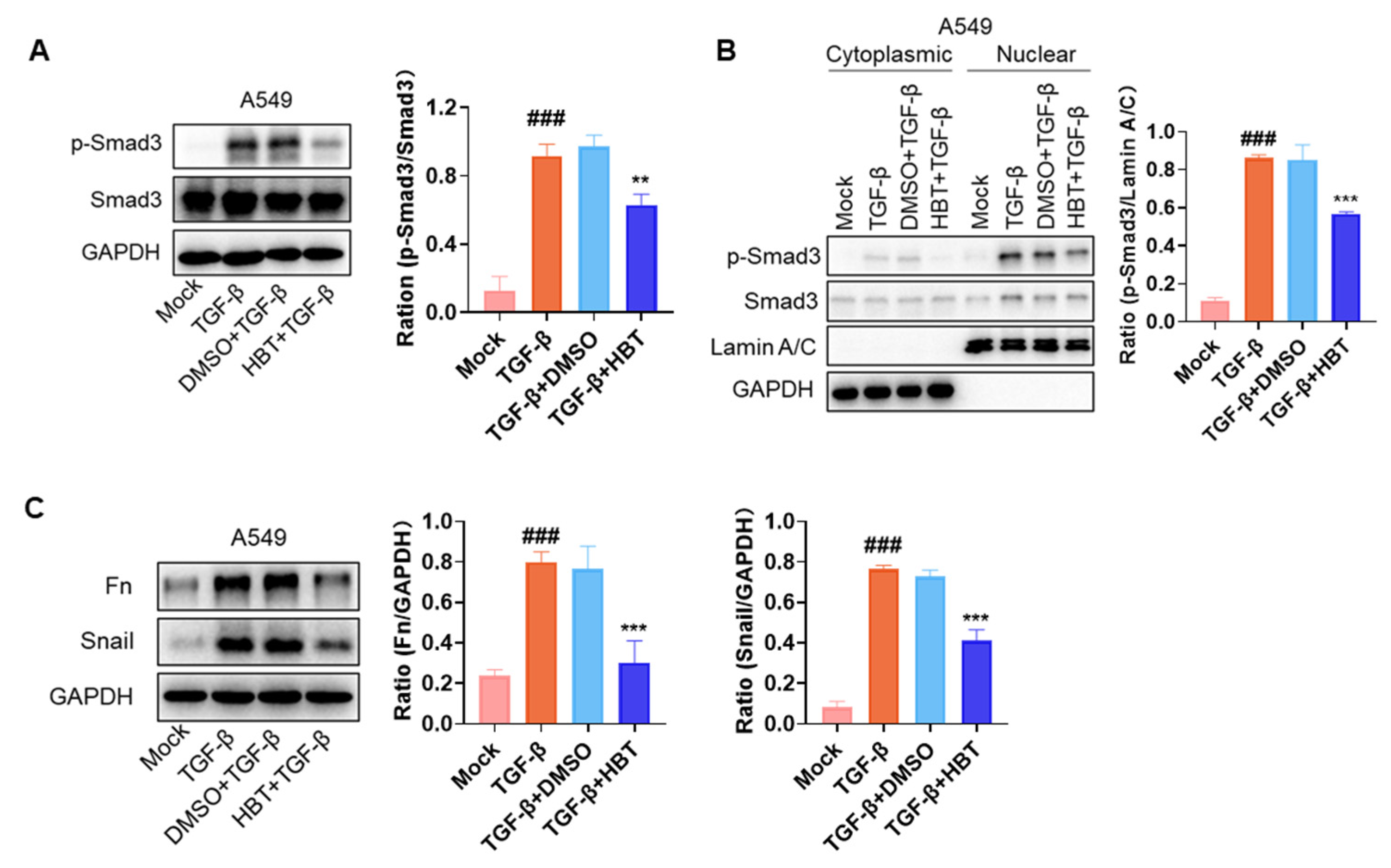
2.4. HBT Inhibited TGF-β-Induced Fibrosis Markers In Vitro
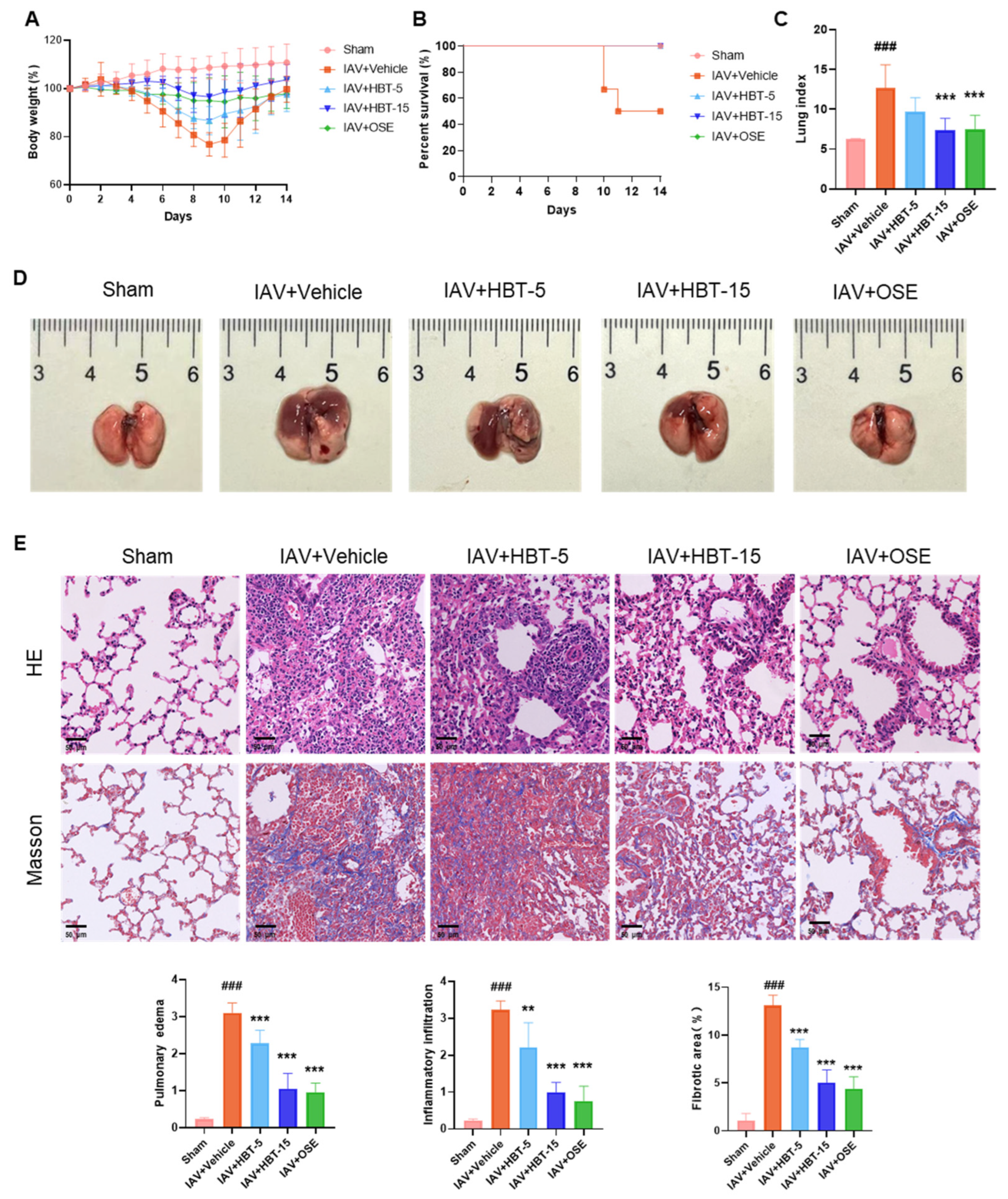
2.5. HBT Reduced Lung Injury and PF Induced by IAV In Vivo
2.6. HBT Reduced TGF-β/Smad3 Activation and Expression of Fibrosis-Related Proteins In Vivo
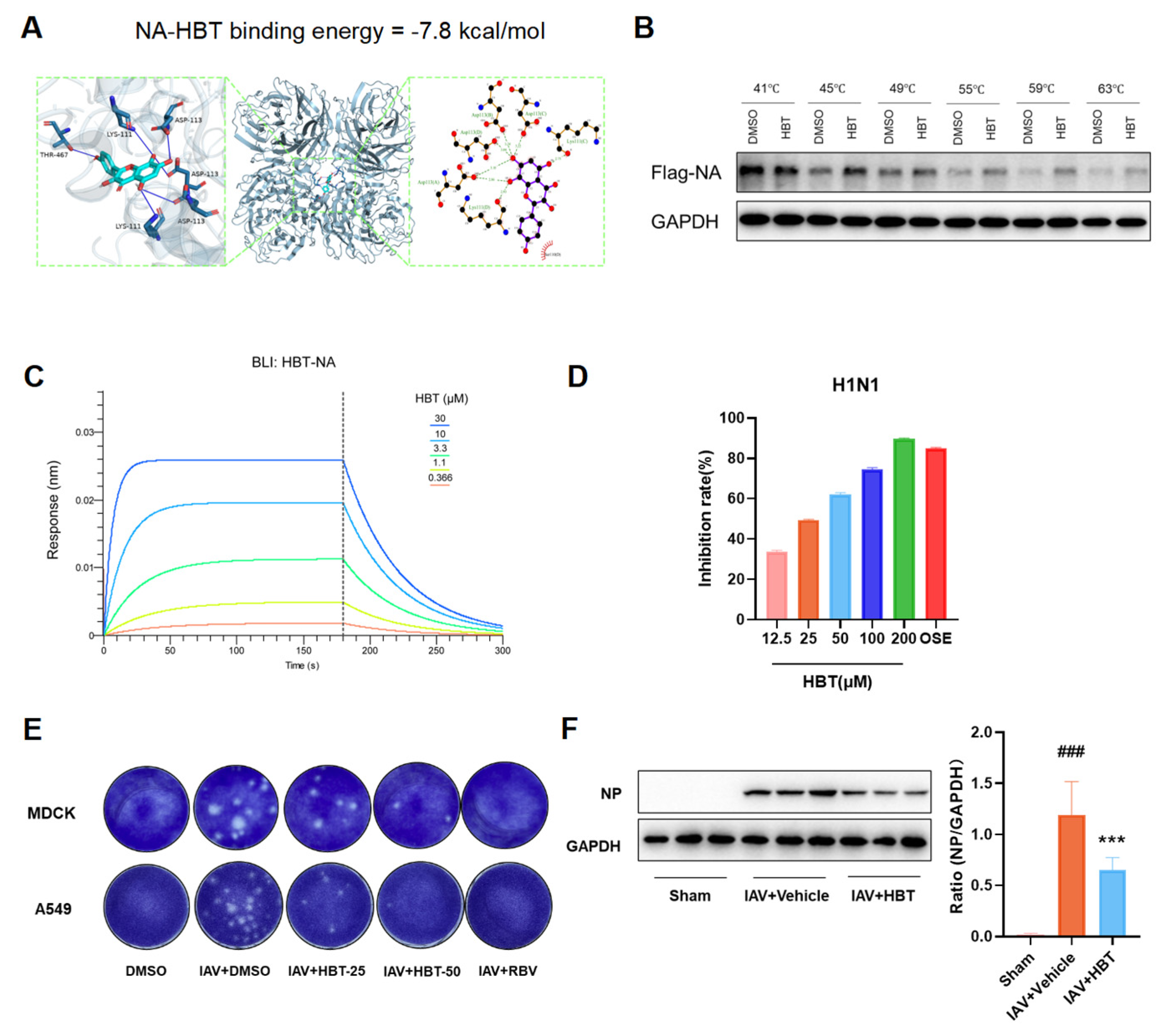
2.7. HBT Reduces IAV Infection by Blocking the NA Protein
3. Discussion
4. Materials and Methods
4.1. Cells and Virus
4.2. Compounds
4.3. Animal Experiment
4.4. Viral Infection Cells and Drug Therapy
4.5. Western Blot
4.6. Quantitative Real-Time PCR (qPCR)
4.7. Cell Counting Kit-8 (CCK8) Assay
4.8. Nuclear and Cytoplasmic Extraction
4.9. Immunohistochemistry
4.10. Plaque Assay
4.11. Molecular Docking
4.12. Biolayer Interferometry (BLI) Assay
4.13. Cellular Thermal Shift Assay (CETSA)
4.14. Neuraminidase Inhibition Assay
4.15. HE and Masson Staining
4.16. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| IAV | Influenza A virus |
| IBV | Influenza B virus |
| ARDS | acute respiratory distress syndrome |
| PF | pulmonary fibrosis |
| R. rosea L. | Rhodiola rosea L. |
| HBT | Herbacetin |
| BLI | BioLayer Interferometry |
| CETSA | cell thermal shift assay |
| NA | neuraminidase |
| TGF-β | transforming growth factor-β |
| Fn | fibronectin |
| TNF-α | tumor necrosis factor-α |
| OSE | oseltamivir |
| DMSO | Dimethyl sulfoxide |
| AIV | Avian Influenza virus |
| RBV | ribavirin |
| MOI | Multiplicity of infection |
| MDCK | Madin-Darby Canine Kidney |
| PBS | phosphate-buffered saline |
| qPCR | Quantitative Real-Time PCR |
| CCK8 | Cell Counting Kit-8 |
| DAB | diamino-benzidine |
References
- Iuliano, A.D.; Roguski, K.M.; Chang, H.H.; Muscatello, D.J.; Palekar, R.; Tempia, S.; Cohen, C.; Gran, J.M.; Schanzer, D.; Cowling, B.J.; et al. Estimates of global seasonal influenza-associated respiratory mortality: A modelling study. Lancet 2018, 391, 1285–1300. [Google Scholar] [CrossRef] [PubMed]
- Lafond, K.E.; Porter, R.M.; Whaley, M.J.; Suizan, Z.; Ran, Z.; Aleem, M.A.; Thapa, B.; Sar, B.; Proschle, V.S.; Peng, Z.; et al. Global burden of influenza-associated lower respiratory tract infections and hospitalizations among adults: A systematic review and meta-analysis. PLoS Med. 2021, 18, e1003550. [Google Scholar] [CrossRef]
- de Courville, C.; Cadarette, S.M.; Wissinger, E.; Alvarez, F.P. The economic burden of influenza among adults aged 18 to 64: A systematic literature review. Influenza Other Respir. Viruses 2022, 16, 376–385. [Google Scholar] [CrossRef]
- Paget, J.; Spreeuwenberg, P.; Charu, V.; Taylor, R.J.; Iuliano, A.D.; Bresee, J.; Simonsen, L.; Viboud, C. Global mortality associated with seasonal influenza epidemics: New burden estimates and predictors from the glamor project. J. Glob. Health 2019, 9, 20421. [Google Scholar] [CrossRef]
- Wang, X.; Li, Y.; O’Brien, K.L.; Madhi, S.A.; Widdowson, M.A.; Byass, P.; Omer, S.B.; Abbas, Q.; Ali, A.; Amu, A.; et al. Global burden of respiratory infections associated with seasonal influenza in children under 5 years in 2018: A systematic review and modelling study. Lancet Glob. Health 2020, 8, e497–e510. [Google Scholar] [CrossRef]
- Uyeki, T.M.; Hui, D.S.; Zambon, M.; Wentworth, D.E.; Monto, A.S. Influenza. Lancet 2022, 400, 693–706. [Google Scholar] [CrossRef]
- Gautam, A.; Boyd, D.F.; Nikhar, S.; Zhang, T.; Siokas, I.; Van de Velde, L.A.; Gaevert, J.; Meliopoulos, V.; Thapa, B.; Rodriguez, D.A.; et al. Necroptosis blockade prevents lung injury in severe influenza. Nature 2024, 628, 835–843. [Google Scholar] [CrossRef]
- Michalski, J.E.; Kurche, J.S.; Schwartz, D.A. From ards to pulmonary fibrosis: The next phase of the COVID-19 pandemic? Transl. Res. 2022, 241, 13–24. [Google Scholar] [CrossRef]
- Nakajima, N.; Sato, Y.; Katano, H.; Hasegawa, H.; Kumasaka, T.; Hata, S.; Tanaka, S.; Amano, T.; Kasai, T.; Chong, J.M.; et al. Histopathological and immunohistochemical findings of 20 autopsy cases with 2009 h1n1 virus infection. Mod. Pathol. 2012, 25, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mineo, G.; Ciccarese, F.; Modolon, C.; Landini, M.P.; Valentino, M.; Zompatori, M. Post-ARDS pulmonary fibrosis in patients with h1n1 pneumonia: Role of follow-up CT. Radiol. Med. 2012, 117, 185–200. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.; Chu, W.; Duan, J.; Li, J.; Ma, W.; Hu, C.; Yao, M.; Xing, L.; Yang, Y. Six-month outcomes of post-ards pulmonary fibrosis in patients with h1n1 pneumonia. Front. Mol. Biosci. 2021, 8, 640763. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Mark, Z.F.; Carbajal, M.P.; Delima, D.S.; Chamberlain, N.; Walzer, J.; Ruban, M.; Chandrasekaran, R.; Daphtary, N.; Aliyeva, M.; et al. The protein disulfide isomerase a3 and osteopontin axis promotes influenza-induced lung remodelling. Br. J. Pharmacol. 2024, 181, 4610–4627. [Google Scholar] [CrossRef]
- Chen, J.; Wu, J.; Hao, S.; Yang, M.; Lu, X.; Chen, X.; Li, L. Long term outcomes in survivors of epidemic influenza a (H7N9) virus infection. Sci. Rep. 2017, 7, 17275. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Bai, X.; Li, H.; Li, H.; Fan, W.; Zhang, H.; Liu, W.; Sun, L. Influenza a and b virus-triggered epithelial-mesenchymal transition is relevant to the binding ability of na to latent tgf-beta. Front. Microbiol. 2022, 13, 841462. [Google Scholar] [CrossRef]
- Narasimhan, H.; Cheon, I.S.; Qian, W.; Hu, S.S.; Parimon, T.; Li, C.; Goplen, N.; Wu, Y.; Wei, X.; Son, Y.M.; et al. An aberrant immune-epithelial progenitor niche drives viral lung sequelae. Nature 2024, 634, 961–969. [Google Scholar] [CrossRef]
- Hutchinson, E.C. Influenza virus. Trends Microbiol. 2018, 26, 809–810. [Google Scholar] [CrossRef]
- Carlson, C.M.; Turpin, E.A.; Moser, L.A.; O’Brien, K.B.; Cline, T.D.; Jones, J.C.; Tumpey, T.M.; Katz, J.M.; Kelley, L.A.; Gauldie, J.; et al. Transforming growth factor-beta: Activation by neuraminidase and role in highly pathogenic h5n1 influenza pathogenesis. PLoS Pathog. 2010, 6, e1001136. [Google Scholar] [CrossRef]
- Liu, M.; van Kuppeveld, F.J.; de Haan, C.A.; de Vries, E. Gradual adaptation of animal influenza a viruses to human-type sialic acid receptors. Curr. Opin. Virol. 2023, 60, 101314. [Google Scholar] [CrossRef]
- Schultz-Cherry, S.; Hinshaw, V.S. Influenza virus neuraminidase activates latent transforming growth factor beta. J. Virol. 1996, 70, 8624–8629. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.H.; Chen, D.Q.; Wang, Y.N.; Feng, Y.L.; Cao, G.; Vaziri, N.D.; Zhao, Y.Y. New insights into tgf-beta/smad signaling in tissue fibrosis. Chem. Biol. Interact. 2018, 292, 76–83. [Google Scholar] [CrossRef]
- Cheng, W.H.; Kao, S.Y.; Chen, C.L.; Yuliani, F.S.; Lin, L.Y.; Lin, C.H.; Chen, B.C. Amphiregulin induces ccn2 and fibronectin expression by tgf-beta through egfr-dependent pathway in lung epithelial cells. Respir. Res. 2022, 23, 381. [Google Scholar] [CrossRef]
- Lv, Q.; Wang, J.; Xu, C.; Huang, X.; Ruan, Z.; Dai, Y. Pirfenidone alleviates pulmonary fibrosis in vitro and in vivo through regulating wnt/gsk-3beta/beta-catenin and tgf-beta1/smad2/3 signaling pathways. Mol. Med. 2020, 26, 49. [Google Scholar] [CrossRef]
- Li, F.; Piattini, F.; Pohlmeier, L.; Feng, Q.; Rehrauer, H.; Kopf, M. Monocyte-derived alveolar macrophages autonomously determine severe outcome of respiratory viral infection. Sci. Immunol. 2022, 7, eabj5761. [Google Scholar] [CrossRef]
- Wynn, T.A.; Vannella, K.M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity 2016, 44, 450–462. [Google Scholar] [CrossRef] [PubMed]
- Gillman, A.; Muradrasoli, S.; Soderstrom, H.; Holmberg, F.; Latorre-Margalef, N.; Tolf, C.; Waldenstrom, J.; Gunnarsson, G.; Olsen, B.; Jarhult, J.D. Oseltamivir-resistant influenza a (H1N1) virus strain with an h274y mutation in neuraminidase persists without drug pressure in infected mallards. Appl. Environ. Microbiol. 2015, 81, 2378–2383. [Google Scholar] [CrossRef] [PubMed]
- Mu, S.; Zou, X.; Wang, Y.; Deng, X.; Cui, D.; Liu, S.; Cao, B. The combined effect of oseltamivir and favipiravir on influenza a virus evolution in patients hospitalized with severe influenza. Antivir. Res. 2023, 216, 105657. [Google Scholar] [CrossRef]
- Zhang, H.; Wang, L.; Compans, R.W.; Wang, B.Z. Universal influenza vaccines, a dream to be realized soon. Viruses 2014, 6, 1974–1991. [Google Scholar] [CrossRef]
- Tao, H.; Wu, X.; Cao, J.; Peng, Y.; Wang, A.; Pei, J.; Xiao, J.; Wang, S.; Wang, Y. Rhodiola species: A comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Med. Res. Rev. 2019, 39, 1779–1850. [Google Scholar] [CrossRef] [PubMed]
- Pu, W.L.; Zhang, M.Y.; Bai, R.Y.; Sun, L.K.; Li, W.H.; Yu, Y.L.; Zhang, Y.; Song, L.; Wang, Z.X.; Peng, Y.F.; et al. Anti-inflammatory effects of Rhodiola rosea L.: A review. Biomed. Pharmacother. 2020, 121, 109552. [Google Scholar] [CrossRef]
- Ivanova, S.E.; Quintela, J.C. The effectiveness of Rhodiola rosea L. Preparations in alleviating various aspects of life-stress symptoms and stress-induced conditions-encouraging clinical evidence. Molecules 2022, 27, 3902. [Google Scholar] [CrossRef]
- Xu, W.; Yang, T.; Zhang, J.; Li, H.; Guo, M. Rhodiola rosea: A review in the context of pppm approach. EPMA J. 2024, 15, 233–259. [Google Scholar] [CrossRef]
- Zhang, K.; Si, X.P.; Huang, J.; Han, J.; Liang, X.; Xu, X.B.; Wang, Y.T.; Li, G.Y.; Wang, H.Y.; Wang, J.H. Preventive effects of Rhodiola rosea L. On bleomycin-induced pulmonary fibrosis in rats. Int. J. Mol. Sci. 2016, 17, 879. [Google Scholar] [CrossRef]
- Doring, K.; Langeder, J.; Duwe, S.; Tahir, A.; Grienke, U.; Rollinger, J.M.; Schmidtke, M. Insights into the direct anti-influenza virus mode of action of Rhodiola rosea. Phytomedicine 2022, 96, 153895. [Google Scholar] [CrossRef]
- Panossian, A.; Wikman, G.; Sarris, J. Rosenroot (Rhodiola rosea): Traditional use, chemical composition, pharmacology and clinical efficacy. Phytomedicine 2010, 17, 481–493. [Google Scholar] [CrossRef]
- Zakharenko, A.M.; Razgonova, M.P.; Pikula, K.S.; Golokhvast, K.S. Simultaneous determination of 78 compounds of Rhodiola rosea extract by supercritical CO2-extraction and hplc-esi-ms/ms spectrometry. Biochem. Res. Int. 2021, 2021, 9957490. [Google Scholar] [CrossRef] [PubMed]
- Peter, Z.Z.; Kusz, N.; Csupor, D.; Peschel, W. Rhodiosin and herbacetin in Rhodiola rosea preparations: Additional markers for quality control? Pharm. Biol. 2019, 57, 295–305. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.; Wang, Y.; Yu, M.; Shang, Y.; Chang, Y.; Zhao, H.; Kang, Y.; Zhao, L.; Xu, L.; Zhao, X.; et al. Discovery of herbacetin as a novel sgk1 inhibitor to alleviate myocardial hypertrophy. Adv. Sci. 2022, 9, e2101485. [Google Scholar] [CrossRef]
- Jo, S.; Kim, S.; Kim, D.Y.; Kim, M.S.; Shin, D.H. Flavonoids with inhibitory activity against SARS-cov-2 3clpro. J. Enzym. Inhib. Med. Chem. 2020, 35, 1539–1544. [Google Scholar] [CrossRef]
- Li, Y.; Zou, H.; Ma, L.; Hu, D.; Long, H.; Lin, J.; Luo, Z.; Zhou, Y.; Liao, F.; Wang, X.; et al. Fuzheng jiedu decoction alleviates h1n1 virus-induced acute lung injury in mice by suppressing the nlrp3 inflammasome activation. J. Ethnopharmacol. 2025, 341, 119314. [Google Scholar] [CrossRef]
- Li, L.; Fan, P.; Chou, H.; Li, J.; Wang, K.; Li, H. Herbacetin suppressed mmp9 mediated angiogenesis of malignant melanoma through blocking egfr-erk/akt signaling pathway. Biochimie 2019, 162, 198–207. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Jiang, Y.H.; Fei, X.; Wang, M.; Liu, A.; Li, X.; Jiang, Z. Herbacetin inhibits asthma development by blocking the sgk1/nf-kappab signaling pathway. J. Asthma Allergy 2024, 17, 703–716. [Google Scholar] [CrossRef]
- Huang, W.J.; Tang, X.X. Virus infection induced pulmonary fibrosis. J. Transl. Med. 2021, 19, 496. [Google Scholar] [CrossRef]
- Gonzales, G.B. In vitro bioavailability and cellular bioactivity studies of flavonoids and flavonoid-rich plant extracts: Questions, considerations and future perspectives. Proc. Nutr. Soc. 2017, 76, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Ge, B.K.; Zhao, L.; Li, X.R.; Xu, P.X.; Xue, M. Glucuronidation is the dominating in vivo metabolism pathway of herbacetin: Elucidation of herbacetin pharmacokinetics after intravenous and oral administration in rats. J. Funct. Foods 2018, 40, 659–669. [Google Scholar] [CrossRef]
- Yuan, D.; Guo, Y.; Pu, F.; Yang, C.; Xiao, X.; Du, H.; He, J.; Lu, S. Opportunities and challenges in enhancing the bioavailability and bioactivity of dietary flavonoids: A novel delivery system perspective. Food Chem. 2024, 430, 137115. [Google Scholar] [CrossRef] [PubMed]
- O’Boyle, N.M.; Banck, M.; James, C.A.; Morley, C.; Vandermeersch, T.; Hutchison, G.R. Open babel: An open chemical toolbox. J. Cheminform. 2011, 3, 33. [Google Scholar] [CrossRef]
- Eberhardt, J.; Santos-Martins, D.; Tillack, A.F.; Forli, S. Autodock vina 1.2.0: New docking methods, expanded force field, and python bindings. J. Chem. Inf. Model. 2021, 61, 3891–3898. [Google Scholar] [CrossRef]
- Volkamer, A.; Kuhn, D.; Grombacher, T.; Rippmann, F.; Rarey, M. Combining global and local measures for structure-based druggability predictions. J. Chem. Inf. Model. 2012, 52, 360–372. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liao, F.; Li, S.; Wu, L.; Chen, J.; Luo, Z.; Zhong, M.; Li, Q.; Wang, W.; Li, G. Herbacetin Alleviates Influenza Virus-Induced Lung Injury and Fibrosis by Targeting the Neuraminidase Protein. Pharmaceuticals 2025, 18, 1306. https://doi.org/10.3390/ph18091306
Liao F, Li S, Wu L, Chen J, Luo Z, Zhong M, Li Q, Wang W, Li G. Herbacetin Alleviates Influenza Virus-Induced Lung Injury and Fibrosis by Targeting the Neuraminidase Protein. Pharmaceuticals. 2025; 18(9):1306. https://doi.org/10.3390/ph18091306
Chicago/Turabian StyleLiao, Feng, Sha Li, Liumei Wu, Jiafan Chen, Ziqing Luo, Ming Zhong, Qiuhong Li, Wenbiao Wang, and Geng Li. 2025. "Herbacetin Alleviates Influenza Virus-Induced Lung Injury and Fibrosis by Targeting the Neuraminidase Protein" Pharmaceuticals 18, no. 9: 1306. https://doi.org/10.3390/ph18091306
APA StyleLiao, F., Li, S., Wu, L., Chen, J., Luo, Z., Zhong, M., Li, Q., Wang, W., & Li, G. (2025). Herbacetin Alleviates Influenza Virus-Induced Lung Injury and Fibrosis by Targeting the Neuraminidase Protein. Pharmaceuticals, 18(9), 1306. https://doi.org/10.3390/ph18091306






