Advances in the Application of Graphene and Its Derivatives in Drug Delivery Systems
Abstract
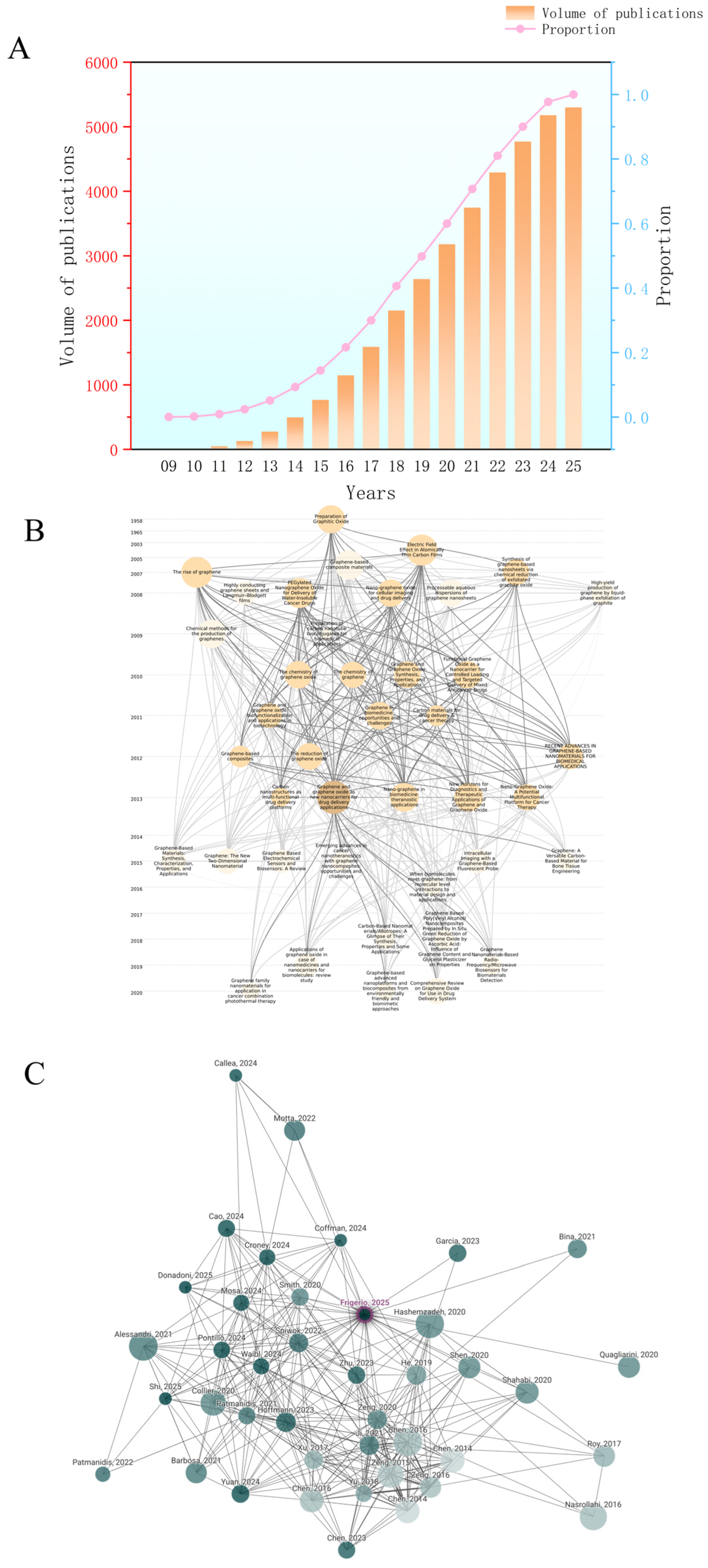
1. Introduction
2. Preparation, Drug Loading, and Targeted Delivery Strategies of GFNs
2.1. Preparation Methods of GFNs
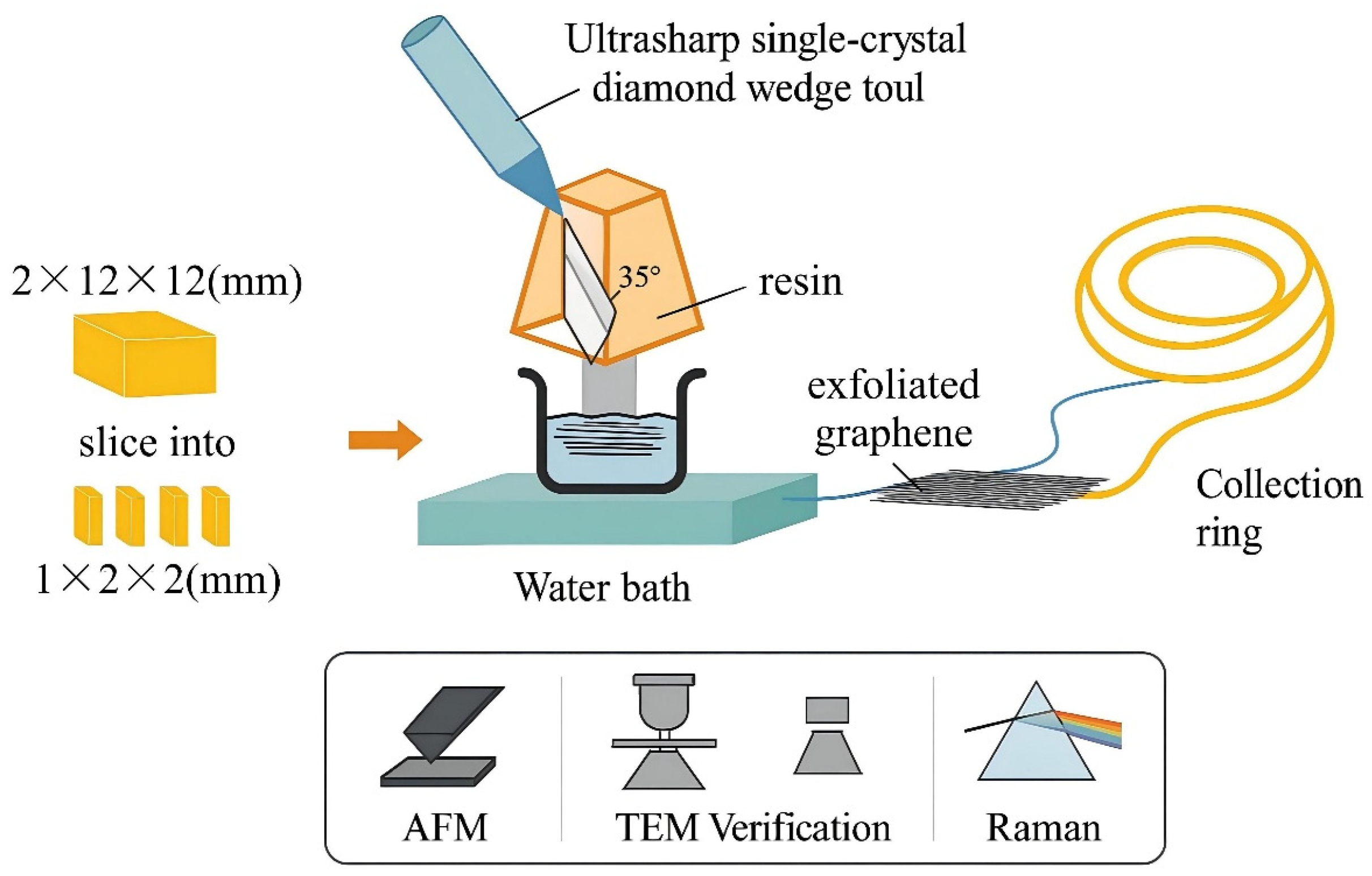
2.1.1. Mechanical Methods for Graphene Synthesis
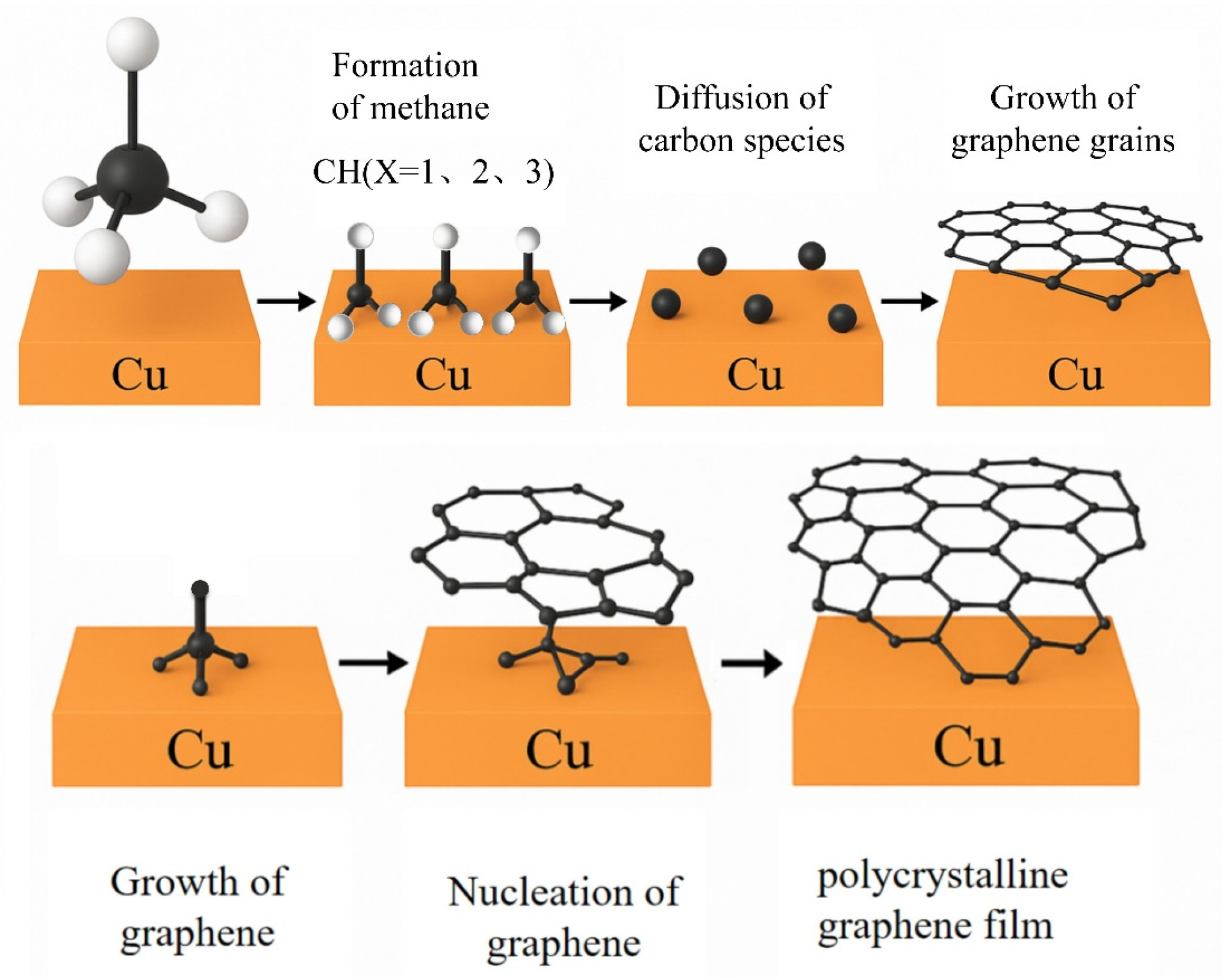
2.1.2. CVD Method
- Adsorption of methane onto the copper surface;
- Partial dehydrogenation of methane to form carbon-containing species, such as CHx (x = 0–3);
- Surface diffusion of these carbon species on the copper substrate;
- Nucleation of graphene at active sites on the copper surface, forming graphene nuclei;
- Adsorption of carbon species at the edges of graphene nuclei, promoting grain growth;
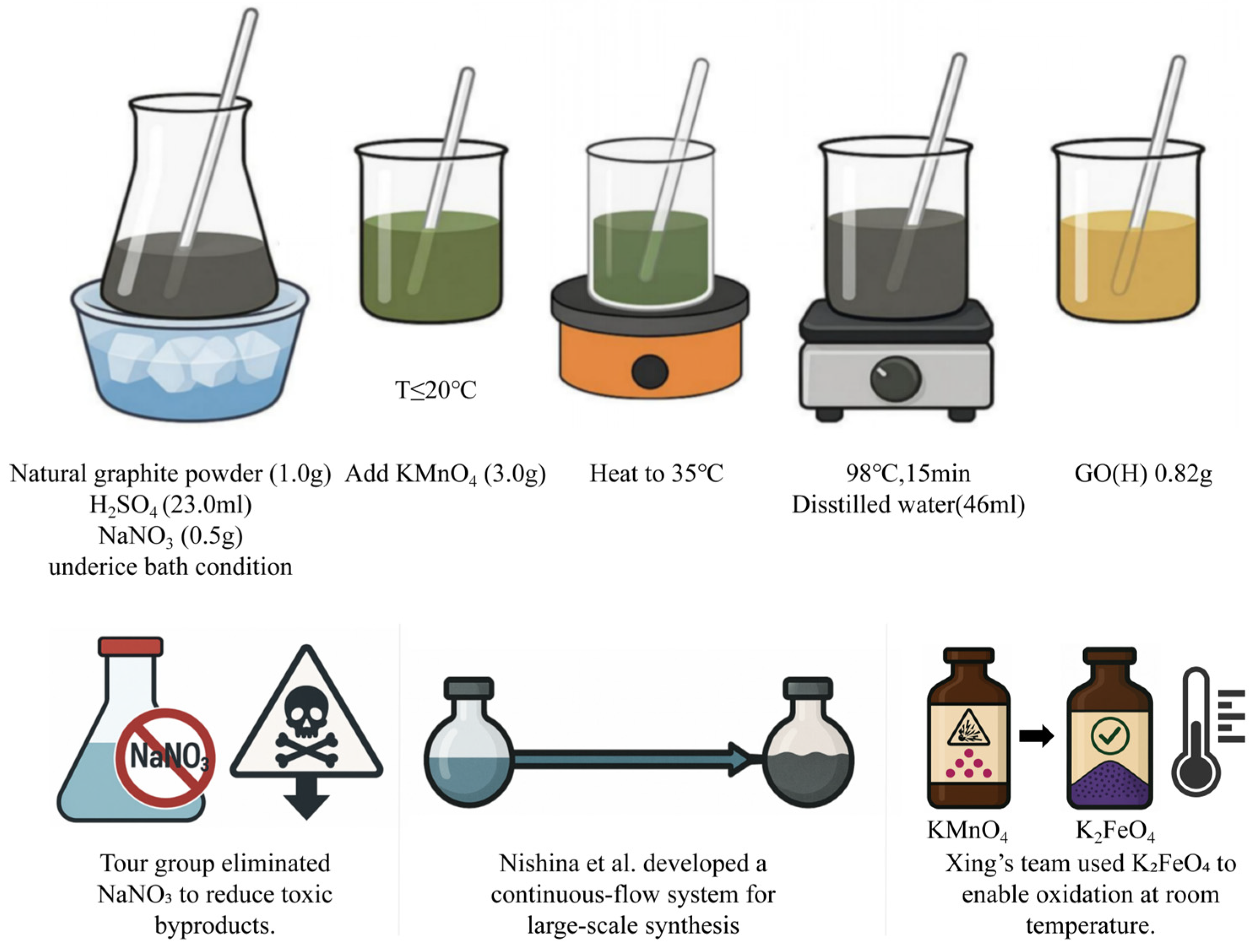
2.1.3. Hummer’s Method for GO Preparation
2.2. Drug Loading of GFNs
- Covalent linkages, including amide bonds, ester bonds, and phosphoester bonds [33].
2.2.1. Delivery of Hydrophobic Small-Molecule Drugs
2.2.2. Delivery of Hydrophilic Small-Molecule Drugs
2.2.3. Delivery of Macromolecular Drugs
- Non-covalent adsorption methods based on mixed adsorption;
- Ultrasonication-assisted dispersion techniques;
- Electrospinning encapsulation methods;
- And covalent grafting approaches aimed at enhancing binding stability.
2.3. Targeted Delivery of GFNs
2.3.1. Enhanced Targeting via Ligand Surface Modification
2.3.2. Stimuli-Responsive Release: pH-Triggered Controlled Release Strategy
3. Applications of GFNs Delivery Systems in Different Diseases
3.1. Cancer Therapy
3.1.1. Breast Cancer
3.1.2. Lung Cancer
- Widespread drug distribution throughout the body,
- Strong side effects and toxicity,
- Poor tumor targeting,
- And high drug resistance.
3.1.3. Cervical Cancer
- Widespread drug distribution in the body,
- Lack of tumor specificity,
- High potential for drug resistance,
- And significant side effects on normal tissues.
3.1.4. Liver Cancer
3.1.5. Other Cancers
3.2. Treatment of Neurological Disorders
3.2.1. Alzheimer’s Disease
3.2.2. Parkinson’s Disease (PD)
3.3. Cardiovascular Diseases
3.4. Bacterial Infections and Inflammation
3.4.1. Bacterial Infections
3.4.2. Inflammation
3.5. Diabetes
3.6. Drug Delivery for Imaging and Diagnostic Functions
4. Preclinical Research and Clinical Translation Challenges
4.1. Biocompatibility Assessment
4.2. Toxicity and Safety
5. Conclusions and Outlook
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| GBNs | Graphene-based nanomaterials |
| GQDs | Graphene quantum dots |
| GO | Graphene oxide |
| rGO | Reduced graphene oxide |
| PTX | Paclitaxel |
| DOX | Doxorubicin |
| GFNs | Graphene-family nanomaterials, |
| CVD | Chemical vapor deposition |
| HOPG | Highly oriented pyrolytic graphite |
| APT | Aptamer |
| AFM | Atomic force microscope |
| TEM | Transmission electron microscopy |
| DEX | Dexamethasone |
| HA | Hyaluronic acid |
| PEG | Polyethylene glycol |
| RFP | Rifampicin |
| QSR | Quercetin |
| CPT | Camptothecin |
| GAD | Gambogic acid D |
| PVA | Polyvinyl alcohol |
| CIP | Ciprofloxacin |
| MTX | Methotrexate |
| PEI | Polyethylenimine |
| EGFR | Epidermal growth factor receptor |
| FA | Folic acid |
| FAP | Fibroblast activation protein |
| NPF@DOX | Doxorubicin-loaded pegylated nano-graphene oxide functionalized with FAP |
| C23 | Targeted nucleolin |
| ADH | Adipic acid dihydrazide |
| DHA | Dihydroartemisinin |
| Tf | Transferrin |
| ROS | Reactive oxygen species |
| PKE | Polymer |
| ATRP | Atom transfer radical polymerization |
| GSH | Glutathione |
| PAMAM | Polyamidoamine |
| DST | Density functional theory |
| NIR | Near-infrared radiation |
| GOMs | Hollow graphene oxide microcapsules |
| BBN-AF750 | bombesin antagonist peptides conjugated to Alexa Fluor 750 |
| PhaL | Phaseolus vulgaris leucoagglutinin |
| NGO-SS-HA | HA-functionalized GO-based gefitinib delivery system |
| PTT | Photothermal Therapy group |
| FGO | Fluorinated graphene oxide |
| CMC | Carboxymethyl chitosan |
| LA | Lactobionic acid |
| OSCC | Oral squamous cell carcinoma |
| MGOMCS | Magnetic graphene oxide microcapsules |
| AD | Alzheimer’s disease |
| DAU | Dauricine |
| BBB | Blood–brain barrier |
| CS | Chitosan |
| AChE | Acetylcholinesterase |
| N-GQDs | Nitrogen-doped graphene quantum dots |
| PD | Parkinson’s disease |
| Lf | Lactoferrin |
| Pue | Puerarin |
| EpCAM | Epithelial cell adhesion molecule |
| CPCs | Cardiac progenitor cell |
| MDG | Methanol derived graphene |
| MGO | Magnetic graphene oxide |
| PEEK | Polyetheretherketone |
| PLA | Polylactic acid |
| Fe3O4 | Magnetite (Iron(II,III) oxide) |
| DEX-P | Dexamethasone phosphomonoester |
| Anti-IL10R | Anti-interleukin-10 receptor antibody |
| PEGDMA | Poly(ethylene glycol) dimethacrylate |
| GNFH | Npn, nh2-peg-nh |
| CD44 | Cluster of Differentiation 44 |
| PEGDA | Polyethylene (glycol) diacrylate |
| IDO | Indoleamine 2,3-dioxygenase |
| ICD | Immunogenic cell death |
| NGO-SS-HA | Nano Graphene Oxide–Disulfide bond–Hyaluronic Acid |
| shRNA | Short hairpin rna |
| CRISPR | Clustered Regularly Interspaced Short Palindromic Repeats |
| Cas9 | CRISPR-associated protein 9 |
| CMCS | Carboxymethyl chitosan |
| DMMA | Dimethylmaleic anhydride |
| shABCG | Short hairpin RNA against ATP-binding cassette sub-family G member 2 |
| GEL | Gelatin |
| PEO | Poly(ethyleneoxide) |
| nGO | Nano graphene oxide |
| Ab | Antibody |
| AuNPs | Gold nanoparticles |
| LDH | Lactate dehydrogenase |
| ALP | Alkaline phosphatase |
| IL-10R | Interleukin-10 receptor |
| GO-PEG@GAD | PEG-modified and EGFR antibody-functionalized GO |
| PAC@HA-ADH-GQDs | Paclitaxel-loaded hyaluronic adipic acid dihydrazide–functionalized graphene quantum dots |
| GO-TD-Fe3O4@PEG | Graphene Oxide–Thiodiglycol–Iron Oxide coated with Polyethylene Glycol |
| GO@LM-SP-FA | Multifunctional graphene oxide nano drug carrier |
| GO-HA | Graphene oxide–hyaluronic acid |
| GO-PEG-EGFR | Graphene oxide-polyethylene glycol-anti-epidermal growth factor receptor |
| FA-GO | Folic acid–graphene oxide |
| MGO@APT | Aptamer-conjugated magnetic graphene oxide nanocarrier |
| DOX@NGOBBN | Doxorubicin-loaded nano-graphene oxide conjugated with BBN-AF750 |
| GO-PAMAM | Graphene oxide–polyamidoamine |
| GO–CD–FA | CDI-activated β-CD and FA co-modified graphene oxide nanocarriers |
| HA-GO/Fe3O4 | Hyaluronic Acid-modified Graphene Oxide/Magnetite Nanocomposite |
| GYSMNP@PF127 | Hydrophilic graphene-based yolk-shell magnetic nanoparticles functionalized with copolymer pluronic F-127 |
| CS-Aco | Chitosan–aconitic anhydride |
| Lac-CMC-GO | Lactic acid–carboxymethyl chitosan–graphene oxide |
| CS–GQDs | Chitosan–graphene quantum dots |
| Clitoria ternatea-–GQDs | Clitoria ternatea Mediated Graphene Quantum Dots |
| PEI-PEG@GO | Polyethyleneimine and polyethylene glycol bonded to graphene oxide |
| SNO-Cys@PGO | S-nitrosocysteamine-functionalised |
| AgNW | Silver nanowire |
| PEGDMA-rGO | Rgo impregnated poly(ethylene glycol) dimethacrylate based hydrogels |
| In@GO NgC | An insulin intercalated GO based nanogel composite |
References
- Paik, P. Graphene Oxide for Biomedical Applications. JNMR 2017, 5. [Google Scholar] [CrossRef]
- Hossain, M. The Fate of Graphene in Biomedical Applications. N. Am. Acad. Res. 2019, 2, 1–13. [Google Scholar]
- Liu, J.; Cui, L.; Losic, D. Graphene and Graphene Oxide as New Nanocarriers for Drug Delivery Applications. Acta Biomater. 2013, 9, 9243–9257. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The Rise of Graphene. Nature Mater. 2007, 6, 183–191. [Google Scholar] [CrossRef]
- Dasari Shareena, T.P.; McShan, D.; Dasmahapatra, A.K.; Tchounwou, P.B. A Review on Graphene-Based Nanomaterials in Biomedical Applications and Risks in Environment and Health. Nano-Micro Lett. 2018, 10, 53. [Google Scholar] [CrossRef]
- Gonçalves, G.; Vila, M.; Portolés, M.-T.; Vallet-Regi, M.; Gracio, J.; Marques, P.A.A.P. Nano-Graphene Oxide: A Potential Multifunctional Platform for Cancer Therapy. Adv. Healthc. Mater. 2013, 2, 1072–1090. [Google Scholar] [CrossRef]
- Maktedar, S.S.; Avashthi, G.; Singh, M. Understanding the Significance of O-Doped Graphene towards Biomedical Applications. RSC Adv. 2016, 6, 114264–114275. [Google Scholar] [CrossRef]
- Karki, N.; Tiwari, H.; Tewari, C.; Rana, A.; Pandey, N.; Basak, S.; Sahoo, N. Functionalized Graphene Oxide as a Vehicle for Targeted Drug Delivery and Bioimaging Applications. J. Mater. Chem. B 2020, 8, 8116–8148. [Google Scholar] [CrossRef]
- Frigerio, G.; Motta, S.; Siani, P.; Donadoni, E.; Di Valentin, C. Unveiling the Drug Delivery Mechanism of Graphene Oxide Dots at the Atomic Scale. J. Control. Release 2025, 379, 344–362. [Google Scholar] [CrossRef]
- Yadav, N.; Lochab, B. A Comparative Study of Graphene Oxide: Hummers, Intermediate and Improved Method. FlatChem 2019, 13, 40–49. [Google Scholar] [CrossRef]
- Novoselov, K.S.; Geim, A.K.; Morozov, S.V.; Jiang, D.; Zhang, Y.; Dubonos, S.V.; Grigorieva, I.V.; Firsov, A.A. Electric Field Effect in Atomically Thin Carbon Films. Science 2004, 306, 666–669. [Google Scholar] [CrossRef]
- Lu, X.; Yu, M.; Huang, H.; Ruoff, R.S. Tailoring Graphite with the Goal of Achieving Single Sheets. Nanotechnology 1999, 10, 269. [Google Scholar] [CrossRef]
- Ci, L.; Song, L.; Jariwala, D.; Elías, A.L.; Gao, W.; Terrones, M.; Ajayan, P.M. Graphene Shape Control by Multistage Cutting and Transfer. Adv. Mater. 2009, 21, 4487–4491. [Google Scholar] [CrossRef]
- Jayasena, B.; Subbiah, S. A Novel Mechanical Cleavage Method for Synthesizing Few-Layer Graphenes. Nanoscale Res. Lett. 2011, 6, 95. [Google Scholar] [CrossRef]
- Anwar, A.; Chang, T.-P.; Chen, C.-T. Graphene Oxide Synthesis Using a Top–down Approach and Discrete Characterization Techniques: A Holistic Review. Carbon. Lett. 2022, 32, 1–38. [Google Scholar] [CrossRef]
- Li, X.; Cai, W.; An, J.; Kim, S.; Nah, J.; Yang, D.; Piner, R.; Velamakanni, A.; Jung, I.; Tutuc, E.; et al. Large-Area Synthesis of High-Quality and Uniform Graphene Films on Copper Foils. Science 2009, 324, 1312–1314. [Google Scholar] [CrossRef]
- Bae, S.; Kim, H.; Lee, Y.; Xu, X.; Park, J.-S.; Zheng, Y.; Balakrishnan, J.; Lei, T.; Ri Kim, H.; Song, Y.I.; et al. Roll-to-Roll Production of 30-Inch Graphene Films for Transparent Electrodes. Nat. Nanotech. 2010, 5, 574–578. [Google Scholar] [CrossRef]
- Sun, Z.; Yan, Z.; Yao, J.; Beitler, E.; Zhu, Y.; Tour, J.M. Growth of Graphene from Solid Carbon Sources. Nature 2010, 468, 549–552. [Google Scholar] [CrossRef]
- Gao, W. The Chemistry of Graphene Oxide. In Graphene Oxide: Reduction Recipes, Spectroscopy, and Applications; Gao, W., Ed.; Springer International Publishing: Cham, Switzerland, 2015; pp. 61–95. ISBN 978-3-319-15500-5. [Google Scholar]
- Artyukhov, V.I.; Liu, Y.; Yakobson, B.I. Equilibrium at the Edge and Atomistic Mechanisms of Graphene Growth. Proc. Natl. Acad. Sci. USA 2012, 109, 15136–15140. [Google Scholar] [CrossRef]
- Hao, Y.; Bharathi, M.S.; Wang, L.; Liu, Y.; Chen, H.; Nie, S.; Wang, X.; Chou, H.; Tan, C.; Fallahazad, B.; et al. The Role of Surface Oxygen in the Growth of Large Single-Crystal Graphene on Copper. Science 2013, 342, 720–723. [Google Scholar] [CrossRef]
- Vlassiouk, I.; Regmi, M.; Fulvio, P.; Dai, S.; Datskos, P.; Eres, G.; Smirnov, S. Role of Hydrogen in Chemical Vapor Deposition Growth of Large Single-Crystal Graphene. ACS Nano 2011, 5, 6069–6076. [Google Scholar] [CrossRef]
- Gao, L.; Guest, J.R.; Guisinger, N.P. Epitaxial Graphene on Cu(111). Nano Lett. 2010, 10, 3512–3516. [Google Scholar] [CrossRef]
- Yan, Z.; Lin, J.; Peng, Z.; Sun, Z.; Zhu, Y.; Li, L.; Xiang, C.; Samuel, E.L.; Kittrell, C.; Tour, J.M. Toward the Synthesis of Wafer-Scale Single-Crystal Graphene on Copper Foils. ACS Nano 2012, 6, 9110–9117. [Google Scholar] [CrossRef]
- Kim, Y.-T.; Han, J.H.; Hong, B.H.; Kwon, Y.-U. Electrochemical Synthesis of CdSe Quantum Dot Array on Graphene Basal Plane Using Mesoporous Silica Thin Film Templates. Adv. Mater. 2010, 22, 515–518. [Google Scholar] [CrossRef]
- Chung, C.; Kim, Y.-K.; Shin, D.; Ryoo, S.-R.; Hong, B.H.; Min, D.-H. Biomedical Applications of Graphene and Graphene Oxide. Available online: https://pubs.acs.org/doi/abs/10.1021/ar300159f (accessed on 14 April 2025).
- Marcano, D.C.; Kosynkin, D.V.; Berlin, J.M.; Sinitskii, A.; Sun, Z.; Slesarev, A.; Alemany, L.B.; Lu, W.; Tour, J.M. Improved Synthesis of Graphene Oxide. ACS Nano 2010, 4, 4806–4814. [Google Scholar] [CrossRef]
- Morimoto, N.; Suzuki, H.; Takeuchi, Y.; Kawaguchi, S.; Kunisu, M.; Bielawski, C.W.; Nishina, Y. Real-Time, in Situ Monitoring of the Oxidation of Graphite: Lessons Learned. Chem. Mater. 2017, 29, 2150–2156. [Google Scholar] [CrossRef]
- Yu, H.; Zhang, B.; Bulin, C.; Li, R.; Xing, R. High-Efficient Synthesis of Graphene Oxide Based on Improved Hummers Method. Sci. Rep. 2016, 6, 36143. [Google Scholar] [CrossRef]
- Bai, R.G.; Husseini, G.A. Graphene-Based Drug Delivery Systems. In Biomimetic Nanoengineered Materials for Advanced Drug Delivery; Elsevier: Amsterdam, The Netherlands, 2019; pp. 149–168. [Google Scholar]
- Shim, G.; Kim, M.G.; Park, J.Y.; Oh, Y.K. Graphene-Based Nanosheets for Delivery of Chemotherapeutics and Biological Drugs. Adv. Drug Deliv. Rev. 2016, 105, 205–227. [Google Scholar] [CrossRef]
- Zheng, S.; Xiong, J.; Wang, L.; Zhai, D.; Xu, Y.; Lin, F. E-Graphene: A Computational Platform for the Prediction of Graphene-Based Drug Delivery System by Quantum Genetic Algorithm and Cascade Protocol. Front. Chem. 2021, 9, 664355. [Google Scholar] [CrossRef]
- Lin, J.H.; Chen, L.; Cho, E.C.; Lee, K.C. Influence of Bonding Variance on Electron Affinity in Graphene Quantum Dot-Barium Titanate Nanocomposites for Drug Delivery System. FlatChem 2024, 47, 100713. [Google Scholar] [CrossRef]
- Hashemzadeh, H.; Raissi, H. Understanding Loading, Diffusion and Releasing of Doxorubicin and Paclitaxel Dual Delivery in Graphene and Graphene Oxide Carriers as Highly Efficient Drug Delivery Systems. Appl. Surf. Sci. 2020, 500, 144220. [Google Scholar] [CrossRef]
- Ji, Y.; Zhu, R.; Shen, Y.; Tan, Q.; Chen, J. Comparison of Loading and Unloading of Different Small Drugs on Graphene and Its Oxide. J. Mol. Liq. 2021, 341, 117454. [Google Scholar] [CrossRef]
- Wang, J.; Yu, Y.; Dong, H.; Ji, Y.; Ning, W.; Li, Y. The Interface Hydrophilic–Hydrophobic Integration of Fluorinated Defective Graphene towards Biomedical Applications. Phys. Chem. Chem. Phys. 2025, 27, 7538–7555. [Google Scholar] [CrossRef]
- Matiyani, M.; Rana, A.; Karki, N.; Garwal, K.; Pal, M.; Sahoo, N.G. Development of Multi-Functionalized Graphene Oxide Based Nanocarrier for the Delivery of Poorly Water Soluble Anticancer Drugs. J. Drug Deliv. Sci. Technol. 2023, 83, 104412. [Google Scholar] [CrossRef]
- Yeh, C.-F.; Lee, S.-W.; Liu, C.-H.; Hsu, Y.-B.; Lan, M.-C.; Lu, Y.-J.; Chen, J.-P.; Lan, M.-Y. EpCAM-Targeted Erlotinib Delivery: Enhancing Nasopharyngeal Carcinoma Treatment With Polyethylene Glycol-Coated Graphene Oxide. Head Neck 2025, 47, 2010–2024. [Google Scholar] [CrossRef]
- Hu, Y.; Ma, L.; Shi, Q.; Li, J.; Lv, Y.; Song, C. Research Progress on Graphene Oxide (GO)/Chitosan (CS) Multifunctional Nanocomposites for Drug Delivery. Inorganics 2025, 13, 98. [Google Scholar] [CrossRef]
- Bai, R.G.; Muthoosamy, K.; Shipton, F.N.; Manickam, S. Acoustic Cavitation Induced Generation of Stabilizer-Free, Extremely Stable Reduced Graphene Oxide Nanodispersion for Efficient Delivery of Paclitaxel in Cancer Cells. Ultrason. Sonochem. 2017, 36, 129–138. [Google Scholar] [CrossRef]
- Shariare, M.H.; Masum, A.-A.; Alshehri, S.; Alanazi, F.K.; Uddin, J.; Kazi, M. Preparation and Optimization of PEGylated Nano Graphene Oxide-Based Delivery System for Drugs with Different Molecular Structures Using Design of Experiment (DoE). Molecules 2021, 26, 1457. [Google Scholar] [CrossRef]
- Mi, X.; Wang, X.; Xu, C.; Zhang, Y.; Tan, X.; Gao, J.; Liu, Y. Alginate Microspheres Prepared by Ionic Crosslinking of Pickering Alginate Emulsions. J. Biomater. Sci. Polym. Ed. 2019, 30, 1083–1096. [Google Scholar] [CrossRef]
- Wei, L.; Lu, Z.; Ji, X.; Jiang, Y.; Ma, L. Self-Assembly of Hollow Graphene Oxide Microcapsules Directed by Cavitation for Loading Hydrophobic Drugs. ACS Appl. Mater. Interfaces 2021, 13, 2988–2996. [Google Scholar] [CrossRef]
- Kamkar, M.; Erfanian, E.; Bazazi, P.; Ghaffarkhah, A.; Sharif, F.; Xie, G.; Kannan, A.; Arjmand, M.; Hejazi, S.H.; Russell, T.P.; et al. Interfacial Assembly of Graphene Oxide: From Super Elastic Interfaces to Liquid-in-Liquid Printing. Adv. Mater. Interfaces 2022, 9, 2101659. [Google Scholar] [CrossRef]
- Xu, Q.; Chen, H.; Zhu, J.; Li, X.; Yang, J.; Deng, X.; Yuan, Q. Graphene Oxide Nanosheets Conjugated PEG-Glu-Lys-Glu Copolymer Drug Delivery System Improves Drug-Loading Rates and Enables Reduction-Sensitive Drug Release and Drug Tracking. J. Biomater. Sci. Polym. Ed. 2024, 35, 330–344. [Google Scholar] [CrossRef]
- Baneshi, M.; Dadfarnia, S.; Haji Shabani, A.M.; Sabbagh, S.K.; Bardania, H. AS1411 Aptamer-Functionalized Graphene Oxide-Based Nano-Carrier for Active-Target and pH-Sensitive Delivery of Curcumin. J. Iran. Chem. Soc. 2022, 19, 2367–2376. [Google Scholar] [CrossRef]
- Morani, D.O.; Patil, P.O. Formulation and Evaluation of Hyaluronic Acid and Adipic Acid Dihydrazide Modified Graphene Quantum Dot-Based Nanotherapeutics for Paclitaxel-Targeted Delivery in Breast Cancer. Futur. J. Pharm. Sci. 2025, 11, 7. [Google Scholar] [CrossRef]
- Xu, Z.; Zhu, S.; Wang, M.; Li, Y.; Shi, P.; Huang, X. Delivery of Paclitaxel Using PEGylated Graphene Oxide as a Nanocarrier. ACS Appl. Mater. Interfaces 2015, 7, 1355–1363. [Google Scholar] [CrossRef]
- Pereira De Sousa, I.; Buttenhauser, K.; Suchaoin, W.; Partenhauser, A.; Perrone, M.; Matuszczak, B.; Bernkop-Schnürch, A. Thiolated Graphene Oxide as Promising Mucoadhesive Carrier for Hydrophobic Drugs. Int. J. Pharm. 2016, 509, 360–367. [Google Scholar] [CrossRef]
- Matiyani, M.; Rana, A.; Pal, M.; Dokwal, S.; Sahoo, N.G. Polyamidoamine Dendrimer Decorated Graphene Oxide as a pH-Sensitive Nanocarrier for the Delivery of Hydrophobic Anticancer Drug Quercetin: A Remedy for Breast Cancer. J. Pharm. Pharmacol. 2023, 75, 859–872. [Google Scholar] [CrossRef]
- Gong, P.; Zhou, Y.; Li, H.; Zhang, J.; Wu, Y.; Zheng, P.; Jiang, Y. Theoretical Study on the Aggregation and Adsorption Behaviors of Anticancer Drug Molecules on Graphene/Graphene Oxide Surface. Molecules 2022, 27, 6742. [Google Scholar] [CrossRef]
- Leganés, J.; Sánchez-Migallón, A.; Merino, S.; Vázquez, E. Stimuli-Responsive Graphene-Based Hydrogel Driven by Disruption of Triazine Hydrophobic Interactions. Nanoscale 2020, 12, 7072–7081. [Google Scholar] [CrossRef]
- Lu, J.; Zhang, A.; Zhang, F.; Linhardt, R.J.; Zhu, Z.; Yang, Y.; Zhang, T.; Lin, Z.; Zhang, S.; Zhao, H.; et al. Ganoderenic Acid D-Loaded Functionalized Graphene Oxide-Based Carrier for Active Targeting Therapy of Cervical Carcinoma. Biomed. Pharmacother. 2023, 164, 114947. [Google Scholar] [CrossRef]
- Khoee, S.; Sadeghi, A. An NIR-Triggered Drug Release and Highly Efficient Photodynamic Therapy from PCL/PNIPAm/Porphyrin Modified Graphene Oxide Nanoparticles with the Janus Morphology. RSC Adv. 2019, 9, 39780–39792. [Google Scholar] [CrossRef]
- Verma, A.; Sharma, G.; Wang, T.; Kumar, A.; Dhiman, P.; Verma, Y.; Bhaskaralingam, A.; García-Penas, A. Graphene Oxide/Chitosan Hydrogels for Removal of Antibiotics. Environ. Technol. 2025, 77, 108518. [Google Scholar] [CrossRef]
- Pourjavadi, A.; Asgari, S.; Hosseini, S.H. Graphene Oxide Functionalized with Oxygen-Rich Polymers as a pH-Sensitive Carrier for Co-Delivery of Hydrophobic and Hydrophilic Drugs. J. Drug Deliv. Sci. Technol. 2020, 56, 101542. [Google Scholar] [CrossRef]
- Zhivkov, A.M.; Popov, T.T.; Hristova, S.H. Composite Hydrogels with Included Solid-State Nanoparticles Bearing Anticancer Chemotherapeutics. Gels 2023, 9, 421. [Google Scholar] [CrossRef]
- Li, F.; Zhang, Z.; Wang, X.; Yin, X.; Fu, M.; Qin, T.; Ji, X.; Yang, G.; Sun, S. A Physical Crosslinked pH-Sensitive Hydrogel Based on Hemicellulose/Graphene Oxide for Controlled Oral Drug Delivery. Int. J. Biol. Macromol. 2025, 289, 138875. [Google Scholar] [CrossRef]
- Das, P.N.; Raj, K.G. Chitosan Coated Graphene Oxide Incorporated Sodium Alginate Hydrogel Beads for the Controlled Release of Amoxicillin. Int. J. Biol. Macromol. 2024, 254, 127837. [Google Scholar] [CrossRef]
- Amiryaghoubi, N.; Pesyan, N.N.; Fathi, M.; Omidi, Y. Injectable Thermosensitive Hybrid Hydrogel Containing Graphene Oxide and Chitosan as Dental Pulp Stem Cells Scaffold for Bone Tissue Engineering. Int. J. Biol. Macromol. 2020, 162, 1338–1357. [Google Scholar] [CrossRef]
- Wu, J.; Qin, Z.; Jiang, X.; Fang, D.; Lu, Z.; Zheng, L.; Zhao, J. ROS-Responsive PPGF Nanofiber Membrane as a Drug Delivery System for Long-Term Drug Release in Attenuation of Osteoarthritis. NPJ Regen. Med. 2022, 7, 66. [Google Scholar] [CrossRef]
- Jariya, S.I.; Babu, A.A.; Narayanan, T.S.; Vellaichamy, E.; Ravichandran, K. Development of a Novel Smart Carrier for Drug Delivery: Ciprofloxacin Loaded Vaterite/Reduced Graphene Oxide/PCL Composite Coating on TiO2 Nanotube Coated Titanium. Ceram. Int. 2022, 48, 9579–9594. [Google Scholar] [CrossRef]
- Zhao, Z.; Gao, J.; Cai, W.; Li, J.; Kong, Y.; Zhou, M. Synthesis of Oxidized Carboxymethyl Cellulose/Chitosan Hydrogels Doped with Graphene Oxide for pH- and NIR-Responsive Drug Delivery. Eur. Polym. J. 2023, 199, 112437. [Google Scholar] [CrossRef]
- Pandey, H.; Parashar, V.; Parashar, R.; Prakash, R.; Ramteke, P.W.; Pandey, A.C. Controlled Drug Release Characteristics and Enhanced Antibacterial Effect of Graphene Nanosheets Containing Gentamicin Sulfate. Nanoscale 2011, 3, 4104. [Google Scholar] [CrossRef]
- Wu, X.; Zhou, J.; Liu, Z.; Shao, W. Gentamicin Sulfate Grafted Magnetic GO Nanohybrids with Excellent Antibacterial Properties and Recyclability. Nanomaterials 2023, 13, 1416. [Google Scholar] [CrossRef]
- Matulewicz, K.; Kaźmierski, Ł.; Wiśniewski, M.; Roszkowski, S.; Roszkowski, K.; Kowalczyk, O.; Roy, A.; Tylkowski, B.; Bajek, A. Ciprofloxacin and Graphene Oxide Combination—New Face of a Known Drug. Materials 2020, 13, 4224. [Google Scholar] [CrossRef]
- Yang, S.; Zhang, X.; Zhang, D. Electrospun Chitosan/Poly (Vinyl Alcohol)/Graphene Oxide Nanofibrous Membrane with Ciprofloxacin Antibiotic Drug for Potential Wound Dressing Application. IJMS 2019, 20, 4395. [Google Scholar] [CrossRef]
- Sarkar, K.; Chatterjee, A.; Bankura, B.; Bank, S.; Paul, N.; Chatterjee, S.; Das, A.; Dutta, K.; Chakraborty, S.; De, S.; et al. Efficacy of Pegylated Graphene Oxide Quantum Dots as a Nanoconjugate Sustained Release Metformin Delivery System in in Vitro Insulin Resistance Model. PLoS ONE 2024, 19, e0307166. [Google Scholar] [CrossRef]
- GV, Y.D.; Prabhu, A.; Anil, S.; Venkatesan, J. Preparation and Characterization of Dexamethasone Loaded Sodium Alginate-Graphene Oxide Microspheres for Bone Tissue Engineering. J. Drug Deliv. Sci. Technol. 2021, 64, 102624. [Google Scholar] [CrossRef]
- Islam, M.S.; Mitra, S. Development of Nano Structured Graphene Oxide Incorporated Dexamethasone with Enhanced Dissolution. Colloid Interface Sci. Commun. 2022, 47, 100599. [Google Scholar] [CrossRef]
- Ouyang, L.; Qi, M.; Wang, S.; Tu, S.; Li, B.; Deng, Y.; Yang, W. Osteogenesis and Antibacterial Activity of Graphene Oxide and Dexamethasone Coatings on Porous Polyetheretherketone via Polydopamine-Assisted Chemistry. Coatings 2018, 8, 203. [Google Scholar] [CrossRef]
- Yanikoglu, R.; Karakas, C.Y.; Ciftci, F.; Insel, M.A.; Karavelioglu, Z.; Varol, R.; Yilmaz, A.; Cakir, R.; Uvet, H.; Ustundag, C.B. Development of Graphene Oxide-Based Anticancer Drug Combination Functionalized with Folic Acid as Nanocarrier for Targeted Delivery of Methotrexate. Pharmaceutics 2024, 16, 837. [Google Scholar] [CrossRef]
- Yaghoubi, F.; Naghib, S.M.; Motlagh, N.S.H.; Haghiralsadat, F.; Jaliani, H.Z.; Tofighi, D.; Moradi, A. Multiresponsive Carboxylated Graphene Oxide-Grafted Aptamer as a Multifunctional Nanocarrier for Targeted Delivery of Chemotherapeutics and Bioactive Compounds in Cancer Therapy. Nanotechnol. Rev. 2021, 10, 1838–1852. [Google Scholar] [CrossRef]
- Tabasi, A.; Noorbakhsh, A.; Sharifi, E. Reduced Graphene Oxide-Chitosan-Aptamer Interface as New Platform for Ultrasensitive Detection of Human Epidermal Growth Factor Receptor 2. Biosens. Bioelectron. 2017, 95, 117–123. [Google Scholar] [CrossRef]
- Campora, S.; Mauro, N.; Griffiths, P.; Giammona, G.; Ghersi, G. Graphene Nanosystems as Support in Sirna Delivery. Chem. Eng. Trans. 2018, 64, 415–420. [Google Scholar] [CrossRef]
- Qin, H.; Ji, Y.; Li, G.; Xu, X.; Zhang, C.; Zhong, W.; Xu, S.; Yin, Y.; Song, J. MicroRNA-29b/Graphene Oxide–Polyethyleneglycol–Polyethylenimine Complex Incorporated within Chitosan Hydrogel Promotes Osteogenesis. Front. Chem. 2022, 10, 958561. [Google Scholar] [CrossRef]
- Ali, A.; Saroj, S.; Saha, S.; Gupta, S.K.; Rakshit, T.; Pal, S. Glucose-Responsive Chitosan Nanoparticle/Poly(Vinyl Alcohol) Hydrogels for Sustained Insulin Release In Vivo. ACS Appl. Mater. Interfaces 2023, 15, 32240–32250. [Google Scholar] [CrossRef]
- Hao, Z.; Zhu, Y.; Wang, X.; Rotti, P.G.; DiMarco, C.; Tyler, S.R.; Zhao, X.; Engelhardt, J.F.; Hone, J.; Lin, Q. Real-Time Monitoring of Insulin Using a Graphene Field-Effect Transistor Aptameric Nanosensor. ACS Appl. Mater. Interfaces 2017, 9, 27504–27511. [Google Scholar] [CrossRef]
- Lee, H.; Kim, J.; Lee, J.; Park, H.; Park, Y.; Jung, S.; Lim, J.; Choi, H.C.; Kim, W.J. In Vivo Self-Degradable Graphene Nanomedicine Operated by DNAzyme and Photo-Switch for Controlled Anticancer Therapy. Biomaterials 2020, 263, 120402. [Google Scholar] [CrossRef]
- Zhang, Q.; Pan, Y.; Pan, J.; Wang, Z.; Lu, R.; Sun, J.; Feng, J. Differential Insulin Response Characteristics of Graphene Oxide–Gold Nanoparticle Composites under Varied Synthesis Conditions. PLoS ONE 2025, 20, e0317126. [Google Scholar] [CrossRef]
- Turcheniuk, K.; Khanal, M.; Motorina, A.; Subramanian, P.; Barras, A.; Zaitsev, V.; Kuncser, V.; Leca, A.; Martoriati, A.; Cailliau, K.; et al. Insulin Loaded Iron Magnetic Nanoparticle–Graphene Oxide Composites: Synthesis, Characterization and Application for in Vivo Delivery of Insulin. RSC Adv. 2014, 4, 865–875. [Google Scholar] [CrossRef]
- Sun, X.; Yang, C.; Liu, W.; Lu, K.; Yin, H. Charge Modifications of Graphene Oxide Enhance the Inhibitory Effect on Insulin Amyloid Fibrillation Based on Electrostatic Interactions. Int. J. Biol. Macromol. 2023, 225, 1140–1151. [Google Scholar] [CrossRef]
- Baloch, S.G.; Shaikh, H.; Shah, S.; Memon, S.; Memon, A.A. Synthesis of an Insulin Intercalated Graphene Oxide Nanogel Composite: Evaluation of Its Release Profile and Stability for Oral Delivery of Insulin. Nanoscale Adv. 2022, 4, 2303–2312. [Google Scholar] [CrossRef]
- Belkhalfa, H.; Teodorescu, F.; Quéniat, G.; Coffinier, Y.; Dokhan, N.; Sam, S.; Abderrahmani, A.; Boukherroub, R.; Szunerits, S. Insulin Impregnated Reduced Graphene Oxide/Ni(OH)2 Thin Films for Electrochemical Insulin Release and Glucose Sensing. Sens. Actuators B Chem. 2016, 237, 693–701. [Google Scholar] [CrossRef]
- Pu, Y.; Zhu, Z.; Han, D.; Liu, H.; Liu, J.; Liao, J.; Zhang, K.; Tan, W. Insulin-Binding Aptamer-Conjugated Graphene Oxide for Insulin Detection. Analyst 2011, 136, 4138. [Google Scholar] [CrossRef]
- Li, L.; Luo, C.; Song, Z.; Reyes-Vargas, E.; Clayton, F.; Huang, J.; Jensen, P.; Chen, X. Association of Anti-HER2 Antibody with Graphene Oxide for Curative Treatment of Osteosarcoma. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 581–593. [Google Scholar] [CrossRef]
- Huang, S.; Li, Y.; Zhang, S.; Chen, Y.; Su, W.; Sanchez, D.J.; Mai, J.D.H.; Zhi, X.; Chen, H.; Ding, X. A Self-Assembled Graphene Oxide Adjuvant Induces Both Enhanced Humoral and Cellular Immune Responses in Influenza Vaccine. J. Control. Release 2024, 365, 716–728. [Google Scholar] [CrossRef]
- Li, S.; Sun, Y.; Du, M.; Shangguan, A.; Liu, Z.; Li, W.; Lina, L.; Liu, W.; Zhang, S.; Han, H. Graphene Oxide Nanoparticles Combined with CRISPR_Cas9 System Enable Efficient Inhibition of Pseudorabies Virus. Bioconjug. Chem. 2023, 34, 326–332. [Google Scholar] [CrossRef]
- Taghizadeh-Tabarsi, R.; Akbari-Birgani, S.; Amjadi, M.; Mohammadi, S.; Nikfarjam, N.; Kusamori, K. Aptamer-Guided Graphene Oxide Quantum Dots for Targeted Suicide Gene Therapy in an Organoid Model of Luminal Breast Cancer. Sci. Rep. 2024, 14, 24104. [Google Scholar] [CrossRef]
- Valimukhametova, A.R.; Lee, B.H.; Topkiran, U.C.; Gries, K.; Gonzalez-Rodriguez, R.; Coffer, J.L.; Akkaraju, G.; Naumov, A. Cancer Therapeutic siRNA Delivery and Imaging by Nitrogen- and Neodymium-Doped Graphene Quantum Dots. ACS Biomater. Sci. Eng. 2023, 9, 3425–3434. [Google Scholar] [CrossRef]
- Han, X.-M.; Zheng, K.-W.; Wang, R.-L.; Yue, S.-F.; Chen, J.; Zhao, Z.-W.; Song, F.; Su, Y.; Ma, Q. Functionalization and Optimization-Strategy of Graphene Oxide-Based Nanomaterials for Gene and Drug Delivery. Am. J. Transl. Res. 2020, 12, 1515–1534. [Google Scholar]
- Obireddy, S.R.; Ayyakannu, A.; Kasi, G.; Sridharan, B.; Lai, W.-F.; Viswanathan, K. Folate-Functionalized CS/rGO/NiO Nanocomposites as a Multifunctional Drug Carrier with Anti-Microbial, Target-Specific, and Stimuli-Responsive Capacities. Int. J. Nanomed. 2025, 20, 1965–1981. [Google Scholar] [CrossRef]
- Wang, Z.; Cheng, H.; Sheng, Y.; Chen, Z.; Zhu, X.; Ren, J.; Zhang, X.; Lv, L.; Zhang, H.; Zhou, J.; et al. Biofunctionalized Graphene Oxide Nanosheet for Amplifying Antitumor Therapy: Multimodal High Drug Encapsulation, Prolonged Hyperthermal Window, and Deep-Site Burst Drug Release. Biomaterials 2022, 287, 121629. [Google Scholar] [CrossRef]
- Li, Y.; Yang, Y.; Shao, Y.; Sun, Y.; Si, H.; Miao, J.; Xu, Y. Chitosan Functionalized Graphene Oxide Nanocomposites for Fluorescence Imaging of Apoptotic Processes and Targeted Anti-Inflammation Study. Carbohydr. Polym. 2021, 269, 118345. [Google Scholar] [CrossRef]
- Khakpour, E.; Salehi, S.; Naghib, S.M.; Ghorbanzadeh, S.; Zhang, W. Graphene-Based Nanomaterials for Stimuli-Sensitive Controlled Delivery of Therapeutic Molecules. Front. Bioeng. Biotechnol. 2023, 11, 1129768. [Google Scholar] [CrossRef]
- Chen, H.; Xing, L.; Guo, H.; Luo, C.; Zhang, X. Dual-Targeting SERS-Encoded Graphene Oxide Nanocarrier for Intracellular Co-Delivery of Doxorubicin and 9-Aminoacridine with Enhanced Combination Therapy. Analyst 2021, 146, 6893–6901. [Google Scholar] [CrossRef]
- Li, R.; Liu, C.; Wan, C.; Liu, T.; Zhang, R.; Du, J.; Wang, X.; Jiao, X.; Gao, R.; Li, B. A Targeted and pH-Responsive Nano-Graphene Oxide Nanoparticle Loaded with Doxorubicin for Synergetic Chemo-Photothermal Therapy of Oral Squamous Cell Carcinoma. Int. J. Nanomed. 2023, 18, 3309–3324. [Google Scholar] [CrossRef]
- Chuang, C.-C.; Lan, Y.-H.; Lu, Y.-J.; Weng, Y.-L.; Chen, J.-P. Targeted Delivery of Irinotecan and SLP2 shRNA with GRP-Conjugated Magnetic Graphene Oxide for Glioblastoma Treatment. Biomater. Sci. 2022, 10, 3201–3222. [Google Scholar] [CrossRef]
- Hussien, N.A.; Işıklan, N.; Türk, M. Aptamer-Functionalized Magnetic Graphene Oxide Nanocarrier for Targeted Drug Delivery of Paclitaxel. Mater. Chem. Phys. 2018, 211, 479–488. [Google Scholar] [CrossRef]
- Liu, B.; Yang, W.; Che, C.; Liu, J.; Si, M.; Gong, Z.; Gao, R.; Yang, G. A Targeted Nano Drug Delivery System of AS1411 Functionalized Graphene Oxide Based Composites. ChemistryOpen 2021, 10, 408–413. [Google Scholar] [CrossRef]
- Esmaeili, Y.; Zarrabi, A.; Mirahmadi-Zare, S.Z.; Bidram, E. Hierarchical Multifunctional Graphene Oxide Cancer Nanotheranostics Agent for Synchronous Switchable Fluorescence Imaging and Chemical Therapy. Microchim. Acta 2020, 187, 553. [Google Scholar] [CrossRef]
- Shahidi, M.; Haghiralsadat, B.F.; Abazari, O.; Hemati, M.; Dayati, P.; Jaliani, H.Z.; Motlagh, N.S.H.; Naghib, S.M.; Moradi, A. HB5 Aptamer-Tagged Graphene Oxide for Co-Delivery of Doxorubicin and Silibinin, and Highly Effective Combination Therapy in Breast Cancer. Cancer Nano 2023, 14, 59. [Google Scholar] [CrossRef]
- Li, R.; Gao, R.; Zhao, Y.; Zhang, F.; Wang, X.; Li, B.; Wang, L.; Ma, L.; Du, J. pH-Responsive Graphene Oxide Loaded with Targeted Peptide and Anticancer Drug for OSCC Therapy. Front. Oncol. 2022, 12, 930920. [Google Scholar] [CrossRef]
- Chen, S.; Zhang, S.; Wang, Y.; Yang, X.; Yang, H.; Cui, C. Anti-EpCAM Functionalized Graphene Oxide Vector for Tumor Targeted siRNA Delivery and Cancer Therapy. Asian J. Pharm. Sci. 2021, 16, 598–611. [Google Scholar] [CrossRef]
- Ma, K.; Li, W.; Zhu, G.; Chi, H.; Yin, Y.; Li, Y.; Zong, Y.; Guo, Z.; Wang, L.; Xu, W.; et al. PEGylated DOX-Coated Nano Graphene Oxide as pH-Responsive Multifunctional Nanocarrier for Targeted Drug Delivery. J. Drug Target. 2021, 29, 884–891. Available online: https://www.tandfonline.com/doi/abs/10.1080/1061186X.2021.1887200 (accessed on 24 April 2025). [CrossRef]
- Liu, L.; Wei, Y.; Zhai, S.; Chen, Q.; Xing, D. Dihydroartemisinin and Transferrin Dual-Dressed Nano-Graphene Oxide for a pH-Triggered Chemotherapy. Biomaterials 2015, 62, 35–46. [Google Scholar] [CrossRef]
- Qiu, J.; Zhang, R.; Li, J.; Sang, Y.; Tang, W.; Rivera Gil, P.; Liu, H. Fluorescent Graphene Quantum Dots as Traceable, pH-Sensitive Drug Delivery Systems. Int. J. Nanomed. 2015, 10, 6709–6724. [Google Scholar] [CrossRef]
- Liang, J.; Chen, B.; Hu, J.; Huang, Q.; Zhang, D.; Wan, J.; Hu, Z.; Wang, B. pH and Thermal Dual-Responsive Graphene Oxide Nanocomplexes for Targeted Drug Delivery and Photothermal-Chemo/Photodynamic Synergetic Therapy. ACS Appl. Bio Mater. 2019, 2, 5859–5871. [Google Scholar] [CrossRef]
- Ryu, K.; Park, J.; Kim, T. Effect of pH-Responsive Charge-Conversional Polymer Coating to Cationic Reduced Graphene Oxide Nanostructures for Tumor Microenvironment-Targeted Drug Delivery Systems. Nanomaterials 2019, 9, 1289. [Google Scholar] [CrossRef]
- Kavitha, T.; Haider Abdi, S.I.; Park, S.-Y. pH-Sensitive Nanocargo Based on Smart Polymer Functionalized Graphene Oxide for Site-Specific Drug Delivery. Phys. Chem. Chem. Phys. 2013, 15, 5176. [Google Scholar] [CrossRef]
- Jurić, B.; Ulamec, M.; Krušlin, B.; Balja, M.P. Breast Cancer Development Coordinated by Changes in Microenvironment Setting. Med. Res. Arch. 2024, 12. [Google Scholar] [CrossRef]
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Mehraj, U.; Dar, A.H.; Wani, N.A.; Mir, M.A. Tumor Microenvironment Promotes Breast Cancer Chemoresistance. Cancer Chemother. Pharmacol. 2021, 87, 147–158. [Google Scholar] [CrossRef]
- Almeida, C.F.; Correia-da-Silva, G.; Teixeira, N.; Amaral, C. Influence of Tumor Microenvironment on the Different Breast Cancer Subtypes and Applied Therapies. Biochem. Pharmacol. 2024, 223, 116178. [Google Scholar] [CrossRef]
- Lima-Sousa, R.; Melo, B.L.; Mendonça, A.G.; Correia, I.J.; de Melo-Diogo, D. Hyaluronic Acid-Functionalized Graphene-Based Nanohybrids for Targeted Breast Cancer Chemo-Photothermal Therapy. Int. J. Pharm. 2024, 651, 123763. [Google Scholar] [CrossRef]
- Jafarizad, A.; Aghanejad, A.; Sevim, M.; Metin, Ö.; Barar, J.; Omidi, Y.; Ekinci, D. Gold Nanoparticles and Reduced Graphene Oxide-Gold Nanoparticle Composite Materials as Covalent Drug Delivery Systems for Breast Cancer Treatment. ChemistrySelect 2017, 2, 6663–6672. [Google Scholar] [CrossRef]
- Cui, G.; Wu, J.; Lin, J.; Liu, W.; Chen, P.; Yu, M.; Zhou, D.; Yao, G. Graphene-Based Nanomaterials for Breast Cancer Treatment: Promising Therapeutic Strategies. J. Nanobiotechnol. 2021, 19, 211. [Google Scholar] [CrossRef]
- Hsieh, C.-J.; Chen, Y.-C.; Hsieh, P.-Y.; Liu, S.-R.; Wu, S.-P.; Hsieh, Y.-Z.; Hsu, H.-Y. Graphene Oxide Based Nanocarrier Combined with a pH-Sensitive Tracer: A Vehicle for Concurrent pH Sensing and pH-Responsive Oligonucleotide Delivery. ACS Appl. Mater. Interfaces 2015, 7, 11467–11475. [Google Scholar] [CrossRef]
- Karimi, S.; Namazi, H. Fe3O4@PEG-Coated Dendrimer Modified Graphene Oxide Nanocomposite as a pH-Sensitive Drug Carrier for Targeted Delivery of Doxorubicin. J. Alloys Compd. 2021, 879, 160426. [Google Scholar] [CrossRef]
- Rad, A.R.; Faramarzi, M.; Heydarinasab, A. Functionalized PEGylated Graphene Oxide with Folic Acid and Hydrazine as a New pH-Responsive Nanocarrier for Doxorubicin Delivery. Mater. Today Commun. 2024, 38, 108447. [Google Scholar] [CrossRef]
- Pramanik, N.; Ranganathan, S.; Rao, S.; Suneet, K.; Jain, S.; Rangarajan, A.; Jhunjhunwala, S. A Composite of Hyaluronic Acid-Modified Graphene Oxide and Iron Oxide Nanoparticles for Targeted Drug Delivery and Magnetothermal Therapy. ACS Omega 2019, 4, 9284–9293. [Google Scholar] [CrossRef]
- Alinejad, A.; Raissi, H.; Hashemzadeh, H. Development and Evaluation of a pH-Responsive and Water-Soluble Drug Delivery System Based on Smart Polymer Coating of Graphene Nanosheets: An in Silico Study. RSC Adv. 2020, 10, 31106–31114. [Google Scholar] [CrossRef]
- Angelopoulou, A.; Voulgari, E.; Diamanti, E.K.; Gournis, D.; Avgoustakis, K. Graphene Oxide Stabilized by PLA–PEG Copolymers for the Controlled Delivery of Paclitaxel. Eur. J. Pharm. Biopharm. 2015, 93, 18–26. [Google Scholar] [CrossRef]
- Liu, J.; Zhang, D.; Lian, S.; Zheng, J.; Li, B.; Li, T.; Jia, L. Redox-Responsive Hyaluronic Acid-Functionalized Graphene Oxide Nanosheets for Targeted Delivery of Water-Insoluble Cancer Drugs. Int. J. Nanomed. 2018, 13, 7457–7472. [Google Scholar] [CrossRef]
- Li, X.; Zhu, H.; Xing, Z.; Gong, T.; Li, M.; Su, D.; Liang, W.; Guo, R. Linear Maltodextrin Polymer–Folic Acid Modified Graphene Oxide Nanoparticles for Targeted Delivery and pH/Photothermal Sensitive Release of Hydrophobic Anticancer Drugs in Tumor Cells. New J. Chem. 2023, 47, 1117–1128. [Google Scholar] [CrossRef]
- Gong, P.; Ji, S.; Wang, J.; Dai, D.; Wang, F.; Tian, M.; Zhang, L.; Guo, F.; Liu, Z. Fluorescence-Switchable Ultrasmall Fluorinated Graphene Oxide with High near-Infrared Absorption for Controlled and Targeted Drug Delivery. Chem. Eng. J. 2018, 348, 438–446. [Google Scholar] [CrossRef]
- Wu, H.; Shi, H.; Wang, Y.; Jia, X.; Tang, C.; Zhang, J.; Yang, S. Hyaluronic Acid Conjugated Graphene Oxide for Targeted Drug Delivery. Carbon 2014, 69, 379–389. [Google Scholar] [CrossRef]
- Rodrigues, R.O.; Baldi, G.; Doumett, S.; Garcia-Hevia, L.; Gallo, J.; Bañobre-López, M.; Dražić, G.; Calhelha, R.C.; Ferreira, I.C.F.R.; Lima, R.; et al. Multifunctional Graphene-Based Magnetic Nanocarriers for Combined Hyperthermia and Dual Stimuli-Responsive Drug Delivery. Mater. Sci. Eng. C 2018, 93, 206–217. [Google Scholar] [CrossRef]
- Zhao, X.; Wei, Z.; Zhao, Z.; Miao, Y.; Qiu, Y.; Yang, W.; Jia, X.; Liu, Z.; Hou, H. Design and Development of Graphene Oxide Nanoparticle/Chitosan Hybrids Showing pH-Sensitive Surface Charge-Reversible Ability for Efficient Intracellular Doxorubicin Delivery. ACS Appl. Mater. Interfaces 2018, 10, 6608–6617. [Google Scholar] [CrossRef]
- He, Y.; Zhang, L.; Chen, Z.; Liang, Y.; Zhang, Y.; Bai, Y.; Zhang, J.; Li, Y. Enhanced Chemotherapy Efficacy by Co-Delivery of shABCG2 and Doxorubicin with a pH-Responsive Charge-Reversible Layered Graphene Oxide Nanocomplex. J. Mater. Chem. B 2015, 3, 6462–6472. [Google Scholar] [CrossRef]
- Pan, Q.; Lv, Y.; Williams, G.R.; Tao, L.; Yang, H.; Li, H.; Zhu, L. Lactobionic Acid and Carboxymethyl Chitosan Functionalized Graphene Oxide Nanocomposites as Targeted Anticancer Drug Delivery Systems. Carbohydr. Polym. 2016, 151, 812–820. [Google Scholar] [CrossRef]
- Song, E.; Han, W.; Li, C.; Cheng, D.; Li, L.; Liu, L.; Zhu, G.; Song, Y.; Tan, W. Hyaluronic Acid-Decorated Graphene Oxide Nanohybrids as Nanocarriers for Targeted and pH-Responsive Anticancer Drug Delivery. ACS Appl. Mater. Interfaces 2014, 6, 11882–11890. [Google Scholar] [CrossRef]
- Zhang, B.; Yang, X.; Wang, Y.; Zhai, G. Heparin Modified Graphene Oxide for pH-Sensitive Sustained Release of Doxorubicin Hydrochloride. Mater. Sci. Eng. C 2017, 75, 198–206. [Google Scholar] [CrossRef]
- Alemi, F.; Maleki, M.; Mir, M.; Ebrahimi-Kalan, A.; Zarei, M.; Yousefi, B.; Rashtchizadeh, N. Synthesis, Characterization, and Evaluation of pH-Sensitive Doxorubicin-Loaded Functionalized Graphene Oxide in Osteosarcoma Cells. Bioimpacts 2022, 13, 207. [Google Scholar] [CrossRef] [PubMed]
- Dong, L.; Shi, C.; Guo, L.; Yang, T.; Sun, Y.; Cui, X. Fabrication of Redox and pH Dual-Responsive Magnetic Graphene Oxide Microcapsules via Sonochemical Method. Ultrason. Sonochem. 2017, 36, 437–445. [Google Scholar] [CrossRef]
- Wang, K.; Wang, L.; Chen, L.; Peng, C.; Luo, B.; Mo, J.; Chen, W. Intranasal Administration of Dauricine Loaded on Graphene Oxide: Multi-Target Therapy for Alzheimer’s Disease. Drug Deliv. 2021, 28, 580–593. [Google Scholar] [CrossRef]
- Mohebichamkhorami, F.; Faizi, M.; Mahmoudifard, M.; Hajikarim-Hamedani, A.; Mohseni, S.S.; Heidari, A.; Ghane, Y.; Khoramjouy, M.; Khayati, M.; Ghasemi, R.; et al. Microfluidic Synthesis of Ultrasmall Chitosan/Graphene Quantum Dots Particles for Intranasal Delivery in Alzheimer’s Disease Treatment. Small 2023, 19, 2207626. [Google Scholar] [CrossRef]
- Tak, K.; Sharma, R.; Dave, V.; Jain, S.; Sharma, S. Clitoria Ternatea Mediated Synthesis of Graphene Quantum Dots for the Treatment of Alzheimer’s Disease. ACS Chem. Neurosci. 2020, 11, 3741–3748. [Google Scholar] [CrossRef]
- Huang, A.; Zhang, L.; Li, W.; Ma, Z.; Shuo, S.; Yao, T. Controlled Fluorescence Quenching by Antibody-Conjugated Graphene Oxide to Measure Tau Protein. R. Soc. Open Sci. 2018, 5, 171808. [Google Scholar] [CrossRef]
- Benítez-Martínez, S.; Caballero-Díaz, E.; Valcárcel, M. Development of a Biosensing System for Tacrine Based on Nitrogen-Doped Graphene Quantum Dots and Acetylcholinesterase. Analyst 2016, 141, 2688–2695. [Google Scholar] [CrossRef]
- Sharma, S.; Singh, N.; Nepovimova, E.; Korabecny, J.; Kuca, K.; Satnami, M.L.; Ghosh, K.K. Interaction of Synthesized Nitrogen Enriched Graphene Quantum Dots with Novel Anti-Alzheimer’s Drugs: Spectroscopic Insights. J. Biomol. Struct. Dyn. 2020, 38, 1822–1837. [Google Scholar] [CrossRef]
- Xiong, S.; Luo, J.; Wang, Q.; Li, Z.; Li, J.; Liu, Q.; Gao, L.; Fang, S.; Li, Y.; Pan, H.; et al. Targeted Graphene Oxide for Drug Delivery as a Therapeutic Nanoplatform against Parkinson’s Disease. Biomater. Sci. 2021, 9, 1705–1715. [Google Scholar] [CrossRef]
- González-Mayorga, A.; López-Dolado, E.; Gutiérrez, M.C.; Collazos-Castro, J.E.; Ferrer, M.L.; del Monte, F.; Serrano, M.C. Favorable Biological Responses of Neural Cells and Tissue Interacting with Graphene Oxide Microfibers. ACS Omega 2017, 2, 8253–8263. [Google Scholar] [CrossRef]
- Xiong, W.; Wang, X.; Guan, H.; Kong, F.; Xiao, Z.; Jing, Y.; Cai, L.; Hou, H.; Qiu, X.; Wang, L. A Vascularized Conductive Elastic Patch for the Repair of Infarcted Myocardium through Functional Vascular Anastomoses and Electrical Integration. Adv. Funct. Mater. 2022, 32, 2111273. [Google Scholar] [CrossRef]
- Li, R.; Duan, N.; Guo, J. Development and Novel Design of Clustery Graphene Oxide Formed Conductive Silk Hydrogel Cell Vesicle to Repair Myocardial Infarction: Investigation of Its Biological Activity for Cell Delivery Applications. J. Drug Deliv. Sci. Technol. 2020, 60, 102001. [Google Scholar]
- Yadav, S.K.; Das, S.; Lincon, A.; Saha, S.; BoseDasgupta, S.; Ray, S.K.; Das, S. Gelatin-Decorated Graphene Oxide: A Nanocarrier for Delivering pH-Responsive Drug for Improving Therapeutic Efficacy against Atherosclerotic Plaque. Int. J. Pharm. 2024, 651, 123737. [Google Scholar] [CrossRef]
- Kaya, D.; Küçükada, K.; Alemdar, N. Modeling the Drug Release from Reduced Graphene Oxide-Reinforced Hyaluronic Acid/Gelatin/Poly(Ethylene Oxide) Polymeric Films. Carbohydr. Polym. 2019, 215, 189–197. [Google Scholar] [CrossRef]
- Tabish, T.A.; Hussain, M.Z.; Zhu, Y.; Xu, J.; Huang, W.E.; Diotallevi, M.; Narayan, R.J.; Crabtree, M.J.; Khademhosseini, A.; Winyard, P.G.; et al. Synthesis and Characterization of Amine-Functionalized Graphene as a Nitric Oxide-Generating Coating for Vascular Stents. Appl. Phys. Rev. 2024, 11, 031422. [Google Scholar] [CrossRef]
- Tabish, T.A.; Hussain, M.Z.; Zervou, S.; Myers, W.K.; Tu, W.; Xu, J.; Beer, I.; Huang, W.E.; Chandrawati, R.; Crabtree, M.J.; et al. S-Nitrosocysteamine-Functionalised Porous Graphene Oxide Nanosheets as Nitric Oxide Delivery Vehicles for Cardiovascular Applications. Redox Biol. 2024, 72, 103144. [Google Scholar] [CrossRef]
- Darabi, N.H.; Kalaee, M.; Mazinani, S.; Khajavi, R. GO/AgNW Aided Sustained Release of Ciprofloxacin Loaded in Starch/PVA Nanocomposite Mats for Wound Dressings Application. Int. J. Biol. Macromol. 2024, 266, 130977. [Google Scholar] [CrossRef]
- Bhattacharya, P.; Sengupta, I.; Dey, A.; Chakraborty, S.; Neogi, S. Antibacterial Effect of Ciprofloxacin Loaded Reduced Graphene Oxide Nanosheets against Pseudomonas aeruginosa Strain. Colloid Interface Sci. Commun. 2021, 40, 100344. [Google Scholar] [CrossRef]
- Sun, H.; Zhang, L.; Xia, W.; Chen, L.; Xu, Z.; Zhang, W. Fabrication of Graphene Oxide-Modified Chitosan for Controlled Release of Dexamethasone Phosphate. Appl. Phys. A 2016, 122, 632. [Google Scholar] [CrossRef]
- Ni, G.; Wang, Y.; Wu, X.; Wang, X.; Chen, S.; Liu, X. Graphene Oxide Absorbed Anti-IL10R Antibodies Enhance LPS Induced Immune Responses in Vitro and in Vivo. Immunol. Lett. 2012, 148, 126–132. [Google Scholar] [CrossRef]
- Teodorescu, F.; Oz, Y.; Quéniat, G.; Abderrahmani, A.; Foulon, C.; Lecoeur, M.; Sanyal, R.; Sanyal, A.; Boukherroub, R.; Szunerits, S. Photothermally Triggered On-Demand Insulin Release from Reduced Graphene Oxide Modified Hydrogels. J. Control. Release 2017, 246, 164–173. [Google Scholar] [CrossRef]
- Huang, W.B.; Huang, X.Y.; Mu, R.; Pei, T.; Guo, C.P.; Bai, X.Q. Insulin-Loaded Graphene Oxide Microparticles: Synthesis, Characterization, Insulin Delivery, and Physical Interactions Analysis. Russ. J. Inorg. Chem. 2024, 69, 1970–1978. [Google Scholar] [CrossRef]
- Soganci, K.; Bingol, H.; Zor, E. Simply Patterned Reduced Graphene Oxide as an Effective Biosensor Platform for Glucose Determination. J. Electroanal. Chem. 2021, 880, 114801. [Google Scholar] [CrossRef]
- Zhang, Y.; Chen, J.; Wang, H.; Gao, X.; Niu, B.; Li, W.; Wang, H. Electrochemical Biosensor Based on Copper Sulfide/Reduced Graphene Oxide/Glucose Oxidase Construct for Glucose Detection. Anal. Biochem. 2025, 696, 115696. [Google Scholar] [CrossRef]
- Li, R.; Gao, R.; Wang, Y.; Liu, Z.; Xu, H.; Duan, A.; Zhang, F.; Ma, L. Gastrin Releasing Peptide Receptor Targeted Nano-Graphene Oxide for near-Infrared Fluorescence Imaging of Oral Squamous Cell Carcinoma. Sci. Rep. 2020, 10, 11434. [Google Scholar] [CrossRef]
- Kwak, S.-Y.; Yang, J.-K.; Jeon, S.-J.; Kim, H.-I.; Yim, J.; Kang, H.; Kyeong, S.; Lee, Y.-S.; Kim, J.-H. Luminescent Graphene Oxide with a Peptide-Quencher Complex for Optical Detection of Cell-Secreted Proteases by a Turn-On Response. Adv. Funct. Mater. 2014, 24, 5119–5128. [Google Scholar] [CrossRef]
- Park, R.; Kang, M.S.; Heo, G.; Shin, Y.C.; Han, D.-W.; Hong, S.W. Regulated Behavior in Living Cells with Highly Aligned Configurations on Nanowrinkled Graphene Oxide Substrates: Deep Learning Based on Interplay of Cellular Contact Guidance. ACS Nano 2024, 18, 1325–1344. [Google Scholar] [CrossRef]
- Chatterjee, N.; Eom, H.-J.; Choi, J. A Systems Toxicology Approach to the Surface Functionality Control of Graphene–Cell Interactions. Biomaterials 2014, 35, 1109–1127. [Google Scholar] [CrossRef]
- Matesanz, M.-C.; Vila, M.; Feito, M.-J.; Linares, J.; Gonçalves, G.; Vallet-Regi, M.; Marques, P.-A.A.P.; Portolés, M.-T. The Effects of Graphene Oxide Nanosheets Localized on F-Actin Filaments on Cell-Cycle Alterations. Biomaterials 2013, 34, 1562–1569. [Google Scholar] [CrossRef]
- Zhang, X.; Yin, J.; Peng, C.; Hu, W.; Zhu, Z.; Li, W.; Fan, C.; Huang, Q. Distribution and Biocompatibility Studies of Graphene Oxide in Mice after Intravenous Administration. Carbon 2011, 49, 986–995. [Google Scholar] [CrossRef]
- Liu, J.-H.; Wang, T.; Wang, H.; Gu, Y.; Xu, Y.; Tang, H.; Jia, G.; Liu, Y. Biocompatibility of Graphene Oxide Intravenously Administrated in Mice—Effects of Dose, Size and Exposure Protocols. Toxicol. Res. 2015, 4, 83–91. [Google Scholar] [CrossRef]
- Yang, K.; Gong, H.; Shi, X.; Wan, J.; Zhang, Y.; Liu, Z. In Vivo Biodistribution and Toxicology of Functionalized Nano-Graphene Oxide in Mice after Oral and Intraperitoneal Administration. Biomaterials 2013, 34, 2787–2795. [Google Scholar] [CrossRef]
- Li, B.; Yang, J.; Huang, Q.; Zhang, Y.; Peng, C.; Zhang, Y.; He, Y.; Shi, J.; Li, W.; Hu, J.; et al. Biodistribution and Pulmonary Toxicity of Intratracheally Instilled Graphene Oxide in Mice. NPG Asia Mater. 2013, 5, e44. [Google Scholar] [CrossRef]
- Duch, M.C.; Budinger, G.R.S.; Liang, Y.T.; Soberanes, S.; Urich, D.; Chiarella, S.E.; Campochiaro, L.A.; Gonzalez, A.; Chandel, N.S.; Hersam, M.C.; et al. Minimizing Oxidation and Stable Nanoscale Dispersion Improves the Biocompatibility of Graphene in the Lung. Nano Lett. 2011, 11, 5201–5207. [Google Scholar] [CrossRef]
- Liu, X.; Wang, Z.; Teng, C.; Wang, Z. Changes in Gut Microbiota and Metabolites of Mice with Intravenous Graphene Oxide-Induced Embryo Toxicity. Toxicol. Res. 2024, 40, 571–584. [Google Scholar] [CrossRef]
- Kim, Y.H.; Jo, M.S.; Kim, J.K.; Shin, J.H.; Baek, J.E.; Park, H.S.; An, H.J.; Lee, J.S.; Kim, B.W.; Kim, H.P.; et al. Short-Term Inhalation Study of Graphene Oxide Nanoplates. Nanotoxicology 2018, 12, 224–238. [Google Scholar] [CrossRef]
- Ding, Z.; Zhang, Z.; Ma, H.; Chen, Y. In Vitro Hemocompatibility and Toxic Mechanism of Graphene Oxide on Human Peripheral Blood T Lymphocytes and Serum Albumin. ACS Appl. Mater. Interfaces 2014, 6, 19797–19807. [Google Scholar] [CrossRef]
- Singh, S.K.; Singh, M.K.; Nayak, M.K.; Kumari, S.; Shrivastava, S.; Grácio, J.J.A.; Dash, D. Thrombus Inducing Property of Atomically Thin Graphene Oxide Sheets. ACS Nano 2011, 5, 4987–4996. [Google Scholar] [CrossRef]
- Sundeep, D.; Varadharaj, E.K. Development of Biocompatible and Water-Soluble Reduced Graphene Oxide Using Non-Toxic Reducing Agents for Biomedical Applications. Meet. Abstr. 2024, MA2024-01, 869. [Google Scholar] [CrossRef]
- Singh, S.K.; Singh, M.K.; Kulkarni, P.P.; Sonkar, V.K.; Grácio, J.J.A.; Dash, D. Amine-Modified Graphene: Thrombo-Protective Safer Alternative to Graphene Oxide for Biomedical Applications. ACS Nano 2012, 6, 2731–2740. [Google Scholar] [CrossRef]
- Das, S.; Singh, S.; Singh, V.; Joung, D.; Dowding, J.M.; Reid, D.; Anderson, J.; Zhai, L.; Khondaker, S.I.; Self, W.T.; et al. Oxygenated Functional Group Density on Graphene Oxide: Its Effect on Cell Toxicity. Part. Part. Syst. Charact. 2013, 30, 148–157. [Google Scholar] [CrossRef]
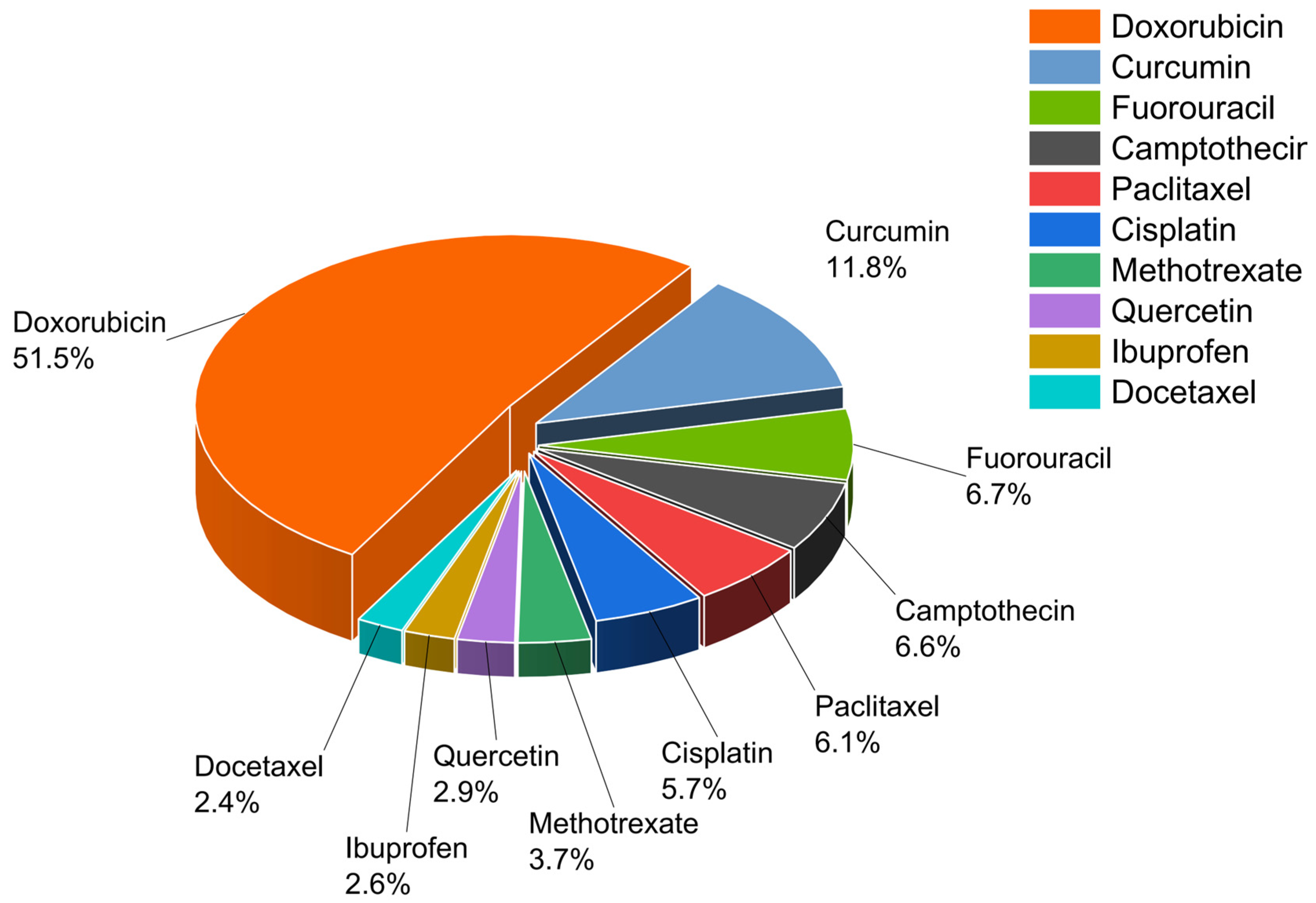
| Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|
| FA-GO | Hummer’s Method; 1070 nm; −29 mV | MTX, DOX | The conventional release of MTX is too rapid, with poor targeting and significant side effects. | Construction of a multifunctional nanoplatform with a clearly defined targeting mechanism and good biocompatibilit. | [72] |
| PAC@HA-ADH-GQDs System | GQDs were synthesized from citric acid. HA–ADH was prepared from HA, ADH, and EDC, HA–ADH–GQDs were formed in PBS; 23 ± 2.62 nm; −26.5 ± 1.32 mV | PTX | Free paclitaxel is poorly water-soluble, requires toxic solvents, causes side effects and releases prematurely in normal tissues. | Specific binding of HA to CD44 receptors enhances targeting; improves PTX anticancer activity. | [47] |
| GO | Modified Hummers’ method; Unavailable; About −30 mV | Oligonu-cleotide Drugs | Difficult to control drug release, lack of responsive mechanisms, oligonucl-eotide drugs are prone to degradation and have low delivery efficiency. | The system can deliver drugs while monitoring the surrounding pH environment in real time, demonstrating potential for “heranostic integration”. | [118] |
| GO-TD-Fe3O4@PEG System | Hummer’s Method; 144.21 nm; −51.1 mV | DOX | Conventional DOX has high toxicity, poor targeting, and nanoparticle aggregation issues. | Magnetic targeting capability, excellent biocompatibility and stability. | [119] |
| GO-PAMAM System | GO was made using a modified Hummer’s method, then linked to PAMAM dendrimer via EDC/NHS coupling; Unavailable; Unavailable | Quercetin | Poor solubility and low delivery efficiency of hydrophobic drugs; poor targeting and low drug utilization. | Developed a pH-sensitive smart nanocarrier; improved solubility and stability of hydrophobic drugs. | [50] |
| DOX@GNFH | Modified Hummer’s method for GO; FA and HZ-PEG conjugation via EDC/NHS amidation; 110 and 172 nm; Unavailable | DOX | Lack of tumor targeting, normal tissue easily affected; limited drug solubility and stability. | Constructed a multifunctional integrated nano delivery platform; FA-mediated active targeting. | [120] |
| MGO@APT Carrier | GO functionalized with β-CD and FA via CDI activation and covalent coupling; Unavailable; −20.4 mV | PTX | Poor water solubility of paclitaxel, rapid metabolic clearance; uncontrollable release, significant side effects. | Dual enhancement of targeting and imaging capabilities, efficient PTX loading and controlled release. | [99] |
| HA-GO/Fe3O4 Nanocomposite | GO was prepared by modified Hummers method, HA grafted via EDC/NHS coupling, and Fe3O4 nanoparticles added to form the composite; 166.8 ± 16.2 nm; −21.5 ± 2.25 mV | PTX, DOX | Limited effectiveness of single treatment modality; inability to deliver multiple drugs simultaneously. | Constructed a multifunctional synergistic treatment platform; combined drug co-delivery for synergistic therapy. | [121] |
| Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|
| Hollow Graphene Oxide Microcapsules | GO was made by the improved Hummers method, then freeze-dried after liquid nitrogen treatment to obtain hollow GOMs; 0.5–3.5 μm; −42.5 mV | Hydrophobic Anticancer Drugs | Low cellular uptake rate, poor membrane permeability, poor solubility of hydrophobic drugs. | Applicable to various hydrophobic drugs; high cellular uptake and rapid intracellular delivery capacity. | [43] |
| GO–PLA–PEG Nano System | PLA–PEG was synthesized by ring-opening polymerization and used to form NGO/PLA–PEG composites via sonication and centrifugation; 534.1 ± 9.24 nm; Unavailable | PTX | Poor water solubility of paclitaxel, requires toxic solubilizing agents. | Efficient PTX loading; enables controlled release. | [123] |
| NGO-SS-HA Nano Delivery System | Conjugation of hyaluronic acid to nano-graphene oxide via disulfide linkers; 125 nm; Unavailable | Gefitinib | Difficult delivery and low bioavailability of water-insoluble drugs; non-targeted release leads to significant side effects. | Constructed a GSH-responsive targeted delivery system; enables delivery of water-insoluble drugs. | [124] |
| Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|
| GO@LM-SP-FA System | GO@LM-SP-FA was prepared by EDC/NHS-mediated conjugation of LM-SP-FA to GO, followed by DOX loading via non-covalent interactions; 257 nm; +5.3 mV | DOX | Hydrophobic anticancer drugs are difficult to dissolve, with low delivery efficiency. | Constructed a dual-responsive (pH and photothermal) smart drug delivery system; possesses photothermal therapy potential. | [125] |
| FGO | Ultrasmall FGO was prepared by alkali activation, mild oxidation, and sonication, then functionalized with folic acid.; ~50 nm; Unavailable | Anticancer Drugs | Drug carriers lacking tracking capability; single-function carriers without responsive control. | Ultra-small material size; excellent fluorescence performance and switchable control capability. | [126] |
| GO-PEG-EGFR | Ultrasmall FGO (~50 nm) was prepared by alkali activation, oxidation, and folic acid conjugation.; 1000 nm; −30.72 mV | GAD | Natural anticancer compound GAD has poor water solubility and low bioavailability; traditional chemotherapy drugs have high toxicity and severe drug resistance issues. | Innovatively utilized natural anticancer compound GAD combined with functionalized GO as a delivery platform. | [53] |
| GO–HA | Prepared by EDC/NHS-mediated ADH amination of GO followed by hyaluronic acid conjugation; 40–350 nm; −34.2 mV | Anticancer Drugs | Poor carrier stability, prone to aggregation or rapid degradation in vivo; insufficient functional sites, making it difficult to load multiple drugs or functional factors simultaneously. | Introduction of amino functional groups to enhance targeting ability and increase the number of active sites, equipped with CD44 targeting capability. | [127] |
| Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|
| GYSMNP@PF127 System | GO–Fe3O4 synthesized via co-precipitation; PVP/PAA grafted via polymerization; 180.0 ± 25.4 nm; −36.8 ± 2.0 mV | DOX | Uncontrolled drug release, narrow therapeutic window; limited anticancer effect with single therapy. | Dual stimulus-responsive capability; magnetic targeting. | [128] |
| GON/CS/CS-DMMA System | Prepared by electrostatic self-assembly of GO with chitosan and DMMA-chitosan; 181 nm; −26.5 mV | DOX | Premature drug release in normal tissues causing side effects. | The drug delivery system features pH responsiveness and charge-switching capability. | [129] |
| GO–PEI–PEG/CS-Aco/shRNA | Fabricated via layer-by-layer self-assembly of functional polymers and therapeutic agents on GO nanosheets; 100–250 nm; 10.97 ± 0.72 mV | shABCG, DOX | Severe chemotherapy resistance in liver cancer; instability and delivery difficulty of siRNA/gene drugs. | Co-delivery of chemotherapy drugs and siRNA; layered structure enables simultaneous loading of both molecules. | [130] |
| LA–CMC–GO Nanocomposite System | GO prepared via modified Hummers’ method, functionalized with CMC, FI, and LA, acetylated; Unavailable; −40.1 ± 1.2 mV | DOX | Poor carrier water solubility, insufficient biocompatibility; premature drug release in non-target areas. | Developed a dual-functionalized graphene oxide nanocarrier; enhances anticancer efficacy while reducing systemic toxicity. | [131] |
| HA-Modified Graphene Oxide Nanocomposite (GO–HA) | DOX was noncovalently loaded onto GO and coated with ADH-modified hyaluronic acid to form HA–GO–DOX; ~250 nm; Unavailable | DOX | Premature drug release in normal tissues reduces therapeutic efficiency; lack of targeting leads to significant side effects. | Active targeting achieved Via HA recognition of CD44 receptors on tumor cell surfaces. | [132] |
| Heparin-Modified GO | GO was carboxylated, ADH-conjugated, heparin-modified, and loaded with DOX; 119.8 nm; −39 mV | DOX | Poor water solubility, Uncontrolled drug release. | Multi-functional integrated platform, Highly versatile and adaptable | [133] |
| Cancer Type | Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|---|
| OSCC | NPF@DOX. | DOX-loaded nano-graphene oxide was functionalized with PEG and conjugated with a FAP-targeting peptide; Unavailable; Unavailable | DOX | Limited efficacy of single treatment modality, prominent drug resistance issues. | Features dual therapeutic mechanisms: pH responsiveness and photothermal conversion. | [97] |
| OSCC | NGO-BBN-AF750 | NGO conjugated with BBN-AF750; via mixing and centrifugation; 0.5–5 μm; −16.6 mV | DOX | Premature drug release at normal pH, low efficiency. | Dual-functional platform design; significantly enhances anticancer activity of the drug in OSCC. | [103] |
| Osteosarcoma | pH-Responsive Functionalized GO Nano System | Prepared by TRIS modification of GO in DMF, hydroxyl activation under alkaline conditions, QIFO coupling, followed by DOX loading; Unavailable; Unavailable | DOX | Premature DOX release in normal tissues, significant side effects. | Simple structural design with clear functionality; complete material characterization providing a reliable reference for future studies. | [134] |
| Malignant Tumors with Acidic and Highly Reductive Microenvironment Characteristics | MGOMCs | Prepared by coupling cysteine-functionalized GO with oleic acid-modified Fe3O4 via high-intensity sonication; 2.0 μm; Unavailable | Hydrop-hobic Chemot-herapy Drugs, such as DOX, PTX | First to form microcapsule structure by polymer coating Fe3O4 with GO; possesses smart release capability. | [135] |
| Disease Type | Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|---|
| AD | GO | GO dispersion was stirred with Dau solution for 24 h, then ultrafiltered to remove free drug; 250.91 ± 15.16 nm; −19.8 ± 0.72 mV | Dauricine | Traditional oral dauricine has low absorption and is easily metabolized and inactivated. | First to load dauricine onto GO for intranasal delivery. | [136] |
| AD | CS–GQDs | Prepared by microfluidic electrostatic crosslinking of chitosan and graphene quantum dots with sodium tripolyphosphate; 10–20 nm; −96.3 mV | Conventional carriers have large particle sizes and non-specific biodistribution. | achieved non-invasive intranasal delivery to cross the BBB. | [137] | |
| AD | Clitoria ternatea GQDs | Prepared via one-step microwave-assisted synthesis from Clitoria ternatea flower extract; 10 ± 1.3 nm; −46 ± 0.4 mV | Traditional GQDs synthesis methods have high toxicity and are not environmentally friendly; neurotoxicity induced by β-amyloid is difficult to reverse. | Green synthesis of GQDs; exhibits strong antioxidant properties and biological safety. | [138] | |
| AD | Antibody Functionalized GO Fluorescent Probe Sensing System | Prepared by EDC/NHS-mediated covalent coupling of antibodies to carboxyl groups on graphene oxide; Unavailable; Unavailable | Lack of rapid and sensitive tau protein detection methods. | high selectivity and specificity. | [139] | |
| AD | N-GQDs | N-GQDs were synthesized hydrothermally from citric acid and ammonia, followed by pH adjustment and dialysis; 4.6 ± 0.78 nm; Unavailable | Tacrine | Traditional drug detection methods are complex, time-consuming, and costly. | Regulate N-GQDs fluorescence intensity using enzymatic reaction products. | [140] |
| AD | Nitrogen-doped Graphene Quantum Dots, N-GQDs | Microwave synthesis from glucose and ammonia, pH adjustment, dialysis; 5–10 nm; −1.06 mV | PC-25, PC-37, PC-48, Tacrine | Challenges in High-Throughput Screening and Enzyme Inhibition Efficiency Monitoring for Anti-AD Drugs. | Established a simple monitoring system for drug-enzyme interaction. | [141] |
| PD | GO | Prepared by loading puerarin onto GO, PEGylation, and covalent conjugation with lactoferrin via EDC/NHS coupling; 236.1 nm; −24.74 mV | PD drugs have difficulty crossing the blood–brain barrier. | traditional carriers lack neuroprotective function First application of the GO platform for PD treatment. | [142] | |
| PD | GO Sheet Materials with Different Microstruct-ures | One-step confined hydrothermal synthesis of rGO microfibers; Unavailable; Unavailable | High toxicity and uncertainty of nanomaterials in the nervous system; drug delivery platforms struggle to form effective interfaces with neural cells. | Demonstrated good biocompatibility of GO with the nervous system. | [143] |
| Disease Type | Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|---|
| Myocardial Infarction | Vascularized Conductive Elastic Patch | A vascularized conductive elastic scaffold was prepared by cryopolymerizing holey graphene oxide/polypyrrole in a poly(hydroxyethyl methacrylate) matrix; Unavailable; Unavailable | Existing myocardial repair methods cannot effectively guide angiogenesis. | The material is composed of conductive polymers, elastic biopolymers, and extracellular matrix composites, combining biocompatibility with functionality. | [144] | |
| Myocardial Infarction | Injectable Silk Fibroin-GO Hydrogel | The clustery GO–silk fibroin composite was obtained by self-assembly, freeze-drying, and crosslinking; Unavailable; Unavailable | Low cell viability in cell delivery | GO was first combined with silk fibroin to create a conductive injectable hydrogel, offering a safe and efficient delivery system for myocardial infarction treatment. | [145] | |
| Atheroscle- rosis | Gelatin-Functionalized GO Nano Platform | Obtained by gelatin–GO conjugation and π–π loading of atorvastatin; 250–400 nm; −17.67 ± 1.9 mV | Atorvastatin | Traditional drugs face low bioavailability and high-dose side effects. | Innovative drug delivery system design; effectively promotes lipid efflux. | [146] |
| Hypertension and Heart Disease | HyA/Gel/PEO | Electroconductive HyA/Gel/PEO films with RGO were prepared by solvent casting with EDC crosslinking; Unavailable; Unavailable | Irbesartan | Uncontrollable or inaccurate drug release process. | Established a mathematical model to predict drug release behavior in rGO-hydrogel systems. | [147] |
| Coronary Heart Disease | PEI-PEG@GO | Prepared by EDC-mediated amide coupling of PEI and PEG to GO, then dip-coated onto PLA stents; Unavailable; Unavailable | S-nitrosoglutathione S-nitroso-N-acetylpenicillamine | Short-acting and uncontrollable NO release; drug-eluting stents delay endothelial repair, leading to thrombosis. | First development of amine-functionalized graphene coating. | [148] |
| Complicatio-ns Caused by Atheroscler-osis | SNO-Cys@PGO | EDC/NHS-mediated cysteamine grafting onto porous GO, followed by NO loading with acidified nitrite; Unavailable; Unavailable | NO | Poor NO release sustainability; partial carrier degradation remains incomplete. | Dual-mechanism controlled NO release. | [149] |
| Disease Type | Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|---|
| infected wounds | GO/AgNW-assisted Starch/PVA nanocomposite film | GO-AgNWs was obtained by ultrasonication of GO and AgNWs for 20 min; 98.9 nm and 773.8 nm; −0.6 mV | CIP | Traditional dressings lack controlled release capability, exhibit poor drug uniformity, and allow rapid diffusion. | A multifunctional dressing platform was constructed with enhanced mechanical strength. | [150] |
| Chronic or Acute Wound Infection | Chitosan/PVA/GO Electrospun Nanofiber Membrane | CS/PVA/GO/CIP nanofibrous membrane was prepared by electrospinning a mixed solution of CS, PVA, GO, and ciprofloxacin; Unavailable; Unavailable | CIP | Traditional wound dressings have short drug release duration and limited antibacterial time. | The incorporation of GO enhances mechanical strength and antibacterial synergistic effects. | [67] |
| Bacterial Infection | Methanol-Derived Graphene | Gentamicin was loaded onto methanol-derived graphene by sonication and stirring at pH 7; Unavailable; Unavailable | Gentamicin Sulfate | Improved controlled-release efficiency of the antibiotic gentamicin sulfate, reduced burst release. | high drug loading capacity up to 2.57 mg/mg; clear drug release mechanism. | [64] |
| Bacterial Infection | Fe3O4@GO Magnetic GO Nanocomposite | MGO was synthesized by loading Fe3O4 NPs onto GO; then GS was grafted via amidation to obtain GS-MGO nanohybrids; Unavailable; Unavailable | Gentamicin Sulfate | Antibiotics easily inactivate, insufficient sustained release. | First exploration of a high-stability strategy combining antibiotics with GO Via covalent grafting. | [65] |
| Pseudom-onas aerugino-sa Infection | rGO | Prepared by mixing rGO with ciprofloxacin hydrochloride; 120 nm; +7.5 mV | CIP | CIP exhibits strong resistance and low efficiency in bacterial infection treatment. | Constructed an efficient antibacterial nanosystem; rGO physically damages bacterial membranes, synergizing with CIP’s antibacterial mechanism to enhance overall efficacy. | [151] |
| Bone Tissue Injury or ImplantAssociat-ed Infection | GO/Polydopamine Functionalized Porous PEEK Surface Coating System DEX | SP plates were coated with polydopamine and modified with GO and Dex-loaded liposomes; Unavailable; Unavailable | DEX | Traditional orthopedic implant materials have poor bioactivity and are prone to infection. | The modified layer provides dual biological functions of antibacterial and osteogenic activity. | [71] |
| Disease Type | Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|---|
| Pneumonia | nGO | Prepared by ultrasonically dispersing GO in water, mixing with dexamethasone for adsorption, and drying; 150 nm; +20 mV to +30 mV | DEX | DEX as a hydrophobic drug, has low solubility, poor absorption, and low bioavailability in the body. | Improved the bioavailability of orally administered drugs; maintained the crystalline properties of the drug. | [70] |
| Inflammation | GO–CS Material | Prepared by dispersing GO in chitosan solution, mixing to form a homogeneous composite, and drying; Unavailable; Unavailable | DEX-P | Enhanced the carrier’s controlled-release performance, reduced toxicity, and prolonged therapeutic efficacy. | voiding high toxicity and extending drug efficacy Significantly suppressed burst drug release, demonstrating good cell compatibility and thermal stability. | [152] |
| Chronic Viral Infection | GO | Prepared by mixing ~20 nm graphene oxide with rat anti-mouse IL-10R antibodies, then washing with PBS; Unavailable; Unavailable | Anti-IL-10 Receptor Antibo-dy | Poor antibody stability, short release duration, insufficient targeting. | First demonstration of GO as a drug delivery platform for anti-IL10R Ab; strong sustained-release capability. | [153] |
| Carrier | Method; Size; and Zeta Potential | Drug | Addressed Drug Delivery Issue | Study Highlight | Reference |
|---|---|---|---|---|---|
| PEGDMA-rGO | Prepared by incorporating reduced graphene oxide into PEGDMA hydrogel; an area of 1 cm2; Unavailable | Insulin | Conventional insulin delivery systems face issues with sustained release and lack of stimulus responsive capability. | Incorporation of intelligent response mechanisms to achieve on-demand insulin release. | [154] |
| GO-AuNPs | prepared by in situ chemical reduction of HAuCl4 on ultrasonically dispersed GO; Unavailable; Unavailable | Insulin | Optimizing Drug Nanomaterial Interactions. | Synthesis of various GO–Au composite materials and systematic comparison. | [80] |
| In@GO NgC | prepared by mixing GO nanogels with insulin for molecular intercalation; 11.0 nm; Unavailable | Insulin | Oral insulin delivery is challenging. | Achieved a breakthrough in oral insulin delivery; developed a pH-responsive controlled-release system. | [83] |
| RGO/Ni(OH)2 film | rGO/Ni(OH)2 films prepared by cathodic EPD after Ni2+-induced zeta potential reversal, then loaded with insulin; Unavailable; +14 mV | Insulin | Conventional insulin therapy lacks real-time feedback mechanisms. | Integrated “sensing–release” dual-function system; electrochemically triggered controlled insulin release. | [84] |
| GO | Synthesized through ultrasonication; 4 μm; –27.5 mV | Insulin | Insulin is easily degraded by gastric acid, with extremely low oral bioavailability. | Improved insulin stability and bioavailability. | [155] |
| Carrier Type | Method of Administration | Main Accumulation Site | Toxicity Manifestations | Reference |
|---|---|---|---|---|
| GO | Intratracheal instillation | Lungs (primary), small amounts in liver, intestine | Dose- and time-dependent acute lung injury (ALI) characterized by increased LDH, increased ALP, protein leakage, pulmonary edema, and neutrophil infiltration; Persistent retention in lungs for up to 3 months, leading to chronic pulmonary fibrosis and collagen deposition | [166] |
| GO | intratracheal instillation | Lungs (alveolar regions, airways for aggregated GO) | Oxidized GO induced severe and persistent lung injury, mitochondrial ROS generation, inflammation, and apoptosis; nanoscale-dispersed pristine graphene exhibited markedly reduced toxicity | [167] |
| PEG-GO | Oral | Gastrointestinal | No significant acute or chronic toxicity; normal hematology, biochemistry, and histopathology | [166] |
| PEG-GO | Intraperitoneal | Liver, spleen, kidney, lung | No significant organ damage despite high tissue accumulation; normal hematology and biochemistry | [166] |
| GO | Intravenous injection | liver, spleen, kidneys, and embryonic tissues. | Increased embryo resorption, decreased survival rate, developmental delay; alterations in maternal gut microbiota and metabolites associated with inflammation and metabolic disruption. | [168] |
| GO | nose inhalation | Alveolar regions of the lungs | High concentration (1.88 mg/m3) caused mild alveolitis and inflammation on day 1, which largely resolved by day 14 without systemic toxicity | [169] |
| Functional Group/Material | Toxicity Type | Dose | Reference |
|---|---|---|---|
| Pristine GO(Epoxy group, Hydroxyl group) | Platelet aggregation via Src kinase activation, calcium release, ROS generation, cytoskeletal changes, mitochondrial potential collapse, apoptosis (high concentration) | 0.5–20 μg/mL in vitro; 250 μg/kg in vivo (mice) | [171] |
| Pristine GO(Epoxy group, Hydroxyl group) | pristine graphene oxide | 10–100 μg/mL | [170] |
| rGO/reduced oxygen functional groups, altered charge distribution | Minor platelet aggregation, weaker activation, minimal thrombus induction | 2–10 μg/mL in vitro; 250 μg/kg in vivo (mice) | [171] |
| Carboxyl-functionalized GO (GO-COOH) | Lower hemolysis rate, reduced oxidative damage compared to GO | 25 μg/mL | [170] |
| Polyethylenimine-functionalized GO, (GO-PEI) | Severe hematotoxicity to T lymphocytes via membrane damage | [171] | |
| Amine-modified graphene (G-NH2) | No thrombogenic or platelet-activating effects; does not compromise red blood cell integrity. | 250 μg/kg body weight | [172] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jin, C.; Zheng, H.; Chen, J. Advances in the Application of Graphene and Its Derivatives in Drug Delivery Systems. Pharmaceuticals 2025, 18, 1245. https://doi.org/10.3390/ph18091245
Jin C, Zheng H, Chen J. Advances in the Application of Graphene and Its Derivatives in Drug Delivery Systems. Pharmaceuticals. 2025; 18(9):1245. https://doi.org/10.3390/ph18091245
Chicago/Turabian StyleJin, Changzhou, Huishan Zheng, and Jianmin Chen. 2025. "Advances in the Application of Graphene and Its Derivatives in Drug Delivery Systems" Pharmaceuticals 18, no. 9: 1245. https://doi.org/10.3390/ph18091245
APA StyleJin, C., Zheng, H., & Chen, J. (2025). Advances in the Application of Graphene and Its Derivatives in Drug Delivery Systems. Pharmaceuticals, 18(9), 1245. https://doi.org/10.3390/ph18091245




