Dual Chemotherapeutic Loading in Oxalate Transferrin-Conjugated Polymersomes Incorporated into Chitosan Hydrogels for Site-Specific Targeting of Melanoma Cells
Abstract
1. Introduction
2. Results and Discussion
2.1. Stability and Characterization of Polymersomes
2.2. Co-Encapsulation of Drugs in Polymersomes
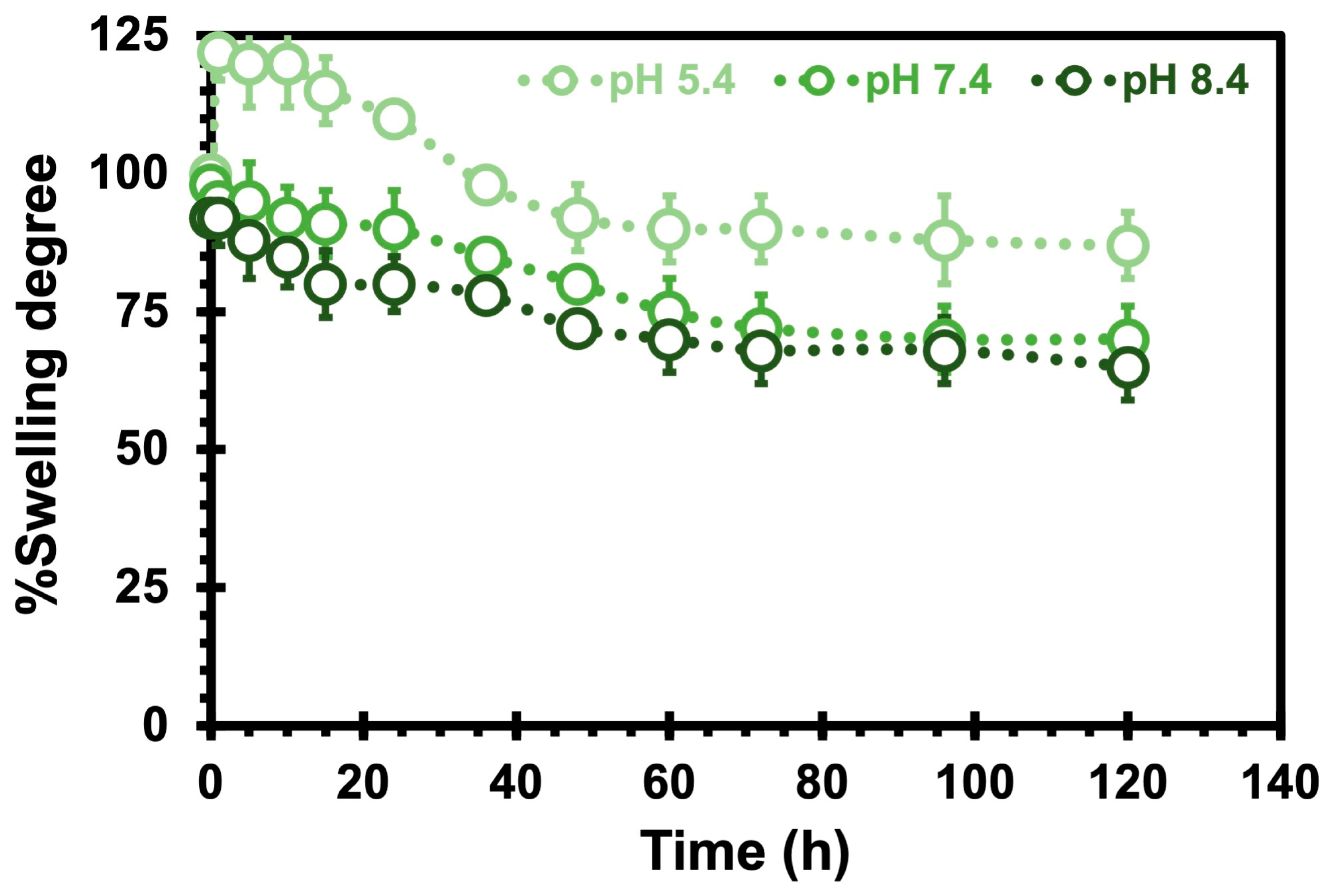
2.3. Studies of Degradation and Swelling of Chitosan Hydrogels
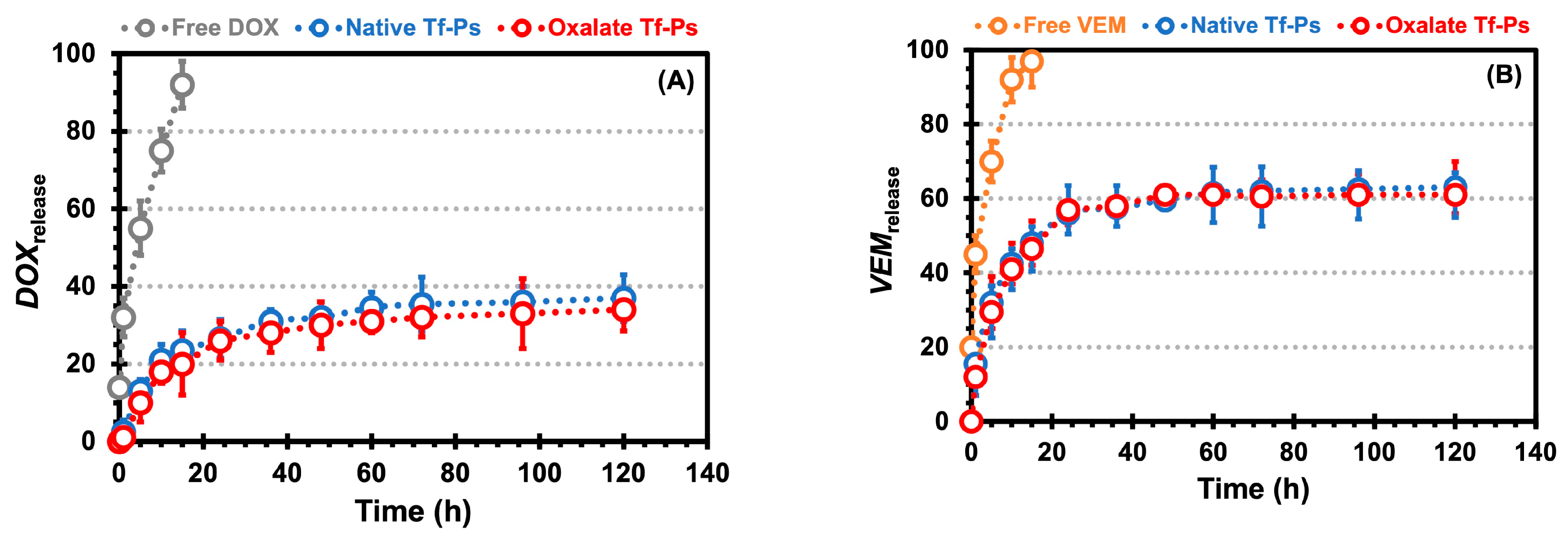
2.4. Studies on Drug Release from Chitosan Hydrogels
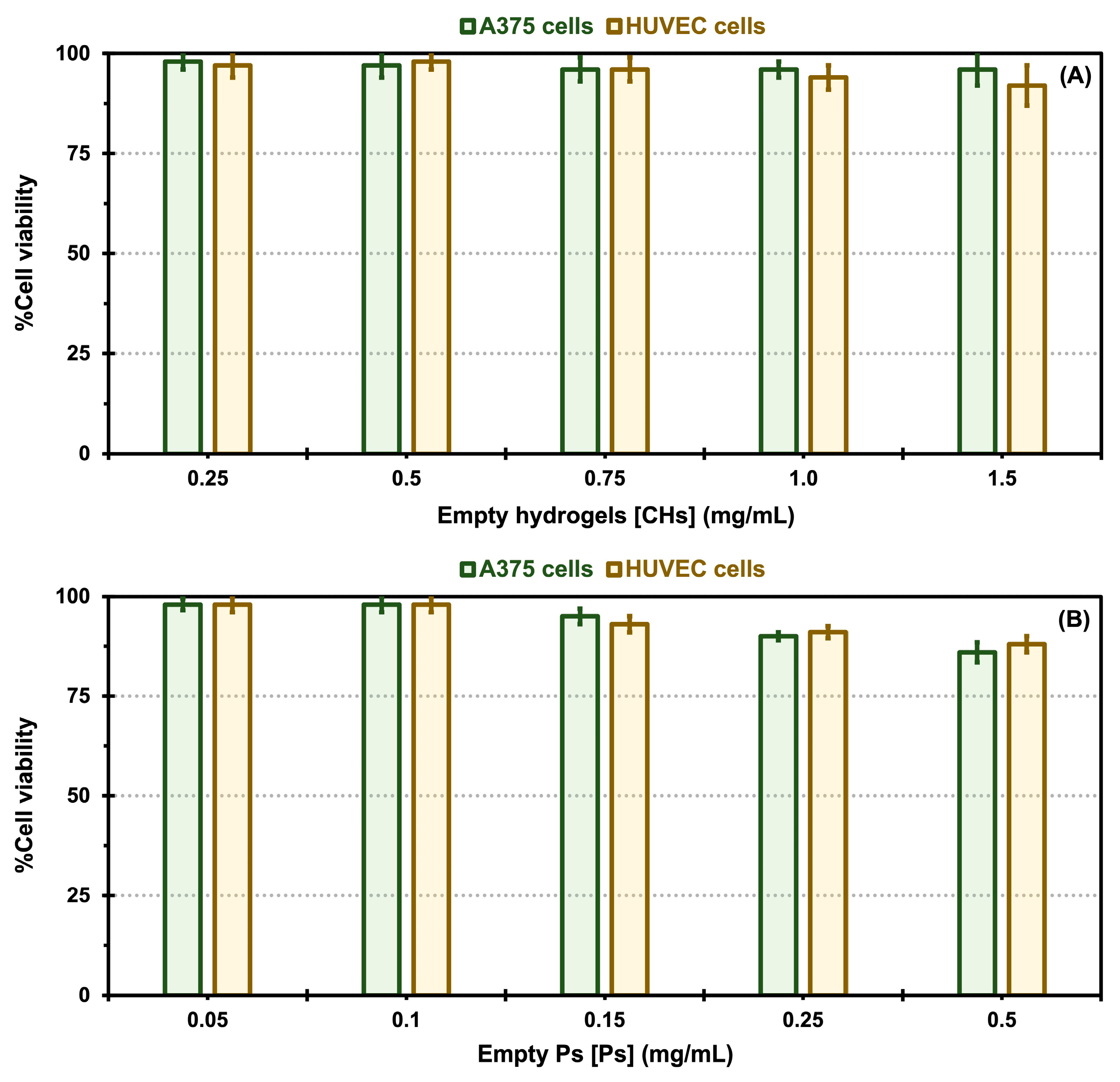
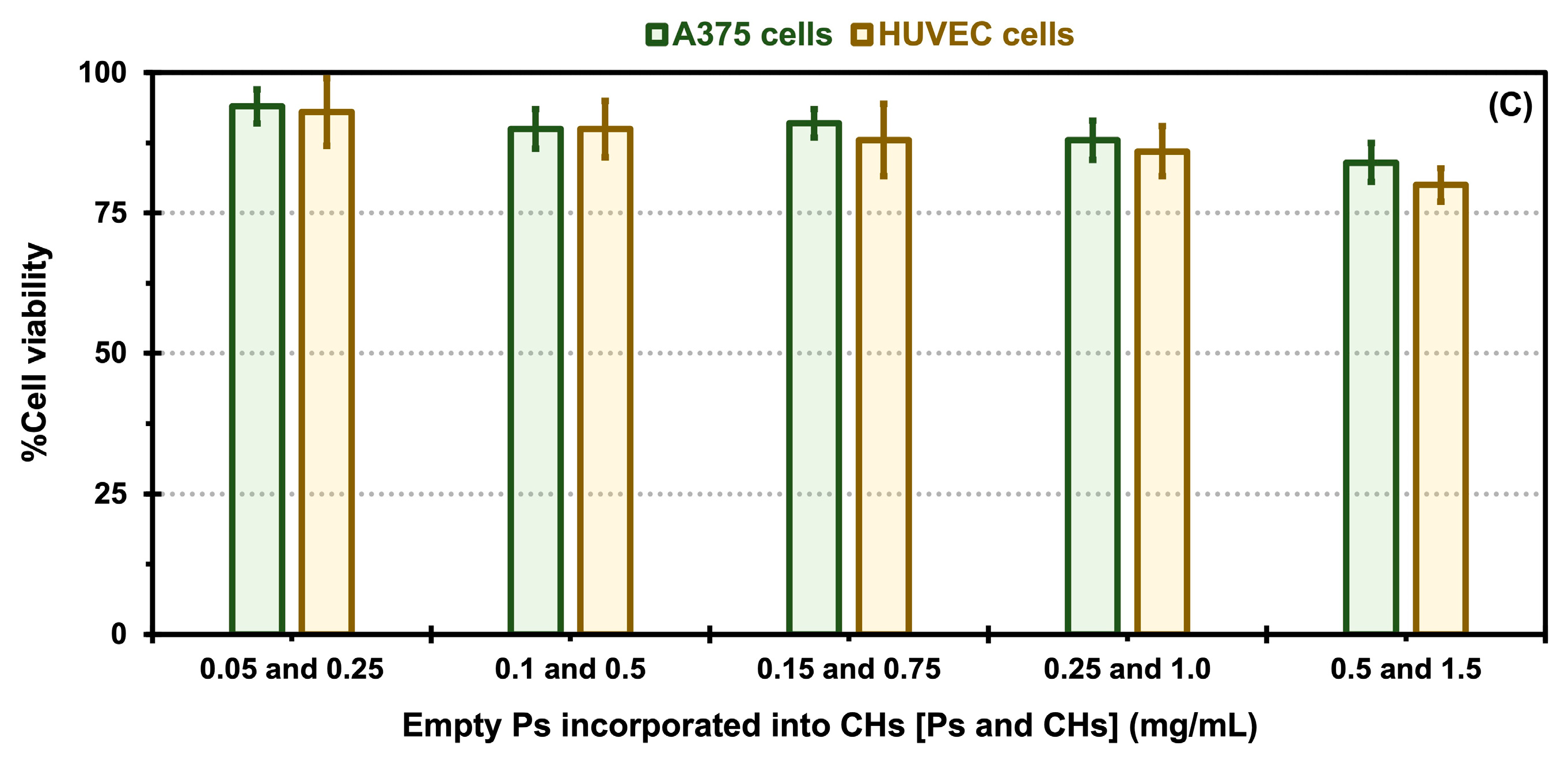
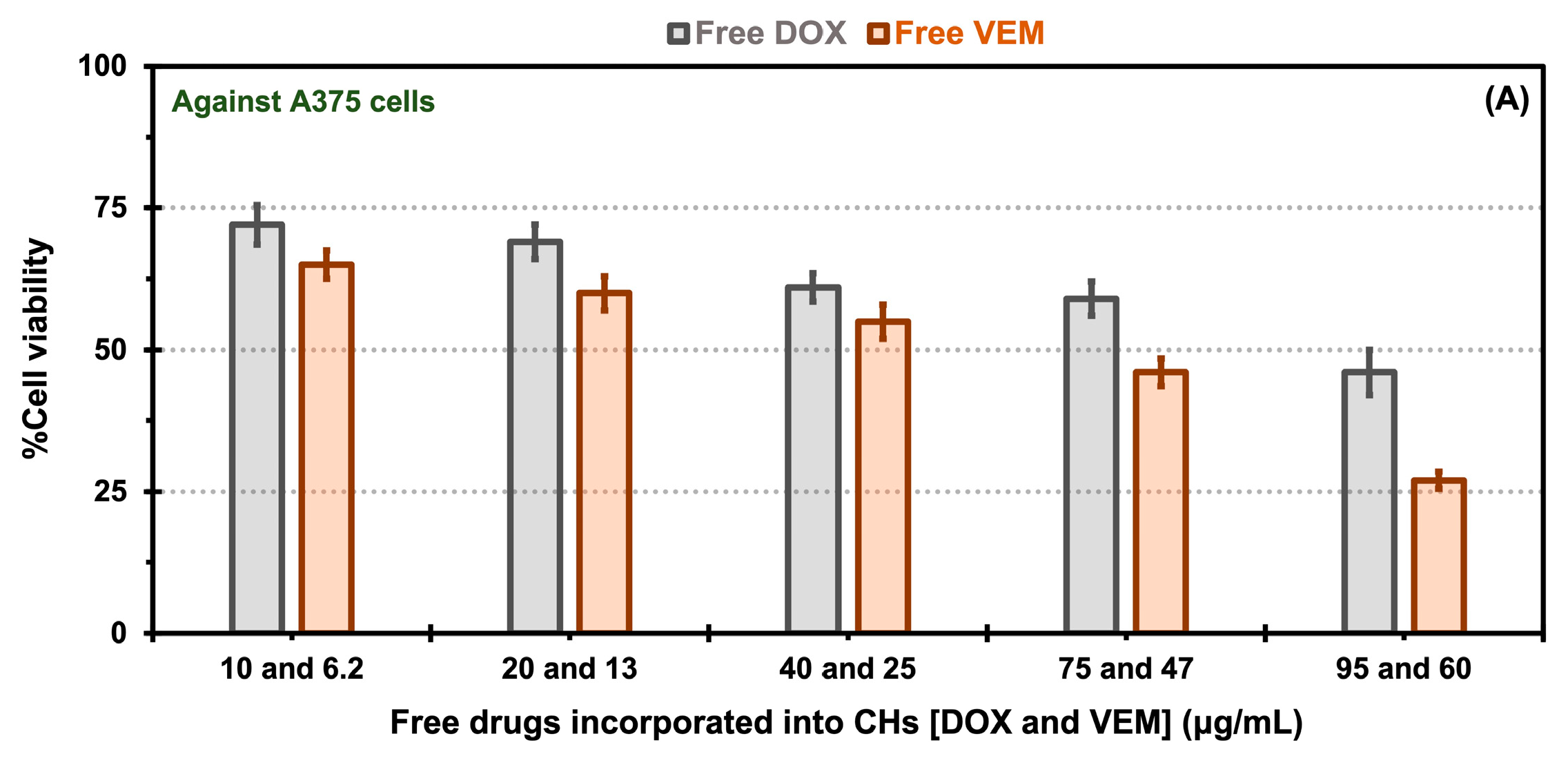
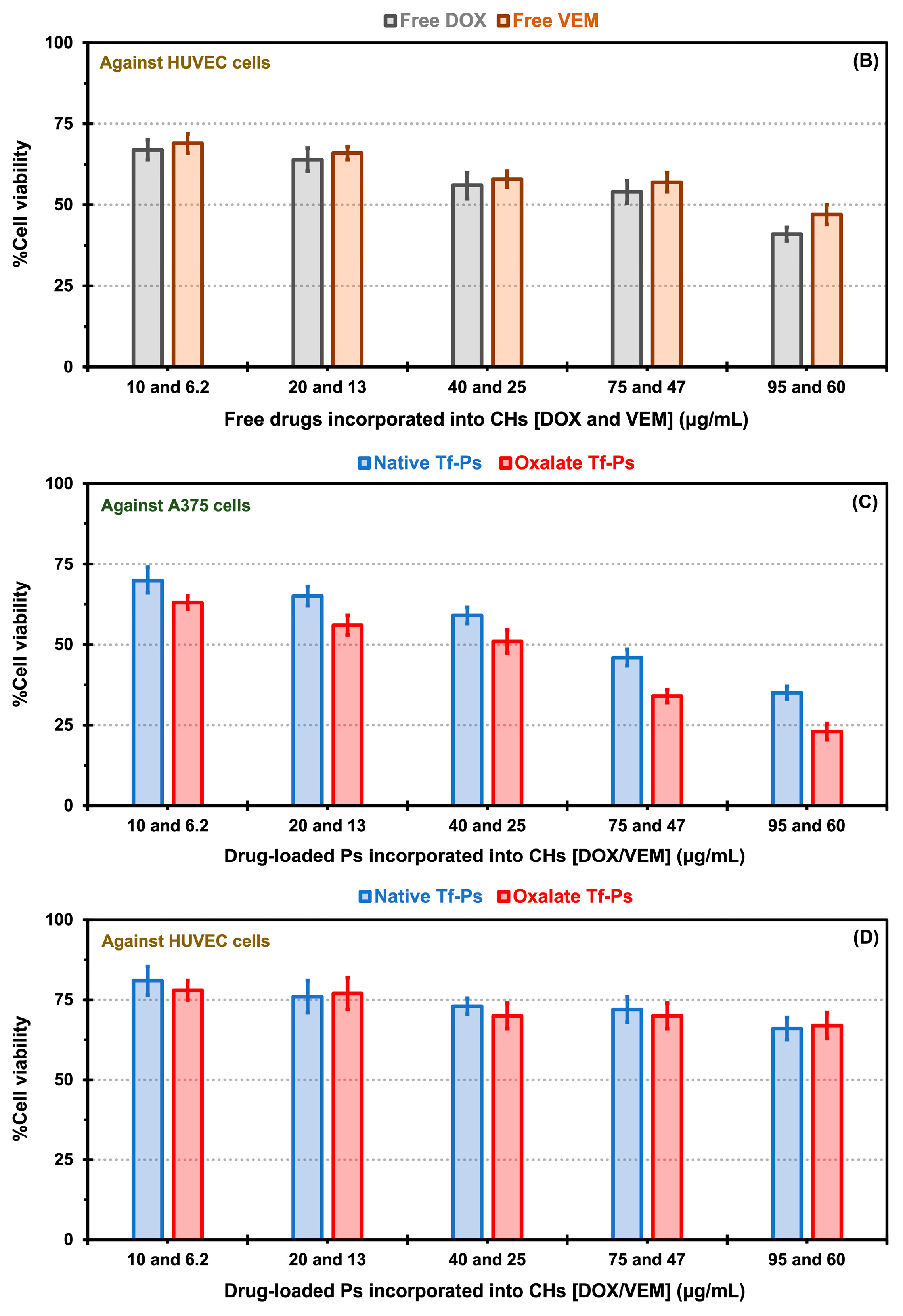
2.5. In Vitro Cytotoxicity Evaluation
3. Materials and Methods
3.1. Materials
3.2. Polymersome Preparation
3.3. Chitosan Hydrogels Preparation and Incorporation of the Polymersomes
3.4. Swelling and Degradation of Chitosan Hydrogels
3.5. Dimensional Stability and Characterization of Polymersomes
3.6. Determination of Encapsulation Parameters
3.7. Drug Quantification
3.8. Drug Release from Chitosan Hydrogels
3.9. In Vitro Cytotoxicity Evaluation
3.10. Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Milojkovic Kerklaan, B.; van Tellingen, O.; Huitema, A.D.R.; Beijnen, J.H.; Boogerd, W.; Schellens JH, M.; Brandsma, D. Strategies to target drugs to gliomas and CNS metastases of solid tumors. J. Neurol. 2016, 263, 428–440. [Google Scholar] [CrossRef] [PubMed]
- Pratt, W.B.; Ruddon, R.W.; Ensminger, W.D.; Maybaum, J. The Anticancer Drugs, 2nd ed.; Oxford University Press: New York, NY, USA, 1994. [Google Scholar]
- Robbins, S.L.; Angell, M.; Kumar, V. Basic Pathology, 3rd ed.; Saunders, W.B.: Philadelphia, PA, USA, 1981. [Google Scholar]
- Hochberg, F.H.; Pruitt, A. Assumptions in the radiotherapy of glioblastoma. Neurology 1980, 30, 907. [Google Scholar] [CrossRef] [PubMed]
- Jain, R.K. Barriers to drug delivery in solid tumors. Sci. Am. 1994, 271, 58–65. [Google Scholar] [CrossRef]
- Nawaz, A.; Ullah, S.; Alnuwaiser, M.A.; Rehman, F.U.; Selim, S.; Al Jaouni, S.K.; Farid, A. Formulation and evaluation of chitosan-gelatin thermosensitive hydrogels containing 5FU-alginate nanoparticles for skin delivery. Gels 2022, 26, 537. [Google Scholar] [CrossRef]
- Fung, L.K.; Saltzman, W.M. Polymeric implants for cancer chemotherapy. Adv. Drug Deliv. Rev. 1997, 26, 209–230. [Google Scholar] [CrossRef] [PubMed]
- De Souza, R.; Zahedi, P.; Allen, C.J.; Piquette-Miller, M. Polymeric drug delivery systems for localized cancer chemotherapy. Drug Deliv. 2010, 17, 365–375. [Google Scholar] [CrossRef] [PubMed]
- Ho, E.A.; Vassileva, V.; Allen, C.; Piquette-Miller, M. In vitro and in vivo characterization of a novel biocompatible polymer–lipid implant system for the sustained delivery of paclitaxel. J. Release 2005, 104, 181–191. [Google Scholar] [CrossRef]
- Brem, H.; Langer, R. Polymer-based drug delivery to the brain. Sci. Med. 1996, 3, 52–61. [Google Scholar]
- DeFail, A.J.; Edington, H.D.; Matthews, S.; Lee, W.-C.C.; Marra, K.G. Controlled release of bioactive doxorubicin from microspheres embedded within gelatin scaffolds. J. Biomed. Mater. Res. A 2006, 79, 954–962. [Google Scholar] [CrossRef]
- Szewczuk, M.; Hrynyk, M.; Ellis, J.P.; Haxho, F.; Allison, S.; Steele JA, M.; Abdulkhalek, S.; Neufeld, R.J. Therapeutic designed poly (lactic-co-glycolic acid) cylindrical oseltamivir phosphate-loaded implants impede tumor neovascularization, growth and metastasis in mouse model of human pancreatic carcinoma. Drug. Des. Devel. Ther. 2015, 9, 4573–4586. [Google Scholar] [CrossRef]
- Liu, J.; Meisner, D.; Kwong, E.; Wu, X.Y.; Johnston, M.R. A Novel trans-lymphatic drug delivery system: Implantable gelatin sponge impregnated with PLGA–paclitaxel microspheres. Biomaterials 2007, 28, 3236–3244. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Li, J.; Jin, K.; Liu, W.; Qiu, X.; Li, C. Fabrication of functional PLGA-based electrospun scaffolds and their applications in biomedical engineering. Mat. Sci. Eng. C 2016, 59, 1181–1194. [Google Scholar] [CrossRef] [PubMed]
- Elstad, N.L.; Fowers, K.D. OncoGel (ReGel/Paclitaxel)—Clinical applications for a novel paclitaxel delivery system. Adv. Drug. Deliv. Rev. 2009, 61, 785–794. [Google Scholar] [CrossRef]
- Yoon, J.J.; Kim, J.H.; Park, T.G. Dexamethasone-releasing biodegradable polymer scaffolds fabricated by a gas-foaming/salt-leaching method. Biomaterials 2003, 24, 2323–2329. [Google Scholar] [CrossRef] [PubMed]
- Makadia, H.K.; Siegel, S.J. Poly lactic-co-glycolic acid (PLGA) as biodegradable controlled drug delivery carrier. Polymers 2011, 3, 1377–1397. [Google Scholar] [CrossRef] [PubMed]
- Jain, J.P.; Modi, S.; Domb, A.J.; Kumar, N. Role of polyanhydrides as localized drug carriers. J. Control Release 2005, 103, 541–563. [Google Scholar] [CrossRef]
- Daniels, T.R.; Bernabeu, E.; Rodríguez, J.A.; Patel, S.; Kozman, M.; Chiappetta, D.A.; Holler, E.; Ljubimova, J.Y.; Helguera, G.; Penichet, M.L. The transferrin receptor and the targeted delivery of therapeutic agents against cancer. Biochim. Biophys. Acta-Gen. Subj. 2012, 1820, 291–317. [Google Scholar] [CrossRef]
- Wei, Y.; Gu, X.; Sun, Y.; Meng, F.; Storm, G.; Zhong, Z. Transferrin-binding peptide functionalized polymersomes mediate targeted doxorubicin delivery to colorectal cancer in vivo. J. Control. Release 2020, 319, 407–415. [Google Scholar] [CrossRef]
- Reckhow Constance, L.; Enns Caroline, A. Characterization of the transferrin receptor in tunicamycin-treated A431 cells. J. Biol. Chem. 1988, 263, 7297–7301. [Google Scholar] [CrossRef]
- De Brabander, M.; Nuydens, R.; Geerts, H.; Hopkins, C.R. Dynamic behavior of the transferrin receptor followed in living epidermoid carcinoma (A431) cells with nanovid microscopy. Cell Motil. 1988, 9, 30–47. [Google Scholar] [CrossRef]
- Tao, J.; Liu, Y.Q.; Li, Y.; Peng, J.L.; Li, L.; Liu, J.; Shen, X.; Shen, G.X.; Tu, Y.T. Hypoxia: Dual effect on the expression of transferrin receptor in human melanoma A375 cell line. Exp. Dermatol. 2007, 16, 899–904. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Tao, J.; Li, Y.; Yang, J.; Yu, Y.; Wang, M.; Xu, X.; Huang, C.; Huang, W.; Dong, J.; et al. Targeting hypoxia-inducible factor-1α with Tf–PEI–shRNA complex via transferrin receptor-mediated endocytosis inhibits melanoma growth. Mol. Ther. 2009, 17, 269–277. [Google Scholar] [CrossRef] [PubMed]
- Mayle, K.M.; Le, A.M.; Kamei, D.T. The intracellular trafficking pathway of transferrin. Biochim. Biophys. Acta-Gen. Subj. 2012, 1820, 264–281. [Google Scholar] [CrossRef]
- Yazdi, P.T.; Murphy, R.M. Quantitative analysis of protein synthesis inhibition by transferrin-toxin conjugates. Cancer Res. 1994, 54, 6387–6394. [Google Scholar]
- Ciechanover, A.; Schwartz, A.L.; Dautry-Varsat, A.; Lodish, H.F. Kinetics of internalization and recycling of transferrin and the transferrin receptor in a human hepatoma cell line. Effect of lysosomotropic agents. J. Biol. Chem. 1983, 258, 9681–9689. [Google Scholar] [CrossRef]
- Lopes, A.M.; Chen, K.Y.; Kamei, D.T. A Transferrin variant as the targeting ligand for polymeric nanoparticles incorporated in 3-D PLGA porous scaffolds. Mat. Sci. Eng. C. 2017, 73, 373–380. [Google Scholar] [CrossRef] [PubMed]
- Chiu RY, T.; Tsuji, T.; Wang, S.J.; Wang, J.; Liu, C.T.; Kamei, D.T. Improving the systemic drug delivery efficacy of nanoparticles using a transferrin variant for targeting. J. Control Release 2014, 180, 33–41. [Google Scholar] [CrossRef]
- Lao, B.J.; Tsai, W.-L.P.; Mashayekhi, F.; Pham, E.A.; Mason, A.B.; Kamei, D.T. Inhibition of transferrin iron release increases in vitro drug carrier efficacy. J. Control Release 2007, 117, 403–412. [Google Scholar] [CrossRef][Green Version]
- D’Angelo, N.A.; Câmara MC, C.; Noronha, M.A.; Grotto, D.; Chorilli, M.; Lourenço, F.R.; Rangel-Yagui, C.O.; Lopes, A.M. Development of PEG-PCL-based polymersomes through design of experiments for co-encapsulation of vemurafenib and doxorubicin as chemotherapeutic drugs. J. Mol. Liq. 2022, 349, 118166. [Google Scholar] [CrossRef]
- Kilár, F.; Simon, I. The Effect of iron binding on the conformation of transferrin. A small angle X-ray scattering study. Biophys. J. 1985, 48, 799–802. [Google Scholar] [CrossRef]
- Grossen, P.; Witzigmann, D.; Sieber, S. Huwyler JPEG-PCL-based nanomedicines: A biodegradable drug delivery system its application. J. Control Release 2017, 260, 46–60. [Google Scholar] [CrossRef]
- Jeong, S.I.; Kim, B.-S.; Lee, Y.M.; Ihn, K.J.; Kim, S.H.; Kim, Y.H. Morphology of elastic poly(L-lactide-co-ε-caprolactone) copolymers and in vitro and in vivo degradation behavior of their scaffolds. Biomacromolecules 2004, 5, 1303–1309. [Google Scholar] [CrossRef]
- Kilàr, F.; Hjertén, S. Separation of the human transferrin forms by carrier- free high-performance zone electrophoresis and isoelectric focusing. J. Chromatogr. A 1989, 480, 351–357. [Google Scholar] [CrossRef] [PubMed]
- Colley, H.E.; Hearnden, V.; Avila-Olias, M.; Cecchin, D.; Canton, I.; Madsen, J.; MacNeil, S.; Warren, N.; Hu, K.; McKeating, J.A.; et al. Polymersome-mediated delivery of combination anticancer therapy to head and neck cancer cells: 2D and 3D in vitro evaluation. Mol. Pharm. 2014, 11, 1176–1188. [Google Scholar] [CrossRef]
- Iatrou, H.; Dimas, K.; Gkikas, M.; Tsimblouli, C.; Sofianopoulou, S. Polymersomes from polypeptide containing triblock co- and terpolymers for drug delivery against pancreatic cancer: Asymmetry of the external hydrophilic blocks. Macromol. Biosci. 2014, 14, 1222–1238. [Google Scholar] [CrossRef] [PubMed]
- Salama, A.; Hasanin, M.; Hesemann, P. Synthesis and antimicrobial properties of new chitosan derivatives containing guanidinium groups. Carbohydr. Polym. 2020, 241, 116363. [Google Scholar] [CrossRef] [PubMed]
- Abou-Yousef, H.; Dacrory, S.; Hasanin, M.; Saber, E.; Kamel, S. Biocompatible hydrogel based on aldehyde-functionalized cellulose and chitosan for potential control drug release. Sustain. Chem. Pharm. 2021, 21, 100419. [Google Scholar] [CrossRef]
- Hasanin, M.S.; Abdelraof, M.; Fikry, M.; Shaker, Y.M.; Sweed, A.M.; Senge, M.O. Development of antimicrobial laser-induced photodynamic therapy based on ethylcellulose/chitosan nanocomposite with 5, 10, 15, 20-tetrakis (m-hydroxyphenyl) porphyrin. Molecules 2021, 26, 3551. [Google Scholar] [CrossRef]
- Wei, L.; Cai, C.; Lin, J.; Chen, T. Dual-drug delivery system based on hydrogel/micelle composites. Biomaterials 2009, 30, 2606–2613. [Google Scholar] [CrossRef]
- Jaklenec, A.; Hinckfuss, A.; Bilgen, B.; Ciombor, D.M.; Aaron, R.; Mathiowitz, E. Sequential release of bioactive IGF-I and TGF-Β1 from PLGA microsphere-based scaffolds. Biomaterials 2008, 29, 1518–1525. [Google Scholar] [CrossRef]
- Jindal, N.; Mehta, S.K. Nevirapine loaded poloxamer 407/pluronic p123 mixed micelles: Optimization of formulation and in vitro evaluation. Colloids Surf. B Biointerfaces 2015, 129, 100–106. [Google Scholar] [CrossRef] [PubMed]
- Ulubayram, K.; Eroglu, I.; Hasirci, N. Gelatin microspheres and sponges for delivery of macromolecules. J. Biomater. Appl. 2002, 16, 227–241. [Google Scholar] [CrossRef]
- Meng, F.; Zhong, Z.; Feijen, J. Stimuli-responsive polymersomes for programmed drug delivery. Biomacromolecules 2009, 10, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Zhou, W.; Meng, F.; Zhang, D.; Otto, C.; Feijen, J. Thermosensitive hydrogel-containing polymersomes for controlled drug delivery. J. Control Release 2010, 146, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Schmaljohann, D. Thermo-and pH-responsive polymers in drug delivery. Adv. Drug Deliv. Rev. 2006, 58, 1655–1670. [Google Scholar] [CrossRef]
- Zhou, Y.; Yan, D.; Dong, W.; Tian, Y. Temperature-responsive phase transition of polymer vesicles: Real-time morphology observation and molecular mechanism. J. Phys. Chem. B 2007, 111, 1262–1270. [Google Scholar] [CrossRef] [PubMed]
- Qu, T.; Wang, A.; Yuan, J.; Gao, Q. Preparation of an amphiphilic triblock copolymer with pH- and thermo-responsiveness and self-assembled micelles applied to drug release. J. Colloid. Interface Sci. 2009, 336, 865–871. [Google Scholar] [CrossRef]
- Kurnik, I.S.; D’Angelo, N.A.; Mazzola, P.G.; Chorilli, M.; Kamei, D.T.; Pereira JF, B.; Vicente, A.A.; Lopes, A.M. Polymeric micelles using cholinium-based ionic liquids for the encapsulation and release of hydrophobic drug molecules. Biomater. Sci. 2021, 9, 2183–2196. [Google Scholar] [CrossRef]
- Salimi, A.; Sharif Makhmal Zadeh, B.; Kazemi, M. Preparation and optimization of polymeric micelles as an oral drug delivery system for deferoxamine mesylate: In vitro and ex vivo studies. Res. Pharm. Sci. 2019, 14, 293. [Google Scholar]
- Castro, K.C.; Coco, J.C.; Santos, É.M.; Ataide, J.A.; Martinez, R.M.; Nascimento MH, M.; Prata, J.; Fonte PR, M.L.; Severino, P.; Mazzola, P.G.; et al. Pluronic® triblock copolymer-based nanoformulations for cancer therapy: A 10-year overview. J. Control Release 2023, 353, 802–822. [Google Scholar] [CrossRef]
- Ma, H.; Huang, Y.; Tian, W.; Liu, J.; Yan, X.; Ma, L.; Lai, J. Endothelial transferrin receptor 1 contributes to thrombogenesis through cascade ferroptosis. Redox Biol. 2024, 70, 103041. [Google Scholar] [CrossRef]
- Johannessen, C.M.; Boehm, J.S.; Kim, S.Y.; Thomas, S.R.; Wardwell, L.; Johnson, L.A.; Emery, C.M.; Stransky, N.; Cogdill, A.P.; Barretina, J.; et al. COT drives resistance to RAF inhibition through MAP kinase pathway reactivation. Nature 2010, 468, 968–972. [Google Scholar] [CrossRef]
- Singh, S. Liposome encapsulation of doxorubicin and celecoxib in combination inhibits progression of human skin cancer cells. Int. J. Nanomed. 2018, 13, 11–13. [Google Scholar] [CrossRef]
- Nahire, R.; Haldar, M.K.; Paul, S.; Ambre, A.H.; Meghnani, V.; Layek, B.; Katti, K.S.; Gange, K.N.; Singh, J.; Sarkar, K.; et al. Multifunctional polymersomes for cytosolic delivery of gemcitabine and doxorubicin to cancer cells. Biomaterials 2014, 35, 6482–6497. [Google Scholar] [CrossRef]
- Zhou, D.; Fei, Z.; Jin, L.; Zhou, P.; Li, C.; Liu, X.; Zhao, C. Dual-responsive polymersomes as anticancer drug carriers for the co-delivery of doxorubicin and paclitaxel. J. Mater. Chem. B 2021, 9, 801–808. [Google Scholar] [CrossRef]
- Curcio, M.; Mauro, L.; Naimo, G.D.; Amantea, D.; Cirillo, G.; Tavano, L.; Casaburi, I.; Nicoletta, F.P.; Alvarez-Lorenzo, C.; Iemma, F. Facile synthesis of pH-responsive polymersomes based on lipidized PEG for intracellular co-delivery of curcumin and methotrexate. Colloids Surf. B Biointerfaces 2018, 167, 568–576. [Google Scholar] [CrossRef]
- Castro, K.C.; Barbosa, L.R.; Greaves, T.L.; Lopes, A.M. Polymersomes-based curcumin delivery as cancer therapeutics. In Curcumin-Based Nanomedicines as Cancer Therapeutics; Chapter 13; Academic Press: Cambridge, MA, USA, 2024; pp. 291–305. [Google Scholar]
- Yang, X.; Hu, C.; Tong, F.; Liu, R.; Zhou, Y.; Qin, L.; Ouyang, L.; Gao, H. Tumor Microenvironment-responsive dual drug dimer-loaded PEGylated bilirubin nanoparticles for improved drug delivery and enhanced immune-chemotherapy of breast cancer. Adv. Funct. Mater. 2019, 29, 1901896. [Google Scholar] [CrossRef]
- Aplin, A.E.; Kaplan, F.M.; Shao, Y. Mechanisms of resistance to RAF inhibitors in melanoma. J. Investig. Dermatol. 2011, 131, 1817–1820. [Google Scholar] [CrossRef]
- Mamalis, A.; Garcha, M.; Jagdeo, J. Targeting the PD-1 pathway: A promising future for the treatment of melanoma. Arch. Dermatol. Res. 2014, 306, 511–519. [Google Scholar] [CrossRef]
- Nikolaou, V.A.; Stratigos, A.J.; Flaherty, K.T.; Tsao, H. Melanoma: New insights and new therapies. J. Investig. Dermatol. 2012, 132, 854–863. [Google Scholar] [CrossRef]
- Yap, T.A.; Omlin, A.; Bono, J.S. Development of therapeutic combinations targeting major cancer signaling pathways. J. Clin. Oncol. 2013, 31, 1592–1605. [Google Scholar] [CrossRef] [PubMed]
- Wande, D.P.; Qiu, Y.; Chen, S.; Yao, L.; Xu, Y.; Yao, J.; Xiong, H. Modified chitosan nanogel-polymersomes for oral co-delivery of oxaliplatin and rapamycin for synergistic chemotherapy. J. Drug Deliv. Sci. Technol. 2022, 77, 103852. [Google Scholar] [CrossRef]
- Li, S.; Amat, D.; Peng, Z.; Vanni, S.; Raskin, S.; De Angulo, G.; Othman, A.M.; Graham, R.M.; Leblanc, R.M. Transferrin conjugated nontoxic carbon dots for doxorubicin delivery to target pediatric brain tumor cells. Nanoscale. 2016, 8, 16662–16669. [Google Scholar] [CrossRef]



| Polymersomes | DOX | VEM | ||
|---|---|---|---|---|
| DL (%) | EE (%) | DL (%) | EE (%) | |
| Ps without Tf | 12.1 ± 3.5 | 35.0 ± 3.0 | 17.3 ± 2.5 | 42.2 ± 4.2 |
| Native Tf-Ps | 10.0 ± 2.5 | 32.2 ± 3.5 | 18.1 ± 4.0 | 40.4 ± 5.0 |
| Oxalate Tf-Ps | 11.6 ± 3.2 | 33.8 ± 4.5 | 16.3 ± 4.5 | 39.5 ± 4.8 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Aranha, M.d.C.; Alencar, L.M.R.; Souto, E.B.; Kamei, D.T.; Lopes, A.M. Dual Chemotherapeutic Loading in Oxalate Transferrin-Conjugated Polymersomes Incorporated into Chitosan Hydrogels for Site-Specific Targeting of Melanoma Cells. Pharmaceuticals 2024, 17, 1177. https://doi.org/10.3390/ph17091177
Aranha MdC, Alencar LMR, Souto EB, Kamei DT, Lopes AM. Dual Chemotherapeutic Loading in Oxalate Transferrin-Conjugated Polymersomes Incorporated into Chitosan Hydrogels for Site-Specific Targeting of Melanoma Cells. Pharmaceuticals. 2024; 17(9):1177. https://doi.org/10.3390/ph17091177
Chicago/Turabian StyleAranha, Mariana de C., Luciana M. R. Alencar, Eliana B. Souto, Daniel T. Kamei, and André M. Lopes. 2024. "Dual Chemotherapeutic Loading in Oxalate Transferrin-Conjugated Polymersomes Incorporated into Chitosan Hydrogels for Site-Specific Targeting of Melanoma Cells" Pharmaceuticals 17, no. 9: 1177. https://doi.org/10.3390/ph17091177
APA StyleAranha, M. d. C., Alencar, L. M. R., Souto, E. B., Kamei, D. T., & Lopes, A. M. (2024). Dual Chemotherapeutic Loading in Oxalate Transferrin-Conjugated Polymersomes Incorporated into Chitosan Hydrogels for Site-Specific Targeting of Melanoma Cells. Pharmaceuticals, 17(9), 1177. https://doi.org/10.3390/ph17091177










