The Effect of a Tribulus-Based Formulation in Alleviating Cholinergic System Impairment and Scopolamine-Induced Memory Loss in Zebrafish (Danio rerio): Insights from Molecular Docking and In Vitro/In Vivo Approaches
Abstract
1. Introduction
2. Results
2.1. Metabolite Profiling of Tribuls Terrestris Extract
2.2. Molecular Docking Study
2.3. Effect of Tt Extract on Locomotor Activity
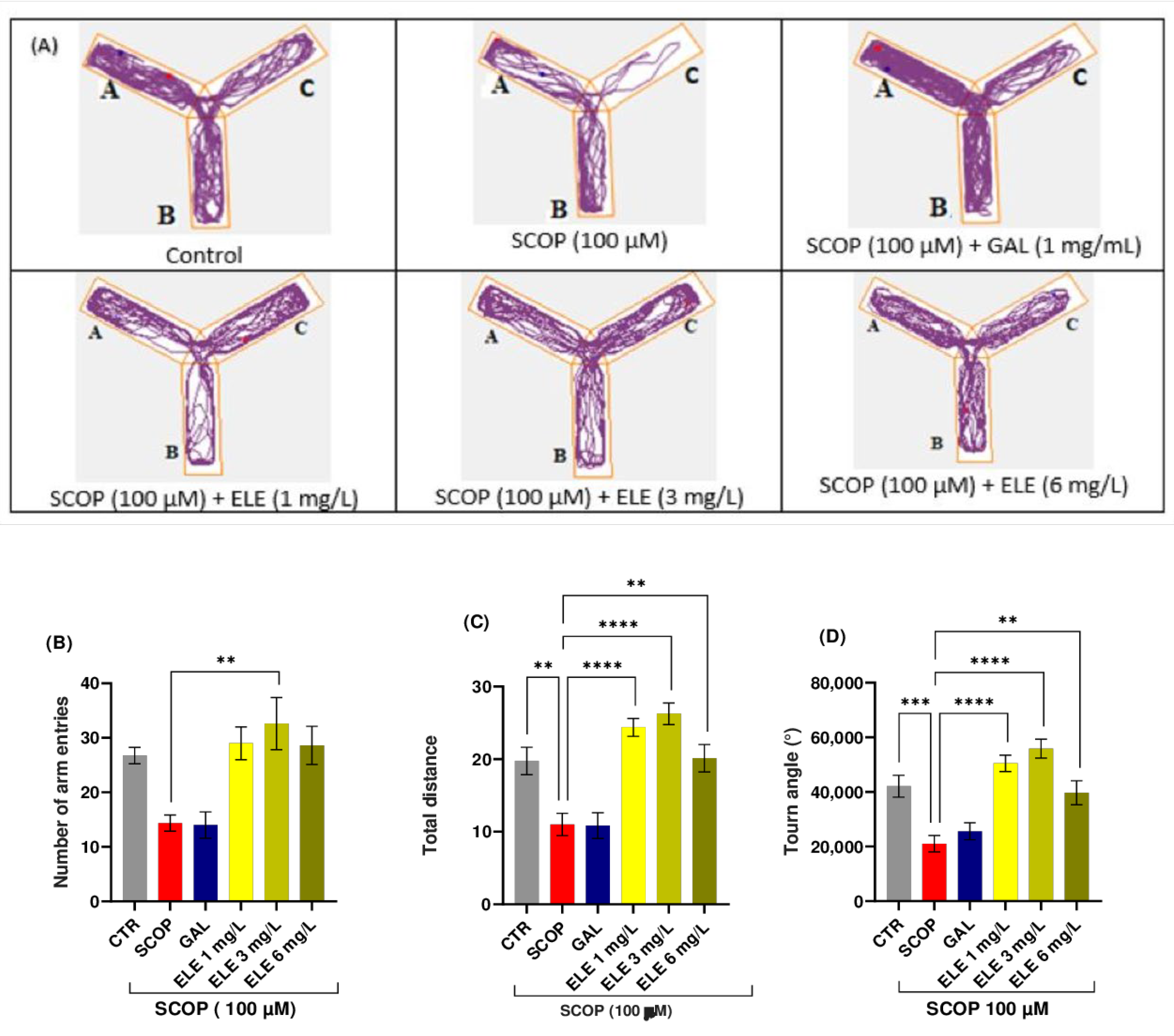
2.4. Effect of Tt Extract on Spatial Memory in Y-Maze
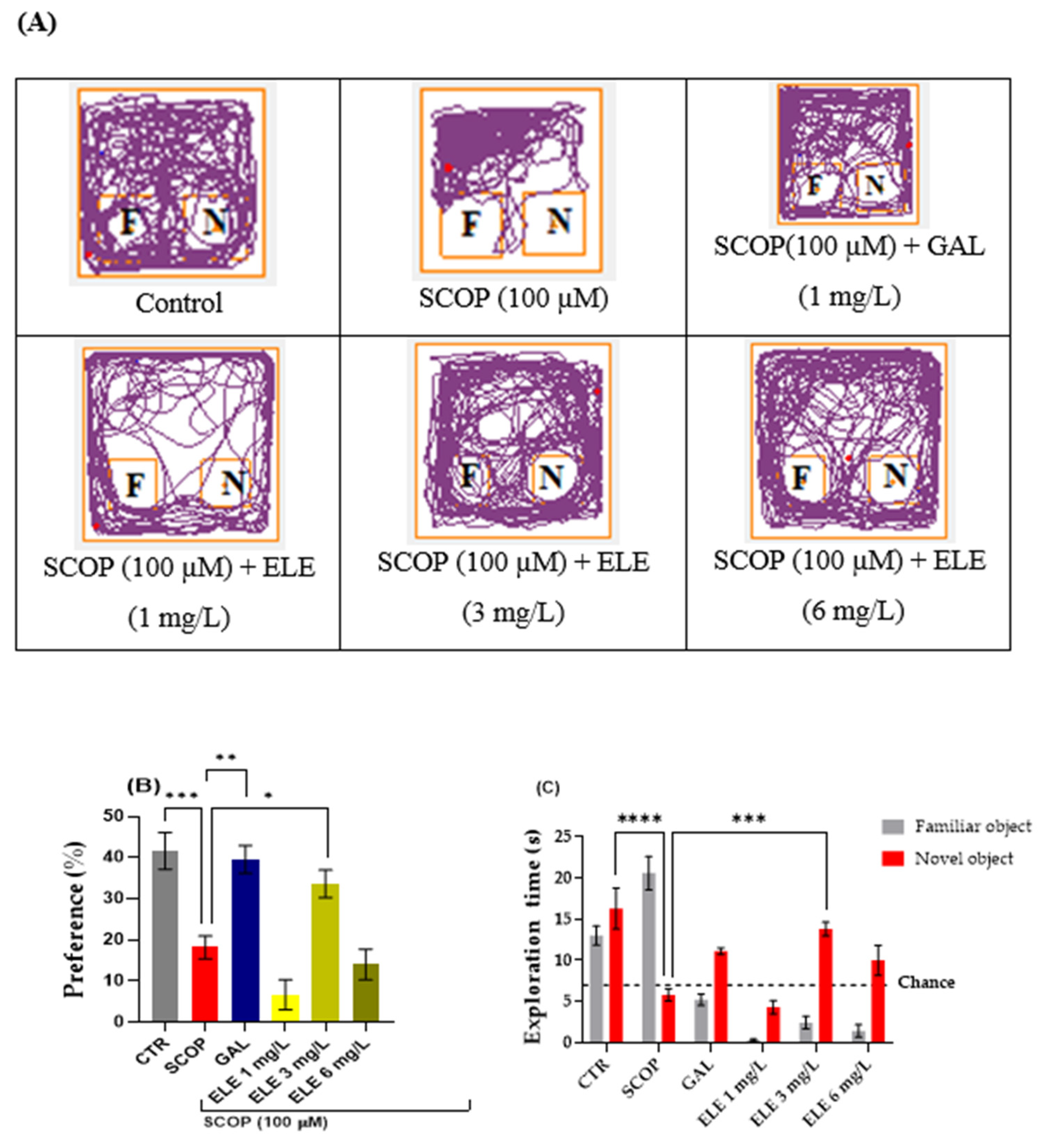
2.5. The Effects of Tt Extract on Recognition Memory in NOR Task
2.6. Effects of Tt Extract on Brain AChE Activity
2.7. Effects of Tt Extract on Brain Oxidative Status
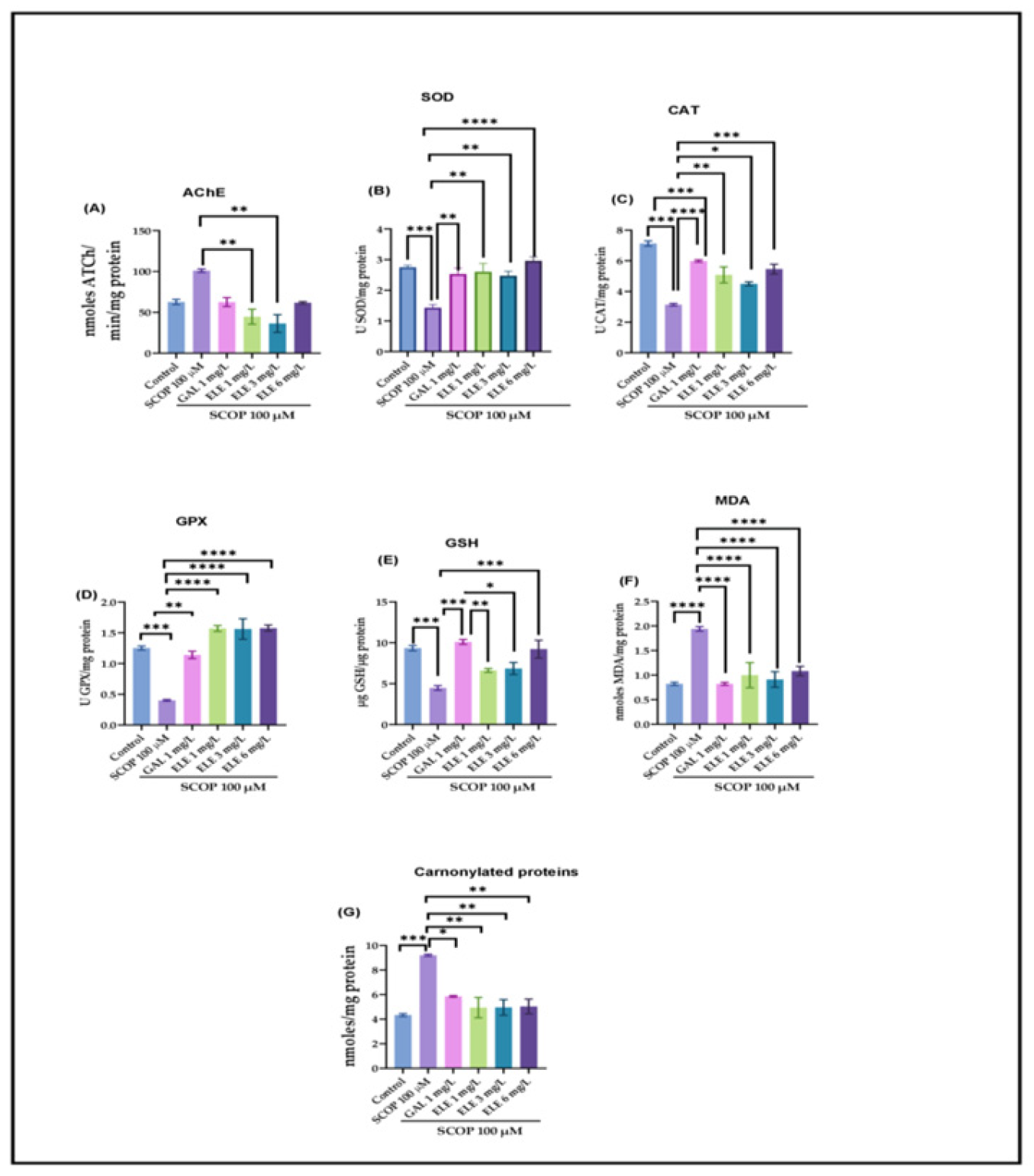
3. Discussion
4. Materials and Methods
4.1. Extraction and Plant Material
4.2. Chemical Analysis of the Investigated Extract
4.2.1. Ultrahigh-Performance Liquid Chromatography–Photodiode Array Detection (UPLC-PDA)
4.2.2. MS Liquid Chromatography (UPLC-ESI/MS)
4.3. Molecular Docking
4.4. Ethical Approval
4.5. Fish Husbandry
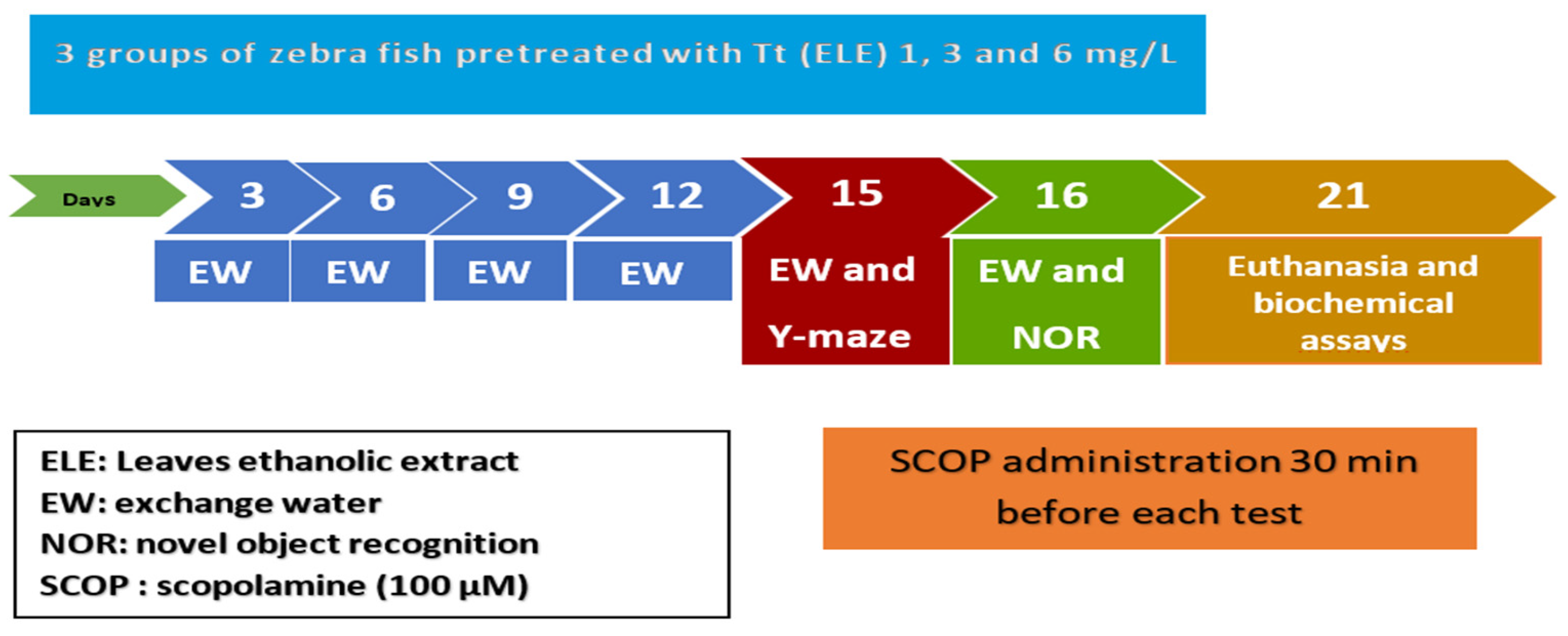
4.6. Drug Treatment and Group Division
4.7. Behavioral Assessment
4.7.1. Y-Maze Test
4.7.2. NOR Test
4.8. Biochemical Analysis
4.9. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Livingston, G.; Huntley, J.; Sommerlad, A.; Ames, D.; Ballard, C.; Banerjee, S.; Brayne, C.; Burns, A.; Cohen-Mansfield, J.; Cooper, C.; et al. Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. Lancet 2020, 396, 413–446. [Google Scholar] [CrossRef]
- Dementia [Fact Sheet]; World Health Organization: Geneva, Switzerland, 2023.
- Rahman, M.A.; Rahman, M.S.; Uddin, M.J.; Mamum-Or-Rashid, A.N.M.; Pang, M.-G.; Rhim, H. Emerging risk of environmental factors: Insight mechanisms of Alzheimer’s diseases. Environ. Sci. Pollut. Res. 2020, 27, 44659–44672. [Google Scholar] [CrossRef] [PubMed]
- Kareti, S.R.; Pharm, S.M. In Silico Molecular Docking Analysis of Potential Anti-Alzheimer’s Compounds Present in Chloroform Extract of Carissa carandas Leaf Using Gas Chromatography MS/MS. Curr. Ther. Res. 2020, 93, 100615. [Google Scholar] [CrossRef]
- Li, G.; Zhang, N.; Geng, F.; Liu, G.; Liu, B.; Lei, X.; Li, G.; Chen, X. High-throughput metabolomics and ingenuity pathway approach reveals the pharmacological effect and targets of Ginsenoside Rg1 in Alzheimer’s disease mice. Sci. Rep. 2019, 9, 7040. [Google Scholar] [CrossRef] [PubMed]
- Dunn, N.R.; Pearce, G.L.; Shakir, S.A. Adverse effects associated with the use of donepezil in general practice in England. J. Psychopharmacol. 2000, 14, 406–408. [Google Scholar] [CrossRef] [PubMed]
- Benninghoff, J.; Perneczky, R. Anti-Dementia Medications and Anti-Alzheimer’s Disease Drugs: Side Effects, Contraindications, and Interactions. In NeuroPsychopharmacotherapy; Riederer, P., Laux, G., Nagatsu, T., Le, W., Riederer, C., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–10. [Google Scholar]
- Wang, S.; Kong, X.; Chen, Z.; Wang, G.; Zhang, J.; Wang, J. Role of Natural Compounds and Target Enzymes in the Treatment of Alzheimer’s Disease. Molecules 2022, 27, 4175. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Lee, H.J.; Lee, K.W. Naturally occurring phytochemicals for the prevention of Alzheimer’s disease. J. Neurochem. 2010, 112, 1415–1430. [Google Scholar] [CrossRef]
- Goozee, K.G.; Shah, T.M.; Sohrabi, H.R.; Rainey-Smith, S.R.; Brown, B.; Verdile, G.; Martins, R.N. Examining the potential clinical value of curcumin in the prevention and diagnosis of Alzheimer’s disease. Br. J. Nutr. 2016, 115, 449–465. [Google Scholar] [CrossRef] [PubMed]
- Yan, Z.-Q.; Chen, J.; Xing, G.-X.; Huang, J.-G.; Hou, X.-H.; Zhang, Y. Salidroside prevents cognitive impairment induced by chronic cerebral hypoperfusion in rats. J. Int. Med. Res. 2015, 43, 402–411. [Google Scholar] [CrossRef]
- Ștefănescu, R.; Tero-Vescan, A.; Negroiu, A.; Aurică, E.; Vari, C.-E. A Comprehensive Review of the Phytochemical, Pharmacological, and Toxicological Properties of Tribulus terrestris L. Biomolecules 2020, 10, 752. [Google Scholar] [CrossRef]
- Chhatre, S.; Nesari, T.; Somani, G.; Kanchan, D.; Sathaye, S. Phytopharmacological overview of Tribulus terrestris. Pharmacogn. Rev. 2014, 8, 45–51. [Google Scholar] [CrossRef]
- Sudheendran, A.; Shajahan, M.A.; Premlal, S. A comparative diuretic evaluation of fruit and root of Gokshura (Tribulus terrestris Linn.) in albino rats. Ayu 2021, 42, 52–56. [Google Scholar] [CrossRef] [PubMed]
- Ara, A.; Vishvkarma, R.; Mehta, P.; Rajender, S. The Profertility and Aphrodisiac Activities of Tribulus terrestris L.: Evidence from Meta-Analyses. Andrologia 2023, 2023, 7118431. [Google Scholar] [CrossRef]
- Phillips, O.A.; Mathew, K.T.; Oriowo, M.A. Antihypertensive and vasodilator effects of methanolic and aqueous extracts of Tribulus terrestris in rats. J. Ethnopharmacol. 2006, 104, 351–355. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.; Algarni, A.S.; Homeida, H.E.; Sultana, S.; Javed, S.A.; Rehman, Z.u.; Abdalla, H.; Alhazmi, H.A.; Albratty, M.; Abdalla, A.N. Phytochemical, Cytotoxic, and Antimicrobial Evaluation of Tribulus terrestris L., Typha domingensis Pers., and Ricinus communis L.: Scientific Evidences for Folkloric Uses. Evid.-Based Complement. Altern. Med. 2022, 2022, 6519712. [Google Scholar] [CrossRef] [PubMed]
- Khalid, A.; Nadeem, T.; Khan, M.A.; Ali, Q.; Zubair, M. In vitro evaluation of immunomodulatory, anti-diabetic, and anti-cancer molecular mechanisms of Tribulus terrestris extracts. Sci. Rep. 2022, 12, 22478. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Tian, X.-C.; Zhang, J.-Q.; Li, T.-T.; Qiao, S.; Jiang, S.-L. Tribulus terrestris L. induces cell apoptosis of breast cancer by regulating sphingolipid metabolism signaling pathways. Phytomedicine 2023, 120, 155014. [Google Scholar] [CrossRef]
- Bouabdallah, S.; Sghaier, R.-M.; Selmi, S.; Khlifi, D.; Laouini, D.; Ben-Attia, M. Current approaches and challenges for chemical characterization of inhibitory effect against cancer cell line isolated from Gokshur extract. J. Chromatogr. B-Anal. Technol. Biomed. Life Sci. 2016, 279–285. [Google Scholar] [CrossRef]
- Bouabdallah, S.; Laouini, D.; Bouzouita, N.; El Bok, S.; Sghaier, R.M.; Selmi, S.; Ben-Attia, M. Separation and evaluation of natural antileishmanial potential against Leishmania major and infantum isolated from the Tunisia strains. Bangladesh J. Pharmacol. 2018, 13, 74–81. [Google Scholar] [CrossRef]
- Bouabdallah, S.; Al-Maktoum, A.; Amin, A. Steroidal Saponins: Naturally Occurring Compounds as Inhibitors of the Hallmarks of Cancer. Cancers 2023, 15, 3900. [Google Scholar] [CrossRef]
- Zhu, W.; Du, Y.; Meng, H.; Dong, Y.; Li, L. A review of traditional pharmacological uses, phytochemistry, and pharmacological activities of Tribulus terrestris. Chem. Cent. J. 2017, 11, 60. [Google Scholar] [CrossRef]
- Semerdjieva, I.B.; Zheljazkov, V.D. Chemical Constituents, Biological Properties, and Uses of Tribulus terrestris: A Review. Nat. Product. Commun. 2019, 14, 1934578X19868394. [Google Scholar] [CrossRef]
- Guan, L.; Mao, Z.; Yang, S.; Wu, G.; Chen, Y.; Yin, L.; Qi, Y.; Han, L.; Xu, L. Dioscin alleviates Alzheimer’s disease through regulating RAGE/NOX4 mediated oxidative stress and inflammation. Biomed. Pharmacother. 2022, 152, 113248. [Google Scholar] [CrossRef]
- Yang, G.-X.; Huang, Y.; Zheng, L.-L.; Zhang, L.; Su, L.; Wu, Y.-H.; Li, J.; Zhou, L.-C.; Huang, J.; Tang, Y.; et al. Design, synthesis and evaluation of diosgenin carbamate derivatives as multitarget anti-Alzheimer’s disease agents. Eur. J. Med. Chem. 2020, 187, 111913. [Google Scholar] [CrossRef]
- Cai, B.; Zhang, Y.; Wang, Z.; Xu, D.; Jia, Y.; Guan, Y.; Liao, A.; Liu, G.; Chun, C.; Li, J. Therapeutic Potential of Diosgenin and Its Major Derivatives against Neurological Diseases: Recent Advances. Oxidative Med. Cell. Longev. 2020, 2020, 3153082. [Google Scholar] [CrossRef]
- Kalueff, A.V.; Stewart, A.M.; Gerlai, R. Zebrafish as an emerging model for studying complex brain disorders. Trends Pharmacol. Sci. 2014, 35, 63–75. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.M.; Collier, A.D.; Meshalkina, D.A.; Kysil, E.V.; Khatsko, S.L.; Kolesnikova, T.; Morzherin, Y.Y.; Warnick, J.E.; Kalueff, A.V.; Echevarria, D.J. Zebrafish models in neuropsychopharmacology and CNS drug discovery. Br. J. Pharmacol. 2017, 174, 1925–1944. [Google Scholar] [CrossRef] [PubMed]
- Levin, E.D.; Bencan, Z.; Cerutti, D.T. Anxiolytic effects of nicotine in zebrafish. Physiol. Behav. 2007, 90, 54–58. [Google Scholar] [CrossRef]
- Johansson, M.; Stomrud, E.; Lindberg, O.; Westman, E.; Johansson, P.M.; van Westen, D.; Mattsson, N.; Hansson, O. Apathy and anxiety are early markers of Alzheimer’s disease. Neurobiol. Aging 2020, 85, 74–82. [Google Scholar] [CrossRef] [PubMed]
- Newman, M.; Ebrahimie, E.; Lardelli, M. Using the zebrafish model for Alzheimer’s disease research. Front. Genet. 2014, 5, 189. [Google Scholar] [CrossRef] [PubMed]
- Stewart, A.M.; Braubach, O.; Spitsbergen, J.; Gerlai, R.; Kalueff, A.V. Zebrafish models for translational neuroscience research: From tank to bedside. Trends Neurosci. 2014, 37, 264–278. [Google Scholar] [CrossRef] [PubMed]
- Saleem, S.; Kannan, R.R. Zebrafish: An emerging real-time model system to study Alzheimer’s disease and neurospecific drug discovery. Cell Death Discov. 2018, 4, 45. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.N.; Yeong, K.Y. Scopolamine, a Toxin-Induced Experimental Model, Used for Research in Alzheimer’s Disease. CNS Neurol. Disord. Drug Targets 2020, 19, 85–93. [Google Scholar] [CrossRef]
- Moreira, N.; Lima, J.; Marchiori, M.F.; Carvalho, I.; Sakamoto-Hojo, E.T. Neuroprotective Effects of Cholinesterase Inhibitors: Current Scenario in Therapies for Alzheimer’s Disease and Future Perspectives. J. Alzheimers Dis. Rep. 2022, 6, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Liao, Y.; Mai, X.; Wu, X.; Hu, X.; Luo, X.; Zhang, G. Exploring the Inhibition of Quercetin on Acetylcholinesterase by Multispectroscopic and In Silico Approaches and Evaluation of Its Neuroprotective Effects on PC12 Cells. Molecules 2022, 27, 7971. [Google Scholar] [CrossRef] [PubMed]
- Brinza, I.; Abd-Alkhalek, A.M.; El-Raey, M.A.; Boiangiu, R.S.; Eldahshan, O.A.; Hritcu, L. Ameliorative Effects of Rhoifolin in Scopolamine-Induced Amnesic Zebrafish (Danio rerio) Model. Antioxidants 2020, 9, 580. [Google Scholar] [CrossRef] [PubMed]
- Capatina, L.; Todirascu-Ciornea, E.; Napoli, E.M.; Ruberto, G.; Hritcu, L.; Dumitru, G. Thymus vulgaris Essential Oil Protects Zebrafish against Cognitive Dysfunction by Regulating Cholinergic and Antioxidants Systems. Antioxidants 2020, 9, 1083. [Google Scholar] [CrossRef]
- Brinza, I.; Raey, M.A.E.; El-Kashak, W.; Eldahshan, O.A.; Hritcu, L. Sweroside Ameliorated Memory Deficits in Scopolamine-Induced Zebrafish (Danio rerio) Model: Involvement of Cholinergic System and Brain Oxidative Stress. Molecules 2022, 27, 5901. [Google Scholar] [CrossRef]
- Dos Santos, T.C.; Gomes, T.M.; Pinto, B.A.S.; Camara, A.L.; Paes, A.M.A. Naturally Occurring Acetylcholinesterase Inhibitors and Their Potential Use for Alzheimer’s Disease Therapy. Front. Pharmacol. 2018, 9, 1192. [Google Scholar] [CrossRef]
- Kassem, I.A.A.; Ragab, M.F.; Nabil, M.; Melek, F.R. Two new acylated triterpenoidal saponins from Gleditsia caspica Desf. and the effect of its saponin content on LPS-induced cognitive impairment in mice. Phytochem. Lett. 2020, 40, 53–62. [Google Scholar] [CrossRef]
- Ramalingayya, G.V.; Nampoothiri, M.; Nayak, P.G.; Kishore, A.; Shenoy, R.R.; Mallikarjuna Rao, C.; Nandakumar, K. Naringin and Rutin Alleviates Episodic Memory Deficits in Two Differentially Challenged Object Recognition Tasks. Pharmacogn. Mag. 2016, 12 (Suppl. 1), S63–S70. [Google Scholar] [PubMed]
- Saleem, U.; Chauhdary, Z.; Raza, Z.; Shah, S.; Rahman, M.-u.; Zaib, P.; Ahmad, B. Anti-Parkinson’s Activity of Tribulus terrestris via Modulation of AChE, α-Synuclein, TNF-α, and IL-1β. ACS Omega 2020, 5, 25216–25227. [Google Scholar] [CrossRef] [PubMed]
- Chauhdary, Z.; Saleem, U.; Ahmad, B.; Shah, S.; Shah, M.A. Neuroprotective evaluation of Tribulus terrestris L. in aluminum chloride induced Alzheimer’s disease. Pak. J. Pharm. Sci. 2019, 32 (Suppl. S2), 805–816. [Google Scholar] [PubMed]
- Sarvin, B.; Stekolshchikova, E.; Rodin, I.; Stavrianidi, A.; Shpigun, O. Optimization and comparison of different techniques for complete extraction of saponins from T. terrestris. J. Appl. Res. Med. Aromat. Plants 2018, 8, 75–82. [Google Scholar] [CrossRef]
- Alam, P.; Parvez, M.K.; Arbab, A.H.; Al-Dosari, M.S. Quantitative analysis of rutin, quercetin, naringenin, and gallic acid by validated RP- and NP-HPTLC methods for quality control of anti-HBV active extract of Guiera senegalensis. Pharm. Biol. 2017, 55, 1317–1323. [Google Scholar] [CrossRef] [PubMed]
- Yang, K.; Chen, Z.; Gao, J.; Shi, W.; Li, L.; Jiang, S.; Hu, H.; Liu, Z.; Xu, D.; Wu, L. The Key Roles of GSK-3beta in Regulating Mitochondrial Activity. Cell Physiol. Biochem. 2017, 44, 1445–1459. [Google Scholar] [CrossRef] [PubMed]
- Jaworski, T.; Banach-Kasper, E.; Gralec, K. GSK-3β at the Intersection of Neuronal Plasticity and Neurodegeneration. Neural Plast. 2019, 2019, 4209475. [Google Scholar] [CrossRef]
- Fu, Y.; Cai, J.; Xi, M.; He, Y.; Zhao, Y.; Zheng, Y.; Zhang, Y.; Xi, J.; He, Y. Neuroprotection Effect of Astragaloside IV from 2-DG-Induced Endoplasmic Reticulum Stress. Oxidative Med. Cell. Longev. 2020, 2020, 9782062. [Google Scholar] [CrossRef]
- Yamazaki, H.; Tanji, K.; Wakabayashi, K.; Matsuura, S.; Itoh, K. Role of the Keap1/Nrf2 pathway in neurodegenerative diseases. Pathol. Int. 2015, 65, 210–219. [Google Scholar] [CrossRef]
- Ramsey, C.P.; Glass, C.A.; Montgomery, M.B.; Lindl, K.A.; Ritson, G.P.; Chia, L.A.; Hamilton, R.L.; Chu, C.T.; Jordan-Sciutto, K.L. Expression of Nrf2 in neurodegenerative diseases. J. Neuropathol. Exp. Neurol. 2007, 66, 75–85. [Google Scholar] [CrossRef]
- Sultana, R.; Perluigi, M.; Butterfield, D.A. Lipid peroxidation triggers neurodegeneration: A redox proteomics view into the Alzheimer disease brain. Free Radic. Biol. Med. 2013, 62, 157–169. [Google Scholar] [CrossRef]
- Cao, P.; Sun, J.; Sullivan, M.A.; Huang, X.; Wang, H.; Zhang, Y.; Wang, N.; Wang, K. Angelica sinensis polysaccharide protects against acetaminophen-induced acute liver injury and cell death by suppressing oxidative stress and hepatic apoptosis in vivo and in vitro. Int. J. Biol. Macromol. 2018, 111, 1133–1139. [Google Scholar] [CrossRef] [PubMed]
- Yin, Y.; Li, H.; Chen, Y.; Zhu, R.; Li, L.; Zhang, X.; Zhou, J.; Wang, Z.; Li, X. Combination of Astragalus membranaceous and Angelica sinensis Ameliorates Vascular Endothelial Cell Dysfunction by Inhibiting Oxidative Stress. Evid. Based Complement. Altern. Med. 2020, 2020, 6031782. [Google Scholar] [CrossRef] [PubMed]
- Song, X.; Kong, J.; Song, J.; Pan, R.; Wang, L. Angelica sinensis Polysaccharide Alleviates Myocardial Fibrosis and Oxidative Stress in the Heart of Hypertensive Rats. Comput. Math. Methods Med. 2021, 2021, 6710006. [Google Scholar] [CrossRef]
- Gonçalves, R.L.G.; Cunha, F.V.M.; Sousa-Neto, B.P.S.; Oliveira, L.S.A.; Lopes, M.E.; Rezende, D.C.; Sousa, I.J.O.; Nogueira, K.M.; Souza, L.K.M.; Medeiros, J.V.R.; et al. α-Phellandrene attenuates tissular damage, oxidative stress, and TNF-α levels on acute model ifosfamide-induced hemorrhagic cystitis in mice. Naunyn Schmiedebergs Arch. Pharmacol. 2020, 393, 1835–1848. [Google Scholar] [CrossRef] [PubMed]
- Cheung, J.; Rudolph, M.J.; Burshteyn, F.; Cassidy, M.S.; Gary, E.N.; Love, J.; Franklin, M.C.; Height, J.J. Structures of human acetylcholinesterase in complex with pharmacologically important ligands. J. Med. Chem. 2012, 55, 10282–10286. [Google Scholar] [CrossRef]
- Abo-Elghiet, F.; Rushdi, A.; Ibrahim, M.H.; Mahmoud, S.H.; Rabeh, M.A.; Alshehri, S.A.; El Menofy, N.G. Chemical Profile, Antibacterial, Antibiofilm, and Antiviral Activities of Pulicaria crispa Most Potent Fraction: An In Vitro and In Silico Study. Molecules 2023, 28, 4184. [Google Scholar] [CrossRef] [PubMed]
- Dumitru, G.; El-Nashar, H.A.S.; Mostafa, N.M.; Eldahshan, O.A.; Boiangiu, R.S.; Todirascu-Ciornea, E.; Hritcu, L.; Singab, A.N.B. Agathisflavone isolated from Schinus polygamus (Cav.) Cabrera leaves prevents scopolamine-induced memory impairment and brain oxidative stress in zebrafish (Danio rerio). Phytomedicine 2019, 58, 152889. [Google Scholar] [CrossRef] [PubMed]
- Cognato Gde, P.; Bortolotto, J.W.; Blazina, A.R.; Christoff, R.R.; Lara, D.R.; Vianna, M.R.; Bonan, C.D. Y-Maze memory task in zebrafish (Danio rerio): The role of glutamatergic and cholinergic systems on the acquisition and consolidation periods. Neurobiol. Learn. Mem. 2012, 98, 321–328. [Google Scholar] [CrossRef]
- Stefanello, F.V.; Fontana, B.D.; Ziani, P.R.; Müller, T.E.; Mezzomo, N.J.; Rosemberg, D.B. Exploring Object Discrimination in Zebrafish: Behavioral Performance and Scopolamine-Induced Cognitive Deficits at Different Retention Intervals. Zebrafish 2019, 16, 370–378. [Google Scholar] [CrossRef]
- Gaspary, K.V.; Reolon, G.K.; Gusso, D.; Bonan, C.D. Novel object recognition and object location tasks in zebrafish: Influence of habituation and NMDA receptor antagonism. Neurobiol. Learn. Mem. 2018, 155, 249–260. [Google Scholar] [CrossRef] [PubMed]
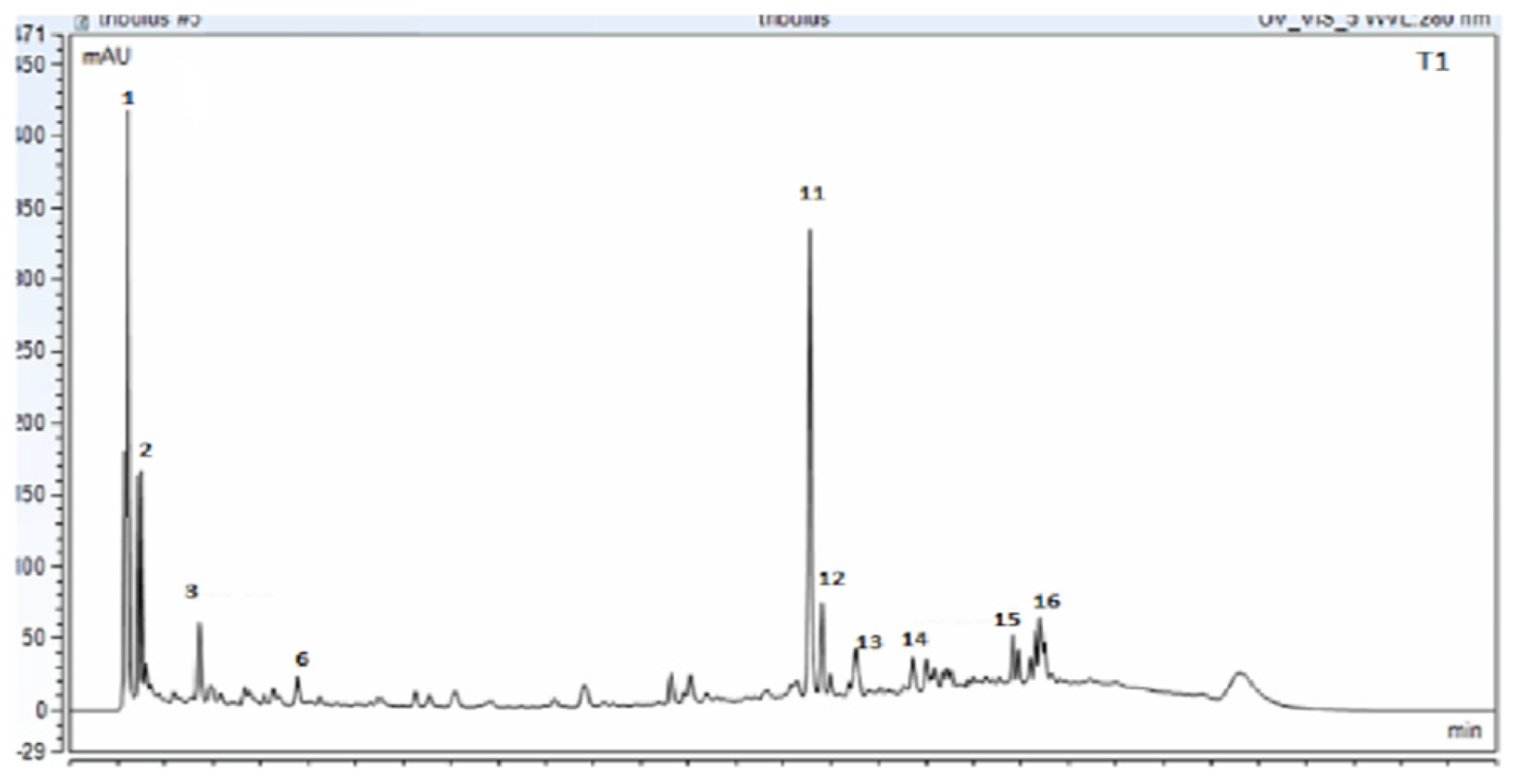
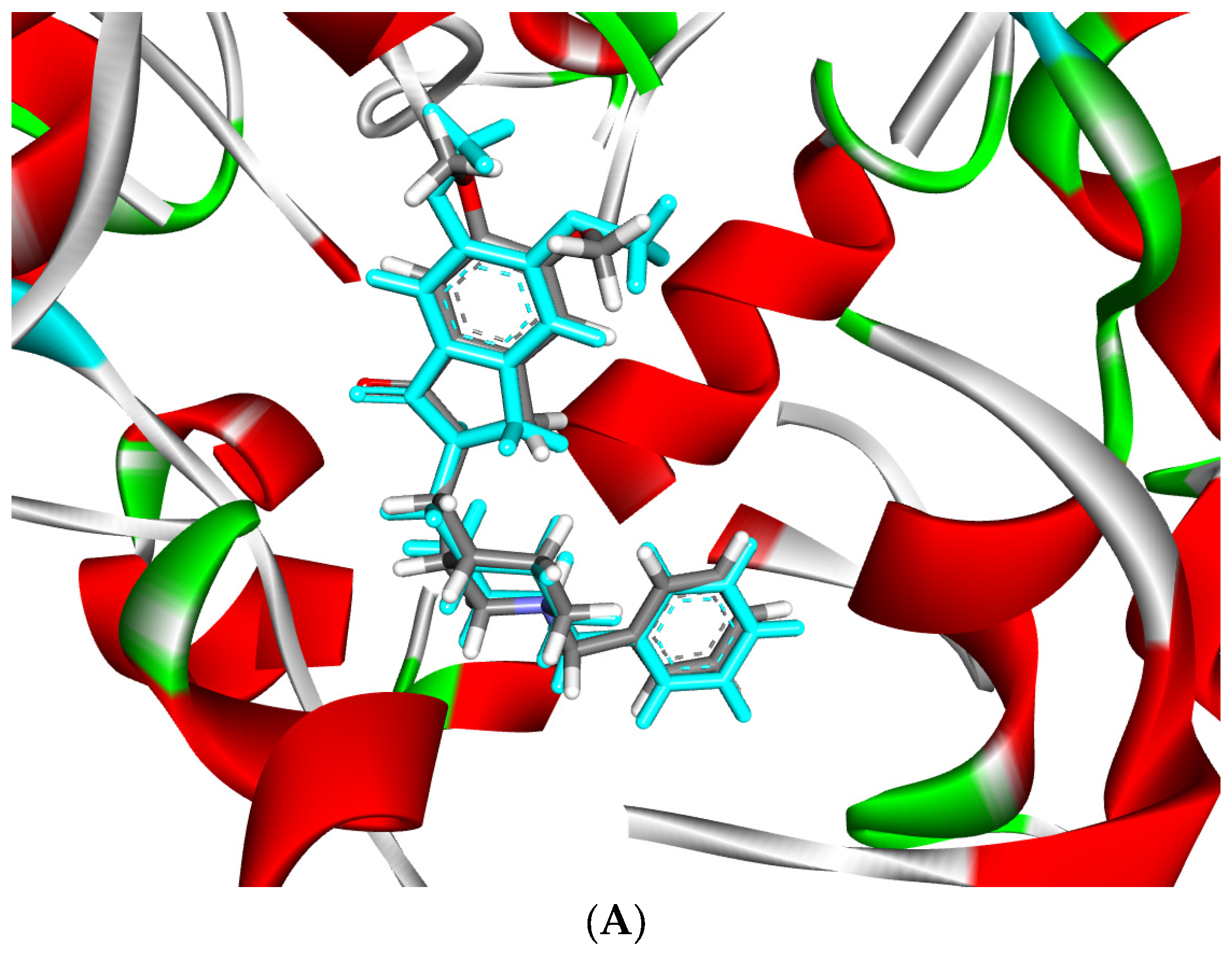

| No. | Formula | Transformations | Error (ppm) | [M−H]−1 Experimental | [M−H]−1 Theoretical | Identification |
|---|---|---|---|---|---|---|
| 1 | C9H10O7 | Hydration, Oxidation | −0.03814 | 180.04225 | 179.03498 | Caffeic acid |
| 2 | C9H8O3 | Hydration, Oxidation | −0.02587 | 198.0402 | 199.0606 | Hydroxycinnamic acid |
| 3 | C21H20O11 | Reduction | −0.38036 | 196.05823 | 195.05095 | Cynarosie |
| 4 | C15H10O6 | Nitro Reduction | −0.21529 | 285.0409 | 286.24 | Kaempferol |
| 5 | C15H10O6 | Nitro Reduction | 0.28338 | 285.04120 | 286.24 | Luteoline |
| 6 | C21H20O11 | Reduction | 0.17143 | 301.0353 | 302.24 | Quercetin |
| 7 | C15H14O7 | Hydration, Oxidation | 0.45039 | 356.07451 | 306.27 | Epigallocatechin |
| 8 | C27H42O4 | Reduction | −0.38036 | 255.08642 | 430.30764 | Hecogenine |
| 9 | C21H20O10 | Reduction | 0.18654 | 435.5 | 432.4 | Apigetrin |
| 10 | C33H52O8 | Desaturation, Nitro Reduction | 2.37949 | 560.22 | 576.761 | Disogluside–Trillin |
| 11 | C27H30O16 | Hydration | −1.45637 | 609.1482 | 610.1084 | Rutin |
| 12 | C39H62O13 | Desaturation, Nitro Reduction | 0.95112 | 738.05805 | 738.05077 | Trillarin |
| 13 | C39H62O14 | Desaturation, Nitro Reduction | 0.3610 | 755.262 | 754.901 | Terreside B |
| 14 | C45H72O17 | Reduction | −4.4 | 915.4550 | 915.4590 | Terreside A |
| 15 | C45H12O15 | Desaturation, Nitro Reduction | −0.38036 | 918.4232 | 915.4590 | Terrestrosin C |
| 16 | C51H84O22 | Oxidation, Nitro Reduction | −0.10032 | 1047.5413 | 1049.2 | Protodioscin |
| Compounds | Docking Energy Scores in kcal/mol | |
|---|---|---|
| Donepezil (Co-crystal ligand) | −14.62 | |
| 1 | Caffeic acid | −11.22 |
| 2 | Disogluside (Trillin) | −18.60 |
| 3 | Apigetrin | −18.60 |
| 4 | Cynaroside | −18.70 |
| 5 | Terreside B | −19.98 |
| 6 | Terrestrosin C | −24.11 |
| 7 | Trillarin | −23.42 |
| 8 | Protodioscin | −24.58 |
| 9 | Epigallocatechin | −20.62 |
| 10 | Rutin | −24.68 |
| 11 | Hecogenin | −14.71 |
| 12 | Saponin C | −24.63 |
| 13 | Quercetin | −15.39 |
| 14 | Kaempferol | −15.48 |
| 15 | Luteoline | −14.65 |
| Ligand | Affinity Energy (kcal/mol) | Interaction | Amino Acid Distance in Angstrom (Å) |
|---|---|---|---|
| Donepezil (Co-crystal ligand) | −14.62 | Attractive Charge | Asp74 (5.40) |
| 1 Hydrogen Bond | Phe295 (1.96) | ||
| 2 Hydrogen Bond | Ser293 (3.06), Tyr72 (3.20) | ||
| π-Cation | Trp86 (4.73), Tyr337 (3.91) | ||
| π-Sigma | Tyr341 (3.59), Trp286 (3.64) | ||
| π–π Stacked | Trp86 (4.46), Trp86 (3.89, 3.82,5.11), Tyr341 (5.05) | ||
| Terrestrosin C | −24.11 | 2 Hydrogen Bond | Gly120 (2.93, 2.88), Asp74 (2.37), Glu202 (2.27), Asn87 (2.33, 2.34), Tyr124 (2.43), Trp86 (2.00) |
| 1 Hydrogen Bond | Tyr124 (2.13), Ser125 (1.76), Trp86 (2.52, 1.68) | ||
| π-Alkyl | Tyr72 (4.65), Tyr124 (4.26), Trp286 (3.20, 4.21, 4.48), Phe297 (4.98), Phe338 (4.91), Tyr341 (4.59, 3.67) | ||
| π-Sigma | Trp286 (2.63) | ||
| Protodioscin | −24.58 | 1 Hydrogen Bond | Tyr72 (2.97), Asp74 (2.82), Tyr124 (2.04), Ser293 (2.15, 2.60), Tyr341 (2.75, 2.81), Trp286 (3.23), Trp86 (3.20), Asn87 (2.80, 2.70), Thr83 (2.57), Asp74 (2.65), Glu202 (2.77) |
| 2 Hydrogen Bond | Thr83 (2.78), Glu292 (2.47) | ||
| π-Sigma | Tyr341 (3.48), Tyr337 (3.00) | ||
| π-Alkyl | Tyr124 (4.94), Trp286 (4.76), Phe297 (5.18, 4.56), Phe338 (5.13, 5.01, 4.31, 5.49), Tyr341 (3.80, 4.53), His447 (4.69, 5.41) | ||
| Rutin | −24.68 | 1 Hydrogen Bond | Tyr124 (1.64), Phe295 (2.93), Tyr341 (2.45, 3.04), His447 (2.85), Asp74 (2.71), Thr83 (2.79), Asn87 (2.50), Trp86 (3.12), Glu202 (3.05, 3.22) |
| 2 Hydrogen Bond | Trp86 (2.83), Asn87 (2.74), Ser125 (3.02), Ser203 (3.03), Val294 (2.41), His447 (2.36) | ||
| 3 Hydrogen Bond | His447 (2.91), Trp286 (3.54) | ||
| π–π Stacked | Trp286 (4.85), Tyr337 (3.44), Tyr341 (3.32, 4.03, 5.61) | ||
| π–π T-shaped | Trp86 (5.92), Tyr124 (5.88), Phe338 (4.80) | ||
| Saponin C | −24.63 | 1 Hydrogen Bond | Asp74 (2.50), Tyr133 (2.96), Ser293 (2.70), Tyr72 (2.90), Asn87 (3.23), Trp286 (3.36), Leu289 (2.99), Gly120 (2.88) |
| 2 Hydrogen Bond | Gly121 (2.60, 2.69, 2.16), Glu292 (2.84), Phe338 (2.38, 2.41), His447 (2.98), Gln291 (3.54) | ||
| 3 Hydrogen Bond | Tyr341 (3.58), Trp86 (3.62, 4.12), Phe338 (3.02, 3.73) | ||
| π-Alkyl | Tyr72 (5.33, 4.87), Trp286 (5.33, 4.63, 5.26, 3.88), His287 (4.67), Phe297 (5.29), Phe338 (5.27), Tyr341 (4.84, 4.88) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bouabdallah, S.; Brinza, I.; Boiangiu, R.S.; Ibrahim, M.H.; Honceriu, I.; Al-Maktoum, A.; Cioanca, O.; Hancianu, M.; Amin, A.; Ben-Attia, M.; et al. The Effect of a Tribulus-Based Formulation in Alleviating Cholinergic System Impairment and Scopolamine-Induced Memory Loss in Zebrafish (Danio rerio): Insights from Molecular Docking and In Vitro/In Vivo Approaches. Pharmaceuticals 2024, 17, 200. https://doi.org/10.3390/ph17020200
Bouabdallah S, Brinza I, Boiangiu RS, Ibrahim MH, Honceriu I, Al-Maktoum A, Cioanca O, Hancianu M, Amin A, Ben-Attia M, et al. The Effect of a Tribulus-Based Formulation in Alleviating Cholinergic System Impairment and Scopolamine-Induced Memory Loss in Zebrafish (Danio rerio): Insights from Molecular Docking and In Vitro/In Vivo Approaches. Pharmaceuticals. 2024; 17(2):200. https://doi.org/10.3390/ph17020200
Chicago/Turabian StyleBouabdallah, Salwa, Ion Brinza, Razvan Stefan Boiangiu, Mona H. Ibrahim, Iasmina Honceriu, Amna Al-Maktoum, Oana Cioanca, Monica Hancianu, Amr Amin, Mossadok Ben-Attia, and et al. 2024. "The Effect of a Tribulus-Based Formulation in Alleviating Cholinergic System Impairment and Scopolamine-Induced Memory Loss in Zebrafish (Danio rerio): Insights from Molecular Docking and In Vitro/In Vivo Approaches" Pharmaceuticals 17, no. 2: 200. https://doi.org/10.3390/ph17020200
APA StyleBouabdallah, S., Brinza, I., Boiangiu, R. S., Ibrahim, M. H., Honceriu, I., Al-Maktoum, A., Cioanca, O., Hancianu, M., Amin, A., Ben-Attia, M., & Hritcu, L. (2024). The Effect of a Tribulus-Based Formulation in Alleviating Cholinergic System Impairment and Scopolamine-Induced Memory Loss in Zebrafish (Danio rerio): Insights from Molecular Docking and In Vitro/In Vivo Approaches. Pharmaceuticals, 17(2), 200. https://doi.org/10.3390/ph17020200












