Pharmacokinetics of Curcumin Delivered by Nanoparticles and the Relationship with Antitumor Efficacy: A Systematic Review
Abstract
1. Introduction
2. Results
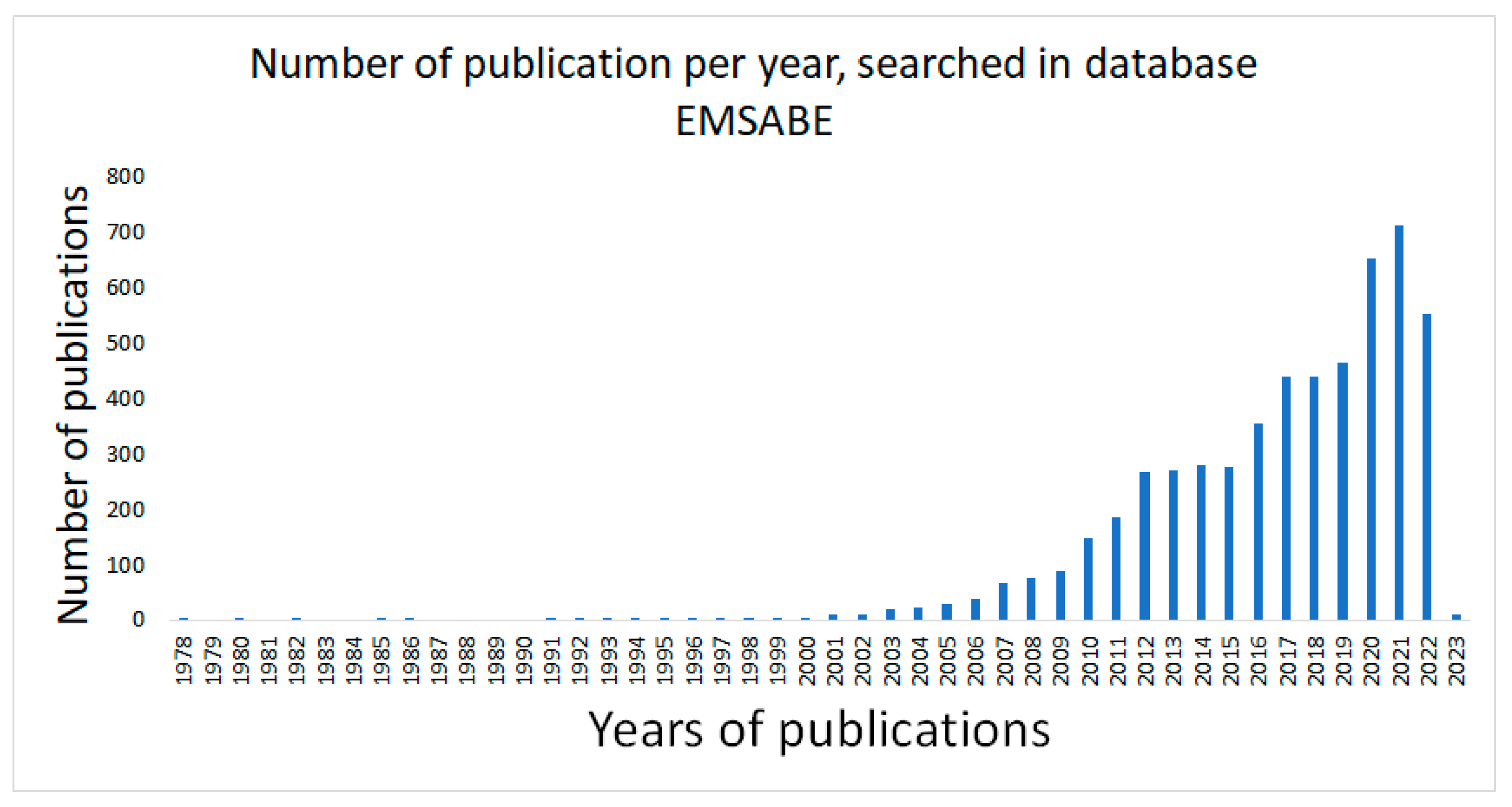
2.1. Selection of Studies
2.2. Characteristics of the Included Studies
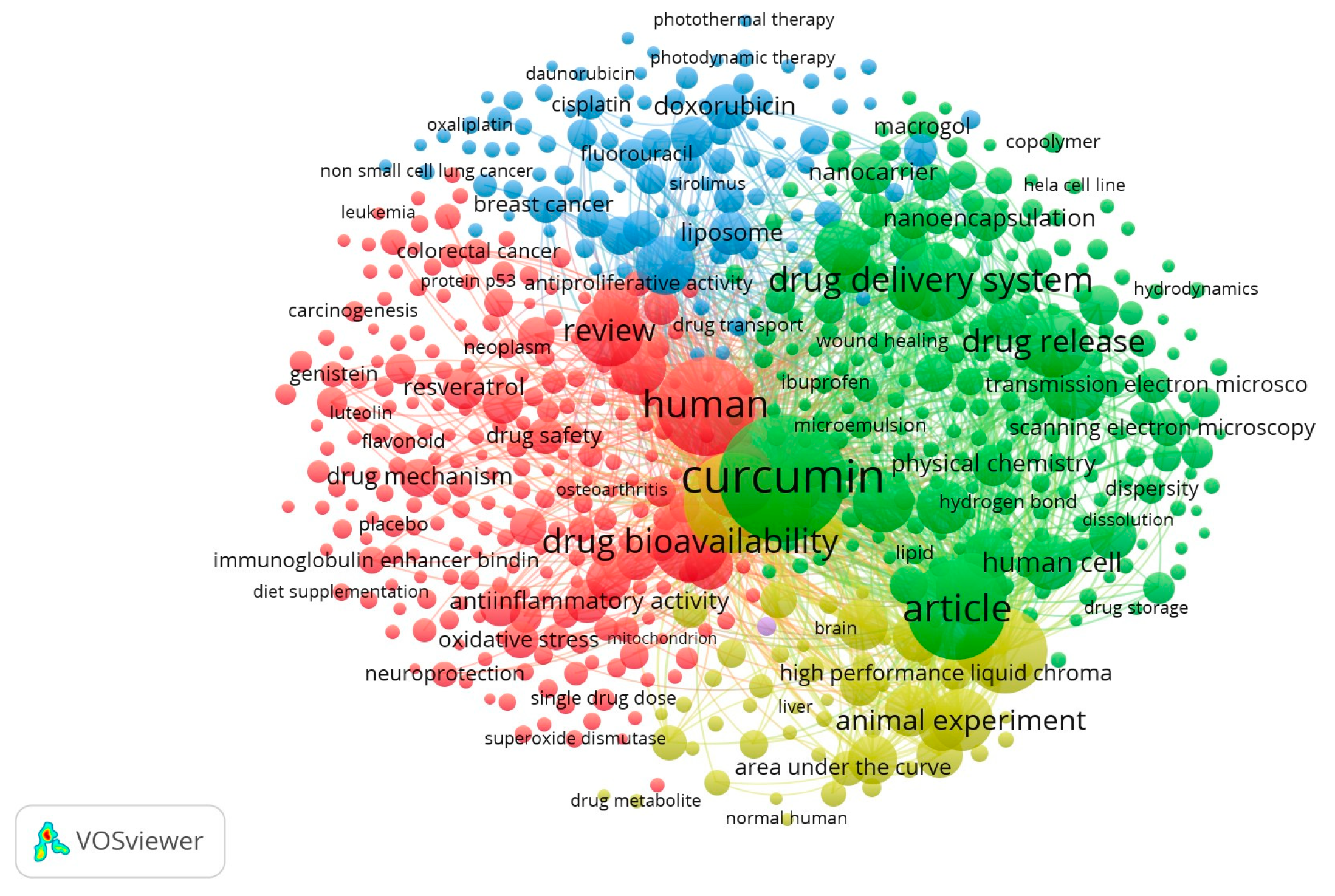
2.3. Synthesis of Results
2.4. Quality and Risk of Bias
3. Discussion
4. Materials and Methods
4.1. Registration Protocol
4.1.1. Inclusion Criteria
4.1.2. Exclusion Criteria
4.2. Information Sources and Research Strategy
4.2.1. Selection Process
4.2.2. Data Collection Process and Quality Analyses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| MDPI | Multidisciplinary Digital Publishing Institute |
| DOAJ | Directory of open-access journals |
| TLA | Three letter acronym |
| CUR | Curcumin |
| Cur-NPs | Curcumin nanoparticles |
| HPLC | High-Performance liquid chromatography |
| NCI | National Cancer Institute |
| PROSPERO | Prospective Register of Systematic Reviews |
| PICOS | Population, Intervention, Comparison, Outcome and Study Design |
| HD | Hydrodynamic diameter |
| PDI | Polydispersity index |
| ZT | Zeta potential |
| UPLC | Ultra-performance liquid chromatography |
| AUC | Area under the curve |
| PLGA | Poly lactic-co-glycolic acid |
| GMO | Chitosan glycerol monooleate |
| PCL | Polycaprolactone |
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A. The nanocomposites designs of phytomolecules from medicinal and aromatic plants: Promising anticancer-antiviral applications. Beni-Suef Univ. J. Basic Appl. Sci. 2022, 11, 17. [Google Scholar] [CrossRef] [PubMed]
- Chandramouli, V.; Niraj, S.K.; Nair, K.G.; Joseph, J.; Aruni, W. Phytomolecules repurposed as COVID-19 inhibitors: Opportunity and challenges. Curr. Microbiol. 2021, 78, 3620–3633. [Google Scholar] [CrossRef] [PubMed]
- Zielińska, A.; Alves, H.; Marques, V.; Durazzo, A.; Lucarini, M.; Alves, T.F.; Morsink, M.; Willemen, N.; Eder, P.; Chaud, M.V.; et al. Medicina Properties, Extraction Methods, and Delivery Systems for Curcumin as a Natural Source of Beneficial Health Effects. Medicina 2020, 56, 336. [Google Scholar] [CrossRef] [PubMed]
- Tomeh, M.A.; Hadianamrei, R.; Zhao, X. Molecular Sciences A Review of Curcumin and Its Derivatives as Anticancer Agents. Int. J. Mol. Sci. 2019, 20, 1033. [Google Scholar] [CrossRef]
- Giordano, A.; Tommonaro, G. Curcumin and Cancer. Nutrients 2019, 11, 2376. [Google Scholar] [CrossRef]
- Hong, J.; Liu, Y.; Xiao, Y.; Yang, X.; Su, W.; Zhang, M.; Liao, Y.; Kuang, H.; Wang, X. High drug payload curcumin nanosuspensions stabilized by mPEG-DSPE and SPC: In vitro and in vivo evaluation. Drug Deliv. 2017, 24, 109–120. [Google Scholar] [CrossRef]
- Ombredane, A.S.; Silva, V.R.P.; Andrade, L.R.; Pinheiro, W.O.; Simonelly, M.; Oliveira, J.V.; Pinheiro, A.C.; Gonçalves, G.F.; Felice, G.J.; Garcia, M.P.; et al. In vivo efficacy and toxicity of curcumin nanoparticles in breast cancer treatment: A systematic review. Front. Oncol. 2021, 11, 612903. [Google Scholar] [CrossRef]
- Mansouri, K.; Rasoulpoor, S.; Daneshkhah, A.; Abolfathi, S.; Salari, N.; Mohammadi, M.; Shabani, S. Clinical effects of curcumin in enhancing cancer therapy: A systematic review. BMC Cancer 2020, 20, 791. [Google Scholar] [CrossRef]
- Mohankumar, K.; Francis, A.P.; Pajaniradje, S.; Rajagopalan, R. Synthetic curcumin analog: Inhibiting the invasion, angiogenesis, and metastasis in human laryngeal carcinoma cells via NF-kB pathway. Mol. Biol. Rep. 2021, 48, 6065–6074. [Google Scholar] [CrossRef]
- Aqil, F.; Munagala, R.; Jeyabalan, J.; Agrawal, A.; Gupta, R. Exosomes for the enhanced tissue bioavailability and efficacy of curcumin. AAPS J. 2017, 19, 1691–1702. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.-P.; Yi, L.; Yu, Z.; Gao-Wei, L. Formulation, characterization and evaluation of curcumin-loaded PLGA-TPGS nanoparticles for liver cancer treatment. Drug Des. Dev. Ther. 2019, 13, 3569. [Google Scholar] [CrossRef]
- Hu, Y.; He, Y.; Ji, J.; Zheng, S.; Cheng, Y. Tumor targeted curcumin delivery by folate-modified MPEG-PCL self-assembly micelles for colorectal cancer therapyy. Int. J. Nanomed. 2020, 15, 1239. [Google Scholar] [CrossRef] [PubMed]
- Xie, X.; Li, Y.; Zhao, D.; Fang, C.; He, D.; Yang, Q.; Yang, L.; Chen, R.; Tan, Q.; Zhang, J. Oral administration of natural polyphenol-loaded natural polysaccharide-cloaked lipidic nanocarriers to improve efficacy against small-cell lung cancer. Nanomed. Nanotechnol. Biol. Med. 2020, 29, 102261. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Ying, X.; Xu, H.; Yan, H.; Li, X.; Tang, H. The functional curcumin liposomes induce apoptosis in C6 glioblastoma cells and C6 glioblastoma stem cells in vitro and in animals. Int. J. Nanomed. 2017, 12, 1369. [Google Scholar] [CrossRef] [PubMed]
- Wang, B.; Liu, X.; Teng, Y.; Yu, T.; Chen, J.; Hu, Y.; Liu, N.; Zhang, L.; Shen, Y. Improving anti-melanoma effect of curcumin by biodegradable nanoparticles. Oncotarget 2017, 8, 108624. [Google Scholar] [CrossRef]
- Yuan, J.-D.; ZhuGe, D.-L.; Tong, M.-Q.; Lin, M.-T.; Xu, X.-F.; Tang, X.; Zhao, Y.-Z.; Xu, H.-L. pH-sensitive polymeric nanoparticles of mPEG-PLGA-PGlu with hybrid core for simultaneous encapsulation of curcumin and doxorubicin to kill the heterogeneous tumour cells in breast cancer. Artif. Cells Nanomed. Biotechnol. 2018, 46, 302–313. [Google Scholar] [CrossRef]
- Sun, L.; Xu, H.; Xu, J.-H.; Wang, S.-N.; Wang, J.-W.; Zhang, H.-F.; Jia, W.-R.; Li, L.-S. Enhanced Antitumor Efficacy of Curcumin-Loaded PLGA Nanoparticles Coated with Unique Fungal Hydrophobin. AAPS PharmSciTech 2020, 21, 171. [Google Scholar] [CrossRef]
- Guo, F.; Fu, Q.; Zhou, K.; Jin, C.; Wu, W.; Ji, X.; Yan, Q.; Yang, Q.; Wu, D.; Li, A. Matrix metalloprotein-triggered, cell penetrating pep-tide-modified star-shaped nanoparticles for tumor targeting and cancer therapy. J. Nanobiotechnol. 2020, 18, 48. [Google Scholar] [CrossRef]
- Guorgui, J.; Wang, R.; Mattheolabakis, G.; Mackenzie, G.G. Curcumin formulated in solid lipid nanoparticles has enhanced efficacy in Hodgkin’s lymphoma in mice. Arch. Biochem. Biophys. 2018, 648, 12–19. [Google Scholar] [CrossRef]
- VOSviewer. Features-Highlights. Available online: https://www.vosviewer.com/features/highlights (accessed on 22 September 2022).
- Guo, F.; Fu, Q.; Jin, C.; Ji, X.; Yan, Q.; Yang, Q.; Wu, D.; Gao, Y.; Hong, W.; Li, A.; et al. Dual functional matrix metalloproteinase-responsive curcumin-loaded nanoparticles for tumor-targeted treatment. Drug Deliv. 2019, 26, 1027–1038. [Google Scholar] [CrossRef] [PubMed]
- Ji, P.; Wang, L.; Chen, Y.; Wang, S.; Wu, Z.; Qi, X. Hyaluronic acid hydrophilic surface rehabilitating curcumin nanocrystals for targeted breast cancer treatment with prolonged biodistribution. Biomater. Sci. 2020, 8, 462–472. [Google Scholar] [CrossRef] [PubMed]
- Risk of Bias. Available online: https://www.riskofbias.info/welcome/robvis-visualization-tool (accessed on 22 September 2022).
- Anand, P.; Kunnumakkara, A.; Newman, R. Bioavailability of curcumin: Problems and promises. Mol. Pharm. 2007, 4, 807–818. [Google Scholar] [CrossRef] [PubMed]
- Jahanbakhshi, F.; Dana, P.M.; Badehnoosh, B.; Yousefi, B.; Mansournia, M.A.; Jahanshahi, M.; Asemi, Z.; Halajzadeh, J. Curcumin anti-tumor effects on endometrial cancer with focus on its molecular targets. Cancer Cell Int. 2021, 21, 120. [Google Scholar] [CrossRef] [PubMed]
- Sharifi-Rad, J.; El Rayess, Y.; Rizk, A.A.; Sadaka, C.; Zgheib, R.; Zam, W.; Sestito, S.; Rapposelli, S.; Neffe-Skocińska, K.; Zielińska, D.; et al. Turmeric and its major compound curcumin on health: Bioactive effects and safety profiles for food, pharmaceutical, biotechnological and medicinal applications. Front. Pharmacol. 2020, 11, 01021. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, S.; Bhattacharjee, P.; Chakraborty, J.; Panda, A.K.; Bandyopadhyay, A.; Banik, S.K.; Sa, G. WITHDRAWN: Curcumin shifts RAS-induced pro-proliferative MEK/ERK-signaling toward pro-apoptotic p38MAPK/JNK1-signaling, triggering p53 activation and apoptosis. J. Biol. Chem. 2017. [Google Scholar] [CrossRef]
- Dou, H.; Shen, R.; Tao, J.; Huang, L.; Shi, H.; Chen, H.; Wang, Y.; Wang, T. Curcumin suppresses the colon cancer proliferation by inhibiting Wnt/β-catenin pathways via miR-130a. Front. Pharmacol. 2017, 8, 877. [Google Scholar] [CrossRef]
- Yance, D.R., Jr.; Sagar, S.M. Targeting angiogenesis with integrative cancer therapies. Integr. Cancer Ther. 2006, 5, 9–29. [Google Scholar] [CrossRef]
- Bagheri, M.; van Nostrum, C.F.; Kok, R.J.; Storm, G.; Hennink, W.E.; Heger, M. Utility of Intravenous Curcumin Nanodelivery Systems for Improving In Vivo Pharmacokinetics and Anticancer Pharmacodynamics. Mol. Pharm. 2022, 19, 3057–3074. [Google Scholar] [CrossRef]
- Liu, Z.; Smart, J.D.; Pannala, A.S. Recent developments in formulation design for improving oral bioavailability of curcumin: A review. J. Drug Deliv. Sci. Technol. 2020, 60, 102082. [Google Scholar] [CrossRef]
- YDizaj, S.M.; Alipour, M.; Abdolahinia, E.D.; Ahmadian, E.; Eftekhari, A.; Forouhandeh, H.; Saadat, Y.R.; Sharifi, S.; Vahed, S.Z. Curcumin nanoformulations: Beneficial nanomedicine against cancer. Phytotherapy Res. PTR 2022, 36, 1156–1181. [Google Scholar] [CrossRef]
- Ramezani Farani, M.; Azarian, M.; Heydari Sheikh Hossein, H.; Abdolvahabi, Z.; Mohammadi Abgarmi, Z.; Moradi, A.; Mousavi, S.M.; Ashrafizadeh, M.; Makvandi, P.; Saeb, M.R.; et al. Folic Acid-Adorned Curcumin-Loaded Iron Oxide Nanoparticles for Cervical Cancer. ACS Appl. Bio Mater. 2022, 5, 1305–1318. [Google Scholar] [CrossRef] [PubMed]
- Hafez Ghoran, S.; Calcaterra, A.; Abbasi, M.; Taktaz, F.; Nieselt, K.; Babaei, E. Curcumin-Based Nanoformulations: A Promising Adjuvant towards Cancer Treatment. Molecules 2022, 27, 5236. [Google Scholar] [CrossRef] [PubMed]



| Study | Population | Intervention | Outcomes | ||
|---|---|---|---|---|---|
| Author, Year/Country | Animal Model | Treatment Regimen | Nanostructure Platform | Pharmacokinetic Analysis | Antitumoral and/or Toxicity Analysis |
| Aqil et al., 2017/USA [11] | Pharmacokinetics analysis: Female Sprague-Dawley rats (n = 4) Antitumoral analysis: Athymic nude mice (n = 5–8) 5 × 106 CaSki cells/subcutaneous injection | Pharmacokinetics analysis: CUR (2.5 mg/kg) and, ExoCUR (1.25, and 2.5 mg/kg) daily by oral gavage for 14 days. Antitumoral analysis: Exo (80 mg/kg), ExoCUR (CUR-20 mg/kg) and Free CUR (60 mg/kg) by oral gavage 3x/week on alternate days. | Exo: HD: 84 ± 7 nm PDI: 0.19 ± 0.02 ExoCUR: PDI: 0.21± 0.04 HD: 93 ± 6 nm DL: 18–24% EE: 53.9 ± 6.7% | ↑ [CUR] in liver (~20 ng/g), lung (~28 ng/g), and brain (~12 ng/g) → (ExoCUR 2.5 mg/kg; p < 0.05). ↑ 6-fold [CUR] in the brain → (ExoCUR 2.5 mg/kg) | Cervical tumor xenograft: ↑ 61% of TGI → (ExoCUR; 20 mg/kg; p < 0.001) ↑ 21% of TGI → (Exo) |
| Chen et al., 2019/China [12] | Pharmacokinetics analysis: Kunming strain mice (n = 6) Antitumoral analysis: Male mice (n = 6) 2 × 107 HCa-F cells/subcutaneous injection on right axilla | Pharmacokinetics analysis: CUR, CPN and CPTN (10 mg/kg) by tail vein single injection. Time of analysis: 0.5, 1, 4, 8, 12, 24 and 36 h. Antitumoral analysis: CUR, CPN and CPTN (10 mg/kg) by tail vein injection daily for 7 days. | CPN: HD: 327.9 ± 14.5 nm ZP: −14.8 ±1.6 mV PDI: <0.21 DL: 6.3 ± 1.1% EE: 71.6 ± 3.4% CPTN: HD: 110.6 ± 2.3 nm ZP: −23.6 ± 2.7 mV PDI: <0.06 DL: 10.1 ± 1.5% EE: 83.2 ± 2.7% | ↑ [CUR] in mice livers → (CPTN, 47 µg/g, 4 h) ↑ CUR AUC → (CPTN, 805.75 μg/mL; p < 0.05) ↑ CUR liver TI → (CPTN; p < 0.05) | Liver cancer: ↓ 31.1% of TV → (CUR) ↓ 56.2% of TV → (CPN; p < 0.05) ↓ 84.4% of TV → (CPTN; p < 0.05) No impact on mice body weight and no damage to major organs → (CPN and CPTN) ↓ 20% of mice body weight → (CUR) |
| Guo et al., 2019/China [22] | Pharmacokinetics analysis: Sprague-Dawley rats (n = 15) Antitumoral analysis: BALB/c nude female mice 107 MCF-7 cells/subcutaneous injection on left armpit | Pharmacokinetics analysis: Free CUR-DMSO, CUR-NP and CUR-P-NP (1.5 mg/kg) by tail vein single injection. Time of analysis: 8 h, 12 h, and 24 h. Tumor biodistribution analysis: CUR-DMSO, CUR-PCL-NPs and CUR-P-PCL-NPs (0.20 mg/kg of CUR) by i.v. single injection. Time of analysis: 2 h, and 6 h. | CUR-NP: HD: 143.9 ± 0.9 PDI: 0.088 ± 0.024 ZP: −14.6 ± 0.6 DL: 9.65 ± 0.32% EE: 94.37 ± 1.36% CUR-P-NP: HD: 176.9 ± 0.5 PDI: 0.116 ± 0.037 ZP: 8.1 ± 0.7 DL: 7.44 ± 0.16% EE: 87.07 ± 0.63% | 1.11% h−1 of CUR release at pH = 7.4 → (CUR-P-NP) 0.939% h−1 of CUR release at pH = 7.4 → (CUR-NP) ↑ T1/2 of CUR → (CUR-P-NP = 0.1229 ± 0.0457 h) ↓ T1/2 of CUR → (CUR-DMSO = 0.0743 ± 0.0349 h) ↑ [CUR] on tumor tissue at 2, 6 and 24 h after injection → (CUR-DMSO, CUR-NP and CUR-P-NP, respectively) | |
| Guo et al., 2020/China [19] | Pharmacokinetics and antitumoral analysis: BALB/c male mice (n = 6) 107 A549 cells/subcutaneous injection on each left fore | Pharmacokinetics analysis: Cur-DMSO, Cur-NPs and Cur-P-NPs (50 µg/mL of CUR) by tail vein single injection. Time of analysis: 1 h, 6 h, and 24 h) Antitumor effect: CUR-DMSO, CUR-NPs and CUR-P-NPs (0.8 mg/kg) by tail vein injection every 2 days for 15 days. | CUR-P-NPs: HD: 215 ± 6.183 nm PDI: 0.141 ± 0.013 ZP: 5.90 ± 0.424 mV DL: 7.11% EE: 82.48% Free P-NPs: HD: 184 ± 2.548 nm PDI: 0.139 ± 0.019 ZP: 4.53 ± 0.512 mV | Collagenase IV accelerates CUR release on CUR-P-NPs group. Tumor 1 h → [CUR-DMSO] 0.139 ng/g, [CUR-NPs] 26.524 ng/g and [CUR-P-NPs] 38.490 ng/g (p < 0.001) Liver 1 h → [CUR-NPs] (6.267 ng/g) and [CUR-P-NPs] (4.292 ng/g) Lung 1 h → [CUR-NPs] (4.476 ng/g) and [CUR-P-NPs] (15.633 ng/g). Tumor 24 h → [Cur-NPs] (4.448 ng/g) and [Cur-P-NPs] (10.380 ng/g). | Lung cancer: ↑ 19.68% of TGI → (Cur-DMSO); ↑ 66.62% of TGI → (Cur-NPs; p < 0.01); ↑ 76.95% of TGI → (Cur-P-NPs; p < 0.001); Presence of tumor necrosis → (Cur-NPs and Cur-P-NPs). |
| Guorgui et al., 2018/US [20] | Pharmacokinetic analysis: BALB/c female mice Antitumoral analysis: SCID female mice (n = 8) 5 × 106 L-540 cells/subcutaneous injection | Pharmacokinetics analysis: Free CUR, SLN-CUR and TPGS-CUR (100 mg/kg) by oral single dose. Time of analysis: 0.5, 1, 2 and 4 h. Antitumoral analysis: Free CUR, SLN-CUR and TPGS-CUR (100 mg/kg) orally 5x/week for 18 days. | SLN-CUR: HD: 125.2 nm PDI: 0.268 ± 0.005 ZP: −19.4 ± 2.2 mV TPGS-CUR: HD: 285 nm PDI: 0.283 ± 0.006 ZP: −21.2 ± 2.6 mV | ↑ SLN-CUR and TPGS-CUR AUC (1.508 and 1.042 ng/mL, respectively) ↓ Free CUR AUC (231.5 ng/mL) ↑ Tumor accumulation of SLN-CUR and TPGS-CUR (4.5 and 3.0-fold increase, respectively; p < 0.05) | Hodgkin’s lymphoma: ↓ 50.5% of TG → (SLN-Cur; p < 0.02) ↓ 43% of TG → (TPGS-Cur; p < 0.04) No significative difference of TG on TPGS-CUR and SLN-CUR compared to free CUR treatment (p = 0.30 and p = 0.13, respectively) No toxicity of SLN-CUR and TPGS-CUR. |
| Hong et al., 2017/China [7] | Pharmacokinetics Sprague male rats (n = 10) Antitumoral (n = 10) and biodistribution (n = 5) analysis ICR mice 107 H22 cells/intraperitoneal injection | Pharmacokinetic analysis: Free CUR and CUR-NSps (10 mg/kg) by i.v. single injection. Time of analysis: 0.08 h, 0.16 h, 0.33 h, 0.5 h, 1 h, 2 h, 4 h, 8 h, 12 h e 24 h. -Biodistribution analysis: Free CUR and CUR-NSps (8 mg/kg) by i.v. single injection. Time of analysis: 0.5, 2, 4, 8, 12, and 24 h. -Antitumoral analysis: Free CUR and CUR-NSps (10 mg/kg) by i.v injection on alternate days for 6 days. | CUR-NSps: HD: 186.33 ± 2.73 nm PDI: 0.22 ZP: −19.00 ± 1.31 mV | ↑ T1/2 35.95-fold → (CUR-NSps; p < 0.001) ↑ AUC0–24 4.5-fold → (CUR-NSps); p < 0.05) ↑ MRT 18.90-fold → (CUR-NSps); p < 0.01) ↑ [CUR] on liver, spleen, kidney, brain, and tumor → (CUR-NSps) No detection of plasma [CUR] after 240 min; → (Free-CUR) | Liver cancer ↑ TIR 70.34% → (CUR-NSps; 10 mg/kg; p < 0.001) ↑ TIR 40.03% → (CUR; 10 mg/kg; p < 0.05) ↑ TIR 55.98% → (CUR- NSps; 5 mg/kg; p < 0.05) ↑ TIR 53.21% → (CUR-NSps; 2.5 mg/kg; p < 0.05) <5% of hemolysis → (CUR-NSps) |
| Hu et al., 2020/China [13] | Pharmacokinetic analysis: BALB/C female mice Antitumoral analysis: BALB/C female mice 1 × 106 CT26 cells inoculated at right flank | Pharmacokinetic analysis: Free CUR, Nano-CUR and FA/Nano-CUR (50 mg/kg) by jugular vein single injection. Time of analysis: 5 h, 10 h, 15 h, 20 h, 25 h, and 30 h. Antitumoral analysis: Free CUR, Nano-CUR, and FA/Nano-CUR (50 mg/kg) by i.v. injection every 2 days for 18 days. | FA/Nano-CUR: HD: 30.47 ± 0.65 nm PDI: 0.17 ZP: −3.55 mV DL: 10% EE: 98% DR: 46.32% Nano-CUR: DR: 44.53% | ↑ T1/2 of 1.58 and 1.42 h → (FA/Nano-CUR and Nano-CUR, respectively) ↓ T1/2 of 0.81 h → (Free-CUR) ↑ AUC of 579.1 and 478.6 µg·mL h → (FA/Nano-CUR and Nano-CUR, respectively) ↓ AUC of 62.28 µg·mL h → (Free-CUR) | Colon-rectal cancer: ↑ TIR of 77.32% → (FA/Nano-CUR) ↓ TIR of 20.77 and 16.25% → (Nano-CUR and Free-CUR, respectively) ↓ PCNA index → (Free-CUR, Nano-Cur and FA/Nano-CUR) ↓ Microvessel density → (FA/Nano-CUR) No toxicity in all treatments |
| Ji et al., 2020/China [23] | Pharmacokinetic analysis: Sprague-Dawley female rats (n = 3) Antitumoral analysis: BALB/c mice female (n = 5) 106 4T1 cells/subcutaneous injection on right flank | Pharmacokinetic analysis: Free CUR, CUR-NC, and CUR-NCHA (2 mg/kg) by tail vein single injection. Time of analysis: 0.25 h, 0.5 h, 1 h, 3 h, 6 h, 12 h, and 24 h. Antitumoral analysis: Free CUR, CUR-NC and CUR-NCHA (5 mg/kg) by i.v. injection every 2 days for 10 days. | CUR-NC: HD: 101.4 ± 7.4 nm PDI: 0.33 CUR-NCHA: HD: 161.9 ± 1.7 nm PDI: 0.25 ZP: −25.0 ± 0.8 mV | ↑ T1/2 of 53.06 ± 18.21 h (CUR-NCHA; p < 0.0001) ↓ T1/2 of 11.14 ± 1.63 h (Free-CUR) <40% CUR release on pH 7.4 (CUR-NCHA) ~80% CUR release on pH 5.0 (CUR-NCHA) | Breast cancer: Deceleration of TG → (↓ CUR-NC and ↓↓ CUR-NCHA; p < 0.05) ↓ TW → (CUR-NCHA) No hemolysis of blood cells → (CUR-NCHA); No damage to major organs → (CUR-NCHA) |
| Sun et al., 2020/China [18] | Pharmacokinetic analysis: Male Sprague-Dawley rats (n = 5) Antitumoral analysis: Balb/c nude xenograft mice 1 × 106 Hela cells/subcutaneous injection on right flank | Pharmacokinetic analysis: Free CUR, CUR-PLGA-NPs and CUR-HPB PLGA-NPs (10 mg/kg) by tail vein single injection. Time of analysis: 0.083, 0.167, 0.33, 0.67, 1, 2, and 3 h. Antitumoral analysis: I.V. administration of CUR (20 mg/kg); PLGA NPs, HPB PLGA NP (10 and 20 mg/kg) every 3 days for 15 days injections by i.v. solution. | PLGA-NPs: HD: 112.8 ± 44 ZP: −6.06 EE: 8.2 ± 0.3% HPB-PLGA NPs: HD: 144.0 ± 44.8 ZP: −1.94 EE: 7.9 ± 0.2% | ↑ AUC0–2 at 0.08 h → (PLGA NPs) ↑ Plasma maximum [CUR] of 13.29 ± 4.473 µg/mL → (PLGA NPs) ↓ Plasma maximum [CUR] of 1.804 ± 0.256 μg/mL → (Free-CUR) Plasma maximum [CUR] 10.425 ± 2.13 μg/mL → (HPB-PLGA NPs) | Cervical cancer ↑ ~75% TGI → (HPB PLGA NPs; 10 or 20 mg/kg) |
| Wang et al., 2017/China [16] | Pharmacokinetic analysis: Sprague-Dawley male rats (n = 5) Antitumoral analysis: C57BL/6 female mice 106 B16F10 cells/subcutaneous injection on right flank BALB/c female mice 107 A375 cells subcutaneous injection on right flank | Pharmacokinetic analysis: Free CUR and CUR-MPEG-PLA (50 mg/kg) by tail vein single injection. Time of analysis: 120 h, 240 h, 360 h, 480 h, 600 h, and 700 h. Antitumoral analysis: Free CUR and CUR-MPEG-PLA (50 mg/kg) | CUR-MPEG-PLA: HD: 34.5 nm PDI: 0.13 ZP: −2.3 mV EE: 98.8% DL: 10% | ↑ 80.6 ± 6.1% CUR release at pH 7.4 after 12 h → (CUR-MPEG-PLA) ↑ AUC: 498.5 mg/L h → (CUR-MPEG-PLA) | B16 melanoma: ↓ 79.4% TV (CUR-MPEG-PLA; p < 0.01) ↓ TW (CUR-MPEG-PLA; p < 0.01) A375 melanoma: ↓ 68.4% TV (CUR-MPEG-PLA; p < 0.01) ↓ TW (CUR-MPEG-PLA; p < 0.01) ↓ Number of microvessels (CUR-MPEG-PLA; p < 0.05) |
| Xie et al., 2020/China [14] | Pharmacokinetic analysis: Male rats (n = 6) Antitumoral analysis: C57BL/6J male mice (n = 6) 106 SCLC H446 cells subcutaneous injection on the left flank | Pharmacokinetic analysis: Free CUR, CLN-CUR and LN-CUR (45 mg/kg) by oral single dose. Time of analysis: 0.5 h, 1 h, 1.5 h, 20 h, 40 h, 60 h, and 80 h. Antitumoral analysis: Free CUR, CLN-CUR and LN-CUR (10 mg/kg) by oral daily dose for 30 consecutively days | CLN-CUR: HD: ~550.1 nm ZP: ~12.7 mV EE: ~79.23% | ↑ Gastrointestinal absorption at 1.35 and 1.34-fold compared to free CUR → (CLN-CUR and LN-CUR, respectively; p < 0.05) ↑ AUC at 8.94 and 1.38-fold compared to free CUR → (CLN-CUR and LN-CUR, respectively; p < 0.05) | Small Cell Lung Cancer: ↓ 58.39% of TV and 59.70% of TW → (CLN-CUR; p < 0.01) ↓ 34.43% of TV and 29.21% of TW → (free-CUR; p < 0.01) No damage or inflammation in major organs → All treatments |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Silvestre, F.; Santos, C.; Silva, V.; Ombredane, A.; Pinheiro, W.; Andrade, L.; Garcia, M.; Pacheco, T.; Joanitti, G.; Luz, G.; et al. Pharmacokinetics of Curcumin Delivered by Nanoparticles and the Relationship with Antitumor Efficacy: A Systematic Review. Pharmaceuticals 2023, 16, 943. https://doi.org/10.3390/ph16070943
Silvestre F, Santos C, Silva V, Ombredane A, Pinheiro W, Andrade L, Garcia M, Pacheco T, Joanitti G, Luz G, et al. Pharmacokinetics of Curcumin Delivered by Nanoparticles and the Relationship with Antitumor Efficacy: A Systematic Review. Pharmaceuticals. 2023; 16(7):943. https://doi.org/10.3390/ph16070943
Chicago/Turabian StyleSilvestre, Fernanda, Carolina Santos, Vitória Silva, Alicia Ombredane, Willie Pinheiro, Laise Andrade, Mônica Garcia, Thyago Pacheco, Graziella Joanitti, Glécia Luz, and et al. 2023. "Pharmacokinetics of Curcumin Delivered by Nanoparticles and the Relationship with Antitumor Efficacy: A Systematic Review" Pharmaceuticals 16, no. 7: 943. https://doi.org/10.3390/ph16070943
APA StyleSilvestre, F., Santos, C., Silva, V., Ombredane, A., Pinheiro, W., Andrade, L., Garcia, M., Pacheco, T., Joanitti, G., Luz, G., & Carneiro, M. (2023). Pharmacokinetics of Curcumin Delivered by Nanoparticles and the Relationship with Antitumor Efficacy: A Systematic Review. Pharmaceuticals, 16(7), 943. https://doi.org/10.3390/ph16070943







