Differential Effects of Cytokine Versus Hypoxic Preconditioning of Human Mesenchymal Stromal Cells in Pulmonary Sepsis Induced by Antimicrobial-Resistant Klebsiella pneumoniae
Abstract
1. Introduction
2. Results
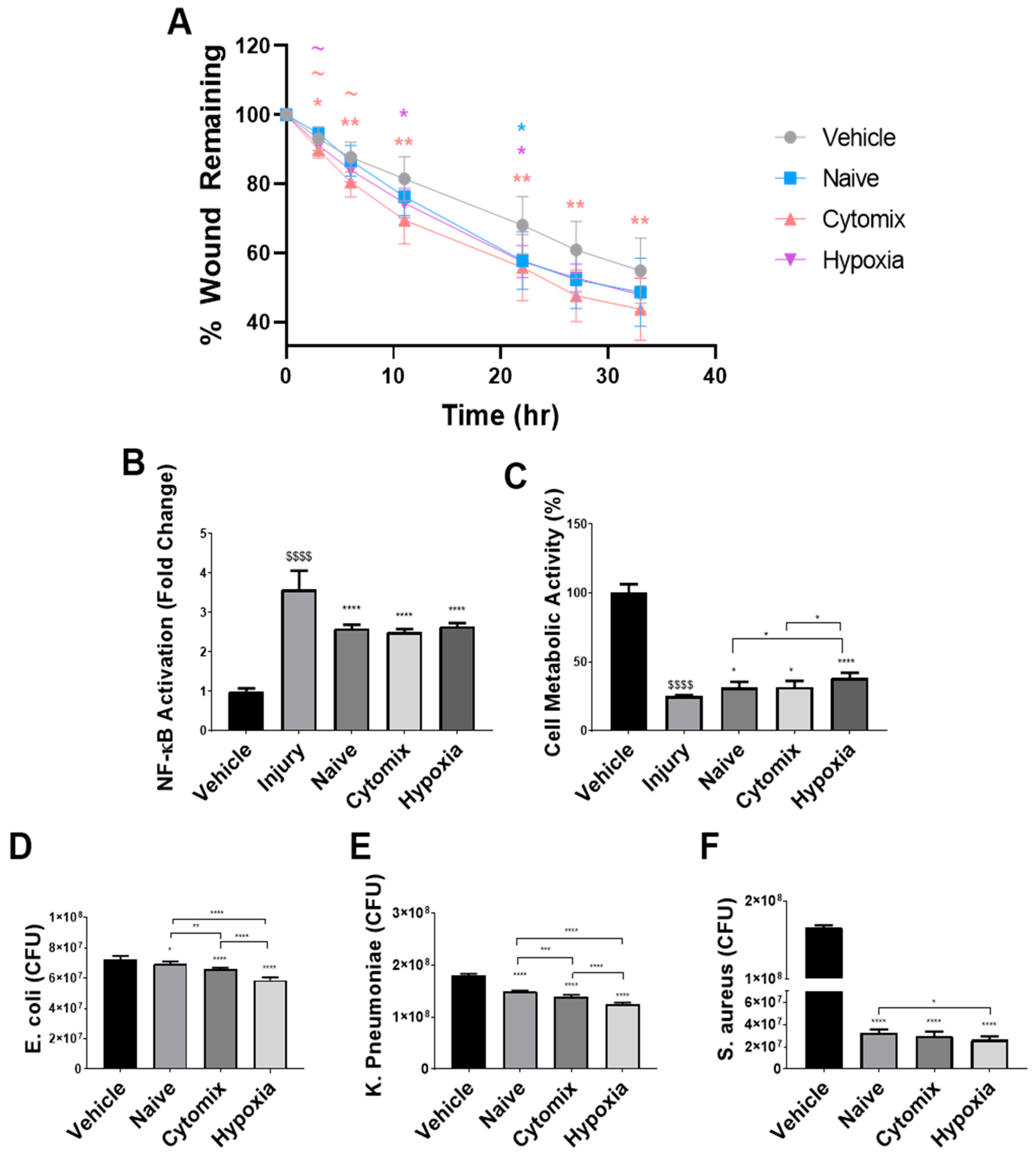
2.1. Pre-Activation Enhances the Functionality of UC-MSCs In Vitro
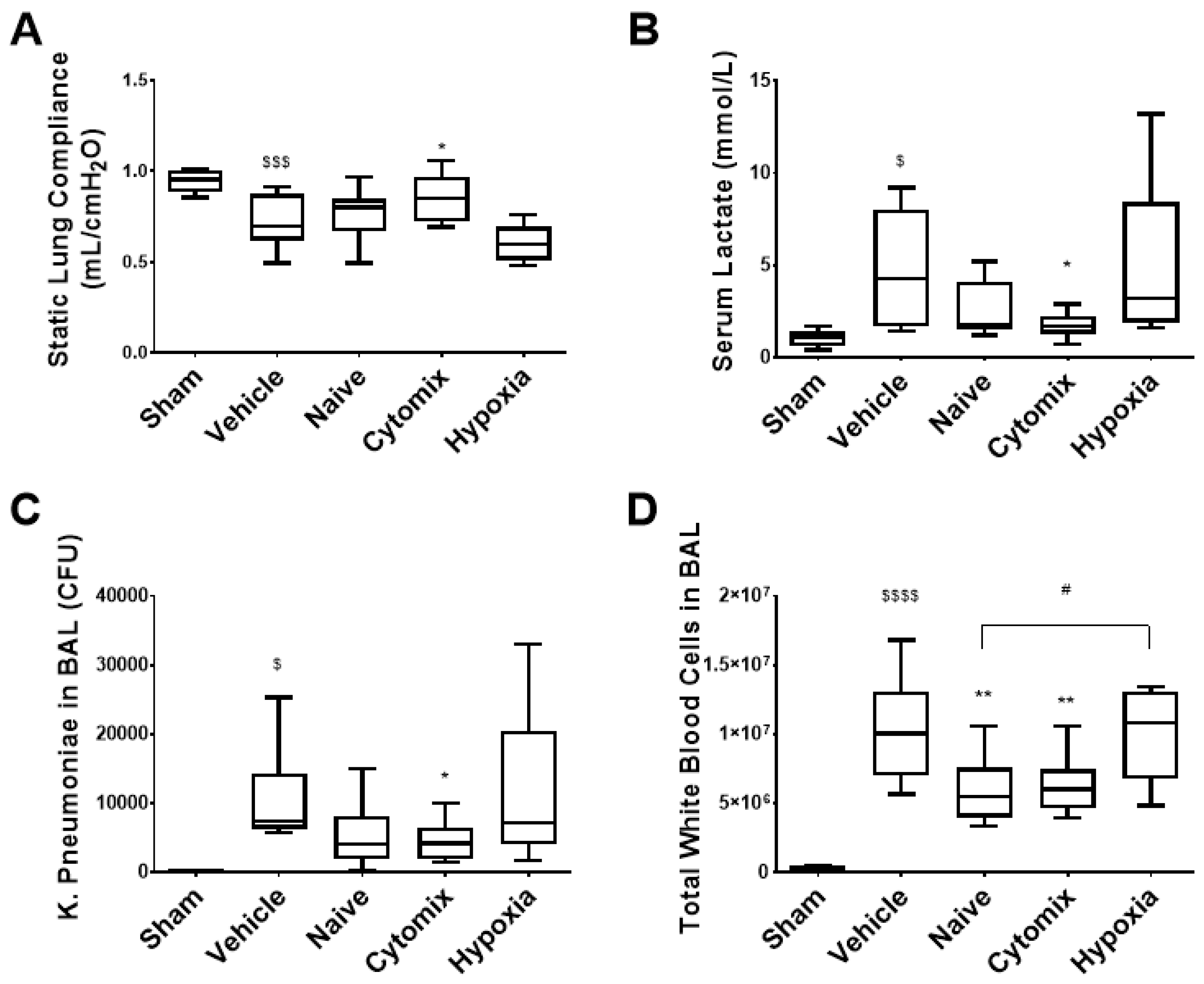
2.2. Pre-Activation of MSCs Attenautes Physiological Dysfunction in Established Klebsiella pneumosepsis
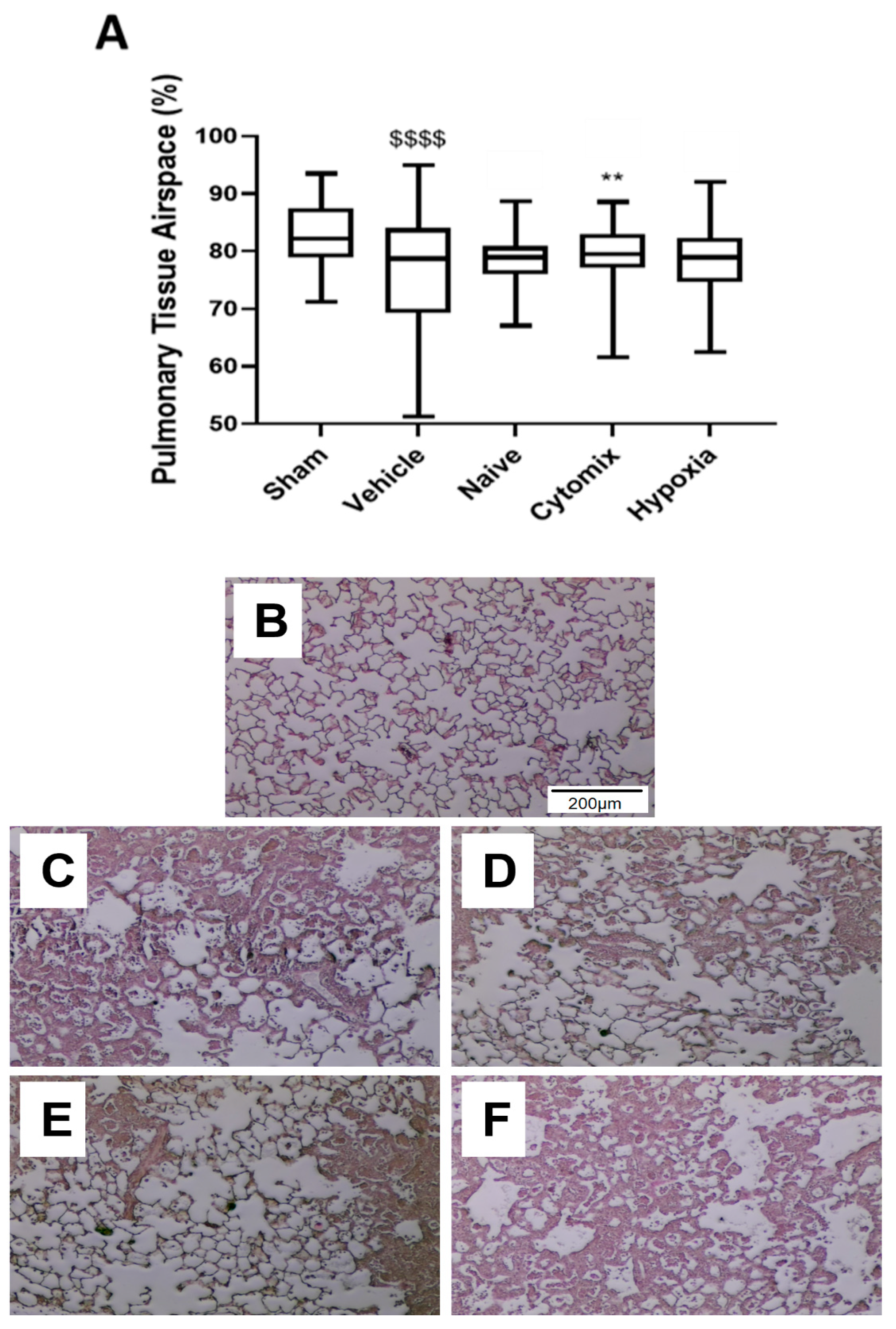
2.3. Naïve and Cytomix-Exposed MSCs Attenuate K. pneumonia-Induced Histologic Injury
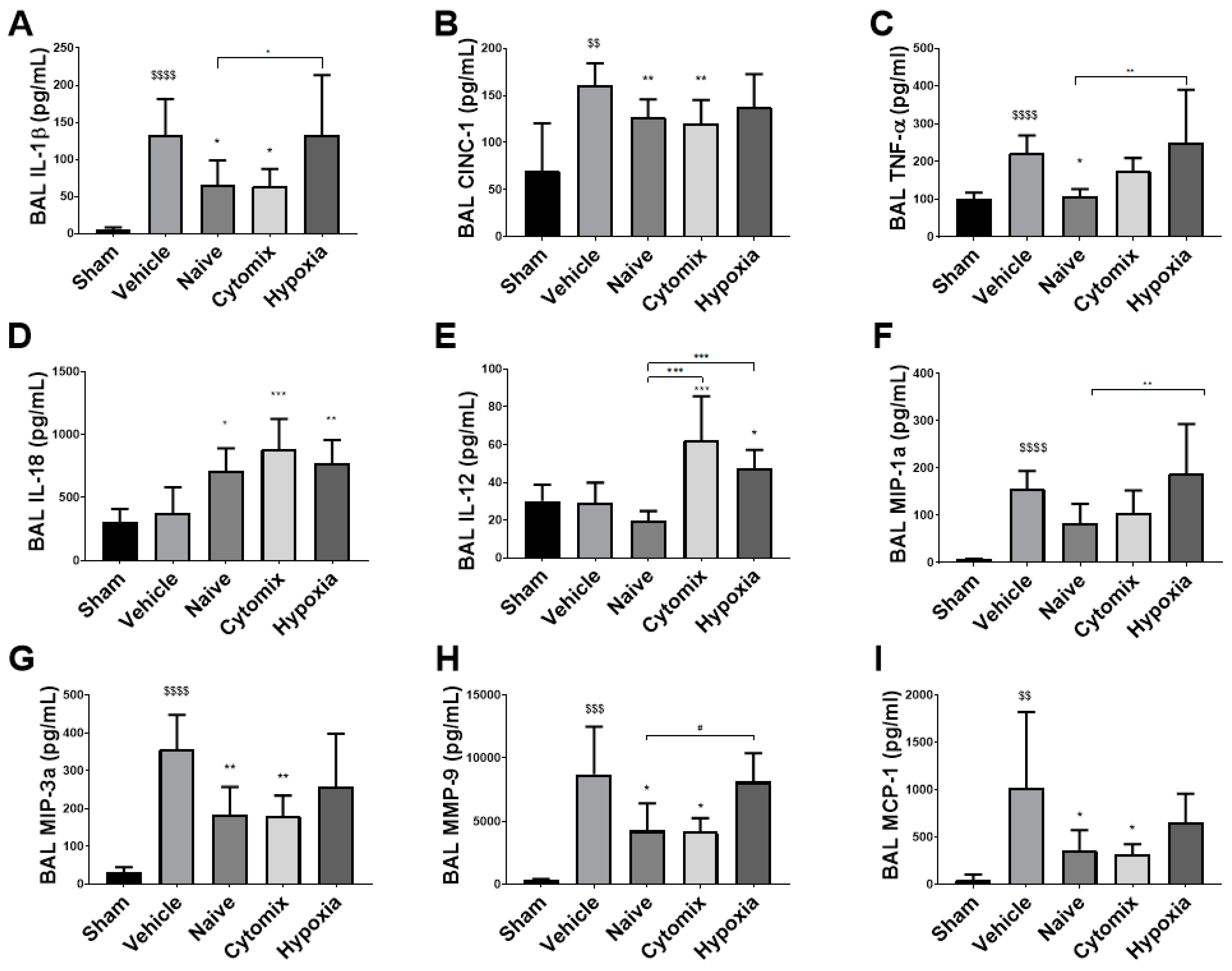
2.4. MSCs Differentually Attenuate the Pulmonary Cytokine Response to K. penumonia Infection
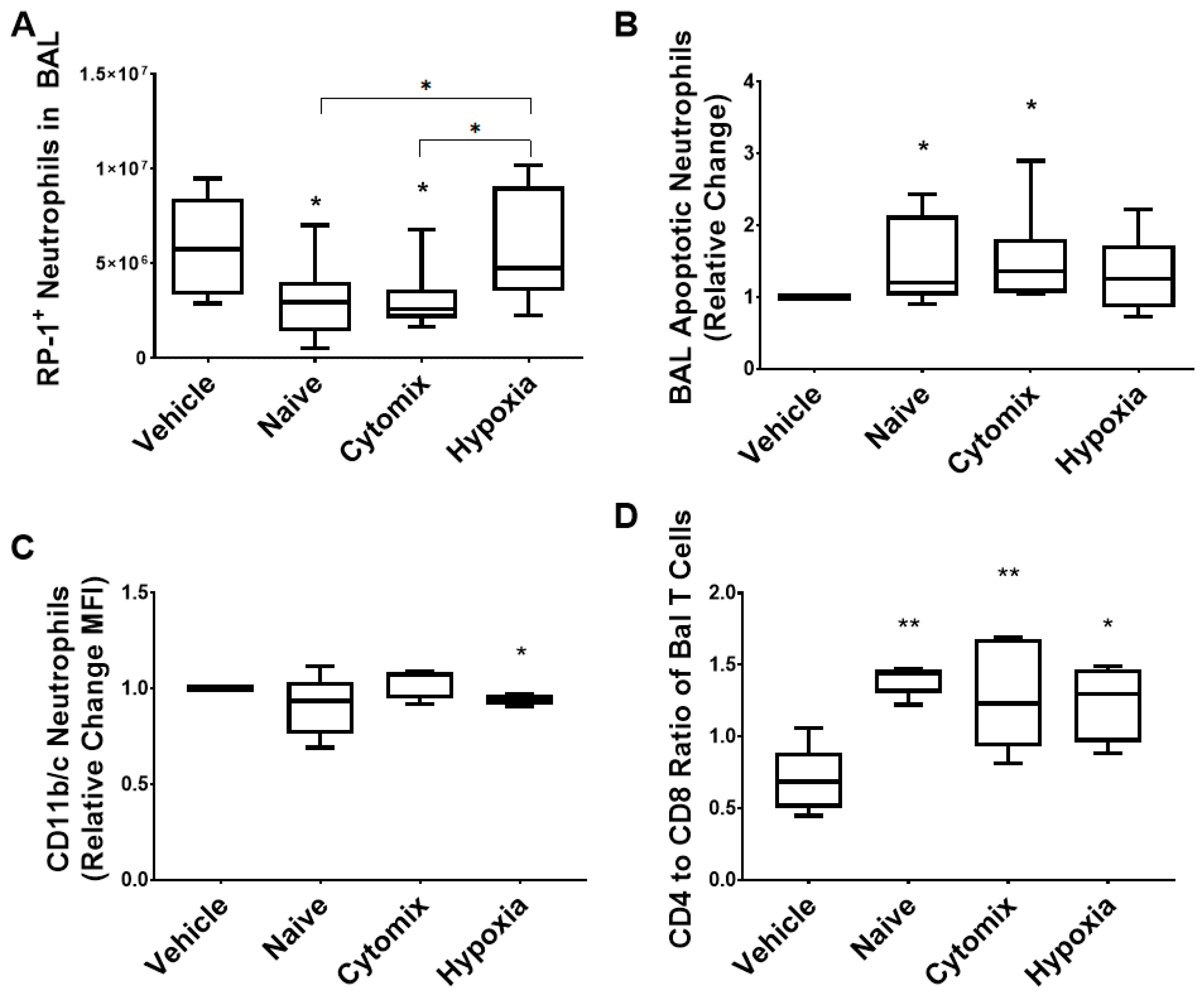
2.5. BAL White Cell Subsets Indicate a Resolution of Pneumonia Following MSC Administration
2.6. BAL Macrophages/Monocytes Show Enhanced Cell Function Following MSC Administration
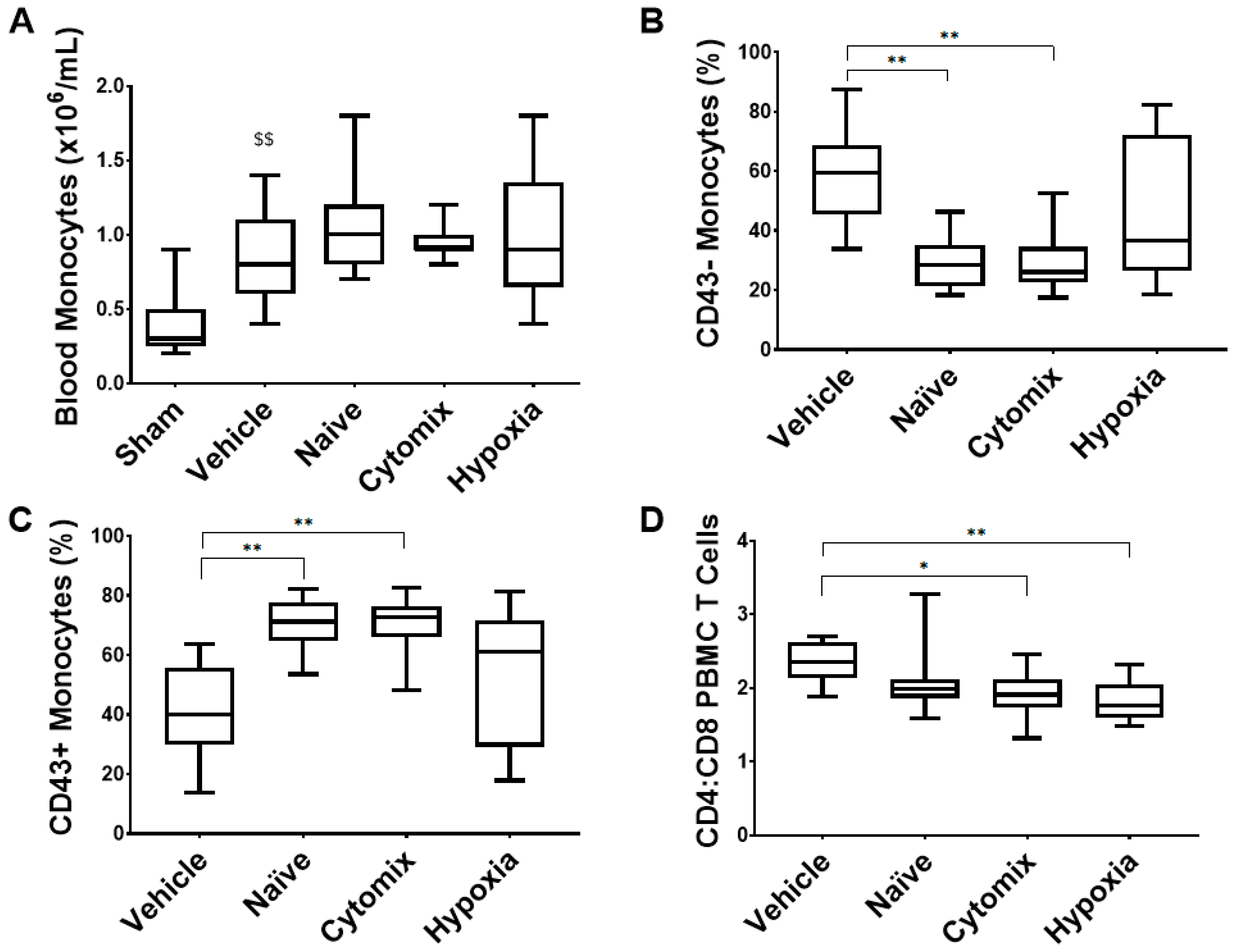
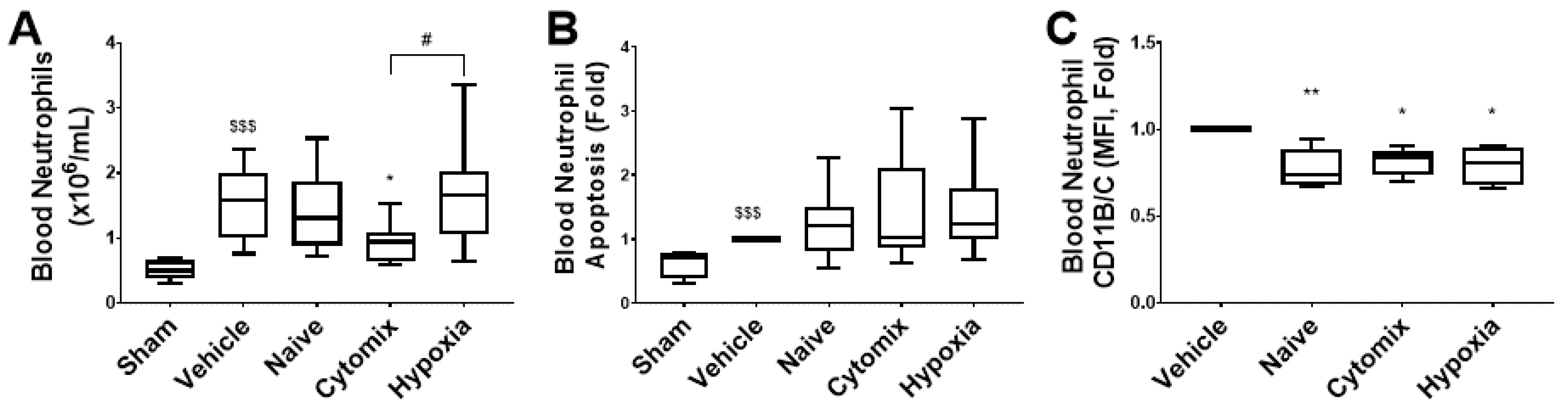
2.7. Systemic Leukocyte and Lymphocyte Number and Function Are Altered by the Administration of Naïve and Cytomix-Pre-Activated MSCs
3. Discussion
4. Materials and Methods
4.1. Human Umbilical Cord-Derived MSCs
4.2. In Vitro Assay Panels to Ascertain MSC Functionality
4.2.1. Wound-Healing Assay
4.2.2. Nuclear Factor κB Activation Assay
4.2.3. Cell Metabolic Activity
4.2.4. Inflammatory Cytokine Production
4.2.5. Bacterial-Killing Assay
4.3. Preclinical Experimental Series
4.3.1. Established Pneumonia
4.3.2. Experimental Design
4.3.3. Premortem Assessment
4.4. Postmortem Assessment
4.4.1. Bacterial Load
4.4.2. Inflammatory Cytokine Profile
4.4.3. Histological Analyses
4.4.4. Flow Cytometry Analysis of PBMCs
4.4.5. Flow Cytometry Analysis of Neutrophils
4.4.6. BAL Macro/Monocyte Phagocytosis and Superoxide Anion Production
4.4.7. BAL Macro/Monocyte Quantification
4.5. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Global Action Plan on Antimicrobial Resistance; World Health Organization: Geneva, Switzerland, 2016; p. 45. Available online: https://www.who.int/publications/i/item/9789241509763 (accessed on 11 November 2022).
- D’Accolti, M.; Soffritti, I.; Mazzacane, S.; Caselli, E. Fighting AMR in the Healthcare Environment: Microbiome-Based Sanitation Approaches and Monitoring Tools. Int. J. Mol. Sci. 2019, 20, 1525. [Google Scholar] [CrossRef] [PubMed]
- Resistance, T.R.O.A. Tackling Drug Resistant Infections Globally: Final Report and Recommendations. 2016. Available online: https://amr-review.org/Publications.html (accessed on 11 November 2022).
- Devaney, J.; Horie, S.; Masterson, C.; Elliman, S.; Barry, F.; O’Brien, T.; Curley, G.F.; O’Toole, D.; Laffey, J.G. Human mesenchymal stromal cells decrease the severity of acute lung injury induced by E. coli in the rat. Thorax 2015, 70, 625–635. [Google Scholar] [CrossRef]
- Sung, D.K.; Chang, Y.S.; Sung, S.I.; Yoo, H.S.; Ahn, S.Y.; Park, W.S. Antibacterial effect of mesenchymal stem cells against Escherichia coli is mediated by secretion of beta- defensin- 2 via toll- like receptor 4 signalling. Cell Microbiol. 2016, 18, 424–436. [Google Scholar] [CrossRef] [PubMed]
- Kniazev, O.V.; Ruchkina, I.N.; Parfenov, A.I.; Konopliannikov, A.G.; Sagynbaeva, V.E. [Complete elimination of cytomegalovirus without antiviral therapy after systemic transplantation of mesenchymal bone marrow stromal cells in a patient with ulcerative colitis]. Eksp Klin Gastroenterol. 2012, 118–123. [Google Scholar]
- Spekker, K.; Leineweber, M.; Degrandi, D.; Ince, V.; Brunder, S.; Schmidt, S.K.; Stuhlsatz, S.; Howard, J.C.; Schares, G.; Degistirici, O.; et al. Antimicrobial effects of murine mesenchymal stromal cells directed against Toxoplasma gondii and Neospora caninum: Role of immunity-related GTPases (IRGs) and guanylate-binding proteins (GBPs). Med. Microbiol. Immunol. 2013, 202, 197–206. [Google Scholar] [CrossRef]
- Alcayaga-Miranda, F.; Cuenca, J.; Khoury, M. Antimicrobial Activity of Mesenchymal Stem Cells: Current Status and New Perspectives of Antimicrobial Peptide-Based Therapies. Front. Immunol. 2017, 8, 339. [Google Scholar] [CrossRef] [PubMed]
- Hayes, M.; Masterson, C.; Devaney, J.; Barry, F.; Elliman, S.; O’Brien, T.; O’Toole, D.; Curley, G.F.; Laffey, J.G. Therapeutic efficacy of human mesenchymal stromal cells in the repair of established ventilator-induced lung injury in the rat. Anesthesiology 2015, 122, 363–373. [Google Scholar] [CrossRef]
- Matthay, M.A.; Calfee, C.S.; Zhuo, H.; Thompson, B.T.; Wilson, J.G.; Levitt, J.E.; Rogers, A.J.; Gotts, J.E.; Wiener-Kronish, J.P.; Bajwa, E.K.; et al. Treatment with allogeneic mesenchymal stromal cells for moderate to severe acute respiratory distress syndrome (START study): A randomised phase 2a safety trial. Lancet Respir. Med. 2019, 7, 154–162. [Google Scholar] [CrossRef]
- Li, H.; Zhu, H.; Ge, T.; Wang, Z.; Zhang, C. Mesenchymal Stem Cell-Based Therapy for Diabetes Mellitus: Enhancement Strategies and Future Perspectives. Stem Cell Rev. Rep. 2021, 17, 1552–1569. [Google Scholar] [CrossRef]
- Alagesan, S.; Brady, J.; Byrnes, D.; Fandino, J.; Masterson, C.; McCarthy, S.; Laffey, J.; O’Toole, D. Enhancement strategies for mesenchymal stem cells and related therapies. Stem Cell Res. Ther. 2022, 13, 75. [Google Scholar] [CrossRef]
- Han, J.; Liu, Y.; Liu, H.; Li, Y. Genetically modified mesenchymal stem cell therapy for acute respiratory distress syndrome. Stem Cell Res. Ther. 2019, 10, 386. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Jiang, Y.; Hou, Q.; Zhao, Y.; Zhong, L.; Fu, X. Potential pre-activation strategies for improving therapeutic efficacy of mesenchymal stem cells: Current status and future prospects. Stem. Cell Res. Ther. 2022, 13, 146. [Google Scholar] [CrossRef] [PubMed]
- Thompson, A.A.; Dickinson, R.S.; Murphy, F.; Thomson, J.P.; Marriott, H.M.; Tavares, A.; Willson, J.; Williams, L.; Lewis, A.; Mirchandani, A.; et al. Hypoxia determines survival outcomes of bacterial infection through HIF-1alpha dependent re-programming of leukocyte metabolism. Sci. Immunol. 2017, 2, eaal2861. [Google Scholar] [CrossRef]
- Matthay, M.A.; Zemans, R.L. The acute respiratory distress syndrome: Pathogenesis and treatment. Annu. Rev. Pathol. 2011, 6, 147–163. [Google Scholar] [CrossRef]
- Rustad, K.C.; Gurtner, G.C. Mesenchymal Stem Cells Home to Sites of Injury and Inflammation. Adv. Wound Care 2012, 1, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Masterson, C.H.; Tabuchi, A.; Hogan, G.; Fitzpatrick, G.; Kerrigan, S.W.; Jerkic, M.; Kuebler, W.M.; Laffey, J.G.; Curley, G.F. Intra-vital imaging of mesenchymal stromal cell kinetics in the pulmonary vasculature during infection. Sci. Rep. 2021, 11, 5265. [Google Scholar] [CrossRef] [PubMed]
- Meakins, J.; Long, C.N. Oxygen Consumption, Oxygen Debt and Lactic Acid in Circulatory Failure. J. Clin. Investig. 1927, 4, 273–293. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Karl, I.E. Reevaluation of the role of cellular hypoxia and bioenergetic failure in sepsis. JAMA 1992, 267, 1503–1510. [Google Scholar] [CrossRef]
- Hespanhol, V.; Barbara, C. Pneumonia mortality, comorbidities matter? Pulmonology 2020, 26, 123–129. [Google Scholar] [CrossRef]
- Assefa, M. Multi-drug resistant gram-negative bacterial pneumonia: Etiology, risk factors, and drug resistance patterns. Pneumonia 2022, 14, 4. [Google Scholar] [CrossRef]
- Capcha, J.M.C.; Rodrigues, C.E.; Moreira, R.S.; Silveira, M.D.; Dourado, P.; Dos Santos, F.; Irigoyen, M.C.; Jensen, L.; Garnica, M.R.; Noronha, I.L.; et al. Wharton’s jelly-derived mesenchymal stem cells attenuate sepsis-induced organ injury partially via cholinergic anti-inflammatory pathway activation. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020, 318, R135–R147. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Mu, G.; Song, C.; Zhou, L.; He, L.; Jin, Q.; Lu, Z. Role of M2 Macrophages in Sepsis-Induced Acute Kidney Injury. Shock 2018, 50, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Pedrazza, L.; Cunha, A.A.; Luft, C.; Nunes, N.K.; Schimitz, F.; Gassen, R.B.; Breda, R.V.; Donadio, M.V.; de Souza Wyse, A.T.; Pitrez, P.M.C.; et al. Mesenchymal stem cells improves survival in LPS-induced acute lung injury acting through inhibition of NETs formation. J. Cell Physiol. 2017, 232, 3552–3564. [Google Scholar] [CrossRef] [PubMed]
- Galipeau, J.; Sensebe, L. Mesenchymal Stromal Cells: Clinical Challenges and Therapeutic Opportunities. Cell Stem Cell 2018, 22, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Hieu Pham, L.; Bich Vu, N.; Van Pham, P. The subpopulation of CD105 negative mesenchymal stem cells show strong immunomodulation capacity compared to CD105 positive mesenchymal stem cells. Biomed. Res. Ther. 2019, 6, 3131–3140. [Google Scholar] [CrossRef]
- Masterson, C.; Devaney, J.; Horie, S.; O’Flynn, L.; Deedigan, L.; Elliman, S.; Barry, F.; O’Brien, T.; O’Toole, D.; Laffey, J.G. Syndecan-2-positive, Bone Marrow-derived Human Mesenchymal Stromal Cells Attenuate Bacterial-induced Acute Lung Injury and Enhance Resolution of Ventilator-induced Lung Injury in Rats. Anesthesiology 2018, 129, 502–516. [Google Scholar] [CrossRef]
- Bouhaddou, M.; Yu, L.J.; Lunardi, S.; Stamatelos, S.K.; Mack, F.; Gallo, J.M.; Birtwistle, M.R.; Walz, A.C. Predicting In Vivo Efficacy from In Vitro Data: Quantitative Systems Pharmacology Modeling for an Epigenetic Modifier Drug in Cancer. Clin. Transl. Sci. 2020, 13, 419–429. [Google Scholar] [CrossRef]
- Rodriguez, L.A., 2nd; Mohammadipoor, A.; Alvarado, L.; Kamucheka, R.M.; Asher, A.M.; Cancio, L.C.; Antebi, B. Preconditioning in an Inflammatory Milieu Augments the Immunotherapeutic Function of Mesenchymal Stromal Cells. Cells 2019, 8, 462. [Google Scholar] [CrossRef]
- Horie, S.; Gaynard, S.; Murphy, M.; Barry, F.; Scully, M.; O’Toole, D.; Laffey, J.G. Cytokine pre-activation of cryopreserved xenogeneic-free human mesenchymal stromal cells enhances resolution and repair following ventilator-induced lung injury potentially via a KGF-dependent mechanism. Intensive Care Med. Exp. 2020, 8, 8. [Google Scholar] [CrossRef]
- Chen, K.; Kolls, J.K. T cell-mediated host immune defenses in the lung. Annu. Rev. Immunol. 2013, 31, 605–633. [Google Scholar] [CrossRef]
- Pechous, R.D. With Friends Like These: The Complex Role of Neutrophils in the Progression of Severe Pneumonia. Front. Cell. Infect. Microbiol. 2017, 7, 160. [Google Scholar] [CrossRef] [PubMed]
- Cesta, M.C.; Zippoli, M.; Marsiglia, C.; Gavioli, E.M.; Mantelli, F.; Allegretti, M.; Balk, R.A. The Role of Interleukin-8 in Lung Inflammation and Injury: Implications for the Management of COVID-19 and Hyperinflammatory Acute Respiratory Distress Syndrome. Front. Pharmacol. 2021, 12, 808797. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Karupiah, G. Targeting tumour necrosis factor to ameliorate viral pneumonia. FEBS J. 2022, 289, 883–900. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.L.; Lee, Y.L.; Chang, K.M.; Chang, G.C.; King, S.L.; Chiang, C.D.; Niederman, M.S. Bronchoalveolar interleukin-1 beta: A marker of bacterial burden in mechanically ventilated patients with community-acquired pneumonia. Crit. Care Med. 2003, 31, 812–817. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, K.; Nakanishi, K.; Tsutsui, H. Interleukin-18 in Health and Disease. Int. J. Mol. Sci. 2019, 20, 649. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.; Xue, Y.; Wang, P.; Lin, L.; Liu, Q.; Li, N.; Xu, J.; Cao, X. IFN-gamma primes macrophage activation by increasing phosphatase and tensin homolog via downregulation of miR-3473b. J. Immunol. 2014, 193, 3036–3044. [Google Scholar] [CrossRef] [PubMed]
- Keane, M.; Belperio, J. Macrophage inflammatory Protein. In Encyclopedia of Respiratory Medicine; Geoffrey, J., Laurent, S.D.S., Eds.; Elsevier Limited: Amsterdam, The Netherlands, 2006; pp. 1–5. [Google Scholar]
- Bradley, L.M.; Douglass, M.F.; Chatterjee, D.; Akira, S.; Baaten, B.J. Matrix metalloprotease 9 mediates neutrophil migration into the airways in response to influenza virus-induced toll-like receptor signaling. PLoS Pathog. 2012, 8, e1002641. [Google Scholar] [CrossRef]
- Singh, S.; Anshita, D.; Ravichandiran, V. MCP-1: Function, regulation, and involvement in disease. Int. Immunopharmacol. 2021, 101, 107598. [Google Scholar] [CrossRef]
- Woodward, E.A.; Prele, C.M.; Nicholson, S.E.; Kolesnik, T.B.; Hart, P.H. The anti-inflammatory effects of interleukin-4 are not mediated by suppressor of cytokine signalling-1 (SOCS1). Immunology 2010, 131, 118–127. [Google Scholar] [CrossRef]
- Silva-Filho, J.L.; Caruso-Neves, C.; Pinheiro, A.A.S. IL-4: An important cytokine in determining the fate of T cells. Biophys. Rev. 2014, 6, 111–118. [Google Scholar] [CrossRef]
- Ross, S.H.; Cantrell, D.A. Signaling and Function of Interleukin-2 in T Lymphocytes. Annu. Rev. Immunol. 2018, 36, 411–433. [Google Scholar] [CrossRef] [PubMed]
- Koya, T.; Takeda, K.; Kodama, T.; Miyahara, N.; Matsubara, S.; Balhorn, A.; Joetham, A.; Dakhama, A.; Gelfand, E.W. RANTES (CCL5) regulates airway responsiveness after repeated allergen challenge. Am. J. Respir. Cell Mol. Biol. 2006, 35, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Steen, E.H.; Wang, X.; Balaji, S.; Butte, M.J.; Bollyky, P.L.; Keswani, S.G. The Role of the Anti-Inflammatory Cytokine Interleukin-10 in Tissue Fibrosis. Adv. Wound Care 2020, 9, 184–198. [Google Scholar] [CrossRef]
- Tanaka, T.; Narazaki, M.; Kishimoto, T. IL-6 in inflammation, immunity, and disease. Cold Spring Harb. Perspect. Biol. 2014, 6, a016295. [Google Scholar] [CrossRef] [PubMed]
- Scheller, J.; Chalaris, A.; Schmidt-Arras, D.; Rose-John, S. The pro- and anti-inflammatory properties of the cytokine interleukin-6. Biochim. Biophys. Acta 2011, 1813, 878–888. [Google Scholar] [CrossRef] [PubMed]
- McClain Caldwell, I.; Hogden, C.; Nemeth, K.; Boyajian, M.; Krepuska, M.; Szombath, G.; MacDonald, S.; Abshari, M.; Moss, J.; Vitale-Cross, L.; et al. Bone Marrow-Derived Mesenchymal Stromal Cells (MSCs) Modulate the Inflammatory Character of Alveolar Macrophages from Sarcoidosis Patients. J. Clin. Med. 2020, 9, 278. [Google Scholar] [CrossRef] [PubMed]
- Connors, T.J.; Ravindranath, T.M.; Bickham, K.L.; Gordon, C.L.; Zhang, F.; Levin, B.; Baird, J.S.; Farber, D.L. Airway CD8(+) T Cells Are Associated with Lung Injury during Infant Viral Respiratory Tract Infection. Am. J. Respir. Cell Mol. Biol. 2016, 54, 822–830. [Google Scholar] [CrossRef]
- McBride, J.A.; Striker, R. Imbalance in the game of T cells: What can the CD4/CD8 T-cell ratio tell us about HIV and health? PLoS Pathog. 2017, 13, e1006624. [Google Scholar] [CrossRef]
- Mukherjee, R.; Kanti Barman, P.; Kumar Thatoi, P.; Tripathy, R.; Kumar Das, B.; Ravindran, B. Non-Classical monocytes display inflammatory features: Validation in Sepsis and Systemic Lupus Erythematous. Sci. Rep. 2015, 5, 13886. [Google Scholar] [CrossRef]
- Kapellos, T.S.; Bonaguro, L.; Gemund, I.; Reusch, N.; Saglam, A.; Hinkley, E.R.; Schultze, J.L. Human Monocyte Subsets and Phenotypes in Major Chronic Inflammatory Diseases. Front. Immunol. 2019, 10, 2035. [Google Scholar] [CrossRef]
- Kiefer, J.; Zeller, J.; Bogner, B.; Horbrand, I.A.; Lang, F.; Deiss, E.; Winninger, O.; Fricke, M.; Kreuzaler, S.; Smudde, E.; et al. An Unbiased Flow Cytometry-Based Approach to Assess Subset-Specific Circulating Monocyte Activation and Cytokine Profile in Whole Blood. Front. Immunol. 2021, 12, 641224. [Google Scholar] [CrossRef] [PubMed]
- Melief, S.M.; Schrama, E.; Brugman, M.H.; Tiemessen, M.M.; Hoogduijn, M.J.; Fibbe, W.E.; Roelofs, H. Multipotent stromal cells induce human regulatory T cells through a novel pathway involving skewing of monocytes toward anti-inflammatory macrophages. Stem Cells 2013, 31, 1980–1991. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Zhang, Y.; Ye, Y.; Zhang, T.; Cheng, J.; Chen, G.; Zhang, Q.; Yang, Y. Umbilical Cord-derived Mesenchymal Stem Cells Instruct Monocytes towards an IL10-producing Phenotype by Secreting IL6 and HGF. Sci. Rep. 2016, 6, 37566. [Google Scholar] [CrossRef] [PubMed]
- Krasnodembskaya, A.; Samarani, G.; Song, Y.; Zhuo, H.; Su, X.; Lee, J.-W.; Gupta, N.; Petrini, M.; Matthay, M.A. Human mesenchymal stem cells reduce mortality and bacteremia in gram-negative sepsis in mice in part by enhancing the phagocytic activity of blood monocytes. Am. J. Physiol. Lung Cell Mol. Physiol. 2012, 302, L1003–L1013. [Google Scholar] [CrossRef]
- Cutler, A.J.; Limbani, V.; Girdlestone, J.; Navarrete, C.V. Umbilical cord-derived mesenchymal stromal cells modulate monocyte function to suppress T cell proliferation. J. Immunol. 2010, 185, 6617–6623. [Google Scholar] [CrossRef]
- Santos, S.S.; Carmo, A.M.; Brunialti, M.K.; Machado, F.R.; Azevedo, L.C.; Assuncao, M.; Trevelin, S.C.; Cunha, F.Q.; Salomao, R. Modulation of monocytes in septic patients: Preserved phagocytic activity, increased ROS and NO generation, and decreased production of inflammatory cytokines. Intensive Care Med. Exp. 2016, 4, 5. [Google Scholar] [CrossRef]
- Bordon, J.; Aliberti, S.; Fernandez-Botran, R.; Uriarte, S.M.; Rane, M.J.; Duvvuri, P.; Peyrani, P.; Morlacchi, L.C.; Blasi, F.; Ramirez, J.A. Understanding the roles of cytokines and neutrophil activity and neutrophil apoptosis in the protective versus deleterious inflammatory response in pneumonia. Int. J. Infect. Dis. 2013, 17, e76–e83. [Google Scholar] [CrossRef]
- Wang, J.F.; Wang, Y.P.; Xie, J.; Zhao, Z.Z.; Gupta, S.; Guo, Y.; Jia, S.H.; Parodo, J.; Marshall, J.C.; Deng, X.M. Upregulated PD-L1 delays human neutrophil apoptosis and promotes lung injury in an experimental mouse model of sepsis. Blood 2021, 138, 806–810. [Google Scholar] [CrossRef]
- Cox, G.; Crossley, J.; Xing, Z. Macrophage engulfment of apoptotic neutrophils contributes to the resolution of acute pulmonary inflammation in vivo. Am. J. Respir. Cell Mol. Biol. 1995, 12, 232–237. [Google Scholar] [CrossRef]
- Liu, J.; He, J.; Ge, L.; Xiao, H.; Huang, Y.; Zeng, L.; Jiang, Z.; Lu, M.; Hu, Z. Hypoxic preconditioning rejuvenates mesenchymal stem cells and enhances neuroprotection following intracerebral hemorrhage via the miR-326-mediated autophagy. Stem Cell Res. Ther. 2021, 12, 413. [Google Scholar] [CrossRef]
- Faulknor, R.A.; Olekson, M.A.; Ekwueme, E.C.; Krzyszczyk, P.; Freeman, J.W.; Berthiaume, F. Hypoxia Impairs Mesenchymal Stromal Cell-Induced Macrophage M1 to M2 Transition. Technol. (Singap. World Sci.) 2017, 5, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Nedeva, C. Inflammation and Cell Death of the Innate and Adaptive Immune System during Sepsis. Biomolecules 2021, 11, 1011. [Google Scholar] [CrossRef] [PubMed]
- Venet, F.; Monneret, G. Advances in the understanding and treatment of sepsis-induced immunosuppression. Nat. Rev. Nephrol. 2018, 14, 121–137. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, S.D.; Horgan, E.; Ali, A.; Masterson, C.; Laffey, J.G.; MacLoughlin, R.; O’Toole, D. Nebulized Mesenchymal Stem Cell Derived Conditioned Medium Retains Antibacterial Properties Against Clinical Pathogen Isolates. J. Aerosol. Med. Pulm. Drug Deliv. 2020, 33, 140–152. [Google Scholar] [CrossRef]


Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Byrnes, D.; Masterson, C.H.; Brady, J.; Alagesan, S.; Gonzalez, H.E.; McCarthy, S.D.; Fandiño, J.; O’Toole, D.P.; Laffey, J.G. Differential Effects of Cytokine Versus Hypoxic Preconditioning of Human Mesenchymal Stromal Cells in Pulmonary Sepsis Induced by Antimicrobial-Resistant Klebsiella pneumoniae. Pharmaceuticals 2023, 16, 149. https://doi.org/10.3390/ph16020149
Byrnes D, Masterson CH, Brady J, Alagesan S, Gonzalez HE, McCarthy SD, Fandiño J, O’Toole DP, Laffey JG. Differential Effects of Cytokine Versus Hypoxic Preconditioning of Human Mesenchymal Stromal Cells in Pulmonary Sepsis Induced by Antimicrobial-Resistant Klebsiella pneumoniae. Pharmaceuticals. 2023; 16(2):149. https://doi.org/10.3390/ph16020149
Chicago/Turabian StyleByrnes, Declan, Claire H. Masterson, Jack Brady, Senthilkumar Alagesan, Hector E. Gonzalez, Sean D. McCarthy, Juan Fandiño, Daniel P. O’Toole, and John G. Laffey. 2023. "Differential Effects of Cytokine Versus Hypoxic Preconditioning of Human Mesenchymal Stromal Cells in Pulmonary Sepsis Induced by Antimicrobial-Resistant Klebsiella pneumoniae" Pharmaceuticals 16, no. 2: 149. https://doi.org/10.3390/ph16020149
APA StyleByrnes, D., Masterson, C. H., Brady, J., Alagesan, S., Gonzalez, H. E., McCarthy, S. D., Fandiño, J., O’Toole, D. P., & Laffey, J. G. (2023). Differential Effects of Cytokine Versus Hypoxic Preconditioning of Human Mesenchymal Stromal Cells in Pulmonary Sepsis Induced by Antimicrobial-Resistant Klebsiella pneumoniae. Pharmaceuticals, 16(2), 149. https://doi.org/10.3390/ph16020149





