Brain Glucose Hypometabolism and Iron Accumulation in Different Brain Regions in Alzheimer’s and Parkinson’s Diseases
Abstract
1. Introduction
2. Methods
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Mosconi, L.; Pupi, A.; De Leon, M.J. Brain Glucose Hypometabolism and Oxidative Stress in Preclinical Alzheimer’s Disease. Ann. N. Y. Acad. Sci. 2008, 1147, 180–195. [Google Scholar] [CrossRef]
- Albrecht, F.; Ballarini, T.; Neumann, J.; Schroeter, M.L. FDG-PET Hypometabolism Is More Sensitive than MRI Atrophy in Parkinson’s Disease: A Whole-Brain Multimodal Imaging Meta-Analysis. NeuroImage Clin. 2019, 21, 101594. [Google Scholar] [CrossRef]
- Herholz, K.; Salmon, E.; Perani, D.; Baron, J.C.; Holthoff, V.; Frölich, L.; Schönknecht, P.; Ito, K.; Mielke, R.; Kalbe, E.; et al. Discrimination between Alzheimer Dementia and Controls by Automated Analysis of Multicenter FDG PET. NeuroImage 2002, 17, 302–316. [Google Scholar] [CrossRef] [PubMed]
- Swerdlow, R.; Marcus, D.L.; Landman, J.; Kooby, D.; Frey, W.; Freedman, M.L. Brain Glucose Metabolism in Alzheimer’s Disease. Am. J. Med. Sci. 1994, 308, 141–144. [Google Scholar] [CrossRef]
- Reiman, E.M.; Caselli, R.J.; Yun, L.S.; Chen, K.; Bandy, D.; Minoshima, S.; Thibodeau, S.N.; Osborne, D. Preclinical Evidence of Alzheimer’s Disease in Persons Homozygous for the Epsilon 4 Allele for Apolipoprotein E. N. Engl. J. Med. 1996, 334, 752–758. [Google Scholar] [CrossRef] [PubMed]
- Strom, A.; Iaccarino, L.; Edwards, L.; Lesman-Segev, O.H.; Soleimani-Meigooni, D.N.; Pham, J.; Baker, S.L.; Landau, S.; Jagust, W.J.; Miller, B.L.; et al. Cortical Hypometabolism Reflects Local Atrophy and Tau Pathology in Symptomatic Alzheimer’s Disease. Brain J. Neurol. 2021, 145, 713–728. [Google Scholar] [CrossRef] [PubMed]
- Meles, S.K.; Renken, R.J.; Pagani, M.; Teune, L.K.; Arnaldi, D.; Morbelli, S.; Nobili, F.; van Laar, T.; Obeso, J.A.; Rodríguez-Oroz, M.C.; et al. Abnormal Pattern of Brain Glucose Metabolism in Parkinson’s Disease: Replication in Three European Cohorts. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 437–450. [Google Scholar] [CrossRef]
- Anandhan, A.; Jacome, M.S.; Lei, S.; Hernandez-Franco, P.; Pappa, A.; Panayiotidis, M.I.; Powers, R.; Franco, R. Metabolic Dysfunction in Parkinson’s Disease: Bioenergetics, Redox Homeostasis and Central Carbon Metabolism. Brain Res. Bull. 2017, 133, 12–30. [Google Scholar] [CrossRef]
- Ward, R.J.; Zucca, F.A.; Duyn, J.H.; Crichton, R.R.; Zecca, L. The Role of Iron in Brain Ageing and Neurodegenerative Disorders. Lancet Neurol. 2014, 13, 1045–1060. [Google Scholar] [CrossRef]
- Fawcett, J.R.; Bordayo, E.Z.; Jackson, K.; Liu, H.; Peterson, J.; Svitak, A.; Frey, W.H. Inactivation of the Human Brain Muscarinic Acetylcholine Receptor by Oxidative Damage Catalyzed by a Low Molecular Weight Endogenous Inhibitor from Alzheimer’s Brain Is Prevented by Pyrophosphate Analogs, Bioflavonoids and Other Antioxidants. Brain Res. 2002, 950, 10–20. [Google Scholar] [CrossRef]
- Atamna, H.; Frey, W.H. A Role for Heme in Alzheimer’s Disease: Heme Binds Amyloid Beta and Has Altered Metabolism. Proc. Natl. Acad. Sci. USA. 2004, 101, 11153–11158. [Google Scholar] [CrossRef] [PubMed]
- Gao, L.; Jiang, Z.; Cai, Z.; Cai, M.; Zhang, Q.; Ma, Y.; Li, G.; Zhao, F.; Ma, Q. Brain Iron Deposition Analysis Using Susceptibility Weighted Imaging and Its Association with Body Iron Level in Patients with Mild Cognitive Impairment. Mol. Med. Rep. 2017, 16, 8209–8215. [Google Scholar] [CrossRef] [PubMed]
- Venters, H.D.; Bonilla, L.E.; Jensen, T.; Garner, H.P.; Bordayo, E.Z.; Najarian, M.M.; Ala, T.A.; Mason, R.P.; Frey, W.H. Heme from Alzheimer’s Brain Inhibits Muscarinic Receptor Binding via Thiyl Radical Generation. Brain Res. 1997, 764, 93–100. [Google Scholar] [CrossRef]
- Schenck, J.F. Magnetic Resonance Imaging of Brain Iron. J. Neurol. Sci. 2003, 207, 99–102. [Google Scholar] [CrossRef]
- Haacke, E.M.; Cheng, N.Y.C.; House, M.J.; Liu, Q.; Neelavalli, J.; Ogg, R.J.; Khan, A.; Ayaz, M.; Kirsch, W.; Obenaus, A. Imaging Iron Stores in the Brain Using Magnetic Resonance Imaging. Magn. Reson. Imaging 2005, 23, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Sofic, E.; Riederer, P.; Heinsen, H.; Beckmann, H.; Reynolds, G.P.; Hebenstreit, G.; Youdim, M.B. Increased Iron (III) and Total Iron Content in Post Mortem Substantia Nigra of Parkinsonian Brain. J. Neural Transm. 1988, 74, 199–205. [Google Scholar] [CrossRef]
- Wan, W.; Jin, L.; Wang, Z.; Wang, L.; Fei, G.; Ye, F.; Pan, X.; Wang, C.; Zhong, C. Iron Deposition Leads to Neuronal α-Synuclein Pathology by Inducing Autophagy Dysfunction. Front. Neurol. 2017, 8, 1. [Google Scholar] [CrossRef]
- Wang, J.-Y.; Zhuang, Q.-Q.; Zhu, L.-B.; Zhu, H.; Li, T.; Li, R.; Chen, S.-F.; Huang, C.-P.; Zhang, X.; Zhu, J.-H. Meta-Analysis of Brain Iron Levels of Parkinson’s Disease Patients Determined by Postmortem and MRI Measurements. Sci. Rep. 2016, 6, 36669. [Google Scholar] [CrossRef] [PubMed]
- Thomas, G.E.C.; Leyland, L.A.; Schrag, A.-E.; Lees, A.J.; Acosta-Cabronero, J.; Weil, R.S. Brain Iron Deposition Is Linked with Cognitive Severity in Parkinson’s Disease. J. Neurol. Neurosurg. Psychiatry 2020, 91, 418–425. [Google Scholar] [CrossRef]
- Freiherr, J.; Hallschmid, M.; Frey, W.H.; Brünner, Y.F.; Chapman, C.D.; Hölscher, C.; Craft, S.; De Felice, F.G.; Benedict, C. Intranasal Insulin as a Treatment for Alzheimer’s Disease: A Review of Basic Research and Clinical Evidence. CNS Drugs 2013, 27, 505–514. [Google Scholar] [CrossRef]
- Ferreira, L.S.S.; Fernandes, C.S.; Vieira, M.N.N.; De Felice, F.G. Insulin Resistance in Alzheimer’s Disease. Front. Neurosci. 2018, 12, 830. [Google Scholar] [CrossRef] [PubMed]
- Frey, I.W.H. Method for Administering Insulin to the Brain. U.S. Patent 6,313,093 B1, 2001. [Google Scholar]
- Jauch-Chara, K.; Friedrich, A.; Rezmer, M.; Melchert, U.H.; Scholand-Engler, H.G.; Hallschmid, M.; Oltmanns, K.M. Intranasal Insulin Suppresses Food Intake via Enhancement of Brain Energy Levels in Humans. Diabetes 2012, 61, 2261–2268. [Google Scholar] [CrossRef] [PubMed]
- Benedict, C.; Hallschmid, M.; Hatke, A.; Schultes, B.; Fehm, H.L.; Born, J.; Kern, W. Intranasal Insulin Improves Memory in Humans. Psychoneuroendocrinology 2004, 29, 1326–1334. [Google Scholar] [CrossRef] [PubMed]
- Reger, M.A.; Watson, G.S.; Frey, W.H.; Baker, L.D.; Cholerton, B.; Keeling, M.L.; Belongia, D.A.; Fishel, M.A.; Plymate, S.R.; Schellenberg, G.D.; et al. Effects of Intranasal Insulin on Cognition in Memory-Impaired Older Adults: Modulation by APOE Genotype. Neurobiol. Aging 2006, 27, 451–458. [Google Scholar] [CrossRef]
- Craft, S.; Baker, L.D.; Montine, T.J.; Minoshima, S.; Watson, G.S.; Claxton, A.; Arbuckle, M.; Callaghan, M.; Tsai, E.; Plymate, S.R.; et al. Intranasal Insulin Therapy for Alzheimer Disease and Amnestic Mild Cognitive Impairment: A Pilot Clinical Trial. Arch. Neurol. 2012, 69, 29–38. [Google Scholar] [CrossRef] [PubMed]
- Novak, P.; Pimentel Maldonado, D.A.; Novak, V. Safety and Preliminary Efficacy of Intranasal Insulin for Cognitive Impairment in Parkinson Disease and Multiple System Atrophy: A Double-Blinded Placebo-Controlled Pilot Study. PLoS ONE 2019, 14, e0214364. [Google Scholar] [CrossRef]
- Kosyakovsky, J.; Fine, J.M.; Frey, W.H.; Hanson, L.R. Mechanisms of Intranasal Deferoxamine in Neurodegenerative and Neurovascular Disease. Pharm. Basel Switz. 2021, 14, 95. [Google Scholar] [CrossRef]
- Giau, V.V.; Bagyinszky, E.; Yang, Y.S.; Youn, Y.C.; An, S.S.A.; Kim, S.Y. Genetic Analyses of Early-Onset Alzheimer’s Disease Using Next Generation Sequencing. Sci. Rep. 2019, 9, 8368. [Google Scholar] [CrossRef]
- Sheerin, U.-M.; Houlden, H.; Wood, N.W. Advances in the Genetics of Parkinson’s Disease: A Guide for the Clinician. Mov. Disord. Clin. Pract. 2014, 1, 3–13. [Google Scholar] [CrossRef]
- Sun, H.; Hu, B.; Yao, Z.; Jackson, M. A PET Study of Discrimination of Cerebral Glucose Metabolism in Alzheimer’s Disease and Mild Cognitive Impairment. In Proceedings of the 2013 6th International Conference on Biomedical Engineering and Informatics, Hangzhou, China, 16–18 December 2013; pp. 6–11. [Google Scholar]
- Fukai, M.; Hirosawa, T.; Kikuchi, M.; Hino, S.; Kitamura, T.; Ouchi, Y.; Yokokura, M.; Yoshikawa, E.; Bunai, T.; Minabe, Y. Different Patterns of Glucose Hypometabolism Underlie Functional Decline in Frontotemporal Dementia and Alzheimer’s Disease: FDG-PET Study. Neuropsychiatry 2018, 8, 441–447. [Google Scholar] [CrossRef]
- Bailly, M.; Destrieux, C.; Hommet, C.; Mondon, K.; Cottier, J.-P.; Beaufils, E.; Vierron, E.; Vercouillie, J.; Ibazizene, M.; Voisin, T.; et al. Precuneus and Cingulate Cortex Atrophy and Hypometabolism in Patients with Alzheimer’s Disease and Mild Cognitive Impairment: MRI and (18)F-FDG PET Quantitative Analysis Using FreeSurfer. BioMed Res. Int. 2015, 2015, 583931. [Google Scholar] [CrossRef]
- Drzezga, A.; Lautenschlager, N.; Siebner, H.; Riemenschneider, M.; Willoch, F.; Minoshima, S.; Schwaiger, M.; Kurz, A. Cerebral Metabolic Changes Accompanying Conversion of Mild Cognitive Impairment into Alzheimer’s Disease: A PET Follow-Up Study. Eur. J. Nucl. Med. Mol. Imaging 2003, 30, 1104–1113. [Google Scholar] [CrossRef] [PubMed]
- Mosconi, L.; Tsui, W.H.; Rusinek, H.; De Santi, S.; Li, Y.; Wang, G.-J.; Pupi, A.; Fowler, J.; de Leon, M.J. Quantitation, Regional Vulnerability, and Kinetic Modeling of Brain Glucose Metabolism in Mild Alzheimer’s Disease. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 1467–1479. [Google Scholar] [CrossRef] [PubMed]
- Chételat, G.; Desgranges, B.; Landeau, B.; Mézenge, F.; Poline, J.B.; de la Sayette, V.; Viader, F.; Eustache, F.; Baron, J.-C. Direct Voxel-Based Comparison between Grey Matter Hypometabolism and Atrophy in Alzheimer’s Disease. Brain 2008, 131, 60–71. [Google Scholar] [CrossRef] [PubMed]
- Yamaji, S.; Ishii, K.; Sasaki, M.; Mori, T.; Kitagaki, H.; Sakamoto, S.; Mori, E. Evaluation of Standardized Uptake Value to Assess Cerebral Glucose Metabolism. Clin. Nucl. Med. 2000, 25, 11. [Google Scholar] [CrossRef]
- Alexander, G.E.; Chen, K.; Pietrini, P.; Rapoport, S.I.; Reiman, E.M. Longitudinal PET Evaluation of Cerebral Metabolic Decline in Dementia: A Potential Outcome Measure in Alzheimer’s Disease Treatment Studies. Am. J. Psychiatry 2002, 159, 738–745. [Google Scholar] [CrossRef]
- Mosconi, L.; Nacmias, B.; Sorbi, S.; Cristofaro, M.T.R.D.; Fayazz, M.; Tedde, A.; Bracco, L.; Herholz, K.; Pupi, A. Brain Metabolic Decreases Related to the Dose of the ApoE E4 Allele in Alzheimer’s Disease. J. Neurol. Neurosurg. Psychiatry 2004, 75, 370–376. [Google Scholar] [CrossRef]
- Fan, X.; Liu, X.; Yan, L.; Mok, V.C.T.; Li, K. Assessment of Brain Iron Accumulation in Alzheimer’s Disease with Quantitative Susceptibility Mapping. Alzheimers Dement. 2020, 16, e038799. [Google Scholar] [CrossRef]
- Raven, E.P.; Lu, P.H.; Tishler, T.A.; Heydari, P.; Bartzokis, G. Increased Iron Levels and Decreased Tissue Integrity in Hippocampus of Alzheimer’s Disease Detected in Vivo with Magnetic Resonance Imaging. J. Alzheimers Dis. 2013, 37, 127–136. [Google Scholar] [CrossRef]
- Bartzokis, G.; Sultzer, D.; Cummings, J.; Holt, L.E.; Hance, D.B.; Henderson, V.W.; Mintz, J. In Vivo Evaluation of Brain Iron in Alzheimer Disease Using Magnetic Resonance Imaging. Arch. Gen. Psychiatry 2000, 57, 47–53. [Google Scholar] [CrossRef]
- Du, L.; Zhao, Z.; Cui, A.; Zhu, Y.; Zhang, L.; Liu, J.; Shi, S.; Fu, C.; Han, X.; Gao, W.; et al. Increased Iron Deposition on Brain Quantitative Susceptibility Mapping Correlates with Decreased Cognitive Function in Alzheimer’s Disease. ACS Chem. Neurosci. 2018, 9, 1849–1857. [Google Scholar] [CrossRef] [PubMed]
- Moon, Y.; Han, S.-H.; Moon, W.-J. Patterns of Brain Iron Accumulation in Vascular Dementia and Alzheimer’s Dementia Using Quantitative Susceptibility Mapping Imaging. J. Alzheimers Dis. 2016, 51, 737–745. [Google Scholar] [CrossRef] [PubMed]
- Ding, B.; Chen, K.-M.; Ling, H.-W.; Sun, F.; Li, X.; Wan, T.; Chai, W.-M.; Zhang, H.; Zhan, Y.; Guan, Y.-J. Correlation of Iron in the Hippocampus with MMSE in Patients with Alzheimer’s Disease. J. Magn. Reson. Imaging 2009, 29, 793–798. [Google Scholar] [CrossRef]
- Qin, Y.; Zhu, W.; Zhan, C.; Zhao, L.; Wang, J.; Tian, Q.; Wang, W. Investigation on Positive Correlation of Increased Brain Iron Deposition with Cognitive Impairment in Alzheimer Disease by Using Quantitative MR R2′ Mapping. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2011, 31, 578. [Google Scholar] [CrossRef]
- Kim, H.-G.; Park, S.; Rhee, H.Y.; Lee, K.M.; Ryu, C.-W.; Rhee, S.J.; Lee, S.Y.; Wang, Y.; Jahng, G.-H. Quantitative Susceptibility Mapping to Evaluate the Early Stage of Alzheimer’s Disease. NeuroImage Clin. 2017, 16, 429–438. [Google Scholar] [CrossRef]
- Jin, R.; Ge, J.; Wu, P.; Lu, J.; Zhang, H.; Wang, J.; Wu, J.; Han, X.; Zhang, W.; Zuo, C. Validation of Abnormal Glucose Metabolism Associated with Parkinson’s Disease in Chinese Participants Based on 18F-Fluorodeoxyglucose Positron Emission Tomography Imaging. Neuropsychiatr. Dis. Treat. 2018, 14, 1981–1989. [Google Scholar] [CrossRef] [PubMed]
- Bohnen, N.I.; Koeppe, R.A.; Minoshima, S.; Giordani, B.; Albin, R.L.; Frey, K.A.; Kuhl, D.E. Cerebral Glucose Metabolic Features of Parkinson Disease and Incident Dementia: Longitudinal Study. J. Nucl. Med. 2011, 52, 848–855. [Google Scholar] [CrossRef]
- Berding, G.; Odin, P.; Brooks, D.J.; Nikkhah, G.; Matthies, C.; Peschel, T.; Shing, M.; Kolbe, H.; van den Hoff, J.; Fricke, H.; et al. Resting Regional Cerebral Glucose Metabolism in Advanced Parkinson’s Disease Studied in the off and on Conditions with [18F]FDG-PET. Mov. Disord. 2001, 16, 1014–1022. [Google Scholar] [CrossRef]
- Hu, M.T.M.; Taylor-Robinson, S.D.; Chaudhuri, K.R.; Bell, J.D.; Labbé, C.; Cunningham, V.J.; Koepp, M.J.; Hammers, A.; Morris, R.G.; Turjanski, N.; et al. Cortical Dysfunction in Non-Demented Parkinson’s Disease Patients: A Combined 31P-MRS and 18FDG-PET Study. Brain 2000, 123, 340–352. [Google Scholar] [CrossRef]
- Teune, L.K.; Renken, R.J.; de Jong, B.M.; Willemsen, A.T.; van Osch, M.J.; Roerdink, J.B.T.M.; Dierckx, R.A.; Leenders, K.L. Parkinson’s Disease-Related Perfusion and Glucose Metabolic Brain Patterns Identified with PCASL-MRI and FDG-PET Imaging. NeuroImage Clin. 2014, 5, 240–244. [Google Scholar] [CrossRef]
- Jokinen, P.; Scheinin, N.M.; Aalto, S.; Någren, K.; Savisto, N.; Parkkola, R.; Rokka, J.; Haaparanta, M.; Röyttä, M.; Rinne, J.O. [11C]PIB-, [18F]FDG-PET and MRI Imaging in Patients with Parkinson’s Disease with and without Dementia. Parkinsonism Relat. Disord. 2010, 16, 666–670. [Google Scholar] [CrossRef] [PubMed]
- Teune, L.K.; Bartels, A.L.; de Jong, B.M.; Willemsen, A.T.M.; Eshuis, S.A.; de Vries, J.J.; van Oostrom, J.C.H.; Leenders, K.L. Typical Cerebral Metabolic Patterns in Neurodegenerative Brain Diseases. Mov. Disord. 2010, 25, 2395–2404. [Google Scholar] [CrossRef]
- Eckert, T.; Barnes, A.; Dhawan, V.; Frucht, S.; Gordon, M.F.; Feigin, A.S.; Eidelberg, D. FDG PET in the Differential Diagnosis of Parkinsonian Disorders. NeuroImage 2005, 26, 912–921. [Google Scholar] [CrossRef] [PubMed]
- Juh, R.; Pae, C.-U.; Lee, C.-U.; Yang, D.; Chung, Y.; Suh, T.; Choe, B. Voxel Based Comparison of Glucose Metabolism in the Differential Diagnosis of the Multiple System Atrophy Using Statistical Parametric Mapping. Neurosci. Res. 2005, 52, 211–219. [Google Scholar] [CrossRef]
- Juh, R.; Kim, J.; Moon, D.; Choe, B.; Suh, T. Different Metabolic Patterns Analysis of Parkinsonism on the 18F-FDG PET. Eur. J. Radiol. 2004, 51, 223–233. [Google Scholar] [CrossRef]
- Granert, O.; Drzezga, A.E.; Boecker, H.; Perneczky, R.; Kurz, A.; Götz, J.; van Eimeren, T.; Häussermann, P. Metabolic Topology of Neurodegenerative Disorders: Influence of Cognitive and Motor Deficits. J. Nucl. Med. 2015, 56, 1916–1921. [Google Scholar] [CrossRef] [PubMed]
- Fan, Z.; Aman, Y.; Ahmed, I.; Chetelat, G.; Landeau, B.; Ray Chaudhuri, K.; Brooks, D.J.; Edison, P. Influence of Microglial Activation on Neuronal Function in Alzheimer’s and Parkinson’s Disease Dementia. Alzheimers Dement. 2015, 11, 608–621.e7. [Google Scholar] [CrossRef]
- Edison, P.; Ahmed, I.; Fan, Z.; Hinz, R.; Gelosa, G.; Ray Chaudhuri, K.; Walker, Z.; Turkheimer, F.E.; Brooks, D.J. Microglia, Amyloid, and Glucose Metabolism in Parkinson’s Disease with and without Dementia. Neuropsychopharmacology 2013, 38, 938–949. [Google Scholar] [CrossRef]
- Berti, V.; Polito, C.; Borghammer, P.; Ramat, S.; Mosconi, L.; Vanzi, E.; De Cristofaro, M.T.; De Leon, M.; Sorbi, S.; Pupi, A. Alternative Normalization Methods Demonstrate Widespread Cortical Hypometabolism in Untreated de Novo Parkinson’s Disease. Q. J. Nucl. Med. Mol. Imaging 2012, 56, 299–308. [Google Scholar]
- Langkammer, C.; Pirpamer, L.; Seiler, S.; Deistung, A.; Schweser, F.; Franthal, S.; Homayoon, N.; Katschnig-Winter, P.; Koegl-Wallner, M.; Pendl, T.; et al. Quantitative Susceptibility Mapping in Parkinson’s Disease. PLoS ONE 2016, 11, e0162460. [Google Scholar] [CrossRef]
- Sjöström, H.; Granberg, T.; Westman, E.; Svenningsson, P. Quantitative Susceptibility Mapping Differentiates between Parkinsonian Disorders. Parkinsonism Relat. Disord. 2017, 44, 51–57. [Google Scholar] [CrossRef]
- Region-specific Disturbed Iron Distribution in Early Idiopathic Parkinson’s Disease Measured by Quantitative Susceptibility Mapping. Hum. Brain Mapp. 2015, 36, 4407–4420. [CrossRef] [PubMed]
- Chen, Q.; Chen, Y.; Zhang, Y.; Wang, F.; Yu, H.; Zhang, C.; Jiang, Z.; Luo, W. Iron Deposition in Parkinson’s Disease by Quantitative Susceptibility Mapping. BMC Neurosci. 2019, 20, 23. [Google Scholar] [CrossRef]
- Sun, H.; Walsh, A.J.; Lebel, R.M.; Blevins, G.; Catz, I.; Lu, J.-Q.; Johnson, E.S.; Emery, D.J.; Warren, K.G.; Wilman, A.H. Validation of Quantitative Susceptibility Mapping with Perls’ Iron Staining for Subcortical Gray Matter. NeuroImage 2015, 105, 486–492. [Google Scholar] [CrossRef] [PubMed]
- Li, D.T.H.; Hui, E.S.; Chan, Q.; Yao, N.; Chua, S.E.; McAlonan, G.M.; Pang, S.Y.Y.; Ho, S.L.; Mak, H.K.F. Quantitative Susceptibility Mapping as an Indicator of Subcortical and Limbic Iron Abnormality in Parkinson’s Disease with Dementia. NeuroImage Clin. 2018, 20, 365–373. [Google Scholar] [CrossRef]
- Shahmaei, V.; Faeghi, F.; Mohammadbeigi, A.; Hashemi, H.; Ashrafi, F. Evaluation of Iron Deposition in Brain Basal Ganglia of Patients with Parkinson’s Disease Using Quantitative Susceptibility Mapping. Eur. J. Radiol. Open 2019, 6, 169–174. [Google Scholar] [CrossRef]
- Acosta-Cabronero, J.; Cardenas-Blanco, A.; Betts, M.J.; Butryn, M.; Valdes-Herrera, J.P.; Galazky, I.; Nestor, P.J. The Whole-Brain Pattern of Magnetic Susceptibility Perturbations in Parkinson’s Disease. Brain 2017, 140, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Ghassaban, K.; He, N.; Sethi, S.K.; Huang, P.; Chen, S.; Yan, F.; Haacke, E.M. Regional High Iron in the Substantia Nigra Differentiates Parkinson’s Disease Patients From Healthy Controls. Front. Aging Neurosci. 2019, 11, 106. [Google Scholar] [CrossRef]
- Azuma, M.; Hirai, T.; Yamada, K.; Yamashita, S.; Ando, Y.; Tateishi, M.; Iryo, Y.; Yoneda, T.; Kitajima, M.; Wang, Y.; et al. Lateral Asymmetry and Spatial Difference of Iron Deposition in the Substantia Nigra of Patients with Parkinson Disease Measured with Quantitative Susceptibility Mapping. Am. J. Neuroradiol. 2016, 37, 782–788. [Google Scholar] [CrossRef]
- Murakami, Y.; Kakeda, S.; Watanabe, K.; Ueda, I.; Ogasawara, A.; Moriya, J.; Ide, S.; Futatsuya, K.; Sato, T.; Okada, K.; et al. Usefulness of Quantitative Susceptibility Mapping for the Diagnosis of Parkinson Disease. Am. J. Neuroradiol. 2015, 36, 1102–1108. [Google Scholar] [CrossRef]
- Barbosa, J.H.O.; Santos, A.C.; Tumas, V.; Liu, M.; Zheng, W.; Haacke, E.M.; Salmon, C.E.G. Quantifying Brain Iron Deposition in Patients with Parkinson’s Disease Using Quantitative Susceptibility Mapping, R2 and R2*. Magn. Reson. Imaging 2015, 33, 559–565. [Google Scholar] [CrossRef]
- Azuma, M.; Hirai, T.; Nakaura, T.; Kitajima, M.; Yamashita, S.; Hashimoto, M.; Yamada, K.; Uetani, H.; Yamashita, Y.; Wang, Y. Combining Quantitative Susceptibility Mapping to the Morphometric Index in Differentiating between Progressive Supranuclear Palsy and Parkinson’s Disease. J. Neurol. Sci. 2019, 406, 116443. [Google Scholar] [CrossRef]
- Dashtipour, K.; Liu, M.; Kani, C.; Dalaie, P.; Obenaus, A.; Simmons, D.; Gatto, N.M.; Zarifi, M. Iron Accumulation Is Not Homogenous among Patients with Parkinson’s Disease. Park. Dis. 2015, 2015, e324843. [Google Scholar] [CrossRef]
- Harper, L.; Bouwman, F.; Burton, E.J.; Barkhof, F.; Scheltens, P.; O’Brien, J.T.; Fox, N.C.; Ridgway, G.R.; Schott, J.M. Patterns of Atrophy in Pathologically Confirmed Dementias: A Voxelwise Analysis. J. Neurol. Neurosurg. Psychiatry 2017, 88, 908–916. [Google Scholar] [CrossRef]
- Braak, H.; Braak, E. Neuropathological Stageing of Alzheimer-Related Changes. Acta Neuropathol. 1991, 82, 239–259. [Google Scholar] [CrossRef]
- Lowe, V.J.; Wiste, H.J.; Senjem, M.L.; Weigand, S.D.; Therneau, T.M.; Boeve, B.F.; Josephs, K.A.; Fang, P.; Pandey, M.K.; Murray, M.E.; et al. Widespread Brain Tau and Its Association with Ageing, Braak Stage and Alzheimer’s Dementia. Brain 2018, 141, 271–287. [Google Scholar] [CrossRef]
- Bousejra-ElGarah, F.; Bijani, C.; Coppel, Y.; Faller, P.; Hureau, C. Iron(II) Binding to Amyloid-β, the Alzheimer’s Peptide. Inorg. Chem. 2011, 50, 9024–9030. [Google Scholar] [CrossRef]
- Liu, J.-L.; Fan, Y.-G.; Yang, Z.-S.; Wang, Z.-Y.; Guo, C. Iron and Alzheimer’s Disease: From Pathogenesis to Therapeutic Implications. Front. Neurosci. 2018, 12, 632. [Google Scholar] [CrossRef]
- Fox, M.D. Mapping Symptoms to Brain Networks with the Human Connectome. N. Engl. J. Med. 2018, 379, 2237–2245. [Google Scholar] [CrossRef]
- Chung, J.Y.; Kim, H.-S.; Song, J. Iron Metabolism in Diabetes-Induced Alzheimer’s Disease: A Focus on Insulin Resistance in the Brain. Biometals Int. J. Role Met. Ions Biol. Biochem. Med. 2018, 31, 705–714. [Google Scholar] [CrossRef]
- Song, T.; Li, J.; Mei, S.; Jia, X.; Yang, H.; Ye, Y.; Yuan, J.; Zhang, Y.; Lu, J. Nigral Iron Deposition Is Associated With Levodopa-Induced Dyskinesia in Parkinson’s Disease. Front. Neurosci. 2021, 15, 647168. [Google Scholar] [CrossRef]
- Crapper McLachlan, D.R.; Dalton, A.J.; Kruck, T.P.; Bell, M.Y.; Smith, W.L.; Kalow, W.; Andrews, D.F. Intramuscular Desferrioxamine in Patients with Alzheimer’s Disease. Lancet Lond. Engl. 1991, 337, 1304–1308. [Google Scholar] [CrossRef]
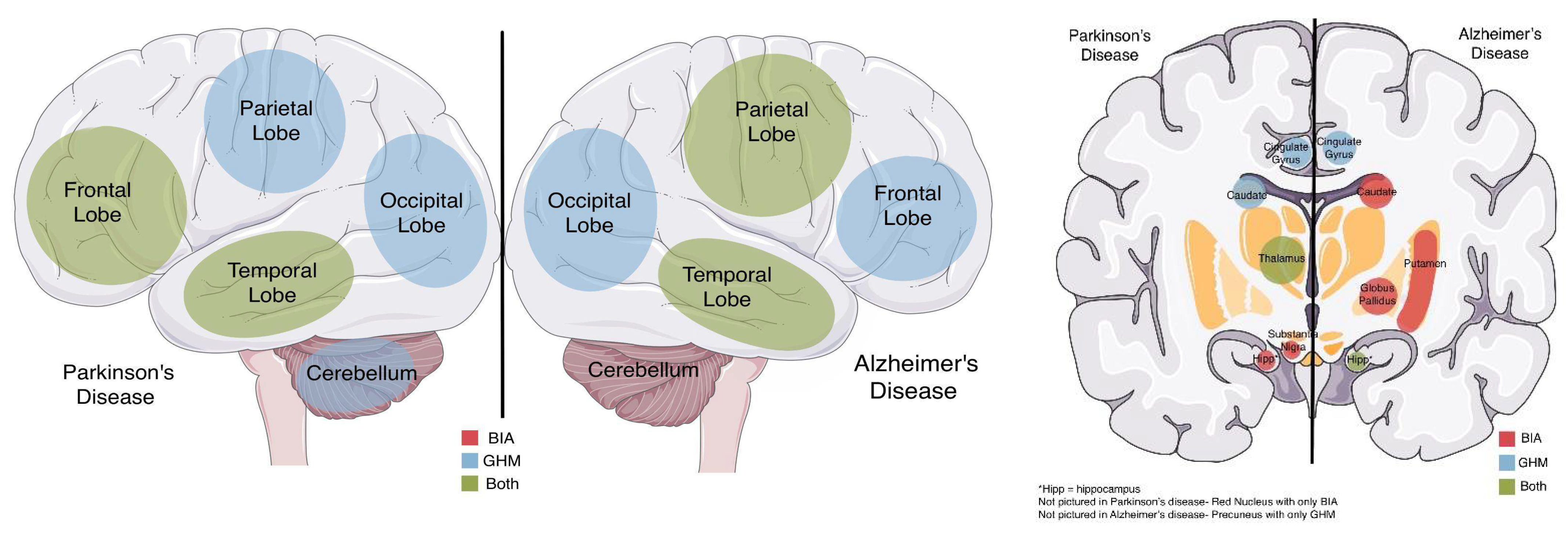
| a. Glucose hypometabolism (GHM) in AD: present (+), absent (−), not reported (NR). Papers reporting parieto–temporal location are shown by merging the columns for the parietal and temporal lobes. Under each brain region is the number of studies reporting GHM present over the number of studies that examined this region. | ||||||||||
| Study Number | Temporal Lobe | Parietal Lobe | Cingulate Gyrus | Precuneus | Hippocampus | Frontal Lobe | Occipital Lobe | Caudate Nucleus | Putamen | Globus Pallidus |
| 10/10 | 9/9 | 9/9 | 5/5 | 3/3 | 3/3 | 2/3 | 1/2 | 0/1 | 0/0 | |
| [3] | + | + | + | NR | + | NR | − | − | NR | |
| [31] | + | NR | + | NR | NR | NR | NR | NR | NR | NR |
| [32] | + | + | + | + | NR | NR | NR | NR | NR | NR |
| [33] | + | + | + | + | + | NR | NR | NR | NR | NR |
| [34] | + | + | NR | NR | NR | NR | NR | NR | NR | |
| [35] | + | + | NR | + | NR | − | NR | NR | NR | |
| [36] | + | + | + | + | + | NR | NR | + | NR | NR |
| [37] | + | + | NR | NR | NR | + | + | NR | NR | NR |
| [38] | + | + | + | NR | NR | + | + | NR | NR | NR |
| [39] | + | + | + | NR | NR | NR | NR | NR | NR | |
| b. Brain iron accumulation (BIA) in AD: present (+), absent (–), not reported (NR). Under each brain region is the number of studies reporting BIA present over the number of studies that examined this region. | ||||||||||
| Study Number | Temporal Lobe | Parietal Lobe | Cingulate Gyrus | Precuneus | Hippocampus | Frontal Lobe | Occipital Lobe | Caudate Nucleus | Putamen | Globus Pallidus |
| 5/5 | 2/2 | 1/1 | 0/0 | 5/5 | 1/3 | 0/0 | 6/7 | 6/6 | 2/6 | |
| [40] | + | NR | NR | NR | + | NR | NR | + | + | NR |
| [41] | + | NR | NR | NR | + | NR | NR | NR | NR | NR |
| [42] | NR | NR | NR | NR | NR | − | NR | + | + | + |
| [43] | NR | NR | NR | NR | NR | NR | NR | + | + | − |
| [44] | NR | NR | NR | NR | NR | NR | NR | + | + | − |
| [45] | + | NR | NR | NR | + | NR | NR | − | + | + |
| [46] | + | + | NR | NR | + | − | NR | + | + | − |
| [47] | + | + | + | NR | + | + | NR | + | NR | − |
| a. Glucose hypometabolism (GHM) in PD: present (+), absent (−), not reported (NR), Parkinson’s disease dementia (PDD). Under each brain region is the number of studies reporting GHM present over the number of studies that examined this region. | |||||||||||||
| Study Number | Substantia Nigra | Red Nucleus | Parietal Lobe | Occipital Lobe | Frontal Lobe | Temporal Lobe | Cerebellum | Cingulate Gyrus | Thalamus | Caudate Nucleus | Putamen | Globus Pallidus | Hippocampus |
| 0/0 | 0/0 | 15/15 | 11/14 | 11/14 | 10/11 | 6/10 | 6/8 | 5/8 | 5/6 | 2/6 | 2/4 | 2/3 | |
| [48] | NR | NR | + | + | − | − | − | NR | − | NR | − | − | NR |
| [7] | NR | NR | + | + | + | NR | + | NR | + | NR | + | + | NR |
| [49] | NR | NR | + | + | + | + | NR | + | + | + | − | NR | + |
| [50] | NR | NR | + | NR | + | + | NR | + | NR | + | NR | NR | NR |
| [51] | NR | NR | + | − | − | + | − | − | − | − | NR | NR | NR |
| [52] | NR | NR | + | + | + | + | − | NR | − | NR | NR | NR | NR |
| [53] (w/PDD) | NR | NR | + | + | + | + | + | + | + | + | − | − | − |
| [53] (w/o PDD) | NR | NR | − | + | − | − | − | − | − | − | − | − | − |
| [54] | NR | NR | + | + | + | + | − | NR | NR | NR | NR | NR | NR |
| [55] | NR | NR | + | + | + | NR | + | NR | + | NR | + | + | NR |
| [56] | NR | NR | + | + | + | NR | + | + | NR | + | NR | NR | NR |
| [57] | NR | NR | + | − | + | + | + | − | + | + | − | NR | NR |
| [58] | NR | NR | + | + | + | NR | + | NR | NR | NR | NR | NR | NR |
| [59] | NR | NR | + | − | − | + | NR | + | NR | NR | NR | NR | + |
| [60] | NR | NR | + | + | NR | + | NR | + | NR | NR | NR | NR | NR |
| [61] | NR | NR | + | + | + | + | NR | NR | NR | NR | NR | NR | NR |
| b. Brain iron accumulation (BIA) in PD: present (+), absent (−), not reported (NR). Under each brain region is the number of studies reporting BIA as a fraction of the number of studies examining and reporting on this brain region. | |||||||||||||
| Study number | Substantia Nigra | Red Nucleus | Parietal Lobe | Occipital Lobe | Frontal Lobe | Temporal Lobe | Cerebellum | Cingulate Gyrus | Thalamus | Caudate Nucleus | Putamen | Globus Pallidus | Hippocampus |
| 13/15 | 6/14 | 1/1 | 0/1 | 2/2 | 2/2 | 0/0 | 0/0 | 3/10 | 1/13 | 3/15 | 5/14 | 2/2 | |
| [19] | + | NR | NR | NR | + | + | NR | NR | NR | NR | + | NR | NR |
| [62] | + | + | NR | NR | NR | NR | NR | NR | + | − | − | + | NR |
| [63] | + | + | NR | NR | NR | NR | NR | NR | NR | NR | + | + | NR |
| [64] | + | + | NR | NR | NR | NR | NR | NR | NR | − | − | − | NR |
| [65] | + | + | NR | NR | NR | NR | NR | NR | − | + | + | + | NR |
| [66] | + | + | NR | NR | NR | NR | NR | NR | − | − | − | + | NR |
| [67] | − | − | NR | NR | NR | NR | NR | NR | + | − | − | − | + |
| [68] | + | + | NR | NR | NR | NR | NR | NR | + | − | − | + | NR |
| [69] | + | − | + | − | + | + | NR | NR | − | − | − | − | + |
| [70] | + | − | NR | NR | NR | NR | NR | NR | − | − | − | − | NR |
| [71] | + | − | NR | NR | NR | NR | NR | NR | NR | − | − | − | NR |
| [72] | + | − | NR | NR | NR | NR | NR | NR | − | − | − | − | NR |
| [73] | + | − | NR | NR | NR | NR | NR | NR | − | − | − | − | NR |
| [74] | + | − | NR | NR | NR | NR | NR | NR | NR | − | − | − | NR |
| [75] | − | − | NR | NR | NR | NR | NR | NR | − | − | − | − | NR |
| Brain Region | AD GHM | AD BIA | Brain Region | PD GHM | PD BIA |
|---|---|---|---|---|---|
| Temporal Lobe | 10/10 | 5/5 | Temporal Lobe | 10/11 | 2/2 |
| Parietal Lobe | 9/9 | 2/2 | Parietal Lobe | 15/15 | 1/1 |
| Frontal Lobe | 3/3 | 1/3 | Frontal Lobe | 11/14 | 2/2 |
| Occipital Lobe | 2/3 | 0/0 | Occipital Lobe | 11/14 | 0/1 |
| Hippocampus | 3/3 | 5/5 | Hippocampus | 2/3 | 2/2 |
| Cingulate Gyrus | 9/9 | 1/1 | Cingulate Gyrus | 6/8 | 0/0 |
| Caudate Nucleus | 1/2 | 6/7 | Caudate Nucleus | 5/6 | 1/13 |
| Putamen | 0/1 | 6/6 | Putamen | 2/6 | 3/15 |
| Globus Pallidus | 0/0 | 2/6 | Globus Pallidus | 2/4 | 5/14 |
| Substantia Nigra | Substantia Nigra | 0/0 | 13/15 | ||
| Red Nucleus | Red Nucleus | 0/0 | 6/14 | ||
| Precuneus | 5/5 | 0/0 | |||
| Thalamus | Thalamus | 5/8 | 3/10 | ||
| Cerebellum | Cerebellum | 6/10 | 0/0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rao, I.Y.; Hanson, L.R.; Johnson, J.C.; Rosenbloom, M.H.; Frey, W.H., II. Brain Glucose Hypometabolism and Iron Accumulation in Different Brain Regions in Alzheimer’s and Parkinson’s Diseases. Pharmaceuticals 2022, 15, 551. https://doi.org/10.3390/ph15050551
Rao IY, Hanson LR, Johnson JC, Rosenbloom MH, Frey WH II. Brain Glucose Hypometabolism and Iron Accumulation in Different Brain Regions in Alzheimer’s and Parkinson’s Diseases. Pharmaceuticals. 2022; 15(5):551. https://doi.org/10.3390/ph15050551
Chicago/Turabian StyleRao, Indira Y., Leah R. Hanson, Julia C. Johnson, Michael H. Rosenbloom, and William H. Frey, II. 2022. "Brain Glucose Hypometabolism and Iron Accumulation in Different Brain Regions in Alzheimer’s and Parkinson’s Diseases" Pharmaceuticals 15, no. 5: 551. https://doi.org/10.3390/ph15050551
APA StyleRao, I. Y., Hanson, L. R., Johnson, J. C., Rosenbloom, M. H., & Frey, W. H., II. (2022). Brain Glucose Hypometabolism and Iron Accumulation in Different Brain Regions in Alzheimer’s and Parkinson’s Diseases. Pharmaceuticals, 15(5), 551. https://doi.org/10.3390/ph15050551






