Up-to-Date Overview of the Use of Natural Ingredients in Sunscreens
Abstract
1. Introduction
2. Results and Discussion
2.1. Overview of the Use of Natural Ingredients in Sunscreens from Terrestrial and Marine Sources
2.2. Scientific Evidence Supporting the Efficacy of the Top 10 Natural Ingredients from Terrestrial and Marine Sources Used in Sunscreens
2.2.1. Natural Ingredients from Terrestrial Organisms
2.2.2. Marine Ingredients
2.2.3. Mechanisms of Action
2.3. Highlights
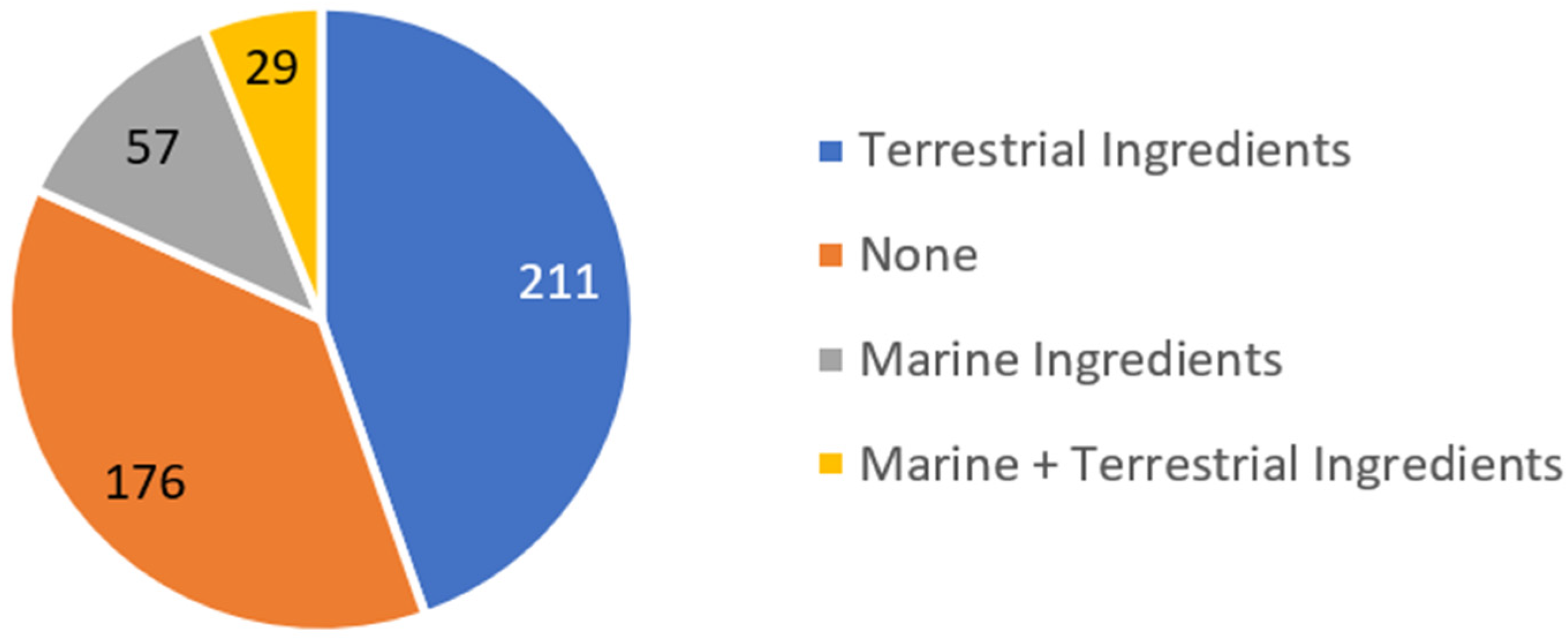
- Two hundred and eleven (48%) of the 444 analyzed sunscreen formulations comprised ingredients from terrestrial plants, while marine ingredients were present in 57 (13%) of the studied formulations.
- The highest prevalence of ingredients from terrestrial plants might be attributed to their biodiversity, easy cultivation, and growth modulation, making them more attractive to the cosmetic industry.
- Twenty-nine (7%) of the total formulations contained both natural ingredients from terrestrial and marine environments, showing a possible synergism of metabolite action towards activity, without loss of the desired properties.
- The most used ingredients from terrestrial sources were obtained from the species H. annuus, G. max, V. paradoxa, P. gratissima, and G. inflata, all of which have been studied as photoprotective agents.
- The antioxidant and photoprotective activities of these extracts were mainly attributed to the presence of tocopherols, flavonoids, and phenolic acids.
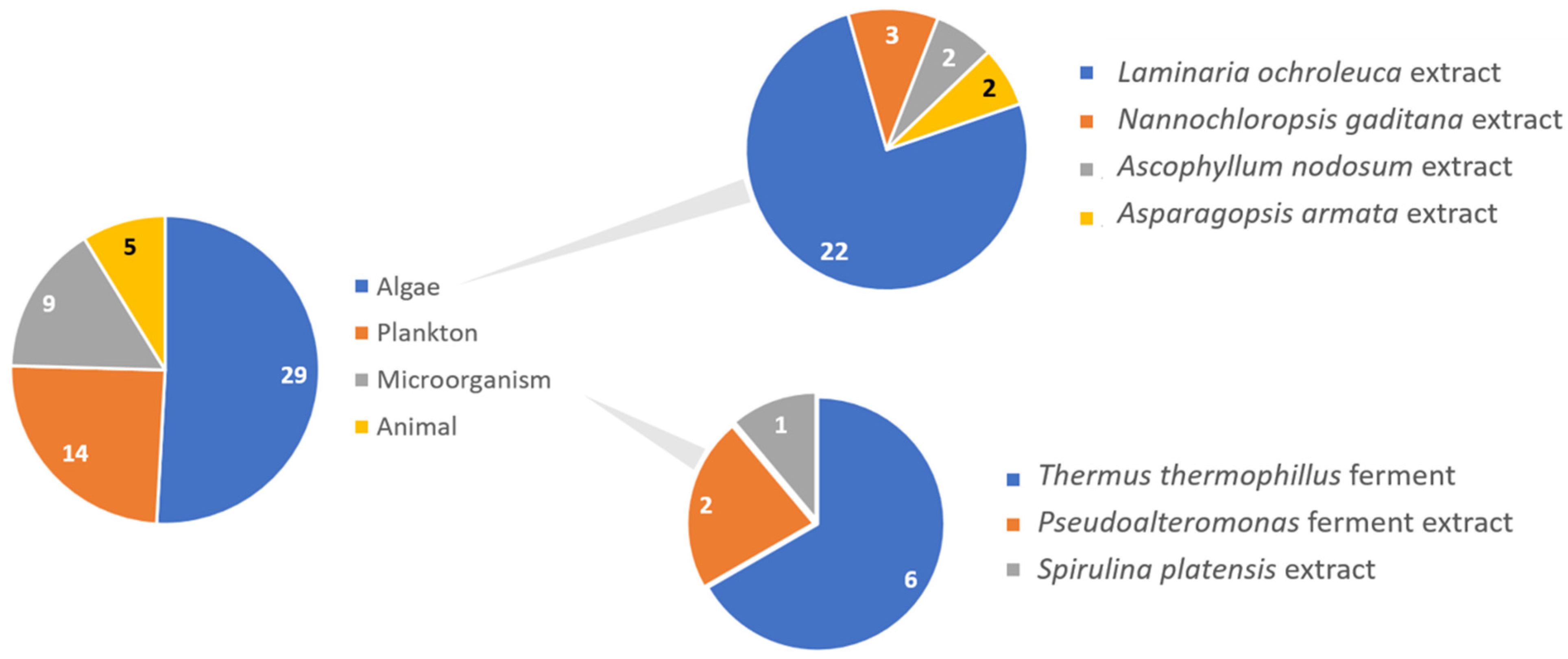
- Algae, due to easy cultivation allied with the development of newer and improved techniques for their cultivation, constitute the most used marine ingredient, which is translated in the number of studies and scientific evidence available in the literature.
- Other marine ingredients such as Artemia or microorganisms were less used, probably due to the scarce scientific evidence of their photoprotective properties to back up their use in sunscreen formulations.
- The photoprotective effects of the analyzed marine ingredients are mainly attributed to MAAs. These low-molecular-weight water-soluble molecules proved to have anti-inflammatory effects by suppressing the expression of pro-inflammatory mediators in response to UV irradiation, therefore constituting a natural promising UV-absorbing alternative to common sunscreens.
3. Materials and Methods
3.1. Data Collection
3.2. Data Analysis
3.2.1. Natural Ingredients Use
3.2.2. Top Natural Ingredients Used in Sunscreens
3.2.3. Scientific Evidence Supporting the Efficacy of Natural Ingredients in Sunscreens
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Radiation: Ultraviolet (UV) Radiation. Available online: https://www.who.int/news-room/q-a-detail/radiation-ultraviolet-(uv) (accessed on 30 August 2021).
- Milito, A.; Castellano, I.; Damiani, E. From Sea to Skin: Is There a Future for Natural Photoprotectants? Mar. Drugs 2021, 19, 379. [Google Scholar] [CrossRef] [PubMed]
- Rittié, L.; Fisher, G.J. Natural and Sun-Induced Aging of Human Skin. Cold Spring Harb. Perspect. Med. 2015, 5, a015370. [Google Scholar] [CrossRef]
- Bosch, R.; Philips, N.; Suárez-Pérez, J.A.; Juarranz, A.; Devmurari, A.; Chalensouk-Khaosaat, J.; González, S. Mechanisms of Photoaging and Cutaneous Photocarcinogenesis, and Photoprotective Strategies with Phytochemicals. Antioxidants 2015, 4, 248–268. [Google Scholar] [CrossRef]
- Orazio, J.; Jarrett, S.; Amaro-Ortiz, A.; Scott, T. UV Radiation and the Skin. Int. J. Mol. Sci. 2013, 14, 12222–12248. [Google Scholar] [CrossRef]
- Martinez, R.M.; Pinho-Ribeiro, F.A.; Steffen, V.S.; Caviglione, C.V.; Vignoli, J.A.; Barbosa, D.S.; Baracat, M.M.; Georgetti, S.R.; Verri, W.A.; Casagrande, R. Naringenin Inhibits UVB Irradiation-Induced Inflammation and Oxidative Stress in the Skin of Hairless Mice. J. Nat. Prod. 2015, 78, 1647–1655. [Google Scholar] [CrossRef] [PubMed]
- Duteil, L.; Cardot-Leccia, N.; Queille-Roussel, C.; Maubert, Y.; Harmelin, Y.; Boukari, F.; Ambrosetti, D.; Lacour, J.-P.; Passeron, T. Differences in visible light-induced pigmentation according to wavelengths: A clinical and histological study in comparison with UVB exposure. Pigment Cell Melanoma Res. 2014, 27, 822–826. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, C.A.; Peres, D.D.A.; Rugno, C.M.; Kojima, M.; Pinto, C.; Consiglieri, V.O.; Kaneko, T.M.; Rosado, C.; Mota, J.; Velasco, M.V.R.; et al. Functional photostability and cutaneous compatibility of bioactive UVA sun care products. J. Photochem. Photobiol. B 2015, 148, 154–159. [Google Scholar] [CrossRef]
- Wolber, R.; Schlenz, K.; Wakamatsu, K.; Smuda, C.; Nakanishi, Y.; Hearing, V.J.; Ito, S. Pigmentation effects of solar simulated radiation as compared with UVA and UVB radiation. Pigment Cell Melanoma Res. 2008, 21, 487–491. [Google Scholar] [CrossRef]
- Leccia, M.-T.; Lebbe, C.; Claudel, J.-P.; Narda, M.; Basset-Seguin, N. New Vision in Photoprotection and Photorepair. Dermatol. Ther. 2019, 9, 103–115. [Google Scholar] [CrossRef]
- Urbach, F. The historical aspects of sunscreens. J. Photochem. Photobiol. B Biol. 2001, 64, 99–104. [Google Scholar] [CrossRef]
- Bens, G. Sunscreens. In Sunlight, Vitamin D and Skin Cancer; Springer: New York, NY, USA, 2014; pp. 429–463. [Google Scholar] [CrossRef]
- Lee, S.-H.; Xiong, J.-Q.; Ru, S.; Patil, S.M.; Kurade, M.B.; Govindwar, S.P.; Oh, S.-E.; Jeon, B.-H. Toxicity of benzophenone-3 and its biodegradation in a freshwater microalga Scenedesmus obliquus. J. Hazard. Mater. 2020, 389, 122149. [Google Scholar] [CrossRef]
- Schneider, S.L.; Lim, H.W. Review of environmental effects of oxybenzone and other sunscreen active ingredients. J. Am. Acad. Dermatol. 2019, 80, 266–271. [Google Scholar] [CrossRef] [PubMed]
- de Miranda, L.L.R.; Harvey, K.E.; Ahmed, A.; Harvey, S.C. UV-filter pollution: Current concerns and future prospects. Environ. Monit. Assess. 2021, 193, 840. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Li, A.; Li, S.; Tang, J.; Li, L.; Xiong, L. Natural components in sunscreens: Topical formulations with sun protection factor (SPF). Biomed. Pharmacother. 2021, 134, 111161. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.S.; Hsia, A.; Miller, J.D.; Hanneman, K.; Scull, H.; Cooper, K.D.; Baron, E. Non-Sunscreen Photoprotection: Antioxidants Add Value to a Sunscreen. J. Investig. Dermatol. Symp. Proc. 2009, 14, 56–59. [Google Scholar] [CrossRef]
- Pangestuti, R.; Siahaan, E.A.; Kim, S.-K. Photoprotective Substances Derived from Marine Algae. Mar. Drugs 2018, 16, 399. [Google Scholar] [CrossRef] [PubMed]
- Amador-Castro, F.; Rodriguez-Martinez, V.; Carrillo-Nieves, D. Robust natural ultraviolet filters from marine ecosystems for the formulation of environmental friendlier bio-sunscreens. Sci. Total Environ. 2020, 749, 141576. [Google Scholar] [CrossRef]
- Gager, L.; Connan, S.; Molla, M.; Couteau, C.; Arbona, J.-F.; Coiffard, L.; Cerantola, S.; Stiger-Pouvreau, V. Active phlorotannins from seven brown seaweeds commercially harvested in Brittany (France) detected by 1H NMR and in vitro assays: Temporal variation and potential valorization in cosmetic applications. J. Appl. Phycol. 2020, 32, 2375–2386. [Google Scholar] [CrossRef]
- Debacq-Chainiaux, F.; Borlon, C.; De Hertogh, B.; Remacle, J.; Morvan, P.Y.; Vallee, R.; Toussaint, O. Identification of potential anti-photoageing algal compounds using an in-vitro model of photoageing. J. Pharm. Pharmacol. 2006, 58, 1577–1583. [Google Scholar] [CrossRef]
- Guinea, M.; Franco, V.; Araujo-Bazán, L.; Rodríguez-Martín, I.; González, S. In vivo UVB-photoprotective activity of extracts from commercial marine macroalgae. Food Chem. Toxicol. 2012, 50, 1109–1117. [Google Scholar] [CrossRef]
- Lalegerie, F.; Lajili, S.; Bedoux, G.; Taupin, L.; Stiger-Pouvreau, V.; Connan, S. Photo-protective compounds in red macroalgae from Brittany: Considerable diversity in mycosporine-like amino acids (MAAs). Mar. Environ. Res. 2019, 147, 37–48. [Google Scholar] [CrossRef] [PubMed]
- Shanuja, S.K.; Iswarya, S.; Sridevi, J.; Gnanamani, A. Exploring the UVB-protective efficacy of melanin precursor extracted from marine imperfect fungus: Featuring characterization and application studies under in vitro conditions. Int. Microbiol. 2018, 21, 59–71. [Google Scholar] [CrossRef] [PubMed]
- Zastrow, L.; Domloge, N.; Golz-Berner, K. Cosmetic Product Based on Artemia Salina Extracts for Regenerating and Stimulating Skin Cells. WO1999038483A1, 5 August 1999. [Google Scholar]
- Cucumel, K.; Farra, C.D.; Domloge, N. Artemia Extract ‘Compensates’ for Age-Related Decrease of Hsp70 in Skin. J. Investig. Dermatol. 2002, 119, 207–350. [Google Scholar] [CrossRef]
- Cucumel, K.; Botto, J.; Bauza, E.; Farra, C.D.; Roetto, R.; Domloge, N. Artemia extract induces Hsp70 in human cells and enhances cell protection from stress. J. Investig. Dermatol. 2001, 117, 456–489. [Google Scholar] [CrossRef]
- Domloge, N.; Bauza, E.; Cucumel, K.; Peyronel, D.; Farra, C.D. Artemia extract toward more extensive sun protection. Cosmet. Toiletries 2002, 2002, 69–78. [Google Scholar]
- Wang, L.; Meng, L.; Ye, S.; Chen, F.; Xie, S.; Wang, X. A Photoprotection Repairing Composition, Preparation Method and Application, and Skin Care Product Thereof [Machine Translation]. CN112535657A, 23 March 2021. [Google Scholar]
- Patil, J.S.; Rodrigues, R.V.; Paul, P.; Sathish, K.; Rafi, M.; Anil, A.C. Benthic dinoflagellate blooms in tropical intertidal rock pools: Elucidation of photoprotection mechanisms. Mar. Biol. 2017, 164, 1–19. [Google Scholar] [CrossRef]
- Moisan, T.A.; Goes, J.; Neale, P.J. Chapter 4—Mycosporine-like amino acids in phytoplankton: Biochemistry, physiology and optics. In Marine Phytoplankton; Kersey, W.T., Munger, S.P., Eds.; Nova Science Publishers: New York, NY, USA, 2009; pp. 119–143. [Google Scholar]
- Laspoumaderes, C.; Bastidas Navarro, M.; Souza, M.S.; Modenutti, B.; Balseiro, E. Effect of ultraviolet radiation on clearance rate of planktonic copepods with different photoprotective strategies. Int. Rev. Hydrobiol. 2019, 104, 34–44. [Google Scholar] [CrossRef]
- Sonntag, B.; Sommaruga, R. Effectiveness of photoprotective strategies in three mixotrophic planktonic ciliate species. Diversity 2020, 12, 252. [Google Scholar] [CrossRef]
- Carreto, J.I.; Carignan, M.O.; Daleo, G.; Marco, S.G.D. Occurrence of mycosporine-like amino acids in the red-tide dinoflagellate Alexandrium excavatum: UV-photoprotective compounds? J. Plankton Res. 1990, 12, 909–921. [Google Scholar] [CrossRef]
- Bashevkin, S.M.; Morgan, S.G.; Christy, J.H. Photoprotective benefits of pigmentation in the transparent plankton community: A comparative species experimental test. Ecology 2019, 100, e02680. [Google Scholar] [CrossRef] [PubMed]
- Hessen, D.O.; Soerensen, K. Photoprotective pigmentation in alpine zooplankton populations. Aqua Fenn. 1990, 20, 165–170. [Google Scholar]
- Karentz, D.; McEuen, F.S.; Land, M.C.; Dunlap, W.C. Survey of mycosporine-like amino acid compounds in Antarctic marine organisms: Potential protection from ultraviolet exposure. Mar. Biol. 1991, 108, 157–166. [Google Scholar] [CrossRef]
- Hylander, S.; Jephson, T. UV protective compounds transferred from a marine dinoflagellate to its copepod predator. J. Exp. Mar. Biol. Ecol. 2010, 389, 38–44. [Google Scholar] [CrossRef]
- Nikitakis, J.; Lange, B. International Cosmetic Ingredient Dictionary and Handbook; Personal Care Products Council: Washington, DC, USA, 2016; Volume 1. [Google Scholar]
- Kuete, V.; Viertel, K.; Efferth, T. 18-Antiproliferative Potential of African Medicinal Plants. In Medicinal Plant Research in Africa; Kuete, V., Ed.; Elsevier: Oxford, UK, 2013; pp. 711–724. [Google Scholar]
- Darqui, F.S.; Radonic, L.M.; Beracochea, V.C.; Hopp, H.E.; López Bilbao, M. Peculiarities of the Transformation of Asteraceae Family Species: The Cases of Sunflower and Lettuce. Front. Plant Sci. 2021, 12, 767459. [Google Scholar] [CrossRef] [PubMed]
- Christaki, E.; Bonos, E.; Giannenas, I.; Florou-Paneri, P. Functional properties of carotenoids originating from algae. J. Sci. Food Agric. 2013, 93, 5–11. [Google Scholar] [CrossRef]
- Resende, D.I.S.P.; Ferreira, M.; Magalhães, C.; Sousa Lobo, J.M.; Sousa, E.; Almeida, I.F. Trends in the use of marine ingredients in anti-aging cosmetics. Algal Res. 2021, 55, 102273. [Google Scholar] [CrossRef]
- Ferreira, M.S.; Magalhães, M.C.; Oliveira, R.; Sousa-Lobo, J.M.; Almeida, I.F. Trends in the Use of Botanicals in Anti-Aging Cosmetics. Molecules 2021, 26, 3584. [Google Scholar] [CrossRef]
- Sánchez-Muniz, F.J.; Bastida, S.; Benedí, J. Sunflower Oil. In Encyclopedia of Food and Health; Caballero, B., Finglas, P.M., Toldrá, F., Eds.; Academic Press: Oxford, UK, 2016; pp. 217–226. [Google Scholar] [CrossRef]
- Pal, D. Sunflower (Helianthus annuus L.) Seeds in Health and Nutrition. In Nuts and Seeds in Health and Disease Prevention; Academic Press: Cambridge, MA, USA, 2011; pp. 1097–1105. [Google Scholar] [CrossRef]
- Adeleke, B.S.; Babalola, O.O. Oilseed crop sunflower (Helianthus annuus) as a source of food: Nutritional and health benefits. Food Sci. Nutr. 2020, 8, 4666–4684. [Google Scholar] [CrossRef]
- Guo, S.; Ge, Y.; Na Jom, K. A review of phytochemistry, metabolite changes, and medicinal uses of the common sunflower seed and sprouts (Helianthus annuus L.). Chem. Cent. J. 2017, 11, 95. [Google Scholar] [CrossRef]
- Salas, J.J.; Bootello, M.A.; Martínez-Force, E.; Venegas Calerón, M.; Garcés, R. High stearic sunflower oil: Latest advances and applications. OCL 2021, 28, 35. [Google Scholar] [CrossRef]
- Mehmood, A.; Zhao, L.; Ishaq, M.; Safdar, B.; Wang, C.; Nadeem, M. Optimization of total phenolic contents, antioxidant, and in-vitro xanthine oxidase inhibitory activity of sunflower head. CYTA J. Food 2018, 16, 957–964. [Google Scholar] [CrossRef]
- Tamara, N.; Angkasa, C.; Ginting, C.L.; Chiuman, L.; Lister, I.N.E. Determination Sunscreen Potential Based on Sunflower Seed Oil (Helianthus annuus) in Cream Preparation with Combination of Oxybenzone and Octyl Methoxycinnamate by in Vitro Method. Am. Sci. Res. J. Eng. Technol. Sci. 2019, 60, 33–39. [Google Scholar]
- Karamac, M.; Kosinska, A.; Estrella, I.; Hernandez, T.; Duenas, M. Antioxidant activity of phenolic compounds identified in sunflower seeds. Eur. Food Res. Technol. 2012, 235, 221–230. [Google Scholar] [CrossRef]
- HELIOXINE®. Sunflowers for a Photoprotection Strategy. Available online: https://www.silab.fr/produit-27-helioxine_usa.html (accessed on 21 September 2021).
- Wang, K.-J.; Li, X.-H.; Zhang, J.-J.; Chen, H.; Zhang, Z.-L.; Yu, G.-D. Natural introgression from cultivated soybean (Glycine max) into wild soybean (Glycine soja) with the implications for origin of populations of semi-wild type and for biosafety of wild species in China. Genet. Resour. Crop Evol. 2010, 57, 747–761. [Google Scholar] [CrossRef]
- Carvalho, D.O.; Curto, A.F.; Guido, L.F. Determination of Phenolic Content in Different Barley Varieties and Corresponding Malts by Liquid Chromatography-diode Array Detection-Electrospray Ionization Tandem Mass Spectrometry. Antioxidants 2015, 4, 563–576. [Google Scholar] [CrossRef] [PubMed]
- An, Y.Y.; Qun, Z.Y.; Yuan, L. Effects of quercetin and enhanced UV-B radiation on the soybean (Glycine max) leaves. Acta Physiol. Plant. 2006, 28, 49–57. [Google Scholar] [CrossRef]
- Soybean Extract—Supplied by Carruba. Available online: https://cosmetics.specialchem.com/product/i-carrubba-soybean-extract (accessed on 9 October 2021).
- Chen, Q.; Yuan, X.; Zhang, C.; Yan, N.; Jing, C.; Wang, X.; Shi, J. Comparison of Phenolic and Flavonoid Compound Profiles and Antioxidant and α-Glucosidase Inhibition Properties of Cultivated Soybean (Glycine max) and Wild Soybean (Glycine soja). Plants 2021, 10, 813. [Google Scholar] [CrossRef]
- Jing, C.; Yuan, Y.; Tang, Q.; Zou, P.; Li, Y.; Zhang, C. Extraction optimization, preliminary characterization and antioxidant activities of polysaccharides from Glycine soja. Int. J. Biol. Macromol. 2017, 103, 1207–1216. [Google Scholar] [CrossRef]
- Machado, A.C.R.; Monteiro, A.C.; Mochi, D.A. Selection of anti-wetting agents and photoprotectants compatible with a soybean weed control bioagent. Afr. J. Microbiol. Res. 2016, 10, 1829–1836. [Google Scholar] [CrossRef][Green Version]
- Tsoyi, K.; Park, H.B.; Kim, Y.M.; Chung, J.I.; Shin, S.C.; Shim, H.J.; Lee, W.S.; Seo, H.G.; Lee, J.H.; Chang, K.C.; et al. Protective Effect of Anthocyanins from Black Soybean Seed Coats on UVB-Induced Apoptotic Cell Death In Vitro and In Vivo. J. Agric. Food Chem. 2008, 56, 10600–10605. [Google Scholar] [CrossRef]
- Honfo, F.G.; Akissoe, N.; Linnemann, A.R.; Soumanou, M.; Van Boekel, M.A.J.S. Nutritional Composition of Shea Products and Chemical Properties of Shea Butter: A Review. Crit. Rev. Food Sci. Nutr. 2014, 54, 673–686. [Google Scholar] [CrossRef] [PubMed]
- Butyrospermum Parkii (Shea) Butter. Available online: https://cosmeticsinfo.org/ingredient/butyrospermum-parkii-shea-butter (accessed on 22 September 2021).
- Maanikuu, P.M.I.; Peker, K. Medicinal and Nutricional Benefits from the Shea-T- (Vitellaria paradoxa). J. Biol. Agric. Healthc. 2017, 7, 51–57. [Google Scholar]
- Fowler, J.; Sivelberg, N. Active Naturals Have a Key Role in Atopic Dermatitis. Semin. Cutan. Med. Surg. 2008, 27, 8–10. [Google Scholar] [CrossRef]
- Maranz, S.; Wiesman, Z.; Garti, N. Phenolic constituents of Shea (Vitellaria paradoxa) kernels. J. Agric. Food Chem. 2003, 51, 6268–6273. [Google Scholar] [CrossRef] [PubMed]
- Morocho-Jacome, A.L.; Freire, T.B.; de Oliveira, A.C.; Velasco, M.V.R.; Baby, A.R.; de, A.T.S.; Rosado, C. In Vivo SPF from multifunctional sunscreen systems developed with natural compounds—A review. J. Cosmet. Dermatol. 2021, 20, 729–737. [Google Scholar] [CrossRef] [PubMed]
- Sarruf, F.D.; Sauce, R.; Candido, T.M.; Oliveira, C.A.; Velasco, M.V.R.; Baby, A.R.; Rosado, C. Butyrospermum parkii butter increased the photostability and in vivo SPF of a molded sunscreen system. J. Cosmet. Dermatol. 2020, 19, 3296–3301. [Google Scholar] [CrossRef] [PubMed]
- Sarruf, F.D.; Candido, T.M.; de Oliveira, C.A.; Bou-Chacra, N.A.; Robles, V.M.V.; Baby, A.R. Influence of Shea (Butyrospermum parkii) butter, TiO2 and ethylhexyl methoxycinnamate on physical parameters and in vitro photoprotective efficacy. J. Cosmet. Dermatol. 2020, 19, 2076–2085. [Google Scholar] [CrossRef]
- Tarras-Wahlberg, N.; Stenhagen, G.; Larko, O.; Rosen, A.; Wennberg, A.M.; Wennerstrom, O. Changes in ultraviolet absorption of sunscreens after ultraviolet irradiation. J. Investig. Dermatol. 1999, 113, 547–553. [Google Scholar] [CrossRef]
- Persea Gratissima (Avocado) Oil. Available online: https://cosmeticsinfo.org/ingredient/persea-gratissima-avocado-oil-0 (accessed on 23 September 2021).
- Dreher, M.L.; Davenport, A.J. Hass avocado composition and potential health effects. Crit. Rev. Food Sci. Nutr. 2013, 53, 738–750. [Google Scholar] [CrossRef]
- Orhan, I.; Sener, B. Fatty Acid Content of Selected Seed Oils. J. Herb. Pharmacother. 2002, 2, 29–33. [Google Scholar] [CrossRef]
- Kruthiventi, A.K.; Krishnaswamy, N.R. Constituents of the flowers of Persea gratissima. Fitoterapia 2000, 71, 94–96. [Google Scholar] [CrossRef]
- Special Chem—Cosmetic Ingredients—Avocado Oil. Available online: https://cosmetics.specialchem.com/product/i-oqema-avocado-oil (accessed on 23 September 2021).
- Joseph, L.B.; Koukouras, K. Persea Gratissima (Avocado) Sterols decrease UVB induced proinflammatory mediators. J. Cosmet. Sci. 2002, 53, 309–311. [Google Scholar] [PubMed]
- Xie, J.; Zhang, Y.; Wang, W. HPLC analysis of glycyrrhizin and licochalcone A in Glycyrrhiza inflata from Xinjiang (China). Chem. Nat. Compd. 2010, 46, 148–151. [Google Scholar] [CrossRef]
- Furusawa, J.; Funakoshi-Tago, M.; Mashino, T.; Tago, K.; Inoue, H.; Sonoda, Y.; Kasahara, T. Glycyrrhiza inflata-derived chalcones, Licochalcone A, Licochalcone B and Licochalcone D, inhibit phosphorylation of NF-kappaB p65 in LPS signaling pathway. Int. Immunopharmacol. 2009, 9, 499–507. [Google Scholar] [CrossRef] [PubMed]
- European Medicines Agency—Committee on Herbal Medicinal Products. Assessment report on Glycyrrhiza glabra L. and/or Glycyrrhiza inflata Bat. and/or Glycyrrhiza uralensis Fisch., Radix; European Medicines Agency—Committee on Herbal Medicinal Products: London, UK, 2013. [Google Scholar]
- Fu, Y.; Chen, J.; Li, Y.-J.; Zheng, Y.-F.; Li, P. Antioxidant and anti-inflammatory activities of six flavonoids separated from licorice. Food Chem. 2013, 141, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Okada, K.; Tamura, Y.; Yamamoto, M.; Inoue, Y.; Takagaki, R.; Takahashi, K.; Demizu, S.; Kajiyama, K.; Hiraga, Y.; Kinoshita, T. Identification of antimicrobial and antioxidant constituents from licorice of Russian and Xinjiang origin. Chem. Pharm. Bull. 1989, 37, 2528–2530. [Google Scholar] [CrossRef] [PubMed]
- Monico, G.; Leo, M.; Ma, B.; Johal, R.S.; Ma, T.; Sivamani, R.K. The use of botanical products and vitamins in sunscreens. Dermatol. Online J. 2015, 21. [Google Scholar] [CrossRef]
- Naser, W. The Cosmetic Effects of Various Natural Biofunctional Ingredients against Skin Aging: A Review. Int. J. Appl. Pharm. 2021, 13, 10–18. [Google Scholar] [CrossRef]
- Castangia, I.; Caddeo, C.; Manca, M.L.; Casu, L.; Latorre, A.C.; Diez-Sales, O.; Ruiz-Sauri, A.; Bacchetta, G.; Fadda, A.M.; Manconi, M. Delivery of liquorice extract by liposomes and hyalurosomes to protect the skin against oxidative stress injuries. Carbohydr. Polym. 2015, 134, 657–663. [Google Scholar] [CrossRef]
- Saeedi, M.; Morteza-Semmani, K.; Ghoreishi, M.-R. The treatment of atopic dermatitis with licorice gel. J. Dermatol. Treat. 2008, 14, 153–157. [Google Scholar] [CrossRef]
- Nguyen, J.K.; Masub, N.; Jagdeo, J. Bioactive ingredients in Korean cosmeceuticals: Trends and research evidence. J. Cosmet. Dermatol. 2020, 19, 1555–1569. [Google Scholar] [CrossRef] [PubMed]
- Pareek, A.; Suthar, M.; Rathore, G.S.; Bansal, V. Feverfew (Tanacetum parthenium L.): A systematic review. Pharmacogn. Rev. 2011, 5, 103–110. [Google Scholar] [CrossRef] [PubMed]
- Grzeszczuk, M.; Grzeszczuk, M.; Wesołowska, A. Composition of essential oils and some antioxidants in flowers of three Chrysanthemum cultivars. J. Elem. 2018, 23, 1331–1342. [Google Scholar] [CrossRef]
- Romo, J.; de Vivar, A.R.; Trevino, R.; Joseph-Nathan, P.; Díaz, E. Constituents of Artemisia and Crysanthemum species—The structures of Chrysartemins A and B. Phytochemistry 1970, 9, 1615–1621. [Google Scholar] [CrossRef]
- Whkomirski, B.; Dubielecka, B. Sterol content as a similarity of different organs of two varietas of Chrysanthemum parthenium. Phytochemistry 1996, 42, 1603–1604. [Google Scholar] [CrossRef]
- Hangaru, D.; Benedec, D.; Vlase, L.; Popica, I.; Bele, C.; Raita, O.; Gheldiu, A.-M.; Mihali, C.V.; Tarmure, V. Polyphenolic content and antioxidant activity of Chrysanthemum Parthenium extract. Farmacia 2016, 64, 498–501. [Google Scholar]
- Williams, C.A.; Hoult, J.R.S.; Harbone, J.B.; Greenham, J.; Eagles, J. A biologically active lipophilic flavonol from Tanacetum Parthenium. Phytochemistry 1995, 38, 267–270. [Google Scholar] [CrossRef]
- Sun, S.; Jiang, P.; Su, W.; Xiang, Y.; Li, J.; Zeng, L.; Yang, S. Wild chrysanthemum extract prevents UVB radiation-induced acute cell death and photoaging. Cytotechnology 2016, 68, 229–240. [Google Scholar] [CrossRef]
- Zhao, Q.; Chen, X.Y.; Martin, C. Scutellaria baicalensis, the golden herb from the garden of Chinese medicinal plants. Sci. Bull. 2016, 61, 1391–1398. [Google Scholar] [CrossRef]
- Seok, J.K.; Kwak, J.Y.; Choi, G.W.; An, S.M.; Kwak, J.H.; Seo, H.H.; Suh, H.J.; Boo, Y.C. Scutellaria radix Extract as a Natural UV Protectant for Human Skin. Phytother. Res. 2016, 30, 374–379. [Google Scholar] [CrossRef]
- Jouy, C.; Ruiz, L.; Pygmalion, M.-J. Photoprotectant Compositions Comprising Baicalin. FR2977490A1, 11 January 2013. [Google Scholar]
- Salehi, B.; Albayrak, S.; Antolak, H.; Kregiel, D.; Pawlikowska, E.; Sharifi-Rad, M.; Uprety, Y.; Tsouh Fokou, P.V.; Yousef, Z.; Amiruddin Zakaria, Z.; et al. Aloe Genus Plants: From Farm to Food Applications and Phytopharmacotherapy. Int. J. Mol. Sci. 2018, 19, 2843. [Google Scholar] [CrossRef]
- Rodrigues, D.; Viotto, A.C.; Checchia, R.; Gomide, A.; Severino, D.; Itri, R.; Baptista, M.S.; Martins, W.K. Mechanism of Aloe Vera extract protection against UVA: Shelter of lysosomal membrane avoids photodamage. Photochem. Photobiol. Sci. 2016, 15, 334–350. [Google Scholar] [CrossRef] [PubMed]
- Fox, C. From aloe vera gel to stabilized vitamin C. Cosmet. Toiletries 2003, 118, 22–28. [Google Scholar]
- Park, H.M.; Moon, E.; Lee, S.; Kim, S.Y.; Do, S.-G.; Kim, J.; Liu, K.-H.; Lee, C.H. Topical application of baby-and adult-aloe on ultraviolet B irradiated mouse skin with metabolite profiling. Metabolomics 2015, 11, 1219–1230. [Google Scholar] [CrossRef]
- Yurin, V.O.; Kim, Y.A.; Muzafarov, E.N. Structural changes in lipid membranes and collagen irradiated with UV light and the protective effect of plant extracts. Biofizika 2004, 49, 666–673. [Google Scholar]
- Lee, H.; Choi, W.; Ro, H.; Kim, G.; Lee, H. Skin Antiaging Effects of the Fermented Outer Layers of Leaf Skin of Aloe barbadensis Miller Associated with the Enhancement of Mitochondrial Activities of UVb-Irradiated Human Skin Fibroblasts. Appl. Sci. 2021, 11, 5660. [Google Scholar] [CrossRef]
- Strickland, F.M.; Pelley, R.P. Plant saccharides and the prevention of sun-induced skin cancer. Med. Aromat. Plants Ind. Profiles 2004, 38, 265–310. [Google Scholar]
- Rodrigues, L.R.; Jose, J. Exploring the photo protective potential of solid lipid nanoparticle-based sunscreen cream containing Aloe vera. Environ. Sci. Pollut. Res. 2020, 27, 20876–20888. [Google Scholar] [CrossRef] [PubMed]
- Savva, S.C.; Kafatos, A. Vegetable Oils: Dietary Importance. In Encyclopedia of Food and Health; Elsevier: Amsterdam, The Netherlands, 2016; pp. 365–372. [Google Scholar] [CrossRef]
- Zhang, Y.; Duan, K.; Wang, W.; Chen, W.; Zhao, S. Antioxidant activities of de-fat coconut endocarp peptide. Zhongguo Liangyou Xuebao 2014, 29, 65–68, 73. [Google Scholar]
- Asghar, M.T.; Yusof, Y.A.; Mokhtar, M.N.; Ya’acob, M.E.; Mohd. Ghazali, H.; Chang, L.S.; Manaf, Y.N. Coconut (Cocos nucifera L.) sap as a potential source of sugar: Antioxidant and nutritional properties. Food Sci. Nutr. 2020, 8, 1777–1787. [Google Scholar] [CrossRef]
- Chakraborty, M.; Mitra, A. The antioxidant and antimicrobial properties of the methanolic extract from Cocos nucifera mesocarp. Food Chem. 2007, 107, 994–999. [Google Scholar] [CrossRef]
- Kaur, C.D.; Saraf, S. In vitro sun protection factor determination of herbal oils used in cosmetics. Pharmacogn. Res. 2010, 2, 22–25. [Google Scholar] [CrossRef]
- Widiyati, E. Determination of ultraviolet filter activity on coconut oil cosmetic cream. In 4th International Conference on Research, Implementation, and Education of Mathematics and Science (4TH ICRIEMS): Research and Education for Developing Scientific Attitude in Sciences And Mathematics; AIP Publishing: Melville, NY, USA, 2017. [Google Scholar] [CrossRef]
- Oliveira, M.B.S.; Valentim, I.B.; Santos, T.R.; Xavier, J.A.; Ferro, J.N.S.; Barreto, E.O.; Santana, A.E.G.; Melo, L.V.; Bottoli, C.B.G.; Goulart, M.O.F. Photoprotective and antiglycation activities of non-toxic Cocos nucifera Linn. (Arecaceae) husk fiber ethanol extract and its phenol chemical composition. Ind. Crops Prod. 2021, 162, 113246. [Google Scholar] [CrossRef]
- Muritala, H.F.; Akolade, J.O.; Akande, S.A.; Abdulazeez, A.T.; Aladodo, R.A.; Bello, A.B. Antioxidant and alpha-amylase inhibitory potentials of Cocos nucifera husk. Food Sci. Nutr. 2018, 6, 1676–1683. [Google Scholar] [CrossRef] [PubMed]
- Segars, K.; Mccarver, V.; Miller, R.A. Dermatologic Applications of Polypodium leucotomos: A Literature Review. J. Clin. Aesthet. Dermatol. 2021, 14, 50–60. [Google Scholar] [PubMed]
- Bhatia, N. Polypodium leucotomos: A potential new photoprotective agent. Am. J. Clin. Dermatol. 2015, 16, 73–79. [Google Scholar] [CrossRef] [PubMed]
- Berman, B.; Ellis, C.; Elmets, C. Polypodium leucotomos—An overview of basic investigative findings. J. Drugs Dermatol. 2016, 15, 224–228. [Google Scholar]
- Rabinovich, L.; Kazlouskaya, V. Herbal sun protection agents: Human studies. Clin. Dermatol. 2018, 36, 369–375. [Google Scholar] [CrossRef]
- El-Haj, N.; Goldstein, N. Sun protection in a pill: The photoprotective properties of Polypodium leucotomos extract. Int. J. Dermatol. 2015, 54, 362–366. [Google Scholar] [CrossRef]
- Gonzalez, S.; Pathak, M.A. Inhibition of ultraviolet-induced formation of reactive oxygen species, lipid peroxidation, erythema and skin photosensitization by Polypodium leucotomos. Photodermatol. Photoimmunol. Photomed. 1996, 12, 45–56. [Google Scholar] [CrossRef]
- Janczyk, A.; Garcia-Lopez, M.A.; Fernandez-Penas, P.; Alonso-Lebrero, J.L.; Benedicto, I.; Lopez-Cabrera, M.; Gonzalez, S. A Polypodium leucotomos extract inhibits solar-simulated radiation-induced TNF-alpha and iNOS expression, transcriptional activation and apoptosis. Exp. Dermatol. 2007, 16, 823–829. [Google Scholar] [CrossRef]
- Gonzalez, S.; Lucena, S.R.; Delgado, P.; Juarranz, A. Comparison of several hydrophilic extracts of Polypodium leucotomos reveals different antioxidant moieties and photoprotective effects in vitro. J. Med. Plants Res. 2018, 12, 336–345. [Google Scholar] [CrossRef]
- Fernandes, F.; Barbosa, M.; Oliveira, A.P.; Azevedo, I.C.; Sousa-Pinto, I.; Valentão, P.; Andrade, P.B. The pigments of kelps (Ochrophyta) as part of the flexible response to highly variable marine environments. J. Appl. Phycol. 2016, 28, 3689–3696. [Google Scholar] [CrossRef]
- Florez, N.; Gonzalez-Munoz, M.J.; Ribeiro, D.; Fernandes, E.; Dominguez, H.; Freitas, M. Algae Polysaccharides’ Chemical Characterization and their Role in the Inflammatory Process. Curr. Med. Chem. 2017, 24, 149–175. [Google Scholar] [CrossRef] [PubMed]
- Fradinho, P.; Flórez-Fernández, N.; Sousa, I.; Raymundo, A.; Domínguez, H.; Torres, M.D. Environmentally friendly processing of Laminaria ochroleuca for soft food applications with bioactive properties. J. Appl. Phycol. 2020, 32, 1455–1465. [Google Scholar] [CrossRef]
- Otero, P.; López-Martínez, M.I.; García-Risco, M.R. Application of pressurized liquid extraction (PLE) to obtain bioactive fatty acids and phenols from Laminaria ochroleuca collected in Galicia (NW Spain). J. Pharm. Biomed. Anal. 2019, 164, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Koch, M.; Glombitza, K.-W.; Eckhard, G. Phlorotannins of phaeophycea Laminaria ochroleuca. Phytochemistry 1980, 19, 1821–1823. [Google Scholar] [CrossRef]
- Vo, T.S.E.A. Brown Algal Polyphenol and Its Pharmaceutical Properties. In Marine-Derived Biomaterials for Tissue Engineering Applications; Choi, A., Ben-Nissan, B., Eds.; Springer: Berlin, Germany, 2019; pp. 223–243. [Google Scholar]
- Abida, H.; Ruchaud, S.; Rios, L.; Humeau, A.; Probert, I.; De Vargas, C.; Bach, S.; Bowler, C. Bioprospecting Marine Plankton. Mar. Drugs 2013, 11, 4594–4611. [Google Scholar] [CrossRef]
- Hylander, S. Mycosporine-Like Amino Acids (MAAs) in Zooplankton. Mar. Drugs 2020, 18, 72. [Google Scholar] [CrossRef]
- Karl, P.L.; Paul, F.L.; Antony, R.Y. Mycosporine-Like Amino Acids for Skin Photoprotection. Curr. Med. Chem. 2018, 25, 5512–5527. [Google Scholar] [CrossRef]
- Rosic, N.N. Mycosporine-Like Amino Acids: Making the Foundation for Organic Personalised Sunscreens. Mar. Drugs 2019, 17, 638. [Google Scholar] [CrossRef]
- Brekhman, I.I.; Golotin, V.G.; Gonenko, V.A.; Molokova, L.P.; Khasina, E.I. Antioxidant activity of neuston and plankton extracts from vostok bay, sea of Japan. Comp. Biochem. Physiol. B 1981, 70, 381–383. [Google Scholar] [CrossRef]
- Niwano, Y.; Sato, E.; Kohno, M.; Matsuyama, Y.; Kim, D.; Oda, T. Antioxidant Properties of Aqueous Extracts from Red Tide Plankton Cultures. Biosci. Biotechnol. Biochem. 2007, 71, 1145–1153. [Google Scholar] [CrossRef]
- Riane, K.; Ouled-Haddar, H.; Alyane, M.; Sifour, M.; Alyane, M.; Espinosa, C.; Angeles, E.M. Assessment of Streptococcus salivarius sp. thermophiles Antioxidant Efficiency and its Role in Reducing Paracetamol Hepatotoxicity. Iran J. Biotechnol. 2019, 17, e2061. [Google Scholar] [CrossRef]
- Afify, A.; Romeilah, R.; Sultan, S.; Hussein, M. Antioxidant activity and biological evaluations of probiotic bacteria strains. Int. J. Acad. Res. 2012, 4, 131–139. [Google Scholar] [CrossRef]
- Spyropoulos, B.G.; Misiakos, E.P.; Fotiadis, C.; Stoidis, C.N. Antioxidant properties of probiotics and their protective effects in the pathogenesis of radiation-induced Enteritis and Colitis. Dig. Dis. Sci. 2011, 56, 285–294. [Google Scholar] [CrossRef] [PubMed]
- Amaretti, A.; di Nunzio, M.; Pompei, A.; Raimondi, S.; Rossi, M.; Bordoni, A. Antioxidant properties of potentially probiotic bacteria: In vitro and in vivo activities. Appl. Microbiol. Biotechnol. 2013, 97, 809–817. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, G.; Traudt, M. Probiotic/Antioxidant Synergistic Blend Applicable in Skin Inflammation. WO2013137899A1, 19 September 2013. [Google Scholar]
- Kim, S.-K.; Ravichandran, Y.D.; Khan, S.B.; Kim, Y.T. Prospective of the cosmeceuticals derived from marine organisms. Biotechnol. Bioprocess Eng. 2008, 13, 511–523. [Google Scholar] [CrossRef]
- Mondon, P.; Ringenbach, C.; Marchand, T.; Ursulet, C. Thermus Thermophilus Fermentation Medium for Use in Prevention and Treatment of Infra-Red Related Skin Damages. EP2986305B1, 22 November 2017. [Google Scholar]
- Venuceane™. Available online: https://cosmetics.specialchem.com/product/i-croda-venuceane (accessed on 23 August 2021).
- Gorouhi, F.; Maibach, H.I. Topical Peptides and Proteins for Aging Skin. In Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; pp. 1–33. [Google Scholar] [CrossRef]
- Bauza, E.; Farra, C.D.; Domloge, N. Hsp70 induction by Artemia extract exhibits anti-inflammatory effect and down-regulates IL-1 and IL-8 synthesis in human hacat cells. J. Investig. Dermatol. 2001, 117, 415. [Google Scholar]
- Letsiou, S.; Kalliampakou, K.; Gardikis, K.; Mantecon, L.; Infante, C.; Chatzikonstantinou, M.; Labrou, N.E.; Flemetakis, E. Skin protective effects of Nannochloropsis gaditana extract on H2O2-stressed human dermal fibroblasts. Front. Mar. Sci. 2017, 4, 221. [Google Scholar] [CrossRef]
- Mourelle, M.L.; Gómez, C.P.; Legido, J.L. The Potential Use of Marine Microalgae and Cyanobacteria in Cosmetics and Thalassotherapy. Cosmetics 2017, 4, 46. [Google Scholar] [CrossRef]
- Chukhutsina, V.U.; Fristedt, R.; Morosinotto, T.; Croce, R. Photoprotection strategies of the alga Nannochloropsis gaditana. Biochim. Biophys. Acta Bioenerg. 2017, 1858, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Carbonera, D.; Agostini, A.; Di Valentin, M.; Gerotto, C.; Basso, S.; Giacometti, G.M.; Morosinotto, T. Photoprotective sites in the violaxanthin-chlorophyll a binding Protein (VCP) from Nannochloropsis gaditana. Biochim. Biophys. Acta Bioenerg. 2014, 1837, 1235–1246. [Google Scholar] [CrossRef] [PubMed]
- Koutsianas, N.; Letsiou, S.; Gardikis, K.; Dragani, P. A Combination of a Nannochloropsis Gaditana Extract and a Propolis Extract as Active Component in Cosmetic Formulations for Dermal Use. WO2017216588A1, 21 December 2017. [Google Scholar]
- Sellimi, S.; Maalej, H.; Rekik, D.M.; Benslima, A.; Ksouda, G.; Hamdi, M.; Sahnoun, Z.; Li, S.; Nasri, M.; Hajji, M. Antioxidant, antibacterial and in vivo wound healing properties of laminaran purified from Cystoseira barbata seaweed. Int. J. Biol. Macromol. 2018, 119, 633–644. [Google Scholar] [CrossRef] [PubMed]
- Agregan, R.; Munekata, P.E.S.; Franco, D.; Carballo, J.; Barba, F.J.; Lorenzo, J.M. Antioxidant potential of extracts obtained from macro- (Ascophyllum nodosum, Fucus vesiculosus and Bifurcaria bifurcata) and micro-algae (Chlorella vulgaris and Spirulina platensis) assisted by ultrasound. Medicines 2018, 5, 33. [Google Scholar] [CrossRef] [PubMed]
- Sineiro Torres, J.; Sanchez Guerrero, M.; Nunez Garcia, M.J. Antioxidant Extracts from Brown Macroalgae. ES2441469A1, 4 February 2014. [Google Scholar]
- Peinado, I.; Giron, J.; Koutsidis, G.; Ames, J.M. Chemical composition, antioxidant activity and sensory evaluation of five different species of brown edible seaweeds. Food Res. Int. 2014, 66, 36–44. [Google Scholar] [CrossRef]
- Blanco-Pascual, N.; Montero, M.P.; Gomez-Guillen, M.C. Antioxidant film development from unrefined extracts of brown seaweeds Laminaria digitata and Ascophyllum nodosum. Food Hydrocoll. 2014, 37, 100–110. [Google Scholar] [CrossRef]
- Abu, R.; Jiang, Z.; Ueno, M.; Okimura, T.; Yamaguchi, K.; Oda, T. In vitro antioxidant activities of sulfated polysaccharide ascophyllan isolated from Ascophyllum nodosum. Int. J. Biol. Macromol. 2013, 59, 305–312. [Google Scholar] [CrossRef]
- Dutot, M.; Fagon, R.; Hemon, M.; Rat, P. Antioxidant, Anti-inflammatory, and Anti-senescence Activities of a Phlorotannin-Rich Natural Extract from Brown Seaweed Ascophyllum nodosum. Appl. Biochem. Biotechnol. 2012, 167, 2234–2240. [Google Scholar] [CrossRef]
- Bourges, C.; Pauquai, T. Antioxidant Composition Containing Extracts of Nettles, Ascophyllum, and Acerola, and Vitamin C. FR2964875A1, 23 March 2012. [Google Scholar]
- Jimenez, J.T.; O’Connell, S.; Lyons, H.; Bradley, B.; Hall, M. Antioxidant, antimicrobial, and tyrosinase inhibition activities of acetone extract of Ascophyllum nodosum. Chem. Pap. 2010, 64, 434–442. [Google Scholar] [CrossRef]
- Connan, S.; Deslandes, E.; Gall, E.A. Influence of day-night and tidal cycles on phenol content and antioxidant capacity in three temperate intertidal brown seaweeds. J. Exp. Mar. Biol. Ecol. 2007, 349, 359–369. [Google Scholar] [CrossRef]
- Le Tutour, B.; Benslimane, F.; Gouleau, M.P.; Gouygou, J.P.; Saadan, B.; Quemeneur, F. Antioxidant and pro-oxidant activities of the brown algae, Laminaria digitata, Himanthalia elongata, Fucus vesiculosus, Fucus serratus and Ascophyllum nodosum. J. Appl. Phycol. 1998, 10, 121–129. [Google Scholar] [CrossRef]
- Ferreira, M.S.; Resende, D.I.S.P.; Lobo, J.M.S.; Sousa, E.; Almeida, I.F. Marine Ingredients for Sensitive Skin: Market Overview. Mar. Drugs 2021, 19, 464. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-J.; Kim, K.B.; Heo, J.; Cho, D.-H.; Kim, H.-S.; Han, S.H.; Ahn, K.J.; An, I.-S.; An, S.; Bae, S. Protective effect of Arthrospira platensis extracts against ultraviolet B-induced cellular senescence through inhibition of DNA damage and matrix metalloproteinase-1 expression in human dermal fibroblasts. J. Photochem. Photobiol. B: Biol. 2017, 173, 196–203. [Google Scholar] [CrossRef]
- Mapoung, S.; Arjsri, P.; Thippraphan, P.; Semmarath, W.; Yodkeeree, S.; Chiewchanvit, S.; Piyamongkol, W.; Limtrakul, P. Photochemoprotective effects of Spirulina platensis extract against UVB irradiated human skin fibroblasts. S. Afr. J. Bot. 2020, 130, 198–207. [Google Scholar] [CrossRef]
- Figueroa, F.L.; Bueno, A.; Korbee, N.; Santos, R.; Mata, L.; Schuenhoff, A. Accumulation of Mycosporine-like Amino Acids in Asparagopsis armata Grown in Tanks with Fishpond Effluents of Gilthead Sea Bream, Sparus aurata. J. World Aquac. Soc. 2008, 39, 692–699. [Google Scholar] [CrossRef]
- Nishigori, C.; Hattori, Y.; Arima, Y.; Miyachi, Y. Photoaging and oxidative stress. Exp. Dermatol. 2003, 12, 18–21. [Google Scholar] [CrossRef] [PubMed]
- Brenneisen, P.; Sies, H.; Scharffetter-Kochanek, K. Ultraviolet-B Irradiation and Matrix Metalloproteinases. Ann. N. Y. Acad. Sci. 2002, 973, 31–43. [Google Scholar] [CrossRef]
- Bennett, M.F.; Robinson, M.K.; Baron, E.D.; Cooper, K.D. Skin Immune Systems and Inflammation: Protector of the Skin or Promoter of Aging? J. Investig. Dermatol. Symp. Proc. 2008, 13, 15–19. [Google Scholar] [CrossRef]
- Ullrich, S.E. Mechanisms underlying UV-induced immune suppression. Mutat Res. 2005, 571, 185–205. [Google Scholar] [CrossRef] [PubMed]
- Yarosh, D.B. DNA Damage and Repair in Skin Aging. In Textbook of Aging Skin; Farage, M.A., Miller, K.W., Maibach, H.I., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 323–331. [Google Scholar] [CrossRef]
- Widmer, R.; Ziaja, I.; Grune, T. Protein oxidation and degradation during aging: Role in skin aging and neurodegeneration. Free Radic. Res. 2006, 40, 1259–1268. [Google Scholar] [CrossRef]
- Niki, E. Lipid oxidation in the skin. Free Rad. Res. 2015, 49, 827–834. [Google Scholar] [CrossRef] [PubMed]
- Farrar, M.D. Advanced glycation end products in skin ageing and photoageing: What are the implications for epidermal function? Exp. Dermatol. 2016, 25, 947–948. [Google Scholar] [CrossRef] [PubMed]
- Park, H.Y.; Kosmadaki, M.; Yaar, M.; Gilchrest, B.A. Cellular mechanisms regulating human melanogenesis. Cell. Mol. Life Sci. 2009, 66, 1493–1506. [Google Scholar] [CrossRef] [PubMed]
- Fitsiou, E.; Pulido, T.; Campisi, J.; Alimirah, F.; Demaria, M. Cellular Senescence and the Senescence-Associated Secretory Phenotype as Drivers of Skin Photoaging. J. Investig. Dermatol. 2021, 141, 1119–1126. [Google Scholar] [CrossRef]

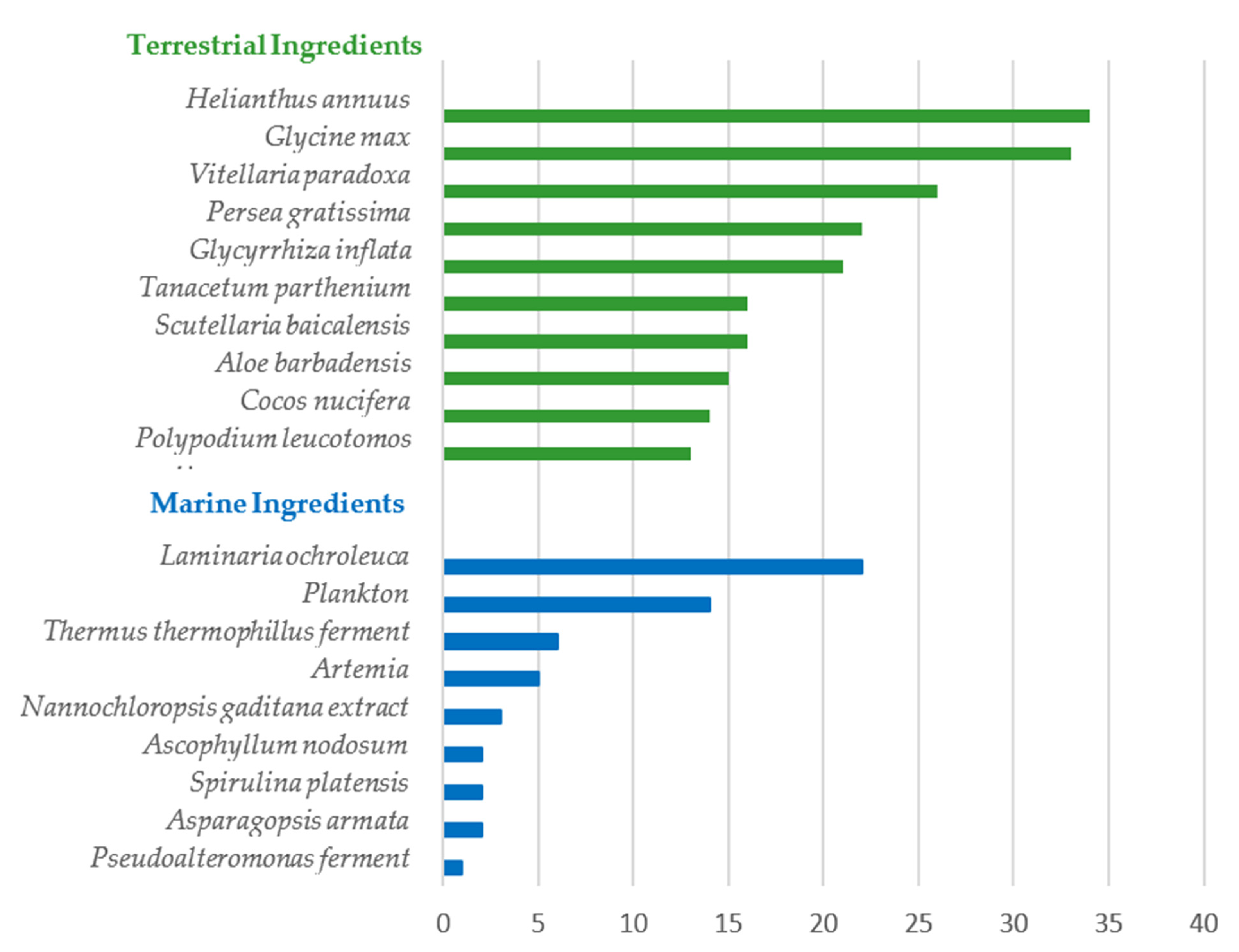

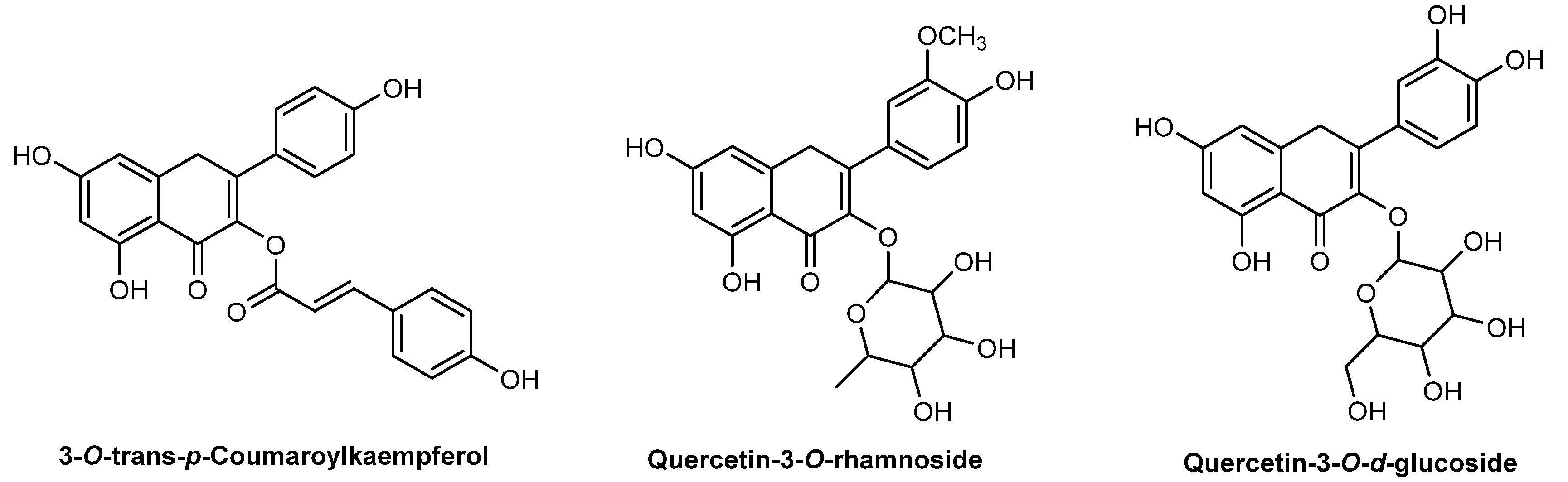

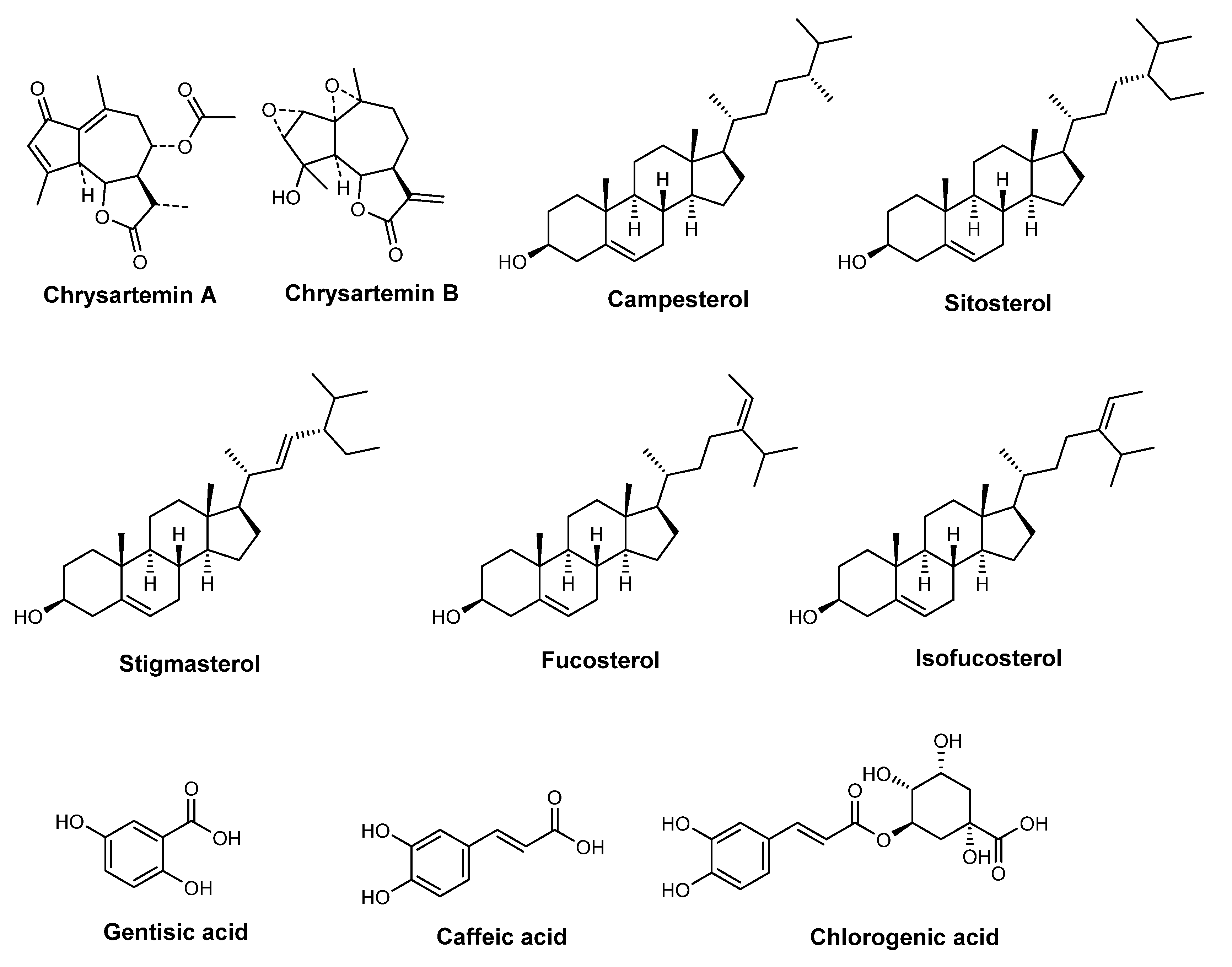
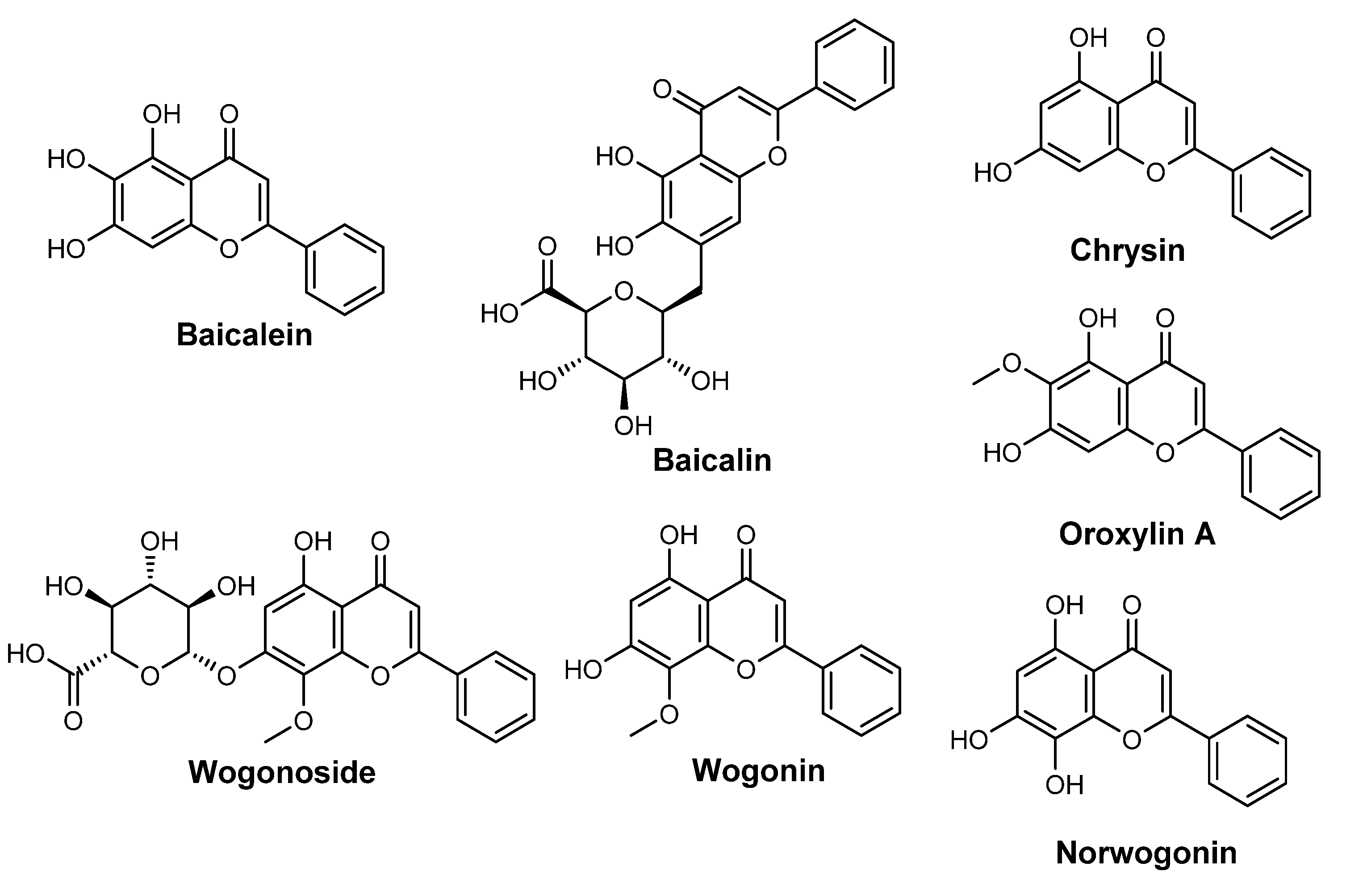
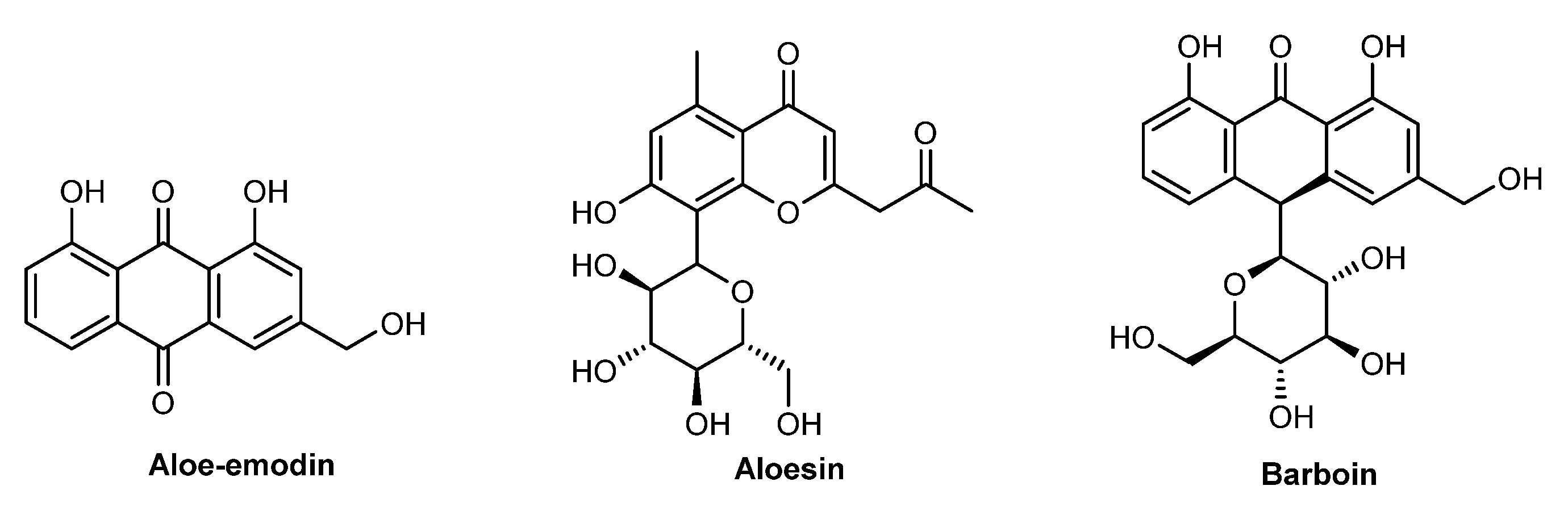
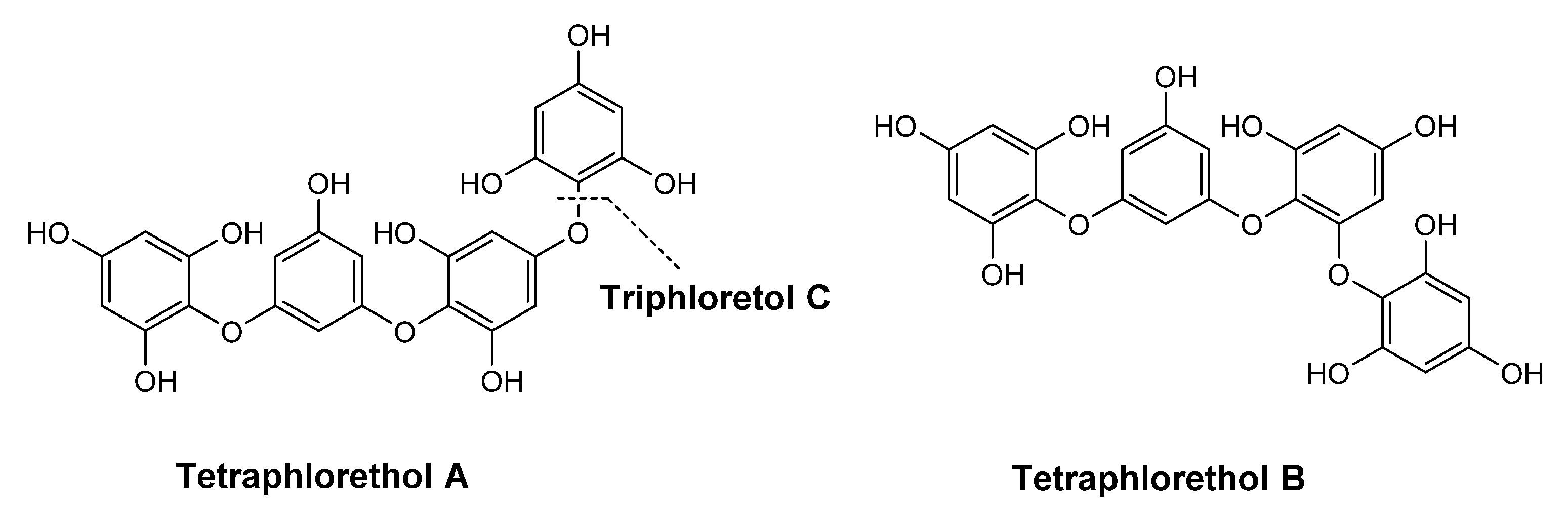
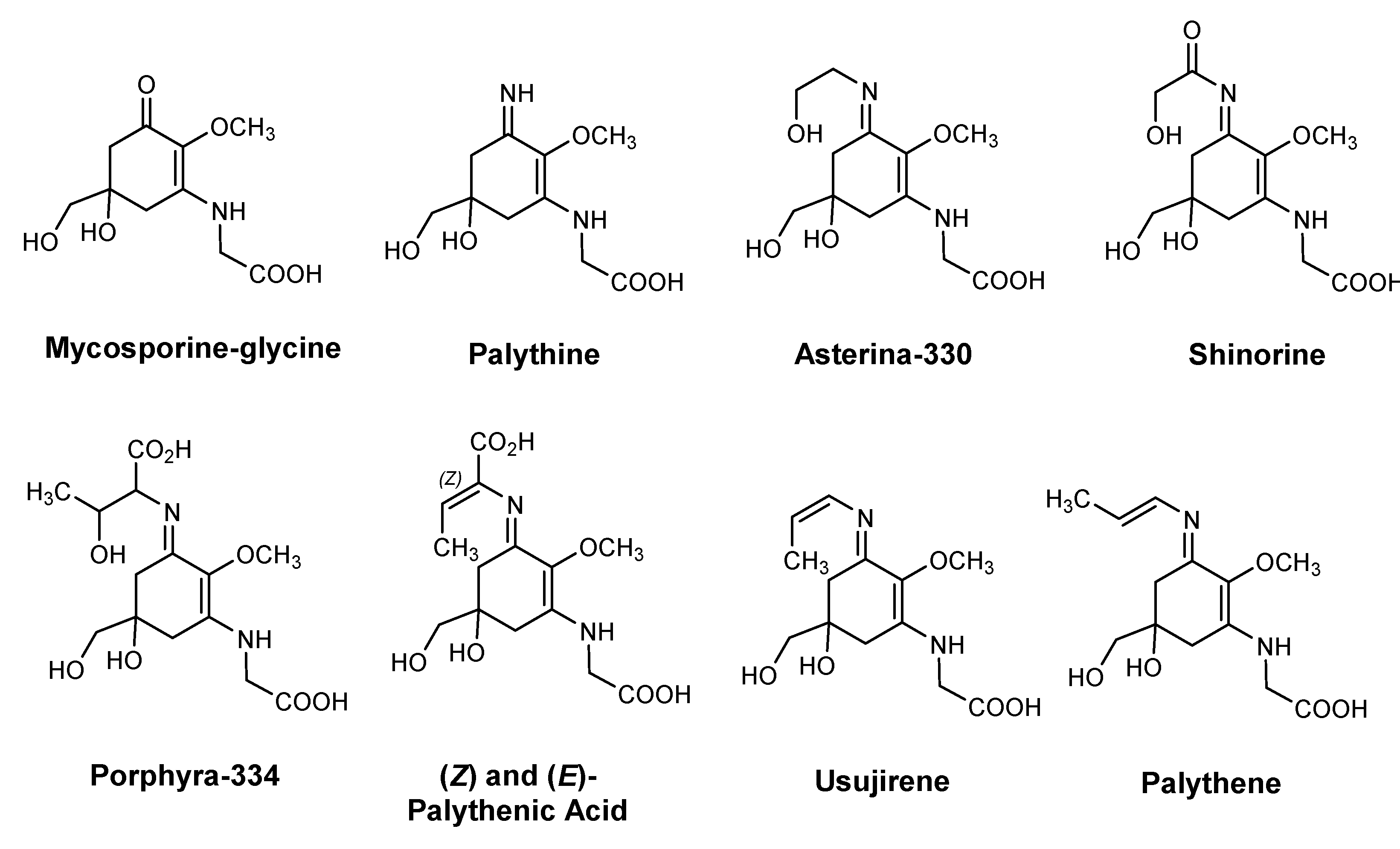
| Species | Usage (%) | INCI | Description |
|---|---|---|---|
| H. annuus (34) | |||
| H. annuus seed oil | 34 (7.7%) | H. annuus seed oil | H. Annuus Seed Oil is the oil expressed from the seeds of the Sunflower, H. annuus L., Compositae |
| Glycine max (33) | |||
| G. max oil | 30 (6.8%) | Glycine soja oil | G. Soja Oil is the oil obtained from the soybean, G. soja, Leguminosae, by extraction or expression. It consists essentially of triglycerides of oleic, linoleic and saturated acids |
| G. max seed extract | 3 (0.7%) | G. soja seed extract | G. Soja Seed is an extract of the Soybean, G. soja, Leguminosae |
| V. paradoxa (26) | |||
| V. paradoxa butter | 16 (3.6%) | Butyrospermum parkii butter | B.Parkii Seedcake Extract is the extract of the seedcake of the Shea Tree, B. parkii, Sapotaceae |
| V. paradoxa butter extract | 8 (1.8%) | B. parkii butter extract | B. Parkii Butter is the fat obtained from the fruit of the Shea Tree, B. parkii, Sapotaceae |
| V. paradoxa butter seedcake extract | 2 (0.5%) | B. parkii butter seedcake extract | B. Parkii Butter Extract is an extract obtained from the Shea Tree, B. parkii, Sapotaceae |
| P. gratissima (22) | |||
| P. gratissima fruit extract | 11 (2.5%) | P. gratissima fruit extract | P. Gratissima Fruit Extract is an extract of the fruit of the Avocado, P. gratissima, Lauraceae |
| P. gratissima oil | 11 (2.5%) | P. gratissima oil | P. Gratissima Oil is the fixed oil obtained by pressing the dehydrated sliced flesh of the avocado pear, P. gratissima, Lauraceae. It consists principally of the glycerides of fatty acids |
| G. inflata (21) | |||
| G. inflata root extract | 21 (4.7%) | G. inflata root extract | G. Inflata Root Extract is an extract of the roots of G. inflata, Leguminosae |
| T. parthenium (16) | |||
| T. parthenium extract | 7 (1.6%) | Chrysanthemum parthenium extract | C. Parthenium Extract is an extract of the herb of the feverfew, C. parthenium, Asteraceae |
| T. parthenium flower extract | 9 (2.0%) | C. parthenium flower extract | C. Parthenium Flower Extract is an extract of the flowers the feverfew, C. parthenium, Asteraceae |
| S. baicalensis (16) | |||
| S. baicalensis extract | 8 (1.8%) | S. baicalensis extract | S. Baicalensis Extract is the extract of the whole plant, S. baicalensis, Lamiaceae |
| S. baicalensis root extract | 8 (1.8%) | S. baicalensis root extract | S. Baicalensis Root Extract is an extract of the roots of the S. baicalensis, Lamiaceae |
| A. barbadensis (15) | |||
| A. barbadensis leaf extract | 2 (0.5%) | A. barbadensis leaf extract | A. Barbadensis Leaf Extract is an extract of the leaves of the aloe, A. barbadensis, Liliaceae |
| A. barbadensis leaf juice | 5 (1.1%) | A. barbadensis leaf juice | A. Barbadensis Leaf Juice is the juice expressed from the leaves of the aloe, A. barbadensis, Liliaceae |
| A. barbadensis leaf juice powder | 6 (1.4%) | A. barbadensis leaf juice powder | A. Barbadensis Leaf Juice Powder is the powder obtained from the dried juice leaves of the aloe, A. barbadensis, Liliaceae |
| A. barbadensis leaf water | 2 (0.5%) | A. barbadensis leaf water | A. Barbadensis Leaf Water is an aqueous solution of the steam distillate obtained from the leaves of the aloe, A. barbadensis, Liliaceae |
| C. nucifera (14) | |||
| C. nucifera oil | 14 (3.2%) | C. nucifera oil | C. Nucifera Oil is the fixed oil obtained by expression of the kernels of the seeds of the Coconut, C. nucifera L., Palmaceae |
| P. leucotomos (13) | |||
| P. leucotomos leaf extract | 13 (2.9%) | P. leucotomos leaf extract | P. Leucotomos Leaf Extract is an extract of the leaves of P. leucotomos, Polypodiaceae |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Resende, D.I.S.P.; Jesus, A.; Sousa Lobo, J.M.; Sousa, E.; Cruz, M.T.; Cidade, H.; Almeida, I.F. Up-to-Date Overview of the Use of Natural Ingredients in Sunscreens. Pharmaceuticals 2022, 15, 372. https://doi.org/10.3390/ph15030372
Resende DISP, Jesus A, Sousa Lobo JM, Sousa E, Cruz MT, Cidade H, Almeida IF. Up-to-Date Overview of the Use of Natural Ingredients in Sunscreens. Pharmaceuticals. 2022; 15(3):372. https://doi.org/10.3390/ph15030372
Chicago/Turabian StyleResende, Diana I. S. P., Ana Jesus, José M. Sousa Lobo, Emília Sousa, Maria T. Cruz, Honorina Cidade, and Isabel F. Almeida. 2022. "Up-to-Date Overview of the Use of Natural Ingredients in Sunscreens" Pharmaceuticals 15, no. 3: 372. https://doi.org/10.3390/ph15030372
APA StyleResende, D. I. S. P., Jesus, A., Sousa Lobo, J. M., Sousa, E., Cruz, M. T., Cidade, H., & Almeida, I. F. (2022). Up-to-Date Overview of the Use of Natural Ingredients in Sunscreens. Pharmaceuticals, 15(3), 372. https://doi.org/10.3390/ph15030372













