Potential Therapeutic Effects of Citrus hystrix DC and Its Bioactive Compounds on Metabolic Disorders
Abstract
:1. Introduction
2. Literature Search
3. Traditional Medicinal Uses
4. Phytochemical Properties
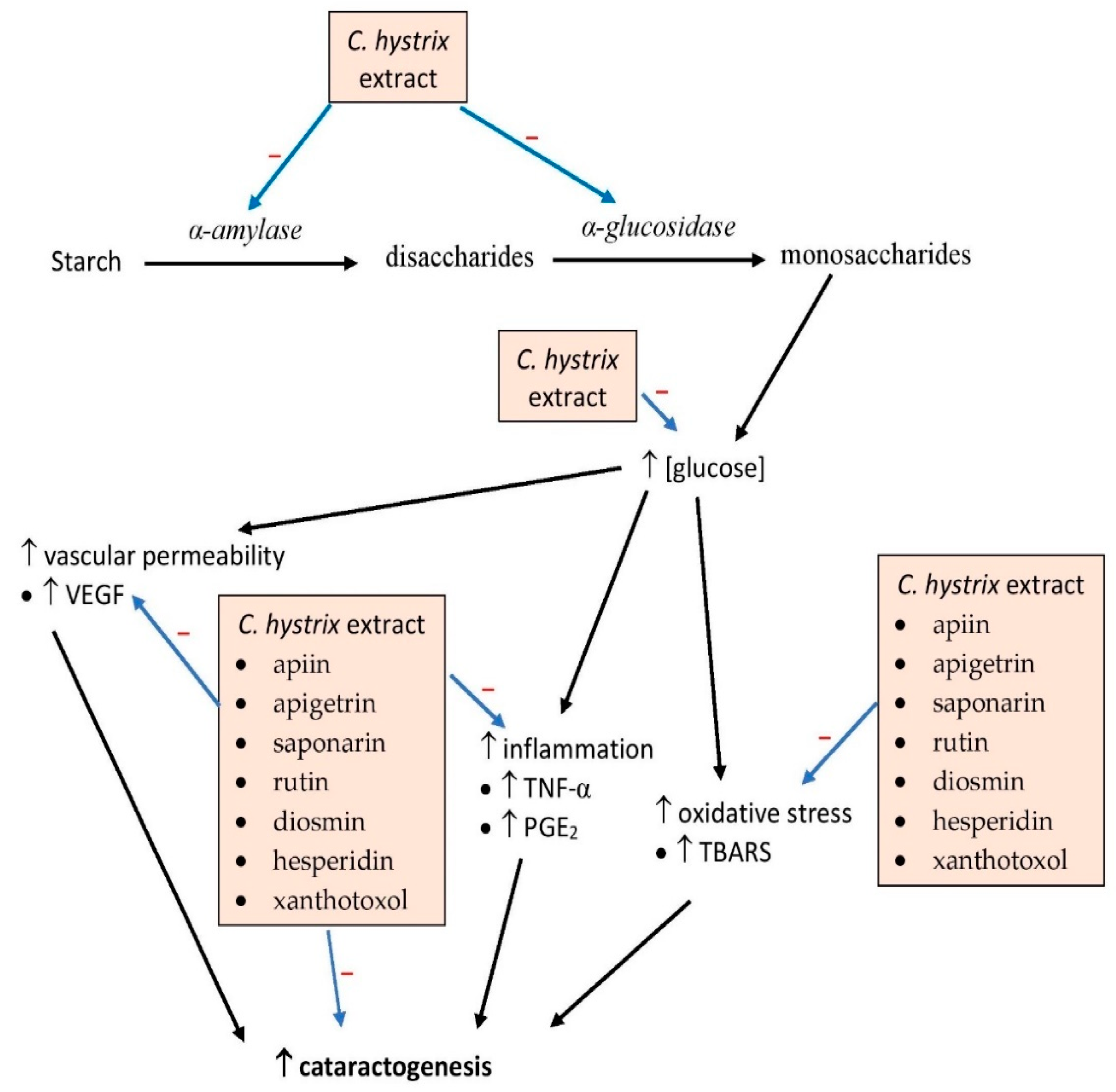
5. Effects on Diabetes
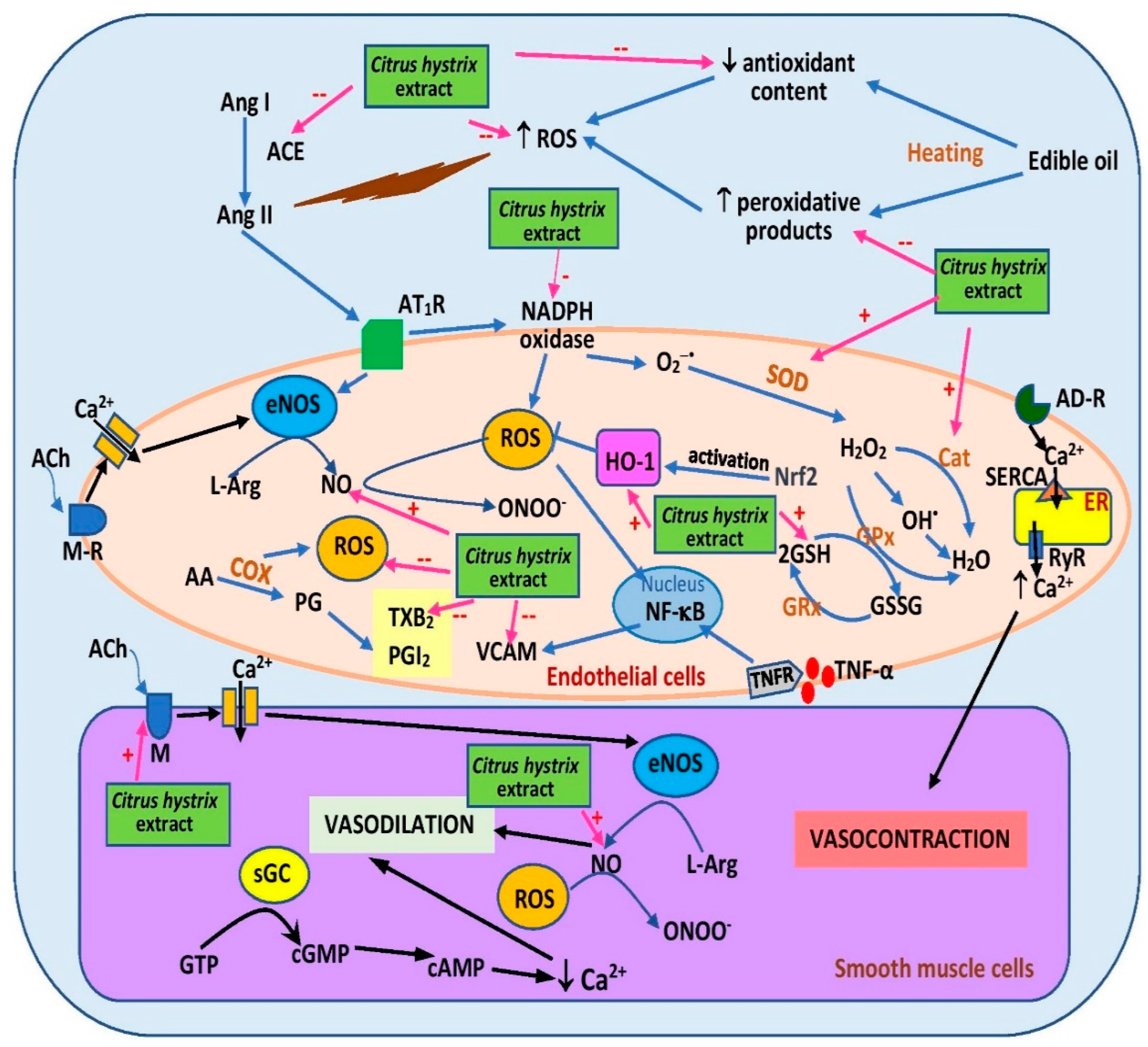
6. Effects on Hypertension
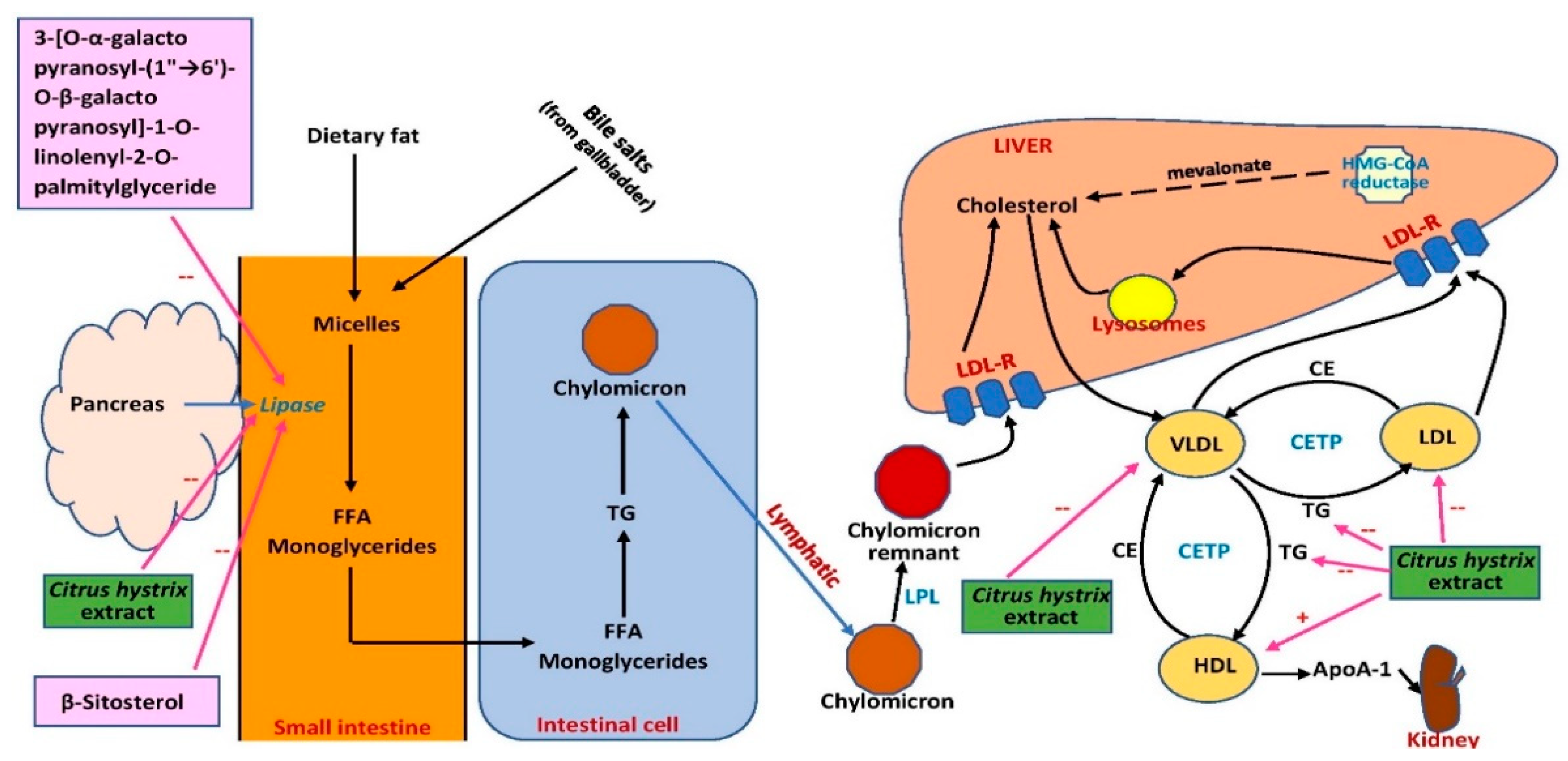
7. Effects on Cholesterol Level and Obesity
8. Pharmacokinetics and Safety
9. Conclusions and Directions for Future Study
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Su, Z.; Guo, Y.; Huang, X.; Feng, B.; Tang, L.; Zheng, G.; Zhu, Y. Phytochemicals: Targeting mitophagy to treat metabolic disorders. Front. Cell. Dev. Biol. 2021, 9, 686820. [Google Scholar] [CrossRef] [PubMed]
- Auriemma, R.S.; Pirchio, R.; Liccardi, A.; Scairati, R.; Del Vecchio, G.; Pivonello, R.; Colao, A. Metabolic syndrome in the era of COVID-19 outbreak: Impact of lockdown on cardiometabolic health. J. Endocrinol. Investig. 2021, 44, 2845–2847. [Google Scholar] [CrossRef] [PubMed]
- Torres, S.; Medina, R.B.; Vasallo Morillas, M.I.; Isla, M.I.; Gauffin-Cano, P. Editorial: Functional foods and bioactive food ingredients in prevention and alleviation of metabolic syndrome. Front. Nutr. 2021, 8, 788941. [Google Scholar] [CrossRef] [PubMed]
- Kamisah, Y.; Zuhair, J.S.F.; Juliana, A.H.; Jaarin, K. Parkia speciosa empty pod prevents hypertension and cardiac damage in rats given N(G)-nitro-l-arginine methyl ester. Biomed. Pharmacother. 2017, 96, 291–298. [Google Scholar] [CrossRef] [PubMed]
- Yu, F.; Wang, Y.; Teng, Y.; Yang, S.; He, Y.; Zhang, Z.; Yang, H.; Ding, C.F.; Zhou, P. Interaction and inhibition of a Ganoderma lucidum proteoglycan on PTP1B activity for anti-diabetes. ACS Omega 2021, 6, 29804–29813. [Google Scholar] [CrossRef] [PubMed]
- de Moraes Arnoso, B.J.; Magliaccio, F.M.; de Araújo, C.A.; de Andrade Soares, R.; Santos, I.B.; de Bem, G.F.; Fernandes-Santos, C.; Ognibene, D.T.; de Moura, R.S.; Resende, A.C.; et al. Acai seed extract (ASE) rich in proanthocyanidins improves cardiovascular remodeling by increasing antioxidant response in obese high-fat diet-fed mice. Chem. Biol. Interact. 2021, 351, 109721. [Google Scholar] [CrossRef]
- Umran, N.S.S.; Mohamed, S.; Lau, S.F.; Mohd Ishak, N.I. Citrus hystrix leaf extract attenuated diabetic-cataract in STZ-rats. J. Food Biochem. 2020, 44, e13258. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Nur Iliyani, M.I.; Mohamed, S.; Jaarin, K. Citrus leaf extract reduces blood pressure and vascular damage in repeatedly heated palm oil diet-induced hypertensive rats. Biomed. Pharmacother. 2017, 87, 451–460. [Google Scholar] [CrossRef]
- Nugraheni, D.M.; Kurniati, I.D.; Deliara, H.; Kusuma, M.A. Kadar LDL tikus betina setelah pemberian ekstrak kulit jeruk purut (Citrus hystrix). Herb. Med. J. 2020, 3, 39–46. [Google Scholar] [CrossRef]
- Lim, T.K. Citrus hystrix. In Edible Medicinal and Non-Medicinal Plants: Fruits; Springer Science + Business Media B.V.: Dordrecht, The Netherlands, 2012; Volume 4, pp. 634–643. [Google Scholar]
- Das, S.C.; Prakash, J.; Deb, A.K.; Biswas, T. Medicinal value of underutilized fruits in hilly Tripura. Acta Hortic. 2013, 972, 135–141. [Google Scholar] [CrossRef]
- Md Othman, S.N.A.; Hassan, M.A.; Nahar, L.; Basar, N.; Jamil, S.; Sarker, S.D. Essential oils from the Malaysian citrus (Rutaceae) medicinal plants. Medicines 2016, 3, 13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silalahi, M.; Nisyawati, N. An ethnobotanical study of traditional steam-bathing by the Batak people of North Sumatra, Indonesia. Pac. Conserv. Biol. 2018, 25, 266–282. [Google Scholar] [CrossRef]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. The medicinal and nutritional role of underutilized citrus fruit Citrus hystrix (Kaffir lime): A review. Drug Invent. Today 2014, 6, 1–5. [Google Scholar]
- Neamsuvan, O.; Komonhiran, P.; Boonming, K. Medicinal plants used for hypertension treatment by folk healers in Songkhla province, Thailand. J. Ethnopharmacol. 2018, 214, 58–70. [Google Scholar] [CrossRef]
- Yabesh, J.E.; Prabhu, S.; Vijayakumar, S. An ethnobotanical study of medicinal plants used by traditional healers in silent valley of Kerala, India. J. Ethnopharmacol. 2014, 154, 774–789. [Google Scholar] [CrossRef]
- Alsarhan, A.; Sultana, N.; Kadir, M.R.A.; Aburjai, T. Ethnopharmacological survey of medicinal plants in Malaysia, the Kangkar Pulai region. Int. J. Pharmacol. 2012, 8, 679–686. [Google Scholar] [CrossRef]
- Anuchapreeda, S.; Anzawa, R.; Viriyaadhammaa, N.; Neimkhum, W.; Chaiyana, W.; Okonogi, S.; Usuki, T. Isolation and biological activity of agrostophillinol from kaffir lime (Citrus hystrix) leaves. Bioorg. Med. Chem. Lett. 2020, 30, 127256. [Google Scholar] [CrossRef]
- Suttisansanee, U.; Thiyajai, P.; Chalermchaiwat, P.; Wongwathanarat, K.; Pruesapan, K.; Charoenkiatkul, S.; Temviriyanukul, P. Phytochemicals and in vitro bioactivities of aqueous ethanolic extracts from common vegetables in Thai food. Plants 2021, 10, 1563. [Google Scholar] [CrossRef]
- Chanthaphon, S.; Chanthachum, S.; Hongpattarakere, T. Antimicrobial activities of essential oils and crude extracts from tropical Citrus spp. against food-related microorganisms. Songklanakarin J. Sci. Technol. 2008, 30, 125–131. [Google Scholar]
- Dertyasasa, E.D.; Tunjung, W.A.S. Volatile organic compounds of kaffir lime (Citrus Hystrix DC.) leaves fractions and their potency as traditional medicine. Biosci. Biotech. Res. Asia 2017, 14, 1235–1250. [Google Scholar] [CrossRef]
- Anuchapreeda, S.; Chueahongthong, F.; Viriyaadhammaa, N.; Panyajai, P.; Anzawa, R.; Tima, S.; Ampasavate, C.; Saiai, A.; Rungrojsakul, M.; Usuki, T.; et al. Antileukemic cell proliferation of active compounds from kaffir lime (Citrus hystrix) leaves. Molecules 2020, 25, 1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buakaew, W.; Pankla Sranujit, R.; Noysang, C.; Thongsri, Y.; Potup, P.; Nuengchamnong, N.; Suphrom, N.; Usuwanthim, K. Phytochemical constituents of Citrus hystrix DC. leaves attenuate inflammation via NF-κB signaling and NLRP3 inflammasome activity in macrophages. Biomolecules 2021, 11, 105. [Google Scholar] [CrossRef] [PubMed]
- Panthong, K.; Srisud, Y.; Rukachaisirikul, V.; Hutadilok-Towatana, N.; Voravuthikunchai, S.P.; Tewtrakul, S. Benzene, coumarin and quinolinone derivatives from roots of Citrus hystrix. Phytochemistry 2013, 88, 79–84. [Google Scholar] [CrossRef] [PubMed]
- Murakami, A.; Nakamura, Y.; Koshimizu, K.; Ohigashi, H. Glyceroglycolipids from Citrus hystrix, a traditional herb in Thailand, potently inhibit the tumor-promoting activity of 12-O-tetradecanoylphorbol 13-acetate in mouse skin. J. Agric. Food Chem. 1995, 43, 2779–2783. [Google Scholar] [CrossRef]
- Ching, L.S.; Mohamed, S. Alpha-Tocopherol content in 62 edible tropical plants. J. Agric. Food Chem. 2001, 49, 3101–3105. [Google Scholar] [CrossRef] [PubMed]
- Butryee, C.; Sungpuag, P.; Chitchumroonchokchai, C. Effect of processing on the flavonoid content and antioxidant capacity of Citrus hystrix leaf. Int. J. Food Sci. Nutr. 2009, 60, 162–174. [Google Scholar] [CrossRef] [PubMed]
- Roowi, S.; Crozier, A. Flavonoids in tropical citrus species. J. Agric. Food Chem. 2011, 59, 12217–12225. [Google Scholar] [CrossRef] [PubMed]
- Ito, C.; Fujiwara, K.; Kajita, M.; Ju-Ichi, M.; Takemura, Y.; Suzuki, Y.; Tanaka, K.; Omura, M.; Furukawa, H. New coumarins from citrus plants. Chem. Pharm. Bull. 1991, 39, 2509–2513. [Google Scholar] [CrossRef] [Green Version]
- Youkwan, J.; Sutthivaiyakit, S.; Sutthivaiyakit, P. Citrusosides A–D and furanocoumarins with cholinesterase inhibitory activity from the fruit peels of Citrus hystrix. J. Nat. Prod. 2010, 73, 1879–1883. [Google Scholar] [CrossRef]
- Sun, S.; Phrutivorapongkul, A.; Dibwe, D.F.; Balachandran, C.; Awale, S. Chemical constituents of Thai Citrus hystrix and their antiausterity activity against the PANC-1 human pancreatic cancer cell line. J. Nat. Prod. 2018, 81, 1877–1883. [Google Scholar] [CrossRef]
- Sadasivam, M.; Kumarasamy, C.; Thangaraj, A.; Govindan, M.; Kasirajan, G.; Vijayan, V.; Devadasan, V.; Chia-Her, L.; Madhusudhanan, G.R.; Ramaraj, T.; et al. Phytochemical constituents from dietary plant Citrus hystrix. Nat. Prod. Res. 2018, 32, 1721–1726. [Google Scholar] [CrossRef] [PubMed]
- Seeka, C.; Sutthivaiyakit, P.; Youkwan, J.; Hertkorn, N.; Harir, M.; Schmitt-Kopplin, P.; Sutthivaiyakit, S. Prenylfuranocoumarin-HMGA-flavonol glucoside conjugates and other constituents of the fruit peels of Citrus hystrix and their anticholinesterase activity. Phytochemistry 2016, 127, 38–49. [Google Scholar] [CrossRef] [PubMed]
- Shaha, R.K.; Punichelvana, Y.N.A.P.; Afandi, A. Optimized extraction condition and characterization of pectin from kaffir lime (Citrus hystrix). Res. J. Agric. Forest. Sci. 2013, 1, 1–11. [Google Scholar]
- Warsito, W.; Palungan, M.H.; Utomo, E.P. Profiling study of the major and minor components of kaffir lime oil (Citrus hystrix DC.) in the fractional distillation process. Pan Afr. Med. J. 2017, 27, 282. [Google Scholar] [CrossRef] [PubMed]
- Waikedre, J.; Dugay, A.; Barrachina, I.; Herrenknecht, C.; Cabalion, P.; Fournet, A. Chemical composition and antimicrobial activity of the essential oils from New Caledonian Citrus macroptera and Citrus hystrix. Chem. Biodivers. 2010, 7, 871–877. [Google Scholar] [CrossRef] [PubMed]
- Hien, T.T.; Quyen, N.T.C.; Truc, T.T.; Quan, P.M. Evaluate the chemical composition of kaffir lime (Citrus hystrix) essential oil using the classical method. IOP Conf. Ser. Mater. Sci. Eng. 2020, 991, 012014. [Google Scholar] [CrossRef]
- Baccati, C.; Gibernau, M.; Paoli, M.; Ollitrault, P.; Tomi, F.; Luro, F. Chemical variability of peel and leaf essential oils in the citrus subgenus papeda (Swingle) and few relatives. Plants 2021, 10, 1117. [Google Scholar] [CrossRef]
- Norkaew, O.; Pitija, K.; Pripdeevech, P.; Sookwong, P.; Wongporncha, S. Supercritical fluid extraction and gas chromatographic-mass spectrometric analysis of terpenoids in fresh kaffir lime leaf oil. Chiang Mai J. Sci. 2013, 40, 240–247. [Google Scholar]
- Kordali, S.; Kotan, R.; Cakir, A. Screening of antifungal activities of 21 oxygenated monoterpenes in-vitro as plant disease control agents. Allelopathy J. 2007, 19, 373–392. [Google Scholar]
- Shepard, B.D. Sex differences in diabetes and kidney disease: Mechanisms and consequences. Am. J. Physiol. Renal Physiol. 2019, 317, F456–F462. [Google Scholar] [CrossRef]
- Luc, K.; Schramm-Luc, A.; Guzik, T.J.; Mikolajczyk, T.P. Oxidative stress and inflammatory markers in prediabetes and diabetes. J. Physiol. Pharmacol. 2019, 70, 809–824. [Google Scholar] [CrossRef]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. Measurement of functional properties and health promoting aspects-glucose retardation index of peel, pulp and peel fiber from Citrus hystrix and Citrus maxima. Bioact. Carbohydr. Diet. Fibre 2014, 4, 16–26. [Google Scholar] [CrossRef]
- Irawaty, W.; Ayucitra, A. Assessment on antioxidant and in vitro antidiabetes activities of different fractions of Citrus hystrix peel. Int. Food Res. J. 2018, 25, 2467–2477. [Google Scholar]
- Abirami, A.; Nagarani, G.; Siddhuraju, P. In vitro antioxidant, anti-diabetic, cholinesterase and tyrosinase inhibitory potential of fresh juice from Citrus hystrix and C. maxima fruits. Food Sci. Hum. Wellness 2014, 3, 16–25. [Google Scholar] [CrossRef] [Green Version]
- Setyabudi, C.; Tanda, S.; Santosa, W.I.; Soetaredjo, F.E. Studi in vitro ekstrak kulit jeruk purut untuk aplikasi terapi diabetes mellitus. J. Ilm. Widya Tek. 2015, 14, 15–19. [Google Scholar]
- Rekasih, M.; Muhandri, T.; Safithri, M.; Wijaya, C.H. Antihyperglycemic Activity of Java tea-based functional drink loaded chitosan nanoparticle in streptozotocin-induced diabetic rats. Hayati J. Biosci. 2021, 28, 212–222. [Google Scholar] [CrossRef]
- Janiak, M.C. Of starch and spit. Elife 2019, 8, e47523. [Google Scholar] [CrossRef]
- Saleh, M.S.M.; Jalil, J.; Mustafa, N.H.; Ramli, F.F.; Asmadi, A.Y.; Kamisah, Y. UPLC-MS-based metabolomics profiling for α-glucosidase inhibiting property of Parkia speciosa pods. Life 2021, 11, 78. [Google Scholar] [CrossRef]
- Crespo, M.E.; Gálvez, J.; Cruz, T.; Ocete, M.A.; Zarzuelo, A. Anti-inflammatory activity of diosmin and hesperidin in rat colitis induced by TNBS. Planta Med. 1999, 65, 651–653. [Google Scholar] [CrossRef]
- Siti, H.N.; Jalil, J.; Asmadi, A.Y.; Kamisah, Y. Rutin modulates MAPK Pathway differently from quercetin in angiotensin ii-induced H9c2 cardiomyocyte hypertrophy. Int. J. Mol. Sci. 2021, 22, 5063. [Google Scholar] [CrossRef]
- Zhang, H.L.; Wu, X.Y.; Mi, J.; Peng, Y.J.; Wang, Z.G.; Liu, Y.; Wu, X.L.; Gao, Y. A new anti-inflammatory alkaloid from roots of Heracleum dissectum. Chem. Biodivers. 2017, 14, e1700184. [Google Scholar] [CrossRef] [PubMed]
- Koysu, P.; Genc, N.; Elmastas, M.; Aksit, H.; Erenler, R. Isolation, identification of secondary metabolites from Salvia absconditiflora and evaluation of their antioxidative properties. Nat. Prod. Res. 2019, 33, 3592–3595. [Google Scholar] [CrossRef] [PubMed]
- Tirumani, P.; Venu, S.; Sridhar, G.; Praveen Kumar, M.; Rajashekhar, A.V.; Naga Raju, T. Delaying of cataract through intervention of Hemidesmus indicus in STZ induced diabetic rats. Nat. Prod. Res. 2018, 32, 1295–1298. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.; Bodakhe, S.H. Biochemical evidence indicates the preventive effect of resveratrol and nicotinamide in the treatment of STZ-induced diabetic cataract. Curr. Eye Res. 2021, 46, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Jang, D.J.; Kim, S.T.; Oh, E.; Lee, K. Enhanced oral bioavailability and antiasthmatic efficacy of curcumin using redispersible dry emulsion. BioMed Mater. Eng. 2014, 24, 917–930. [Google Scholar] [CrossRef]
- Gumprecht, J.; Domek, M.; Lip, G.Y.H.; Shantsila, A. Invited review: Hypertension and atrial fibrillation: Epidemiology, pathophysiology, and implications for management. J. Hum. Hypertens. 2019, 33, 824–836. [Google Scholar] [CrossRef]
- Siti, H.N.; Kamisah, Y.; Kamsiah, J. The role of oxidative stress, antioxidants and vascular inflammation in cardiovascular disease (a review). Vascul. Pharmacol. 2015, 71, 40–56. [Google Scholar] [CrossRef]
- Khalid, N.M.; Babji, A.S. Antioxidative and antihypertensive activities of selected Malaysian ulam (salad), vegetables and herbs. J. Food Res. 2018, 7, 27–37. [Google Scholar] [CrossRef]
- Tejpal, S.; Wemyss, A.M.; Bastie, C.C.; Klein-Seetharaman, J. Lemon extract reduces angiotensin converting enzyme (ACE) Expression and activity and increases insulin sensitivity and lipolysis in mouse adipocytes. Nutrients 2020, 12, 2348. [Google Scholar] [CrossRef]
- Jaarin, K.; Mustafa, M.R.; Leong, X.F. The effects of heated vegetable oils on blood pressure in rats. Clinics 2011, 6, 2125–2132. [Google Scholar] [CrossRef] [Green Version]
- Ng, C.Y.; Kamisah, Y.; Faizah, O.; Jaarin, K. The role of repeatedly heated soybean oil in the development of hypertension in rats: Association with vascular inflammation. Int. J. Exp. Pathol. 2012, 93, 377–387. [Google Scholar] [CrossRef] [PubMed]
- Suhaimi, N.H.; Zuhair, J.S.F.; Azlan, M.A.; Juliana, A.H.; Mustazil, M.N.M.; Zainalabidin, S.; Jaarin, K.; Mohamed, S.; Kamisah, Y. Addition of citrus leaf extract into frying oil prevents hypertension and improves vascular reactivity in heated oil-fed rats. J. Food Nutr. Res. 2020, 8, 417–423. [Google Scholar] [CrossRef]
- Sukalingam, K.; Jaarin, K.; Saad, Q.H.M.; Mohamed, S.; Othman, F. Consumption of ADD-X and repeatedly heated palm oil on the blood pressure and oxidative stress markers in ovarectemized rats. Int. J. Pharmacol. 2016, 12, 514–522. [Google Scholar] [CrossRef] [Green Version]
- Adam, S.K.; Sulaiman, N.A.; Mat Top, A.G.; Jaarin, K. Heating reduces vitamin E content in palm and soy oils. J. Biochem. Mol. Biol. 2007, 5, 76–79. [Google Scholar]
- Siti, H.N.; Kamisah, Y.; Mohamed, S.; Jaarin, K. Effects of citrus leaf extract on aortic vascular reactivity in hypertensive rats fed repeatedly heated vegetable oil. Appl. Physiol. Nutr. Metab. 2019, 44, 373–380. [Google Scholar] [CrossRef]
- Sukalingam, K.; Jaarin, K.; Saad, Q.H.M.; Mohamed, S.; Othman, F. Effect of Rutacea plant extract (ADD-X) on inflammatory biomarkers, cardiac ldl, troponin t and histological changes in ovariectomized rats fed with heated palm oil. Int. J. Toxicol. Pharmacol. Res. 2016, 8, 223–231. [Google Scholar]
- Li, C.J.; Barkath, A.A.; Abdullah, M.Z.; Lingkan, N.; Ismail, N.H.M.; Pauzi, S.H.M.; Kamisah, Y.; Qodriyah, H.M.S.; Jaarin, K.; Mohamed, S.; et al. The effects of citrus leaf extract on renal oxidative stress, renal function and histological changes in rats fed with heated palm oil. Biomed. Pharmacol. J. 2019, 12, 363–373. [Google Scholar]
- Imai, T.; Morita, T.; Shindo, T.; Nagai, R.; Yazaki, Y.; Kurihara, H.; Suematsu, M.; Katayama, S. Vascular smooth muscle cell-directed overexpression of heme oxygenase-1 elevates blood pressure through attenuation of nitric oxide induced vasodilation in mice. Circ. Res. 2001, 89, 55–62. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Yip, S.; Wang, Z.; Wang, W.; Xing, D.; Du, L. Brazilein induced contraction of rat arterial smooth muscle involves activation of Ca2+ entry and ROK, ERK pathways. Eur. J. Pharmacol. 2008, 580, 366–371. [Google Scholar] [CrossRef]
- Lüneburg, N.; Harbaum, L.; Hennigs, J.K. The endothelial ADMA/NO pathway in hypoxia-related chronic respiratory diseases. Biomed Res Int. 2014, 2014, 501612. [Google Scholar] [CrossRef]
- Muñoz-Durango, N.; Fuentes, C.A.; Castillo, A.E.; González-Gómez, L.M.; Vecchiola, A.; Fardella, C.E.; Kalergis, A.M. Role of the renin-angiotensin-aldosterone system beyond blood pressure regulation: Molecular and cellular mechanisms involved in end-organ damage during arterial hypertension. Int. J. Mol. Sci. 2016, 17, 797. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gui, J.S.; Mustafa, N.; Jalil, J.; Jubri, Z.; Kamisah, Y. Modulation of NOX4 and MAPK signalling pathways by Parkia speciosa empty pods in H9c2 cardiomyocytes exposed to H2O2. Indian J. Pharm. Sci. 2019, 81, 1029–1035. [Google Scholar] [CrossRef]
- Gao, L.; Yuan, P.; Zhang, Q.; Fu, Y.; Hou, Y.; Wei, Y.; Zheng, X.; Feng, W. Taxifolin improves disorders of glucose metabolism and water-salt metabolism in kidney via PI3K/AKT signaling pathway in metabolic syndrome rats. Life Sci. 2020, 263, 118713. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Kurth, T.; Zheleznova, N.N.; Yang, C.; Cowley, A.W., Jr. NOX4/H(2)O(2)/mTORC1 pathway in salt-induced hypertension and kidney injury. Hypertension 2020, 76, 133–143. [Google Scholar] [CrossRef] [PubMed]
- Karr, S. Epidemiology and management of hyperlipidemia. Am. J. Manag. Care 2017, 23, S139–S148. [Google Scholar] [PubMed]
- Deliara, H.; Kartikadewi, A.; Nugraheni, D.M. Kaffir lime peel ethanol extract (Citrus hystrix) as a cholesterol reducing agent: In vivo study. Med. Art. 2020, 2, 1–9. [Google Scholar]
- State-Ease. Least Significant Difference (LSD). Available online: https://www.statease.com/docs/v11/contents/model-graphs/least-significant-difference-bars/ (accessed on 21 December 2021).
- Zajmi, A.; Ramli, L.H.; Al-Abd, N.; Othman, Z. Anti-hyperlipidemia activity of key lime (Citrus aurantifolia) and kaffir lime (Citrus hystrix) peel extracts on high fat diet induced hyperlipidemic rats. Solid State Tech. 2020, 63, 2925–2932. [Google Scholar]
- Watanabe, D.; Kerakawati, R.; Morita, T.; Nakamura, T.; Ueno, K.; Kumamoto, T.; Nakanishi, W.; Ishikawa, T.; Uzawa, J.; Seki, H.; et al. Isolation of β-sitosterol and digalactopyranosyl-diacylglyceride from Citrus hystrix, a Thai traditional herb, as pancreatic lipase inhibitors. Heterocycles 2009, 78, 1497–1505. [Google Scholar]
- Tirawanchai, N.; Kengkoom, K.; Isarangkul, D.; Burana-Osot, J.; Kanjanapruthipong, T.; Chantip, S.; Phattanawasin, P.; Sotanaphun, U.; Ampawong, S. A combination extract of kaffir lime, galangal, and lemongrass maintains blood lipid profiles, hepatocytes, and liver mitochondria in rats with nonalcoholic steatohepatitis. Biomed. Pharmacother. 2020, 124, 109843. [Google Scholar] [CrossRef]
- de la Garza, A.L.; Milagro, F.I.; Boque, N.; Campión, J.; Martínez, J.A. Natural inhibitors of pancreatic lipase as new players in obesity treatment. Planta Med. 2011, 77, 773–785. [Google Scholar] [CrossRef] [Green Version]
- Piyachaturawat, P.; Glinsukon, T.; Chanjarunee, A. Antifertility effect of Citrus hystrix DC. J. Ethnopharmacol. 1985, 13, 105–110. [Google Scholar] [CrossRef]
- Koh, D.; Ong, C.N. Phytophotodermatitis due to the application of citrus hystrix as a folk remedy. Br. J. Dermatol. 1999, 140, 737–738. [Google Scholar] [CrossRef] [PubMed]

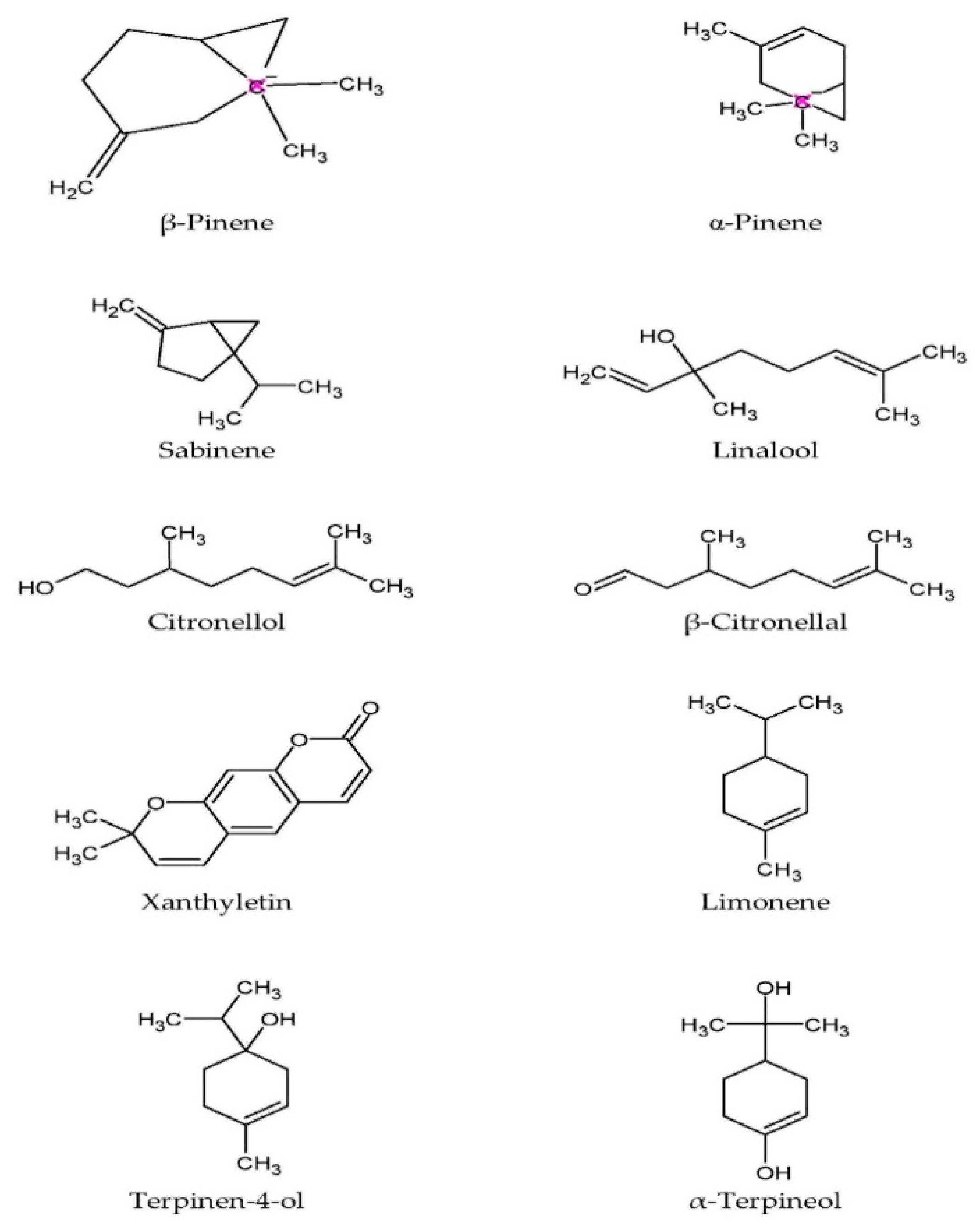
| Plant Parts | Medicinal Uses | Reference |
|---|---|---|
| Rinds | Worm infestation Headache | [10] |
| Fruit juice | Cancer | [16,17] |
| Skin diseases | ||
| Dandruff | ||
| Hair loss | ||
| Fruit juice and rinds | An ingredient in jamu for promoting physical and general health | [10] |
| Fruits | Stomachache | [11] |
| Leaves and fruits | Steam bath Aphrodisiac Diabetes mellitus Fever Rheumatism | [13,14] |
| Flu Hypertension Diarrhea Heart disease Dizziness Indigestion | ||
| Leaves | Stomachache | [10,18] |
| To maintain healthy teeth and gums Scurvy | ||
| Roots and leaves | Hypertension | [15] |
| Plant Part | Type | Phytochemical | Potential Activity * | Reference |
|---|---|---|---|---|
| Leaves | Monoterpenoids | Citronellal | Antimicrobial | [20] |
| Linalool oxide | [21] | |||
| Citronellol | ||||
| Terpeneol | ||||
| Geraniol | ||||
| α-Thujene | ||||
| α-Cubene | ||||
| β-Cubene | ||||
| Diterpene | Phytol | Antileukemic | [22] | |
| Triterpene | Lupeol | Antileukemic | [21,22,23] | |
| Agrostophillinol | Anti-inflammatory | [18] | ||
| Squalene | [21] | |||
| Tetranortriterpenoids (limonins) | Limonexin | [24] | ||
| Limonexic acid | ||||
| Limonin | Antibacterial | |||
| Sesquiterpenes | Spathulenol | [21] | ||
| Nerolidol | ||||
| Germacrene | ||||
| Caryophyllene oxide | ||||
| Longipinenepoxide | ||||
| α-Cedrane | ||||
| Globulol | ||||
| Phytosterols | Stigmasterol | |||
| Sitosterol | ||||
| Lanost-7-en-3-one | ||||
| Glyceroglycolipids | l,2-di-O-a-linolenoyl-3-O-galactopyranosyl-sn-glycerol l-O-a-Linolenoyl-2-O-palmitoyl-3-O-galactopyranosyl-sn-glycerol | Viral tumor- promoting inhibitors | [25] | |
| Phenol | α-Tocopherol | Antioxidant | [21,26] | |
| Flavonoids | Cyanidin | Antioxidant | [27,28] | |
| Myricetin | ||||
| Peonidin | ||||
| Quercetin | ||||
| Luteolin | ||||
| Hesperetin | ||||
| Apigenin | ||||
| Isorhamnetin | ||||
| Hesperidin | [7] | |||
| Diosmin | ||||
| Apiin | ||||
| Saponarin | ||||
| Apigetrin | ||||
| Rutin | ||||
| Eriodictyol rutinoside | [28] | |||
| Eriodictyol neohesperidoside | ||||
| Phloretin | ||||
| Diosmetin | ||||
| Roots | Coumarins | Hystrixarin | [7,24,29] | |
| Hopeyhopin | ||||
| Peroxytamarin | ||||
| Tamarin | Antibacterial | |||
| Trans-decursidinol | ||||
| Suberosin | ||||
| Scoparone | ||||
| Scopoletin | ||||
| Umbelliferone | ||||
| cis-Khellactone | ||||
| Oxypeucedanin hydrate | ||||
| cis-Casegravol | ||||
| Citrusarin A-B | ||||
| Seselin | ||||
| 5-Hydroxy-seselin | ||||
| Dipetalolactone | ||||
| Xanthyletin | ||||
| Osthenol | ||||
| Isoimperatorin | ||||
| Xanthotoxol | ||||
| Benzenoid | Hystroxene-I | |||
| Quinolinone | Hystrolinone 1-Methyl-2-n-nonyl-4-quinolinone | |||
| Cinnamyl alcohol | Etrogol | |||
| Alkyl phenylketones | Xanthoxylin (Brevifolin) | Antibacterial | ||
| Flavonoids | Crenulatin | |||
| Yukovanol | Antibacterial | |||
| P-hydroxy-benzaldehyde | ||||
| Acridone | Baiyumine-A | |||
| 5-Hydroxy-noracronycine | Anti-HIV-1 protease Antioxidant | |||
| Citracridone-I | ||||
| Citpressine-I | ||||
| Citramine | ||||
| Benzoic acid | Valencic acid | |||
| p-Hydroxybenzoic acid | ||||
| Lignan | Syringaresinol | Antioxidant Antibacterial | ||
| Suberin | 20,30-dihydroxydihydrosuberin | |||
| Phenolic | Vanillic acid | |||
| Tyrosol | ||||
| Rinds | Coumarins | Citrusosides B–D | Anticholinesterase | [30] |
| Oxypeucedanin methanolate | [31] | |||
| Oxypeucedanin hydrate | ||||
| Isoimperatorin | ||||
| Oxypeucedanin | ||||
| Pabulenol | ||||
| Bergamottin | ||||
| Hydroxy-7′-methoxy-bergamottin | ||||
| 6′,7′-dihydroxy-bergamottin | ||||
| 7-hydroxycoumarin | ||||
| Umbelliferone | α-Amylase inhibitor | [32] | ||
| Bergamottin | [33] | |||
| Oxypeucedanin | ||||
| Citrusoside E-G | ||||
| Citrusoside H | Anticholinesterase | |||
| Citrusoside I-O | ||||
| Bergaptol | Antioxidant Anticholinesterase Antitumor α-Amylase inhibitor | [32] | ||
| Isoimperatorin | Antioxidant Antitumor α-Amylase inhibitor | |||
| Pectin | Anhydrouronic acid (85%) | [34] | ||
| Monoterpenes | β-pinene Limonene Sabinene | Antimicrobial | [20] | |
| Tetranor-triterpenoids | Limonin | Metal chelator α-Amylase inhibitor | [32] | |
| Acridone | Citracridone I | Antioxidant Metal chelator Antitumor α-Amylase inhibitor | ||
| Phytosterol | Daucosterol | Antioxidant Antitumor α-Amylase inhibitor | ||
| Stigmasterol | Antioxidant Metal chelator Antitumor α-Amylase inhibitor | |||
| Flavonoids | Trihydroxypyranoflavone | α-Amylase inhibitor Anticholinesterase Antitumor | ||
| Dimethyl-6-prenylpyranoflavone | Anticholinesterase Antitumor α-Amylase inhibitor | |||
| Tangeretin | Antioxidant Antitumor α-Amylase inhibitor | |||
| Nobiletin | Antioxidant Metal chelator Antitumor α-Amylase inhibitor | |||
| Tetramethoxyflavone | Antioxidant Metal chelator Antitumor α-Amylase inhibitor | |||
| Natsudaidain | Antioxidant Metal chelator | |||
| Quercetin | [19] | |||
| Hesperidin | ||||
| Hesperitin | [33] | |||
| Fruits | Coumarins | Bergamottin | Nitric oxide synthesis inhibitor, antitumor | [25,31] |
| Oxypeucedanin | Nitric oxide synthesis inhibitor | |||
| 5-[(6′,7′-Dihydroxy-3′,7′-dimethyl-2-octenyl)oxy]psoralen | ||||
| Twig Essential oil | Monoterpenes | β-citronellal | [35] | |
| 4-Terpeneol | ||||
| L-linalool | ||||
| β-Citronellol | ||||
| Citronelyl acetate | ||||
| Sabinene | ||||
| β-Pinene | ||||
| β-Micrene | ||||
| Tran-β-ocimene | ||||
| (-)-Isopulegol | ||||
| Cis-Linalol oxide | ||||
| Sesquiterpenes | Trans-β-caryopilene | |||
| Nerolidol | ||||
| Leaf essential oil | Monoterpenes | α-Pinene | [36,37,38,39] | |
| Camphene | ||||
| β-Pinene | ||||
| β-Myrcene | ||||
| α-Phellandrene | ||||
| β-Phellandrene | ||||
| β-Car-3-ene | ||||
| p-Cymene | ||||
| Limonene | ||||
| 1,8-Cineole | ||||
| p-Mentha-2,4(8)-diene | ||||
| Linalool | ||||
| endo-Fenchol | ||||
| cis-p-Menth-2-en-ol | ||||
| Sabinene | ||||
| α-Terpinene | ||||
| (E)-β-Ocimene | ||||
| γ-Terpinene | ||||
| Terpinolene | ||||
| trans-p-Menth-2-en-ol | ||||
| p-Menth-8-en-3-ol | ||||
| Citronellal | ||||
| Geranial | ||||
| Isopulegol | ||||
| Isoneopulegol | ||||
| Borneol | ||||
| Terpinen-4-ol | ||||
| p-Cymen-8-ol | ||||
| Geranyl acetate | ||||
| α-Terpineol | ||||
| Myrtenol | ||||
| trans-Piperitol | ||||
| Citronellol | ||||
| Geraniol | ||||
| Bornyl acetate | ||||
| Carvacrol | ||||
| Citronellyl acetate | ||||
| Neryl acetate | ||||
| trans-p-Menth-6-ene-2,8-diol | ||||
| Sesquiterpenes | α-Copaene | |||
| cis-Cadina-1,4-diene | ||||
| Hedycaryol | ||||
| Caryophyllene oxide | ||||
| Cubenol | ||||
| β-Eudesmol | ||||
| (E)-β-Caryophyllene | ||||
| trans-α-Bergamotene | ||||
| α-Humulene | ||||
| Germacrene D | ||||
| Bicyclogermacrene | ||||
| (E,E)-α-Farnesene | ||||
| β-Bisabolene | ||||
| δ-Cadinene | ||||
| (E)-Nerolidol | ||||
| Spathulenol | ||||
| Globulol | ||||
| Rind essential oil | Monoterpenes | α-Thujene | [38] | |
| α-Pinene | ||||
| Camphene | ||||
| Sabinene | ||||
| β-Pinene | ||||
| Myrcene | ||||
| p-Cymene | ||||
| β-Phellandrene | ||||
| Limonene | ||||
| trans-Linalool oxide | ||||
| Linalool | ||||
| Citronellal | ||||
| Terpinen-4-ol | ||||
| α-Terpineol | ||||
| trans-Carveol | ||||
| Citronellol | ||||
| Geranyl acetate | ||||
| Sesquiterpenes | α-Copaene | |||
| β-Elemene | ||||
| δ-Cadinene | ||||
| Ester | Hexyl hexanoate |
| Plant Extract | Model | Outcomes | Study |
|---|---|---|---|
| Rinds and pulps (Powdered form) | In vitro | ↑ glucose adsorption capacity ↓ glucose dialysis retardation index ↓ starch digestibility | [43] |
| Rinds (Ethyl acetate fraction and water residue) | In vitro | ↓ α-amylase activity ↓ α-glucosidase activity ↓ Starch digestibility | [44] |
| Fresh fruit juice | In vitro | ↓ α-amylase activity ↓ α-glucosidase activity Almost comparable to acarbose | [45] |
| Rinds (Ethanol) | In vitro | ↓ α-amylase activity | [46] |
| Leaves (Ethanol) | In vivo STZ-induced diabetes in rats | Extract at 150 and 300 mg/kg bw in drinking water for 8 weeks: ↓ fasting blood glucose ↓ cataract incidence ↓ oxidative stress and inflammation ↓ vascular leakage | [7] |
| Fruit juice in combination with other extracts (functional drink) | In vivo STZ-induced diabetes in rats | Functional drink (18.2 mL/kg bw) for 14 days: ↓ fasting blood glucose ↑ pancreatic β-cell viability ↑ Langerhans islet viability | [47] |
| Plant Extract | Model | Mode of Administration | Outcomes | Study |
|---|---|---|---|---|
| Leaves (Aqueous) | In vitro | - | Good ACE-inhibiting activity (~90%) | [59] |
| Leaves (Ethanol) | OVX rats fed a high-fat diet for 6 months | Addition of extract into the frying oil (1%) | Both in 5HPO and 10HPO groups: ↓ PV in oil, ↓ BP, ↓ serum oxidative product, ↑ serum antioxidant enzymes | [64] |
| Leaves (Ethanol) | Heated oil diet-induced in rats for 16 weeks | Addition of extract into the frying oil (1%) | Both in 5HPO and 10HPO groups: ↓ PV and ↑ TPC in oil ↓ BP, ↑ plasma NO, ↓ vasoconstriction response to PE ↑ vasorelaxation response to ACh and SNP, ↓ TI/TM. ↓ VCAM | [63] |
| Leaves (Ethanol) | Heated oil diet-induced in rats for 16 weeks | Dietary (0.15%) | In 5HPO group but not in 10HPO group: ↓ BP, ↓ plasma TBARS, ↓ serum ACE, ↓ plasma TXB2, ↓ IMA, ↓ IMT, ↓ CWT In both 5HPO and 10HPO groups: ↑ serum HO-1, ↔ plasma PGI2 | [8] |
| Leaves (Ethanol) | Heated oil diet-induced in rats for 16 weeks | Dietary (0.15%) | In 5HPO group but not in 10HPO group: ↑ plasma NO, ↓ vasoconstriction response to PE ↔ vasorelaxation response to ACh and SNP | [66] |
| Leaves (Ethanol) | OVX rats fed a high-fat diet for 6 months | Addition of extract into the frying oil (1%) | In both 5HPO and 10HPO groups: ↓ serum CRP, ↓ serum TNF-α, ↓ cardiac troponin, ↓ cardiac LDH | [67] |
| Leaves (Ethanol) | Heated oil diet-induced in rats for 16 weeks | Dietary (0.15%) | Both 5HPO and 10HPO groups: ↓ renal TBARS, ↓ renal NOX, ↑ renal HO, ↓ serum creatinine in 5HPO group | [68] |
| Plant Extract | Model | Mode of Administration | Outcomes | Study |
|---|---|---|---|---|
| Rinds (Ethanol) | A high-fat diet- induced hyperlipidemia in rats | Oral (35, 70, and 140 mg/kg bw) for 3 weeks | No significant change was noted in serum LDL level | [9] |
| Rinds (Ethanol) | A high-fat diet- induced hyperlipidemia in rats | Oral (35, 70, and 140 mg/kg bw) for 3 weeks | Extract at 70 and 140 mg/kg: ↓ serum TC | [77] |
| Rinds (Methanol) | A high-fat diet- induced hyperlipidemia in rats | Oral (500 mg/kg) for 14 days | ↓ serum TC ↓ serum TG ↑ serum HDL ↓ serum LDL | [79] |
| Leaves (Ethanol) | OVX rats fed a high-fat diet for 6 months | Addition of extract into the frying oil (1%) | ↓ cardiac free fatty acid ↓ cardiac TG | [67] |
| Leaves (Ethanol) | In vitro | - | Pancreatic lipase activity inhibition: Extract: 58%. Isolate β-sitosterol (100 μg/mL): 79% Isolate 3-[O-α-galactopyranosyl-(1”→ 6’)-O-β- galactopyranosyl]-1-O-linolenyl-2-O-palmitylglyceride (100 μg/mL): 88% | [80] |
| Leaves in combination with galangal rhizomes and lemongrass extracts (Aqueous) | Hyper- cholesterolemic diet (3% cholesterol) in rats | Oral mixed extract at 400 mg/kg/day for 28 days | Compared to simvastatin-treated group: ↔ serum lipid profile ↔ centrilobular steatotis ↔ periportal steatosis ↔ hepatitis | [81] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Siti, H.N.; Mohamed, S.; Kamisah, Y. Potential Therapeutic Effects of Citrus hystrix DC and Its Bioactive Compounds on Metabolic Disorders. Pharmaceuticals 2022, 15, 167. https://doi.org/10.3390/ph15020167
Siti HN, Mohamed S, Kamisah Y. Potential Therapeutic Effects of Citrus hystrix DC and Its Bioactive Compounds on Metabolic Disorders. Pharmaceuticals. 2022; 15(2):167. https://doi.org/10.3390/ph15020167
Chicago/Turabian StyleSiti, Hawa Nordin, Suhaila Mohamed, and Yusof Kamisah. 2022. "Potential Therapeutic Effects of Citrus hystrix DC and Its Bioactive Compounds on Metabolic Disorders" Pharmaceuticals 15, no. 2: 167. https://doi.org/10.3390/ph15020167
APA StyleSiti, H. N., Mohamed, S., & Kamisah, Y. (2022). Potential Therapeutic Effects of Citrus hystrix DC and Its Bioactive Compounds on Metabolic Disorders. Pharmaceuticals, 15(2), 167. https://doi.org/10.3390/ph15020167






