Immunomodulatory Effect of Polysaccharides from the Mushroom-Forming Basidiomycete Gymnopilus imperialis (Agaricomycetes, Basidiomycota)
Abstract
1. Introduction
2. Results and Discussion
2.1. Taxonomy
2.2. Characterization of the Polysaccharide Fractions
2.3. Immunomodulatory Potential
3. Materials and Methods
3.1. Mushroom Collection and Taxonomy
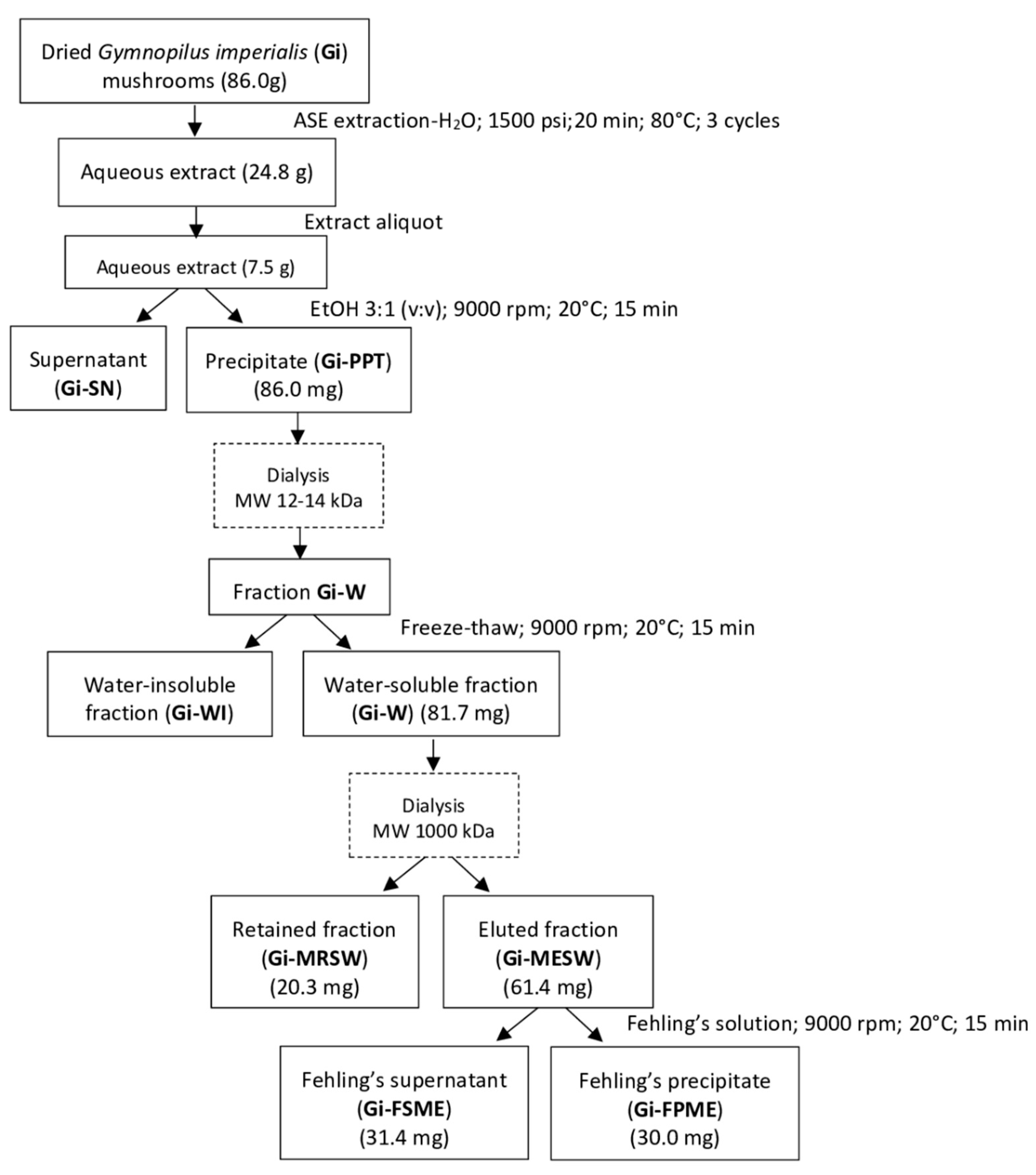
3.2. Extraction of the Compounds
3.3. Fractionation of the Compounds
3.4. Monosaccharide Composition
3.5. NMR Measurement
3.6. Cell Line and Culture Conditions
3.7. Measurement of Nitric Oxide (NO) Production
3.8. Transcript Level Expression of Target Genes
3.9. Statistical Analysis
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Guzmán-dávalos, A.L.; Mueller, G.M.; Cifuentes, J.; Andrew, N.; Santerre, A.; Guzman-divalos, L.; Mueller, G.M. Mycological Society of America Traditional Infrageneric Classification of Gymnopilus Is Not Supported by Ribosomal DNA Sequence Data. Mycologia 2003, 95, 1204–1214. [Google Scholar] [CrossRef] [PubMed]
- He, M.Q.; Zhao, R.L.; Hyde, K.D.; Begerow, D.; Kemler, M.; Yurkov, A.; McKenzie, E.H.C.; Raspé, O.; Kakishima, M.; Sánchez-Ramírez, S.; et al. Notes, Outline and Divergence Times of Basidiomycota. Fungal Divers. 2019, 99, 105–367. [Google Scholar]
- Pegler, D.N. Agaric Flora of the Lesser Antilles. Kew Bull. Add 1983, 9, 1–668. [Google Scholar]
- Buck, R.B. Psychodelic Effect of Pholiota spectabilis. N. Engl. J. Med. 1967, 321, 1306–1311. [Google Scholar]
- Romagnesi, H. Champignons Toxiques Au Japon. Bull. Soc. Mye. Fr. 1964, 80, 4–5. [Google Scholar]
- Isaka, M.; Sappan, M.; Suvannakad, R.; Boonpratuang, T.; Thummarukcharoen, T. Drimane Sesquiterpenoids from Cultures of the Basidiomycete Gymnopilus sp. Phytochem. Lett. 2020, 35, 141–146. [Google Scholar] [CrossRef]
- Thorn, R.G.; Malloch, D.W.; Saar, I.; Lamoureux, Y.; Nagasawa, E.; Redhead, S.A.; Margaritescu, S.; Moncalvo, J.M. New Species in the Gymnopilus junonius Group (Basidiomycota: Agaricales). Botany 2020, 98, 293–315. [Google Scholar] [CrossRef]
- Caldas, L.A.; Soares, D.M.M.; Menolli, N.; Stevani, C.V.; Sartorelli, P. Metabolomics of the Wild Mushroom Gymnopilus imperialis (Agaricomycetes, Basidiomycota) by UHPLC-HRMS/MS Analysis and Molecular Network. Fungal Biol. 2022, 126, 132–138. [Google Scholar] [CrossRef]
- Kayano, T.; Kitamura, N.; Miyazaki, S.; Ichiyanagi, T.; Shimomura, N.; Shibuya, I.; Aimi, T. Gymnopilins, a Product of a Hallucinogenic Mushroom, Inhibit the Nicotinic Acetylcholine Receptor. Toxicon 2014, 81, 23–31. [Google Scholar] [CrossRef]
- Tanaka, M.S.; Hashimoto, K.; Okuno, T.; Shirahama, H. Neurotoxic oligoisoprenoids of the hallucinogenic mushroom, Gymnopilus imperialis. Phytoch 1993, 34, 661–664. [Google Scholar] [CrossRef]
- Wasser, S.P. Medicinal Mushroom Science: Current Perspectives, Advances, Evidences, and Challenges. Biomed. J. 2014, 37, 345–356. [Google Scholar] [CrossRef] [PubMed]
- Boukes, G.J.; Koekemoer, T.C.; van de Venter, M.; Govender, S. Cytotoxicity of Thirteen South African Macrofungal Species against Five Cancer Cell Lines. S. Afr. J. Bot. 2017, 113, 62–67. [Google Scholar] [CrossRef]
- Hatfield, G.M.; Brady, L.R. Occurrence of Bis-noryangonin in Gymnopilus spectabilis. J. Pharm. Sci. 1969, 58, 1298–1299. [Google Scholar] [CrossRef] [PubMed]
- Lee, I.-K.; Cho, S.-M.; Seok, S.-J.; Yun, B.-S. Chemical Constituents of Gymnopilus spectabilis and Their Antioxidant Activity. Mycobiology 2008, 36, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.H.; Choi, S.U.; Lee, K.R. Gymnopilin K: A New Cytotoxic Gymnopilin from Gymnopilus spectabilis. J. Antibiot. 2012, 65, 135–137. [Google Scholar] [CrossRef]
- Brady, L.R.; Benedict, R.G. Occurrence of Bisnoryangonin in Pholiota squarroso-adiposa. J. Pharm. Sci. 1972, 61, 318. [Google Scholar] [CrossRef]
- Singh, R.S.; Walia, A.K.; Kennedy, J.F. Mushroom Lectins in Biomedical Research and Development. Int. J. Biol. Macromol. 2020, 151, 1340–1350. [Google Scholar] [CrossRef]
- Alborés, S.; Mora, P.; Bustamante, M.J.; Cerdeiras, M.P.; Franco Fraguas, L. Purification and Applications of a Lectin from the Mushroom Gymnopilus Spectabilis. App. Biochem. Biotechnol. 2014, 172, 2081–2090. [Google Scholar] [CrossRef]
- Ismaya, W.T.; Tjandrawinata, R.R.; Rachmawati, H. 2020 Lectins from the Edible Mushoom Agaricus bisporus and Their Therapeutic Potentials. Molecules 2018, 388, 539–547. [Google Scholar]
- Wang, Q.; Wang, F.; Xu, Z.; Ding, Z. Bioactive Mushroom Polysaccharides: A Review on Monosaccharide Composition, Biosynthesis and Regulation. Molecules 2017, 22, 955. [Google Scholar] [CrossRef]
- Ruiz-Herrera, J.; Ortiz-Castellanos, L. Cell Wall Glucans of Fungi. A Review. Cell Surf. 2019, 5, 100022. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Cui, S.W.; Cheung, P.C.K.; Wang, Q. Antitumor Polysaccharides from Mushrooms: A Review on Their Isolation Process, Structural Characteristics and Antitumor Activity. Trends Food Sci. Technol. 2007, 18, 4–19. [Google Scholar] [CrossRef]
- Castro-Alves, V.C.; Gomes, D.; Menolli, N.; Sforça, M.L.; do Nascimento, J.R.O. Characterization and Immunomodulatory Effects of Glucans from Pleurotus Albidus, a Promising Species of Mushroom for Farming and Biomass Production. Int. J. Biol. Macromol. 2017, 95, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Maity, P.; Sen, I.K.; Chakraborty, I.; Mondal, S.; Bar, H.; Bhanja, S.K.; Mandal, S.; Maity, G.N. Biologically Active Polysaccharide from Edible Mushrooms: A Review. Int. J. Biol. Macromol. 2021, 172, 408–417. [Google Scholar] [CrossRef]
- Rout, D.; Mondal, S.; Chakraborty, I.; Pramanik, M.; Islam, S.S. Chemical Analysis of a New (1→3)-, (1→6)-Branched Glucan from an Edible Mushroom, Pleurotus florida. Carbohydr. Res. 2005, 340, 2533–2539. [Google Scholar] [CrossRef]
- Bogdan, C. Nitric Oxide Synthase in Innate and Adaptive Immunity: An Update. Trends Immunol. 2015, 36, 161–178. [Google Scholar] [CrossRef]
- Barad, A.; Mackedenski, S.; Li, W.M.; Li, X.J.; Lim, B.C.C.; Rashid, F.; Tackaberry, L.E.; Massicotte, H.B.; Egger, K.N.; Reimer, K.; et al. Anti-Proliferative Activity of a Purified Polysaccharide Isolated from the Basidiomycete Fungus Paxillus involutus. Carbohydr. Polym. 2018, 181, 923–930. [Google Scholar] [CrossRef]
- Harada, K. Accumulation of Aberrant CpG Hypermethylation by Helicobacter pylori Infection Promotes Development. Int. J. Oncol. 2009, 35, 547–557. [Google Scholar] [CrossRef]
- Lee, W.T.; Lee, T.H.; Cheng, C.H.; Chen, K.C.; Chen, Y.C.; Lin, C.W. Antroquinonol from Antrodia camphorata Suppresses Breast Tumor Migration/Invasion through Inhibiting ERK-AP-1- and AKT-NF-ΚB-Dependent MMP-9 and Epithelial-Mesenchymal Transition Expressions. Food Chem. Toxicol. 2015, 78, 33–41. [Google Scholar] [CrossRef]
- Carbonero, E.R.; Cordeiro, L.M.C.; Mellinger, C.G.; Sassaki, G.L.; Stocker-Wörgötter, E.; Gorin, P.A.J.; Iacomini, M. Galactomannans with Novel Structures from the Lichen Roccella decipiens Darb. Carbohydr. Res. 2005, 340, 1699–1705. [Google Scholar] [CrossRef]
- Kono, H.; Kondo, N.; Hirabayashi, K.; Ogata, M.; Totani, K.; Ikematsu, S.; Osada, M. Two-Dimensional NMR Data of a Water-Soluble β-(1→3, 1→6)-Glucan from Aureobasidium pullulans and Schizophyllan from Schizophyllum commune. Data Brief. 2017, 15, 382–388. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, R.S.; Biscaia, S.M.P.; Bellan, D.L.; Viana, S.R.F.; Di-Medeiros Leal, M.C.; Vasconcelos, A.F.D.; Lião, L.M.; Trindade, E.S.; Carbonero, E.R. Structure Elucidation of a Bioactive Fucomannogalactan from the Edible Mushroom Hypsizygus marmoreus. Carbohydr. Polym. 2019, 225, 115203. [Google Scholar] [CrossRef] [PubMed]
- Nyman, A.A.T.; Aachmann, F.L.; Rise, F.; Ballance, S.; Samuelsen, A.B.C. Structural Characterization of a Branched (1 → 6)-α-Mannan and β-Glucans Isolated from the Fruiting Bodies of Cantharellus cibarius. Carbohydr. Polym. 2016, 146, 197–207. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Cai, Z.; Mao, H.; Hu, P.; Li, X. Isolation and Structure Elucidation of Polysaccharides from Fruiting Bodies of Mushroom Coriolus versicolor and Evaluation of Their Immunomodulatory Effects. Int. J. Biol. Macromol. 2021, 166, 1387–1395. [Google Scholar] [CrossRef]
- Dong, Z.; Zhang, M.; Li, H.; Zhan, Q.; Lai, F.; Wu, H. Structural Characterization and Immunomodulatory Activity of a Novel Polysaccharide from Pueraria lobata (Willd.) Ohwi root. Int. J. Biol. Macromol. 2019, 64, 1921–1931. [Google Scholar] [CrossRef]
- Mishra, V.; Tomar, S.; Yadav, P.; Singh, M.P. Promising Anticancer Activity of Polysaccharides and Other Macromolecules Derived from Oyster Mushroom (Pleurotus sp.): An Updated Review. Int. J. Biol. Macromol. 2021, 182, 1628–1637. [Google Scholar] [CrossRef]
- Zhong, R.F.; Yang, J.J.; Geng, J.H.; Chen, J. Structural Characteristics, Anti-Proliferative and Immunomodulatory Activities of a Purified Polysaccharide from Lactarius volemus. Fr. Int. J. Biol. Macromol. 2021, 192, 967–977. [Google Scholar] [CrossRef]
- Rodrigues Barbosa, J.; dos Santos Freitas, M.M.; da Silva Martins, L.H.; de Carvalho, R.N. Polysaccharides of Mushroom Pleurotus spp.: New Extraction Techniques, Biological Activities and Development of New Technologies. Carbohydr. Polym. 2019, 229, 115550. [Google Scholar] [CrossRef]
- Zhao, S.; Gao, Q.; Rong, C.; Wang, S.; Zhao, Z.; Liu, Y.; Xu, J. Immunomodulatory Effects of Edible and Medicinal Mushrooms and Their Bioactive Immunoregulatory Products. J. Fungi 2020, 6, 269. [Google Scholar] [CrossRef]
- Inagawa, H.; Saitoh, F.; Iguchi, M.; Nishizawa, T.; Okutomi, T.; Morikawa, A.; Soma, G.-I.; Mizuno, D. Homeostasis as Regulated by Activated Macrophage. III. Protective Effect of LPSw (Lipopolysaccharide (LPS) of Wheat Flour) on Gastric Ulcer in Mice as Compared with Those of Other LPS from Various Sources. Chem. Pharm. Bull. 1992, 40, 998–1000. [Google Scholar] [CrossRef][Green Version]
- Balta, M.G.; Papathanasiou, E.; Christopoulos, P.F. Specialized Pro-Resolving Mediators as Potential Regulators of Inflammatory Macrophage Responses in COVID-19. Front. Immunol. 2021, 12, 632238. [Google Scholar] [CrossRef] [PubMed]
- Mantovani, A.; Locati, M. Tumor-Associated Macrophages as a Paradigm of Macrophage Plasticity, Diversity, and Polarization Lessons and Open Questions. Arterioscler. Thromb. Vasc. Biol. 2013, 33, 1478–1483. [Google Scholar] [CrossRef] [PubMed]
- Schildberger, A.; Rossmanith, E.; Eichhorn, T.; Strassl, K.; Weber, V. Monocytes, Peripheral Blood Mononuclear Cells, and THP-1 Cells Exhibit Different Cytokine Expression Patterns Following Stimulation with Lipopolysaccharide. Mediat. Inflamm. 2013, 2013, 697972. [Google Scholar] [CrossRef] [PubMed]
- Han, C.K.; Chiang, H.C.; Lin, C.Y.; Tang, C.H.; Lee, H.; Huang, D.D.; Zeng, Y.R.; Chuang, T.N.; Huang, Y.L. Comparison of Immunomodulatory and Anticancer Activities in Different Strains of Tremella fuciformis Berk. Am. J. Chin. Med. 2015, 43, 1637–1655. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Luo, M.; Liu, F.; Feng, X.; Ibrahim, S.A.; Cheng, L.; Huang, W. Effects of Freeze Drying and Hot-Air Drying on the Physicochemical Properties and Bioactivities of Polysaccharides from Lentinula edodes. Int. J. Biol. Macromol. 2020, 145, 476–483. [Google Scholar] [CrossRef]
- Mao, G.H.; Ren, Y.; Feng, W.W.; Li, Q.; Wu, H.Y.; Jin, D.; Zhao, T.; Xu, C.Q.; Yang, L.Q.; Wu, X.Y. Antitumor and Immunomodulatory Activity of a Water-Soluble Polysaccharide from Grifola frondosa. Carbohydr. Polym. 2015, 134, 406–412. [Google Scholar] [CrossRef]
- Zhang, Y.; Zeng, Y.; Men, Y.; Zhang, J.; Liu, H.; Sun, Y. Structural Characterization and Immunomodulatory Activity of Exopolysaccharides from Submerged Culture of Auricularia auricula-judae. Int. J. Biol. Macromol. 2018, 115, 978–984. [Google Scholar] [CrossRef]
- Campi, M.; Maubet, Y.; Grassi, E.; Niveiro, N.; Guzmán-dávalos, L.; Alto, D. First Contribution to the Genus Gymnopilus (Agaricales, Strophariaceae) in Paraguay. Rodriguésia 2021, 72, e00752019. [Google Scholar] [CrossRef]
- Hesler, L.R. North American Species of Gymnopilus. Mycol. Mem. 1969, 3, 112–113. [Google Scholar]
- Schoch, C.L.; Seifert, K.A.; Huhndorf, S.; Robert, V.; Spouge, J.L.; Levesque, C.A.; Chen, W.; Bolchacova, E.; Voigt, K.; Crous, P.W.; et al. Nuclear Ribosomal Internal Transcribed Spacer (ITS) Region as a Universal DNA Barcode Marker for Fungi. Proc. Nat. Acad. Sci. USA 2012, 109, 6241–6246. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J.W. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. PCR Protoc. Guide Methods Appl. 1990, 18, 315–322. [Google Scholar]
- Katoh, K.; Rozewicki, J.; Yamada, K.D. MAFFT Online Service: Multiple Sequence Alignment, Interactive Sequence Choice and Visualization. Brief. Bioinforma. 2018, 20, 1160–1166. [Google Scholar] [CrossRef] [PubMed]
- Varga, T.; Krizsán, K.; Földi, C.; Dima, B.; Sánchez-García, M.; Sánchez-Ramírez, S.; Szöllősi, G.J.; Szarkándi, J.G.; Papp, V.; Albert, L.; et al. Megaphylogeny Resolves Global Patterns of Mushroom Evolution. Nat. Ecol. Evol. 2019, 3, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Kearse, M.; Moir, R.; Wilson, A.; Stones-Havas, S.; Cheung, M.; Sturrock, S.; Buxton, S.; Cooper, A.; Markowitz, S.; Duran, C.; et al. Geneious Basic: An Integrated and Extendable Desktop Software Platform for the Organization and Analysis of Sequence Data. Bioinformatics 2012, 28, 1647–1649. [Google Scholar] [CrossRef] [PubMed]
- Stamatakis, A. RAxML Version 8: A Tool for Phylogenetic Analysis and Post-Analysis of Large Phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef] [PubMed]
- Palanisamy, M.; Aldars-García, L.; Gil-Ramírez, A.; Ruiz-Rodríguez, A.; Marín, F.R.; Reglero, G.; Soler-Rivas, C. Pressurized Water Extraction of β-Glucan Enriched Fractions with Bile Acids-Binding Capacities Obtained from Edible Mushrooms. Biotechnol. Prog. 2014, 30, 391–400. [Google Scholar] [CrossRef]
- Gorin, P.A.J.; Iacomini, M. Polysaccharides of the Lichens Cetraria islandica and Ramalina usnea. Carbohydr. Res. 1984, 128, 119–132. [Google Scholar] [CrossRef]
- Santos-Neves, J.C.; Pereira, M.I.; Carbonero, E.R.; Gracher, A.H.P.; Alquini, G.; Gorin, P.A.J.; Sassaki, G.L.; Iacomini, M. A Novel Branched Aβ-Glucan Isolated from the Basidiocarps of the Edible Mushroom Pleurotus florida. Carbohydr. Polym. 2008, 73, 309–314. [Google Scholar] [CrossRef]
- Wolfrom, M.L.; Thompson, A. Reduction with Sodium Borohydride. Methods Carbohydr. Chem. 1963, 2, 65–68. [Google Scholar]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wishnok, J.S.; Tannenbaum, S.R. Analysis of Nitrate, Nitrite, and [15N] Nitrate in Biological Fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Volman, J.J.; Johannes Helsper, P.F.G.; Wei, S.; Baars, J.J.P.; van Griensven, L.J.L.D.; Sonnenberg, A.S.M.; Mensink, R.P.; Plat, J. Effects of Mushroom-Derived Beta-Glucan-Rich Polysaccharide Extracts on Nitric Oxide Production by Bone Marrow-Derived Macrophages and Nuclear Factor-KappaB Transactivation in Caco-2 Reporter Cells: Can Effects Be Explained by Structure? Mol. Nutr. Food Res. 2010, 54, 268–276. [Google Scholar] [CrossRef] [PubMed]
- Vandesompele, J.; De Preter, K.; Pattyn, F.; Poppe, B.; Van Roy, N.; De Paepe, A.; Speleman, F. Accurate Normalization of Real-Time Quantitative RT-PCR Data by Geometric Averaging of Multiple Internal Control Genes. Genome Biol. 2002, 3, research0034.1. [Google Scholar] [CrossRef] [PubMed]
- Pfaffl, M.W. A New Mathematical Model for Relative Quantification in Real-Time RT-PCR. Nucleic Acids Res. 2001, 29, e54. [Google Scholar] [CrossRef] [PubMed]
- Ramakers, C.; Ruijter, J.M.; Lekanne Deprez, R.H.; Moorman, A.F.M. Assumption-Free Analysis of Quantitative Real-Time Polymerase Chain Reaction (PCR) Data. Neurosci. Lett. 2003, 339, 62–66. [Google Scholar] [CrossRef]



| Fractions | Monosaccharides (%) | |||
|---|---|---|---|---|
| Fucose | Mannose | Galactose | Glucose | |
| Gi-MRSW | - | 14.1 | 21.9 | 64.0 |
| Gi-PFME | 7.1 | 30.7 | 58.8 | 3.4 |
| Gi-SFME | - | 22.8 | 11.4 | 65.8 |
| Gene | Sequence | Reference |
|---|---|---|
| IL6 | F 5′-CTGCAAGAGACTTCCATCCAG -3′ | NM_031168.2 |
| R 5′-AGTGGTATAGACAGGTCTGTTGG -3′ | ||
| IL10 | F 5′-CTTACTGACTGGCATGAGGATCA -3′ | NM_010548.2 |
| R 5′-GCAGCTCTAGGAGCATGTGG -3′ | ||
| iNOS | F 5′-GTTCTCAGCCCAACAATACAAGA -3′ | NM_010927.4 |
| R 5′-GTGGACGGGTCGATGTCAC -3′ | ||
| TNFα | F 5′-CTGAACTTCGGGGTGATCGG -3′ | NM_013693.3 |
| R 5′-GGCTTGTCACTCGAATTTTGAGA -3′ | ||
| GAPDH | F 5′-AACGACCCCTTCATTGAC -3′ | NM_001289726.1 |
| R 5′-TCCACGACATACTCAGCA -3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Caldas, L.A.; Santos, P.D.; Carbonero, E.R.; Ionta, M.; Miyazawa, M.; Caixeta, E.S.; Fregnan, A.M.; Nóbrega, B.B.; Di Medeiros, M.C.B.; Menolli, N., Jr.; et al. Immunomodulatory Effect of Polysaccharides from the Mushroom-Forming Basidiomycete Gymnopilus imperialis (Agaricomycetes, Basidiomycota). Pharmaceuticals 2022, 15, 1179. https://doi.org/10.3390/ph15101179
Caldas LA, Santos PD, Carbonero ER, Ionta M, Miyazawa M, Caixeta ES, Fregnan AM, Nóbrega BB, Di Medeiros MCB, Menolli N Jr., et al. Immunomodulatory Effect of Polysaccharides from the Mushroom-Forming Basidiomycete Gymnopilus imperialis (Agaricomycetes, Basidiomycota). Pharmaceuticals. 2022; 15(10):1179. https://doi.org/10.3390/ph15101179
Chicago/Turabian StyleCaldas, Lhaís A., Patricia D. Santos, Elaine R. Carbonero, Marisa Ionta, Marta Miyazawa, Ester S. Caixeta, Antonio M. Fregnan, Bianca Barros Nóbrega, Maria Carolina B. Di Medeiros, Nelson Menolli, Jr., and et al. 2022. "Immunomodulatory Effect of Polysaccharides from the Mushroom-Forming Basidiomycete Gymnopilus imperialis (Agaricomycetes, Basidiomycota)" Pharmaceuticals 15, no. 10: 1179. https://doi.org/10.3390/ph15101179
APA StyleCaldas, L. A., Santos, P. D., Carbonero, E. R., Ionta, M., Miyazawa, M., Caixeta, E. S., Fregnan, A. M., Nóbrega, B. B., Di Medeiros, M. C. B., Menolli, N., Jr., Soares, D. M. M., Stevani, C. V., & Sartorelli, P. (2022). Immunomodulatory Effect of Polysaccharides from the Mushroom-Forming Basidiomycete Gymnopilus imperialis (Agaricomycetes, Basidiomycota). Pharmaceuticals, 15(10), 1179. https://doi.org/10.3390/ph15101179







