Structure, Function, and Pharmaceutical Ligands of 5-Hydroxytryptamine 2B Receptor
Abstract
1. Introduction
2. Function
2.1. Cardiovascular System
2.1.1. Cardiomyopathy
2.1.2. VHD
2.1.3. PAH
2.2. Fibrosis Disorders
2.2.1. Liver Fibrosis
2.2.2. Pulmonary Fibrosis
2.2.3. Systemic Sclerosis (SSc)
2.3. Cancer
2.3.1. Hepatocellular Carcinoma (HCC)
2.3.2. Neuroendocrine Tumor (NET)
2.3.3. Pancreatic Tumor
2.4. Gastrointestinal (GI) Tract
2.5. Nervous System
2.5.1. Regulation of Pain Disorders
2.5.2. Regulation of Neuroglia Function
2.5.3. Regulation of the Dopaminergic Pathway
3. Structure
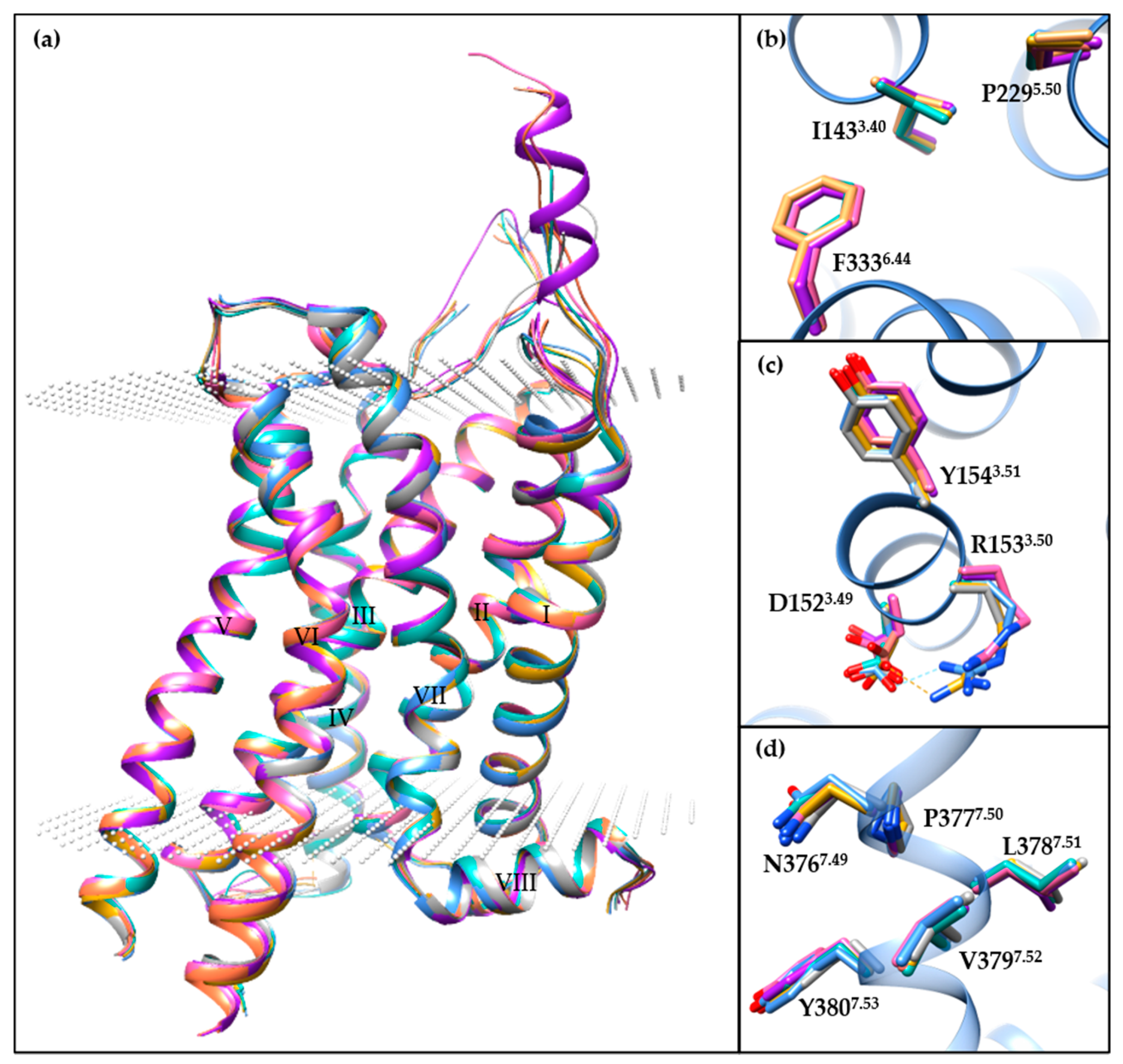
3.1. Crystal Structures
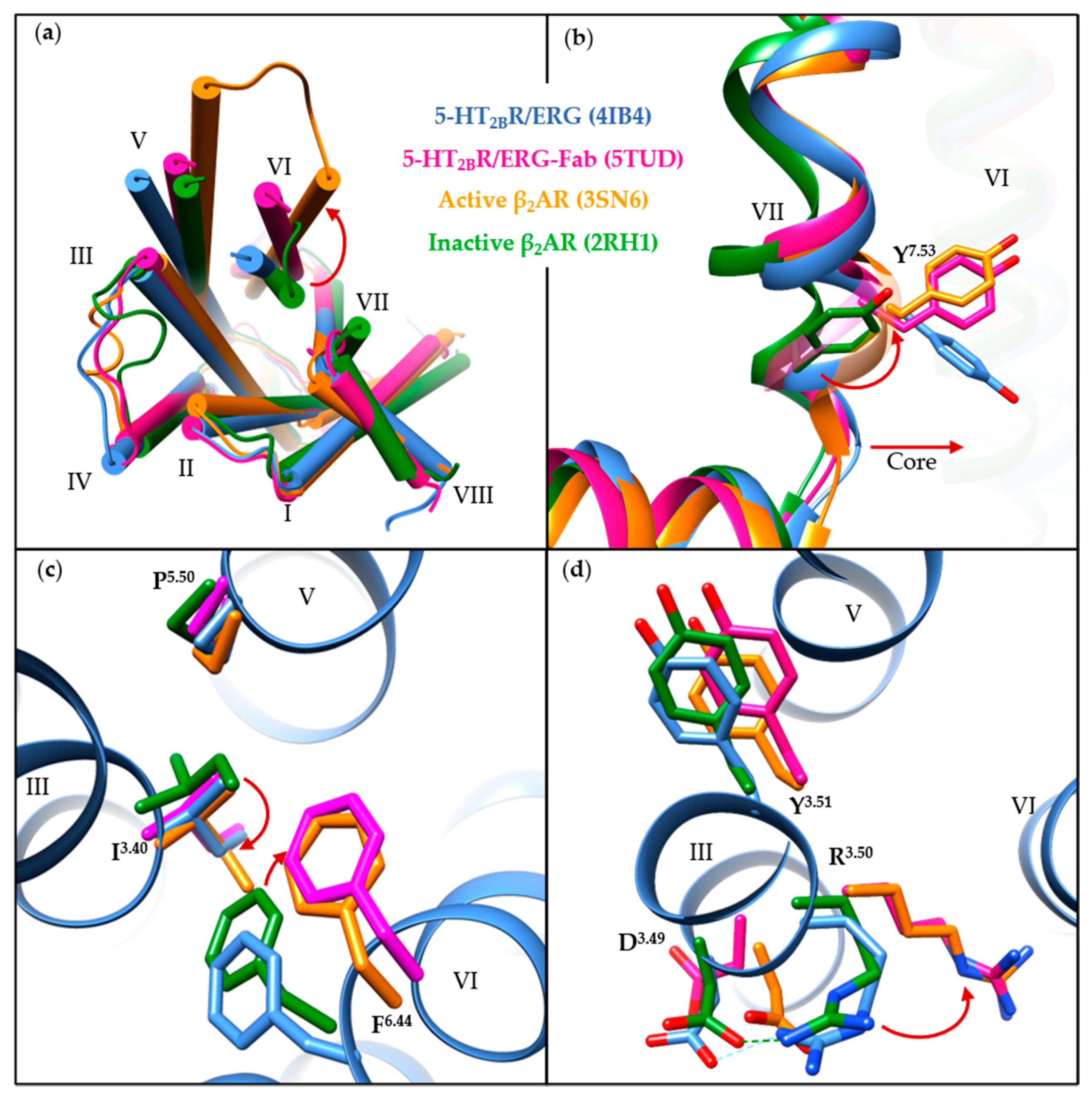
3.2. Conformational States
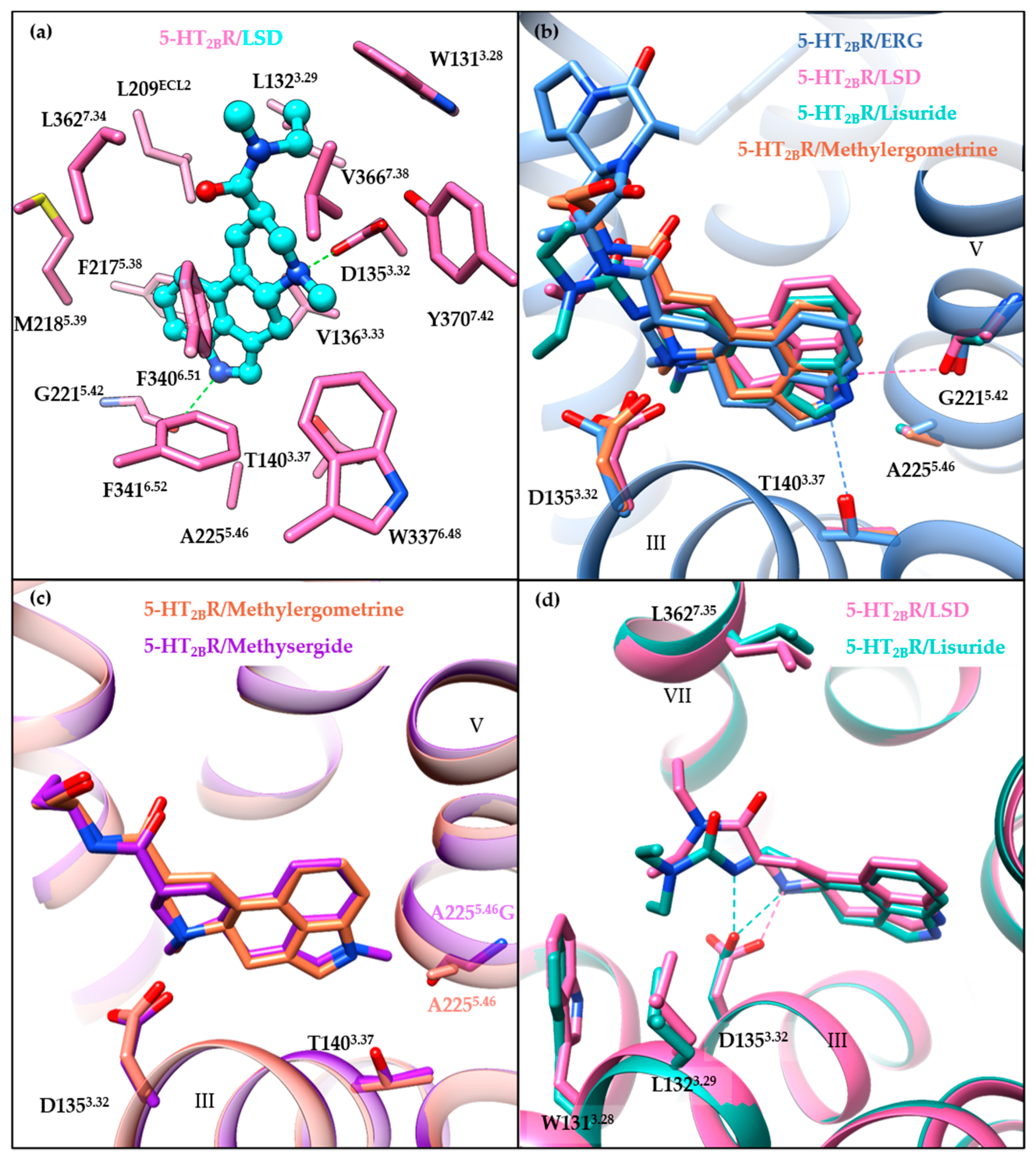
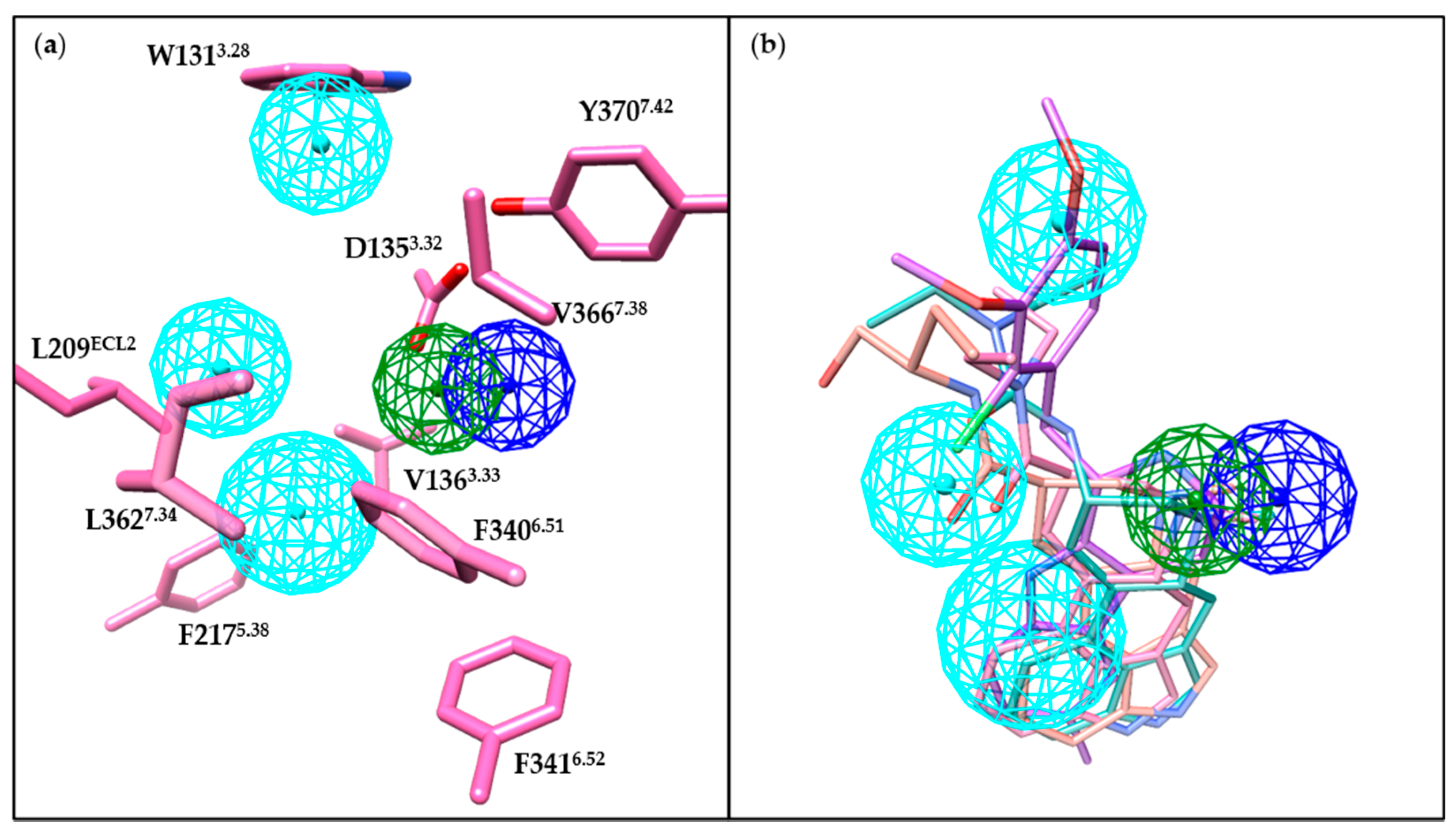
3.3. Orthosteric Binding Pocket (OBP)
3.4. Extended Binding Pocket (EBP)
4. Pharmaceutical Ligands
4.1. Clinical-Related Pharmaceutical Ligands of the 5-HT2BR
4.1.1. MT-500
4.1.2. PRX-08066
4.1.3. BF-1
4.1.4. AMAP102
4.1.5. AM1030
4.1.6. RQ-00310941
4.2. Representative Pharmaceutical Ligands of the 5-HT2BR
4.2.1. EXT5, EXT9, AM1125, and AM1476
4.2.2. Bis-Amino-Triazine Derivatives
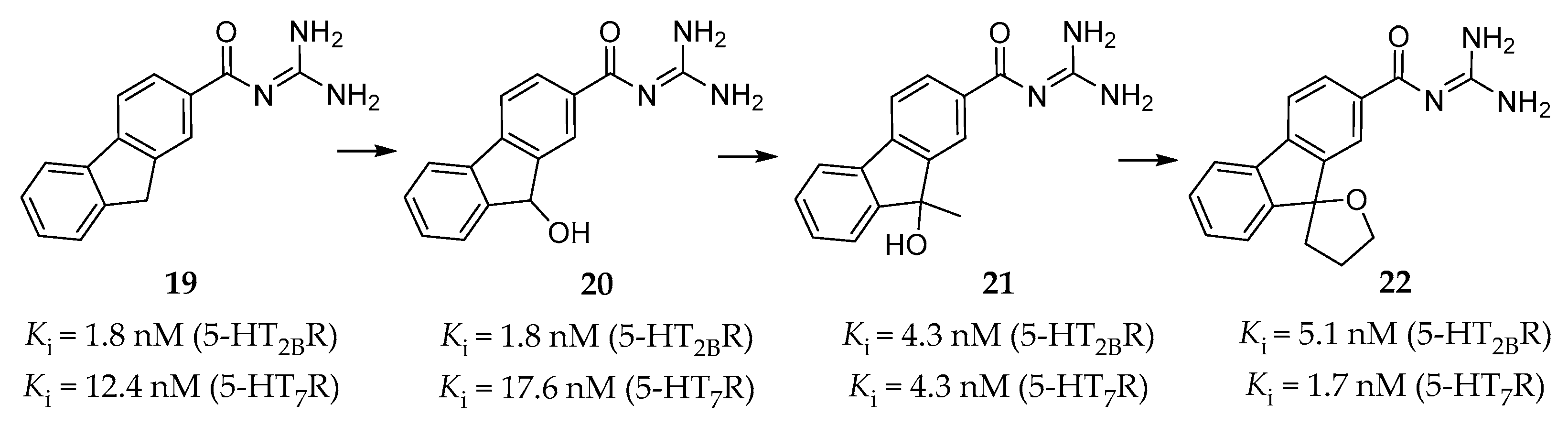
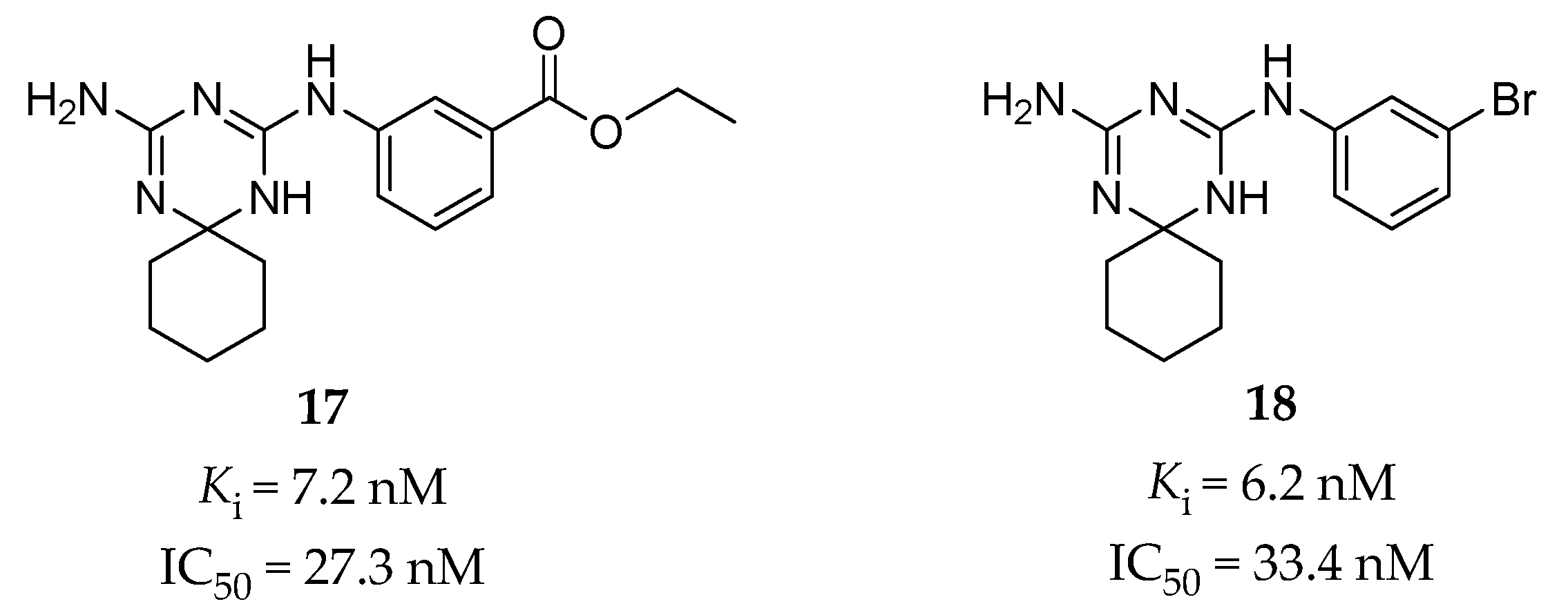
4.2.3. Guanidine Derivatives
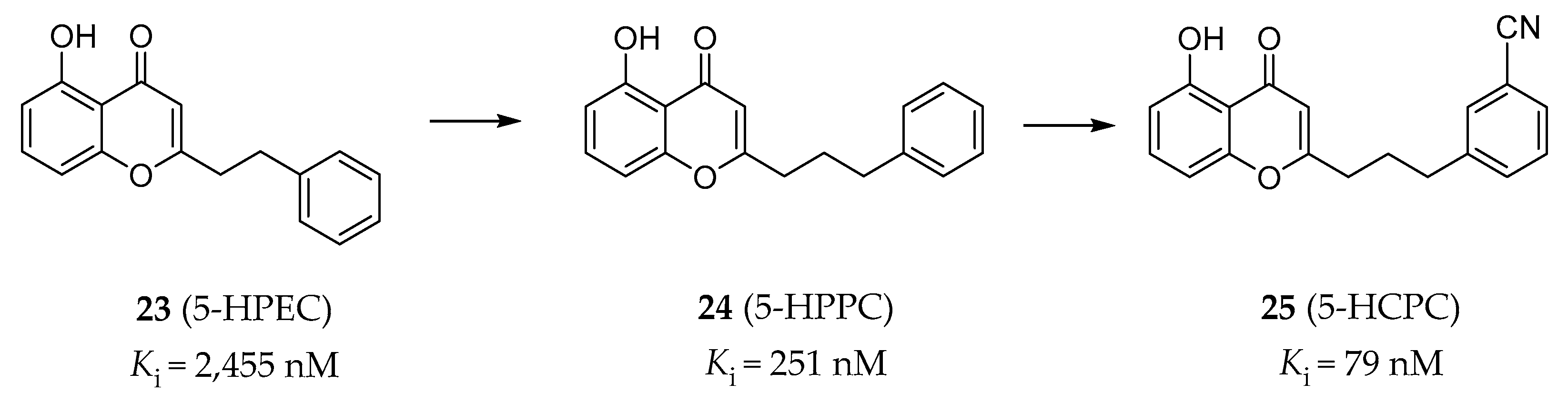
4.2.4. Chromone Derivatives
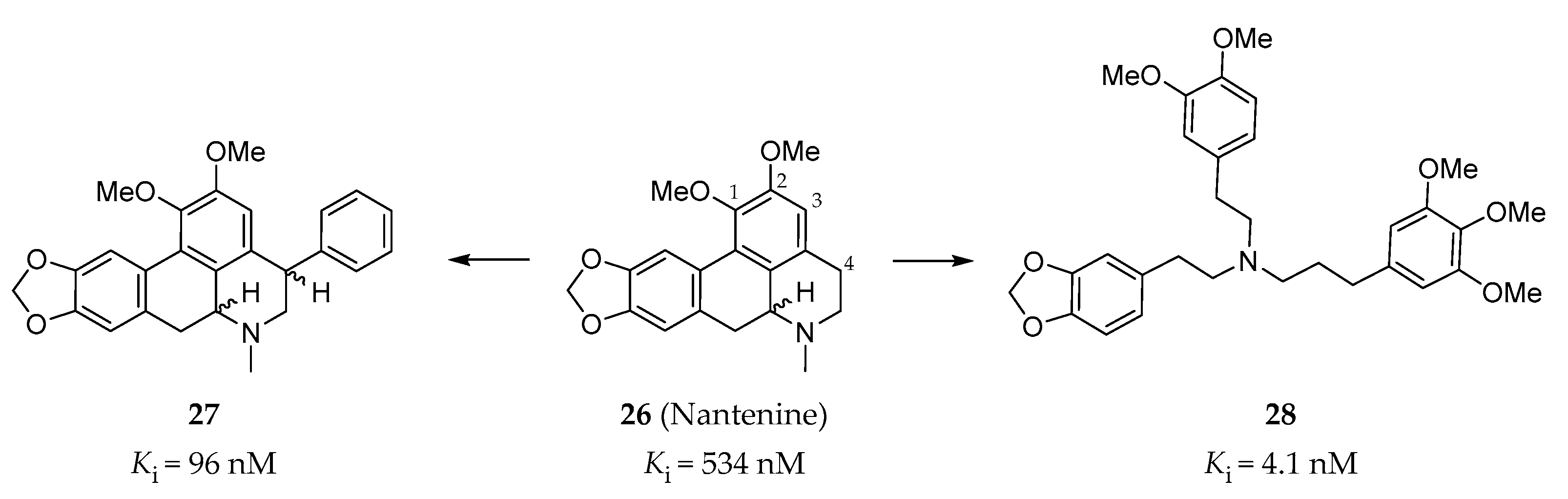
4.2.5. C4 Phenyl Aporphines and Tris-(phenylalkyl)amines
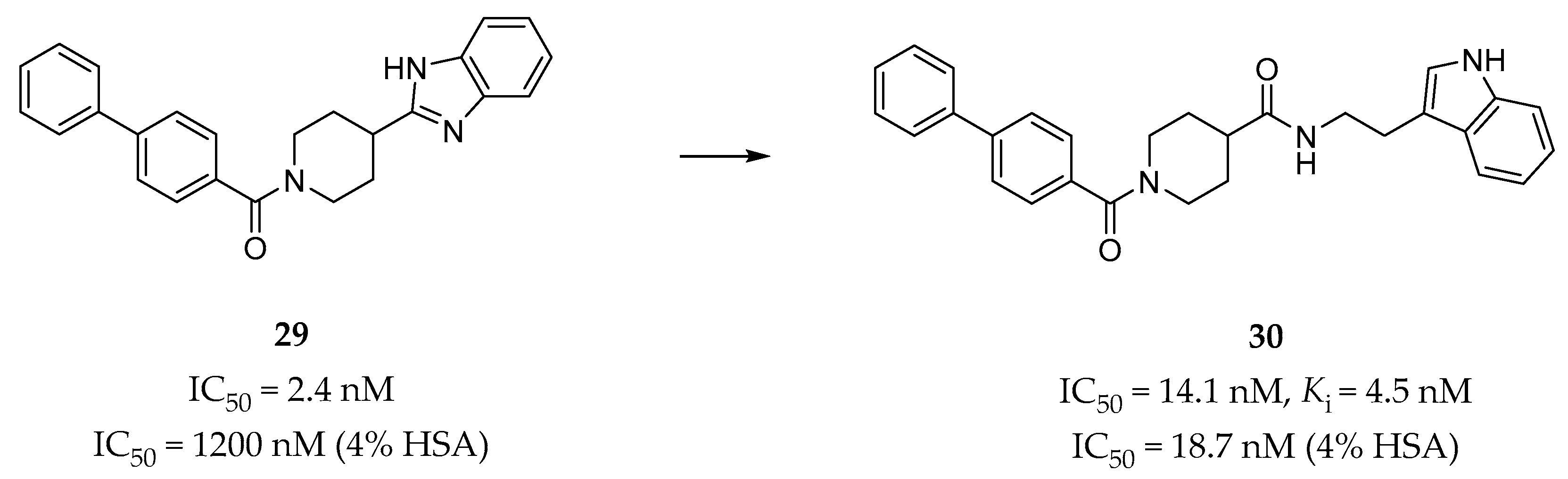
4.2.6. Biphenyl Amide Derivatives
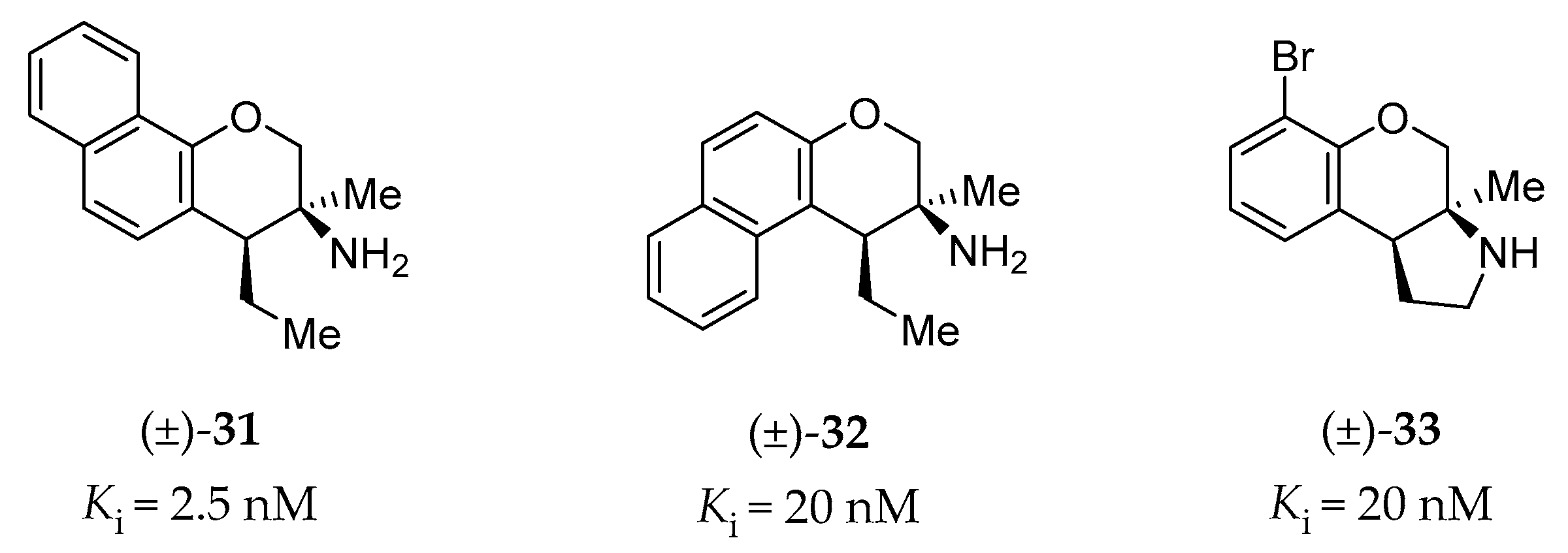
4.2.7. Chromane Derivatives
4.2.8. Other Pharmaceutical Ligands of the 5-HT2BR
4.3. Summary of Binding Features of 5-HT2BR Ligands
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
References
- Rapport, M.M.; Green, A.A.; Page, I.H. Crystalline Serotonin. Science 1948, 108, 329–330. [Google Scholar] [CrossRef] [PubMed]
- Padhariya, K.; Bhandare, R.; Canney, D.; Velingkar, V. Cardiovascular Concern of 5-HT2B Receptor and Recent Vistas in the Development of Its Antagonists. Cardiovasc. Hematol. Disord. Drug Targets. 2017, 17, 86–104. [Google Scholar] [CrossRef] [PubMed]
- Göthert, M.; Bönisch, H.; Malinowska, B.; Schlicker, E. Serotonin Discovery and Stepwise Disclosure of 5-HT Receptor Complexity over Four Decades. Part II. Some Contributions of Manfred Göthert. Pharmacol. Rep. 2020, 72, 271–284. [Google Scholar] [CrossRef] [PubMed]
- Foguet, M.; Nguyen, H.; Le, H.; Lübbert, H. Structure of the Mouse 5-HT1C, 5-HT2 and Stomach Fundus Serotonin Receptor Genes. Neuroreport 1992, 3, 345–348. [Google Scholar] [CrossRef] [PubMed]
- Schmuck, K.; Ullmer, C.; Engels, P.; Lübbert, H. Cloning and Functional Characterization of the Human 5-HT2B Serotonin Receptor. FEBS Lett. 1994, 342, 85–90. [Google Scholar] [CrossRef]
- Kursar, J.D.; Nelson, D.L.; Wainscott, D.B.; Baez, M. Molecular Cloning, Functional Expression, and MRNA Tissue Distribution of the Human 5-Hydroxytryptamine2B Receptor. Mol. Pharmacol. 1994, 46, 227–234. [Google Scholar] [PubMed]
- Devroye, C.; Cathala, A.; Piazza, P.V.; Spampinato, U. The Central Serotonin2B Receptor as a New Pharmacological Target for the Treatment of Dopamine-Related Neuropsychiatric Disorders: Rationale and Current Status of Research. Pharmacol. Ther. 2018, 181, 143–155. [Google Scholar] [CrossRef]
- Wirth, A.; Holst, K.; Ponimaskin, E. How Serotonin Receptors Regulate Morphogenic Signalling in Neurons. Prog. Neurobiol. 2017, 151, 35–56. [Google Scholar] [CrossRef]
- Nebigil, C.G.; Choi, D.-S.; Dierich, A.; Hickel, P.; Meur, M.L.; Messaddeq, N.; Launay, J.-M.; Maroteaux, L. Serotonin 2B Receptor Is Required for Heart Development. Proc. Natl. Acad. Sci. USA 2000, 97, 9508–9513. [Google Scholar] [CrossRef]
- Maroteaux, L.; Ayme-Dietrich, E.; Aubertin-Kirch, G.; Banas, S.; Quentin, E.; Lawson, R.; Monassier, L. New Therapeutic Opportunities for 5-HT2 Receptor Ligands. Pharmacol. Ther. 2017, 170, 14–36. [Google Scholar] [CrossRef]
- Nebigil, C.; Jaffré, F.; Messaddeq, N.; Hickel, P.; Monassier, L.; Launay, J.-M.; Maroteaux, L. Overexpression of the Serotonin 5-HT 2B Receptor in Heart Leads to Abnormal Mitochondrial Function and Cardiac Hypertrophy. Circulation 2003, 107, 3223–3229. [Google Scholar] [CrossRef] [PubMed]
- Jaffré, F.; Callebert, J.; Sarre, A.; Etienne, N.; Nebigil, C.G.; Launay, J.-M.; Maroteaux, L.; Monassier, L. Involvement of the Serotonin 5-HT2B Receptor in Cardiac Hypertrophy Linked to Sympathetic Stimulation: Control of Interleukin-6, Interleukin-1beta, and Tumor Necrosis Factor-Alpha Cytokine Production by Ventricular Fibroblasts. Circulation 2004, 110, 969–974. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Monassier, L.; Laplante, M.-A.; Jaffré, F.; Bousquet, P.; Maroteaux, L.; de Champlain, J. Serotonin 5-HT(2B) Receptor Blockade Prevents Reactive Oxygen Species-Induced Cardiac Hypertrophy in Mice. Hypertension 2008, 52, 301–307. [Google Scholar] [CrossRef] [PubMed]
- Bai, C.-F.; Liu, J.-C.; Zhao, R.; Cao, W.; Liu, S.-B.; Zhang, X.-N.; Guo, H.-J.; Yang, Q.; Yi, D.-H.; Zhao, M.-G. Role of 5-HT2B Receptors in Cardiomyocyte Apoptosis in Noradrenaline-Induced Cardiomyopathy in Rats. Clin. Exp. Pharmacol. Physiol. 2010, 37, e145–e151. [Google Scholar] [CrossRef] [PubMed]
- Fonfara, S.; Hetzel, U.; Oyama, M.A.; Kipar, A. The Potential Role of Myocardial Serotonin Receptor 2B Expression in Canine Dilated Cardiomyopathy. Vet. J. 2014, 199, 406–412. [Google Scholar] [CrossRef]
- Ceron, C.; Goyal, A.; Makaryus, A.N. Drug Induced Valvular Heart Disease. In StatPearls; StatPearls Publishing: Treasure Island, FL, USA, 2020. [Google Scholar]
- Fortier, J.H.; Pizzarotti, B.; Shaw, R.E.; Levy, R.J.; Ferrari, G.; Grau, J. Drug-Associated Valvular Heart Diseases and Serotonin-Related Pathways: A Meta-Analysis. Heart 2019, 105, 1140–1148. [Google Scholar] [CrossRef]
- Ayme-Dietrich, E.; Lawson, R.; Côté, F.; de Tapia, C.; Silva, S.D.; Ebel, C.; Hechler, B.; Gachet, C.; Guyonnet, J.; Rouillard, H.; et al. The Role of 5-HT2B Receptors in Mitral Valvulopathy: Bone Marrow Mobilization of Endothelial Progenitors. Br. J. Pharmacol. 2017, 174, 4123–4139. [Google Scholar] [CrossRef]
- Papoian, T.; Jagadeesh, G.; Saulnier, M.; Simpson, N.; Ravindran, A.; Yang, B.; Laniyonu, A.A.; Khan, I.; Szarfman, A. Regulatory Forum Review*: Utility of in Vitro Secondary Pharmacology Data to Assess Risk of Drug-Induced Valvular Heart Disease in Humans: Regulatory Considerations. Toxicol. Pathol. 2017, 45, 381–388. [Google Scholar] [CrossRef]
- Roth, B.L. Drugs and Valvular Heart Disease. N. Engl. J. Med. 2007, 356, 6–9. [Google Scholar] [CrossRef]
- Driesbaugh, K.H.; Branchetti, E.; Grau, J.B.; Keeney, S.J.; Glass, K.; Oyama, M.A.; Rioux, N.; Ayoub, S.; Sacks, M.S.; Quackenbush, J.; et al. Serotonin Receptor 2B Signaling with Interstitial Cell Activation and Leaflet Remodeling in Degenerative Mitral Regurgitation. J. Mol. Cell. Cardiol. 2018, 115, 94–103. [Google Scholar] [CrossRef]
- Hutcheson, J.D.; Ryzhova, L.M.; Setola, V.; Merryman, W.D. 5-HT2B Antagonism Arrests Non-Canonical TGF-Β1-Induced Valvular Myofibroblast Differentiation. J. Mol. Cell. Cardiol. 2012, 53, 707–714. [Google Scholar] [CrossRef] [PubMed]
- Joll, J.E.; Clark, C.R.; Peters, C.S.; Raddatz, M.A.; Bersi, M.R.; Merryman, W.D. Genetic Ablation of Serotonin Receptor 2B Improves Aortic Valve Hemodynamics of Notch1 Heterozygous Mice in a High-Cholesterol Diet Model. PLoS ONE 2020, 15, e0238407. [Google Scholar] [CrossRef] [PubMed]
- Launay, J.-M.; Hervé, P.; Peoc’h, K.; Tournois, C.; Callebert, J.; Nebigil, C.G.; Etienne, N.; Drouet, L.; Humbert, M.; Simonneau, G.; et al. Function of the Serotonin 5-Hydroxytryptamine 2B Receptor in Pulmonary Hypertension. Nat. Med. 2002, 8, 1129–1135. [Google Scholar] [CrossRef] [PubMed]
- Dumitrascu, R.; Kulcke, C.; Konigshoff, M.; Kouri, F.; Yang, X.; Morrell, N.; Ghofrani, H.A.; Weissmann, N.; Reiter, R.; Seeger, W.; et al. Terguride Ameliorates Monocrotaline-Induced Pulmonary Hypertension in Rats. Eur. Respir. J. 2011, 37, 1104–1118. [Google Scholar] [CrossRef]
- Watts, S.W.; Fink, G.D. 5-HT2B-Receptor Antagonist LY-272015 Is Antihypertensive in DOCA-Salt-Hypertensive Rats. Am. J. Physiol.-Heart Circ. Physiol. 1999, 276, H944–H952. [Google Scholar] [CrossRef]
- Bhat, L.; Hawkinson, J.; Cantillon, M.; Reddy, D.G.; Bhat, S.R.; Laurent, C.E.; Bouchard, A.; Biernat, M.; Salvail, D. RP5063, a Novel, Multimodal, Serotonin Receptor Modulator, Prevents Monocrotaline-Induced Pulmonary Arterial Hypertension in Rats. Eur. J. Pharmacol. 2017, 810, 92–99. [Google Scholar] [CrossRef]
- West, J.D.; Carrier, E.J.; Bloodworth, N.C.; Schroer, A.K.; Chen, P.; Ryzhova, L.M.; Gladson, S.; Shay, S.; Hutcheson, J.D.; Merryman, W.D. Serotonin 2B Receptor Antagonism Prevents Heritable Pulmonary Arterial Hypertension. PLoS ONE 2016, 11, e0148657. [Google Scholar] [CrossRef]
- Launay, J.-M.; Hervé, P.; Callebert, J.; Mallat, Z.; Collet, C.; Doly, S.; Belmer, A.; Diaz, S.L.; Hatia, S.; Côté, F.; et al. Serotonin 5-HT2B Receptors Are Required for Bone-Marrow Contribution to Pulmonary Arterial Hypertension. Blood 2012, 119, 1772–1780. [Google Scholar] [CrossRef]
- Bloodworth, N.C.; Clark, C.R.; West, J.D.; Snider, J.C.; Gaskill, C.; Shay, S.; Scott, C.; Bastarache, J.; Gladson, S.; Moore, C.; et al. Bone Marrow-Derived Proangiogenic Cells Mediate Pulmonary Arteriole Stiffening via Serotonin 2B Receptor Dependent Mechanism. Circ. Res. 2018, 123, e51–e64. [Google Scholar] [CrossRef]
- Ebrahimkhani, M.R.; Oakley, F.; Murphy, L.B.; Mann, J.; Moles, A.; Perugorria, M.J.; Ellis, E.; Lakey, A.F.; Burt, A.D.; Douglass, A.; et al. Stimulating Healthy Tissue Regeneration by Targeting the 5-HT2B Receptor in Chronic Liver Disease. Nat. Med. 2011, 17, 1668–1673. [Google Scholar] [CrossRef]
- Li, M.; Sun, Q.; Li, S.; Zhai, Y.; Wang, J.; Chen, B.; Lu, J. Chronic Restraint Stress Reduces Carbon Tetrachloride-induced Liver Fibrosis. Exp. Ther. Med. 2016, 11, 2147–2152. [Google Scholar] [CrossRef] [PubMed]
- Xiang, Y.; Ma, Y.-S.; Liu, J.-B.; Wu, Z.-J.; Wang, X.-P.; Liu, L.-P.; Wang, G.-R.; Fu, D.; Shi, W. Serotonin-Induced MiR-221/222 Contribute to the Activation of Hepatic Stellate Cells. Biologia (Bratisl.) 2020, 75, 997–1007. [Google Scholar] [CrossRef]
- Fabre, A.; Marchal-Sommé, J.; Marchand-Adam, S.; Quesnel, C.; Borie, R.; Dehoux, M.; Ruffié, C.; Callebert, J.; Launay, J.-M.; Hénin, D.; et al. Modulation of Bleomycin-Induced Lung Fibrosis by Serotonin Receptor Antagonists in Mice. Eur. Respir. J. 2008, 32, 426–436. [Google Scholar] [CrossRef] [PubMed]
- Königshoff, M.; Dumitrascu, R.; Udalov, S.; Amarie, O.V.; Reiter, R.; Grimminger, F.; Seeger, W.; Schermuly, R.T.; Eickelberg, O. Increased Expression of 5-Hydroxytryptamine2A/B Receptors in Idiopathic Pulmonary Fibrosis: A Rationale for Therapeutic Intervention. Thorax 2010, 65, 949–955. [Google Scholar] [CrossRef] [PubMed]
- Löfdahl, A.; Rydell-Törmänen, K.; Müller, C.; Holst, C.M.; Thiman, L.; Ekström, G.; Wenglén, C.; Larsson-Callerfelt, A.-K.; Westergren-Thorsson, G. 5-HT2B Receptor Antagonists Attenuate Myofibroblast Differentiation and Subsequent Fibrotic Responses in Vitro and in Vivo. Physiol. Rep. 2016, 4, e12873. [Google Scholar] [CrossRef]
- Abd-Alla, S.; Elaidy, S.; Essawy, S. Evaluation of the Antifibrotic Effect of Serotonin Receptor Antagonists on Bleomycin Induced Pulmonary Fibrosis in Rats. Egypt. J. Basic Clin. Pharmacol. 2017, 7, 35–46. [Google Scholar] [CrossRef]
- Löfdahl, A.; Rydell-Törmänen, K.; Larsson-Callerfelt, A.-K.; Wenglén, C.; Westergren-Thorsson, G. Pulmonary Fibrosis in vivo Displays Increased P21 Expression Reduced by 5-HT 2B Receptor Antagonists in Vitro—A Potential Pathway Affecting Proliferation. Sci. Rep. 2018, 8, 1927. [Google Scholar] [CrossRef]
- Dees, C.; Akhmetshina, A.; Zerr, P.; Reich, N.; Palumbo, K.; Horn, A.; Jüngel, A.; Beyer, C.; Krönke, G.; Zwerina, J.; et al. Platelet-Derived Serotonin Links Vascular Disease and Tissue Fibrosis. J. Exp. Med. 2011, 208, 961–972. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Misra, D.P.; Prasad, N.; Rastogi, K.; Singh, H.; Rai, M.K.; Agarwal, V. 5-HT2 and 5-HT2B Antagonists Attenuate pro-Fibrotic Phenotype in Human Adult Dermal Fibroblasts by Blocking TGF-Β1 Induced Non-Canonical Signaling Pathways Including STAT3: Implications for Fibrotic Diseases like Scleroderma. Int. J. Rheum. Dis. 2018, 21, 2128–2138. [Google Scholar] [CrossRef]
- Chaturvedi, S.; Rai, M.; Singh, H.; Misra, D.; Prasad, N.; Agrawal, V.; Agarwal, V. Dual Inhibition by Phosphodiesterase 5 and 5-HT 2B Inhibitor Leads to near Complete Amelioration of Fibrotic Potential of Human Adult Dermal Fibroblasts Isolated from a Scleroderma Patient. Indian J. Rheumatol. 2020. [Google Scholar] [CrossRef]
- Wenglén, C.; Pettersson, L.; Arozenius, H.; Ekström, G. SAT0314 A Novel Highly Selective 5-Hydroxytryptamine 2B (5-HT2B) Receptor Antagonist Ameliorating Fibrosis in Preclinical Models of Systemic Sclerosis. Ann. Rheum. Dis. 2017, 76, 891. [Google Scholar] [CrossRef]
- Wenglén, C.; Arozenius, H.; Pettersson, L.; Ekstrom, G. THU0353 AN ORALLY AVAILABLE HIGHLY SELECTIVE 5-HYDROXYTRYPTAMINE 2B (5-HT2B) RECEPTOR ANTAGONIST AMELIORATING PULMONARY AND DERMAL FIBROSIS IN PRECLINICAL MODELS OF SYSTEMIC SCLEROSIS. Ann. Rheum. Dis. 2019, 78, 457–458. [Google Scholar] [CrossRef]
- Sharma, S.; Pande, G.; Rai, M.; Misra, D.; Gupta, L.; Agarwal, V. The Convergence of Fibrosis and Immune Pathways: 5-HT2 and 5-HT2B Antagonists Attenuate Profibrotic Phenotype in Human Pancreatic Stellate Cells by Modulating Signal Transducer and Activator of Transcription 3 Phosphorylation. Indian J. Rheumatol. 2020. [Google Scholar] [CrossRef]
- Higashi, T.; Friedman, S.L.; Hoshida, Y. Hepatic Stellate Cells as Key Target in Liver Fibrosis. Adv. Drug Deliv. Rev. 2017, 121, 27–42. [Google Scholar] [CrossRef] [PubMed]
- Khomich, O.; Ivanov, A.V.; Bartosch, B. Metabolic Hallmarks of Hepatic Stellate Cells in Liver Fibrosis. Cells 2020, 9, 24. [Google Scholar] [CrossRef] [PubMed]
- Elaidy, S.M.; Essawy, S.S. The Antifibrotic Effects of Alveolar Macrophages 5-HT2C Receptors Blockade on Bleomycin-Induced Pulmonary Fibrosis in Rats. Pharmacol. Rep. PR 2016, 68, 1244–1253. [Google Scholar] [CrossRef]
- Tawfik, M.K.; Makary, S. 5-HT7 Receptor Antagonism (SB-269970) Attenuates Bleomycin-Induced Pulmonary Fibrosis in Rats via Downregulating Oxidative Burden and Inflammatory Cascades and Ameliorating Collagen Deposition: Comparison to Terguride. Eur. J. Pharmacol. 2017, 814, 114–123. [Google Scholar] [CrossRef]
- Sarrouilhe, D.; Mesnil, M. Serotonin and Human Cancer: A Critical View. Biochimie 2019, 161, 46–50. [Google Scholar] [CrossRef]
- Soll, C.; Riener, M.-O.; Oberkofler, C.E.; Hellerbrand, C.; Wild, P.J.; DeOliveira, M.L.; Clavien, P.-A. Expression of Serotonin Receptors in Human Hepatocellular Cancer. Clin. Cancer Res. 2012, 18, 5902–5910. [Google Scholar] [CrossRef]
- Liang, C.; Chen, W.; Zhi, X.; Ma, T.; Xia, X.; Liu, H.; Zhang, Q.; Hu, Q.; Zhang, Y.; Bai, X.; et al. Serotonin Promotes the Proliferation of Serum-Deprived Hepatocellular Carcinoma Cells via Upregulation of FOXO3a. Mol. Cancer 2013, 12, 14. [Google Scholar] [CrossRef]
- Yang, Q.; Yan, C.; Yin, C.; Gong, Z. Serotonin Activated Hepatic Stellate Cells Contribute to Sex Disparity in Hepatocellular Carcinoma. Cell. Mol. Gastroenterol. Hepatol. 2017, 3, 484–499. [Google Scholar] [CrossRef] [PubMed]
- Yang, Q.; Yan, C.; Gong, Z. Interaction of Hepatic Stellate Cells with Neutrophils and Macrophages in the Liver Following Oncogenic Kras Activation in Transgenic Zebrafish. Sci. Rep. 2018, 8, 8495. [Google Scholar] [CrossRef] [PubMed]
- Hassan, S.A.; Banchs, J.; Iliescu, C.; Dasari, A.; Lopez-Mattei, J.; Yusuf, S.W. Carcinoid Heart Disease. Heart 2017, 103, 1488–1495. [Google Scholar] [CrossRef] [PubMed]
- Hayes, A.R.; Davar, J.; Caplin, M.E. Carcinoid Heart Disease: A Review. Endocrinol. Metab. Clin. 2018, 47, 671–682. [Google Scholar] [CrossRef] [PubMed]
- Svejda, B.; Kidd, M.; Giovinazzo, F.; Eltawil, K.; Gustafsson, B.I.; Pfragner, R.; Modlin, I.M. The 5-HT2B Receptor Plays a Key Regulatory Role in Both Neuroendocrine Tumor Cell Proliferation and the Modulation of the Fibroblast Component of the Neoplastic Microenvironment. Cancer 2010, 116, 2902–2912. [Google Scholar] [CrossRef] [PubMed]
- Jiang, S.-H.; Li, J.; Dong, F.-Y.; Yang, J.-Y.; Liu, D.-J.; Yang, X.-M.; Wang, Y.-H.; Yang, M.-W.; Fu, X.-L.; Zhang, X.-X.; et al. Increased Serotonin Signaling Contributes to the Warburg Effect in Pancreatic Tumor Cells Under Metabolic Stress and Promotes Growth of Pancreatic Tumors in Mice. Gastroenterology 2017, 153, 277–291.e19. [Google Scholar] [CrossRef] [PubMed]
- Borman, R.A.; Tilford, N.S.; Harmer, D.W.; Day, N.; Ellis, E.S.; Sheldrick, R.L.G.; Carey, J.; Coleman, R.A.; Baxter, G.S. 5-HT 2B Receptors Play a Key Role in Mediating the Excitatory Effects of 5-HT in Human Colon in Vitro. Br. J. Pharmacol. 2002, 135, 1144–1151. [Google Scholar] [CrossRef]
- Wouters, M.M.; Gibbons, S.J.; Roeder, J.L.; Distad, M.; Ou, Y.; Strege, P.R.; Szurszewski, J.H.; Farrugia, G. Exogenous Serotonin Regulates Proliferation of Interstitial Cells of Cajal in Mouse Jejunum Through 5-HT2B Receptors. Gastroenterology 2007, 133, 897–906. [Google Scholar] [CrossRef]
- Tharayil, V.S.; Wouters, M.M.; Stanich, J.E.; Roeder, J.L.; Lei, S.; Beyder, A.; Gomez-Pinilla, P.J.; Gershon, M.D.; Maroteaux, L.; Gibbons, S.J.; et al. Lack of Serotonin 5-HT2B Receptor Alters Proliferation and Network Volume of Interstitial Cells of Cajal in Vivo. Neurogastroenterol. Motil. Off. J. Eur. Gastrointest. Motil. Soc. 2010, 22, 462.e110. [Google Scholar] [CrossRef]
- Wouters, M.M.; Roeder, J.L.; Tharayil, V.S.; Stanich, J.E.; Strege, P.R.; Lei, S.; Bardsley, M.R.; Ordog, T.; Gibbons, S.J.; Farrugia, G. Protein Kinase Cγ Mediates Regulation of Proliferation by the Serotonin 5-Hydroxytryptamine Receptor 2B. J. Biol. Chem. 2009, 284, 21177–21184. [Google Scholar] [CrossRef]
- Bassil, A.K.; Taylor, C.M.; Bolton, V.J.N.; Gray, K.M.; Brown, J.D.; Cutler, L.; Summerfield, S.G.; Bruton, G.; Winchester, W.J.; Lee, K.; et al. Inhibition of Colonic Motility and Defecation by RS-127445 Suggests an Involvement of the 5-HT2B Receptor in Rodent Large Bowel Physiology. Br. J. Pharmacol. 2009, 158, 252–258. [Google Scholar] [CrossRef] [PubMed]
- O’mahony, S.M.; Bulmer, D.C.; Coelho, A.-M.; Fitzgerald, P.; Bongiovanni, C.; Lee, K.; Winchester, W.; Dinan, T.G.; Cryan, J.F. 5-HT2B Receptors Modulate Visceral Hypersensitivity in a Stress-Sensitive Animal Model of Brain-Gut Axis Dysfunction. Neurogastroenterol. Motil. 2010, 22, 573.e124. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Inagaki, K.; Taniguchi, K.; Sakaguchi, Y.; Kawamura, K. The Novel 5-HT2B Receptor Antagonist, RQ-00310941, Attenuates Visceral Hypersensitivity and Abnormal Defecation in Rat Models. Gastroenterology 2011, 140, S-607. [Google Scholar] [CrossRef]
- Zhou, Y.; Ma, J.; Lin, X.; Huang, X.-P.; Wu, K.; Huang, N. Structure-Based Discovery of Novel and Selective 5-Hydroxytryptamine 2B Receptor Antagonists for the Treatment of Irritable Bowel Syndrome. J. Med. Chem. 2016, 59, 707–720. [Google Scholar] [CrossRef] [PubMed]
- Morita, H.; Mochiki, E.; Takahashi, N.; Kawamura, K.; Watanabe, A.; Sutou, T.; Ogawa, A.; Yanai, M.; Ogata, K.; Fujii, T.; et al. Effects of 5-HT2B, 5-HT3 and 5-HT4 Receptor Antagonists on Gastrointestinal Motor Activity in Dogs. World J. Gastroenterol. WJG 2013, 19, 6604–6612. [Google Scholar] [CrossRef] [PubMed]
- Robson, M.J.; Quinlan, M.A.; Blakely, R.D. Immune System Activation and Depression: Roles of Serotonin in the Central Nervous System and Periphery. ACS Chem. Neurosci. 2017, 8, 932–942. [Google Scholar] [CrossRef]
- Bacqué-Cazenave, J.; Bharatiya, R.; Barrière, G.; Delbecque, J.-P.; Bouguiyoud, N.; Di Giovanni, G.; Cattaert, D.; De Deurwaerdère, P. Serotonin in Animal Cognition and Behavior. Int. J. Mol. Sci. 2020, 21, 1649. [Google Scholar] [CrossRef]
- Radke, A.K.; Piantadosi, P.T.; Uhl, G.R.; Hall, F.S.; Holmes, A. Improved Visual Discrimination Learning in Mice with Partial 5-HT2B Gene Deletion. Neurosci. Lett. 2020, 738, 135378. [Google Scholar] [CrossRef]
- Gibbs, M.E. Role of Glycogenolysis in Memory and Learning: Regulation by Noradrenaline, Serotonin and ATP. Front. Integr. Neurosci. 2016, 9, 70. [Google Scholar] [CrossRef]
- Meneses, A.; Nieto-Vera, R.; Anaya-Jiménez, R.M. 5-HT2A/2B/2C Receptors, Memory and Therapeutic Targets. The Receptors. In 5-HT2A Receptors in the Central Nervous System; Guiard, B.P., Di Giovanni, G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; Volume 32, pp. 259–271. ISBN 978-3-319-70474-6. [Google Scholar]
- Borkowski, L.F.; Craig, T.A.; Stricklin, O.E.; Johnson, K.A.; Nichols, N.L. 5-HT2A/B Receptor Expression in the Phrenic Motor Nucleus in a Rat Model of ALS (SOD1G93A). Respir. Physiol. Neurobiol. 2020, 279, 103471. [Google Scholar] [CrossRef]
- Perim, R.R.; Fields, D.P.; Mitchell, G.S. Cross-Talk Inhibition between 5-HT2B and 5-HT7 Receptors in Phrenic Motor Facilitation via NADPH Oxidase and PKA. Am. J. Physiol.-Regul. Integr. Comp. Physiol. 2018, 314, R709–R715. [Google Scholar] [CrossRef] [PubMed]
- Tadjalli, A.; Mitchell, G.S. Cervical Spinal 5-HT2A and 5-HT2B Receptors Are Both Necessary for Moderate Acute Intermittent Hypoxia-Induced Phrenic Long-Term Facilitation. J. Appl. Physiol. 2019, 127, 432–443. [Google Scholar] [CrossRef] [PubMed]
- Segelcke, D.; Messlinger, K. Putative Role of 5-HT2B Receptors in Migraine Pathophysiology. Cephalalgia Int. J. Headache 2017, 37, 365–371. [Google Scholar] [CrossRef] [PubMed]
- Villalón, M.; Maassen, C.; Van Den Brink, A. The Role of 5-Hydroxytryptamine in the Pathophysiology of Migraine and Its Relevance to the Design of Novel Treatments. Mini Rev. Med. Chem. 2017, 17, 928–938. [Google Scholar] [CrossRef] [PubMed]
- Liu, Q.Q.; Yao, X.X.; Gao, S.H.; Li, R.; Li, B.J.; Yang, W.; Cui, R.J. Role of 5-HT Receptors in Neuropathic Pain: Potential Therapeutic Implications. Pharmacol. Res. 2020, 159, 104949. [Google Scholar] [CrossRef] [PubMed]
- Fozard, J.R.; Gray, J.A. 5-HT1C Receptor Activation: A Key Step in the Initiation of Migraine? Trends Pharmacol. Sci. 1989, 10, 307–309. [Google Scholar] [CrossRef]
- Schmitz, B.; Ullmer, C.; Segelcke, D.; Gwarek, M.; Zhu, X.-R.; Lübbert, H. BF-1—A Novel Selective 5-HT2B Receptor Antagonist Blocking Neurogenic Dural Plasma Protein Extravasation in Guinea Pigs. Eur. J. Pharmacol. 2015, 751, 73–80. [Google Scholar] [CrossRef]
- Hunfeld, A.; Segelcke, D.; Bäcker, I.; Mecheri, B.; Hemmer, K.; Dlugosch, E.; Andriske, M.; Paris, F.; Zhu, X.; Lübbert, H. Hypoxia Facilitates Neurogenic Dural Plasma Protein Extravasation in Mice: A Novel Animal Model for Migraine Pathophysiology. Sci. Rep. 2015, 5, 17845. [Google Scholar] [CrossRef]
- Lin, S.-Y.; Chang, W.-J.; Lin, C.-S.; Huang, C.-Y.; Wang, H.-F.; Sun, W.-H. Serotonin Receptor 5-HT2B Mediates Serotonin-Induced Mechanical Hyperalgesia. J. Neurosci. 2011, 31, 1410–1418. [Google Scholar] [CrossRef]
- Sun, W.-H. Commentary: Serotonin Receptor 2B Mediates Mechanical Hyperalgesia by Regulating Transient Receptor Potential Vanilloid 1. J. Neurol. Neuromedicine 2016, 1, 23–26. [Google Scholar] [CrossRef]
- Cervantes-Durán, C.; Vidal-Cantú, G.C.; Barragán-Iglesias, P.; Pineda-Farias, J.B.; Bravo-Hernández, M.; Murbartián, J.; Granados-Soto, V. Role of Peripheral and Spinal 5-HT2B Receptors in Formalin-Induced Nociception. Pharmacol. Biochem. Behav. 2012, 102, 30–35. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Durán, C.; Pineda-Farias, J.B.; Bravo-Hernández, M.; Quiñonez-Bastidas, G.N.; Vidal-Cantú, G.C.; Barragán-Iglesias, P.; Granados-Soto, V. Evidence for the Participation of Peripheral 5-HT2A, 5-HT2B, and 5-HT2C Receptors in Formalin-Induced Secondary Mechanical Allodynia and Hyperalgesia. Neuroscience 2013, 232, 169–181. [Google Scholar] [CrossRef] [PubMed]
- Cervantes-Durán, C.; Vidal-Cantú, G.C.; Godínez-Chaparro, B.; Granados-Soto, V. Role of Spinal 5-HT2 Receptors Subtypes in Formalin-Induced Long-Lasting Hypersensitivity. Pharmacol. Rep. 2016, 68, 434–442. [Google Scholar] [CrossRef] [PubMed]
- Pineda-Farias, J.B.; Velázquez-Lagunas, I.; Barragán-Iglesias, P.; Cervantes-Durán, C.; Granados-Soto, V. 5-HT2B Receptor Antagonists Reduce Nerve Injury-Induced Tactile Allodynia and Expression of 5-HT2B Receptors. Drug Dev. Res. 2015, 76, 31–39. [Google Scholar] [CrossRef] [PubMed]
- Jiang, M.; Bo, J.; Lei, Y.; Hu, F.; Xia, Z.; Liu, Y.; Lu, C.; Sun, Y.; Hou, B.; Ni, K.; et al. Anxiety-Induced Hyperalgesia in Female Rats Is Mediated by Cholecystokinin 2 Receptor in Rostral Ventromedial Medulla and Spinal 5-Hydroxytryptamine 2B Receptor. J. Pain Res. 2019, 12, 2009–2026. [Google Scholar] [CrossRef] [PubMed]
- Urtikova, N.; Berson, N.; Van Steenwinckel, J.; Doly, S.; Truchetto, J.; Maroteaux, L.; Pohl, M.; Conrath, M. Antinociceptive Effect of Peripheral Serotonin 5-HT2B Receptor Activation on Neuropathic Pain. Pain 2012, 153, 1320–1331. [Google Scholar] [CrossRef] [PubMed]
- D’Andrea, I.; Béchade, C.; Maroteaux, L. Serotonin and 5-HT2B receptors in microglia control of behavior. In Handbook of Behavioral Neuroscience; Müller, C.P., Cunningham, K.A., Eds.; Handbook of the Behavioral Neurobiology of Serotonin; Elsevier: Amsterdam, The Netherlands, 2020; Chapter 34; Volume 31, pp. 589–599. [Google Scholar]
- Kolodziejczak, M.; Béchade, C.; Gervasi, N.; Irinopoulou, T.; Banas, S.M.; Cordier, C.; Rebsam, A.; Roumier, A.; Maroteaux, L. Serotonin Modulates Developmental Microglia via 5-HT2B Receptors: Potential Implication during Synaptic Refinement of Retinogeniculate Projections. ACS Chem. Neurosci. 2015, 6, 1219–1230. [Google Scholar] [CrossRef] [PubMed]
- Béchade, C.; D’Andrea, I.; Etienne, F.; Verdonk, F.; Moutkine, I.; Banas, S.M.; Kolodziejczak, M.; Diaz, S.L.; Parkhurst, C.N.; Gan, W.B.; et al. The Serotonin 2B Receptor Is Required in Neonatal Microglia to Limit Neuroinflammation and Sickness Behavior in Adulthood. Glia 2020. [Google Scholar] [CrossRef]
- Zhang, S.; Li, B.; Lovatt, D.; Xu, J.; Song, D.; Goldman, S.A.; Nedergaard, M.; Hertz, L.; Peng, L. 5-HT2B Receptors Are Expressed on Astrocytes from Brain and in Culture and Are a Chronic Target for All Five Conventional “Serotonin-Specific Reuptake Inhibitors”. Neuron Glia Biol. 2010, 6, 113–125. [Google Scholar] [CrossRef]
- Diaz, S.L.; Doly, S.; Narboux-Nême, N.; Fernández, S.; Mazot, P.; Banas, S.M.; Boutourlinsky, K.; Moutkine, I.; Belmer, A.; Roumier, A.; et al. 5-HT 2B Receptors Are Required for Serotonin-Selective Antidepressant Actions. Mol. Psychiatry 2012, 17, 154–163. [Google Scholar] [CrossRef]
- Li, B.; Zhang, S.; Zhang, H.; Nu, W.; Cai, L.; Hertz, L.; Peng, L. Fluoxetine-Mediated 5-HT2B Receptor Stimulation in Astrocytes Causes EGF Receptor Transactivation and ERK Phosphorylation. Psychopharmacology (Berl.) 2008, 201, 443–458. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Zhang, S.; Li, M.; Hertz, L.; Peng, L. Serotonin Increases ERK1/2 Phosphorylation in Astrocytes by Stimulation of 5-HT2B and 5-HT2C Receptors. Neurochem. Int. 2010, 57, 432–439. [Google Scholar] [CrossRef] [PubMed]
- Peng, L.; Song, D.; Li, B.; Verkhratsky, A. Astroglial 5-HT2B Receptor in Mood Disorders. Expert Rev. Neurother. 2018, 18, 435–442. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Song, D.; Gu, L.; Ren, Y.; Verkhratsky, A.; Peng, L. Decrease of Gene Expression of Astrocytic 5-HT2B Receptors Parallels Development of Depressive Phenotype in a Mouse Model of Parkinson’s Disease. Front. Cell. Neurosci. 2015, 9. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Ma, K.; Verkhratsky, A.; Peng, L. L-Dopa and Fluoxetine Upregulate Astroglial 5-HT2B Receptors and Ameliorate Depression in Parkinson’s Disease Mice. Neuroglia 2018, 1, 6. [Google Scholar] [CrossRef]
- Xia, M.; Li, Z.; Li, S.; Liang, S.; Li, X.; Chen, B.; Zhang, M.; Dong, C.; Verkhratsky, A.; Guan, D.; et al. Sleep Deprivation Selectively Down-Regulates Astrocytic 5-HT2B Receptors and Triggers Depressive-Like Behaviors via Stimulating P2X7 Receptors in Mice. Neurosci. Bull. 2020, 36, 1259–1270. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Liang, S.; Li, Z.; Li, S.; Xia, M.; Verkhratsky, A.; Li, B. Leptin Increases Expression of 5-HT2B Receptors in Astrocytes Thus Enhancing Action of Fluoxetine on the Depressive Behavior Induced by Sleep Deprivation. Front. Psychiatry 2019, 9, 734. [Google Scholar] [CrossRef]
- Spampinato, U.; Cathala, A.; Devroye, C. The serotonin2B receptor and neurochemical regulation in the brain. In Handbook of Behavioral Neuroscience; Müller, C.P., Cunningham, K.A., Eds.; Handbook of the Behavioral Neurobiology of Serotonin; Elsevier: Amsterdam, The Netherlands, 2020; Chapter 7; Volume 31, pp. 147–156. [Google Scholar]
- Devroye, C.; Haddjeri, N.; Cathala, A.; Rovera, R.; Drago, F.; Piazza, P.V.; Artigas, F.; Spampinato, U. Opposite Control of Mesocortical and Mesoaccumbal Dopamine Pathways by Serotonin2B Receptor Blockade: Involvement of Medial Prefrontal Cortex Serotonin1A Receptors. Neuropharmacology 2017, 119, 91–99. [Google Scholar] [CrossRef]
- Auclair, A.L.; Cathala, A.; Sarrazin, F.; Depoortère, R.; Piazza, P.V.; Newman-Tancredi, A.; Spampinato, U. The Central Serotonin2B Receptor: A New Pharmacological Target to Modulate the Mesoaccumbens Dopaminergic Pathway Activity. J. Neurochem. 2010, 114, 1323–1332. [Google Scholar] [CrossRef]
- Devroye, C.; Cathala, A.; Di Marco, B.; Caraci, F.; Drago, F.; Piazza, P.V.; Spampinato, U. Central Serotonin2B Receptor Blockade Inhibits Cocaine-Induced Hyperlocomotion Independently of Changes of Subcortical Dopamine Outflow. Neuropharmacology 2015, 97, 329–337. [Google Scholar] [CrossRef]
- Devroye, C.; Cathala, A.; Haddjeri, N.; Rovera, R.; Vallée, M.; Drago, F.; Piazza, P.V.; Spampinato, U. Differential Control of Dopamine Ascending Pathways by Serotonin2B Receptor Antagonists: New Opportunities for the Treatment of Schizophrenia. Neuropharmacology 2016, 109, 59–68. [Google Scholar] [CrossRef] [PubMed]
- Pitychoutis, P.M.; Belmer, A.; Moutkine, I.; Adrien, J.; Maroteaux, L. Mice Lacking the Serotonin Htr 2B Receptor Gene Present an Antipsychotic-Sensitive Schizophrenic-Like Phenotype. Neuropsychopharmacology 2015, 40, 2764–2773. [Google Scholar] [CrossRef] [PubMed]
- Doly, S.; Valjent, E.; Setola, V.; Callebert, J.; Hervé, D.; Launay, J.-M.; Maroteaux, L. Serotonin 5-HT2B Receptors Are Required for 3,4-Methylenedioxymethamphetamine-Induced Hyperlocomotion and 5-HT Release In Vivo and In Vitro. J. Neurosci. 2008, 28, 2933–2940. [Google Scholar] [CrossRef] [PubMed]
- Cathala, A.; Devroye, C.; Robert, É.; Vallée, M.; Revest, J.-M.; Artigas, F.; Spampinato, U. Serotonin2B Receptor Blockade in the Rat Dorsal Raphe Nucleus Suppresses Cocaine-Induced Hyperlocomotion through an Opposite Control of Mesocortical and Mesoaccumbens Dopamine Pathways. Neuropharmacology 2020, 180, 108309. [Google Scholar] [CrossRef]
- Cong, X.; Topin, J.; Golebiowski, J. Class A GPCRs: Structure, Function, Modeling and Structure-Based Ligand Design. Curr. Pharm. Des. 2017, 23, 4390–4409. [Google Scholar] [CrossRef]
- Wacker, D.; Wang, C.; Katritch, V.; Han, G.W.; Huang, X.-P.; Vardy, E.; McCorvy, J.D.; Jiang, Y.; Chu, M.; Siu, F.Y.; et al. Structural Features for Functional Selectivity at Serotonin Receptors. Science 2013, 340, 615–619. [Google Scholar] [CrossRef]
- Liu, W.; Wacker, D.; Gati, C.; Han, G.W.; James, D.; Wang, D.; Nelson, G.; Weierstall, U.; Katritch, V.; Barty, A.; et al. Serial Femtosecond Crystallography of G Protein–Coupled Receptors. Science 2013, 342, 1521–1524. [Google Scholar] [CrossRef]
- Wacker, D.; Wang, S.; McCorvy, J.D.; Betz, R.M.; Venkatakrishnan, A.J.; Levit, A.; Lansu, K.; Schools, Z.L.; Che, T.; Nichols, D.E.; et al. Crystal Structure of an LSD-Bound Human Serotonin Receptor. Cell 2017, 168, 377–389. [Google Scholar] [CrossRef]
- Ishchenko, A.; Wacker, D.; Kapoor, M.; Zhang, A.; Han, G.W.; Basu, S.; Patel, N.; Messerschmidt, M.; Weierstall, U.; Liu, W.; et al. Structural Insights into the Extracellular Recognition of the Human Serotonin 2B Receptor by an Antibody. Proc. Natl. Acad. Sci. USA 2017, 114, 8223–8228. [Google Scholar] [CrossRef]
- McCorvy, J.D.; Wacker, D.; Wang, S.; Agegnehu, B.; Liu, J.; Lansu, K.; Tribo, A.R.; Olsen, R.H.J.; Che, T.; Jin, J.; et al. Structural Determinants of 5-HT 2B Receptor Activation and Biased Agonism. Nat. Struct. Mol. Biol. 2018, 25, 787–796. [Google Scholar] [CrossRef]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A Visualization System for Exploratory Research and Analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, S.G.F.; DeVree, B.T.; Zou, Y.; Kruse, A.C.; Chung, K.Y.; Kobilka, T.S.; Thian, F.S.; Chae, P.S.; Pardon, E.; Calinski, D.; et al. Crystal Structure of the β 2 Adrenergic Receptor–Gs Protein Complex. Nature 2011, 477, 549–555. [Google Scholar] [CrossRef] [PubMed]
- Cherezov, V.; Rosenbaum, D.M.; Hanson, M.A.; Rasmussen, S.G.F.; Thian, F.S.; Kobilka, T.S.; Choi, H.-J.; Kuhn, P.; Weis, W.I.; Kobilka, B.K.; et al. High-Resolution Crystal Structure of an Engineered Human Β2-Adrenergic G Protein–Coupled Receptor. Science 2007, 318, 1258–1265. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Q.; Yang, D.; Wu, M.; Guo, Y.; Guo, W.; Zhong, L.; Cai, X.; Dai, A.; Jang, W.; Shakhnovich, E.I.; et al. Common Activation Mechanism of Class A GPCRs. eLife 2019, 8, e50279. [Google Scholar] [CrossRef]
- Manglik, A.; Kruse, A.C. Structural Basis for G Protein-Coupled Receptor Activation. Biochemistry 2017, 56, 5628–5634. [Google Scholar] [CrossRef]
- Wang, C.; Jiang, Y.; Ma, J.; Wu, H.; Wacker, D.; Katritch, V.; Han, G.W.; Liu, W.; Huang, X.-P.; Vardy, E.; et al. Structural Basis for Molecular Recognition at Serotonin Receptors. Science 2013, 340, 610–614. [Google Scholar] [CrossRef]
- Bermudez, M.; Nguyen, T.N.; Omieczynski, C.; Wolber, G. Strategies for the Discovery of Biased GPCR Ligands. Drug Discov. Today 2019, 24, 1031–1037. [Google Scholar] [CrossRef]
- Weïwer, M.; Xu, Q.; Gale, J.P.; Lewis, M.; Campbell, A.J.; Schroeder, F.A.; Van de Bittner, G.C.; Walk, M.; Amaya, A.; Su, P.; et al. Functionally Biased D2R Antagonists: Targeting the β-Arrestin Pathway to Improve Antipsychotic Treatment. ACS Chem. Biol. 2018, 13, 1038–1047. [Google Scholar] [CrossRef]
- Poissonnet, G.; Parmentier, J.G.; Boutin, J.A.; Goldstein, S. The Emergence of Selective 5-HT 2B Antagonists Structures, Activities and Potential Therapeutic Applications. Mini Rev. Med. Chem. 2004, 4, 325–330. [Google Scholar] [CrossRef]
- Brea, J.; Castro-Palomino, J.; Yeste, S.; Cubero, E.; Parraga, A.; Dominguez, E.; Loza, M.I. Emerging Opportunities and Concerns for Drug Discovery at Serotonin 5-5-HT2B Receptors. Curr. Top. Med. Chem. 2010, 10, 493–503. [Google Scholar] [CrossRef]
- Bonhaus, D.W.; Flippin, L.A.; Greenhouse, R.J.; Jaime, S.; Rocha, C.; Dawson, M.; Natta, K.V.; Chang, L.K.; Pulido-Rios, T.; Webber, A.; et al. RS-127445: A Selective, High Affinity, Orally Bioavailable 5-HT2B Receptor Antagonist. Br. J. Pharmacol. 1999, 127, 1075–1082. [Google Scholar] [CrossRef] [PubMed]
- Porvasnik, S.L.; Germain, S.; Embury, J.; Gannon, K.S.; Jacques, V.; Murray, J.; Byrne, B.J.; Shacham, S.; Al-Mousily, F. PRX-08066, a Novel 5-Hydroxytryptamine Receptor 2B Antagonist, Reduces Monocrotaline-Induced Pulmonary Arterial Hypertension and Right Ventricular Hypertrophy in Rats. J. Pharmacol. Exp. Ther. 2010, 334, 364–372. [Google Scholar] [CrossRef] [PubMed]
- Aurilia, C.; Siller, M.; Arozenius, H.; Palmqvist, N.; Ryde, A.-C.; Klint, C.; Sjödin, A.; Wenglén, C.; Ekström, G.; Seifert, E.; et al. AMAP102: AN ORALLY AVAILABLE SMALL MOLECULAR DRUG WITH BENEFICIAL EFFECTS ON ARTHRITIS AND INFLAMMATORY PAIN. Ann. Rheum. Dis. 2009, 68, 585. [Google Scholar]
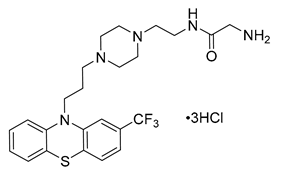
- Palmqvist, N.; Siller, M.; Klint, C.; Sjödin, A. A Human and Animal Model-Based Approach to Investigating the Anti-Inflammatory Profile and Potential of the 5-HT2B Receptor Antagonist AM1030. J. Inflamm. 2016, 13, 20. [Google Scholar] [CrossRef] [PubMed]
- Zai, H.; Ando, A.; Sato, N. Sa1762—First-In-Human Study of the Novel 5-HT2B Receptor Antagonist, Rq-00310941, Demonstrated Safety and Tolerability Both in Healthy Subjects and Ulcerative Colitis Patients, with Improvement of a Sense of Abnormal Bowel Movement. Gastroenterology 2018, 154, S-385. [Google Scholar] [CrossRef]
- Pettersson, L. Novel 5-Ht2 Antagonists. WO2016207231, 29 December 2016. [Google Scholar]
- Löfdahl, A.; Wenglén, C.; Rydell-Törmänen, K.; Westergren-Thorsson, G.; Larsson-Callerfelt, A.-K. Effects of 5-Hydroxytryptamine Class 2 Receptor Antagonists on Bronchoconstriction and Pulmonary Remodeling Processes. Am. J. Pathol. 2018, 188, 1113–1119. [Google Scholar] [CrossRef]
- Wenglén, C.; Pettersson, L.; Arozenius, H.; Ekström, G. P012 A Novel Highly Selective 5-HT2B Receptor Antagonist Reduces Myofibroblast Differentiation and Extracellular Matrix Deposition in Models of Lung Fibrosis. QJM Int. J. Med. 2016, 109, S25–S26. [Google Scholar] [CrossRef]
- Huang, N.; Kalyanaraman, C.; Irwin, J.J.; Jacobson, M.P. Physics-Based Scoring of Protein−Ligand Complexes: Enrichment of Known Inhibitors in Large-Scale Virtual Screening. J. Chem. Inf. Model. 2006, 46, 243–253. [Google Scholar] [CrossRef]
- Zhou, Y.; Wang, Y.; Li, P.; Huang, X.-P.; Qi, X.; Du, Y.; Huang, N. Exploring Halogen Bonds in 5-Hydroxytryptamine 2B Receptor–Ligand Interactions. ACS Med. Chem. Lett. 2018, 9, 1019–1024. [Google Scholar] [CrossRef]
- Moritomo, A.; Yamada, H.; Watanabe, T.; Itahana, H.; Akuzawa, S.; Okada, M.; Ohta, M. Synthesis and Structure–Activity Relationships of New Carbonyl Guanidine Derivatives as Novel Dual 5-HT2B and 5-HT7 Receptor Antagonists. Bioorg. Med. Chem. 2013, 21, 7841–7852. [Google Scholar] [CrossRef]
- Moritomo, A.; Yamada, H.; Watanabe, T.; Itahana, H.; Koga, Y.; Akuzawa, S.; Okada, M. Synthesis and Structure–Activity Relationships of New Carbonyl Guanidine Derivatives as Novel Dual 5-HT2B and 5-HT7 Receptor Antagonists. Part 2. Bioorg. Med. Chem. 2014, 22, 4323–4337. [Google Scholar] [CrossRef] [PubMed]
- Moritomo, A.; Yamada, H.; Matsuzawa-Nomura, T.; Watanabe, T.; Itahana, H.; Oku, M.; Akuzawa, S.; Okada, M. Synthesis and Pharmacological Evaluation of Optically Pure, Novel Carbonyl Guanidine Derivatives as Dual 5-HT2B and 5-HT7 Receptor Antagonists. Bioorg. Med. Chem. 2014, 22, 6026–6038. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.A.; Zaidi, S.A.; Zhang, Y. 5-Hydroxy-2-(2-Phenylethyl)Chromone (5-HPEC): A Novel Non-Nitrogenous Ligand for 5-HT2B Receptor. Bioorg. Med. Chem. Lett. 2014, 24, 1489–1492. [Google Scholar] [CrossRef] [PubMed]
- Williams, D.A.; Zaidi, S.A.; Zhang, Y. Synthesis and Characterization of 5-Hydroxy-2-(2-Phenylethyl)Chromone (5-HPEC) and Its Analogues as Non-Nitrogenous 5-HT2B Ligands. J. Nat. Prod. 2015, 78, 1859–1867. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Truss, M.; Pagare, P.P.; Essandoh, M.A.; Zhang, Y.; Williams, D.A. Structure Activity Relationship Exploration of 5-Hydroxy-2-(3-Phenylpropyl)Chromones as a Unique 5-HT2B Receptor Antagonist Scaffold. Bioorg. Med. Chem. Lett. 2020, 30, 127511. [Google Scholar] [CrossRef] [PubMed]
- Kapadia, N.; Harding, W. Aporphine Alkaloids as Ligands for Serotonin Receptors. Med. Chem. 2016, 6, 241–249. [Google Scholar] [CrossRef]
- Kapadia, N.; Harding, W.W. C4 Phenyl Aporphines with Selective H5-HT2B Receptor Affinity. Bioorg. Med. Chem. Lett. 2015, 25, 3451–3454. [Google Scholar] [CrossRef]
- Ponnala, S.; Kapadia, N.; Harding, W.W. Identification of Tris-(Phenylalkyl)Amines as New Selective H5-HT2B Receptor Antagonists. MedChemComm 2015, 6, 601–605. [Google Scholar] [CrossRef]
- Gabr, M.T.; Abdel-Raziq, M.S. Pharmacophore-Based Tailoring of Biphenyl Amide Derivatives as Selective 5-Hydroxytryptamine 2B Receptor Antagonists. MedChemComm 2018, 9, 1069–1075. [Google Scholar] [CrossRef]
- Moss, N.; Choi, Y.; Cogan, D.; Flegg, A.; Kahrs, A.; Loke, P.; Meyn, O.; Nagaraja, R.; Napier, S.; Parker, A.; et al. A New Class of 5-HT2B Antagonists Possesses Favorable Potency, Selectivity, and Rat Pharmacokinetic Properties. Bioorg. Med. Chem. Lett. 2009, 19, 2206–2210. [Google Scholar] [CrossRef]
- Porter, M.R.; Xiao, H.; Wang, J.; Smith, S.B.; Topczewski, J.J. 3-Amino-Chromanes and Tetrahydroquinolines as Selective 5-HT2B, 5-HT7, or Σ1 Receptor Ligands. ACS Med. Chem. Lett. 2019, 10, 1436–1442. [Google Scholar] [CrossRef] [PubMed]
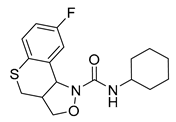
- Kwon, Y.J.; Saubern, S.; Macdonald, J.M.; Huang, X.-P.; Setola, V.; Roth, B.L. N-Tetrahydrothiochromenoisoxazole-1-Carboxamides as Selective Antagonists of Cloned Human 5-HT2B. Bioorg. Med. Chem. Lett. 2010, 20, 5488–5490. [Google Scholar] [CrossRef] [PubMed]
- Zopf, D.A.; das Neves, L.A.A.; Nikula, K.J.; Huang, J.; Senese, P.B.; Gralinski, M.R. C-122, a Novel Antagonist of Serotonin Receptor 5-HT2B, Prevents Monocrotaline-Induced Pulmonary Arterial Hypertension in Rats. Eur. J. Pharmacol. 2011, 670, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, T.; Hauser, N.; Reker, D.; Reutlinger, M.; Wunderlin, T.; Hamon, J.; Koch, G.; Schneider, G. Multidimensional De Novo Design Reveals 5-HT2B Receptor-Selective Ligands. Angew. Chem. 2015, 127, 1571–1575. [Google Scholar] [CrossRef]
- Tosh, D.K.; Ciancetta, A.; Warnick, E.; Crane, S.; Gao, Z.-G.; Jacobson, K.A. Structure-Based Scaffold Repurposing for G Protein-Coupled Receptors: Transformation of Adenosine Derivatives into 5HT2B/5HT2C Serotonin Receptor Antagonists. J. Med. Chem. 2016, 59, 11006–11026. [Google Scholar] [CrossRef]
- Rataj, K.; Kelemen, Á.A.; Brea, J.; Loza, M.I.; Bojarski, A.J.; Keserű, G.M. Fingerprint-Based Machine Learning Approach to Identify Potent and Selective 5-HT2BR Ligands. Molecules 2018, 23, 1137. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, S.; Hu, Q.; Gao, S.; Ma, X.; Zhang, W.; Shen, Y.; Chen, F.; Lai, L.; Pei, J. CavityPlus: A Web Server for Protein Cavity Detection with Pharmacophore Modelling, Allosteric Site Identification and Covalent Ligand Binding Ability Prediction. Nucleic Acids Res. 2018, 46, W374–W379. [Google Scholar] [CrossRef]
- Kim, K.; Che, T.; Panova, O.; DiBerto, J.F.; Lyu, J.; Krumm, B.E.; Wacker, D.; Robertson, M.J.; Seven, A.B.; Nichols, D.E.; et al. Structure of a Hallucinogen-Activated Gq-Coupled 5-HT2A Serotonin Receptor. Cell 2020, 182, 1574–1588.e19. [Google Scholar] [CrossRef]
- Kimura, K.T.; Asada, H.; Inoue, A.; Kadji, F.M.N.; Im, D.; Mori, C.; Arakawa, T.; Hirata, K.; Nomura, Y.; Nomura, N.; et al. Structures of the 5-HT 2A Receptor in Complex with the Antipsychotics Risperidone and Zotepine. Nat. Struct. Mol. Biol. 2019, 26, 121–128. [Google Scholar] [CrossRef]
- Peng, Y.; McCorvy, J.D.; Harpsøe, K.; Lansu, K.; Yuan, S.; Popov, P.; Qu, L.; Pu, M.; Che, T.; Nikolajsen, L.F.; et al. 5-HT2C Receptor Structures Reveal the Structural Basis of GPCR Polypharmacology. Cell 2018, 172, 719–730.e14. [Google Scholar] [CrossRef]
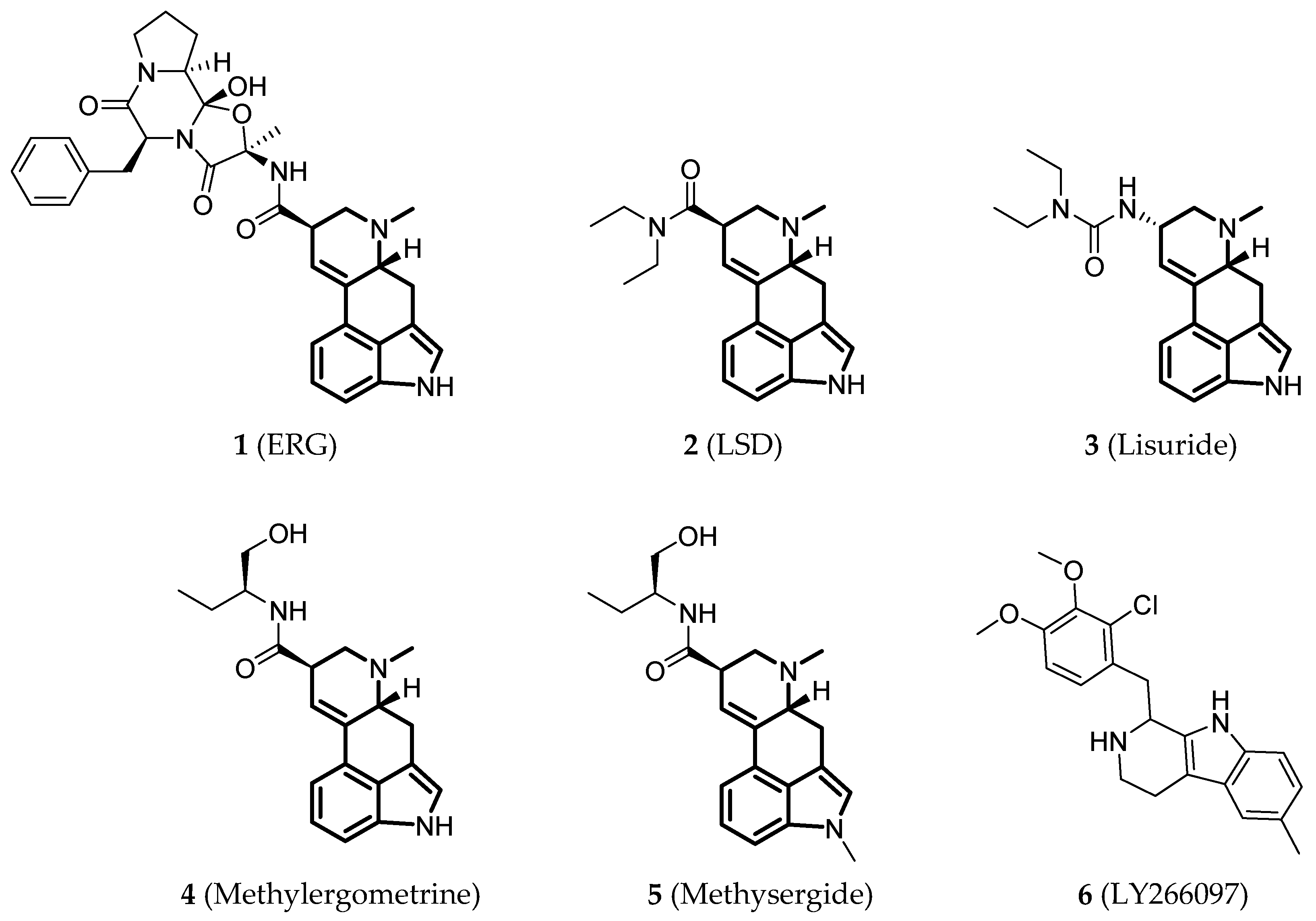
| PDB ID | Ligand | State | Ligand Function | Resolution (Å) |
|---|---|---|---|---|
| 4IB4 [110] | ERG | Intermediate | β-arrestin-biased agonist | 2.7 |
| 4NC3 [111] | ERG | Intermediate | β-arrestin-biased agonist | 2.8 |
| 5TVN [112] | LSD | Intermediate | β-arrestin-biased agonist | 2.9 |
| 5TUD [113] | ERG + antibody Fab fragment | Active | β-arrestin-biased agonist | 3.0 |
| 6DRX [114] | Lisuride | Intermediate | Antagonist | 3.1 |
| 6DRY [114] | Methylergometrine | Intermediate | Agonist | 2.9 |
| 6DRZ [114] | Methysergide | Intermediate | Antagonist | 3.1 |
| 6DS0 [114] | LY266097 | Intermediate | Antagonist (Gq partial agonist, β-arrestin2 antagonist) | 3.2 |
| Compound Name | Structure | Activity | Highest Clinical State | Indication |
|---|---|---|---|---|
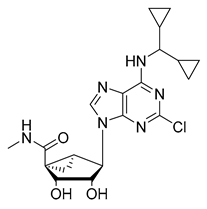
| 7 [125] (MT-500, RS-127445) | 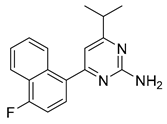 | Ki = 0.3 nM | Phase 1 | Migraine |
| 8 [126] (PRX-08066) | 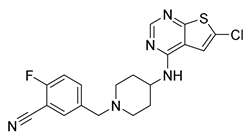 | Ki ~3.4 nM | Phase 2 | PH, COPD |
| 9 [79] (BF-1) | 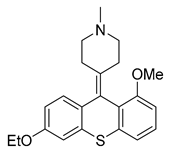 | Ki = 2.7 nM | Phase 1 | Migraine |
| 10 [127] (AMAP102) | Undisclosed | Undisclosed | Phase 2 | Osteoarthritis |
| 11 [128] (AM-1030) | Aminoguanidine derivative | Ki = 330 nM | Phase 2 | AD |
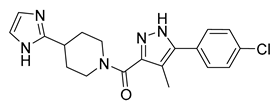
| 12 [64] (RQ-00310941) | 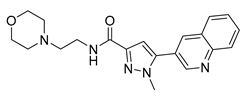 | Ki = 2.0 nM | Phase 1 | UC |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Q.; Zhou, Y.; Huang, J.; Huang, N. Structure, Function, and Pharmaceutical Ligands of 5-Hydroxytryptamine 2B Receptor. Pharmaceuticals 2021, 14, 76. https://doi.org/10.3390/ph14020076
Wang Q, Zhou Y, Huang J, Huang N. Structure, Function, and Pharmaceutical Ligands of 5-Hydroxytryptamine 2B Receptor. Pharmaceuticals. 2021; 14(2):76. https://doi.org/10.3390/ph14020076
Chicago/Turabian StyleWang, Qing, Yu Zhou, Jianhui Huang, and Niu Huang. 2021. "Structure, Function, and Pharmaceutical Ligands of 5-Hydroxytryptamine 2B Receptor" Pharmaceuticals 14, no. 2: 76. https://doi.org/10.3390/ph14020076
APA StyleWang, Q., Zhou, Y., Huang, J., & Huang, N. (2021). Structure, Function, and Pharmaceutical Ligands of 5-Hydroxytryptamine 2B Receptor. Pharmaceuticals, 14(2), 76. https://doi.org/10.3390/ph14020076