Status and Challenges of Plant-Anticancer Compounds in Cancer Treatment
Abstract
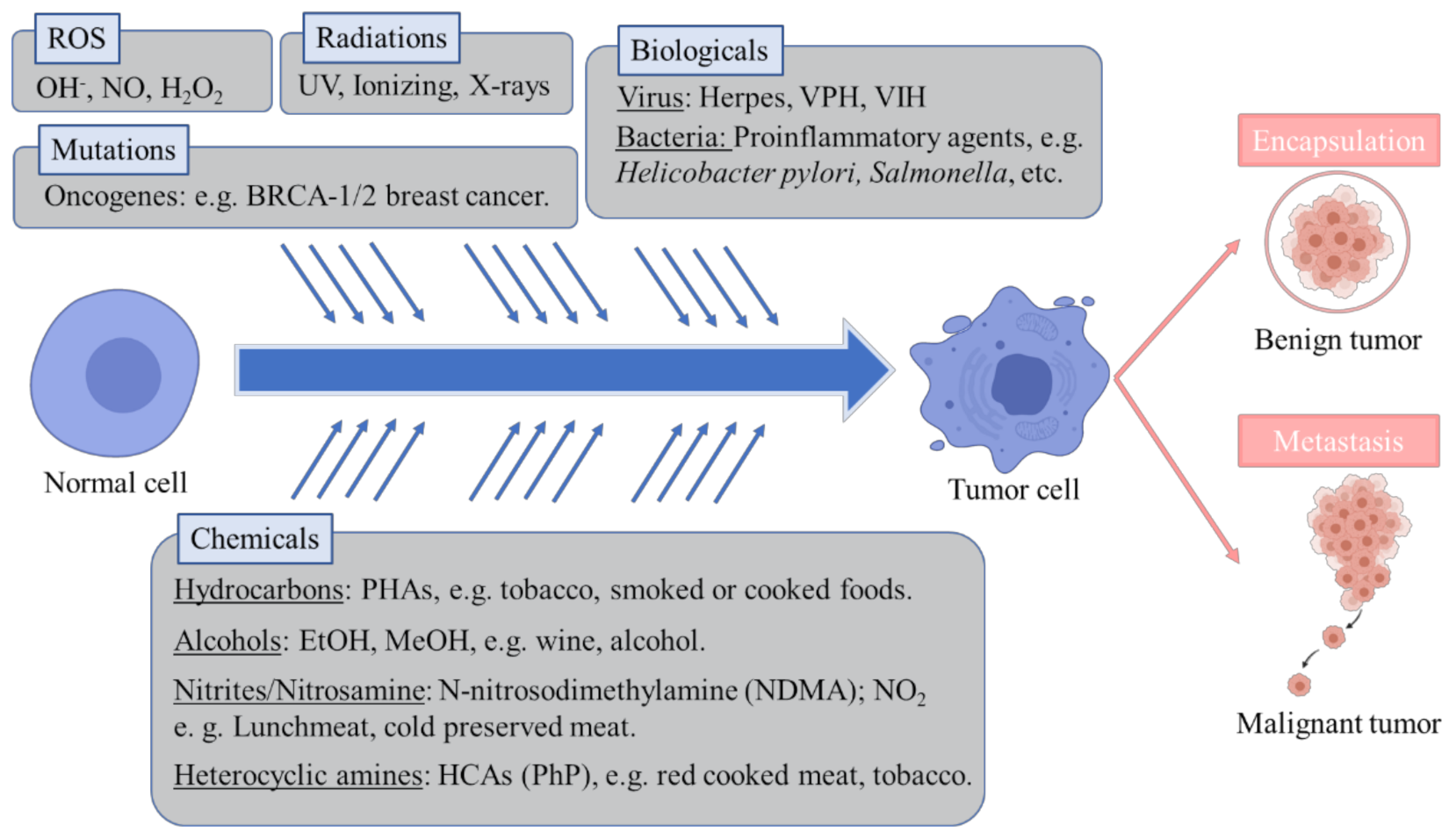
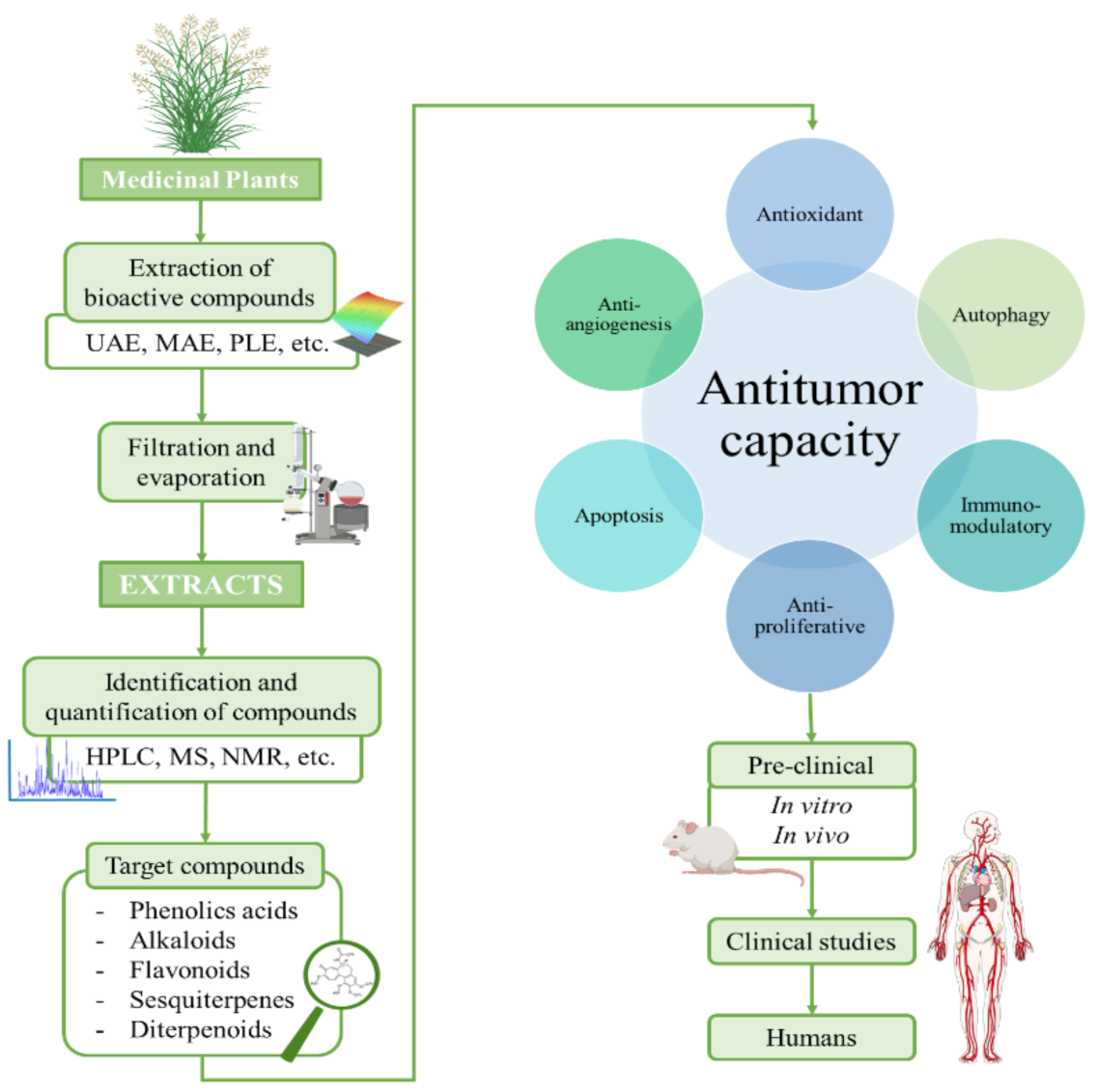
1. Introduction
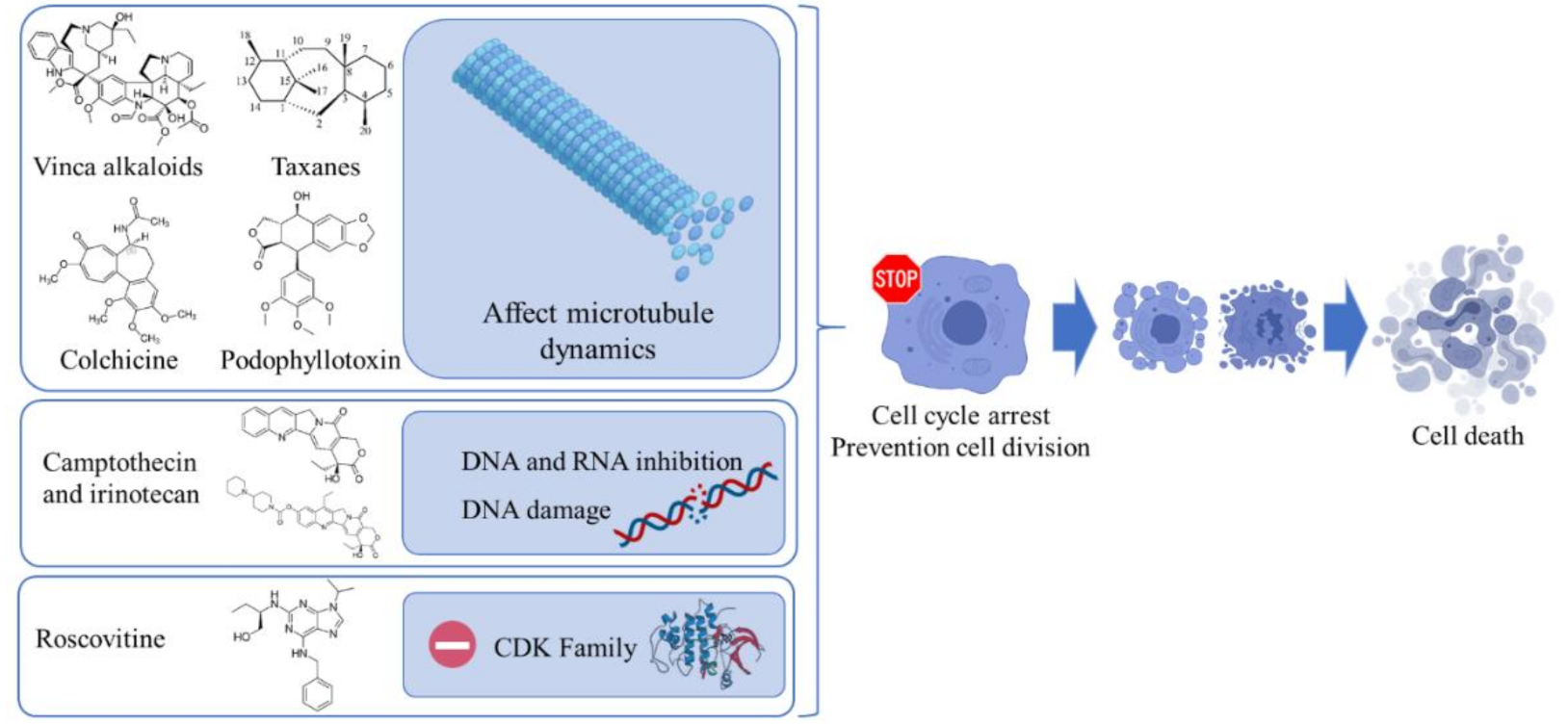
2. Phytochemicals Currently Used in Cancer Therapy
2.1. Vinca Alkaloids
2.2. Paclitaxel
2.3. Camptothecin and Irinotecan
2.4. Podophyllotoxin and Analogues
2.5. Roscovitine
3. Compounds in Pre-Clinical and Clinical Stages
3.1. Colchicine Derivates
3.2. Sulforaphane
3.3. Phenolic Compounds
3.3.1. Resveratrol
3.3.2. Curcumin
3.3.3. Quercetin
3.3.4. Gingerol
3.3.5. Kaempferol
3.4. Other Plant Derived Compounds
4. Current Challenges of Anticancer Phytochemicals
4.1. Limited Solubility and Poor Absortion
4.2. Combination of Phytochemicals with Conventional Chemotherapeutics
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- International Agency for Research on Cancer Global Cancer Observatory. Available online: https://gco.iarc.fr/today (accessed on 31 December 2020).
- Valent, P.; Bonnet, D.; De Maria, R.; Lapidot, T.; Copland, M.; Melo, J.V.; Chomienne, C.; Ishikawa, F.; Schuringa, J.J.; Stassi, G.; et al. Cancer stem cell definitions and terminology: The devil is in the details. Nat. Rev. Cancer 2012, 12, 767–775. [Google Scholar] [CrossRef]
- Fernald, K.; Kurokawa, M. Evading apoptosis in cancer. Trends Cell Biol. 2013, 23, 620–633. [Google Scholar] [CrossRef]
- Stewart, B.W.; Wild, C.P. World Cancer Report 2014; WHO Press; World Health Organization: Geneva, Switzerland, 2014; ISBN 978-92-832-0443-5. [Google Scholar]
- Narayanan, D.L.; Saladi, R.N.; Fox, J.L. Ultraviolet radiation and skin cancer. Int. J. Dermatol. 2010, 49, 978–986. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer. Biological Agents, Volume 100B: A Review on Human Carcinogens; IARC: Lyon, France, 2012. [Google Scholar]
- Zhao, L.H.; Liu, X.; Yan, H.X.; Li, W.Y.; Zeng, X.; Yang, Y.; Zhao, J.; Liu, S.P.; Zhuang, X.H.; Lin, C.; et al. Genomic and oncogenic preference of HBV integration in hepatocellular carcinoma. Nat. Commun. 2016, 7, 1–10. [Google Scholar] [CrossRef]
- Martin, D.; Gutkind, J.S. Human tumor-associated viruses and new insights into the molecular mechanisms of cancer. Oncogene 2008, 27, S31–S42. [Google Scholar] [CrossRef] [PubMed]
- Hansen, A.; Henderson, S.; Lagos, D.; Nikitenko, L.; Coulter, E.; Roberts, S.; Gratrix, F.; Plaisance, K.; Renne, R.; Bower, M.; et al. KSHV-encoded miRNAs target MAF to induce endothelial cell reprogramming. Genes Dev. 2010, 24, 195–205. [Google Scholar] [CrossRef]
- Wen, S.; Moss, S.F. Helicobacter pylori virulence factors in gastric carcinogenesis. Cancer Lett. 2009, 282, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Scanu, T.; Spaapen, R.M.; Bakker, J.M.; Pratap, C.B.; Wu, L.E.; Hofland, I.; Broeks, A.; Shukla, V.K.; Kumar, M.; Janssen, H.; et al. Salmonella Manipulation of Host Signaling Pathways Provokes Cellular Transformation Associated with Gallbladder Carcinoma. Cell Host Microbe 2015, 17, 763–774. [Google Scholar] [CrossRef] [PubMed]
- Elsland, D.; Neefjes, J. Bacterial infections and cancer. EMBO Rep. 2018, 19, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Mesri, E.A.; Cesarman, E.; Boshoff, C. Kaposi’s sarcoma and its associated herpesvirus. Nat. Rev. Cancer 2010, 10, 707–719. [Google Scholar] [CrossRef]
- Reuter, S.; Gupta, S.C.; Chaturvedi, M.M.; Aggarwal, B.B. Oxidative stress, inflammation, and cancer: How are they linked? Free Radic. Biol. Med. 2010, 49, 1603–1616. [Google Scholar] [CrossRef]
- Moloney, J.N.; Cotter, T.G. ROS signalling in the biology of cancer. Semin. Cell Dev. Biol. 2018, 80, 50–64. [Google Scholar] [CrossRef]
- Crusz, S.M.; Balkwill, F.R. Inflammation and cancer: Advances and new agents. Nat. Rev. Clin. Oncol. 2015, 12, 584–596. [Google Scholar] [CrossRef] [PubMed]
- Masferrer, J.L.; Leahy, K.M.; Koki, A.T.; Zweifel, B.S.; Settle, S.L.; Woerner, B.M.; Edwards, D.A.; Flickinger, A.G.; Moore, R.J.; Seibert, K. Antiangiogenic and antitumor activities of cyclooxygenase-2 inhibitors. Cancer Res. 2000, 60, 1306–1311. [Google Scholar] [PubMed]
- Qu, X.; Tang, Y.; Hua, S. Immunological approaches towards cancer and inflammation: A cross talk. Front. Immunol. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Tao, S.; Liu, J.; Huang, Y.; Chen, H.; Li, W.; Zhang, Y.; Chen, Y.; Su, S.; Lin, N.; et al. Global lung cancer risk from PAH exposure highly depends on emission sources and individual susceptibility. Sci. Rep. 2014, 4, 1–8. [Google Scholar] [CrossRef]
- Bansal, V.; Kim, K.H. Review of PAH contamination in food products and their health hazards. Environ. Int. 2015, 84, 26–38. [Google Scholar] [CrossRef] [PubMed]
- Sugimura, T.; Wakabayashi, K.; Nakagama, H.; Nagao, M. Heterocyclic amines: Mutagens/carcinogens produced during cooking of meat and fish. Gann Monogr. Cancer Res. 2004, 52, 71–96. [Google Scholar] [CrossRef]
- Puangsombat, K.; Gadgil, P.; Houser, T.A.; Hunt, M.C.; Smith, J.S. Occurrence of heterocyclic amines in cooked meat products. Meat Sci. 2012, 90, 739–746. [Google Scholar] [CrossRef] [PubMed]
- WHO Report on the Global Tobacco Epidemic, 2011: Warning About the Dangers of Tobacco, 3rd ed.; World Health Organization: Geneva, Switzerland, 2011.
- Song, P.; Wu, L.; Guan, W. Dietary nitrates, nitrites, and nitrosamines intake and the risk of gastric cancer: A meta-analysis. Nutrients 2015, 7, 9872. [Google Scholar] [CrossRef]
- Praud, D.; Rota, M.; Rehm, J.; Shield, K.; Zatoński, W.; Hashibe, M.; La Vecchia, C.; Boffetta, P. Cancer incidence and mortality attributable to alcohol consumption. Int. J. Cancer 2016, 138, 1380–1387. [Google Scholar] [CrossRef] [PubMed]
- Litzenburger, U.M.; Buenrostro, J.D.; Wu, B.; Shen, Y.; Sheffield, N.C.; Kathiria, A.; Greenleaf, W.J.; Chang, H.Y. Single-cell epigenomic variability reveals functional cancer heterogeneity. Genome Biol. 2017, 18, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.Y.; Cheng, Y.J.; Lei, Q.; Zhang, A.Q.; Zhang, X.Z. Combinational strategy for high-performance cancer chemotherapy. Biomaterials 2018, 171, 178–197. [Google Scholar] [CrossRef] [PubMed]
- Gautam, L.; Jain, A.; Shrivastava, P.; Vyas, S.; Vyas, S.P. Emergence of novel targeting systems and conventional therapies for effective cancer treatment. In Nano Drug Delivery Strategies for the Treatment of Cancers; Elsevier: Amsterdam, the Netherlands, 2021; pp. 1–35. [Google Scholar]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse effects of cancer chemotherapy: Anything new to improve tolerance and reduce sequelae? Front. Pharmacol. 2018, 9, 1–3. [Google Scholar] [CrossRef]
- Glass, C.K.; Mitchell, R.N. Winning the battle, but losing the war: Mechanisms and morphology of cancer-therapy-associated cardiovascular toxicity. Cardiovasc. Pathol. 2017, 30, 55–63. [Google Scholar] [CrossRef]
- Lichota, A.; Gwozdzinski, K. Anticancer Activity of Natural Compounds from Plant and Marine Environment. Int. J. Mol. Sci. 2018, 19, 3533. [Google Scholar] [CrossRef]
- Babaei, G.; Aliarab, A.; Abroon, S.; Rasmi, Y.; Aziz, S.G.G. Application of sesquiterpene lactone: A new promising way for cancer therapy based on anticancer activity. Biomed. Pharmacother. 2018, 106, 239–246. [Google Scholar] [CrossRef]
- Garcia-Oliveira, P.; Fraga-Corral, M.; Pereira, A.G.; Lourenço-Lopes, C.; Jimenez-Lopez, C.; Prieto, M.A.; Simal-Gandara, J. Scientific basis for the industrialization of traditionally used plants of the Rosaceae family. Food Chem. 2020, 330, 127197. [Google Scholar] [CrossRef]
- Lopes, C.M.; Dourado, A.; Oliveira, R. Phytotherapy and Nutritional Supplements on Breast Cancer. Biomed Res. Int. 2017, 2017, 1–42. [Google Scholar] [CrossRef]
- Choudhari, A.S.; Mandave, P.C.; Deshpande, M.; Ranjekar, P.; Prakash, O. Phytochemicals in cancer treatment: From preclinical studies to clinical practice. Front. Pharmacol. 2020, 10, 1–17. [Google Scholar] [CrossRef]
- Iqbal, J.; Abbasi, B.A.; Mahmood, T.; Kanwal, S.; Ali, B.; Shah, S.A.; Khalil, A.T. Plant-derived anticancer agents: A green anticancer approach. Asian Pac. J. Trop. Biomed. 2017, 7, 1129–1150. [Google Scholar] [CrossRef]
- Mao, Q.Q.; Xu, X.Y.; Shang, A.; Gan, R.Y.; Wu, D.T.; Atanasov, A.G.; Li, H. Bin Phytochemicals for the prevention and treatment of gastric cancer: Effects and mechanisms. Int. J. Mol. Sci. 2020, 21, 570. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Blanco, S.; Fernández, J.; Gutiérrez-del-Río, I.; Villar, C.J.; Lombó, F. New insights toward colorectal cancer chemotherapy using natural bioactive compounds. Front. Pharmacol. 2017, 8, 1–22. [Google Scholar] [CrossRef] [PubMed]
- Clardy, J.; Walsh, C. Lessons from natural molecules. Nature 2004, 432, 829–837. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs from 1981 to 2014. J. Nat. Prod. 2016, 79, 629–661. [Google Scholar] [CrossRef]
- Desai, A.; Qazi, G.; Ganju, R.; El-Tamer, M.; Singh, J.; Saxena, A.; Bedi, Y.; Taneja, S.; Bhat, H. Medicinal Plants and Cancer Chemoprevention. Curr. Drug Metab. 2008, 9, 581–591. [Google Scholar] [CrossRef]
- Moraes, D.F.C.; de Mesquita, L.S.S.; do Amaral, F.M.M.; de Sousa Ribeiro, M.N.; Malik, S. Anticancer drugs from plants. In Biotechnology and Production of Anti-Cancer Compounds; Springer International Publishing: Berlin/Heidelberg, Germany, 2017; pp. 121–142. [Google Scholar]
- Greenwell, M.; Rahman, P.K.S. Medicinal Plants: Their Use in Anticancer Treatment. Int. J. Pharm. Sci. Res. 2015, 6, 4103–4112. [Google Scholar] [CrossRef]
- FDA Food and Drud Administration Approved Drug Products—Orange Book. Available online: https://www.accessdata.fda.gov/scripts/cder/ob/index.cfm (accessed on 31 December 2020).
- Chu, I.-H.; Bodnar, J.A.; Bowman, R.N.; White, E.L. Determination of Vincristine and Vinblastine in Catharanthus roseus Plants by High Performance Liquid Chromatography/Electrospray Ionization Mass Spectrometry. J. Liq. Chromatogr. Relat. Technol. 1997, 20, 1159–1174. [Google Scholar] [CrossRef]
- Lee, C.-T.; Huang, Y.-W.; Yang, C.-H.; Huang, K.-S. Drug Delivery Systems and Combination Therapy by Using Vinca Alkaloids. Curr. Top. Med. Chem. 2015, 15, 1491–1500. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, H.; Colby, D.A.; Seto, S.; Va, P.; Tam, A.; Kakei, H.; Rayl, T.J.; Hwang, I.; Boger, D.L. Total synthesis of vinblastine, vincristine, related natural products, and key structural analogues. J. Am. Chem. Soc. 2009, 131, 4904–4916. [Google Scholar] [CrossRef] [PubMed]
- Martino, E.; Casamassima, G.; Castiglione, S.; Cellupica, E.; Pantalone, S.; Papagni, F.; Rui, M.; Siciliano, A.M.; Collina, S. Vinca alkaloids and analogues as anti-cancer agents: Looking back, peering ahead. Bioorganic Med. Chem. Lett. 2018, 28, 2816–2826. [Google Scholar] [CrossRef] [PubMed]
- Jordan, M.A.; Himes, R.H.; Wilson, L. Comparison of the effects of vinblastine, vincristine, vindesine, and vinepidine on microtubule dynamics and cell proliferation in vitro. Cancer Res. 1985, 45, 2741–2747. [Google Scholar]
- Lobert, S.; Vulevic, B.; Correia, J.J. Interaction of Vinca Alkaloids with Tubulin: A Comparison of Vinblastine, Vincristine, and Vinorelbine. Biochemistry 1996, 35, 6806–6814. [Google Scholar] [CrossRef]
- Sertel, S.; Fu, Y.; Zu, Y.; Rebacz, B.; Konkimalla, B.; Plinkert, P.K.; Krämer, A.; Gertsch, J.; Efferth, T. Molecular docking and pharmacogenomics of Vinca alkaloids and their monomeric precursors, vindoline and catharanthine. Biochem. Pharmacol. 2011, 81, 723–735. [Google Scholar] [CrossRef] [PubMed]
- Taher, M.A.; Foundation, I.B. Vinca alkaloid—The second most used alkaloid for cancer treatment—A review. Int. J. Physiol. Nutr. Phys. Educ. 2017; 2, 723–727. [Google Scholar]
- Almagro, L.; Fernández-Pérez, F.; Pedreño, M.A. Indole Alkaloids from Catharanthus roseus: Bioproduction and Their Effect on Human Health. Molecules 2015, 20, 2973–3000. [Google Scholar] [CrossRef] [PubMed]
- Seca, A.M.L.; Pinto, D.C.G.A. Plant Secondary Metabolites as Anticancer Agents: Successes in Clinical Trials and Therapeutic Application. Int. J. Mol. Sci. 2018, 19, 263. [Google Scholar] [CrossRef]
- Kuruvilla, J.; Song, K.; Mollee, P.; Panzarella, T.; McCrae, J.; Nagy, T.; Crump, M.; Keating, A. A phase II study of thalidomide and vinblastine for palliative patients with Hodgkin’s lymphoma. Hematology 2006, 11, 25–29. [Google Scholar] [CrossRef] [PubMed]
- Young, S.D.; Whissell, M.; Noble, J.C.S.; Cano, P.O.; Lopez, P.G.; Germond, C.J. Phase II clinical trial results involving treatment with low-dose daily oral cyclophosphamide, weekly vinblastine, and rofecoxib in patients with advanced solid tumors. Clin. Cancer Res. 2006, 12, 3092–3098. [Google Scholar] [CrossRef] [PubMed]
- Kitamura, H.; Tsukamoto, T.; Shibata, T.; Masumori, N.; Fujimoto, H.; Hirao, Y.; Fujimoto, K.; Kitamura, Y.; Tomita, Y.; Tobisu, K.; et al. Randomised phase III study of neoadjuvant chemotherapy with methotrexate, doxorubicin, vinblastine and cisplatin followed by radical cystectomy compared with radical cystectomy alone for muscle-invasive bladder cancer: Japan Clinical Oncology Group Study. Ann. Oncol. 2014, 25, 1192–1198. [Google Scholar] [CrossRef]
- Gerullis, H.; Wawroschek, F.; Köhne, C.H.; Ecke, T.H. Vinflunine in the treatment of advanced urothelial cancer: Clinical evidence and experience. Ther. Adv. Urol. 2017, 9, 28–35. [Google Scholar] [CrossRef]
- PubChem Compound Summary for CID 16760674, Taxol®. Available online: https://pubchem.ncbi.nlm.nih.gov/compound/16760674 (accessed on 30 December 2020).
- Bergman, M.E.; Davis, B.; Phillips, M.A. Medically useful plant terpenoids: Biosynthesis, occurrence, and mechanism of action. Molecules 2019, 24, 3961. [Google Scholar] [CrossRef]
- Bernabeu, E.; Cagel, M.; Lagomarsino, E.; Moretton, M.; Chiappetta, D.A. Paclitaxel: What has been done and the challenges remain ahead. Int. J. Pharm. 2017, 526, 474–495. [Google Scholar] [CrossRef]
- Li, F.; Jiang, T.; Li, Q.; Ling, X. Camptothecin (CPT) and its derivatives are known to target topoisomerase I (Top1) as their mechanism of action: Did we miss something in CPT analogue molecular targets for treating human disease such as cancer? Am. J. Cancer Res. 2017, 7, 2350–2394. [Google Scholar]
- Jones, R. Cancer therapies utilizing the camptothecins: A Review of the in vivo literature. Mol. Pharm. 2010, 7, 307–349. [Google Scholar] [CrossRef]
- Kciuk, M.; Marciniak, B.; Kontek, R. Irinotecan—Still an important player in cancer chemotherapy: A comprehensive overview. Int. J. Mol. Sci. 2020, 21, 4919. [Google Scholar] [CrossRef] [PubMed]
- Fujita, K.I.; Sasaki, Y. Optimization of cancer chemotherapy on the basis of pharmacokinetics and pharmacodynamics: From patients enrolled in clinical trials to those in the “real world”. Drug Metab. Pharmacokinet. 2014, 29, 20–28. [Google Scholar] [CrossRef]
- Wahid, M. Camptothecin and its analogs antitumor activity by poisoning topoisomerase I, their structure activity relationship and clinical development perspective of analogs. J. App. Pharm. 2016; 6, 286–295. [Google Scholar]
- Zhang, X.; Rakesh, K.P.; Shantharam, C.S.; Manukumar, H.M.; Asiri, A.M.; Marwani, H.M.; Qin, H. Bioorganic & Medicinal Chemistry Podophyllotoxin derivatives as an excellent anticancer aspirant for future chemotherapy: A key current imminent needs. Bioorg. Med. Chem. 2018, 26, 340–355. [Google Scholar] [CrossRef] [PubMed]
- Carolina, N.; Hill, C.; Carolina, N. Recent progress on C-4-modified podophyllotoxin analogs as potent antitumor agents. Med. Res. Rev. 2016, 35, 1–62. [Google Scholar] [CrossRef]
- Ardalani, H.; Avan, A.; Ghayour-mobarhan, M. Podophyllotoxin: A novel potential natural anticancer agent. Avicenna J. Phytomed. 2017, 7, 285–294. [Google Scholar]
- Cicenas, J.; Kalyan, K.; Sorokinas, A.; Stankunas, E.; Levy, J.; Meskinyte, I.; Stankevicius, V.; Kaupinis, A.; Valius, M. Roscovitine in cancer and other diseases. Ann. Transl. Med. 2015, 3, 135. [Google Scholar]
- Delehouzé, C.; Godl, K.; Loaëc, N.; Bruyère, C.; Desban, N.; Oumata, N.; Galons, H.; Roumeliotis, T.I.; Giannopoulou, E.G.; Grenet, J.; et al. CDK/CK1 inhibitors roscovitine and CR8 downregulate amplified MYCN in neuroblastoma cells. Oncogene 2014, 33, 5675–5687. [Google Scholar] [CrossRef]
- Khalil, H.S.; Mitev, V.; Vlaykova, T.; Cavicchi, L.; Zhelev, N. Discovery and development of Seliciclib. How systems biology approaches can lead to better drug performance. J. Biotechnol. 2015, 202, 40–49. [Google Scholar]
- Theodoropoulou, M.; Reincke, M. Tumor-Directed Therapeutic Targets in Cushing Disease. J. Clin. Endocrinol. Metab. 2019, 104, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Gary, C.; Hajek, M.; Biktasova, A.; Bellinger, G.; Wendell, G. Selective antitumor activity of roscovitine in head and neck cancer. Oncotarget 2016, 7, 38598. [Google Scholar] [CrossRef] [PubMed]
- Cipolla, B.G.; Mandron, E.; Marc Lefort, J.; Coadou, Y.; Della Negra, E.; Corbel, L.; Le Scodan, R.; Rahmene Azzouzi, A.; Mottet, N. Effect of sulforaphane in men with biochemical recurrence after radical prostatectomy. Cancer Prev. Res. 2015, 8, 712–719. [Google Scholar] [CrossRef] [PubMed]
- Alumkal, J.J.; Slottke, R.; Schwartzman, J.; Cherala, G.; Munar, M.; Graff, J.N.; Beer, T.M.; Ryan, C.W.; Koop, D.R.; Gibbs, A.; et al. A phase II study of sulforaphane-rich broccoli sprout extracts in men with recurrent prostate cancer. Investig. New Drugs 2015, 33, 480–489. [Google Scholar] [CrossRef]
- Rong, Y.; Huang, L.; Yi, K.; Chen, H.; Liu, S.; Zhang, W.; Yuan, C.; Song, X.; Wang, F. Co-administration of sulforaphane and doxorubicin attenuates breast cancer growth by preventing the accumulation of myeloid-derived suppressor cells. Cancer Lett. 2020, 493, 189–196. [Google Scholar] [CrossRef] [PubMed]
- Howells, L.M.; Berry, D.P.; Elliott, P.J.; Jacobson, E.W.; Hoffmann, E.; Hegarty, B.; Brown, K.; Steward, W.P.; Gescher, A.J. Phase I randomized, double-blind pilot study of micronized resveratrol (SRT501) in patients with hepatic metastases—Safety, pharmacokinetics, and pharmacodynamics. Cancer Prev. Res. 2011, 4, 1419–1425. [Google Scholar] [CrossRef]
- Zhu, W.; Qin, W.; Zhang, K.; Rottinghaus, G.E.; Chen, Y.C.; Kliethermes, B.; Sauter, E.R. Trans-resveratrol alters mammary promoter hypermethylation in women at increased risk for breast cancer. Nutr. Cancer 2012, 64, 393–400. [Google Scholar] [CrossRef]
- Paller, C.J.; Rudek, M.A.; Zhou, X.C.; Wagner, W.; Hudson, T.S.; Anders, N.; Hammers, H.; Dowling, D.; King, S.; Antoranakis, E.; et al. A Phase I Study of Muscadine Grape Skin Extract in Men with Biochemically Recurrent Prostate Cancer: Safety, Tolerability, and Dose Determination. Prostate 2015, 75, 1518–1525. [Google Scholar] [CrossRef]
- Paller, C.J.; Zhou, X.C.; Heath, E.I.; Taplin, M.E.; Mayer, T.M.; Stein, M.N.; Bubley, G.J.; Pili, R.; Hudson, T.S.; Kakarla, R.; et al. Muscadine Grape Skin Extract (MPX) in Men with Biochemically Recurrent Prostate Cancer: A Randomized, Multicenter, Placebo- Controlled Clinical Trial. Clin. Cancer Res. 2018, 24, 306–315. [Google Scholar] [CrossRef]
- Pastorelli, D.; Fabricio, A.S.C.; Giovanis, P.; D’Ippolito, S.; Fiduccia, P.; Soldà, C.; Buda, A.; Sperti, C.; Bardini, R.; Da Dalt, G.; et al. Phytosome complex of curcumin as complementary therapy of advanced pancreatic cancer improves safety and efficacy of gemcitabine: Results of a prospective phase II trial. Pharmacol. Res. 2018, 132, 72–79. [Google Scholar] [CrossRef]
- Saghatelyan, T.; Tananyan, A.; Janoyan, N.; Tadevosyan, A.; Petrosyan, H.; Hovhannisyan, A.; Hayrapetyan, L.; Arustamyan, M.; Arnhold, J.; Rotmann, A.R.; et al. Efficacy and safety of curcumin in combination with paclitaxel in patients with advanced, metastatic breast cancer: A comparative, randomized, double-blind, placebo-controlled clinical trial. Phytomedicine 2020, 70, 153218. [Google Scholar] [CrossRef]
- Ekström, A.M.; Serafini, M.; Nyrén, O.; Wolk, A.; Bosetti, C.; Bellocco, R. Dietary quercetin intake and risk of gastric cancer: Results from a population-based study in Sweden. Ann. Oncol. 2011, 22, 438–443. [Google Scholar] [CrossRef]
- Citronberg, J.; Bostick, R.; Ahearn, T.; Turgeon, D.K.; Ruf, M.T.; Djuric, Z.; Sen, A.; Brenner, D.E.; Zick, S.M. Effects of Ginger Supplementation on Cell-Cycle Biomarkers in the Normal-Appearing Colonic Mucosa of Patients at Increased Risk for Colorectal Cancer: Results from a Pilot, Randomized, and Controlled Trial. Cancer Pre. 2013, 6, 271–282. [Google Scholar] [CrossRef]
- Danwilai, K.; Konmun, J.; Sripanidkulchai, B.; Subongkot, S. Antioxidant activity of ginger extract as a daily supplement in cancer patients receiving adjuvant chemotherapy: a pilot study. Cancer Manag. Res. 2017, 9, 11–18. [Google Scholar] [CrossRef]
- Konmun, J.; Danwilai, K.; Ngamphaiboon, N.; Sripanidkulchai, B.; Sookprasert, A.; Subongkot, S. A phase II randomized double-blind placebo-controlled study of 6-gingerol as an anti-emetic in solid tumor patients receiving moderately to highly emetogenic chemotherapy. Med. Oncol. 2017, 34, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Gates, M.A.; Tworoger, S.S.; Hecht, J.L.; De Vivo, I.; Rosner, B.; Hankinson, S.E. A prospective study of dietary flavonoid intake and incidence of epithelial ovarian cancer. Int. J. Cancer 2007, 121, 2225–2232. [Google Scholar] [CrossRef] [PubMed]
- Nöthlings, U.; Murphy, S.P.; Wilkens, L.R.; Henderson, B.E.; Kolonel, L.N. Flavonols and pancreatic cancer risk: The multiethnic cohort study. Am. J. Epidemiol. 2007, 166, 924–931. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Jiang, S.; Li, X.; Liu, Y.; Su, J.; Chen, J. Recent advances in trimethoxyphenyl (TMP) based tubulin inhibitors targeting the colchicine binding site. Eur. J. Med. Chem. 2018, 151, 482–494. [Google Scholar] [CrossRef]
- Ghawanmeh, A.A.; Chong, K.F.; Sarkar, S.M.; Bakar, M.A.; Othaman, R.; Khalid, R.M. Colchicine prodrugs and codrugs: Chemistry and bioactivities. Eur. J. Med. Chem. 2018, 144, 229–242. [Google Scholar] [CrossRef]
- Majcher, U.; Klejborowska, G.; Kaik, M.; Maj, E.; Wietrzyk, J.; Barl, F.; Predo, J.; Tuszynski, J.A.; Huczynski, A. Antiproliferative Activity and Molecular Docking of Novel Double-Modified Colchicine Derivatives. Cells 2018, 7, 192. [Google Scholar] [CrossRef]
- Majcher, U.; Klejborowska, G.; Kaik, M.; Maj, E.; Wietrzyk, J.; Moshari, M.; Predo, J.; Tuszynski, J.A.; Huczynski, A. Synthesis and Biological Evaluation of Novel Triple-Modified Colchicine Derivatives as Potent Tubulin-Targeting. Cells 2018, 7, 216. [Google Scholar] [CrossRef]
- Akhtar, M.S.; Swammy, M.K. Anticancer Plants: Clinical Trials and Nanotechnology; Springer: Singapore, 2017; Volume 3. [Google Scholar]
- Chartoumpekis, D.V.; Ziros, P.G.; Chen, J.; Groopman, J.D.; Kensler, T.W.; Sykiotis, G.P. Broccoli sprout beverage is safe for thyroid hormonal and autoimmune status: results of a 12-week randomized trial. Food Chem. Toxicol. 2020, 126, 1–6. [Google Scholar] [CrossRef]
- Kuran, D.; Pogorzelska, A. Breast Cancer Prevention-Is there a Future for Sulforaphane and Its Analogs? Nutrients 2020, 12, 1559. [Google Scholar] [CrossRef] [PubMed]
- Soundararajan, P.; Kim, J.S. Vegetables and Their Antagonistic Effects on Prevention of Cancers. Molecules 2018, 23, 2983. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Wade, K.L.; Wehage, S.L.; Holtzclaw, W.D.; Liu, H.; Talalay, P.; Fuchs, E.; Stephenson, K.K. Stabilized sulforaphane for clinical use: Phytochemical delivery efficiency. Mol. Nutr. Food Res. 2017, 61, 1–10. [Google Scholar] [CrossRef]
- Su, X.; Jiang, X.; Meng, L.; Dong, X.; Shen, Y.; Xin, Y. Anticancer activity of sulforaphane: The epigenetic mechanisms and the Nrf2 signaling pathway. Oxid. Med. Cell. Longev. 2018, 2018, 5438179. [Google Scholar] [CrossRef]
- Leone, A.; Diorio, G.; Sexton, W.; Schell, M.; Alexandrow, M.; Fahey, J.W.; Kumar, N.B. Sulforaphane for the chemoprevention of bladder cancer: Molecular mechanism targeted approach. Oncotarget 2017, 8, 35412–35424. [Google Scholar] [CrossRef]
- Jabbarzadeh, P.; Afzalipour, M.; Mohammadi, M. Targets and mechanisms of sulforaphane derivatives obtained from cruciferous plants with special focus on breast cancer—Contradictory effects and future perspectives. Biomed. Pharmacother. 2020, 121, 109635. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Li, H.U.A.; Lee, E.D.; Grandis, J.R.; Bauman, J.E.; Johnson, D.E. Gene targets of sulforaphane in head and neck squamous cell carcinoma. Mol. Med. Rep. 2019, 5335–5344. [Google Scholar] [CrossRef]
- Scarano, A.; Chieppa, M.; Santino, A. Plant polyphenols-biofortified foods as a novel tool for the prevention of human gut diseases. Antioxidants 2020, 9, 1225. [Google Scholar] [CrossRef]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem. 2002, 13, 572–584. [Google Scholar] [CrossRef]
- Ravishankar, D.; Rajora, A.K.; Greco, F.; Osborn, H.M.I. Flavonoids as prospective compounds for anti-cancer therapy. Int. J. Biochem. Cell Biol. 2013, 45, 2821–2831. [Google Scholar] [CrossRef]
- Dai, F.; Yan, W.J.; Du, Y.T.; Bao, X.Z.; Li, X.Z.; Zhou, B. Structural basis, chemical driving forces and biological implications of flavones as Cu(II) ionophores. Free Radic. Biol. Med. 2017, 108, 554–563. [Google Scholar] [CrossRef]
- Tungmunnithum, D.; Thongboonyou, A.; Pholboon, A.; Yangsabai, A. Flavonoids and Other Phenolic Compounds from Medicinal Plants for Pharmaceutical and Medical Aspects: An Overview. Medicines 2018, 5, 93. [Google Scholar] [CrossRef] [PubMed]
- Procházková, D.; Boušová, I.; Wilhelmová, N. Antioxidant and prooxidant properties of flavonoids. Fitoterapia 2011, 82, 513–523. [Google Scholar] [CrossRef]
- Malaguarnera, L. Influence of resveratrol on the immune response. Nutrients 2019, 11, 946. [Google Scholar] [CrossRef]
- Czop, M.; Bogucka-Kocka, A.; Kubrak, T.; Knap-Czop, K.; Makuch-Kocka, A.; Galkowski, D.; Wawer, J.; Kocki, T.; Kocki, J. Imaging flow cytometric analysis of stilbene-dependent apoptosis in drug resistant human leukemic cell lines. Molecules 2019, 24, 1896. [Google Scholar] [CrossRef]
- Wang, H.; Zhang, H.; Tang, L.; Chen, H.; Wu, C.; Zhao, M.; Yang, Y.; Chen, X.; Liu, G. Resveratrol inhibits TGF-β1-induced epithelial-to-mesenchymal transition and suppresses lung cancer invasion and metastasis. Toxicology 2013, 303, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Li, J.; Yang, Y.; Zhao, X.; Liu, Y.; Jiang, Y.; Zhou, L.; Feng, Y.; Yu, Y.; Cheng, Y. Resveratrol modulates the apoptosis and autophagic death of human lung adenocarcinoma A549 cells via a p53-dependent pathway: Integrated bioinformatics analysis and experimental validation. Int. J. Oncol. 2020, 57, 926–938. [Google Scholar] [CrossRef]
- Ganapathy, S.; Chen, Q.; Singh, K.P.; Shankar, S.; Srivastava, R.K. Resveratrol enhances antitumor activity of TRAIL in prostate cancer xenografts through activation of FOXO transcription factor. PLoS ONE 2010, 5, 15627. [Google Scholar] [CrossRef] [PubMed]
- Rai, G.; Mishra, S.; Suman, S.; Shukla, Y. Resveratrol improves the anticancer effects of doxorubicin in vitro and in vivo models: A mechanistic insight. Phytomedicine 2016, 23, 233–242. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Xu, Z.; Dai, S.; Qian, L.; Sun, L.; Gong, Z. Targeting autophagy to sensitive glioma to temozolomide treatment. J. Exp. Clin. Cancer Res. 2016, 35, 1–14. [Google Scholar] [CrossRef]
- Patra, S.; Pradhan, B.; Nayak, R.; Behera, C.; Rout, L.; Jena, M.; Efferth, T.; Bhutia, S.K. Chemotherapeutic efficacy of curcumin and resveratrol against cancer: Chemoprevention, chemoprotection, drug synergism and clinical pharmacokinetics. Semin. Cancer Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gregoriou, Y.; Gregoriou, G.; Yilmaz, V.; Kapnisis, K.; Prokopi, M. Resveratrol loaded polymeric micelles for theranostic targeting of breast cancer cells. Nanotheranostics 2021, 5, 113–124. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.; Huang, Y.-W.; Oshima, K.; Martha Yearsley, J.Z.; Arnold, M.; Yu, J.; Wang, L.-S. The Immunomodulatory Potential of Natural Compounds in Tumor-Bearing Mice and Humans. Crit. Rev. Food Sci. Nutr. 2019, 59, 992–1007. [Google Scholar]
- Kumar, G.; Mittal, S.; Sak, K.; Tuli, H.S. Molecular mechanisms underlying chemopreventive potential of curcumin: Current challenges and future perspectives. Life Sci. 2016, 148, 313–328. [Google Scholar] [CrossRef] [PubMed]
- Pourhanifeh, M.H.; Darvish, M.; Tabatabaeian, J.; Fard, M.R.; Mottaghi, R.; Azadchehr, M.J.; Jahanshahi, M.; Sahebkar, A.; Mirzaei, H. Therapeutic role of curcumin and its novel formulations in gynecological cancers. J. Ovarian Res. 2020, 13, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Chen, Q.; Gao, Q.; Chen, K.; Wang, Y.; Chen, L.; Li, X. Curcumin suppresses migration and invasion of human endometrial carcinoma cells. Oncol. Lett. 2015, 10, 1297–1302. [Google Scholar] [CrossRef]
- Masuelli, L.; Benvenuto, M.; Di Stefano, E.; Mattera, R.; Fantini, M.; De Feudis, G.; De Smaele, E.; Tresoldi, I.; Giganti, M.G.; Modesti, A.; et al. Curcumin blocks autophagy and activates apoptosis of malignant mesothelioma cell lines and increases the survival of mice intraperitoneally transplanted with a malignant mesothelioma cell line. Oncotarget 2017, 8, 34405–34422. [Google Scholar] [CrossRef]
- Taverna, S.; Giallombardo, M.; Pucci, M.; Flugy, A.; Manno, M.; Raccosta, S.; Rolfo, C.; De Leo, G.; Alessandro, R. Curcumin inhibits in vitro and in vivo chronic myelogenous leukemia cells growth: A possible role for exosomal disposal of miR-21. Oncotarget 2015, 6, 21918–21933. [Google Scholar] [CrossRef] [PubMed]
- Koohpar, Z.K.; Entezari, M.; Movafagh, A.; Hashemi, M. Anticancer activity of curcumin on human breast adenocarcinoma: Role of Mcl-1 gene. Int. J. Cancer Manag. 2015, 8, 2331. [Google Scholar] [CrossRef]
- Bimonte, S.; Barbieri, A.; Leongito, M.; Piccirillo, M.; Giudice, A.; Pivonello, C.; De Angelis, C.; Granata, V.; Palaia, R.; Izzo, F. Curcumin anticancer studies in pancreatic cancer. Nutrients 2016, 8, 433. [Google Scholar] [CrossRef]
- Chen, W.; Li, L.; Zhang, X.; Liang, Y.; Pu, Z.; Wang, L.; Mo, J. Curcumin: A calixarene derivative micelle potentiates anti-breast cancer stem cells effects in xenografted, triple-negative breast cancer mouse models. Drug Deliv. 2017, 24, 1470–1481. [Google Scholar] [CrossRef]
- Sen, G.S.; Mohanty, S.; Hossain, D.M.S.; Bhattacharyya, S.; Banerjee, S.; Chakraborty, J.; Saha, S.; Ray, P.; Bhattacharjee, P.; Mandal, D.; et al. Curcumin enhances the efficacy of chemotherapy by tailoring p65NFκB-p300 cross-talk in favor of p53-p300 in breast cancer. J. Biol. Chem. 2011, 286, 42232–42247. [Google Scholar] [CrossRef] [PubMed]
- Su, P.; Yang, Y.; Wang, G.; Chen, X.; Ju, Y. Curcumin attenuates resistance to irinotecan via induction of apoptosis of cancer stem cells in chemoresistant colon cancer cells. Int. J. Oncol. 2018, 53, 1343–1353. [Google Scholar] [CrossRef] [PubMed]
- Dihal, A.A.; De Boer, V.C.J.; Van Der Woude, H.; Tilburgs, C.; Bruijntjes, J.P.; Alink, G.M.; Rietjens, I.M.C.M.; Woutersen, R.A.; Stierum, R.H. Quercetin, but not its glycosidated conjugate rutin, inhibits azoxymethane-induced colorectal carcinogenesis in F344 rats. J. Nutr. 2006, 136, 2862–2867. [Google Scholar] [CrossRef] [PubMed]
- Tang, S.M.; Deng, X.T.; Zhou, J.; Li, Q.P.; Ge, X.X.; Miao, L. Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomed. Pharmacother. 2020, 121, 109604. [Google Scholar] [CrossRef]
- Khursheed, R.; Singh, S.K.; Wadhwa, S.; Gulati, M.; Awasthi, A. Enhancing the potential preclinical and clinical benefits of quercetin through novel drug delivery systems. Drug Discov. Today 2020, 25, 209–222. [Google Scholar] [CrossRef]
- Duo, J.; Ying, G.G.; Wang, G.W.; Zhang, L. Quercetin inhibits human breast cancer cell proliferation and induces apoptosis via Bcl-2 and Bax regulation. Mol. Med. Rep. 2012, 5, 1453–1456. [Google Scholar] [CrossRef] [PubMed]
- Zheng, S.Y.; Li, Y.; Jiang, D.; Zhao, J.; Ge, J.F. Anticancer effect and apoptosis induction by quercetin in the human lung cancer cell line A-549. Mol. Med. Rep. 2012, 5, 822–826. [Google Scholar] [CrossRef] [PubMed]
- Albrecht, C.; Cittadini, M.C.; Soria, E.A. Pharmacological Activity of Quercetin and 5 caffeoylquinic Acid Oral Intake in Male Balb/c Mice with Lung Adenocarcinoma. Arch. Med. Res. 2020, 51, 8–12. [Google Scholar] [CrossRef] [PubMed]
- Calgarotto, A.K.; Maso, V.; Junior, G.C.F.; Nowill, A.E.; Filho, P.L.; Vassallo, J.; Saad, S.T.O. Antitumor activities of Quercetin and Green Tea in xenografts of human leukemia HL60 cells. Sci. Rep. 2018, 8, 3–9. [Google Scholar] [CrossRef]
- Jia, L.; Huang, S.; Yin, X.; Zan, Y.; Guo, Y.; Han, L. Quercetin suppresses the mobility of breast cancer by suppressing glycolysis through Akt-mTOR pathway mediated autophagy induction. Life Sci. 2018, 208, 123–130. [Google Scholar] [CrossRef]
- Lei, C.S.; Hou, Y.C.; Pai, M.H.; Lin, M.T.; Yeh, S.L. Effects of quercetin combined with anticancer drugs on metastasis-associated factors of gastric cancer cells: in vitro and in vivo studies. J. Nutr. Biochem. 2018, 51, 105–113. [Google Scholar] [CrossRef]
- Shitole, A.A.; Sharma, N.; Giram, P.; Khandwekar, A.; Baruah, M.; Garnaik, B.; Koratkar, S. LHRH-conjugated, PEGylated, poly-lactide-co-glycolide nanocapsules for targeted delivery of combinational chemotherapeutic drugs Docetaxel and Quercetin for prostate cancer. Mater. Sci. Eng. C 2020, 114, 111035. [Google Scholar] [CrossRef] [PubMed]
- Kooshyar, M.M.; Mozafari, P.M.; Amirchaghmaghi, M.; Pakfetrat, A.; Karoos, P.; Mohasel, M.R.; Orafai, H.; Azarian, A.A. A randomized placebo- controlled double blind clinical trial of quercetin in the prevention and treatment of chemotherapy-induced oral mucositis. J. Clin. Diagnostic Res. 2017, 11, ZC46–ZC50. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.C.; Syu, J.J.; Chen, J.C.; Wang, T.J.; Chang, P.Y.; Chen, C.Y.; Jian, Y.T.; Jian, Y.J.; Lin, Y.W. Resveratrol Enhances Etoposide-Induced Cytotoxicity through Down-Regulating ERK1/2 and AKT-Mediated X-ray Repair Cross-Complement Group 1 (XRCC1) Protein Expression in Human Non-Small-Cell Lung Cancer Cells. Basic Clin. Pharmacol. Toxicol. 2015, 117, 383–391. [Google Scholar] [CrossRef] [PubMed]
- De Lima, R.M.T.; dos Reis, A.C.; de Menezes, A.A.P.M.; Santos, J.V.; Filho, J.W.G.D.O.; Ferreira, J.R.D.O.; de Alencar, M.V.O.B.; da Mata, A.M.O.F.; Khan, I.N.; Islam, A.; et al. Protective and therapeutic potential of ginger (Zingiber officinale) extract and [6]-gingerol in cancer: A comprehensive review. Phyther. Res. 2018, 32, 1885–1907. [Google Scholar] [CrossRef]
- De Lima, R.M.T.; dos Reis, A.C.; de Oliveira Santos, J.V.; de Oliveira Ferreira, J.R.; de Oliveira Filho, J.W.G.; Dias, A.C.S.; de Menezes, A.A.P.M.; da Mata, A.M.O.; Ferreira; de Alencar, M.V.O.B.; et al. Antitumoral effects of [6]-gingerol [(S)-5-hydroxy-1-(4-hydroxy-3-methoxyphenyl) -3-decanone] in sarcoma 180 cells through cytogenetic mechanisms. Biomed. Pharmacother. 2020, 126, 110004. [Google Scholar] [CrossRef]
- De Lima, R.M.T.; dos Reis, A.C.; de Oliveira Santos, J.V.; de Oliveira Ferreira, J.R.; de Oliveira Filho, J.W.G.; de Menezes, A.A.P.M.; da Mata, A.M.O.F.; de Alencar, M.V.O.B.; Rodrigues, D.C.N.; Ferreira, P.M.P.; et al. Toxic, cytogenetic and antitumor evaluations of [6] -gingerol in non-clinical in vitro studies. Biomed. Pharmacother. 2019, 115, 108873. [Google Scholar] [CrossRef]
- Martin, A.C.B.M.; Fuzer, A.M.; Becceneri, A.B.; Almada, J.; Tomasin, R.; Denoyer, D.; Kim, S.; Mcintyre, K.A.; Pearson, H.B.; Yeo, B.; et al. [10]-gingerol induces apoptosis and inhibits metastatic dissemination of triple negative breast cancer in vivo. Oncotarget 2017, 8, 72260–72271. [Google Scholar] [CrossRef]
- Martin, A.C.B.M.; Tomasin, R.; Luna-Dulcey, L.; Graminha, A.E.; Naves, M.A.; Teles, R.H.G.; da Silva, V.D.; da Silva, J.A.; Vieira, P.C.; Annabi, B.; et al. [10]-Gingerol improves doxorubicin anticancer activity and decreases its side effects in triple negative breast cancer models. Cell. Oncol. 2020, 43, 915–929. [Google Scholar] [CrossRef] [PubMed]
- Luo, Y.; Zha, L.; Luo, L.; Chen, X.; Zhang, Q.; Gao, C.; Zhuang, X.; Yuan, S.; Qiao, T. [6]-Gingerol enhances the cisplatin sensitivity of gastric cancer cells through inhibition of proliferation and invasion via PI3K/AKT signaling pathway. Phyther. Res. 2019, 33, 1353–1362. [Google Scholar] [CrossRef]
- Wang, Q.; Wei, Q.; Yang, Q.; Cao, X.; Li, Q.; Shi, F.; Tong, S.S.; Feng, C.; Yu, Q.; Yu, J.; et al. A novel formulation of [6] -gingerol: Proliposomes with enhanced oral bioavailability and antitumor effect. Int. J. Pharm. 2018, 535, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Wei, Q.; Yang, Q.; Wang, Q.; Sun, C.; Zhu, Y.; Niu, Y.; Yu, J.; Xu, X. Formulation, Characterization, and Pharmacokinetic Studies of 6-Gingerol-Loaded Nanostructured Lipid Carriers. AAPS PharmSciTech 2018, 19, 3661–3669. [Google Scholar] [CrossRef]
- Xu, Y.; Wang, Q.; Feng, Y.; Firempong, C.K.; Zhu, Y.; Omari-Siaw, E.; Zheng, Y.; Pu, Z.; Xu, X.; Yu, J. Enhanced oral bioavailability of [6]-Gingerol-SMEDDS: Preparation, in vitro and in vivo evaluation. J. Funct. Foods 2016, 27, 703–710. [Google Scholar] [CrossRef]
- Chen, A.Y.; Chen, Y.C. A review of the dietary flavonoid, kaempferol on human health and cancer chemoprevention. Food Chem. 2013, 138, 2099–2107. [Google Scholar] [CrossRef]
- Rajendran, P.; Rengarajan, T.; Nandakumar, N.; Palaniswami, R.; Nishigaki, Y.; Nishigaki, I. Kaempferol, a potential cytostatic and cure for inflammatory disorders. Eur. J. Med. Chem. 2014, 86, 103–112. [Google Scholar] [CrossRef] [PubMed]
- Kashyap, D.; Sharma, A.; Singh, H.; Sak, K.; Punia, S.; Mukherjee, T.K. Kaempferol—A dietary anticancer molecule with multiple mechanisms of action: Recent trends and advancements. J. Funct. Foods 2017, 30, 203–219. [Google Scholar] [CrossRef] [PubMed]
- Wang, L.; Lee, I.M.; Zhang, S.M.; Blumberg, J.B.; Buring, J.E.; Sesso, H.D. Dietary intake of selected flavonols, flavones, and flavonoid-rich foods and risk of cancer in middle-aged and older women. Am. J. Clin. Nutr. 2009, 89, 905–912. [Google Scholar] [CrossRef] [PubMed]
- Gates, M.A.; Vitonis, A.F.; Tworoger, S.S.; Rosner, B.; Titus-Ernstoff, L.; Hankinson, S.E.; Cramer, D.W. Flavonoid intake and ovarian cancer risk in a population-based case-control study. Int. J. Cancer 2009, 124, 1918–1925. [Google Scholar] [CrossRef]
- Adebamowo, C.A.; Cho, E.; Sampson, L.; Katan, M.B.; Spiegelman, D.; Willett, W.C.; Holmes, M.D. Dietary flavonols and flavonol-rich foods intake and the risk of breast cancer. Int. J. Cancer 2005, 114, 628–633. [Google Scholar] [CrossRef]
- Thangavel, P.; Viswanath, B.; Kim, S. Synthesis and characterization of kaempferol-based ruthenium (II) complex: A facile approach for superior anticancer application. Mater. Sci. Eng. C 2018, 89, 87–94. [Google Scholar] [CrossRef] [PubMed]
- Pecere, T.; Gazzola, M.V.; Mucignat, C.; Parolin, C.; Vecchia, F.D.; Cavaggioni, A.; Basso, G.; Diaspro, A.; Salvato, B.; Carli, M.; et al. Aloe-emodin is a new type of anticancer agent with selective activity against neuroectodermal tumors. Cancer Res. 2000, 60, 2800–2804. [Google Scholar] [PubMed]
- Xing, J.Y.; Song, G.P.; Deng, J.P.; Jiang, L.Z.; Xiong, P.; Yang, B.J.; Liu, S.S. Antitumor effects and mechanism of novel emodin rhamnoside derivatives against human cancer cells in vitro. PLoS ONE 2015, 10, 144781. [Google Scholar] [CrossRef]
- Slezakova, S.; Ruda-Kucerova, J. Anticancer Activity of Artemisinin and its Derivatives. Anticancer Res. 2017, 37, 5995–6003. [Google Scholar]
- Efferth, T.; Dunstan, H.; Sauerbrey, A.; Miyachi, H.; Chitambar, C.R. The anti-malarial artesunate is also active against cancer. Int. J. Oncol. 2001, 18, 767–773. [Google Scholar] [CrossRef]
- Bukhari, S.N.A.; Kumar, G.B.; Revankar, H.M.; Qin, H.L. Development of combretastatins as potent tubulin polymerization inhibitors. Bioor. Chem. 2017, 72, 130–147. [Google Scholar] [CrossRef]
- Liu, W.M.; Gravett, A.M.; Dalgleish, A.G. The antimalarial agent artesunate possesses anticancer properties that can be enhanced by combination strategies. Int. J. Cancer 2011, 128, 1471–1480. [Google Scholar] [CrossRef]
- Faujan, N.H.; Alitheen, N.B.; Yeap, S.K.; Ali, A.M.; Muhajir, A.H.; Ahmad, F.B.H. Cytotoxic effect of betulinic acid and betulinic acid acetate isolated from Melaleuca cajuput on human myeloid leukemia (HL-60) cell line. African J. Biotechnol. 2010, 9, 6387–6396. [Google Scholar] [CrossRef]
- Surowiak, P.; Drag, M.; Materna, V.; Dietel, M.; Lage, H. Betulinic acid exhibits stronger cytotoxic activity on the normal melanocyte NHEM-neo cell line than on drug-resistant and drug-sensitive MeWo melanoma cell lines. Mol. Med. Rep. 2009, 2, 543–548. [Google Scholar] [CrossRef] [PubMed]
- Damle, A.A.; Pawar, Y.P.; Narkar, A.A. Anticancer activity of betulinic acid on MCF-7 tumors in nude mice. Indian J. Exp. Biol. 2013, 51, 485–491. [Google Scholar]
- Zhang, X.; Hu, J.; Chen, Y. Betulinic acid and the pharmacological effects of tumor suppression (Review). Mol. Med. Rep. 2016, 14, 4489–4495. [Google Scholar] [CrossRef]
- Król, S.K.; Kiełbus, M.; Rivero-Müller, A.; Stepulak, A. Comprehensive review on betulin as a potent anticancer agent. Biomed Res. Int. 2015, 2015, 584189. [Google Scholar] [CrossRef]
- Xu, J.; Long, Y.; Ni, L.; Yuan, X.; Yu, N.; Wu, R.; Tao, J.; Zhang, Y. Anticancer effect of berberine based on experimental animal models of various cancers: A systematic review and meta-analysis. BMC Cancer 2019, 19, 589. [Google Scholar] [CrossRef]
- Barzegar, E.; Fouladdel, S.; Movahhed, T.K.; Atashpour, S.; Ghahremani, M.H.; Ostad, S.N.; Azizi, E. Effects of berberine on proliferation, cell cycle distribution and apoptosis of human breast cancer T47D and MCF7 cell lines. Iran. J. Basic Med. Sci. 2015, 18, 334–342. [Google Scholar] [PubMed]
- Babich, H.; Krupka, M.E.; Nissim, H.A.; Zuckerbraun, H.L. Differential in vitro cytotoxicity of (-)-epicatechin gallate (ECG) to cancer and normal cells from the human oral cavity. Toxicol. Vitr. 2005, 19, 231–242. [Google Scholar] [CrossRef]
- Park, K.D.; Lee, S.G.; Kim, S.U.; Kim, S.H.; Sun, W.S.; Cho, S.J.; Jeong, D.H. Anticancer activity of 3-O-acyl and alkyl-(-)-epicatechin derivatives. Bioorganic Med. Chem. Lett. 2004, 14, 5189–5192. [Google Scholar] [CrossRef]
- Yang, G.Y.; Liao, J.; Kim, K.; Yurkow, E.J.; Yang, C.S. Inhibition of growth and induction of apoptosis in human cancer cell lines by tea polyphenols. Carcinogenesis 1998, 19, 611–616. [Google Scholar] [CrossRef]
- Karatoprak, G.Ş.; Küpeli Akkol, E.; Genç, Y.; Bardakci, H.; Yücel, Ç.; Sobarzo-Sánchez, E. Combretastatins: An Overview of Structure, Probable Mechanisms of Action and Potential Applications. Molecules 2020, 25, 2560. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, J.M.; Kim, S.C.; Park, C.B.; Lee, P.C. Proposed cytotoxic mechanisms of the saffron carotenoids crocin and crocetin on cancer cell lines. Biochem. Cell Biol. 2014, 92, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Hire, R.R.; Srivastava, S.; Davis, M.B.; Kumar Konreddy, A.; Panda, D. Antiproliferative Activity of Crocin Involves Targeting of Microtubules in Breast Cancer Cells. Sci. Rep. 2017, 7, 1–11. [Google Scholar] [CrossRef]
- Hoshyar, R.; Mollaei, H. A comprehensive review on anticancer mechanisms of the main carotenoid of saffron, crocin. J. Pharm. Pharmacol. 2017, 69, 1419–1427. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhao, Y.; Guo, J.; Cui, H.; Liu, S. Anticancer activity of safranal against colon carcinoma is due to induction of apoptosis and G2/M cell cycle arrest mediated by suppression of mTOR/PI3K/Akt pathway. J. B.U.ON. 2018, 23, 574–578. [Google Scholar]
- Stahlhut, M.; Bertelsen, M.; Høyer-Hansen, M.; Zibert, J.R. Ingenol Mebutate-Induced Cell Death Patterns in Normal and Cancer Epithelial Cells. J. Drugs Dermatol. 2012, 11, 707–712. [Google Scholar]
- Silva, V.A.O.; Rosa, M.N.; Martinho, O.; Tanuri, A.; Lima, J.P.; Pianowski, L.F.; Reis, R.M. Modified ingenol semi-synthetic derivatives from Euphorbia tirucalli induce cytotoxicity on a large panel of human cancer cell lines. Investig. New Drugs 2019, 37, 1029–1035. [Google Scholar] [CrossRef]
- Chen, Z.; Liu, W.; Qin, Z.; Liang, X.; Tian, G. Geniposide exhibits anticancer activity to medulloblastoma cells by downregulating microRNA-373. J. Biochem. Mol. Toxicol. 2020, 34. [Google Scholar] [CrossRef]
- Shanmugam, M.K.; Shen, H.; Tang, F.R.; Arfuso, F.; Rajesh, M.; Wang, L.; Kumar, A.P.; Bian, J.; Goh, B.C.; Bishayee, A.; et al. Potential role of genipin in cancer therapy. Pharmacol. Res. 2018, 133, 195–200. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.S.; Bi, W.C.; Chan, G.K.L.; Jin, Y.; Wong, C.W.; Zhou, Z.Y.; Wang, H.Y.; Yao, P.; Dong, T.T.X.; Tsim, K.W.K. Ginkgetin induces autophagic cell death through p62/SQSTM1- mediated autolysosome formation and redox setting in nonsmall cell lung cancer. Oncotarget 2017, 8, 93131–93148. [Google Scholar] [CrossRef]
- Jeon, Y.J.; Jung, S.N.; Yun, J.; Lee, C.W.; Choi, J.; Lee, Y.J.; Han, D.C.; Kwon, B.M. Ginkgetin inhibits the growth of DU-145 prostate cancer cells through inhibition of signal transducer and activator of transcription 3 activity. Cancer Sci. 2015, 106, 413–420. [Google Scholar] [CrossRef]
- Park, Y.; Woo, S.H.; Seo, S.K.; Kim, H.; Noh, W.C.; Lee, J.K.; Kwon, B.M.; Min, K.N.; Choe, T.B.; Park, I.C. Ginkgetin induces cell death in breast cancer cells via downregulation of the estrogen receptor. Oncol. Lett. 2017, 14, 5027–5033. [Google Scholar] [CrossRef]
- Li, M.; Li, B.; Xia, Z.M.; Tian, Y.; Zhang, D.; Rui, W.J.; Dong, J.X.; Xiao, F.J. Anticancer effects of five biflavonoids from ginkgo biloba l. Male flowers in vitro. Molecules 2019, 24, 1496. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, Y.S.; Thakur, K.; Hussain, S.S.; Zhang, J.G.; Xiao, G.R.; Wei, Z.J. Licochalcone A from licorice root, an inhibitor of human hepatoma cell growth via induction of cell apoptosis and cell cycle arrest. Food Chem. Toxicol. 2018, 120, 407–417. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.J.; Cho, I.A.; Kang, K.R.; Jung, Y.R.; Cho, S.S.; Yoon, G.; Oh, J.S.; You, J.S.; Seo, Y.S.; Lee, G.J.; et al. Licochalcone-E induces caspase-dependent death of human pharyngeal squamous carcinoma cells through the extrinsic and intrinsic apoptotic signaling pathways. Oncol. Lett. 2017, 13, 3662–3668. [Google Scholar] [CrossRef][Green Version]
- Peng, J.; Fan, G.; Wu, Y. Preparative isolation of four new and two known flavonoids from the leaf of Patrinia villosa Juss. by counter-current chromatography and evaluation of their anticancer activities in vitro. J. Chromatogr. A 2006, 1115, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, D.; Ganguli, A.; Dastidar, D.G.; Acharya, B.R.; Das, A.; Chakrabarti, G. Apigenin shows synergistic anticancer activity with curcumin by binding at different sites of tubulin. Biochimie 2013, 95, 1297–1309. [Google Scholar] [CrossRef]
- Al-Dabbagh, B.; Elhaty, I.A.; Elhaw, M.; Murali, C.; Al Mansoori, A.; Awad, B.; Amin, A. Antioxidant and anticancer activities of chamomile (Matricaria recutita L.). BMC Res. Notes 2019, 12, 3. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, J.K.; Gupta, S. Antiproliferative and apoptotic effects of chamomile extract in various human cancer cells. J. Agric. Food Chem. 2007, 55, 9470–9478. [Google Scholar] [CrossRef]
- Li, X.L.; Wang, C.Z.; Mehendale, S.R.; Sun, S.; Wang, Q.; Yuan, C.S. Panaxadiol, a purified ginseng component, enhances the anti-cancer effects of 5-fluorouracil in human colorectal cancer cells. Cancer Chemother. Pharmacol. 2009, 64, 1097–1104. [Google Scholar] [CrossRef]
- Sun, M.; Ye, Y.; Xiao, L.; Duan, X.; Zhang, Y.; Zhang, H. Anticancer effects of ginsenoside Rg3 (Review). Int. J. Mol. Med. 2017, 39, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, M.; Reddy, P.; Rawat, D. Noscapine and its analogues as anti- cancer agents. Chem. Biol. Interface 2014, 4, 1–22. [Google Scholar]
- Falodun, A.; Engel, N.; Kragl, U.; Nebe, B.; Langer, P. Novel anticancer alkene lactone from Persea americana. Pharm. Biol. 2013, 51, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Hu, C.X.; Zuo, Z.L.; Xiong, B.; Ma, J.G.; Geng, M.Y.; Lin, L.P.; Jiang, H.L.; Ding, J. Salvicine functions as novel topoisomerase II poison by binding to ATP pocket. Mol. Pharmacol. 2006, 70, 1593–1601. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.H.; Ding, J. Salvicine, a novel topoisomerase II inhibitor, exerts its potent anticancer activity by ROS generation. Acta Pharmacol. Sin. 2007, 28, 1460–1465. [Google Scholar] [CrossRef]
- Deng, F.; Lu, J.J.; Liu, H.Y.; Lin, L.P.; Ding, J.; Zhang, J.S. Synthesis and antitumor activity of novel salvicine analogues. Chinese Chem. Lett. 2011, 22, 25–28. [Google Scholar] [CrossRef]
- Wu, C.F.; Klauck, S.M.; Efferth, T. Anticancer activity of cryptotanshinone on acute lymphoblastic leukemia cells. Arch. Toxicol. 2016, 90, 2275–2286. [Google Scholar] [CrossRef]
- Chen, L.; Wang, H.J.; Xie, W.; Yao, Y.; Zhang, Y.S.; Wang, H. Cryptotanshinone inhibits lung tumorigenesis and induces apoptosis in cancer cells in vitro and in vivo. Mol. Med. Rep. 2014, 9, 2447–2452. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Wang, H.; Hong, L.; Liu, W.; Huang, F.; Wang, J.; Wang, P.; Zhang, X.; Zhou, J. Cryptotanshinone inhibits breast cancer cell growth by suppressing estrogen receptor signaling. Cancer Biol. Ther. 2015, 16, 176–184. [Google Scholar] [CrossRef]
- Qin, T.; Rasul, A.; Sarfraz, A.; Sarfraz, I.; Hussain, G.; Anwar, H.; Riaz, A.; Liu, S.; Wei, W.; Li, J.; et al. Salvianolic acid A & B: Potential cytotoxic polyphenols in battle against cancer via targeting multiple signaling pathways. Int. J. Biol. Sci. 2019, 15, 2256–2264. [Google Scholar]
- Deep, G.; Oberlies, N.H.; Kroll, D.J.; Agarwal, R. Isosilybin B and isosilybin A inhibit growth, induce G1 arrest and cause apoptosis in human prostate cancer LNCaP and 22Rv1 cells. Carcinogenesis 2007, 28, 1533–1542. [Google Scholar] [CrossRef] [PubMed]
- Dhanalakshmi, S.; Agarwal, C.; Singh, R.P.; Agarwal, R. Silibinin up-regulates DNA-protein kinase-dependent p53 activation to enhance UVB-induced apoptosis in mouse epithelial JB6 cells. J. Biol. Chem. 2005, 280, 20375–20383. [Google Scholar] [CrossRef]
- Scambia, G.; De Vincenzo, R.; Ranelletti, F.O.; Panici, P.B.; Ferrandina, G.; D’Agostino, G.; Fattorossi, A.; Bombardelli, E.; Mancuso, S. Antiproliferative effect of silybin on gynaecological malignancies: Synergism with cisplatin and doxorubicin. Eur. J. Cancer 1996, 32, 877–882. [Google Scholar] [CrossRef]
- Ramasamy, K.; Agarwal, R. Multitargeted therapy of cancer by silymarin. Cancer Lett. 2008, 269, 352–362. [Google Scholar] [CrossRef] [PubMed]
- Zi, X.; Feyes, D.K.; Agarwal, R. Anticarcinogenic effect of a flavonoid antioxidant, silymarin, in human breast cancer cells MDA-MB 468: Induction of G1 arrest through an increase in Cip1/p21 concomitant with a decrease in kinase activity of cyclin-dependent kinases and associated cyclins. Clin. Cancer Res. 1998, 4, 1055–1064. [Google Scholar]
- Shukla, Y.; Singh, M. Cancer preventive properties of ginger: A brief review. Food Chem. Toxicol. 2007, 45, 683–690. [Google Scholar] [CrossRef]
- Al-Abbasi, F.A.; Alghamdi, E.A.; Baghdadi, M.A.; Alamoudi, A.J.; El-Halawany, A.M.; El-Bassossy, H.M.; Aseeri, A.H.; Al-Abd, A.M. Gingerol synergizes the cytotoxic effects of doxorubicin against liver cancer cells and protects from its vascular toxicity. Molecules 2016, 21, 886. [Google Scholar] [CrossRef]
- Zhu, Y.; Warin, R.F.; Soroka, D.N.; Chen, H.; Sang, S. Metabolites of Ginger Component [6]-Shogaol Remain Bioactive in Cancer Cells and Have Low Toxicity in Normal Cells: Chemical Synthesis and Biological Evaluation. PLoS ONE 2013, 8, 54677. [Google Scholar] [CrossRef] [PubMed]
- Jafri, A.; Amjad, S.; Bano, S.; Kumar, S.; Serajuddin, M.; Arshad, M. Efficacy of Nano-phytochemicals Over Pure Phytochemicals Against Various Cancers: Current Trends and Future Prospects. In Nanomaterials and Environmental Biotechnology; Bhushan, I., Singh, V.K., Tripathi, D.K., Eds.; Springer: New York, NY, USA, 2020; pp. 407–424. [Google Scholar]
- Singh, V.K.; Arora, D.; Ansari, M.I.; Sharma, P.K. Phytochemicals based chemopreventive and chemotherapeutic strategies and modern technologies to overcome limitations for better clinical applications. Phyther. Res. 2019, 33, 3064–3089. [Google Scholar] [CrossRef]
- Zhao, J.; Koay, E.J.; Li, T.; Wen, X.; Li, C. A hindsight reflection on the clinical studies of poly(l-glutamic acid)-paclitaxel. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2018, 10, 1–18. [Google Scholar] [CrossRef]
- Lagoa, R.; Silva, J.; Rui, J.; Bishayee, A. Advances in phytochemical delivery systems for improved anticancer activity. Biotechnol. Adv. 2020, 38, 107382. [Google Scholar] [CrossRef] [PubMed]
- Manjili, H.K.; Ma, L.; Tavaddod, S.; Mashhadikhan, M. D, L-Sulforaphane Loaded Fe3O4@ Gold Core Shell Nanoparticles: A Potential Sulforaphane Delivery System. PLoS ONE 2016, 11, e0151344. [Google Scholar] [CrossRef]
- Ghosh, S.; Dutta, S.; Sarkar, A.; Kundu, M.; Sil, P.C. Targeted delivery of curcumin in breast cancer cells via hyaluronic acid modified mesoporous silica nanoparticle to enhance anticancer efficiency. Colloids Surf. B Biointerfaces 2021, 197, 111404. [Google Scholar] [CrossRef]
- Zhou, X.; Seto, S.W.; Chang, D.; Kiat, H.; Razmovski-Naumovski, V.; Chan, K.; Bensoussan, A. Synergistic effects of Chinese herbal medicine: A comprehensive review of methodology and current research. Front. Pharmacol. 2016, 7, 1–16. [Google Scholar] [CrossRef]
- Singh, S.; Sharma, B.; Kanwar, S.S.; Kumar, A. Lead phytochemicals for anticancer drug development. Front. Plant Sci. 2016, 7, 1–13. [Google Scholar] [CrossRef]
- Zhang, M.; Chen, X.; Radacsi, N. New tricks of old drugs: Repurposing non-chemo drugs and dietary phytochemicals as adjuvants in anti-tumor therapies. J. Control. Release 2021, 329, 96–120. [Google Scholar] [CrossRef]
- Aung, T.N.; Qu, Z.; Kortschak, R.D.; Adelson, D.L. Understanding the effectiveness of natural compound mixtures in cancer through their molecular mode of action. Int. J. Mol. Sci. 2017, 18, 656. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.Q.; Bi, H.; Feng, J.Q.; Cao, J.G. Effect of curcumin on multidrug resistance in resistant human gastric carcinoma cell line SGC7901/VCR. Acta Pharmacol. Sin. 2005, 26, 1009–1016. [Google Scholar] [CrossRef]
- Ganta, S.; Amiji, M. Coadministration of paclitaxel and curcumin in nanoemulsion formulations to overcome multidrug resistance in tumor cells. Mol. Pharm. 2009, 6, 928–939. [Google Scholar] [CrossRef]
| Compounds | Source-Extraction | Mechanism Action | Clinical Development | Commercial Name |
|---|---|---|---|---|
| Vinca alkaloids | Catharanthus roseu (Leaves) Isolated by semi-synthetic routes | Inhibit the tubulin polymerization of tumor cells and also cause mitotic spindle destruction | In clinical use; combination trials | Vinorelbine, Vincristine, Vinblastine, Vindesine, Vinflunine, Vincamine, Vintafolide |
| Paclitaxel, docetaxel | Taxus spp. (Bark) Synthesis, semi-synthesis, and plant cell culture | Stabilization of microtubules and inhibition of depolymerization into tubulin, which stops the cell cycle in the G2/M phase leading to cell death | In clinical use; Phase I-III clinical trials; early treatment settings; non-small lung cancer, breast cancer, ovarian cancer, Kaposi sarcoma. Research and development in alternative drug administration using nanoparticles, naocochealtes and nanoliposomes. | Taxol®, Taxotere®, Abraxane®, Jevtana®, Taxoprexin®, Xytotax® |
| Camptotecin, irinotecan | Camptotheca acuminata (leaves) Water extraction | Binding to the TOP1 cleavage complex, leading to an accumulation of DNA strand breaks upon replication, causing apoptosis during the S phase of the cell cycle | Ovarian, lung, colorectal and pediatric cancer | Topotecan, irinotecan, belotecan |
| Podophyllotoxin and analogues | Podophyllum spp. (rhizome, roots) Alcohol extraction | Blockage of cell division metaphase of mitosis | Lymphomas and testicular cancer trials | No rentable |
| Roscovitine | Raphanus sativus (Radish) Chloroform extraction | Inhibition of cyclin dependent kinases; reduction of cell cycle progression | Phase II clinical trials in Europe | Roscovitine, seliciclib |
| Compound | Type of Cancer | Main Results | Refs. |
|---|---|---|---|
| Sulforaphane | Prostate | Reduction of prostate-specific antigen after prostatectomy/Lengthening of the on-treatment prostate-specific antigen doubling time. | [75,76] |
| Breast | Improved efficacy of doxorubicin, without any cytotoxic effect. | [77] | |
| Resveratrol | Colorectal | Induction of apoptosis in malignant cells in hepatic metastasis. | [78] |
| Breast | Reduction of DNA methylation of RASSF-1α and prostaglandin E2 expression. | [79] | |
| Prostate | Reduction of cancer recurrence. | [80,81] | |
| Curcumin | Pancreas | Improved efficacy of gemcitabine, without any cytotoxic effect. | [82] |
| Breast | Improved efficacy of paclitaxel, without any cytotoxic effect. | [83] | |
| Quercetin | Gastric | High dietary intake is inversely related to the risk of cancer development. | [84] |
| Gingerol | Colorectal | Reduction of the risk of cancer development. | [85] |
| Solid tumor | Enhanced antioxidant status of patients receiving chemotherapy/Improvement of general quality of cancer patients receiving chemotherapy. | [86,87] | |
| Kaempferol | Ovarian | Reduction of the risk of cancer development. | [88,89] |
| Pancreatic |
| Plant | Compound | Structure | Results (IC50 µM) | Refs. |
|---|---|---|---|---|
| Aloe vera | Emodin | 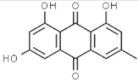 | In vitro: Breast (8.6); Lung (19.6); Liver (12.8) Induction of apoptosis, specific energy-dependent pathway of drug incorporation. | [157,158] |
| Artemisia annua | Artemisinin | 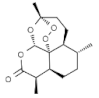 | In vitro: Lung (0.9) Production of reactive oxygen species, inhibition of cell cycle in G0/G1 phase, induction of apoptosis, inhibition of angiogenesis. | [159] |
| Artesunate | 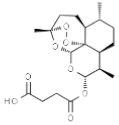 | In vitro: Breast (2.3); colon (1.9); lung (9.8); leukemia (2); ovarian (15.0) Antiangiogenic effect; inhibition of VEGF expression. | [160,161,162] | |
| Betula sp. | Betulinic acid |  | In vitro: Human leukemia (2.6); murine leukemia (2.1); cervix (2.5); colon (10.0); lung (4.3); skin (8.0); gastric (13.0) In vivo: (mice): Breast (13.5) Activation of extrinsic apoptosis pathway via up regulation of DR4, DR5 and PARP cleavage. | [163,164,165,166] |
| Betulin | 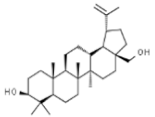 | In vitro: Breast (30.7); colon (45.2); lung (45.2); leukemia (14.5); skin (6.8); prostate (17.9) Synergistic effect with chemotherapeutic drugs. | [167] | |
| Berberis vulgaris | Berberine | 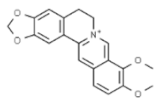 | In vitro: Breast (25)/In vivo: Breast (25.0); colon (25.0) Induction of cell cycle arrest, synergistic effect with drugs/Reduction of tumor volume and angiogensis in a dose-dependent manner. | [168,169] |
| Camellia sinensis | Epicatechin gallate |  | In vitro: Breast (350.0); colon (1000); lung (78.0) Induce apoptosis of cancer cell lines. | [170] |
| Epicatechin | 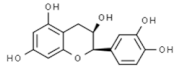 | In vitro: Colon (60); prostate (8.9); ovarian (7.9) Growth inhibition. | [171] | |
| Epigallocatechin | 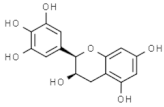 | In vitro: Breast (22.0); colon (75.0); lung (70.0) Growth inhibition. | [172] | |
| Combretum caffrum | Combretastatin | 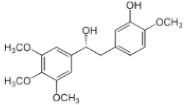 | In vivo (mice): Breast (80-190); colon (8.4;) lung (7.1); leukemia (1.9) β-tubulin binding, causing the destabilization of the microtubules. | [161,173] |
| Crocus sativus | Crocetin | 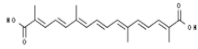 | In vitro: Colon (0.2); lung (0.4); liver (0.6) Activation of Nrf2, reduction of lactate dehydrogenase A expression. | [174] |
| Crocin | 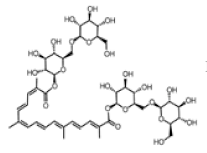 | Inhibition of cancer cells’ proliferation or/and induction of apoptosis, both in vitro and in vivo studies. | [174,175,176] | |
| Safranal | 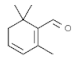 | In vitro: Colon (20.0) Apoptosis and G2/M cell cycle arrest mediated by suppression of mTOR/PI3K/Akt pathway. | [177] | |
| Euphorbia peplus | Ingenol mebutate | 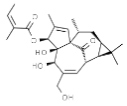 | In vitro: Breast (23.9); colon (22.2); lung (15.2); skin (200.0); prostate (29.8) Necrotic mechanism. | [178,179] |
| Gardenia jasminoides | Geniposide |  | In vitro: Lung (351.5); medullobalstome (293.5) Activation of mitochondrial execution pathway by Caspase-9 and -3; increase levels of Bax in response to p38MAPK signaling initiation of the mitochondrial death cascade. | [161,180] |
| Genipin | 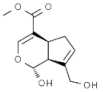 | Inhibitor of mitochondrial uncoupling protein 2, induction caspase-dependent apoptosis in vitro and in in vivo models. | [181] | |
| Ginkgo biloba | Ginkgetin | 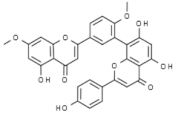 | In vivo (mouse): Breast 10.0; colon 5.0; lung 10.0; prostate 5.0 p62/SQSTM1-mediated autolysosome formation and redox setting; inhibition of signal transducer and activator of transcription 3 activity. | [182,183,184] |
| Bilobetin | 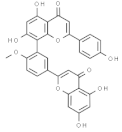 | In vitro: Breast 57.6; lung 36.4; leukemia 46.0 Arresting the G2/M phase of the cell cycle. | [185] | |
| Isoginkgetin | 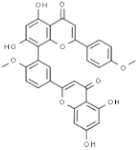 | In vitro: Breast 91.2; lung 43.0; skin 18.8 Arresting the G2/M phase of the cell cycle. | [185] | |
| Glycyrrhiza glabra | Licochalcone | 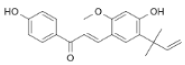 | In vitro: Lung (30.0); skin (50.0); liver (44.13); gastric (42) Increased mRNA expression of DR3, DR5, caspases-3, caspases-8, caspases-10, Fas, Bad, Bax, Bcl-2, Bak, and PUMA; decreased level of PKCε, p70S6K, Akt. | [186,187] |
| Licoagrochalcone | 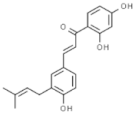 | In vitro: Breast (28.6;) colon (25.4); leukemia (12.2); kidney (35.21) Induction of apoptosis, inhibition of proliferation. | [188] | |
| Matricaria chamomilla | Apigenin | 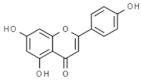 | In vitro: Breast (100;) lung (93.7); liver (38.9) Up-regulation of DR5 pathway. | [189] |
| Chamomillol | 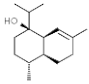 | In vitro: Breast (300.0); colon (165.0); prostate (200.0); liver (300.0) Regulation angiogenesis by down regulating expression proteins. | [190,191] | |
| Panax ginseng | Panaxadiol |  | In vitro: Colon (25.0); liver (1.2) Regulation of cell cycle transition and the induction of apoptotic cells. | [192] |
| Ginsenoside | 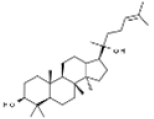 | In vitro: Breast (30.0); colon (100.0); lung (50.0); skin (25–100) Induction of apoptosis, inhibition of proliferation. | [193] | |
| Papaver somniferum | Noscapine |  | In vivo (mice): Breast (45.0); lung (73.0); leukemia (20.0); pancreas (70.0) Activating multiple signaling pathways including apoptosis. | [194] |
| Persea americana | Borbonol | 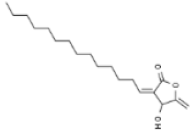 | In vitro: Breast (20.5) Inhibition of proliferation. | [195] |
| Salvia prionitis | Salvicine | 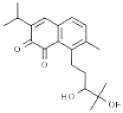 | In vitro: Breast (1.4); leukemia (3.6); stomach (1.8) Breaks two strands of DNA by facilitating TOP2 activity; inhibits re-ligation | [196,197,198] |
| Cryptotanshinone | 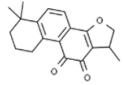 | In vivo (mice): Breast (1.1;) lung (20.0); leukemia (4.8) Multispecific modes of actions: G2/M arrest, apoptosis, inhibition of cellular movement. Inhibition of NFκB orchestrates all these mechanisms. | [199,200,201] | |
| Salvianolic acid | 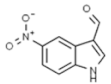 | In vitro studies. Prompting apoptosis, disturbance of cell cycle and adjourning metastasis. | [202] | |
| Silybum marianum | Silibinin | 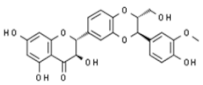 | In vitro: Breast (24.0); skin (100.0); prostate (55.6); ovarian (25.0) Induction of apoptosis and cell cycle arrest. | [203,204,205] |
| Silymarin | 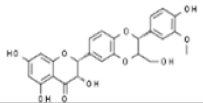 | In vitro: Breast (75); prostate (67.9) Interference with expressions of cell cycle regulators and proteins involved in apoptosis; induction of cell cycle arrest. | [203,206,207] | |
| Zingiber officinale | Paradol | 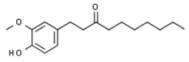 | In vitro: Breast (20.4;) colon (10.4); leukemia (100); liver (>100). Reduction of cell viability. | [208,209] |
| Shogaol |  | In vitro: Breast (24.4); colon (3.1); lung (25.8); liver (18.7) Inhibitory activity. | [209,210] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Oliveira, P.; Otero, P.; Pereira, A.G.; Chamorro, F.; Carpena, M.; Echave, J.; Fraga-Corral, M.; Simal-Gandara, J.; Prieto, M.A. Status and Challenges of Plant-Anticancer Compounds in Cancer Treatment. Pharmaceuticals 2021, 14, 157. https://doi.org/10.3390/ph14020157
Garcia-Oliveira P, Otero P, Pereira AG, Chamorro F, Carpena M, Echave J, Fraga-Corral M, Simal-Gandara J, Prieto MA. Status and Challenges of Plant-Anticancer Compounds in Cancer Treatment. Pharmaceuticals. 2021; 14(2):157. https://doi.org/10.3390/ph14020157
Chicago/Turabian StyleGarcia-Oliveira, Paula, Paz Otero, Antia Gonzalez Pereira, Franklin Chamorro, Maria Carpena, Javier Echave, Maria Fraga-Corral, Jesus Simal-Gandara, and Miguel Angel Prieto. 2021. "Status and Challenges of Plant-Anticancer Compounds in Cancer Treatment" Pharmaceuticals 14, no. 2: 157. https://doi.org/10.3390/ph14020157
APA StyleGarcia-Oliveira, P., Otero, P., Pereira, A. G., Chamorro, F., Carpena, M., Echave, J., Fraga-Corral, M., Simal-Gandara, J., & Prieto, M. A. (2021). Status and Challenges of Plant-Anticancer Compounds in Cancer Treatment. Pharmaceuticals, 14(2), 157. https://doi.org/10.3390/ph14020157












