Brain Perivascular Macrophages Do Not Mediate Interleukin-1-Induced Sickness Behavior in Rats
Abstract
1. Introduction
2. Results
2.1. Ip IL-1 β-Alexa Conjugate Decreased Food Intake
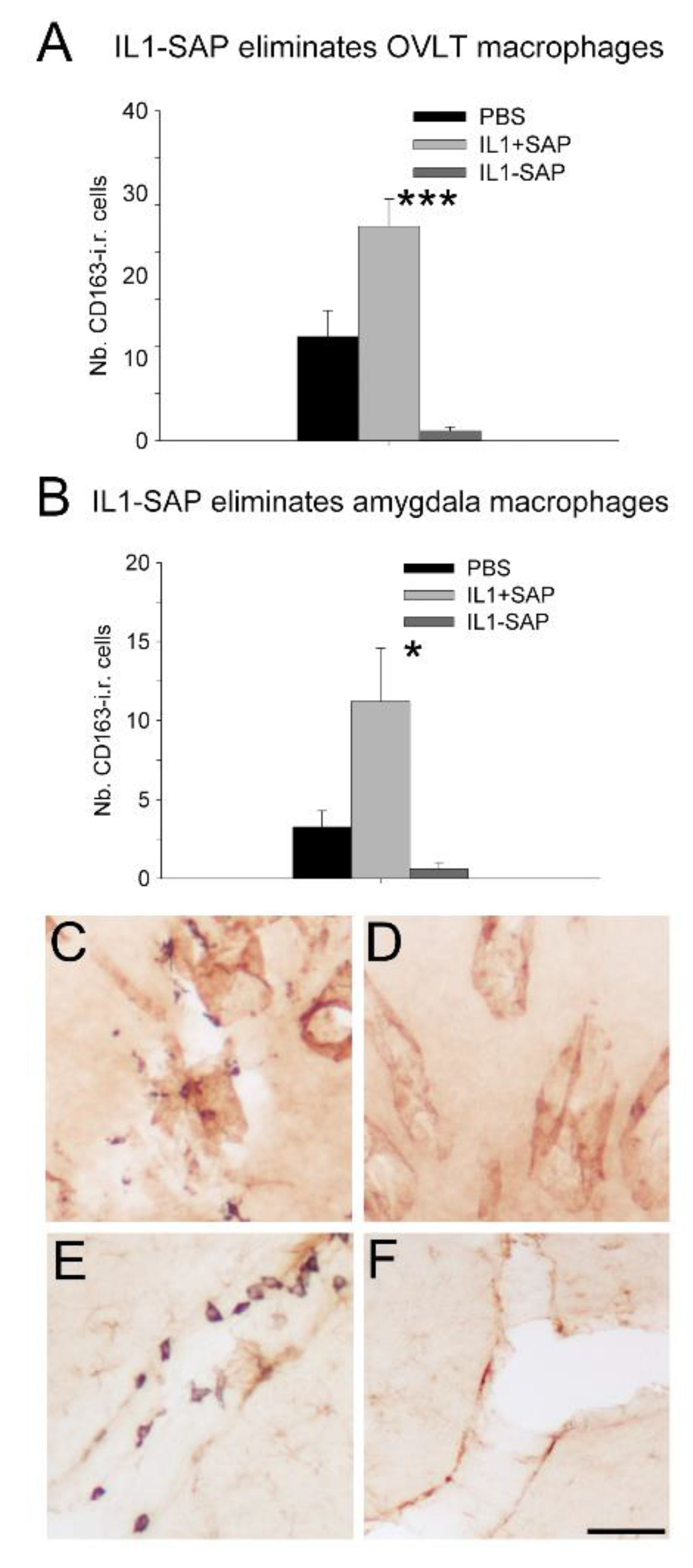
2.2. Icv IL-1β-Saporin Conjugate Eliminated Brain Perivascular Macrophages, but Not Endothelial Cells
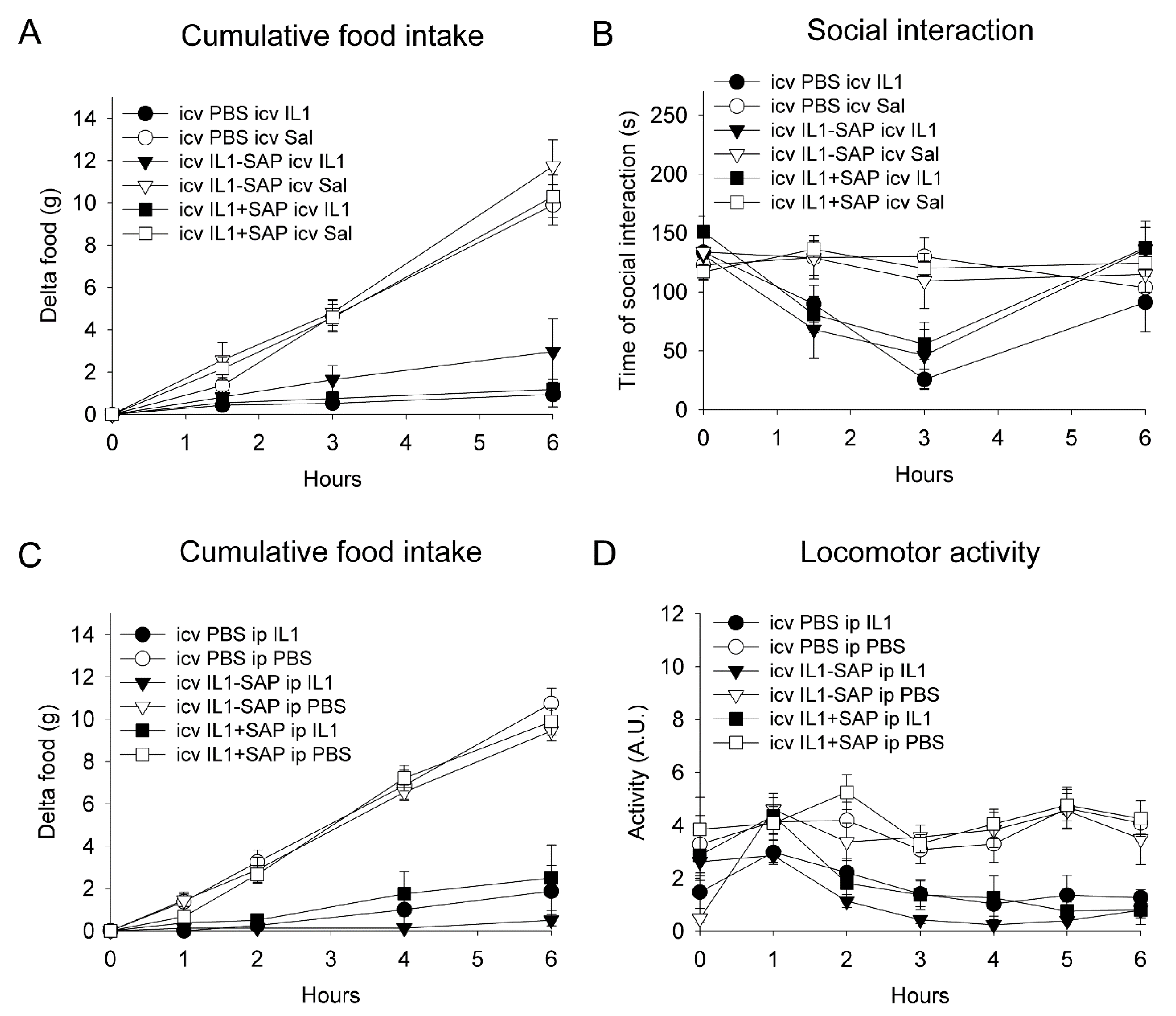
2.3. Icv IL-1β-Saporin Conjugate Did Not Alter Behavioral Responses to Subsequent IL-1β Injection
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Biologically Active Molecules
4.3. Sickness Behavior Testing after Intraperitoneal Injection of Interleukin-1β-Alexa-488
4.4. Intracerebroventricular Injection of Interleukin-1β ± Saporin
4.5. Sickness Behavior Assessment after Intracerebroventricular Interleukin-1β Injection
4.6. Sickness Behavior Assessment after Intraperitoneal Interleukin-1β Injection
4.7. Brain Tissue Preparation
4.8. Immunohistochemistry
4.9. Microscopy
4.10. Data Presentation and Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Hart, B.L. Biological basis of the behavior of sick animals. Neurosci. Biobehav. Rev. 1988, 12, 123–137. [Google Scholar] [CrossRef]
- Sylvia, K.E.; Demas, G.E. A return to wisdom: Using sickness behaviors to integrate ecological and translational research. Integr. Comp. Biol. 2017, 57, 1204–1213. [Google Scholar] [CrossRef]
- Lasselin, J.; Schedlowski, M.; Karshikoff, B.; Engler, H.; Lekander, M.; Konsman, J.P. Comparison of bacterial lipopolysaccharide-induced sickness behavior in rodents and humans: Relevance for symptoms of anxiety and depression. Neurosci. Biobehav. Rev. 2020, 115, 15–24. [Google Scholar] [CrossRef]
- Rietschel, E.T.; Schletter, J.; Weidemann, B.; El-Samalouti, V.; Mattern, T.; Zähringer, U.; Seydel, U.; Brade, H.; Flad, H.D.; Kusumoto, S.; et al. Lipopolysaccharide and peptidoglycan: Cd14-dependent bacterial inducers of inflammation. Microb. Drug Resist. 1998, 4, 37–44. [Google Scholar] [CrossRef]
- Pedra, J.H.; Cassel, S.L.; Sutterwala, F.S. Sensing pathogens and danger signals by the inflammasome. Curr. Opin. Immunol. 2009, 21, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Dinarello, C.A. Il-1: Discoveries, controversies and future directions. Eur. J. Immunol. 2010, 40, 599–606. [Google Scholar] [CrossRef] [PubMed]
- McCarthy, D.O.; Kluger, M.J.; Vander, A.J. Suppression of food intake during infection: Is interleukin-1 involved? Am. J. Clin. Nutr. 1985, 42, 1179–1182. [Google Scholar] [CrossRef] [PubMed]
- Moldawer, L.L.; Andersson, C.; Gelin, J.; Lundholm, K.G. Regulation of food intake and hepatic protein synthesis by recombinant-derived cytokines. Am. J. Physiol. 1988, 254, G450–G456. [Google Scholar] [CrossRef] [PubMed]
- Hellerstein, M.K.; Meydani, S.N.; Meydani, M.; Wu, K.; Dinarello, C.A. Interleukin-1-induced anorexia in the rat. Influence of prostaglandins. J. Clin. Investig. 1989, 84, 228–235. [Google Scholar] [CrossRef]
- Crestani, F.; Seguy, F.; Dantzer, R. Behavioural effects of peripherally injected interleukin-1: Role of prostaglandins. Brain Res. 1991, 542, 330–335. [Google Scholar] [CrossRef]
- Bluthé, R.M.; Dantzer, R.; Kelley, K.W. Effects of interleukin-1 receptor antagonist on the behavioral effects of lipopolysaccharide in rat. Brain Res. 1992, 573, 318–320. [Google Scholar] [CrossRef]
- Swiergiel, A.H.; Smagin, G.N.; Johnson, L.J.; Dunn, A.J. The role of cytokines in the behavioral responses to endotoxin and influenza virus infection in mice: Effects of acute and chronic administration of the interleukin-1-receptor antagonist (il-1ra). Brain Res. 1997, 776, 96–104. [Google Scholar] [CrossRef]
- Konsman, J.P.; Parnet, P.; Dantzer, R. Cytokine-induced sickness behaviour: Mechanisms and implications. Trends Neurosci. 2002, 25, 154–159. [Google Scholar] [CrossRef]
- Ching, S.; Zhang, H.; Belevych, N.; He, L.; Lai, W.; Pu, X.A.; Jaeger, L.B.; Chen, Q.; Quan, N. Endothelial-specific knockdown of interleukin-1 (IL-1) type 1 receptor differentially alters CNS responses to IL-1 depending on its route of administration. J. Neurosci. 2007, 27, 10476–10486. [Google Scholar] [CrossRef]
- Chaskiel, L.; Bristow, A.D.; Bluthe, R.M.; Dantzer, R.; Blomqvist, A.; Konsman, J.P. Interleukin-1 reduces food intake and body weight in rat by acting in the arcuate hypothalamus. Brain Behav. Immun. 2019, 81, 560–573. [Google Scholar] [CrossRef]
- Cheunsuang, O.; Stewart, A.L.; Morris, R. Differential uptake of molecules from the circulation and csf reveals regional and cellular specialisation in cns detection of homeostatic signals. Cell Tissue Res. 2006, 325, 397–402. [Google Scholar] [CrossRef] [PubMed]
- Morita, S.; Miyata, S. Different vascular permeability between the sensory and secretory circumventricular organs of adult mouse brain. Cell Tissue Res. 2012, 349, 589–603. [Google Scholar] [CrossRef]
- Konsman, J.P.; Vigues, S.; Mackerlova, L.; Bristow, A.; Blomqvist, A. Rat brain vascular distribution of interleukin-1 type-1 receptor immunoreactivity: Relationship to patterns of inducible cyclooxygenase expression by peripheral inflammatory stimuli. J. Comp. Neurol. 2004, 472, 113–129. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Nemeth, D.P.; McKim, D.B.; Zhu, L.; DiSabato, D.J.; Berdysz, O.; Gorantla, G.; Oliver, B.; Witcher, K.G.; Wang, Y.; et al. Cell-type-specific interleukin 1 receptor 1 signaling in the brain regulates distinct neuroimmune activities. Immunity 2019, 50, 317–333 e316. [Google Scholar] [CrossRef] [PubMed]
- Van Dam, A.M.; Brouns, M.; Louisse, S.; Berkenbosch, F. Appearance of interleukin-1 in macrophages and in ramified microglia in the brain of endotoxin-treated rats: A pathway for the induction of non-specific symptoms of sickness? Brain Res. 1992, 588, 291–296. [Google Scholar] [CrossRef]
- Konsman, J.P.; Kelley, K.; Dantzer, R. Temporal and spatial relationships between lipopolysaccharide-induced expression of fos, interleukin-1beta and inducible nitric oxide synthase in rat brain. Neuroscience 1999, 89, 535–548. [Google Scholar] [CrossRef]
- Quan, N.; Sundar, S.K.; Weiss, J.M. Induction of interleukin-1 in various brain regions after peripheral and central injections of lipopolysaccharide. J. Neuroimmunol. 1994, 49, 125–134. [Google Scholar] [CrossRef]
- Konsman, J.P.; Tridon, V.; Dantzer, R. Diffusion and action of intracerebroventricularly injected interleukin-1 in the CNS. Neuroscience 2000, 101, 957–967. [Google Scholar] [CrossRef]
- Knoll, J.G.; Krasnow, S.M.; Marks, D.L. Interleukin-1beta signaling in fenestrated capillaries is sufficient to trigger sickness responses in mice. J. Neuroinflammation 2017, 14, 219. [Google Scholar] [CrossRef] [PubMed]
- Spadaro, F.; Dunn, A.J. Intracerebroventricular administration of interleukin-1 to mice alters investigation of stimuli in a novel environment. Brain Behav. Immun. 1990, 4, 308–322. [Google Scholar] [CrossRef]
- Kent, S.; Bluthe, R.M.; Dantzer, R.; Hardwick, A.J.; Kelley, K.W.; Rothwell, N.J.; Vannice, J.L. Different receptor mechanisms mediate the pyrogenic and behavioral effects of interleukin 1. Proc. Natl. Acad. Sci. USA 1992, 89, 9117–9120. [Google Scholar] [CrossRef]
- Plata-Salamán, C.R. Meal patterns in response to the intracerebroventricular administration of interleukin-1 beta in rats. Physiol. Behav. 1994, 55, 727–733. [Google Scholar] [CrossRef]
- Schiltz, J.C.; Sawchenko, P.E. Distinct brain vascular cell types manifest inducible cyclooxygenase expression as a function of the strength and nature of immune insults. J. Neurosci. 2002, 22, 5606–5618. [Google Scholar] [CrossRef]
- Schnurch, H.; Risau, W. Expression of tie-2, a member of a novel family of receptor tyrosine kinases, in the endothelial cell lineage. Development 1993, 119, 957–968. [Google Scholar] [CrossRef]
- Gomez Perdiguero, E.; Klapproth, K.; Schulz, C.; Busch, K.; Azzoni, E.; Crozet, L.; Garner, H.; Trouillet, C.; de Bruijn, M.F.; Geissmann, F.; et al. Tissue-resident macrophages originate from yolk-sac-derived erythro-myeloid progenitors. Nature 2015, 518, 547–551. [Google Scholar] [CrossRef]
- Nishiyori, A.; Minami, M.; Ohtani, Y.; Takami, S.; Yamamoto, J.; Kawaguchi, N.; Kume, T.; Akaike, A.; Satoh, M. Localization of fractalkine and Cx3cr1 mrnas mRNAs in rat brain: Does fractalkine play a role in signaling from neuron to microglia? FEBS Lett. 1998, 429, 167–172. [Google Scholar] [CrossRef]
- Goldmann, T.; Wieghofer, P.; Jordao, M.J.; Prutek, F.; Hagemeyer, N.; Frenzel, K.; Amann, L.; Staszewski, O.; Kierdorf, K.; Krueger, M.; et al. Origin, fate and dynamics of macrophages at central nervous system interfaces. Nat. Immunol. 2016, 17, 797–805. [Google Scholar] [CrossRef]
- Sugiyama, D.; Kusuhara, H.; Taniguchi, H.; Ishikawa, S.; Nozaki, Y.; Aburatani, H.; Sugiyama, Y. Functional characterization of rat brain-specific organic anion transporter (oatp14) at the blood-brain barrier: High affinity transporter for thyroxine. J. Biol. Chem. 2003, 278, 43489–43495. [Google Scholar] [CrossRef] [PubMed]
- Roberts, L.M.; Woodford, K.; Zhou, M.; Black, D.S.; Haggerty, J.E.; Tate, E.H.; Grindstaff, K.K.; Mengesha, W.; Raman, C.; Zerangue, N. Expression of the thyroid hormone transporters monocarboxylate transporter-8 (slc16a2) and organic ion transporter-14 (slco1c1) at the blood-brain barrier. Endocrinology 2008, 149, 6251–6261. [Google Scholar] [CrossRef] [PubMed]
- Lang, M.F.; Salinin, S.; Ridder, D.A.; Kleesiek, J.; Hroudova, J.; Berger, S.; Schutz, G.; Schwaninger, M. A transgenic approach to identify thyroxine transporter-expressing structures in brain development. J. Neuroendocrinol. 2011, 23, 1194–1203. [Google Scholar] [CrossRef]
- Faraco, G.; Park, L.; Anrather, J.; Iadecola, C. Brain perivascular macrophages: Characterization and functional roles in health and disease. J. Mol. Med. 2017, 95, 1143–1152. [Google Scholar] [CrossRef]
- Prinz, M.; Masuda, T.; Wheeler, M.A.; Quintana, F.J. Microglia and central nervous system-associated macrophages-from origin to disease modulation. Annu. Rev. Immunol. 2021, 39, 251–277. [Google Scholar] [CrossRef]
- Yang, T.; Guo, R.; Zhang, F. Brain perivascular macrophages: Recent advances and implications in health and diseases. CNS Neurosci. Ther. 2019, 25, 1318–1328. [Google Scholar] [CrossRef]
- Ferreras, J.M.; Barbieri, L.; Girbes, T.; Battelli, M.G.; Rojo, M.A.; Arias, F.J.; Rocher, M.A.; Soriano, F.; Mendez, E.; Stirpe, F. Distribution and properties of major ribosome-inactivating proteins (28 s rRNA n-glycosidases) of the plant saponaria officinalis l. (caryophyllaceae). Biochim. Biophys. Acta 1993, 1216, 31–42. [Google Scholar] [CrossRef]
- Santanche, S.; Bellelli, A.; Brunori, M. The unusual stability of saporin, a candidate for the synthesis of immunotoxins. Biochem. Biophys. Res. Commun. 1997, 234, 129–132. [Google Scholar] [CrossRef]
- Wiley, R.G. Targeting toxins to neural antigens and receptors. Methods Mol. Biol. 2001, 166, 267–276. [Google Scholar]
- Munafo, M.R.; Davey Smith, G. Robust research needs many lines of evidence. Nature 2018, 553, 399–401. [Google Scholar] [CrossRef]
- Zhao, X.F.; Alam, M.M.; Liao, Y.; Huang, T.; Mathur, R.; Zhu, X.; Huang, Y. Targeting microglia using Cx3cr1-Cre lines: Revisiting the specificity. eNeuro 2019, 6. [Google Scholar] [CrossRef]
- Polfliet, M.M.; Goede, P.H.; van Kesteren-Hendrikx, E.M.; van Rooijen, N.; Dijkstra, C.D.; van den Berg, T.K. A method for the selective depletion of perivascular and meningeal macrophages in the central nervous system. J. Neuroimmunol. 2001, 116, 188–195. [Google Scholar] [CrossRef]
- Michels, M.; Avila, P.; Pescador, B.; Vieira, A.; Abatti, M.; Cucker, L.; Borges, H.; Goulart, A.I.; Junior, C.C.; Barichello, T.; et al. Microglial cells depletion increases inflammation and modifies microglial phenotypes in an animal model of severe sepsis. Mol. Neurobiol. 2019, 56, 7296–7304. [Google Scholar] [CrossRef]
- Takagi, S.; Murayama, S.; Torii, K.; Takemura-Morita, S.; Kurganov, E.; Nagaoka, S.; Wanaka, A.; Miyata, S. Depletion of microglia and macrophages with clodronate liposomes attenuates zymosan-induced fos expression and hypothermia in the adult mouse. J. Neuroimmunol. 2020, 344, 577244. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.C.; Corey, S.; Westmoreland, S.V.; Pauley, D.; Knight, H.; DeBakker, C.; Alvarez, X.; Lackner, A.A. Perivascular macrophages are the primary cell type productively infected by simian immunodeficiency virus in the brains of macaques: Implications for the neuropathogenesis of aids. J. Exp. Med. 2001, 193, 905–915. [Google Scholar] [CrossRef] [PubMed]
- Kim, W.K.; Alvarez, X.; Fisher, J.; Bronfin, B.; Westmoreland, S.; McLaurin, J.; Williams, K. CD163 identifies perivascular macrophages in normal and viral encephalitic brains and potential precursors to perivascular macrophages in blood. Am. J. Pathol. 2006, 168, 822–834. [Google Scholar] [CrossRef]
- Ransohoff, R.M.; Engelhardt, B. The anatomical and cellular basis of immune surveillance in the central nervous system. Nat. Rev. Immunol. 2012, 12, 623–635. [Google Scholar] [CrossRef]
- Konsman, J.P.; Veeneman, J.; Combe, C.; Poole, S.; Luheshi, G.N.; Dantzer, R. Central nervous action of interleukin-1 mediates activation of limbic structures and behavioural depression in response to peripheral administration of bacterial lipopolysaccharide. Eur. J. Neurosci. 2008, 28, 2499–2510. [Google Scholar] [CrossRef] [PubMed]
- Kanai, T.; Watanabe, M.; Okazawa, A.; Sato, T.; Yamazaki, M.; Okamoto, S.; Ishii, H.; Totsuka, T.; Iiyama, R.; Okamoto, R.; et al. Macrophage-derived IL-18-mediated intestinal inflammation in the murine model of crohn’s disease. Gastroenterology 2001, 121, 875–888. [Google Scholar] [CrossRef] [PubMed]
- Kanai, T.; Uraushihara, K.; Totsuka, T.; Nemoto, Y.; Fujii, R.; Kawamura, T.; Makita, S.; Sawada, D.; Yagita, H.; Okumura, K.; et al. Ameliorating effect of saporin-conjugated anti-CD11b monoclonal antibody in a murine T-cell-mediated chronic colitis. J. Gastroenterol. Hepatol. 2006, 21, 1136–1142. [Google Scholar] [CrossRef] [PubMed]
- Hozain, S.; Cottrell, J. CDllb+ targeted depletion of macrophages negatively affects bone fracture healing. Bone 2020, 138, 115479. [Google Scholar] [CrossRef]
- Anforth, H.R.; Bluthe, R.M.; Bristow, A.; Hopkins, S.; Lenczowski, M.J.; Luheshi, G.; Lundkvist, J.; Michaud, B.; Mistry, Y.; Van Dam, A.M.; et al. Biological activity and brain actions of recombinant rat interleukin-1alpha and interleukin-1beta. Eur. Cytokine Netw. 1998, 9, 279–288. [Google Scholar]
- Terao, A.; Matsumura, H.; Saito, M. Interleukin-1 induces slow-wave sleep at the prostaglandin D2-sensitive sleep-promoting zone in the rat brain. J. Neurosci. 1998, 18, 6599–6607. [Google Scholar] [CrossRef]
- Inoue, A.; Ikoma, K.; Morioka, N.; Kumagai, K.; Hashimoto, T.; Hide, I.; Nakata, Y. Interleukin-1beta induces substance p release from primary afferent neurons through the cyclooxygenase-2 system. J. Neurochem. 1999, 73, 2206–2213. [Google Scholar] [PubMed]
- Serou, M.J.; DeCoster, M.A.; Bazan, N.G. Interleukin-1 beta activates expression of cyclooxygenase-2 and inducible nitric oxide synthase in primary hippocampal neuronal culture: Platelet-activating factor as a preferential mediator of cyclooxygenase-2 expression. J. Neurosci. Res. 1999, 58, 593–598. [Google Scholar] [CrossRef]
- Boraschi, D.; Bossu, P.; Macchia, G.; Ruggiero, P.; Tagliabue, A. Structure-function relationship in the IL-1 family. Front. Biosci. 1996, 1, d270–d308. [Google Scholar] [CrossRef][Green Version]
- Van Dijk, G.; Strubbe, J.H. Time-dependent effects of neuropeptide Y infusion in the paraventricular hypothalamus on ingestive and associated behaviors in rats. Physiol. Behav. 2003, 79, 575–580. [Google Scholar] [CrossRef]
- Bolognesi, A.; Tazzari, P.L.; Olivieri, F.; Polito, L.; Lemoli, R.; Terenzi, A.; Pasqualucci, L.; Falini, B.; Stirpe, F. Evaluation of immunotoxins containing single-chain ribosome-inactivating proteins and an anti-CD22 monoclonal antibody (om124): In vitro and in vivo studies. Br. J. Haematol. 1998, 101, 179–188. [Google Scholar] [CrossRef]
- Pistillo, M.P.; Tazzari, P.L.; Palmisano, G.L.; Pierri, I.; Bolognesi, A.; Ferlito, F.; Capanni, P.; Polito, L.; Ratta, M.; Pileri, S.; et al. CTLA-4 is not restricted to the lymphoid cell lineage and can function as a target molecule for apoptosis induction of leukemic cells. Blood 2003, 101, 202–209. [Google Scholar] [CrossRef] [PubMed]
- Scremin, O.U. Cerebral vascular system. In The Rat Nervous System; Paxinos, G., Ed.; Academic Press: San Diego, CA, USA, 1995; pp. 3–35. [Google Scholar]
- Mato, M.; Ookawara, S.; Sakamoto, A.; Aikawa, E.; Ogawa, T.; Mitsuhashi, U.; Masuzawa, T.; Suzuki, H.; Honda, M.; Yazaki, Y.; et al. Involvement of specific macrophage-lineage cells surrounding arterioles in barrier and scavenger function in brain cortex. Proc. Natl. Acad. Sci. USA 1996, 93, 3269–3274. [Google Scholar] [CrossRef] [PubMed]
- Ookawara, S.; Mitsuhashi, U.; Suminaga, Y.; Mato, M. Study on distribution of pericyte and fluorescent granular perithelial (fgp) cell in the transitional region between arteriole and capillary in rat cerebral cortex. Anat. Rec. 1996, 244, 257–264. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaskiel, L.; Dantzer, R.; Konsman, J.P. Brain Perivascular Macrophages Do Not Mediate Interleukin-1-Induced Sickness Behavior in Rats. Pharmaceuticals 2021, 14, 1030. https://doi.org/10.3390/ph14101030
Chaskiel L, Dantzer R, Konsman JP. Brain Perivascular Macrophages Do Not Mediate Interleukin-1-Induced Sickness Behavior in Rats. Pharmaceuticals. 2021; 14(10):1030. https://doi.org/10.3390/ph14101030
Chicago/Turabian StyleChaskiel, Léa, Robert Dantzer, and Jan Pieter Konsman. 2021. "Brain Perivascular Macrophages Do Not Mediate Interleukin-1-Induced Sickness Behavior in Rats" Pharmaceuticals 14, no. 10: 1030. https://doi.org/10.3390/ph14101030
APA StyleChaskiel, L., Dantzer, R., & Konsman, J. P. (2021). Brain Perivascular Macrophages Do Not Mediate Interleukin-1-Induced Sickness Behavior in Rats. Pharmaceuticals, 14(10), 1030. https://doi.org/10.3390/ph14101030





