Combination Therapies with PRRT
Abstract
:1. Introduction
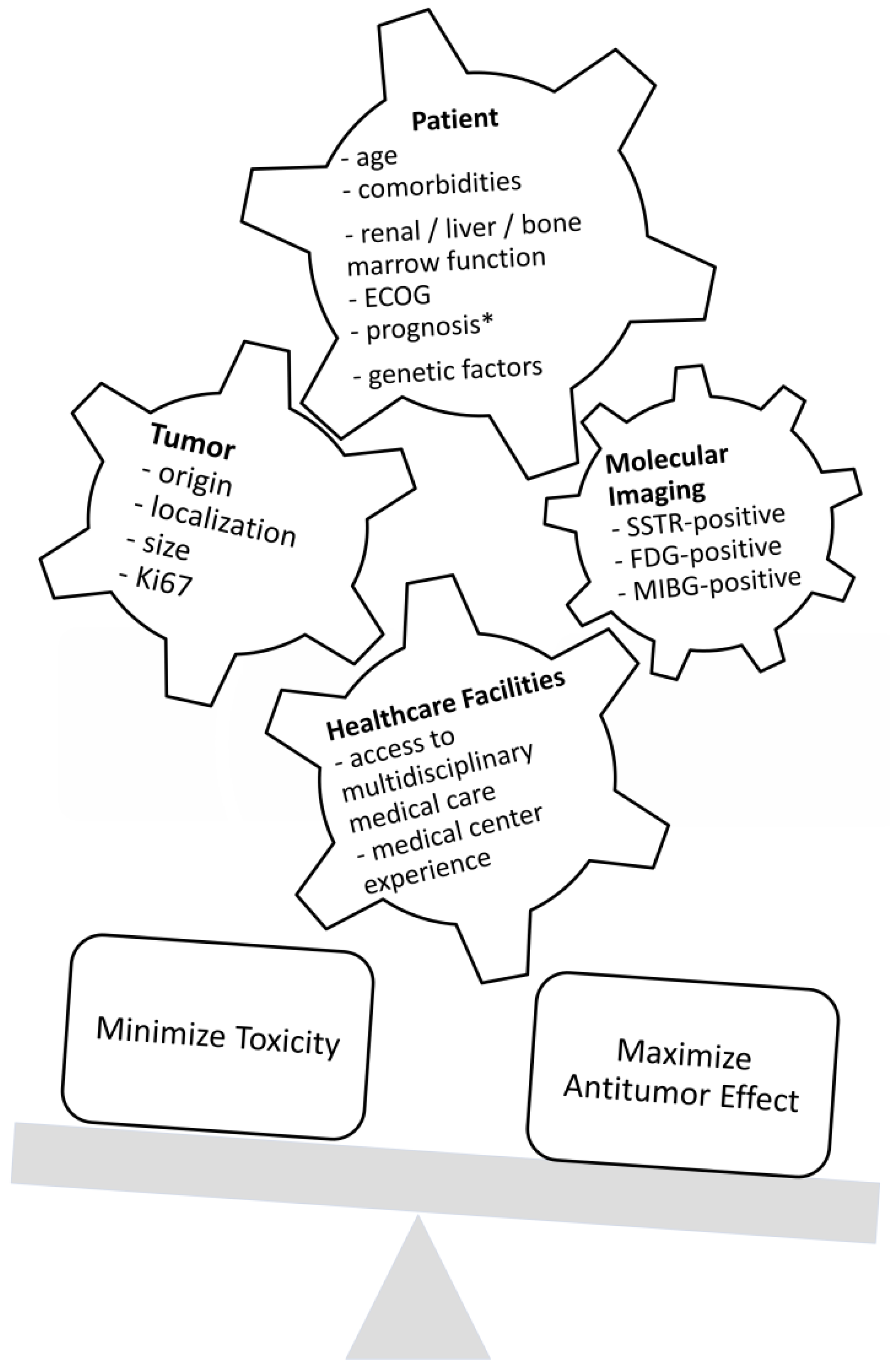
Combination Treatment Decision Making
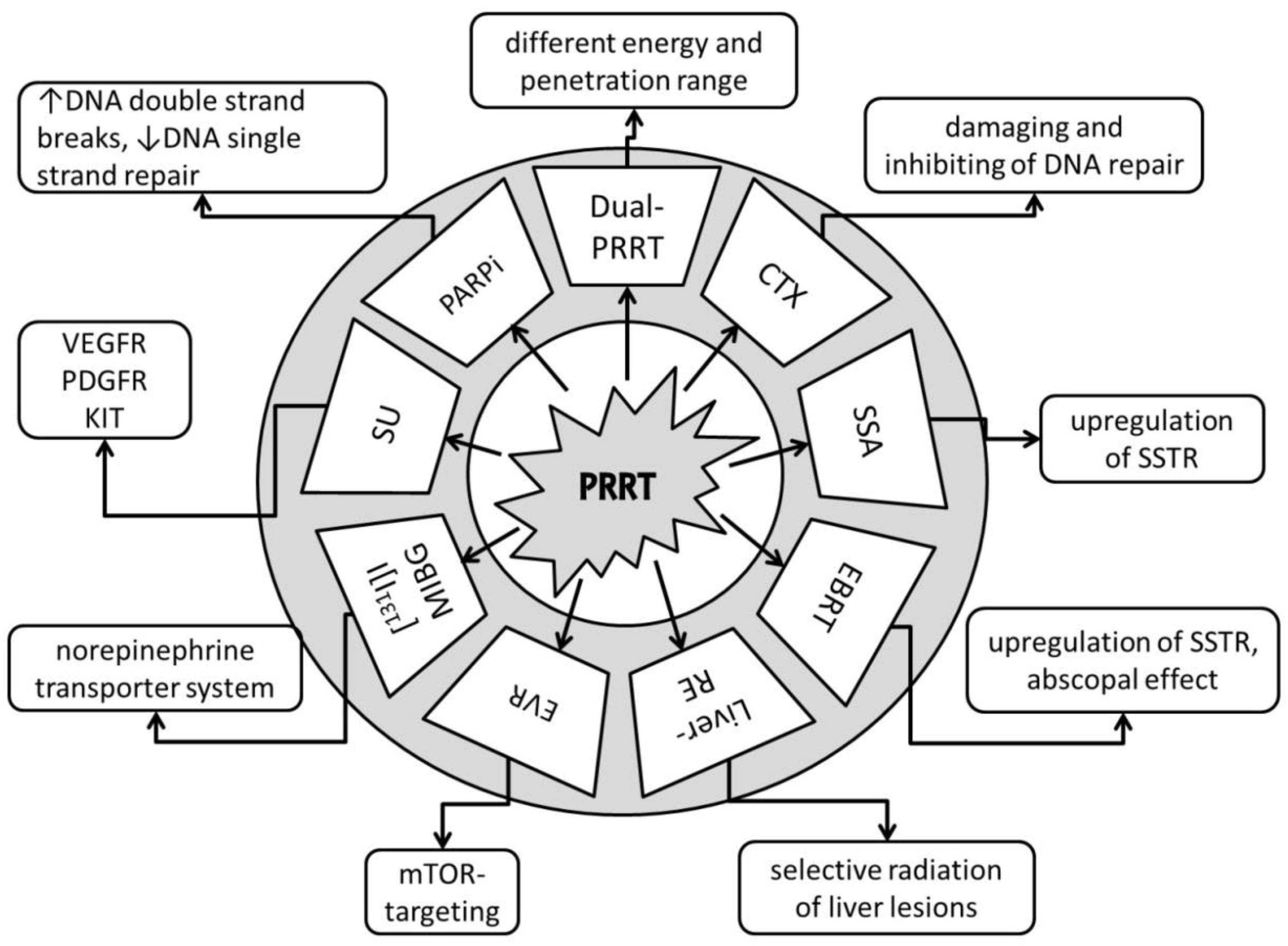
2. Dual PRRT
3. Chemotherapy
4. Somatostatin Analogues
5. Fractionated External Beam Radiotherapy
6. Liver Radioembolization
7. Everolimus
8. [131. I]I-MIBG
9. Promising Future Combination Therapies
10. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Das, S.; Dasari, A. Epidemiology, Incidence, and Prevalence of Neuroendocrine Neoplasms: Are There Global Differences? Curr. Oncol. Rep. 2021, 23, 43. [Google Scholar] [CrossRef] [PubMed]
- Riihimäki, M.; Hemminki, A.; Sundquist, K.; Sundquist, J.; Hemminki, K. The epidemiology of metastases in neuroendocrine tumors. Int. J. Cancer 2016, 139, 2679–2686. [Google Scholar] [CrossRef] [PubMed]
- Pedraza-Arévalo, S.; Gahete, M.D.; Alors-Pérez, E.; Luque, R.M.; Castaño, J.P. Multilayered heterogeneity as an intrinsic hallmark of neuroendocrine tumors. Rev. Endocr. Metab. Disord. 2018, 19, 179–192. [Google Scholar] [CrossRef] [PubMed]
- Grozinsky-Glasberg, S.; Shimon, I.; Korbonits, M.; Grossman, A.B. Somatostatin analogues in the control of neuroendocrine tumours: Efficacy and mechanisms. Endocr. Relat. Cancer 2008, 15, 701–720. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oberg, K.; Casanovas, O.; Castaño, J.P.; Chung, D.; Delle Fave, G.; Denèfle, P.; Harris, P.; Khan, M.S.; Kulke, M.H.; Scarpa, A.; et al. Molecular pathogenesis of neuroendocrine tumors: Implications for current and future therapeutic approaches. Clin. Cancer Res. 2013, 19, 2842–2849. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adant, S.; Shah, G.M.; Beauregard, J.-M. Combination treatments to enhance peptide receptor radionuclide therapy of neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 907–921. [Google Scholar] [CrossRef]
- Hennrich, U.; Kopka, K. Lutathera®: The First FDA- and EMA-Approved Radiopharmaceutical for Peptide Receptor Radionuclide Therapy. Pharmaceuticals 2019, 12, 114. [Google Scholar] [CrossRef] [Green Version]
- Strosberg, J.; El-Haddad, G.; Wolin, E.; Hendifar, A.; Yao, J.; Chasen, B.; Mittra, E.; Kunz, P.L.; Kulke, M.H.; Jacene, H.; et al. Phase 3 Trial of (177)Lu-Dotatate for Midgut Neuroendocrine Tumors. N. Engl. J. Med. 2017, 376, 125–135. [Google Scholar] [CrossRef]
- Strosberg, J.; Wolin, E.; Chasen, B.; Kulke, M.; Bushnell, D.; Caplin, M.; Baum, R.P.; Kunz, P.; Hobday, T.; Hendifar, A.; et al. Health-Related Quality of Life in Patients With Progressive Midgut Neuroendocrine Tumors Treated With (177)Lu-Dotatate in the Phase III NETTER-1 Trial. J. Clin. Oncol. 2018, 36, 2578–2584. [Google Scholar] [CrossRef]
- Sabet, A.; Haslerud, T.; Pape, U.-F.; Sabet, A.; Ahmadzadehfar, H.; Grünwald, F.; Guhlke, S.; Biersack, H.-J.; Ezziddin, S. Outcome and toxicity of salvage therapy with 177Lu-octreotate in patients with metastatic gastroenteropancreatic neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 205–210. [Google Scholar] [CrossRef]
- Yordanova, A.; Mayer, K.; Brossart, P.; Gonzalez-Carmona, M.A.; Strassburg, C.P.; Essler, M.; Ahmadzadehfar, H. Safety of multiple repeated cycles of 177Lu-octreotate in patients with recurrent neuroendocrine tumour. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1207–1214. [Google Scholar] [CrossRef] [PubMed]
- Werner, R.A.; Weich, A.; Higuchi, T.; Schmid, J.S.; Schirbel, A.; Lassmann, M.; Wild, V.; Rudelius, M.; Kudlich, T.; Herrmann, K.; et al. Imaging of Chemokine Receptor 4 Expression in Neuroendocrine Tumors—A Triple Tracer Comparative Approach. Theranostics 2017, 7, 1489–1498. [Google Scholar] [CrossRef] [PubMed]
- Soukup, T.; Lamb, B.W.; Weigl, M.; Green, J.S.A.; Sevdalis, N. An Integrated Literature Review of Time-on-Task Effects with a Pragmatic Framework for Understanding and Improving Decision-Making in Multidisciplinary Oncology Team Meetings. Front. Psychol. 2019, 10, 1245. [Google Scholar] [CrossRef] [PubMed]
- Yordanova, A. Peptide Receptor Radionuclide Therapy. In Clinical Nuclear Medicine; Ahmadzadehfar, H., Biersack, H.-J., Freeman, L.M., Zuckier, L.S., Eds.; Springer International Publishing: Cham, Switzerland, 2020; pp. 867–898. ISBN 978-3-030-39455-4. [Google Scholar]
- Yordanova, A.; Ahmadzadehfar, H.; Gonzalez-Carmona, M.; Strassburg, C.; Mayer, K.; Feldmann, G.; Schmidt-Wolf, I.; Lingohr, P.; Fischer, S.; Kristiansen, G.; et al. A Step-by-Step Clinical Approach for the Management of Neuroendocrine Tumours. Horm. Metab. Res. 2017, 49, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Hindié, E. The NETPET Score: Combining FDG and Somatostatin Receptor Imaging for Optimal Management of Patients with Metastatic Well-Differentiated Neuroendocrine Tumors. Theranostics 2017, 7, 1159–1163. [Google Scholar] [CrossRef] [PubMed]
- Chan, D.L.; Pavlakis, N.; Schembri, G.P.; Bernard, E.J.; Hsiao, E.; Hayes, A.; Barnes, T.; Diakos, C.; Khasraw, M.; Samra, J.; et al. Dual Somatostatin Receptor/FDG PET/CT Imaging in Metastatic Neuroendocrine Tumours: Proposal for a Novel Grading Scheme with Prognostic Significance. Theranostics 2017, 7, 1149–1158. [Google Scholar] [CrossRef]
- Has Simsek, D.; Kuyumcu, S.; Turkmen, C.; Sanlı, Y.; Aykan, F.; Unal, S.; Adalet, I. Can complementary 68Ga-DOTATATE and 18F-FDG PET/CT establish the missing link between histopathology and therapeutic approach in gastroenteropancreatic neuroendocrine tumors? J. Nucl. Med. 2014, 55, 1811–1817. [Google Scholar] [CrossRef] [Green Version]
- Parghane, R.V.; Mitra, A.; Bannore, T.U.; Rakshit, S.; Banerjee, S.; Basu, S. Initial clinical evaluation of indigenous (90)Y-DOTATATE in sequential duo-PRRT approach ((177)Lu-DOTATATE and (90)Y-DOTATATE) in neuroendocrine tumors with large bulky disease: Observation on tolerability, (90)Y-DOTATATE post-PRRT imaging characteristics (bremsstrahlung and PETCT) and early adverse effects. World J. Nucl. Med. 2020, 20, 73–81. [Google Scholar] [CrossRef]
- de Jong, M.; Breeman, W.A.P.; Valkema, R.; Bernard, B.F.; Krenning, E.P. Combination radionuclide therapy using 177Lu- and 90Y-labeled somatostatin analogs. J. Nucl. Med. 2005, 46 (Suppl. S1), 13S–17S. [Google Scholar]
- Bison, S.M.; Konijnenberg, M.W.; Melis, M.; Pool, S.E.; Bernsen, M.R.; Teunissen, J.J.M.; Kwekkeboom, D.J.; de Jong, M. Peptide receptor radionuclide therapy using radiolabeled somatostatin analogs: Focus on future developments. Clin. Transl. Imaging 2014, 2, 55–66. [Google Scholar] [CrossRef] [Green Version]
- Presky, D.H.; Schonbrunn, A. Somatostatin pretreatment increases the number of somatostatin receptors in GH4C1 pituitary cells and does not reduce cellular responsiveness to somatostatin. J. Biol. Chem. 1988, 263, 714–721. [Google Scholar] [CrossRef]
- Oddstig, J.; Bernhardt, P.; Nilsson, O.; Ahlman, H.; Forssell-Aronsson, E. Radiation induces up-regulation of somatostatin receptors 1, 2, and 5 in small cell lung cancer in vitro also at low absorbed doses. Cancer Biother. Radiopharm. 2011, 26, 759–765. [Google Scholar] [CrossRef] [PubMed]
- Reynders, K.; Illidge, T.; Siva, S.; Chang, J.Y.; De Ruysscher, D. The abscopal effect of local radiotherapy: Using immunotherapy to make a rare event clinically relevant. Cancer Treat. Rev. 2015, 41, 503–510. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ahmadzadehfar, H.; Biersack, H.-J.; Ezziddin, S. Radioembolization of Liver Tumors with Yttrium-90 Microspheres. Semin. Nucl. Med. 2010, 40, 105–121. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Edeline, J.; Rolland, Y.; Garin, E. Abscopal Effect After SIRT: It Exists, but How Could We Use It? CardioVasc. Interv. Radiol. 2020, 43, 1650–1651. [Google Scholar] [CrossRef]
- Multhoff, G.; Radons, J.; Vaupel, P. Critical role of aberrant angiogenesis in the development of tumor hypoxia and associated radioresistance. Cancers 2014, 6, 813–828. [Google Scholar] [CrossRef] [Green Version]
- D’Onofrio, M.; Cingarlini, S.; Ortolani, S.; Crosara, S.; de Robertis, R.; Vallerio, P.; Grego, E.; Ciaravino, V.; Ruzzenente, A.; Landoni, L.; et al. Perfusion CT Changes in Liver Metastases from Pancreatic Neuroendocrine Tumors During Everolimus Treatment. Anticancer Res. 2017, 37, 1305–1311. [Google Scholar] [CrossRef]
- Pandit-Taskar, N.; Modak, S. Norepinephrine Transporter as a Target for Imaging and Therapy. J. Nucl. Med. 2017, 58, 39S. [Google Scholar] [CrossRef] [Green Version]
- Faivre, S.; Niccoli, P.; Castellano, D.; Valle, J.W.; Hammel, P.; Raoul, J.-L.; Vinik, A.; van Cutsem, E.; Bang, Y.-J.; Lee, S.-H.; et al. Sunitinib in pancreatic neuroendocrine tumors: Updated progression-free survival and final overall survival from a phase III randomized study. Ann. Oncol. 2017, 28, 339–343. [Google Scholar] [CrossRef]
- Caron, M.-C.; Sharma, A.K.; O’Sullivan, J.; Myler, L.R.; Ferreira, M.T.; Rodrigue, A.; Coulombe, Y.; Ethier, C.; Gagné, J.-P.; Langelier, M.-F.; et al. Poly(ADP-ribose) polymerase-1 antagonizes DNA resection at double-strand breaks. Nat. Commun. 2019, 10, 2954. [Google Scholar] [CrossRef] [Green Version]
- Yao, J.C.; Fazio, N.; Singh, S.; Buzzoni, R.; Carnaghi, C.; Wolin, E.; Tomasek, J.; Raderer, M.; Lahner, H.; Voi, M.; et al. Everolimus for the treatment of advanced, non-functional neuroendocrine tumours of the lung or gastrointestinal tract (RADIANT-4): A randomised, placebo-controlled, phase 3 study. Lancet 2016, 387, 968–977. [Google Scholar] [CrossRef]
- Villard, L.; Romer, A.; Marincek, N.; Brunner, P.; Koller, M.T.; Schindler, C.; Ng, Q.K.T.; Mäcke, H.R.; Müller-Brand, J.; Rochlitz, C.; et al. Cohort study of somatostatin-based radiopeptide therapy with (90)Y-DOTA-TOC versus (90)Y-DOTA-TOC plus (177)Lu-DOTA-TOC in neuroendocrine cancers. J. Clin. Oncol. 2012, 30, 1100–1106. [Google Scholar] [CrossRef] [PubMed]
- Seregni, E.; Maccauro, M.; Chiesa, C.; Mariani, L.; Pascali, C.; Mazzaferro, V.; de Braud, F.; Buzzoni, R.; Milione, M.; Lorenzoni, A.; et al. Treatment with tandem 90YDOTA-TATE and 177LuDOTA-TATE of neuroendocrine tumours refractory to conventional therapy. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 223–230. [Google Scholar] [CrossRef] [PubMed]
- Kunikowska, J.; Królicki, L.; Hubalewska-Dydejczyk, A.; Mikołajczak, R.; Sowa-Staszczak, A.; Pawlak, D. Clinical results of radionuclide therapy of neuroendocrine tumours with 90Y-DOTATATE and tandem 90Y/177Lu-DOTATATE: Which is a better therapy option? Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 1788–1797. [Google Scholar] [CrossRef] [Green Version]
- Kunikowska, J.; Pawlak, D.; Bąk, M.I.; Kos-Kudła, B.; Mikołajczak, R.; Królicki, L. Long-term results and tolerability of tandem peptide receptor radionuclide therapy with (90)Y/(177)Lu-DOTATATE in neuroendocrine tumors with respect to the primary location: A 10-year study. Ann. Nucl. Med. 2017, 31, 347–356. [Google Scholar] [CrossRef]
- Pfeifer, A.K.; Gregersen, T.; Grønbæk, H.; Hansen, C.P.; Müller-Brand, J.; Herskind Bruun, K.; Krogh, K.; Kjær, A.; Knigge, U. Peptide receptor radionuclide therapy with Y-DOTATOC and (177)Lu-DOTATOC in advanced neuroendocrine tumors: Results from a Danish cohort treated in Switzerland. Neuroendocrinology 2011, 93, 189–196. [Google Scholar] [CrossRef]
- van Essen, M.; Krenning, E.P.; Kam, B.L.; de Herder, W.W.; van Aken, M.O.; Kwekkeboom, D.J. Report on short-term side effects of treatments with 177Lu-octreotate in combination with capecitabine in seven patients with gastroenteropancreatic neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 743–748. [Google Scholar] [CrossRef] [Green Version]
- Claringbold, P.G.; Brayshaw, P.A.; Price, R.A.; Turner, J.H. Phase II study of radiopeptide 177Lu-octreotate and capecitabine therapy of progressive disseminated neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2011, 38, 302–311. [Google Scholar] [CrossRef]
- Nicolini, S.; Bodei, L.; Bongiovanni, A.; Sansovini, M.; Grassi, I.; Ibrahim, T.; Monti, M.; Caroli, P.; Sarnelli, A.; Diano, D.; et al. Combined use of 177Lu-DOTATATE and metronomic capecitabine (Lu-X) in FDG-positive gastro-entero-pancreatic neuroendocrine tumors. Eur. J. Nucl. Med. Mol. Imaging 2021, 48, 3260–3267. [Google Scholar] [CrossRef]
- Claringbold, P.G.; Price, R.A.; Turner, J.H. Phase I-II study of radiopeptide 177Lu-octreotate in combination with capecitabine and temozolomide in advanced low-grade neuroendocrine tumors. Cancer Biother. Radiopharm. 2012, 27, 561–569. [Google Scholar] [CrossRef]
- Claringbold, P.G.; Turner, J.H. Pancreatic Neuroendocrine Tumor Control: Durable Objective Response to Combination 177Lu-Octreotate-Capecitabine-Temozolomide Radiopeptide Chemotherapy. Neuroendocrinology 2016, 103, 432–439. [Google Scholar] [CrossRef]
- Ostwal, V.; Basu, S.; Bhargava, P.; Shah, M.; Parghane, R.V.; Srinivas, S.; Chaudhari, V.; Bhandare, M.S.; Shrikhande, S.V.; Ramaswamy, A. Capecitabine-Temozolomide (CAPTEM) in advanced Grade 2 and grade 3 Neuroendocrine neoplasms (NENs)—Benefits of chemotherapy in NENs with significant 18FDG uptake. Neuroendocrinology 2020, 111, 998–1004. [Google Scholar] [CrossRef] [PubMed]
- Kong, G.; Thompson, M.; Collins, M.; Herschtal, A.; Hofman, M.S.; Johnston, V.; Eu, P.; Michael, M.; Hicks, R.J. Assessment of predictors of response and long-term survival of patients with neuroendocrine tumour treated with peptide receptor chemoradionuclide therapy (PRCRT). Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 1831–1844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prasad, V.; Srirajaskanthan, R.; Toumpanakis, C.; Grana, C.M.; Baldari, S.; Shah, T.; Lamarca, A.; Courbon, F.; Scheidhauer, K.; Baudin, E.; et al. Lessons from a multicentre retrospective study of peptide receptor radionuclide therapy combined with lanreotide for neuroendocrine tumours: A need for standardised practice. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2358–2371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yordanova, A.; Wicharz, M.M.; Mayer, K.; Brossart, P.; Gonzalez-Carmona, M.A.; Strassburg, C.P.; Fimmers, R.; Essler, M.; Ahmadzadehfar, H. The Role of Adding Somatostatin Analogues to Peptide Receptor Radionuclide Therapy as a Combination and Maintenance Therapy. Clin. Cancer Res. 2018, 24, 4672–4679. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hartrampf, P.E.; Hänscheid, H.; Kertels, O.; Schirbel, A.; Kreissl, M.C.; Flentje, M.; Sweeney, R.A.; Buck, A.K.; Polat, B.; Lapa, C. Long-term results of multimodal peptide receptor radionuclide therapy and fractionated external beam radiotherapy for treatment of advanced symptomatic meningioma. Clin. Transl. Radiat. Oncol. 2020, 22, 29–32. [Google Scholar] [CrossRef] [Green Version]
- Braat, A.J.A.T.; Ahmadzadehfar, H.; Kappadath, S.C.; Stothers, C.L.; Frilling, A.; Deroose, C.M.; Flamen, P.; Brown, D.B.; Sze, D.Y.; Mahvash, A.; et al. Radioembolization with (90)Y Resin Microspheres of Neuroendocrine Liver Metastases After Initial Peptide Receptor Radionuclide Therapy. CardioVasc. Interv. Radiol. 2020, 43, 246–253. [Google Scholar] [CrossRef] [Green Version]
- Braat, A.J.; Bruijnen, R.C.; van Rooij, R.; Braat, M.N.; Wessels, F.J.; van Leeuwaarde, R.S.; van Treijen, M.J.; de Herder, W.W.; Hofland, J.; Tesselaar, M.E.; et al. Additional holmium-166 radioembolisation after lutetium-177-dotatate in patients with neuroendocrine tumour liver metastases (HEPAR PLuS): A single-centre, single-arm, open-label, phase 2 study. Lancet Oncol. 2020, 21, 561–570. [Google Scholar] [CrossRef]
- Yilmaz, E.; Engin, M.N.; Özkan, Z.G.; Kovan, B.; Büyükkaya, F.; Poyanli, A.; Sağlam, S.; Başaran, M.; Türkmen, C. Y90 selective internal radiation therapy and peptide receptor radionuclide therapy for the treatment of metastatic neuroendocrine tumors: Combination or not? Nucl. Med. Commun. 2020, 41, 1242–1249. [Google Scholar] [CrossRef]
- Claringbold, P.G.; Turner, J.H. NeuroEndocrine Tumor Therapy with Lutetium-177-octreotate and Everolimus (NETTLE): A Phase I Study. Cancer Biother. Radiopharm. 2015, 30, 261–269. [Google Scholar] [CrossRef]
- Bushnell, D.L.; Bodeker, K.L.; O’Dorisio, T.M.; Madsen, M.T.; Menda, Y.; Graves, S.A.; O’Dorisio, M.S.; Zamba, G.K.D. Addition Of (131)I MIBG To PRRT ((90)Y DOTATOC) For Personalized Treatment of Selected Patients with Neuroendocrine Tumors. J. Nucl. Med. 2021, 62. [Google Scholar] [CrossRef] [PubMed]
- Anthony, L.B.; Woltering, E.A.; Espenan, G.D.; Cronin, M.D.; Maloney, T.J.; McCarthy, K.E. Indium-111-pentetreotide prolongs survival in gastroenteropancreatic malignancies. Semin. Nucl. Med. 2002, 32, 123–132. [Google Scholar] [CrossRef] [PubMed]
- Buscombe, J.R.; Caplin, M.E.; Hilson, A.J.W. Long-term efficacy of high-activity 111in-pentetreotide therapy in patients with disseminated neuroendocrine tumors. J. Nucl. Med. 2003, 44, 1–6. [Google Scholar] [PubMed]
- Kunikowska, J.; Zemczak, A.; Kołodziej, M.; Gut, P.; Łoń, I.; Pawlak, D.; Mikołajczak, R.; Kamiński, G.; Ruchała, M.; Kos-Kudła, B.; et al. Tandem peptide receptor radionuclide therapy using (90)Y/(177)Lu-DOTATATE for neuroendocrine tumors efficacy and side-effects—Polish multicenter experience. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 922–933. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kratochwil, C.; Giesel, F.L.; Bruchertseifer, F.; Mier, W.; Apostolidis, C.; Boll, R.; Murphy, K.; Haberkorn, U.; Morgenstern, A. 213Bi-DOTATOC receptor-targeted alpha-radionuclide therapy induces remission in neuroendocrine tumours refractory to beta radiation: A first-in-human experience. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 2106–2119. [Google Scholar] [CrossRef] [Green Version]
- Chan, H.S.; Konijnenberg, M.W.; Daniels, T.; Nysus, M.; Makvandi, M.; de Blois, E.; Breeman, W.A.; Atcher, R.W.; de Jong, M.; Norenberg, J.P. Improved safety and efficacy of (213)Bi-DOTATATE-targeted alpha therapy of somatostatin receptor-expressing neuroendocrine tumors in mice pre-treated with L-lysine. EJNMMI Res. 2016, 6, 83. [Google Scholar] [CrossRef] [Green Version]
- Miederer, M.; Henriksen, G.; Alke, A.; Mossbrugger, I.; Quintanilla-Martinez, L.; Senekowitsch-Schmidtke, R.; Essler, M. Preclinical evaluation of the alpha-particle generator nuclide 225Ac for somatostatin receptor radiotherapy of neuroendocrine tumors. Clin. Cancer Res. 2008, 14, 3555–3561. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Kulkarni, H.R.; Baum, R.P. Peptide Receptor Radionuclide Therapy Using 225Ac-DOTATOC Achieves Partial Remission in a Patient with Progressive Neuroendocrine Liver Metastases After Repeated β-Emitter Peptide Receptor Radionuclide Therapy. Clin. Nucl. Med. 2020, 45, 241–243. [Google Scholar] [CrossRef]
- Ballal, S.; Yadav, M.P.; Bal, C.; Sahoo, R.K.; Tripathi, M. Broadening horizons with (225)Ac-DOTATATE targeted alpha therapy for gastroenteropancreatic neuroendocrine tumour patients stable or refractory to (177)Lu-DOTATATE PRRT: First clinical experience on the efficacy and safety. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 934–946. [Google Scholar] [CrossRef]
- Lee, D.; Graves, S.; Liu, D.; Wen, H.; Zepeda-Orozco, D.; Madsen, M.; Walsh, S.; Watkins, G.; Menda, Y.; Schultz, M.; et al. Preclinical evaluation of 212Pb-based alpha-particle therapy for neuroendocrine tumors: Dosimetry and potential toxicities. J. Nucl. Med. 2019, 60, 663. [Google Scholar]
- Delpassand, E.; Tworowska, I.; Esfandiari, R.; Torgue, J.; Hurt, J.D.; Nunez, R. Phase I dose-escalation study of AlphaMedix for targeted-alpha-emitter therapy of PRRT-naive neuroendocrine patients. J. Clin. Oncol. 2021, 39, 4117. [Google Scholar] [CrossRef]
- Stallons, T.A.R.; Saidi, A.; Tworowska, I.; Delpassand, E.S.; Torgue, J.J. Preclinical Investigation of (212)Pb-DOTAMTATE for Peptide Receptor Radionuclide Therapy in a Neuroendocrine Tumor Model. Mol. Cancer Ther. 2019, 18, 1012–1021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sorbye, H.; Welin, S.; Langer, S.W.; Vestermark, L.W.; Holt, N.; Osterlund, P.; Dueland, S.; Hofsli, E.; Guren, M.G.; Ohrling, K.; et al. Predictive and prognostic factors for treatment and survival in 305 patients with advanced gastrointestinal neuroendocrine carcinoma (WHO G3): The NORDIC NEC study. Ann. Oncol. 2013, 24, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Thang, S.P.; Lung, M.S.; Kong, G.; Hofman, M.S.; Callahan, J.; Michael, M.; Hicks, R.J. Peptide receptor radionuclide therapy (PRRT) in European Neuroendocrine Tumour Society (ENETS) grade 3 (G3) neuroendocrine neoplasia (NEN)—A single-institution retrospective analysis. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 262–277. [Google Scholar] [CrossRef] [PubMed]
- Nicolini, S.; Severi, S.; Ianniello, A.; Sansovini, M.; Ambrosetti, A.; Bongiovanni, A.; Scarpi, E.; Di Mauro, F.; Rossi, A.; Matteucci, F.; et al. Investigation of receptor radionuclide therapy with (177)Lu-DOTATATE in patients with GEP-NEN and a high Ki-67 proliferation index. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 923–930. [Google Scholar] [CrossRef] [PubMed]
- Yordanova, A.; Ahrens, H.; Feldmann, G.; Brossart, P.; Gaertner, F.C.; Fottner, C.; Weber, M.M.; Ahmadzadehfar, H.; Schreckenberger, M.; Miederer, M.; et al. Peptide Receptor Radionuclide Therapy Combined with Chemotherapy in Patients With Neuroendocrine Tumors. Clin. Nucl. Med. 2019, 44, e329–e335. [Google Scholar] [CrossRef]
- Özdirik, B.; Amthauer, H.; Schatka, I.; Goretzki, P.E.; Mogl, M.T.; Fehrenbach, U.; Tacke, F.; Jann, H.; Roderburg, C. A rare case of a patient with a high grade neuroendocrine tumor developing neutropenic sepsis after receiving PRRT combined with Capecitabine or Temozolomide: A case report. Mol. Clin. Oncol. 2021, 14, 20. [Google Scholar] [CrossRef]
- Hicks, R.J.; Kwekkeboom, D.J.; Krenning, E.; Bodei, L.; Grozinsky-Glasberg, S.; Arnold, R.; Borbath, I.; Cwikla, J.; Toumpanakis, C.; Kaltsas, G.; et al. ENETS Consensus Guidelines for the Standards of Care in Neuroendocrine Neoplasia: Peptide Receptor Radionuclide Therapy with Radiolabeled Somatostatin Analogues. Neuroendocrinology 2017, 105, 295–309. [Google Scholar] [CrossRef]
- Haug, A.R.; Rominger, A.; Mustafa, M.; Auernhammer, C.; Göke, B.; Schmidt, G.P.; Wängler, B.; Cumming, P.; Bartenstein, P.; Hacker, M. Treatment with Octreotide Does Not Reduce Tumor Uptake of 68Ga-DOTATATE as Measured by PET/CT in Patients with Neuroendocrine Tumors. J. Nucl. Med. 2011, 52, 1679–1683. [Google Scholar] [CrossRef] [Green Version]
- Cherk, M.H.; Kong, G.; Hicks, R.J.; Hofman, M.S. Changes in biodistribution on (68)Ga-DOTA-Octreotate PET/CT after long acting somatostatin analogue therapy in neuroendocrine tumour patients may result in pseudoprogression. Cancer Imaging 2018, 18, 3. [Google Scholar] [CrossRef] [Green Version]
- Aalbersberg, E.A.; de Wit-van der Veen, B.J.; Versleijen, M.W.J.; Saveur, L.J.; Valk, G.D.; Tesselaar, M.E.T.; Stokkel, M.P.M. Influence of lanreotide on uptake of (68)Ga-DOTATATE in patients with neuroendocrine tumours: A prospective intra-patient evaluation. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 696–703. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.R.; Caplin, M.E.; Kunz, P.L.; Ruszniewski, P.B.; Bodei, L.; Hendifar, A.E.; Mittra, E.; Wolin, E.M.; Yao, J.C.; Pavel, M.E.; et al. Final overall survival in the phase 3 NETTER-1 study of lutetium-177-DOTATATE in patients with midgut neuroendocrine tumors. J. Clin. Oncol. 2021, 39, 4112. [Google Scholar] [CrossRef]
- Braat, A.J.A.T.; Snijders, T.J.; Seute, T.; Vonken, E.P.A. Will 177Lu-DOTATATE Treatment Become More Effective in Salvage Meningioma Patients, When Boosting Somatostatin Receptor Saturation? A Promising Case on Intra-arterial Administration. CardioVasc. Interv. Radiol. 2019, 42, 1649–1652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ezziddin, S.; Khalaf, F.; Vanezi, M.; Haslerud, T.; Mayer, K.; Al Zreiqat, A.; Willinek, W.; Biersack, H.-J.; Sabet, A. Outcome of peptide receptor radionuclide therapy with 177Lu-octreotate in advanced grade 1/2 pancreatic neuroendocrine tumours. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 925–933. [Google Scholar] [CrossRef] [PubMed]
- Strosberg, J.; Kunz, P.L.; Hendifar, A.; Yao, J.; Bushnell, D.; Kulke, M.H.; Baum, R.P.; Caplin, M.; Ruszniewski, P.; Delpassand, E. Impact of liver tumour burden, alkaline phosphatase elevation, and target lesion size on treatment outcomes with 177 Lu-Dotatate: An analysis of the NETTER-1 study. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 2372–2382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, J.C.; Shah, M.H.; Ito, T.; Bohas, C.L.; Wolin, E.M.; van Cutsem, E.; Hobday, T.J.; Okusaka, T.; Capdevila, J.; de Vries, E.G.E.; et al. Everolimus for advanced pancreatic neuroendocrine tumors. N. Engl. J. Med. 2011, 364, 514–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zellmer, J.; Vomacka, L.; Boening, G.; Gildehaus, F.J.; Mille, E.; Hacker, M.; Bartenstein, P.; Haug, A.; Ilhan, H. Combination of Peptide Receptor Radionuclide Therapy with Lu-177 DOTATATE and the m-TOR inhibitor RAD001 (Everolimus) in AR42J tumor bearing mice and response assessment by Ga-68 DOTATATE PET. J. Nucl. Med. 2018, 59, 1346b. [Google Scholar]
- Hamming, L.C.; Slotman, B.J.; Verheul, H.M.W.; Thijssen, V.L. The clinical application of angiostatic therapy in combination with radiotherapy: Past, present, future. Angiogenesis 2017, 20, 217–232. [Google Scholar] [CrossRef] [Green Version]
- Kim, C.; Liu, S.V.; Subramaniam, D.S.; Torres, T.; Loda, M.; Esposito, G.; Giaccone, G. Phase I study of the (177)Lu-DOTA(0)-Tyr(3)-Octreotate (lutathera) in combination with nivolumab in patients with neuroendocrine tumors of the lung. J. Immunother. Cancer 2020, 8. [Google Scholar] [CrossRef]
- Cullinane, C.; Waldeck, K.; Kirby, L.; Rogers, B.E.; Eu, P.; Tothill, R.W.; Hicks, R.J. Enhancing the anti-tumour activity of 177Lu-DOTA-octreotate radionuclide therapy in somatostatin receptor-2 expressing tumour models by targeting PARP. Sci. Rep. 2020, 10, 10196. [Google Scholar] [CrossRef]
- Adant, S.; Purohit, N.; Shah, R.; Shah, G.; Beauregard, J.-M. Potentiation of 177Lu-octreotate PRRT by PARP inhibitors in a 3D spheroid model of human-derived neuroendocrine tumor cell lines. J. Nucl. Med. 2017, 58, 243. [Google Scholar]
| Combination Partner | ORR (%) | OS (Month) | PFS (Month) | SAE (%) | Ref |
|---|---|---|---|---|---|
| Dual PRRT Lu-177 and Y-90 | 42 | 66–127 | - | 2% MDS, 2% nephrotoxicity, 7% hematotoxicity | [20,32,33,34,35,36,37] |
| Capecitabine | 24–30 | not reached | 31 | <15% anemia/thrombocytopenia/neutropenia 5% fatigue/diarrhea | [38,39,40] |
| CAPTEM | 53–70 | not reached | 22–48 | 6% neutropenia, 3% nausea | [41,42,43] |
| 5-fluorouracil | 25 | not reached | - | - | [44] |
| SSA | 37 | 91 | 48 | 3% hepatotoxicity | [45,46] |
| EBRT | 0 | not reached | 108 | 0% | [47] |
| Liver embolization | 16 (Y-90) 43 (Ho-166) | 42–68 | - | 10% abdominal pain, 3% fatigue/nausea, >20% lymphocytopenia, 5% radiation-induced gastric ulceration, 2% radiation pneumonitis, 2% liver abscess, 2% cholangitis, 50% liver enzyme elevation, <5% liver failure (2–3% fatal) | [48,49,50] |
| Everolimus | 44 | not reached | not reached (63% at 24 months) | mainly hematotoxicity (thrombocytopenia, anemia) in the 10 mg/d everolimus dose group 100%, one case (6%) hepatotoxicity | [51] |
| [131I]I-MIBG | 0 | - | - | one case of three (33%) thrombocytopenia | [52] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yordanova, A.; Ahmadzadehfar, H. Combination Therapies with PRRT. Pharmaceuticals 2021, 14, 1005. https://doi.org/10.3390/ph14101005
Yordanova A, Ahmadzadehfar H. Combination Therapies with PRRT. Pharmaceuticals. 2021; 14(10):1005. https://doi.org/10.3390/ph14101005
Chicago/Turabian StyleYordanova, Anna, and Hojjat Ahmadzadehfar. 2021. "Combination Therapies with PRRT" Pharmaceuticals 14, no. 10: 1005. https://doi.org/10.3390/ph14101005
APA StyleYordanova, A., & Ahmadzadehfar, H. (2021). Combination Therapies with PRRT. Pharmaceuticals, 14(10), 1005. https://doi.org/10.3390/ph14101005





