Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability
Abstract
1. Introduction
2. Results
2.1. Participants
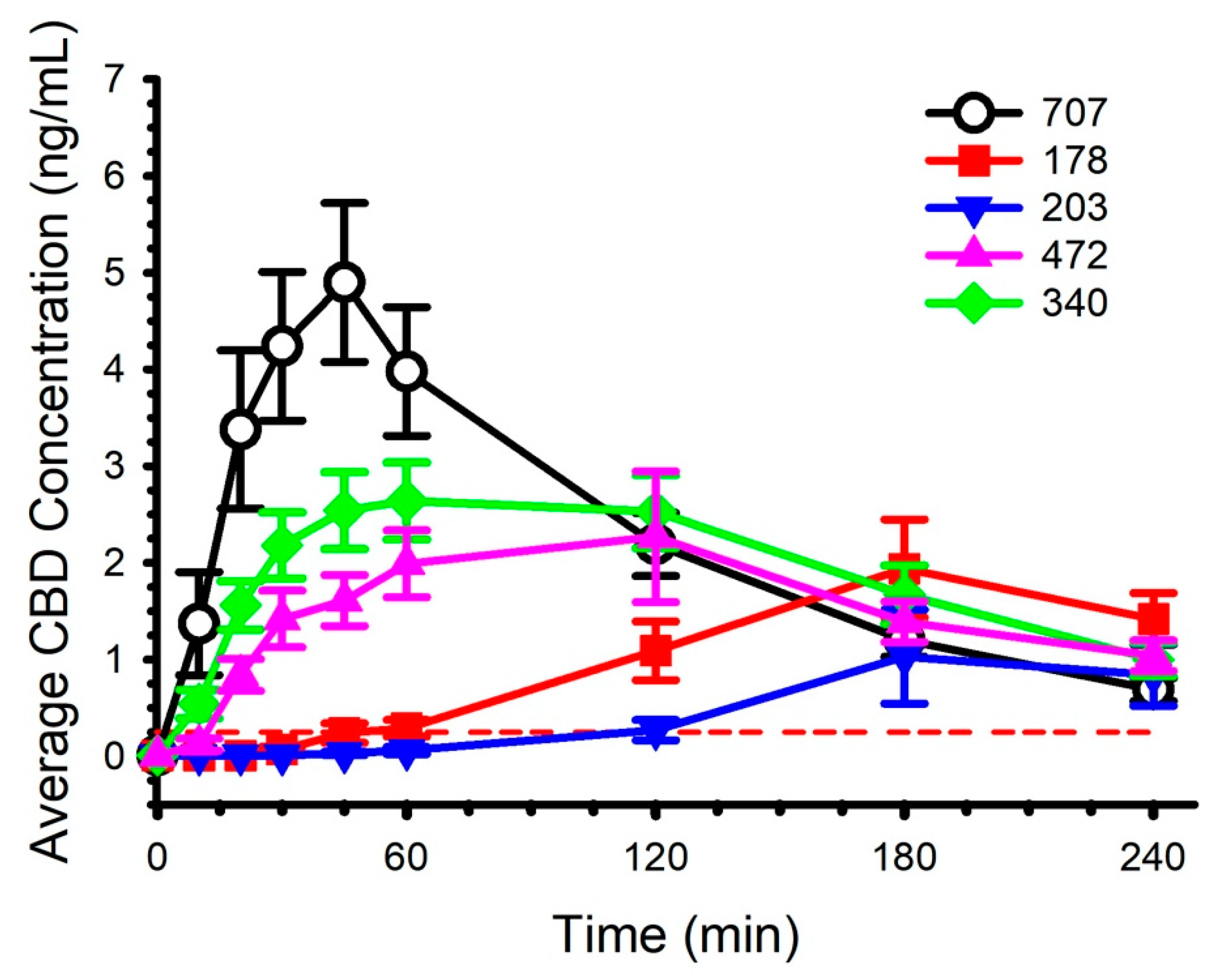
2.2. Circulating CBD and Pharmacokinetics
2.3. Body Composition
2.4. Heart Rate Variability
3. Discussion
4. Materials and Methods
4.1. Participants
4.2. Protocol Overview
4.3. Procedures
4.4. Cannabidiol Preparations
4.5. Cannabidiol Analysis
4.6. Pharmacokinetic Analysis
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Cather, J.C.; Cather, J.C. Cannabidiol primer for healthcare professionals. Proc. Bayl. Univ. Med. Cent. 2020, 33, 376–379. [Google Scholar] [CrossRef]
- VanDolah, H.J.; Bauer, B.A.; Mauck, K.F. Clinicians’ Guide to Cannabidiol and Hemp Oils. Mayo Clin. Proc. 2019, 94, 1840–1851. [Google Scholar] [CrossRef]
- Millan-Guerrero, R.O.; Isais-Millan, S. Cannabis and the exocannabinoid and endocannabinoid systems. Their use and controversies. Gac. Med. Mex. 2019, 155, 471–474. [Google Scholar] [CrossRef]
- Millar, S.A.; Maguire, R.F.; Yates, A.S.; O’Sullivan, S.E. Towards Better Delivery of Cannabidiol (CBD). Pharmaceuticals 2020, 13, 219. [Google Scholar] [CrossRef]
- Davy, K.P.; Seals, D.R. Total blood volume in healthy young and older men. J. Appl. Physiol. 1994, 76, 2059–2062. [Google Scholar] [CrossRef]
- Jones, P.P.; Davy, K.P.; DeSouza, C.A.; van Pelt, R.E.; Seals, D.R. Absence of age-related decline in total blood volume in physically active females. Am. J. Physiol. 1997, 272, H2534–H2540. [Google Scholar] [CrossRef] [PubMed]
- Wong, A.; Montebello, M.E.; Norberg, M.M.; Rooney, K.; Lintzeris, N.; Bruno, R.; Booth, J.; Arnold, J.C.; McGregor, I.S. Exercise increases plasma THC concentrations in regular cannabis users. Drug Alcohol Depend. 2013, 133, 763–767. [Google Scholar] [CrossRef]
- Johansson, E.; Noren, K.; Sjovall, J.; Halldin, M.M. Determination of delta 1-tetrahydrocannabinol in human fat biopsies from marihuana users by gas chromatography-mass spectrometry. Biomed. Chromatogr. 1989, 3, 35–38. [Google Scholar] [CrossRef] [PubMed]
- Sultan, S.R.; Millar, S.A.; England, T.J.; O’Sullivan, S.E. A Systematic Review and Meta-Analysis of the Haemodynamic Effects of Cannabidiol. Front. Pharmacol. 2017, 8, 81. [Google Scholar] [CrossRef] [PubMed]
- Jadoon, K.A.; Tan, G.D.; O’Sullivan, S.E. A single dose of cannabidiol reduces blood pressure in healthy volunteers in a randomized crossover study. JCI Insight 2017, 2. [Google Scholar] [CrossRef] [PubMed]
- Sultan, S.R.; O’Sullivan, S.E.; England, T.J. The effects of acute and sustained cannabidiol dosing for seven days on the haemodynamics in healthy men: A randomised controlled trial. Br. J. Clin. Pharmacol. 2020, 86, 1125–1138. [Google Scholar] [CrossRef] [PubMed]
- Hobbs, J.M.; Vazquez, A.R.; Remijan, N.D.; Trotter, R.E.; McMillan, T.V.; Freedman, K.E.; Wei, Y.; Woelfel, K.A.; Arnold, O.R.; Wolfe, L.M.; et al. Evaluation of pharmacokinetics and acute anti-inflammatory potential of two oral cannabidiol preparations in healthy adults. Phytother. Res. 2020, 34, 1696–1703. [Google Scholar] [CrossRef] [PubMed]
- De Faria, S.M.; de Morais Fabricio, D.; Tumas, V.; Castro, P.C.; Ponti, M.A.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.S.; Chagas, M.H.N. Effects of acute cannabidiol administration on anxiety and tremors induced by a Simulated Public Speaking Test in patients with Parkinson’s disease. J. Psychopharmacol. 2020, 34, 189–196. [Google Scholar] [CrossRef]
- Tsuji, H.; Larson, M.G.; Venditti, F.J., Jr.; Manders, E.S.; Evans, J.C.; Feldman, C.L.; Levy, D. Impact of reduced heart rate variability on risk for cardiac events. The Framingham Heart Study. Circulation 1996, 94, 2850–2855. [Google Scholar] [CrossRef] [PubMed]
- Zbilut, J.P.; Lawson, L. Decreased heart rate variability in significant cardiac events. Crit. Care Med. 1988, 16, 64–66. [Google Scholar] [CrossRef] [PubMed]
- Hernandez-Vicente, A.; Hernando, D.; Santos-Lozano, A.; Rodriguez-Romo, G.; Vicente-Rodriguez, G.; Pueyo, E.; Bailon, R.; Garatachea, N. Heart Rate Variability and Exceptional Longevity. Front. Physiol. 2020, 11, 566399. [Google Scholar] [CrossRef]
- Lopez, H.L.; Cesareo, K.R.; Raub, B.; Kedia, A.W.; Sandrock, J.E.; Kerksick, C.M.; Ziegenfuss, T.N. Effects of Hemp Extract on Markers of Wellness, Stress Resilience, Recovery and Clinical Biomarkers of Safety in Overweight, But Otherwise Healthy Subjects. J. Diet Suppl. 2020, 17, 561–586. [Google Scholar] [CrossRef]
- Knaub, K.; Sartorius, T.; Dharsono, T.; Wacker, R.; Wilhelm, M.; Schon, C. A Novel Self-Emulsifying Drug Delivery System (SEDDS) Based on VESIsorb((R)) Formulation Technology Improving the Oral Bioavailability of Cannabidiol in Healthy Subjects. Molecules 2019, 24, 2967. [Google Scholar] [CrossRef]
- Millar, S.A.; Stone, N.L.; Yates, A.S.; O’Sullivan, S.E. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Front. Pharmacol. 2018, 9, 1365. [Google Scholar] [CrossRef]
- Izgelov, D.; Davidson, E.; Barasch, D.; Regev, A.; Domb, A.J.; Hoffman, A. Pharmacokinetic investigation of synthetic cannabidiol oral formulations in healthy volunteers. Eur. J. Pharm. Biopharm. 2020, 154, 108–115. [Google Scholar] [CrossRef]
- Atsmon, J.; Heffetz, D.; Deutsch, L.; Deutsch, F.; Sacks, H. Single-Dose Pharmacokinetics of Oral Cannabidiol Following Administration of PTL101: A New Formulation Based on Gelatin Matrix Pellets Technology. Clin. Pharmacol. Drug Dev. 2018, 7, 751–758. [Google Scholar] [CrossRef]
- Verrico, C.D.; Wesson, S.; Konduri, V.; Hofferek, C.J.; Vazquez-Perez, J.; Blair, E.; Dunner, K., Jr.; Salimpour, P.; Decker, W.K.; Halpert, M.M. A randomized, double-blind, placebo-controlled study of daily cannabidiol for the treatment of canine osteoarthritis pain. Pain 2020. [Google Scholar] [CrossRef]
- Stott, C.G.; White, L.; Wright, S.; Wilbraham, D.; Guy, G.W. A phase I study to assess the single and multiple dose pharmacokinetics of THC/CBD oromucosal spray. Eur. J. Clin. Pharmacol. 2013, 69, 1135–1147. [Google Scholar] [CrossRef]
- Lamsen, M.R.L.; Wang, T.; D’Souza, D.; Dia, V.; Chen, G.; Zhong, Q. Encapsulation of vitamin D3 in gum arabic to enhance bioavailability and stability for beverage applications. J. Food Sci. 2020. [Google Scholar] [CrossRef]
- Feng, J.; Huang, M.; Chai, Z.; Li, C.; Huang, W.; Cui, L.; Li, Y. The influence of oil composition on the transformation, bioaccessibility, and intestinal absorption of curcumin in nanostructured lipid carriers. Food Funct. 2020, 11, 5223–5239. [Google Scholar] [CrossRef]
- Nadulski, T.; Pragst, F.; Weinberg, G.; Roser, P.; Schnelle, M.; Fronk, E.M.; Stadelmann, A.M. Randomized, double-blind, placebo-controlled study about the effects of cannabidiol (CBD) on the pharmacokinetics of Delta9-tetrahydrocannabinol (THC) after oral application of THC verses standardized cannabis extract. Ther. Drug Monit. 2005, 27, 799–810. [Google Scholar] [CrossRef]
- Nadulski, T.; Sporkert, F.; Schnelle, M.; Stadelmann, A.M.; Roser, P.; Schefter, T.; Pragst, F. Simultaneous and sensitive analysis of THC, 11-OH-THC, THC-COOH, CBD, and CBN by GC-MS in plasma after oral application of small doses of THC and cannabis extract. J. Anal. Toxicol. 2005, 29, 782–789. [Google Scholar] [CrossRef]
- Guy, G.W.; Robson, P.J. A Phase I, Open Label, Four-Way Crossover Study to Compare the Pharmacokinetic Profiles of a Single Dose of 20 mg of a Cannabis Based Medicine Extract (CBME) Administered on 3 Different Areas of the Buccal Mucosa and to Investigate the Pharmacokinetics of CBME per Oral in Healthy Male and Female Volunteers (GWPK0112). J. Cannabis Ther. 2004, 3, 79–120. [Google Scholar] [CrossRef]
- Cherniakov, I.; Izgelov, D.; Barasch, D.; Davidson, E.; Domb, A.J.; Hoffman, A. Piperine-pro-nanolipospheres as a novel oral delivery system of cannabinoids: Pharmacokinetic evaluation in healthy volunteers in comparison to buccal spray administration. J. Control. Release 2017, 266, 1–7. [Google Scholar] [CrossRef]
- Atsmon, J.; Cherniakov, I.; Izgelov, D.; Hoffman, A.; Domb, A.J.; Deutsch, L.; Deutsch, F.; Heffetz, D.; Sacks, H. PTL401, a New Formulation Based on Pro-Nano Dispersion Technology, Improves Oral Cannabinoids Bioavailability in Healthy Volunteers. J. Pharm. Sci. 2018, 107, 1423–1429. [Google Scholar] [CrossRef]
- Taylor, L.; Crockett, J.; Tayo, B.; Morrison, G. A Phase 1, Open-Label, Parallel-Group, Single-Dose Trial of the Pharmacokinetics and Safety of Cannabidiol (CBD) in Subjects with Mild to Severe Hepatic Impairment. J. Clin. Pharmacol. 2019, 59, 1110–1119. [Google Scholar] [CrossRef]
- Tayo, B.; Taylor, L.; Sahebkar, F.; Morrison, G. A Phase I, Open-Label, Parallel-Group, Single-Dose Trial of the Pharmacokinetics, Safety, and Tolerability of Cannabidiol in Subjects with Mild to Severe Renal Impairment. Clin. Pharm. 2020, 59, 747–755. [Google Scholar] [CrossRef]
- Manini, A.F.; Yiannoulos, G.; Bergamaschi, M.M.; Hernandez, S.; Olmedo, R.; Barnes, A.J.; Winkel, G.; Sinha, R.; Jutras-Aswad, D.; Huestis, M.A.; et al. Safety and pharmacokinetics of oral cannabidiol when administered concomitantly with intravenous fentanyl in humans. J. Addict. Med. 2015, 9, 204–210. [Google Scholar] [CrossRef] [PubMed]
- Crockett, J.; Critchley, D.; Tayo, B.; Berwaerts, J.; Morrison, G. A phase 1, randomized, pharmacokinetic trial of the effect of different meal compositions, whole milk, and alcohol on cannabidiol exposure and safety in healthy subjects. Epilepsia 2020, 61, 267–277. [Google Scholar] [CrossRef]
- Taylor, L.; Gidal, B.; Blakey, G.; Tayo, B.; Morrison, G. A Phase I, Randomized, Double-Blind, Placebo-Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs 2018, 32, 1053–1067. [Google Scholar] [CrossRef]
- Schoedel, K.A.; Szeto, I.; Setnik, B.; Sellers, E.M.; Levy-Cooperman, N.; Mills, C.; Etges, T.; Sommerville, K. Abuse potential assessment of cannabidiol (CBD) in recreational polydrug users: A randomized, double-blind, controlled trial. Epilepsy Behav. 2018, 88, 162–171. [Google Scholar] [CrossRef]
- Devinsky, O.; Cilio, M.R.; Cross, H.; Fernandez-Ruiz, J.; French, J.; Hill, C.; Katz, R.; Di Marzo, V.; Jutras-Aswad, D.; Notcutt, W.G.; et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 2014, 55, 791–802. [Google Scholar] [CrossRef]
- Hosseini, A.; McLachlan, A.J.; Lickliter, J.D. A phase I trial of the safety, tolerability and pharmacokinetics of cannabidiol administered as single-dose oil solution and single and multiple doses of a sublingual wafer in healthy volunteers. Br. J. Clin. Pharmacol. 2020. [Google Scholar] [CrossRef]
- Schmid, K.; Schonlebe, J.; Drexler, H.; Mueck-Weymann, M. The effects of cannabis on heart rate variability and well-being in young men. Pharmacopsychiatry 2010, 43, 147–150. [Google Scholar] [CrossRef]
- Brennan, M.; Palaniswami, M.; Kamen, P. Poincare plot interpretation using a physiological model of HRV based on a network of oscillators. Am. J. Physiol. Heart Circ. Physiol. 2002, 283, H1873–H1886. [Google Scholar] [CrossRef]
- Blake, R.R.; Shaw, D.J.; Culshaw, G.J.; Martinez-Pereira, Y. Poincare plots as a measure of heart rate variability in healthy dogs. J. Vet. Cardiol. 2018, 20, 20–32. [Google Scholar] [CrossRef] [PubMed]
- Ryan, S.P.P.; Newman, A.A.; Wilburn, J.R.; Rhoades, L.D.; Trikha, S.R.J.; Godwin, E.C.; Schoenberg, H.M.; Battson, M.L.; Ewell, T.R.; Luckasen, G.J.; et al. Sodium Glucose Co-Transporter 2 Inhibition Does Not Favorably Modify the Physiological Responses to Dietary Counselling in Diabetes-Free, Sedentary Overweight and Obese Adult Humans. Nutrients 2020, 12, 510. [Google Scholar] [CrossRef] [PubMed]
- Paxton, R.J.; Malcolm, M.P.; Newsom, S.A.; Richards, J.C.; Rynn, G.M.; Bell, C. Sympathetic responses to repetitive trans-spinal magnetic stimulation. Clin. Auton. Res. 2011, 21, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Tarvainen, M.P.; Niskanen, J.P.; Lipponen, J.A.; Ranta-Aho, P.O.; Karjalainen, P.A. Kubios HRV--heart rate variability analysis software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef]
- Tarvainen, M.P.; Ranta-Aho, P.O.; Karjalainen, P.A. An advanced detrending method with application to HRV analysis. IEEE Trans. Biomed. Eng. 2002, 49, 172–175. [Google Scholar] [CrossRef]
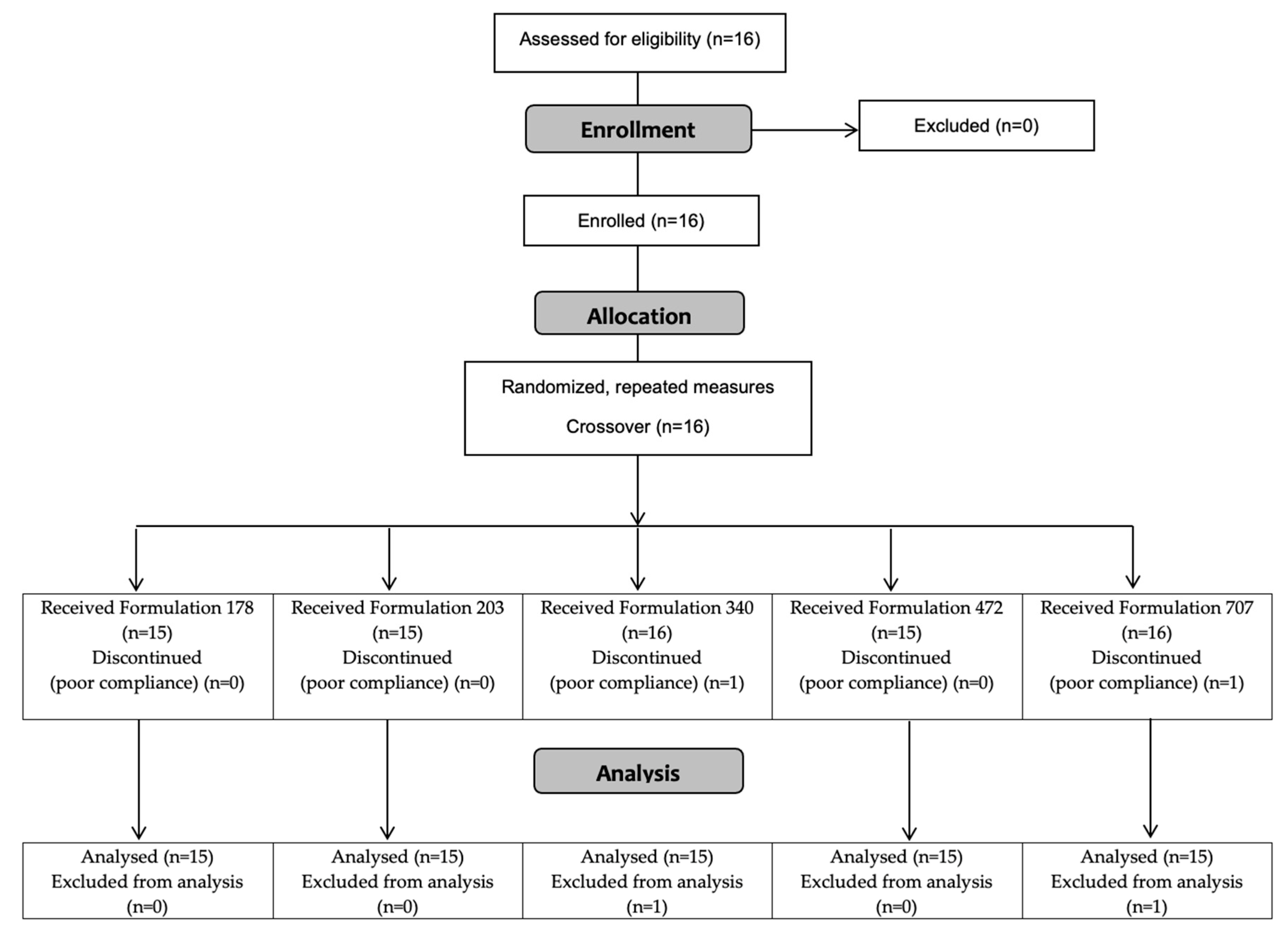
| Characteristic | Mean ± SD | Range |
|---|---|---|
| Sex (M/F) | 9/6 | - |
| Age (years) | 29 ± 11 | 21–62 |
| Height (cm) | 174 ± 9 | 155–191 |
| Body Mass (kg) | 75.5 ± 13.8 | 58.6–100.1 |
| Body Mass Index (kg/m2) | 24.6 ± 3.5 | 18.8–31.4 |
| Fat Mass (kg) | 19.4 ± 4.8 | 12.0–30.1 |
| Body Fat (%) | 26.2 ± 6.6 | 18.0–40.7 |
| Lean Mass (kg) | 53.7 ± 12.3 | 33.3–70.8 |
| Bone Mineral Content (kg) | 2.4 ± 0.4 | 1.6–2.9 |
| Code | Preparation | Composition and Administration |
|---|---|---|
| 178 | CBD Tincture Base | MCT Oil droplet containing CBD isolate; administered in 227 mL (8 oz) of water |
| 203 | CBD Powder in Water | CBD as powder, suspended in reverse osmosis water; administered in 227 mL (8 oz) of water |
| 340 | 20% CBD Concentration Liquid | Reverse osmosis water, CBD, MCT oil, quillaja extract; administered in 227 mL (8 oz) of water |
| 472 | 5% CBD Concentration Powder | Water soluble CBD, sorbitol, modified food starch, natural flavors, MCT; oil administered in 227 mL (8 oz) of water |
| 707 | 5% CBD Concentration Liquid | Reverse osmosis water, gum arabic, CBD, MCT oil, citric acid; administered in 227 mL (8 oz) of water |
| Parameter | 178 | 203 | 340 | 472 | 707 |
|---|---|---|---|---|---|
| Tmax (h) | 3.29 ± 0.61 a,b n = 14 | 3.39 ± 0.65 c n = 13 | 1.28 ± 0.62 a n = 15 | 1.53 ± 1.02 b n = 15 | 0.70 ± 0.23 a,c n = 15 |
| Cmax (ng/mL) | 2.20 ± 1.88 a n = 14 | 1.29 ± 1.93 b,c n = 13 | 3.54 ± 1.65 b n = 15 | 2.88 ± 2.48 d n = 15 | 5.57 ± 3.32 a,c,d n = 15 |
| AUC0-4 (h × ng/mL) | 4.58 ± 3.88 a n = 14 | 2.30 ± 2.77 b,c n = 13 | 7.81 ± 3.91 a,b n = 15 | 6.32 ± 4.57 n = 15 | 9.12 ± 5.21 c n = 15 |
| AUC0-inf (h × ng/mL) | - n = 0 | - n = 0 | 13.81 ± 8.2n = 9 | 9.96 ± 8.11 n = 8 | 10.77 ± 5.71 n = 15 |
| t½ (h) | - n = 0 | - n = 0 | 2.20 ± 1.14 n = 9 | 5.18 ± 7.07 n = 8 | 1.42 ± 0.52 n = 15 |
| Ka (1/h) | 0.32 ± 0.24 n = 5 | 0.24 ± 0.00 n = 2 | 1.19 ± 0.80 n = 15 | 1.87 ± 2.23 n = 14 | 1.43 ± 0.65 n = 12 |
| Ke (1/h) | - n = 0 | - n = 15 | 0.40 ± 0.21 n = 9 | 0.27 ± 0.16 a n = 8 | 0.56 ± 0.22 a n = 15 |
| Vd (L) | - n = 0 | - n = 0 | 7428 ± 2232 n = 9 | 20,178 ± 9989 a n = 8 | 8024 ± 6630 a n = 15 |
| Parameter | 178 | 203 | 340 | 472 | 707 | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| 0 | 60 | 0 | 60 | 0 | 60 | 0 | 60 | 0 | 60 | |
| Time Domain | ||||||||||
| HR (b/min) a | 57 ± 9 | 56 ± 10 | 61 ± 10 | 59 ± 10 | 58 ± 11 | 55 ± 10 | 58 ± 10 | 56 ± 9 | 57 ± 9 | 55 ± 9 |
| R-to-RInt (ms) a | 1076 ± 173 | 1110 ± 194 | 1011 ± 183 | 1049 ± 200 | 1061 ± 190 | 1113 ± 187 | 1069 ± 190 | 1106 ± 195 | 1075 ± 170 | 1119 ± 180 |
| SDNN (ms) | 112 ± 50 | 115 ± 57 | 126 ± 53 | 129 ± 53 | 126 ± 53 | 125 ± 46 | 133 ± 64 | 132 ± 53 | 123 ± 55 | 129 ± 47 |
| RMSSD (ms) | 100 ± 43 | 108 ± 56 | 118 ± 57 | 116 ± 50 | 113 ± 47 | 117 ± 41 | 121 ± 60 | 125 ± 55 | 113 ± 54 | 126 ± 49 |
| R-to-R Triangular Index (ms) | 19 ± 8 | 19 ± 6 | 17 ± 5 | 21 ± 6 | 21 ± 7 | 22 ± 8 | 22 ± 8 | 22 ± 10 | 21 ± 7 | 20 ± 8 |
| TINN (ms) b | 466 ± 174 | 525 ± 237 | 537 ± 167 | 605 ± 188 | 592 ± 252 | 624 ± 219 | 619 ± 215 | 708 ± 255 | 598 ± 242 | 697 ± 239 |
| Frequency Domain | ||||||||||
| VLFpeak (Hz) | 0.037 ± 0.003 | 0.035 ± 0.005 | 0.037 ± 0.005 | 0.037 ± 0.004 | 0.035 ± 0.004 | 0.035 ± 0.004 | 0.036 ± 0.003 | 0.035 ± 0.005 | 0.037 ± 0.003 | 0.036 ± 0.005 |
| LFpeak (Hz) | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 | 0.1 ± 0.0 |
| HFpeak (Hz) a | 0.21 ± 0.03 | 0.20 ± 0.04 | 0.21 ± 0.03 | 0.20 ± 0.01 | 0.22 ± 0.04 | 0.19 ± 0.02 | 0.21 ± 0.04 | 0.19 ± 0.01 | 0.21 ± 0.02 | 0.20 ± 0.02 |
| Non-linear | ||||||||||
| SI b | 5.2 ± 2.4 | 4.6 ± 2.5 | 4.8 ± 1.9 | 3.9 ± 1.4 | 4.4 ± 2.2 | 3.9 ± 1.7 | 4.0 ± 1.6 | 3.5 ± 1.3 | 4.3 ± 2.0 | 3.5 ± 1.3 |
| SD1 (%) a | 34.3 ± 4.9 | 35.0 ± 3.8 | 34.5 ± 5.6 | 33.9 ± 4.0 | 33.7 ± 3.1 | 34.8 ± 3.8 | 34.2 ± 4.1 | 35.0 ± 3.6 | 34.5 ± 4.6 | 36.2 ± 5.4 |
| SD2 (%) a | 65.7 ± 4.9 | 65.2 ± 3.8 | 65.5 ± 5.6 | 66.1 ± 4.0 | 66.3 ± 3.1 | 65.2 ± 3.8 | 65.9 ± 4.1 | 65.0 ± 3.6 | 65.5 ± 4.6 | 63.9 ± 5.4 |
| SD2/SD1 b | 1.96 ± 0.37 | 1.89 ± 0.30 | 1.96 ± 0.37 | 1.98 ± 0.33 | 1.99 ± 0.26 | 1.91 ± 0.29 | 1.97 ± 0.35 | 1.89 ± 0.29 | 1.94 ± 0.36 | 1.82 ± 0.41 |
| Time | Variable | Cannabidiol Preparation (30 mg) | ||||
|---|---|---|---|---|---|---|
| 178 | 203 | 340 | 472 | 707 | ||
| Base | HRBP | 53 ± 8 115/70 ± 9/6 | 58 ± 9 116/67 ± 12/8 | 56 ± 16 115/71 ± 8/6 | 54 ± 10 115/70 ± 9/6 | 54 ± 10 115/72 ± 11/8 |
| 30 | HRBP | 57 ± 9 121/70 ± 11/8 | 58 ± 9 120/71 ± 12/5 | 54 ± 10 117/72 ± 11/7 | 56 ± 12 117/70 ± 12/9 | 55 ± 11 121/69 ± 12/9 |
| 60 | HRBP | 58 ± 9 119/72 ± 10/6 | 56 ± 10 119/68 ± 5/7 | 54 ± 9 118/71 ± 10/7 | 55 ± 10 119/69 ± 11/5 | 55 ± 12 120/71 ± 11/8 |
| 120 | HRBP | 64 ± 10 125/68 ± 9/5 | 65 ± 11 122/70 ± 11/6 | 62 ± 10 124/68 | 64 ± 10 118/66 ± 10/7 | 64 ± 12 125/69 ± 9/7 |
| 180 | HRBP | 65 ± 10 123/66 ± 12/9 | 66 ± 11 121/68 ± 10/8 | 65 ± 12 122/66 ± 12/7 | 64 ± 11 123/68 ± 11/9 | 63 ± 10 120/67 ± 12/7 |
| 240 | HRBP | 65 ± 12 124/66 ± 9/5 | 67 ± 10 125/67 ± 9/4 | 64 ± 13 122/67 ± 7/8 | 66 ± 11 124/66 ± 11/7 | 63 ± 9 119/68 ± 12/8 |
| Study | Formulation, Administration, CBD Single Dose (mg) | Tmax (h) | Cmax (ng/mL) | AUC0-t (h × ng/mL) | AUC0-inf (h × ng/mL) | t½ (h) | Ka (1/h) | Ke (1/h) | Vd (L) |
|---|---|---|---|---|---|---|---|---|---|
| [26] | Oral capsule (CBD + THC) 5.4 mg | 0.99 | 0.93 | 4.35 | |||||
| [27] | Oral capsule (CBD + THC) 5.4 mg | 1.0 | 0.95 | ||||||
| [28] | GW oral capsule (CBD + THC) 10 mg | 1.27 | 2.47 | 5.76 | 6.03 | 1.09 | |||
| [29] | Oral capsule (CBD + THC) 10 mg | 1 | 2.1 | 6.9 | |||||
| [21] | PTL101 * CBD oral capsule 10 mg | 3 | 3.22 | 9.64 | 10.31 | 2.95 | 0.1 | ||
| [30] | PTL401 * (CBD + THC) oral capsule 10 mg | 1.25 | 2.94 | 9.85 | 10.52 | 3.21 | 0.29 | ||
| [18] | Oral capsule MCT-CBD ** 25 mg | 3.0 | 3.05 | 9.51 | 19.23 | ||||
| [18] | Oral capsule SEEDS-CBD *** 25 mg | 13.53 | 27.15 | 32.63 | |||||
| [12] | Caliper CBD **** water soluble 30 mg | 0.9 | 2.82 | 6.80 | 7.94 | 2.54 | 1.68 | 0.66 | |
| [12] | Caliper CBD lipid soluble 30 mg | 1.5 | 0.65 | 1.51 | 1.64 | 2.30 | 1.14 | 0.72 | |
| Preparation 178 CBD 30 mg | 3.29 | 2.20 | 4.58 | - | - | 0.32 | - | - | |
| Preparation 203 CBD 30 mg | 3.39 | 1.29 | 2.30 | - | - | 0.24 | - | - | |
| Preparation 340 CBD 30 mg | 1.28 | 3.54 | 7.81 | 13.81 | 2.20 | 1.19 | 0.40 | 7428 | |
| Preparation 472 CBD 30 mg | 1.53 | 2.88 | 6.32 | 9.96 | 5.18 | 1.87 | 0.27 | 20,178 | |
| Preparation 707 CBD 30 mg | 0.70 | 5.57 | 9.12 | 10.77 | 1.42 | 1.43 | 0.56 | 8024 | |
| [21] | PTL101 CBD oral capsule 100 mg | 3.5 | 47.44 | 150 | 153 | 3.59 | |||
| [31] | (Epidiolex®) 200 mg | 2.3 | 148.0 | 449 | 474 | 8.58 | 4105 | ||
| [32] | (Epidiolex®) 200 mg | 2.5 | 200.0 | 671 | 600 | 15.5 | 6661 | ||
| [32] | (Epidiolex®) 200 mg | 2.0 | 172.0 | 530 | 522 | 14.6 | 7778 | ||
| [32] | (Epidiolex®) 200 mg | 2.5 | 155.0 | 532 | 601 | 13.1 | 6016 | ||
| [32] | (Epidiolex®) 200 mg | 2.5 | 153.0 | 464 | 499 | 11.2 | 5800 | ||
| [33] | GW oral CBD capsule 400 mg | 3 | 181.2 | 704 | |||||
| [33] | GW oral CBD capsule 400 mg | 1.5 | 114.2 | 482 | |||||
| [34] | (Epidiolex®) 750 mg | 4.0 | 187.0 | 1077 | 1190 | 39.7 | |||
| [33] | GW oral CBD capsule 800 mg | 3 | 221.1 | 867 | |||||
| [33] | GW oral CBD capsule 800 mg | 4 | 157.1 | 722 | |||||
| [35] | GW oral solution 1500 mg | 4 | 292.4 | 1517 | 1618 | 14.43 | 20,963 | ||
| [35] | GW oral solution 1500 mg | 3.5 | 335.4 | 1987 | 2198 | 30.33 | 34,101 | ||
| [36] | (Epidiolex®) 1500 mg | 6.13 | 524.5 | 2650 | 2713 | ||||
| [35] | GW oral solution 3000 mg | 5 | 533.0 | 2669 | 2802 | 14.39 | 23,357 | ||
| [35] | GW oral solution 4500 mg | 5 | 722.1 | 3215 | 3426 | 16.61 | 36,575 | ||
| [36] | (Epidiolex®) 4500 mg | 4.07 | 426.9 | 2339 | |||||
| [35] | GW oral solution 6000 mg | 5 | 782 | 3696 | 3900 | 15.42 | 42,849 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Williams, N.N.B.; Ewell, T.R.; Abbotts, K.S.S.; Harms, K.J.; Woelfel, K.A.; Dooley, G.P.; Weir, T.L.; Bell, C. Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability. Pharmaceuticals 2021, 14, 35. https://doi.org/10.3390/ph14010035
Williams NNB, Ewell TR, Abbotts KSS, Harms KJ, Woelfel KA, Dooley GP, Weir TL, Bell C. Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability. Pharmaceuticals. 2021; 14(1):35. https://doi.org/10.3390/ph14010035
Chicago/Turabian StyleWilliams, Natasha N. Bondareva, Taylor Russell Ewell, Kieran Shay Struebin Abbotts, Kole Jerel Harms, Keith A. Woelfel, Gregory P. Dooley, Tiffany L. Weir, and Christopher Bell. 2021. "Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability" Pharmaceuticals 14, no. 1: 35. https://doi.org/10.3390/ph14010035
APA StyleWilliams, N. N. B., Ewell, T. R., Abbotts, K. S. S., Harms, K. J., Woelfel, K. A., Dooley, G. P., Weir, T. L., & Bell, C. (2021). Comparison of Five Oral Cannabidiol Preparations in Adult Humans: Pharmacokinetics, Body Composition, and Heart Rate Variability. Pharmaceuticals, 14(1), 35. https://doi.org/10.3390/ph14010035






