Eluxadoline Loaded Solid Lipid Nanoparticles for Improved Colon Targeting in Rat Model of Ulcerative Colitis
Abstract
1. Introduction
2. Results and Discussion
2.1. Particles Characterization
2.2. Percent Drug Entrapment and Loading
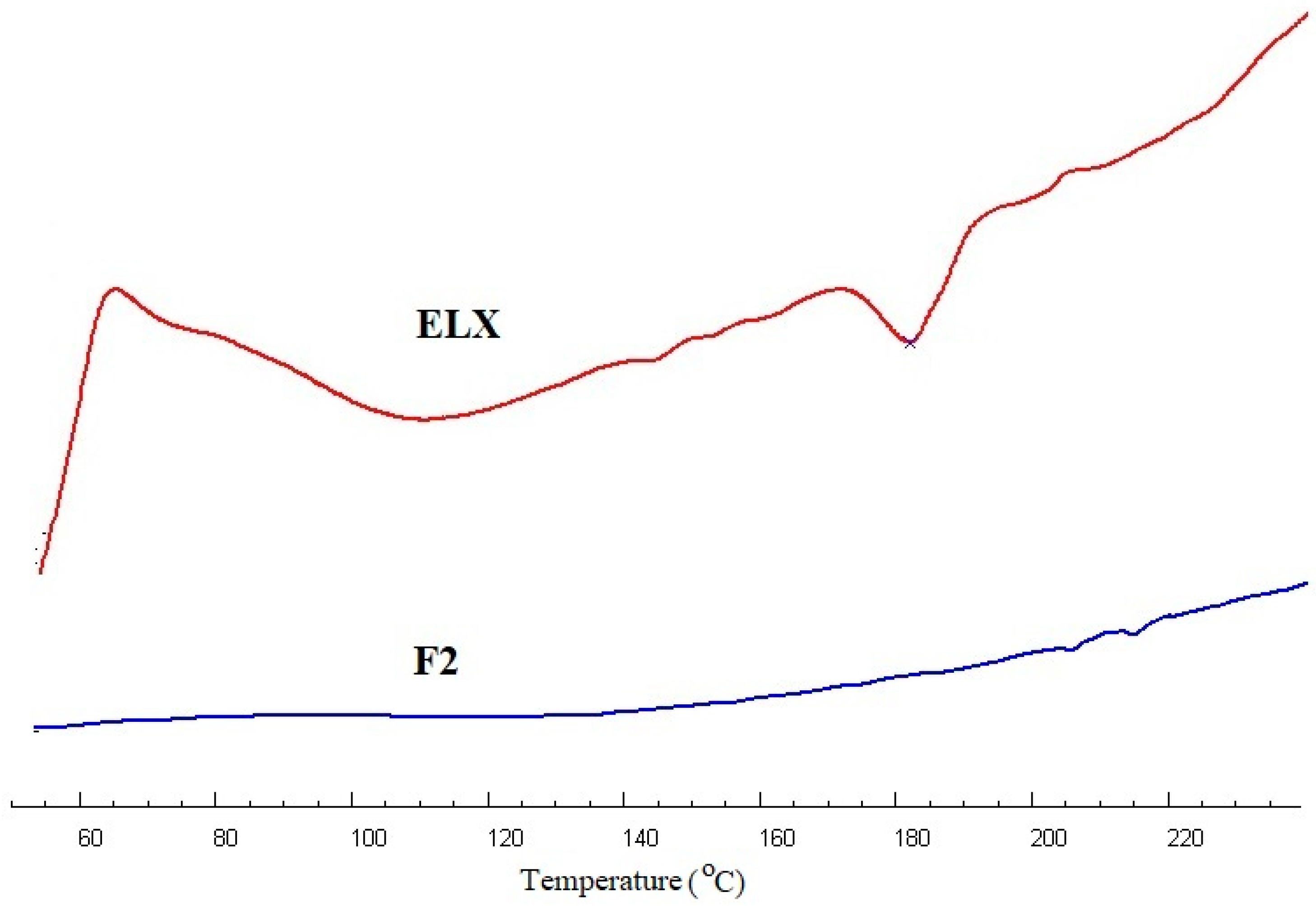
2.3. Thermal Analysis
2.4. FTIR Spectra Analysis
2.5. In-Vitro Drug Release Profile
2.6. Morphology
2.7. Stability Studies
2.8. In Vivo Studies: Assessment of Ulcerative Colitis
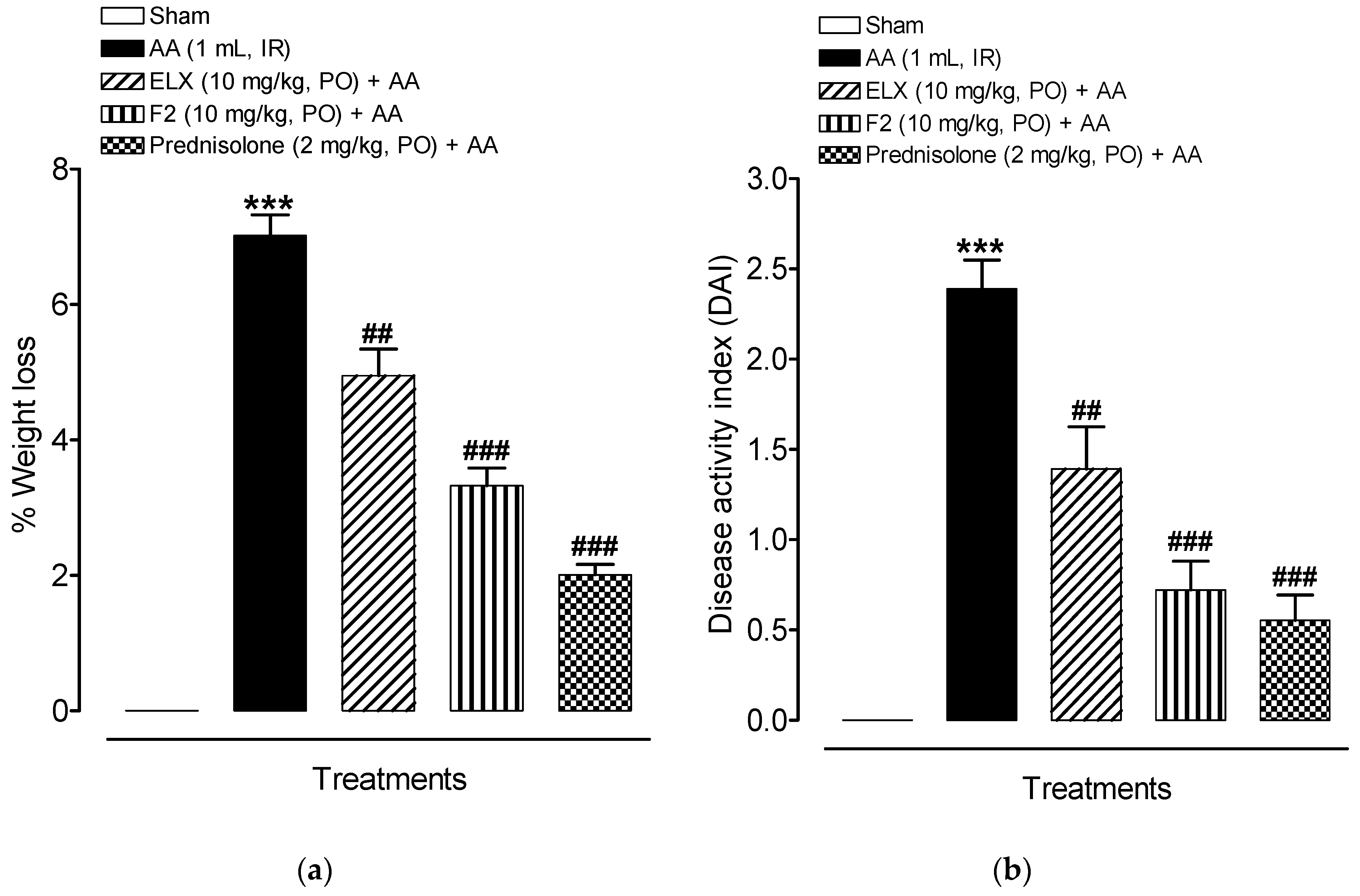
2.8.1. Effect of ELX and F2 on Disease Activity Index (DAI)
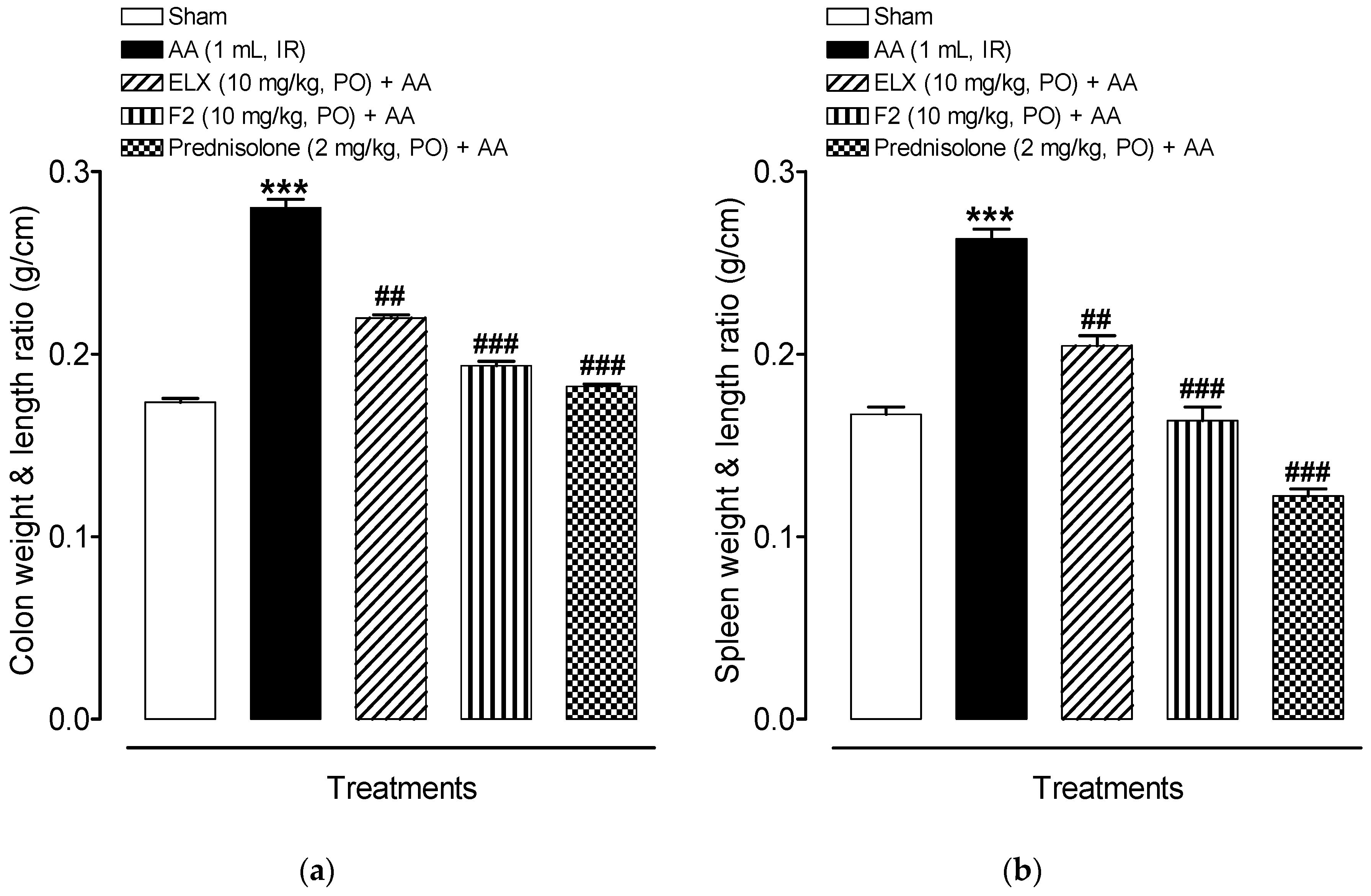
2.8.2. Effect of ELX and F2 on Macroscopic Damage
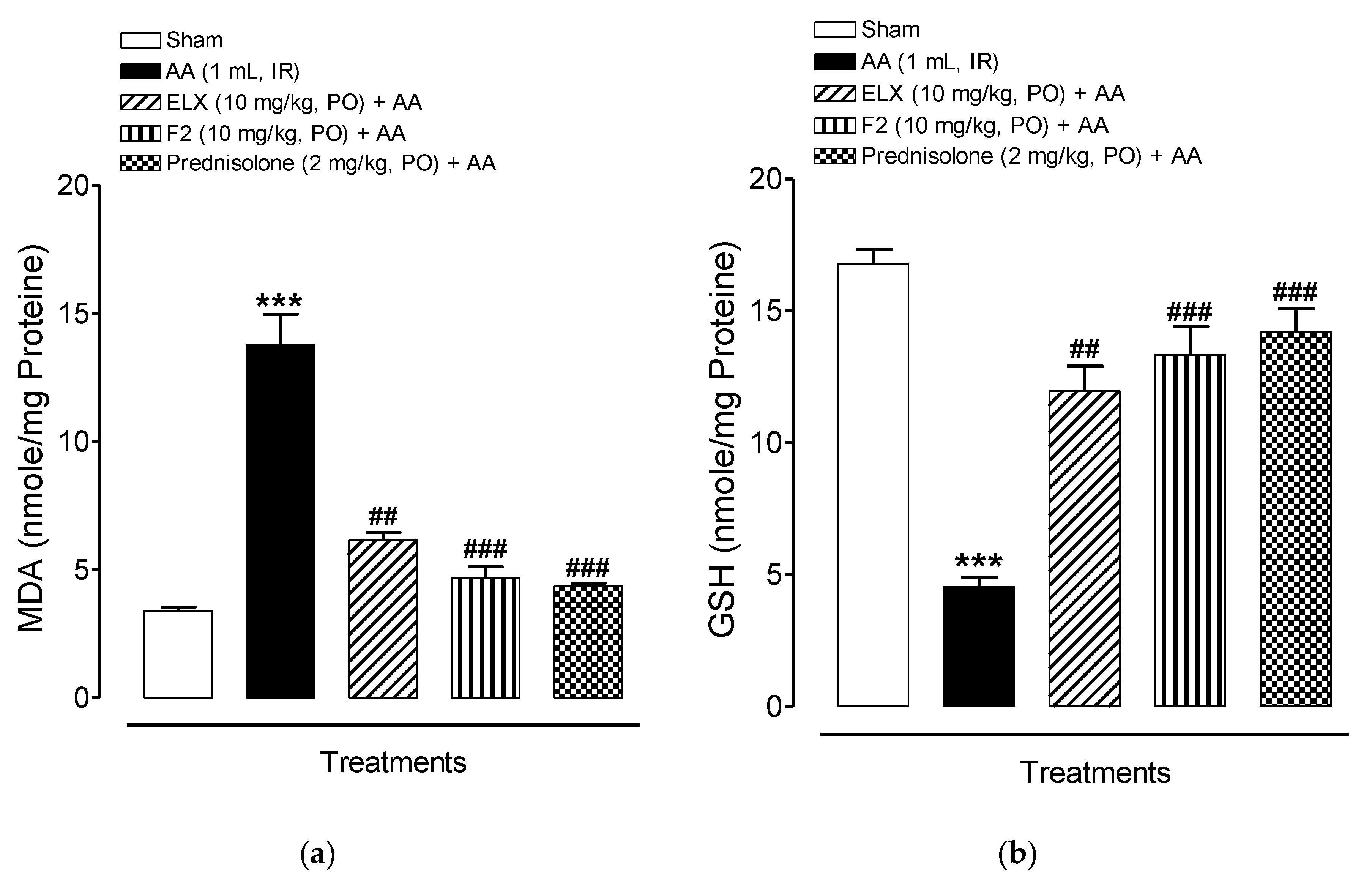
2.8.3. Effect of ELX and F2 on Lipid Peroxidation Activity
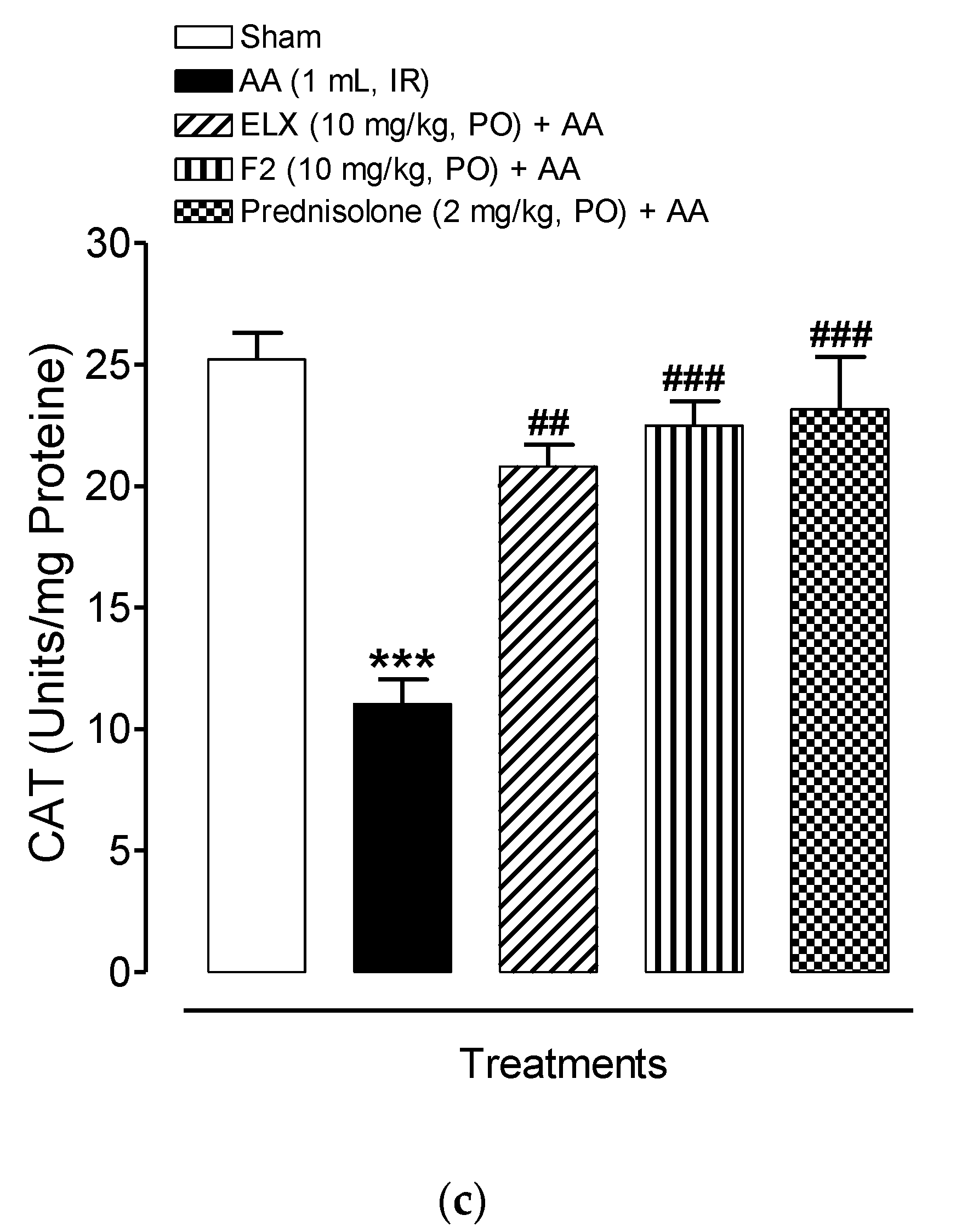
2.8.4. Effect of ELX and F2 on Antioxidant Activity
2.8.5. Effect of ELX and F2 on Microscopic Damage
3. Materials and Method
3.1. Materials
3.2. Preparation of ELX Loaded Solid Lipid Nanoparticle (SLNs)
3.3. Particles Characterization
3.4. Percent Drug Entrapment (%EE) and Loading (%DL)
3.5. Thermal Analysis
3.6. FTIR Spectra Analysis
3.7. Morphology
3.8. In-Vitro Release Studies
3.9. Stability Studies
3.10. In Vivo Studies: Assessment of Ulcerative Colitis
3.10.1. Experimental Animals
3.10.2. Study Design
3.10.3. Induction of Acetic Acid Colitis
3.10.4. Assessment of Disease Activity Index (DAI)
3.10.5. Assessment of Macroscopic Damage
3.10.6. Assessment of Lipid Peroxidation Activity
3.10.7. Assessment of Antioxidant Activity
3.10.8. Microscopic Assessment of UC
3.11. Statistical Analysis
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Saha, L. Irritable bowel syndrome: Pathogenesis, diagnosis, treatment, and evidence-based medicine. World. J. Gastroenterol. 2014, 20, 6759–6773. [Google Scholar] [CrossRef] [PubMed]
- Longstreth, G.F.; Thompson, W.G.; Chey, W.D.; Houghton, L.A.; Mearin, F.; Spiller, R.C. Functional bowel disorders. Gastroenterology 2006, 130, 1480–1491. [Google Scholar] [CrossRef] [PubMed]
- Anwer, M.K.; Al-Shdefat, R.; Ezzeldin, E.; Alshahrani, S.M.; Alshetaili, A.S.; Iqbal, M. Preparation, Evaluation and Bioavailability Studies of Eudragit Coated PLGA Nanoparticles for Sustained Release of Eluxadoline for the Treatment of Irritable Bowel Syndrome. Front. Pharmacol. 2017, 8, 844. [Google Scholar] [CrossRef] [PubMed]
- Covington, P.S.; Dove, S. Methods of Treatment Using Eluxadoline. U.S. Patent No. WO2018069770A1, 19 April 2018. [Google Scholar]
- Satti, M.B.; Al-Quorain, A.; Al-Gindan, Y.; Al-Hamdan, A. Chronic idiopathic ulcerative colitis in Saudi Arabia: A clinicopathological study of 76 cases. Ann. Saudi Med. 1996, 16, 637–640. [Google Scholar] [CrossRef] [PubMed]
- Lacy, B.E.; Harris, L.A.; Chang, L.; Lucak, S.; Gutman, C.; Dove, L.S.; Covington, P.S.; Lembo, A. Impact of patient and disease characteristics on the efficacy and safety of eluxadoline for IBS-D: A subgroup analysis of phase III trials. Ther. Adv. Gastroenterol. 2019, 12. [Google Scholar] [CrossRef]
- Vera, I.; Júdez, F.J. Eluxadoline in the treatment of diarrhea-predominant irritable bowel syndrome. The SEPD perspective. Rev. Esp. Enferm. Dig. 2017, 109, 788–794. [Google Scholar] [CrossRef]
- Davenport, J.M.; Covington, P.; Bonifacio, L.; McIntyre, G.; Venitz, J. Effect of uptake transporters OAT3 and OATP1B1 and efflux transporter MRP2 on the pharmacokinetics of eluxadoline. J. Clin. Pharm. 2015, 55, 534–542. [Google Scholar] [CrossRef]
- Iqbal, M.; Ezzeldin, E.; Al-Rashood, K.A.; Al-Shdefat, R.; Anwer, M.K. High throughput μ-SPE based elution coupled with UPLC-MS/MS for determination of eluxadoline in plasma sample: Application in pharmacokinetic characterization of PLGA nanoparticle formulations in rats. J. Pharm. Biomed. Anal. 2018, 149, 172–178. [Google Scholar] [CrossRef]
- Marbury, T.C.; Berg, J.K.; Dove, L.S.; Covington, P.S. Effect of hepatic impairment on eluxadoline pharmacokinetics. J. Clin. Pharm. 2017, 57, 1454. [Google Scholar] [CrossRef]
- Kumari, P.; Ghosh, B.; Biswas, S. Nanocarriers for cancer-targeted drug delivery. J. Drug. Target. 2016, 24, 179–191. [Google Scholar] [CrossRef]
- Palmerston, M.L.; Pan, J.; Torchilin, V.P. Dendrimers as Nanocarriers for Nucleic Acid and Drug Delivery in Cancer Therapy. Molecules 2017, 22, 1401. [Google Scholar] [CrossRef]
- Daeihamed, M.; Dadashzadeh, S.; Haeri, A.; Akhlaghi, M.F. Potential of Liposomes for Enhancement of Oral Drug Absorption. Curr. Drug Deliv. 2017, 14, 289–303. [Google Scholar] [CrossRef] [PubMed]
- de Araújo, D.R.; Ribeiro, L.N.M.; de Paula, E. Lipid-based carriers for the delivery of local anesthetics. Expert. Opin. Drug Deliv. 2019, 16, 701–714. [Google Scholar] [CrossRef] [PubMed]
- Huang, Z.R.; Hua, S.C.; Yang, Y.L.; Fang, J.Y. Development and evaluation of lipid nanoparticles for camptothecin delivery: A comparison of solid lipid nanoparticles, nanostructured lipid carriers, and lipid emulsion. Acta. Pharm. Sin. 2008, 29, 1094–1102. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Ng, W.K.; Tan, R.B. Are nanostructured lipid carriers (NLCs) better than solid lipid nanoparticles (SLNs): Development, characterizations and comparative evaluations of clotrimazole-loaded SLNs and NLCs? Eur. J. Pharm. Sci. 2012, 47, 139–151. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, J.; Ye, N.; Luo, Z.; Lai, W.; Cai, X.; Lin, Y. Absorption, pharmacokinetics and disposition properties of solid lipid nanoparticles (SLNs). Curr. Drug Metab. 2012, 13, 447–456. [Google Scholar] [CrossRef]
- Tapeinos, C.; Battaglini, M.; Ciofani, G. Advances in the design of solid lipid nanoparticles and nanostructured lipid carriers for targeting brain diseases. J. Contrl. Rel. 2017, 264, 306–332. [Google Scholar] [CrossRef]
- Uner, M.; Yener, G. Importance of solid lipid nanoparticles (SLN) in various administration routes and future perspectives. Int. J. Nanomed. 2007, 2, 289–300. [Google Scholar]
- Yassin, A.E.; Anwer, M.K.; Mowafy, H.A.; El-Bagory, I.M.; Bayomi, M.A.; Alsarra, I.A. Optimization of 5-flurouracil solid-lipid nanoparticles: A preliminary study to treat colon cancer. Int. J. Med. Sci. 2010, 7, 398–408. [Google Scholar] [CrossRef]
- Ozturk, A.A.; Aygul, A.; Senel, B. Influence of glyceryl behenate, tripalmitin and stearic acid on the properties of clarithromycin incorporated solid lipid nanoparticles (SLNs): Formulation, characterization, antibacterial activity and cytotoxicity. J. Drug Deliv. Sci. Technol. 2019, 54, 101240. [Google Scholar] [CrossRef]
- Center for Drug Evaluation and Research. Chemistry Review. Application Number. 206940Orig1s000. 2015. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/nda/2015/206940Orig1s000ChemR.pdf (accessed on 10 September 2020).
- Anwer, M.K.; Mohammad, M.; Ezzeldin, E.; Fatima, F.; Alalaiwe, A.; Iqbal, M. Preparation of sustained release apremilast-loaded PLGA nanoparticles: In vitro characterization and in vivo pharmacokinetic study in rats. Int. J. Nanomed. 2019, 14, 1587–1595. [Google Scholar] [CrossRef] [PubMed]
- Prasad, P.U.; Kumar, R.A.S. A novel validated RP-HPLC-DAD method for the estimation of Eluxadoline in bulk and pharmaceutical dosage form. Res. J. Pharm. Technol. 2015, 8, 1469–1476. [Google Scholar] [CrossRef]
- Anwer, M.K.; Jamil, S.; Ansari, M.J.; Al-Shdefat, R.; Ali, B.E.; Ganaie, M.A.; Abdel-Kader, M.S.; Shakeel, F. Water soluble binary and ternary complexes of diosmin with β-cyclodextrin: Spectroscopic characterization, release studies and anti-oxidant activity. J. Mol. Liq. 2014, 199, 35–41. [Google Scholar] [CrossRef]
- Ali, H.; Weigmann, B.; Neurath, M.F.; Collnot, E.M.; Windbergs, M.; Lehr, C.M. Budesonide loaded nanoparticles with pH-sensitive coating for improved mucosal targeting in mouse models of inflammatory bowel diseases. J. Contrl. Rel. 2014, 183, 167–177. [Google Scholar] [CrossRef]
- del Pozo-Rodríguez, A.; Solinís, M.A.; Gascón, A.R.; Pedraz, J.L. Short- and long-term stability study of lyophilized solid lipid nanoparticles for gene therapy. Eur. J. Pharm. Biopharm. 2009, 71, 181–189. [Google Scholar] [CrossRef]
- National Research Council. Guide for the Care and Use of Laboratory Animals; National Academy Press: Washington, DC, USA, 1996; pp. 1–7. [Google Scholar]
- MacPherson, B.R.; Pfeiffer, C.J. Experimental production of diffuse colitis in rats. Digestion 1978, 17, 135–150. [Google Scholar] [CrossRef]
- Wirtz, S.; Neufert, C.; Weigmann, B.; Neurath, M.F. Chemically induced mouse models of intestinal inflammation. Nat. Protoc. 2007, 2, 541–546. [Google Scholar] [CrossRef]
- Minaiyan, M.; Ghannadi, A.; Karimzadeh, A. Anti-ulcerogenic effect of ginger (rhizome of Zingiber officinale Roscoe) on cystemine induced duodenal ulcer in rats. Daru J. Pharm Sci. 2006, 14, 97–101. [Google Scholar]
- Morris, G.P.; Beck, P.L.; Herridge, M.S.; Depew, W.T.; Szewczuk, M.R.; Wallace, J.L. Hapten-induced model of chronic inflammation and ulceration in the rat colon. Gastroenterology 1989, 96, 795–803. [Google Scholar] [CrossRef]
- Esterbauer, H.; Cheeseman, K.H. Determination of aldehydic lipid peroxidation products: Malonaldehyde and 4-hydroxynonenal. Methods Enzym. 1990, 186, 407–421. [Google Scholar] [CrossRef]
- Jollow, D.J.; Mitchell, J.R.; Zampaglione, N.; Gillette, J.R. Bromobenzene-induced liver necrosis. Protective role of glutathione and evidence for 3,4-bromobenzene oxide as the hepatotoxic metabolite. Pharmacology 1974, 11, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Claiborne, A.L. Assay of Catalase. In Handbook of Methods of Oxygen Radical Research; Greenwald, R.A., Ed.; CRC Press: Boca Raton, FL, USA, 1985; pp. 283–285. [Google Scholar]






| SLNs | SIZE ± SD (nm) | PDI | ZP ± SD (mV) | %EE ± SD | %DL ± SD |
|---|---|---|---|---|---|
| F1 | 394.3 ± 8.4 | 0.226 ± 0.03 | 26.8 ± 5.41 | 73.0 ± 3.2 | 5.13 ± 1.2 |
| F2 | 266.0 ± 6.4 | 0.217 ± 0.04 | 31.2 ± 5.19 | 65.0 ± 4.8 | 4.60 ± 0.8 |
| F3 | 1570.5 ± 14.2 | 0.882 ± 0.06 | 25.7 ± 6.11 | 54.1 ± 2.6 | 3.82 ± 0.7 |
| Months | Conditions | Particle Size (nm ± SD) | PDI (± SD) | ZP (mV) (mV ± SD) | Entrapment Efficiency (% ± SD) | Release (% ± SD) |
|---|---|---|---|---|---|---|
| 0 | - | 266 ± 6.4 | 0.217 ± 0.04 | 31.2 ± 5.1 | 65.0 ± 4.8 | 95.7 ± 4.7 |
| 1 | 30 ± 2 °C/65 ± 5% RH | 268 ± 4.5 | 0.243 ± 0.02 | 30.3 ± 6.3 | 67.2 ± 2.7 | 96.5 ± 3.1 |
| 2 | 272 ± 7.4 | 0.249 ± 0.06 | 27.2 ± 5.8 | 63.6 ± 3.9 | 94.6 ± 4.4 | |
| 3 | 276 ± 8.5 | 0.212 ± 0.04 | 30.5 ± 4.2 | 62.4 ± 8.3 | 91.4 ± 7.3 | |
| 1 | 40 ± 2 °C/75 ± 5% RH | 267 ± 2.5 | 0.223 ± 0.07 | 33.6 ± 9.4 | 65.2 ± 4.8 | 93.6 ± 6.4 |
| 2 | 273 ± 6.8 | 0.279 ± 0.09 | 29.2 ± 6.3 | 63.6 ± 2.6 | 93.2 ± 7.3 | |
| 3 | 276 ± 7.5 | 0.272 ± 0.08 | 25.7 ± 6.7 | 61.5 ± 5.9 | 90.8 ± 6.2 |
| SLNs | ELX (mg) | Lipids (400 mg) | Soyalecithin (%w/v) |
|---|---|---|---|
| F1 | 40 | Glyceryl monostearate | 0.5 |
| F2 | 40 | Stearic acid | 0.5 |
| F3 | 40 | Glyceryl trioctanoate | 0.5 |
| Weight Loss (%) | Stool Consistency | Occult/Gross Bleeding | Score |
|---|---|---|---|
| Normal | Normal | Normal | 0 |
| 1–5 | Soft but still formed | - | 1 |
| 5–10 | Loose stools | Hemo-occult positive | 2 |
| 10–20 | Diarrhea | - | 3 |
| >20 | - | Gross bleeding | 4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anwer, M.K.; Ahmed, M.M.; Aldawsari, M.F.; Alshahrani, S.; Fatima, F.; Ansari, M.N.; Rehman, N.U.; Al-Shdefat, R.I. Eluxadoline Loaded Solid Lipid Nanoparticles for Improved Colon Targeting in Rat Model of Ulcerative Colitis. Pharmaceuticals 2020, 13, 255. https://doi.org/10.3390/ph13090255
Anwer MK, Ahmed MM, Aldawsari MF, Alshahrani S, Fatima F, Ansari MN, Rehman NU, Al-Shdefat RI. Eluxadoline Loaded Solid Lipid Nanoparticles for Improved Colon Targeting in Rat Model of Ulcerative Colitis. Pharmaceuticals. 2020; 13(9):255. https://doi.org/10.3390/ph13090255
Chicago/Turabian StyleAnwer, Md. Khalid, Mohammed Muqtader Ahmed, Mohammed F. Aldawsari, Saad Alshahrani, Farhat Fatima, Mohd Nazam Ansari, Najeeb Ur Rehman, and Ramadan I. Al-Shdefat. 2020. "Eluxadoline Loaded Solid Lipid Nanoparticles for Improved Colon Targeting in Rat Model of Ulcerative Colitis" Pharmaceuticals 13, no. 9: 255. https://doi.org/10.3390/ph13090255
APA StyleAnwer, M. K., Ahmed, M. M., Aldawsari, M. F., Alshahrani, S., Fatima, F., Ansari, M. N., Rehman, N. U., & Al-Shdefat, R. I. (2020). Eluxadoline Loaded Solid Lipid Nanoparticles for Improved Colon Targeting in Rat Model of Ulcerative Colitis. Pharmaceuticals, 13(9), 255. https://doi.org/10.3390/ph13090255







