Antibacterial and Antibiofilm Activity of Myrtenol against Staphylococcus aureus
Abstract
1. Introduction
2. Results and Discussion
2.1. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC) of Myrtenol against S. aureus
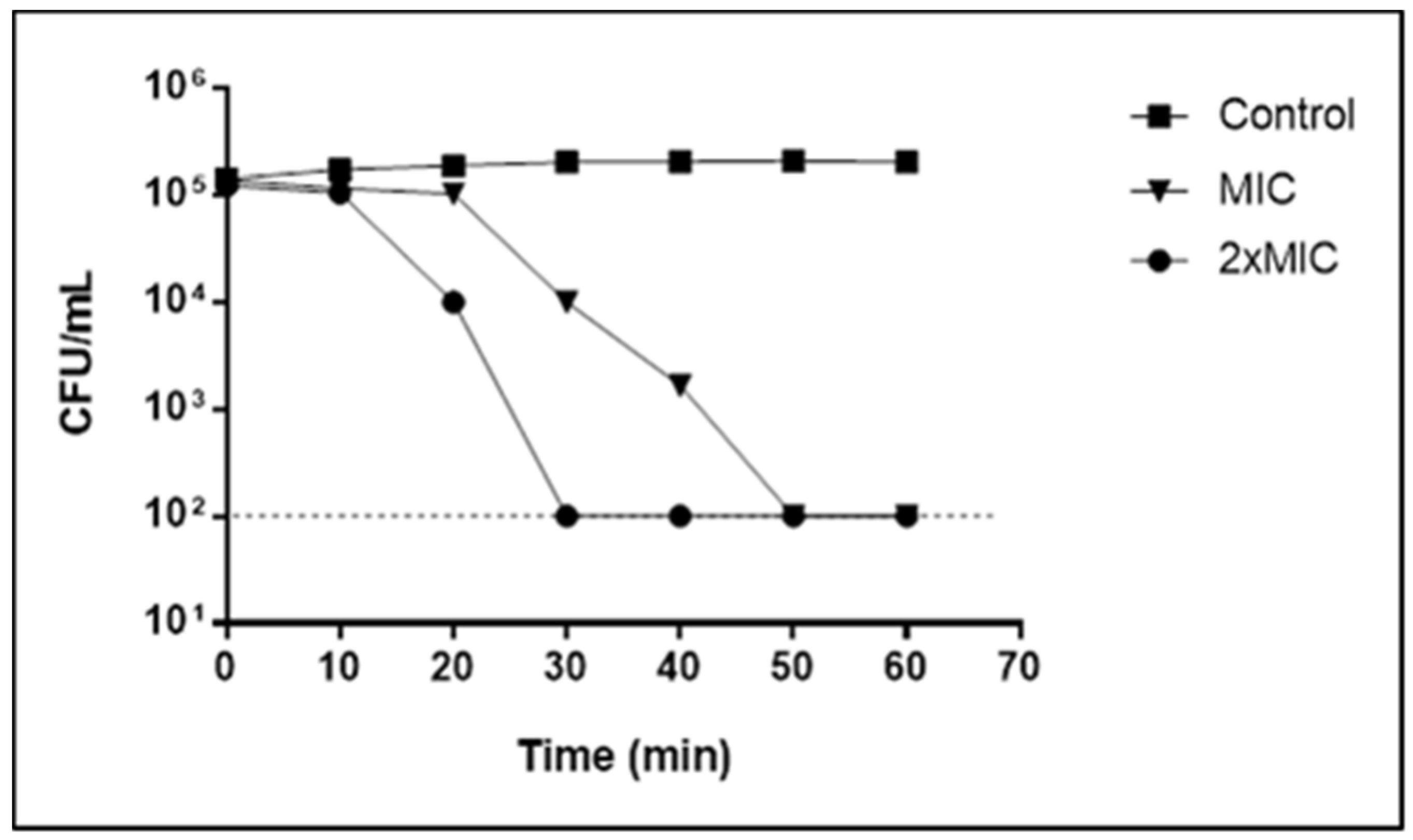
2.2. Myrtenol Time-Kill Kinetics against S. aureus
2.3. Molecular Docking Analysis
2.4. Association Test of Myrtenol with Antibacterial Drugs against S. aureus
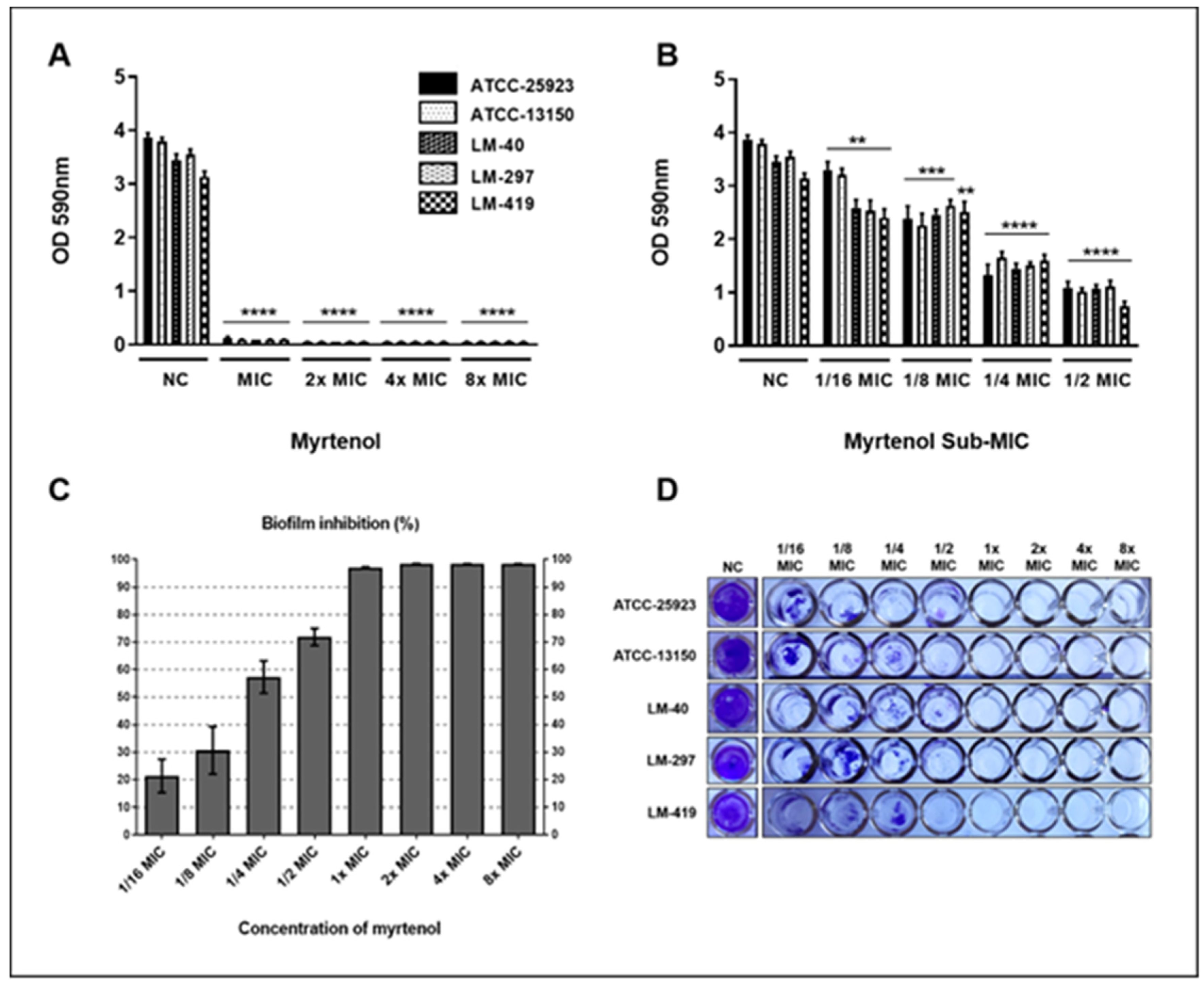
2.5. Antibiofilm Effect of Myrtenol against S. aureus
2.6. In Silico Studies of Myrtenol Lipinski’s Parameters
3. Materials and Methods
3.1. Substances
3.2. Strains
3.3. Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
3.4. Time-Kill Kinetics
3.5. Molecular Docking
3.6. Association Test
3.7. Antibiofilm Effect
3.8. In Silico Studies of Lipinski’s Parameters
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATCC | American Type Culture Collection |
| BHI | Cerebral Heart Infusion (Broth) |
| CFU | Colony Forming Units |
| DMSO | Dimethyl Sulfoxide |
| FIC | Fractional Inhibitory Concentration |
| FICI | Fractional Inhibitory Concentration Index |
| FIC | Fractional Inhibitory Concentration |
| MBC | Minimum Bactericidal Concentration |
| MIC | Minimum Inhibitory Concentration |
| MVD | Molegro Virtual Docker |
| MW | Molecular Weight |
| PDB | Protein Data Bank |
| RMSD | Root Mean Standard Deviation |
| TPSA | Topological Polar Surface Area |
References
- Al-Mebairik, N.F.; El-Kersh, T.A.; Al-Sheikh, Y.A.; Marie, M.A.M. A review of virulence factors, pathogenesis, and antibiotic resistance in Staphylococcus aureus. Rev. Med. Microbiol. 2016, 27, 50–56. [Google Scholar] [CrossRef]
- Venkatesh, V. Staphylococcus aureus and MRSA: Do we know the true burden? Clin. Epidemiol. Glob. Health 2018, 6, 103–104. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Global Priority List of Antibiotic-Resistant Bacteria to Guide Research, Discovery, and Development of New Antibiotics. 2017. Available online: http://www.who.int/medicines/publications/global-priority-list-antibiotic-resistant-bacteria/en/ (accessed on 16 April 2020).
- Ciofu, O.; Rojo-Molinero, E.; Macià, M.D.; Oliver, A. Antibiotic treatment of biofilm infections. APMIS 2017, 125, 304–319. [Google Scholar] [CrossRef] [PubMed]
- Stewart, P.S. Antimicrobial tolerance in biofilms. Microbiol. Spectr. 2015, 3, MB-0010-2014. [Google Scholar] [CrossRef] [PubMed]
- Ranieri, M.R.; Whitchurch, C.B.; Burrows, L.L. Mechanisms of biofilm stimulation by subinhibitory concentrations of antimicrobials. Curr. Opin. Microbiol. 2018, 45, 164–169. [Google Scholar] [CrossRef] [PubMed]
- Chandra, H.; Bishnoi, P.; Yadav, A.; Patni, B.; Mishra, A.P.; Nautiyal, A.R. Antimicrobial resistance and the alternative resources with special emphasis on plant-based antimicrobials—A review. Plants 2017, 6, 16. [Google Scholar] [CrossRef]
- Simões, M.; Bennett, R.N.; Rosa, E.A. Understanding antimicrobial activities of phytochemicals against multidrug resistant bacteria and biofilms. Nat. Prod. Rep. 2009, 26, 746–757. [Google Scholar] [CrossRef]
- Asllani, U. Chemical composition of Albanian myrtle oil (Myrtus communis L.). J. Essent. Oil Res. 2000, 12, 140–142. [Google Scholar] [CrossRef]
- Sisay, M.; Gashaw, T. Ethnobotanical, ethnopharmacological, and phytochemical studies of Myrtus communis Linn: A popular herb in Unani system of medicine. Evid.-Based Complementary Altern. Med. 2017, 22, 1035–1043. [Google Scholar] [CrossRef]
- Moreira, M.R.C.; Salvadori, M.G.D.S.S.; de Almeida, A.A.C.; de Sousa, D.P.; Jordán, J.; Satyal, P.; Freitas, R.M.; de Almeida, R.N. Anxiolytic-like effects and mechanism of (−)-myrtenol: A monoterpene alcohol. Neurosci. Lett. 2014, 579, 119–124. [Google Scholar] [CrossRef]
- Gomes, B.S.; Neto, B.P.; Lopes, E.M.; Cunha, F.V.; Araújo, A.R.; Wanderley, C.W.; Wong, D.V.; Júnior, R.C.P.; Ribeiro, R.A.; De Sousa, D.P.; et al. Anti-inflammatory effect of the monoterpene myrtenol is dependent on the direct modulation of neutrophil migration and oxidative stress. Chem. Interact. 2017, 273, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Viana, A.F.S.C.; da Silva, F.V.; Fernandes, H.D.B.; Oliveira, I.S.; Braga, M.A.; Nunes, P.I.G.; Viana, D.A.; Sousa, D.P.; Rao, V.S.; Oliveira, R.C.M.; et al. Gastroprotective effect of (-)-myrtenol against ethanol-induced acute gastric lesions: Possible mechanisms. J. Pharm. Pharmacol. 2016, 68, 1085–1092. [Google Scholar] [CrossRef] [PubMed]
- Selvaraj, A.; Jayasree, T.; Valliammai, A.; Pandian, S.K. Myrtenol attenuates MRSA biofilm and virulence by suppressing sarA expression dynamism. Front. Microbiol. 2019, 10, 2027. [Google Scholar] [CrossRef]
- Al-Mariri, A.; Swied, G.; Oda, A.; Al Hallab, L. Variation in Myrtus communis L. Essential oil composition and its antibacterial activities components. Biol. Sci. PJSIR 2016, 59, 6–11. [Google Scholar]
- İşcan, G. Antibacterial and Anticandidal Activities of Common Essential Oil Constituents. Rec. Nat. Prod. 2017, 11, 374–388. [Google Scholar]
- Clinical and Laboratory Standards Institute (CLSI). Methods for Determining Bactericidal Activity of Antimicrobial Agents. Approved Guideline M26-A; Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 1999; pp. 1–32. [Google Scholar]
- Pankey, G.A.; Sabath, L.D. Clinical relevance of bacteriostatic versus bactericidal mechanisms of action in the treatment of Gram-positive bacterial infections. Clin. Infect. Dis. 2004, 38, 864–870. [Google Scholar] [CrossRef]
- Thomsen, R.; Christensen, M.H. MolDock: A new technique for high-accuracy molecular docking. J. Med. Chem. 2006, 49, 3315–3321. [Google Scholar] [CrossRef]
- Lim, D.; Strynadka, N.C.J. Structural basis for the β-lactam resistance of PBP2a from methicillin-resistant Staphylococcus aureus. Nat. Struct. Mol. Biol. 2002, 9, 870–876. [Google Scholar] [CrossRef]
- Yap, P.S.X.; Yiap, B.C.; Ping, H.C.; Lim, S.H.E. Essential oils, a new horizon in combating bacterial antibiotic resistance. Open Microbiol. J. 2014, 8, 6. [Google Scholar] [CrossRef]
- Ayaz, M.; Ullah, F.; Sadiq, A.; Ullah, F.; Ovais, M.; Ahmed, J.; Devkota, H.P. Synergistic interactions of phytochemicals with antimicrobial agents: Potential strategy to counteract drug resistance. Chem. Biol. Interact. 2019, 308, 294–303. [Google Scholar] [CrossRef]
- Kwasny, S.M.; Opperman, T.J. Static biofilm cultures of Gram-positive pathogens grown in a microtiter format used for anti-biofilm drug discovery. Curr. Protoc. Pharmacol. 2010, 50, 13A.8.1–13A.8.23. [Google Scholar] [CrossRef]
- Kaplan, J.B.; Izano, E.A.; Gopal, P.; Karwacki, M.T.; Kim, S.; Bose, J.L.; Bayles, K.W.; Horswill, A.R. Low levels of β-lactam antibiotics induce extracellular DNA release and biofilm formation in Staphylococcus aureus. MBio 2012, 3, e00198-12. [Google Scholar] [CrossRef]
- Evren, E.; Yurtcu, E. In vitro effects on biofilm viability and antibacterial and antiadherent activities of silymarin. Folia Microbiol. 2015, 60, 351–356. [Google Scholar] [CrossRef]
- Savage, V.J.; Chopra, I.; O’Neill, A.J. Staphylococcus aureus biofilms promote horizontal transfer of antibiotic resistance. Antimicrob. Agents Chemother. 2013, 57, 1968–1970. [Google Scholar] [CrossRef]
- Mohammed, Y.H.E.; Manukumar, H.; Rakesh, K.; Karthik, C.; Mallu, P.; Qin, H.-L.; Eissa, M.Y.H. Vision for medicine: Staphylococcus aureus biofilm war and unlocking key’s for anti-biofilm drug development. Microb. Pathog. 2018, 123, 339–347. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 46, 3–26. [Google Scholar] [CrossRef]
- Ghose, A.K.; Viswanadhan, V.N.; Wendoloski, J.J. A Knowledge-based approach in designing combinatorial or medicinal chemistry libraries for drug discovery. A qualitative and quantitative characterization of known drug databases. J. Comb. Chem. 1999, 1, 55–68. [Google Scholar] [CrossRef]
- Veber, D.F.; Johnson, S.R.; Cheng, H.U.; Smith, B.R.; Ward, K.W.; Kopple, K.D. Molecular properties that influence the oral Bioavailabillity of Drug candidates. J. Med. Chem. 2002, 45, 2615–2623. [Google Scholar] [CrossRef]
- Egan, W.J.; Merz, K.M.; Baldwin, J.J. Prediction of Drug absorption using multivariate Statistics. J. Med. Chem. 2000, 43, 3867–3877. [Google Scholar] [CrossRef]
- Ali, J.; Camilleri, P.; Brown, M.B.; Hutt, A.J.; Kirton, S.B. In Silico Prediction of Aqueous Solubility Using Simple QSPR Models: The importance of Phenol and Phenol-like moieties. J. Chem. Inf. Model. 2012, 52, 2950–2957. [Google Scholar] [CrossRef]
- Clinical Laboratory Standards Institute (CLSI). Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically. Approved standard M07; Clinical and Laboratory Standards Institute (CLSI): Wayne, PA, USA, 2018; pp. 1–112. [Google Scholar]
- Silva, D.; Diniz-Neto, H.; Cordeiro, L.; Silva-Neta, M.; Silva, S.; Andrade-Júnior, F.; Leite, M.; Nóbrega, J.; Morais, M.; Souza, J.; et al. (R)-(+)-β-Citronellol and (S)-(−)-β-Citronellol in Combination with Amphotericin B against Candida spp. Int. J. Mol. Sci. 2020, 21, 1785. [Google Scholar] [CrossRef] [PubMed]
- Flamm, R.K.; Farrell, D.J.; Rhomberg, P.R.; Scangarella-Oman, N.E.; Sader, H.S. Gepotidacin (GSK2140944) in vitro activity against Gram-positive and Gram-negative bacteria. Antimicrob. Agents Chemother. 2017, 61, e00468-17. [Google Scholar] [CrossRef]
- Wu, X.; Li, Z.; Li, X.; Tian, Y.; Fan, Y.; Yu, C.; Zhou, B.; Liu, Y.; Xiang, R.; Yang, L. Synergistic effects of antimicrobial peptide DP7 combined with antibiotics against multidrug-resistant bacteria. Drug Des. Dev. Ther. 2017, 11, 939–946. [Google Scholar] [CrossRef]
- Balasubramanian, D.; Schneper, L.; Merighi, M.; Smith, R.; Narasimhan, G.; Lory, S.; Mathee, K. The Regulatory Repertoire of Pseudomonas aeruginosa AmpC ß-Lactamase Regulator AmpR Includes Virulence Genes. PLoS ONE 2012, 7, e34067. [Google Scholar] [CrossRef]

| S. aureus | Myrtenol | |||
|---|---|---|---|---|
| MIC | MBC | MIC:MBC | Effect | |
| ATCC-25923 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| ATCC-13150 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-02 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-40 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-45 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-182 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-232 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-297 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-314 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-418 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-419 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| LM-443 | 128 µg/mL | 128 µg/mL | 1:1 | Bactericidal |
| Strains and Drugs | FIC | FICI | Effect 1 |
|---|---|---|---|
| ATCC-25923 | |||
| Myrtenol | 0.25 | 0.50 | Synergism |
| Gentamycin | 0.25 | ||
| Myrtenol | 0.06 | 0.56 | Additivity |
| Ciprofloxacin | 0.50 | ||
| Myrtenol | 0.06 | 1.06 | Indifference |
| Oxacillin | 1.00 | ||
| ATCC-13150 | |||
| Myrtenol | 0.25 | 0.50 | Synergism |
| Gentamycin | 0.25 | ||
| Myrtenol | 0.06 | 0.56 | Additivity |
| Ciprofloxacin | 0.50 | ||
| Myrtenol | 0.06 | 1.06 | Indifference |
| Oxacillin | 1.00 | ||
| LM-40 | |||
| Myrtenol | 0.25 | 0.50 | Synergism |
| Gentamycin | 0.25 | ||
| Myrtenol | 0.25 | 0.75 | Additivity |
| Ciprofloxacin | 0.50 | ||
| Myrtenol | 0.12 | 1.12 | Indifference |
| Oxacillin | 1.00 | ||
| LM-297 | |||
| Myrtenol | 0.25 | 0.50 | Synergism |
| Gentamycin | 0.25 | ||
| Myrtenol | 0.12 | 0.62 | Additivity |
| Ciprofloxacin | 0.50 | ||
| Myrtenol | 0.06 | 1.06 | Indifference |
| Oxacillin | 1.00 | ||
| LM-419 | |||
| Myrtenol | 0.25 | 0.50 | Synergism |
| Gentamycin | 0.25 | ||
| Myrtenol | 0.25 | 0.75 | Additivity |
| Ciprofloxacin | 0.50 | ||
| Myrtenol | 0.12 | 1.12 | Indifference |
| Oxacillin | 1.00 |
| Parameters | Myrtenol |
|---|---|
| Physicochemical Properties | |
| Formula | C10H16O |
| Molecular Weigth | 152.23 g/mol |
| Num. Heavy atoms | 11 |
| Fraction Csp3 | 0.80 |
| Num. Rotatable Bonds | 1 |
| Num. H-bonds acceptors | 1 |
| Num. H-bonds donors | 1 |
| Molar Refractivity | 46.38 |
| TPSA 1 | 20.23 Å2 |
| Lipophilicity | |
| Consensus 2 Log Po/w 3 | 2.40 |
| Water Solubility | |
| Log S (Ali) | −3.32 |
| Class 4 | Soluble |
| Druglikeness | |
| Lipinski 5 | Yes; 0 violation |
| Ghose 6 | No; 1 violation: MW < 160 |
| Veber 7 | Yes; 0 violation |
| Egan 8 | Yes; 0 violation |
| Bioavailability Score | 0.55 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cordeiro, L.; Figueiredo, P.; Souza, H.; Sousa, A.; Andrade-Júnior, F.; Barbosa-Filho, J.; Lima, E. Antibacterial and Antibiofilm Activity of Myrtenol against Staphylococcus aureus. Pharmaceuticals 2020, 13, 133. https://doi.org/10.3390/ph13060133
Cordeiro L, Figueiredo P, Souza H, Sousa A, Andrade-Júnior F, Barbosa-Filho J, Lima E. Antibacterial and Antibiofilm Activity of Myrtenol against Staphylococcus aureus. Pharmaceuticals. 2020; 13(6):133. https://doi.org/10.3390/ph13060133
Chicago/Turabian StyleCordeiro, Laísa, Pedro Figueiredo, Helivaldo Souza, Aleson Sousa, Francisco Andrade-Júnior, José Barbosa-Filho, and Edeltrudes Lima. 2020. "Antibacterial and Antibiofilm Activity of Myrtenol against Staphylococcus aureus" Pharmaceuticals 13, no. 6: 133. https://doi.org/10.3390/ph13060133
APA StyleCordeiro, L., Figueiredo, P., Souza, H., Sousa, A., Andrade-Júnior, F., Barbosa-Filho, J., & Lima, E. (2020). Antibacterial and Antibiofilm Activity of Myrtenol against Staphylococcus aureus. Pharmaceuticals, 13(6), 133. https://doi.org/10.3390/ph13060133