3.2. Chemical Methods
Chemical reagents were commercially obtained from Merck Life Science S.r.l Milano, Italy and were utilized without purification. Monitoring of the reactions occurred via thin-layer chromatography (TLC; silica gel, UV254) with UV light (short-wave ultraviolet 254 nm and long-wave ultraviolet 365 nm). Inert atmosphere of N2 or Ar was used to carry out all reactions that required an anhydrous environment. Column chromatography was conducted using Fluka silica gel 60 Å (63–200 µm) or silica gel Si 60 (40–63 µm) (Merck Life Science S.r.l Milano, Italy). Mass spectrometry was conducted on a HP MS 6890-5973 MDS spectrometer, electron impact 70 eV, equipped with a HP ChemStation or with an Agilent 6530 Series Accurate-Mass Quadrupole Time-of-FLIFHT (Q-TOF) LC/MS (Agilent, Santa Clara CA, USA). A Bruker micro-TOF QII mass spectrometer (Bruker, Billerica, MA, USA) equipped with an electrospray ion source (ESI) was used to carry out high-resolution mass spectroscopy (HRMS) analyses. 1H NMR spectra were obtained deuterated solvents on a Varian Mercury 300 (Varian Inc., Palo Alto, CA, USA) or an Agilent VNMRS500 spectrometer (Agilent, Santa Clara CA, USA). Chemical shifts (δ) are expressed as parts per million (ppm) and coupling constants (J) in Hertz (Hz). Melting points were measured on a Gallenkamp electrothermal apparatus (Fisons Erba Science Ltd. Guildford, UK) in open capillaries and are uncorrected.
3.2.1. General Procedure for the Preparation of Sulfonamide Intermediates
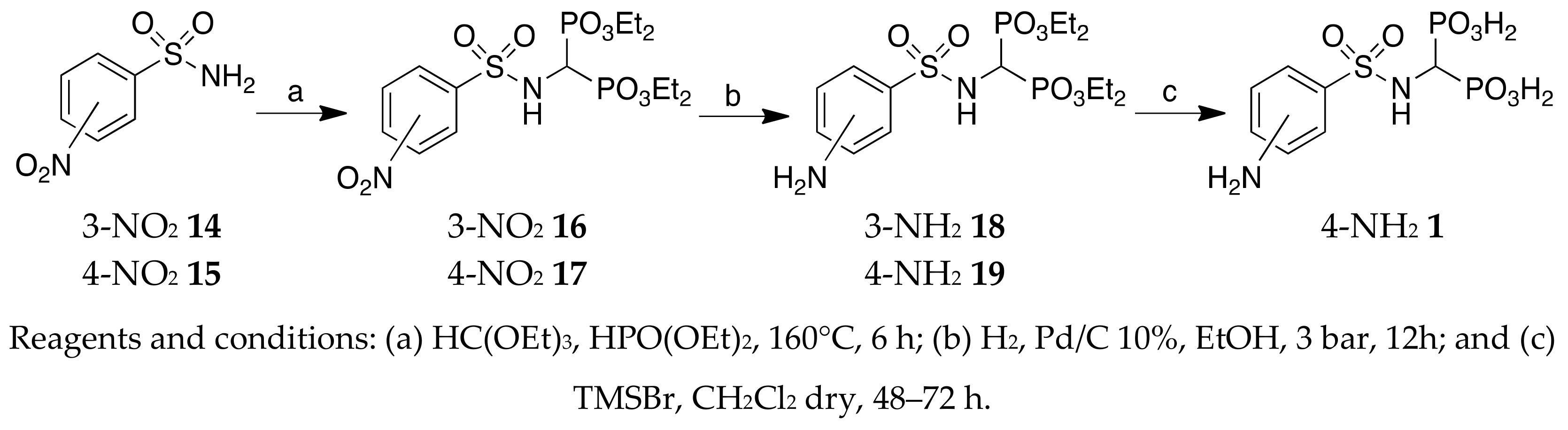
A suspension of the appropriate sulfonyl chloride (4.41 mmol) and NH4OH (110.25 mmol) was stirred for 15 min at 0 °C and at room temperature for 3–24 h. Subsequently, the mixture was partitioned between EtOAc and NaHCO3, and the layers were separated. The organic phase was washed with brine, dried over anhydrous Na2SO4, filtered, and the filtrate was evaporated in vacuo to give a crude product that was used for the next step without any purification.
3-Nitrobenzenesulfonamide 14: Yellow solid, 50% yield. 1H NMR (500 MHz, [D6]DMSO): δ = 7.65 (bs, 2H, NH2), 7.88 (t, J = 7.83, 1H, aromatic), 8.21–8.23 (m, 1H, aromatic), 8.43–8.45(m, 1H, aromatic), 8.57–8.58 (m, 1H, aromatic). GC-MS: m/z (%): 202(97), 186(33), 156(42), 138(59), 122(55), 108(25), 92(57), 75(100).
4-Nitrobenzenesulfonamide 15: Yellow solid, 50% yield; 1H NMR (400 MHz, [D6]DMSO): δ = 8.42–8.40 (m, 2H, aromatics), 8.07–8.05 (m, 2H, aromatics), 7.73 (bs, 2H, NH2). GC-MS: m/z (%): 202(100), 186(44), 156(20), 138(83), 122(60), 108(25), 92(35), 75(95).
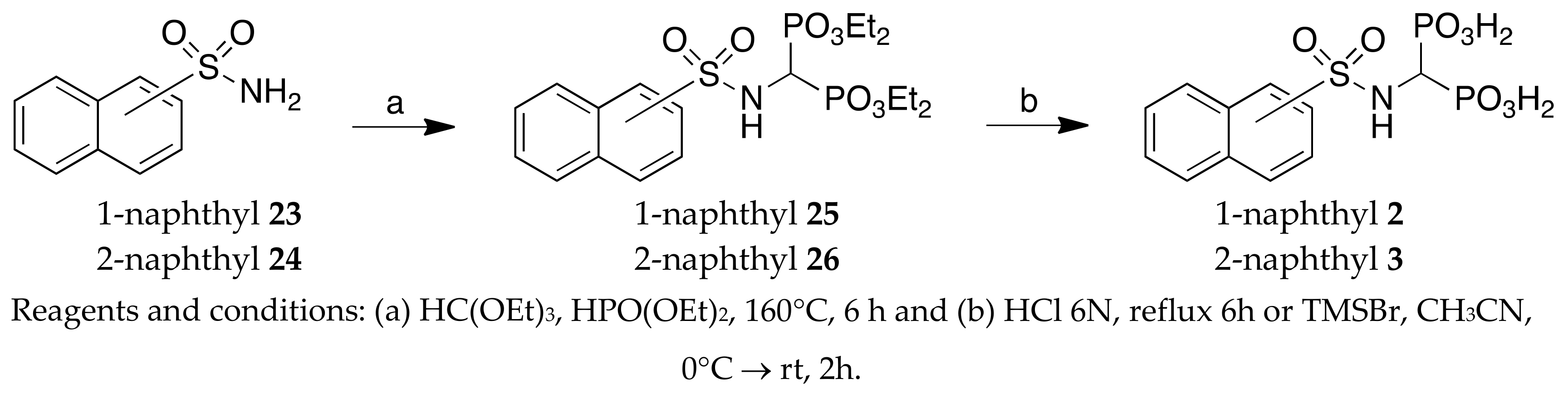
Naphthalene-1-sulfonamide 23: White solid, 93% yield. 1H NMR (300 MHz, [D6]DMSO): δ = 7.59–7.68 (m, 3H, aromatics), 7.63 (bs, 2H, NH2), 8.06–8.16 (m, 3H, aromatics), 8.59–8.61 (m, 1H, aromatic). GC-MS: m/z (%): 207 (80), 143 (68), 127 (100), 115 (33).
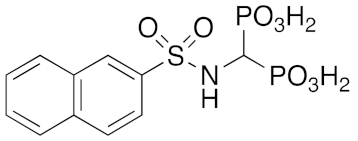
Naphthalene-2-sulfonamide 24: White solid, 98% yield. 1H NMR (300 MHz, [D6]DMSO): δ = 7.49 (bs, 2H, NH2), 7.60–7.70 (m, 2H, aromatics), 7.85–7.87 (m, 1H, aromatic), 7.99–8.03 (m, 1H, aromatic), 8.09–8.14 (m, 2H, aromatics), 8.40–8.41 (m, 1H, aromatic). GC-MS: m/z (%): 207 (35), 143 (40), 127 (100), 115 (20), 77 (10).
3.2.2. General Procedure for the Preparation of Tetraethyl Bisphosphonates 16, 17, 25, and 26
Triethyl orthoformate, diethyl phosphite, and the opportune sulfonamide were mixed in a 1.2:3:1 stoichiometric ratio. The reaction was heated at 160 °C until the evolution of EtOH was complete; then, EtOAc was used to dissolve the residue. Distillation of the solvent afforded a crude yellow oil, which was purified by column chromatography on silica gel (eluent EtOAc). The titled compounds were obtained as white solids in 35–50% yield.
Tetraethyl ((3-nitro-phenylsulfonylamino)methyl)-1,1-bisphosphonate 16: Yellow solid, 35% yield. 1H NMR (500 MHz, CDCl3): δ = 1.26–1.31 (m, 12H, CH3), 4.05–4.20 (m, 8H, CH2), 4.26 (td, JHP = 22.00, JHH = 9.78, 1H, CH), 5.80 (br, 1H, NH), 7.72 (t, J = 7.83, 1H, aromatic), 8.23–8.26 (m, 1H, aromatic), 8.43–8.431 (m, 1H, aromatic), 8.75 (t, J = 1.71, 1H, aromatic). MS(ESI): m/z: 511 [M + Na]+, 489 [M + H]+; MS2 (489): m/z (%): 377 (35), 298 (64), 295 (64), 204 (26), 110 (100).
Tetraethyl ((4-nitro-phenylsulfonylamino)methyl)-1,1-bisphosphonate 17: Yellow solid, 47% yield; mp: 122–125 °C. 1H NMR (500 MHz, CDCl3): δ = 1.23–1.31 (m, 12 H, CH3), 4.11–4.21 (m, 8H, CH2), 4.25 (td, JHP = 22.10, JHH = 9.60, 1H, PCHP), 6.08 (br, 1H, NH), 8.08–8.12 (m, 2H, aromatics), 8.32–8.35 (m, 2H, aromatics); MS (ESI): m/z: 511 (M + Na]+; MS2: m/z (%): 373 (100).
Tetraethyl ((1-naphthylsulfonylamino)methyl)-1,1-bisphosphonate 25: White solid, 35% yield. 1H NMR(300 MHz, CDCl3): δ = 1.07–1.18 (m, 12H, CH3 ), 3.81–4.00 (m, 8H, CH2), 4.15 (td, JHP = 22.00, J = 9.57, 1H, PCHP), 5.58 (br, 1H, NH), 7.54 (t, J = 7.84, 1H, aromatic), 7.58–7.63 (m, 1H, aromatic), 7.67–7.73 (m, 1H, aromatic), 7.92–7.96 (m, 1H, aromatic), 8.07 (d, J = 8.25, 1H, aromatic), 8.25 (dd, J1 = 7.43, J2 = 1.10, 1H, aromatic), 8.56 (d, J = 8.53, 1H, aromatic). MS (ESI): m/z: 516 [M + Na]+; MS2: m/z (%): 488 (39), 378 (100). 492[M − H]−; MS2: m/z (%): 191 (100).
Tetraethyl ((2-naphthylsulfonylamino)methyl)-1,1-bisphosphonate 26: White solid, 50% yield. 1H NMR(300 MHz, CDCl3): δ = 1.12–1.23 (m, 12H, CH3), 3.90–4.12 (m, 8H, CH2), 4.23 (td, JHP = 22.00, JHH = 9.2, 1H, PCHP), 5.29 (br, 1H, NH), 7.57–7.67 (m, 2H, aromatics), 7.85–7.97 (m, 4H, aromatics), 8.43–8.44 (m, 1H, aromatic). MS (ESI): m/z: 516 [M + Na]+; MS2: m/z (%): 488 (28),378 (100). 492[M − H]−; MS2: m/z (%): 191 (100).
3.2.3. General Procedure for Preparation of Amino Derivates 19 and 20
To a solution of the nitro compound (0.532 mmol) in 8.5 mL EtOH, 10% Pd/C (0.20 mmol) was added. The hydrogenation of the mixture was carried out at room temperature at a pressure of 3 bar for 12–24 h. The reaction mixture was filtered through a celite pad, and the resulting solution was evaporated in vacuo, giving an oil. The crude was purified by chromatography on silica gel (eluent: CHCl3/MeOH 9.5:0.5 v/v), affording the desired amino derivate.
Tetraethyl ((3-amino-phenylsulfonylamino))methyl-1,1-bisphosphonate 18: White solid, 75% yield. 1H NMR (300 MHz, CDCl3): δ = 1.11–1.43 (m, 12H, CH3), 3.99–4.07 (m, 1H, PCHP), 4.11–4.19 (m, 8H, CH2), 5.64 (br, 1H, NH), 6.85–6.87 (m, 1H, aromatic), 7.23–7.25 (m, 3H, aromatics). MS (ESI): m/z: 481[M + Na]+. 459[M + H]+; MS2(459): m/z (%): 183(100), 138(62).
Tetraethyl ((4-amino-phenylsulfonylamino)methyl)-1,1-bisphosphonate 19: White solid, 77% yield. 1H NMR (500 MHz, CDCl3): δ = 1.26–1.31 (m, 12H, CH3), 4.04–4.12 (m, 1H, PCHP), 4.13–4.20 (m, 8H, CH2), 5.05 (br, 1H, NH), 6.65 (dl, J = 8.81, 2H, aromatics), 7.65 (dl, J = 8.81, 2H, aromatics). MS (ESI): m/z: 481[M + Na]+, 459[M + H]+; MS2(469): m/z (%): 156(100), 138(28), 108(50).
3.2.4. General Procedure for the Preparation of N-acylate Derivates
The opportune acyl chloride (1.1–2 mmol) and triethylamine (2 mmol) were added to the solution of amino derivate (18 and 19) (1 mmol) in anhydrous THF. Stirring of the mixture occurred at room temperature under inert atmosphere (N2 or Ar) for 3–12 h. After a given time, the mixture was dried under vacuum and dissolved in EtOAc; the resulting solution was washed with NaHCO3 and then with HCl 1N, NH4Cl ss, and brine. The organic phase was finally dried over anhydrous Na2SO4, filtered, and the filtrate was evaporated in vacuo. The residue was purified by chromatography on silica gel (eluent: CHCl3/MeOH 98: 2 v/v or AcOEt /MeOH 9: 1 v/v) or crystallized with AcOEt to give the desired product.
Tetraethyl((3-benzamidophenylsulfonylamino)methyl)-1,1-bisphosphonate 6a:
White solid, 64% yield, (AcOEt). 1H NMR (500MHz, [D6] DMSO): δ = 1.11–1.88 (m, 12H, CH3), 3.87–4.03 (m, 8H, CH2), 4.11 (t, JHP = 23.00, 1H, PCHP), 7.50–7.61 (m, 5H, aromatics), 7.95–7.97 (m, 3H, aromatics), 8.30 (s, 1H, aromatic), 8.80 (br, 1H, NH), 10.50 (s, 1H, NH). MS (ESI): m/z: 561[M − H]−; MS2: m/z (%): 288(54), 260(37), 134 (100), 106 (34). 563 [M + H]+.
Tetraethyl((4-benzamidophenylsulfonylamino)methyl)-1,1-bisphosphonate 7a:
White solid, 65% yield (chromatography, eluent: CHCl3/MeOH 98: 2 v/v). 1H NMR (500 MHz, [D6] DMSO): δ = 1.13–1.17 (m, 12H, CH3), 3.89–4.05 (m, 8H, CH2), 4.14 (t, JHP = 23.00, 1H, PCHP), 7.52–7.62 (m, 3H, aromatics), 7.78 (d, J = 8.81, 2H, aromatics), 7.90–7.96 (m, 4H, aromatics), 8.63 (br, 1H, NH), 10.53 (s, 1H, NH). MS (ESI): m/z: 585[M + Na]+, 563[M + H]+; MS2(563): m/z (%): 351(22), 282(10), 261(20), 260(100), 196(50), 166(63), 138(74), 110(42).
Tetraethyl((4-(4-methylbenzamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 8a:
White solid, 79% yield (AcOEt). 1H NMR(500 MHz, CDCl3): δ = 1.25–1.34 (m, 12H, CH3), 2.43 (s, 3H, CH3), 4.06–4.20 (m, 8H, CH2), 4.23 (td, JHP = 22.00, J = 9.79, 1H, PCHP), 5.31 (br, 1H, NH), 7.30 (dl, J = 7.83, 2H, aromatics), 7.78–7.88 (m, 6H, aromatics), 8.15 (bs, 1H, NH). MS (ESI): m/z: 599 [M + Na]+, 577[M + H]+; MS2(577): m/z (%): 393(18), 365(23), 291(21), 275(26), 274(100), 166(87), 138(74), 138(58), 119 (96), 110 (36). 575[M − H]−; MS2: m/z (%): 437(25), 302(16), 210(32), 134(100).
Tetraethyl((4-(4-bromobenzamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 9a:
White solid, 55% yield (AcOEt). 1H NMR(500 MHz, CDCl3): δ = 1.14–1.18 (m, 12H, CH3), 3.94- 4.04 (m, 8H, CH2), 4.14 (t, JHP = 23.00, 1H, PCHP), 7.74 (d, J = 8.32, 2H, aromatics), 7.79 (d, J = 8.81, 2H, aromatics), 7.80–7.92 (m, 4H, aromatics), 8.55 (br, 1H, NH), 10.54 (s, 1H, NH). MS (ESI): m/z: 643[M + 2 + H]+, 641[M + H]+; MS2(641): m/z (%): 458(18), 431(18), 340(59), 338(53), 276 (11), 274(15), 185(24), 183(32), 166(100), 138(82), 110(39).
Tetraethyl((4-(3-bromobenzamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 10a:
White solid, 73% yield (AcOEt). 1H NMR(500 MHz, [D6] DMSO): δ = 1.13–1.96 (m, 12H, CH3), 3.97–4.05 (m, 8H, CH2), 4.14 (t, JHP = 23.00, 1H, PCHP), 7.51 (t, J = 8.08, 1H, aromatic), 7.80–7.82 (m, 3H, aromatics), 7.89–7.96 (m, 3H, aromatics), 8.14 (t, J = 1.96, 1H, aromatic), 8.63 (br, 1H, NH), 10.62 (s, 1H, NH). MS (ESI): m/z: 643[M+2+H]+, 641 [M + H]+; MS2: m/z (%): 459(12), 430(12), 340(70), 338(43), 276(20), 274(18), 185(18), 183 (42), 166(88), 138(100).
Tetraethyl((4-(4-nitrobenzamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 11a:
White solid, 57% yield (AcOEt). 1H NMR(500 MHz, [D6] DMSO): δ = 1.15–1.17 (m, 12H, CH3), 3.90–4.05 (m, 8H, CH2), 4.14 (t, JHP = 23.00, 1H, PCHP), 7.81 (d, J = 8.56, 2H, aromatics), 7.92 (d, J = 8.56, 2H, aromatics), 8.17–8.20 (m, 2H, aromatics), 8.36–8.39 (m, 2H, aromatics), 8.66 (br, 1H, NH), 10.85 (s, 1H, NH). MS (ESI): m/z: 630[M + Na]+, 608 [M + H]+; MS2(630): m/z (%): 492(100). 606 [M − H]−; MS2: m/z (%): 468(86), 333(42), 305(45), 240 (100), 134(61).
Tetraethyl((4-(1-naphthamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 12a:
White solid, 59% yield (AcOEt). 1H NMR(500 MHz, CDCl3): δ = 1.16–1.43 (m, 12H, CH3), 4.04–4.15 (m, 8H, CH2), 4.23 (t, JHP = 22.02, JHH = 9.79, 1H, PCHP), 5.29 (br, 1H, NH), 7.52 (t, J = 7.59, 1H, aromatic), 7.55–7.62 (m, 2H, aromatics), 7.74 (d, J = 6.85, 1H, aromatics), 7.81–7.88 (m, 2H, aromatics), 7.88–7.97 (m, 3H, aromatics), 7.99 (d, J = 8.32, 1H, aromatics), 8.10 (s, 1H, NH), 8.30 (d, J = 7.83, 1H, aromatic). MS (ESI): m/z: 635[M + Na]+, 613 [M + H]+; MS2(613): m/z (%): 310(47), 166(55), 155(100), 138(45), 110(22). 611[M − H]−; MS2: m/z (%): 473(45), 388(67), 310(42), 246 (33), 245(39), 134 (100), 106(29).
Tetraethyl((4-(2-naphthamido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 13a:
White solid, 49% yield (chromatography, eluent: AcOEt/MeOH 9:1 v/v). 1H NMR (500 MHz, [D6] DMSO): δ = 1.04–1.29 (m, 12H, CH3), 3.89–4.09 (m, 8H, CH2), 4.16 (t, JHP = 22.75, 1H, PCHP), 7.60–7.67 (m, 2H, aromatics), 7.82 (d, J = 8.81, 2H, aromatics), 7.96–8.10 (m, 6H, aromatics), 8.60 (s, 1H, aromatic), 8.63 (br, 1H, NH), 10.72 (s, 1H, NH). MS (ESI): m/z: 635[M + Na]+, 613 [M + H]+; MS2(613): m/z (%): 310(76), 166(61), 134(54), 110(18). 611[M − H]−; MS2: m/z (%): 473(43), 388(70), 310(30), 246 (25), 245(33), 134 (100), 106(29).
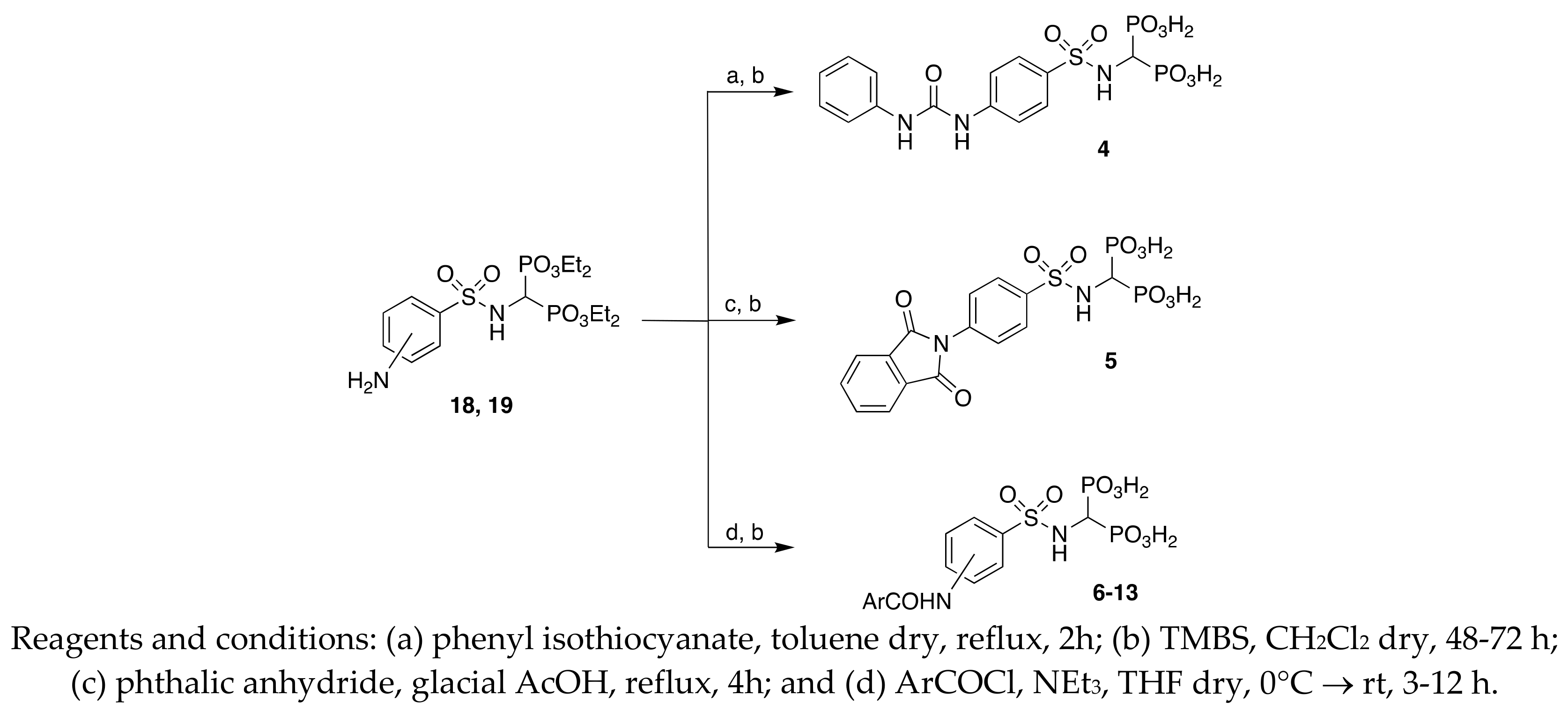
Tetraethyl((4-(3 phenylureido)phenylsulfonylamino)methyl)-1,1-bisphosphonate 4a:
A solution of phenyl isothiocyanate (1.2 mmol) in anhydrous toluene (2 mL) was added to the suspension of 19 (1 mmol) in anhydrous toluene (2 mL), and the mixture was heated to reflux for 2 h. The eluent was evaporated in vacuo, and the residue was purified by chromatography on silica gel (eluent AcOEt/MeOH 9: 1 v/v) to obtain the desired product.
White solid, 71% yield. 1H NMR (500 MHz, CDCl3): δ = 1.17–1.81 (m, 12H, CH3), 4.05–4.16 (m, 8H, CH2), 4.31 (td, JHP = 22.26, JHH = 9.30, 1H, PCHP), 5.75 (br, 1H, NH), 7.03 (t, J = 7.34, 1H, aromatics), 7.26–7.28 (m, 2H, aromatics), 7.42 (d, J = 7.34, 2H, aromatics), 7.58 (d, J = 6.85, 2H, aromatics), 7.72 (d, J = 7.34, 2H, aromatics), 8.09 (bs, 1H, NH), 8.39 (bs, 1H, NH). MS(ESI): m/z: 600[M + Na]+, 578[M + H]+; MS2(578): m/z (%): 366(13), 293(22), 275 (100), 211(36), 166 (84), 156(24), 138 (49), 108(40).
Tetraethyl((4-(1,3-dioxoisoindolin-2-yl)phenylsulfonylamino)methyl)-1,1-bisphosphonate 5a:
A mixture of 19 (1 mmol) and phthalic anhydride (1.07 mmol) in 6 ml of glacial acetic acid was refluxed for 4 h. The resulting solution was dried under vacuum. EtOAc was subsequently used to dilute the residue; then, 6-M NaOH was added until pH = 6. After separation of the layers, the organic phase was washed with brine, dried over anhydrous Na2SO4, filtered, and the filtrate was dried in vacuo. Column chromatography on silica gel (eluent: CHCl3/MeOH 9: 1 v/v) was used to purify the residue, affording the desired product.
White solid, 47% yield. 1H NMR (500 MHz, [D6]DMSO): δ = 1.15–1.17 (m, 12H, CH3), 3.87–4.05 (m, 8H, CH2), 4.16 (t, J = 23.00, 1H, PCHP), 7.61(d, J = 8.32, 2H, aromatics), 7.90–7.98 (m, 6H, aromatics), 8.89 (br, 1H, NH). MS (ESI): m/z: 611[M + Na]+; MS2: m/z (%): 473 (100), 445(11), 324(10), 270(23), 160 (12). 587[M − H]−; MS2: m/z %: 287(12), 286(60), 222 (31), 137(100), 108(30).
3.2.5. General Procedure for the Preparation of 1,1-bisphosphonic Acids
Method A: A solution of the appropriate tetraethyl bisphosphonate (1 mmol) in 4-mL 2N HCl solution was refluxed for 12–24 h. The aqueous phase was removed under reduced pressure, and the crude bisphosphonic acids were then triturated with the opportune solvent and filtered to afford the final compounds as white solids in 20–96% yield.
Method B: Anhydrous trimethylsilylbromide (17–32 mmol) was carefully added to a solution of the corresponding tetraethyl bisphosphonate (1 mmol) in anhydrous acetonitrile (6 mL) at 0 °C under argon, and the resulting mixture was stirred at room temperature for 24–48 h. Two milliliters of MeOH were added, and the mixture was stirred for 5 min; the solvent was distilled off, and the crude bisphosphonic acids were triturated with the opportune solvent and filtered to afford the desired compounds as white solids in a 33–99% yield.
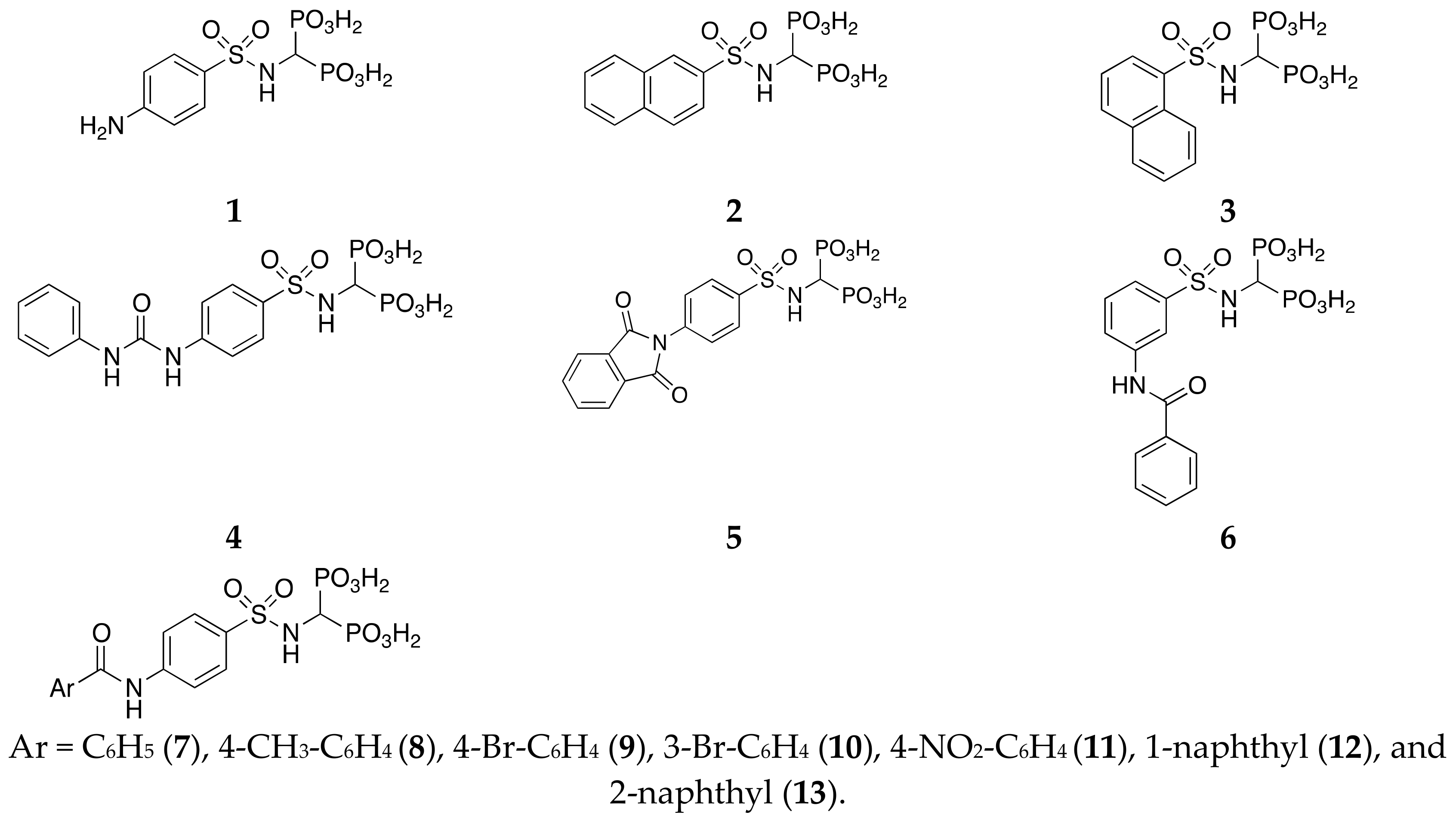
(4-aminophenylsulfonylamino)methyl-1,1-bisphosphonic acid (1). Method B:
White solid, mp: 243°C (dec)(Acetone/ H2O). 1H NMR (500 MHz, [D6]DMSO): δ = 3.78 (td, JHP= 21.53, J = 9.30, 1H, PCHP), 5.38–6.42 (br, 6H, OH, NH2), 6.50 (d, J = 8.32, 2H, aromatics), 6.81(d, J = 9.30, 1H, NH), 7.45 (d, J = 8.32, 2H, aromatics). 31P NMR (500 MHz, [D6] DMSO): δ = 15.03 (d, JPH = 22.84). MS (ESI): m/z: 345 [M − H]−; MS2: m/z (%): 280 (100). HRMS [M − H]−: calculated 344.9717; found 344.9709.
(2-naphthylsulfonylamino)methyl-1,1-bisphosphonic acid (2). Method B:
White solid, 43% yield. (IPA) mp: 107–110 °C; 1H NMR (300 MHz, [D6]DMSO): δ = 3.88 (td, JHP= 21.86, JHH = 9.63, 1H, PCHP), 6.2–7.00 (br, 4H, OH), 7.60–7.66 (m, 2H, aromatics), 7.90–8.07 (m, 5H, 4H aromatics, NH), 8.40 (s, 1H, aromatic). 31P NMR (300 MHz, [D6] DMSO): δ = 14.96 (d, JPH = 21.37). MS (ESI): m/z: 380 [M − H]; MS2: m/z (%): 362 (100), 298(19).
(1-naphthylsulfonylamino)methyl-1,1-bisphosphonic acid (3). Method A:
White solid, 20% yield; mp: 183 °C (dec) (MeOH/Et2O 1:1 v/v). 1H NMR (300MHz, [D6]DMSO): δ = 3.71 (t, JHP= 19.52, 1H, PCHP), 6.10–6.50 and 6.99–7.38 (br, 5H, 4 OH and NH), 7.51 (t, J = 7.70, 1H, aromatic), 7.55–7.65 (m, 2H, aromatics), 7.98–8.02 (m, 1H, aromatic), 8.08 (d, J = 8.25, 1H, aromatic), 8.36 (d, J = 6.88, 1H, aromatic), 8.64 (d, J = 7.98, 1H, aromatic). 31P NMR (300 MHz [D6] DMSO): δ = 14.14 (d, JPH = 18.34). MS (ESI): m/z: 380 [M − H]−; MS2: m/z (%): 362 (100), 298 (21).
(4-(3-phenylureido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (4). Method B:
White solid, 33% yield; mp: > 243 °C (dec). 1H NMR (500 MHz, [D6]DMSO): δ = 3.82 (td, JHP = 22.00, J = 9.30, 1H, PCHP), 5.65–6.80 (br, 5H, OH and NH), 6.97 (t, J = 7.34, 1H, aromatic), 7.27 (t, J = 7.34, 2H, aromatics), 7.42–7.54 (m, 4H, aromatics), 7.72 (d, J = 7.82, 2H, aromatics), 8.76 (s, 1H, NH), 9.01(s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 15.04 (d, JPH = 21.25). MS (ESI): m/z: 464[M − H]−; MS2: m/z (%): 382(56), 275(27), 263(95), 106(71), 79(100). HRMS [M − H]−: calculated 464.0088; found 464.0081.
(4-(1,3-dioxoisoindolin-2-yl)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (5). Method B:
White solid, 47% yield; mp: > 250 °C (MeOH). 1H NMR (500 MHz, [D6] DMSO): δ = 3.87–3.97 (td, JHP = 21.53, J = 9.46, 1H, PCHP), 4.25–5.40 (br, 5H, OH and NH), 7.56 (d, J = 8.30, 2H, aromatics), 7.89–7.93 (m, 3H, aromatics), 7.96–8.00 (m, 3H, aromatics). 31P NMR (500 MHz, [D6]DMSO): δ = 14.90 (d, JPH = 21.36). MS (ESI): m/z: 475[M − H]−; MS2: m/z (%): 393(100), 286(65), 106(100). HRMS [M − H]−: calculated 474.9772; found 474.9768.
(4-benzamidophenylsulfonylamino)methyl-1,1-bisphosphonic acid (6). Method B:
White solid, 99% yield; mp: 243–245 °C (Acetone). 1H NMR (500 MHz, [D6]DMSO): δ = 3.83 (td, 1H, JHP = 21.53, J = 9.54, 1H, PCHP), 4.0–5.5 (br, 4H, OH), 7.53 (t, J = 7.34, 2H, aromatics), 7.58–7.60 (m, 2H, 1H aromatic, NH), 7.79 (d, J = 8.81, 2H, aromatics), 7.87 (d, J = 8.87, 2H, aromatics), 7.94 (d, J = 7.34, 2H, aromatics), 10.54 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 14.82 (s). MS (ESI): m/z: 449[M − H]−; MS2: m/z (%): 368(20), 367(100), 260(45), 106(76). HRMS [M − H]−: calculated 448.9979; found 448.9972.
(3-benzamidophenylsulfonylamino)methyl-1,1-bisphosphonic acid (7). Method B:
White solid, 53% yield; mp: > 250 °C (IPA). 1H NMR (300 MHz, [D6]DMSO): δ = 3.75–3.97 (m, 1H, PCHP), 5.65–6.57 (br, 4H, OH), 7.41–7.65 (m, 5H, aromatics), 7.67–7.80 (br, 1H, NH), 7.95–7.98 (m, 3H, aromatics), 8.25 (s, 1H, aromatic), 10.43 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 14.87 (d, JPH = 19.82). MS (ESI): m/z: 449 [M − H]−; MS2: m/z (%): 367(94), 260(34), 106(100). HRMS [M − H]−: calculated 448.9979; found 448.9973.
(4-(4-methylbenzamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (8). Method B:
White solid, 61% yield; mp: > 250 °C (MeOH). 1H NMR (500 MHz, [D6]DMSO): δ = 2.38 (s, 3H, CH3), 3.84 (td, JHP = 21.75, J = 8.90, 1H, PCHP), 5.18–6.05 (br, 5H, OH, NH), 7.34 (d, J = 7.83, 2H, aromatics), 7.58 (d, J = 8.32, 1H, aromatic), 7.78–7.87 (m, 5H, aromatics), 10.40 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 14.02 (d, JPH = 21.25). MS (ESI): m/z: 463 [M − H]−; MS2: m/z (%): 382(14), 381(62), 274(34), 106(60), 79(100). HRMS [M − H]−: calculated 463.0135; found 463.0130.
(4-(4-bromobenzamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (9) Method B:
White solid, 80% yield; mp: > 250 °C (MeOH). 1H NMR (300 MHz, [D6]DMSO): δ = 3.74–3.92 (td, JHP = 21.67, J = 9.66, 1H, PCHP), 7.46–7.92 (m, 12H, 4OH, 8 H aromatics), 7.67 (d, J = 9.66, 1H, NH), 10.56 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ =14.95 (d, JPH = 21.36). MS(ESI): m/z: 529[M + 2 − H]−, 527[M − H]−. MS2: m/z (%): 447(100), 445(69), 340(24), 338(19). HRMS [M − H]−: calculated 526.9084; found 526.9077.
(4-(3-bromobenzamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (10) Method B:
White solid, 68% yield; mp: >246 °C (IPA). 1H NMR (500 MHz, [D6]DMSO): δ = 3.83 (td, 1H, JHP = 21.76, J= 9.29, 1H, PCHP), 6.20–7.07 (br, 4H, OH), 7.50 (t, J = 7.83, 1H, aromatic), 7.63 (d, J = 9.29, 1H, NH), 7.80–7.86 (m, 5H, aromatics), 7.94 (d, J = 7.83, 1H, aromatic), 8.12–8.13 (m, 1H, aromatic), 10.58 (s, 1H, NH). 31P NMR (500 MHz [D6]DMSO): δ = 14.92 (d, JPH = 21.24). MS (ESI): m/z: 529[M + 2 − H]−, 527[M − H]−; MS2: m/z (%): 447(98), 445 (69), 340(23), 338(20), 106 (72), 79 (100). HRMS [M − H]−: calculated 526.9084; found 526.9076.
(4-(4-nitrobenzamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (11) Method B:
White solid, 71% yield; mp: > 250 °C (MeOH). 1H NMR (500 MHz, [D6]DMSO): δ = 3.79–3.90 (td, JHP = 22.00, J = 9.45, 1H, PCHP), 6.00–7.20 (br, 4H, OH), 7.69 (d, J = 9.45, 1H, NH), 7.81–7.88 (m, 4H, aromatics), 8.18 (d, J = 8.81, 2H, aromatics), 8.37 (d, J = 8.81, 2H, aromatics), 10.80 (s, 1H, NH). 31P NMR (500 MHz [D6]DMSO): δ = 14.96 (d, JPH = 21.36). MS (ESI): m/z: 494[M − H]−MS2: m/z (%): 458 (8), 412(100), 305 (56), 106 (76). HRMS [M − H]−: calculated 493.9830; found 493.9824.
(4-(1-naphthamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (12) Method B:
White solid, 44% yield; mp: 237–240 °C (IPA). 1H NMR (500 MHz, [D6]DMSO): δ = 3.86 (t, JHP = 21.52, J = 9.27, 1H, PCHP), 4.25–6.22 (br, 4H, OH), 7.57–7.62 (m, 4H, 3H aromatics, NH), 7.76 (d, J = 6.85, 1H, aromatic), 7.82 (d, J = 8.81, 2H, aromatics), 7.88 (d, J = 8.81, 2H, aromatics), 8.00–8.03 (m, 1H, aromatic), 8.09 (d, J = 8.39, 1H, aromatic), 8.13–8.17 (m, 1H, aromatic), 10.85 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 13.91 (d, JPH = 21.45). MS (ESI): m/z: 499 [M − H]−; MS2: m/z (%): 417 (94), 310(39), 106(66), 79(100). HRMS [M − H]−: calculated 499.0135; found 499.0130.
(4-(2-naphthamido)phenylsulfonylamino)methyl-1,1-bisphosphonic acid (13) Method B:
White solid, 68% yield; mp: 248–250 °C (IPA). 1H NMR (500 MHz, [D6]DMSO): δ = 3.85 (td, JHP = 22.01, J = 9.78, 1H, PCHP), 5.15–5.95 (br, 4H, OH), 7.53–7.58 (br, 1H), 7.60–7.66 (m, 2H, aromatics), 7.83 (d, J = 9.30, 2H, aromatics), 7.92 (d, J = 8.81, 2H, aromatics), 7.99–8.09 (m, 4H, aromatics), 8.58 (s, 1H), 10.67 (s, 1H, NH). 31P NMR (500 MHz, [D6]DMSO): δ = 13.97 (d, JPH = 21.25). MS (ESI): m/z: 499 [M − H]−; MS2: m/z (%): 417 (90), 310 (37), 106 (66), 79 (100). HRMS [M − H]−: calculated 499.0135; found 499.0127.