An Overview of Recent Strategies in Pathogen Sensing
Abstract
:1. Introduction
2. Recent Sensing Strategies for Pathogen Detection Based on Microfluidics
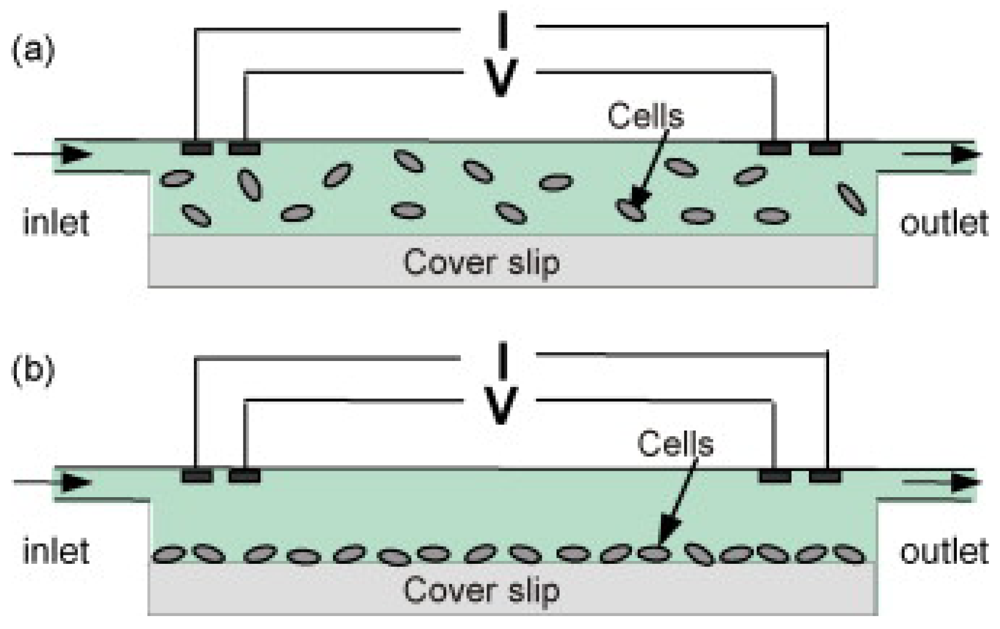
2.1. Label-free bacterial sensor based on electrical and electrochemical detection
Impedance based detection
Electrochemical detection
Interdigitated array electrodes
2.2. Nucleic acid-based detection
PCR-based pathogen sensor
Nucleic acid sensor without PCR
2.3. Focusing strategies in microfluidic devices
3. Recent Sensing Strategies for Pathogen Detection Based on Nanomaterials
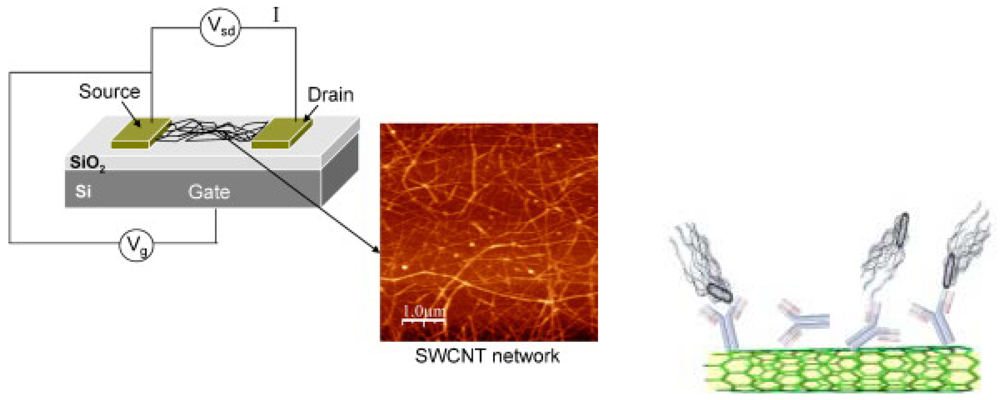
3.1. Nanofabricated electrical sensors: nanowell, nanotube and nanowire
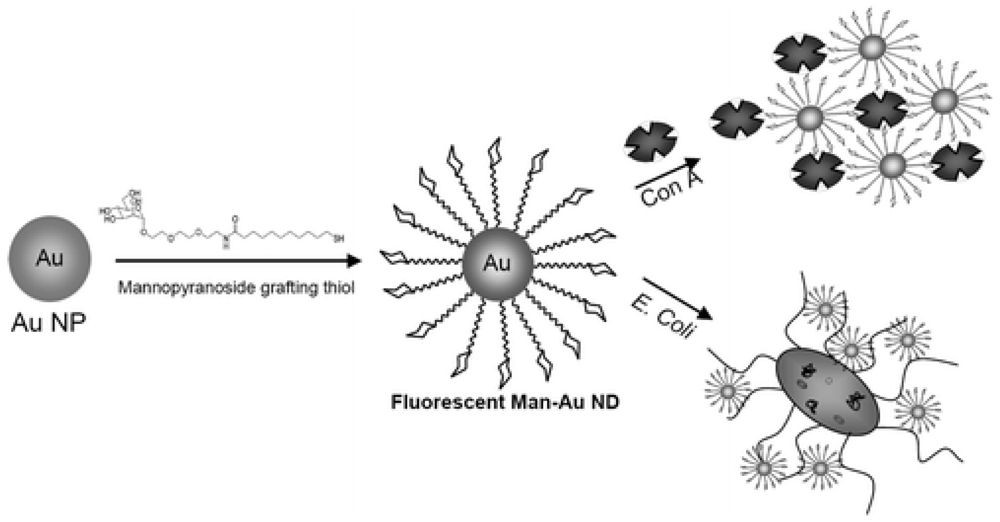
3.2. Nanoparticles
Nanoparticles as biomarkers
Nanoparticles as a signal amplifier
4. Conclusions and Prospects
Acknowledgments
References and Notes
- Deisingh, A.K.; Thompson, M. Detection of infectious and toxigenic bacteria. Analyst 2002, 127, 567–581. [Google Scholar]
- Ivnitski, D.; Abdel-Hamid, I.; Atanasov, P.; Wilkins, E. Biosensors for detection of pathogenic bacteria. Biosens. Bioelectron 1999, 14, 599–624. [Google Scholar]
- Nugen, S.R.; Baeumner, A.J. Trends and opportunities in food pathogen detection. Anal. Bioanal. Chem 2008, 391, 451–454. [Google Scholar]
- Batt, C.A. Food pathogen detection. Science 2007, 316, 1579–1580. [Google Scholar]
- Lazcka, O.; Del Campo, F.J.; Munoz, F.X. Pathogen detection: a perspective of traditional methods and biosensors. Biosens. Bioelectron 2007, 22, 1205–1217. [Google Scholar]
- Yang, L.; Bashir, R. Electrical/electrochemical impedance for rapid detection of foodborne pathogenic bacteria. Biotechnol. Adv. 2008, 26, 135–150. [Google Scholar]
- Skottrup, P.D.; Nicolaisen, M.; Justesen, A.F. Towards on-site pathogen detection using antibody-based sensors. Biosens. Bioelectron. 2008, 24, 339–348. [Google Scholar]
- Fritz, J. Cantilever biosensors. Analyst 2008, 133, 855–863. [Google Scholar]
- Mujika, M.; Arana, S.; Castano, E.; Tijero, M.; Vilares, R.; Ruano-Lopez, J.M.; Cruz, A.; Sainz, L.; Berganza, J. Magnetoresistive immunosensor for the detection of Escherichia coli O157:H7 including a microfluidic network. Biosens. Bioelectron. 2009, 24, 1253–1258. [Google Scholar]
- Fritzsche, W. DNA-gold conjugates for the detection of specific molecular interactions. Rev. Mol. Biotechnol 2001, 82, 37–46. [Google Scholar]
- de Lumley-Woodyear, T.; Campbell, C.N.; Heller, A. Direct enzyme-amplified electrical recognition of a 30-base model oligonucleotide. J. Am. Chem. Soc. 1996, 118, 5504–5505. [Google Scholar]
- Millan, K.M.; Mikkelsen, S.R. Sequence-selective biosensor for DNA based on electroactive hybridization indicators. Anal. Chem. 1993, 65, 2317–2323. [Google Scholar]
- Millan, K.M.; Saraullo, A.; Mikkelsen, S R. Voltammetric DNA biosensor for cystic fibrosis based on a modified carbon paste electrode. Anal. Chem. 1994, 66, 2943–2948. [Google Scholar]
- Xu, C.; Cai, H.; He, P.; Fang, Y. Electrochemical detection of sequence-specific DNA using a DNA probe labeled with aminoferrocene and chitosan modified electrode immobilized with ssDNA. Analyst 2001, 126, 62–65. [Google Scholar]
- Takenaka, S.; Yamashita, K.; Takagi, M.; Uto, Y.; Kondo, H. DNA sensing on a DNA probe-modified electrode using ferrocenylnaphthalene diimide as the electrochemically active ligand. Anal. Chem. 2000, 72, 1334–1341. [Google Scholar]
- Boehm, D.A.; Gottlieb, P.A.; Hua, S.Z. On-chip microfluidic biosensor for bacterial detection and identification. Sens. Actuators B 2007, B126, 508–514. [Google Scholar]
- Hua, S.Z.; Thomas, P. A microfluidic chip for real-time studies of the volume of single cells. Lab. Chip 2009, 9, 251–256. [Google Scholar]
- Javanmard, M.; Talasaz, A.H.; Nemat-Gorgani, M.; Pease, F.; Ronaghi, M.; Davis, R.W. Direct electrical detection of target cells on a microfluidic biochip. Proc. SPIE 2008, 6886, 68860B/68861–68860B/68810. [Google Scholar]
- Richter, L.; Stepper, C.; Mak, A.; Reinthaler, A.; Heer, R.; Kast, M.; Brueckl, H.; Ertl, P. Development of a microfluidic biochip for online monitoring of fungal biofilm dynamics. Lab. Chip 2007, 7, 1723–1731. [Google Scholar]
- Carbonaro, A.; Mohanty, S.K.; Huang, H.; Godley, L.A.; Sohn, L.L. Cell characterization using a protein-functionalized pore. Lab. Chip 2008, 8, 1478–1485. [Google Scholar]
- Cheng, X.; Liu, Y.-S.; Irimia, D.; Demirci, U.; Yang, L.; Zamir, L.; Rodriguez, W.R.; Toner, M.; Bashir, R. Cell detection and counting through cell lysate impedance spectroscopy in microfluidic devices. Lab. Chip 2007, 7, 746–755. [Google Scholar]
- Mantzila, A.G.; Maipa, V.; Prodromidis, M.I. Development of a Faradic impedimetric immunosensor for the detection of Salmonella typhimurium in milk. Anal. Chem. 2008, 80, 1169–1175. [Google Scholar]
- Tully, E.; Higson, S.P.; O'Kennedy, R. The development of a “labeless” immunosensor for the detection of Listeria monocytogenes cell surface protein, Internalin B. Biosens. Bioelectron. 2008, 23, 906–912. [Google Scholar]
- Bayoudh, S.; Othmane, A.; Ponsonnet, L.; Ben Ouada, H. Electrical detection and characterization of bacterial adhesion using electrochemical impedance spectroscopy-based flow chamber. Colloids Surf. A 2008, 318, 291–300. [Google Scholar]
- Liao, J.C.; Mastali, M.; Gau, V.; Suchard, M.A.; Moller, A.K.; Bruckner, D.A.; Babbitt, J.T.; Li, Y.; Gornbein, J.; Landaw, E.M.; McCabe, E.R.B.; Churchill, B.M.; Haake, D.A. Use of electrochemical DNA biosensor for rapid molecular identification of uropathogens in clinical urine specimens. J. Clin. Microbiol 2006, 44, 561–570. [Google Scholar]
- Lazcka, O.; Baldrich, E.; Munoz, F.X.; del Campo, F.J. Detection of Escherichia coli and Salmonella typhimurium using interdigitated microelectrode capacitive immunosensors: the importance of transducer geometry. Anal. Chem. 2008, 80, 7239–7247. [Google Scholar]
- Radke, S.M.; Alocilja, E.C. A high density microelectrode array biosensor for detection of E. coli O157:H7. Biosens. Bioelectron. 2005, 20, 1662–1667. [Google Scholar]
- Yang, L.; Li, Y.; Griffis, C.L.; Johnson, M.G. Interdigitated microelectrode (IME) impedance sensor for the detection of viable Salmonella typhimurium. Biosens. Bioelectron. 2004, 19, 1139–1147. [Google Scholar]
- Varshney, M.; Li, Y.; Srinivasan, V.; Tung, S. A label-free, microfluidics and interdigitated array microelectrode-based impedance biosensor in combination with nanoparticles immunoseparation for detection of Escherichia coli O157:H7 in food samples. Sens. Actuat. A 2007, 128, 99–107. [Google Scholar]
- Lu, Y.-C.; Chuang, Y.-S.; Chen, Y.-Y.; Shu, A.-C.; Hsu, H.-Y.; Chang, H.-Y.; Yew, T.-R. Bacterial detection utilizing electrical conductivity. Biosens. Bioelectron. 2008, 23, 1856–1861. [Google Scholar]
- Yang, L.; Li, Y.; Erf, G.F. Interdigitated array microelectrode-based electrochemical impedance immunosensor for detection of Escherichia coli O157:H7. Anal. Chem. 2004, 76, 1107–1113. [Google Scholar]
- Chen, L.; Manz, A.; Day, P.J.R. Total nucleic acid analysis integrated on microfluidic devices. Lab. Chip 2007, 7, 1413–1423. [Google Scholar]
- Zhu, L.; Zhang, Q.; Feng, H.; Ang, S.; Chau, F.S.; Liu, W.T. Filter-based microfluidic device for immunofluorescent assay of microbial cells. Lab. Chip 2004, 4, 337–341. [Google Scholar]
- Beyor, N.; Seo, T.S.; Liu, P.; Mathies, R.A. Immunomagnetic bead-based cell concentration microdevice for dilute pathogen detection. Biomed. Microdev. 2008, 10, 909–917. [Google Scholar]
- Iliescu, C.; Xu, G.L.; Samper, V.; Tay, F.E.H. Fabrication of a dielectrophoretic chip with 3D silicon electrodes. J. Micromech. Microeng. 2005, 15, 494–500. [Google Scholar]
- Xu, C.X.; Wang, Y.; Cao, M.; Lu, Z.H. Dielectrophoresis of human red cells in microchips. Electrophoresis 1999, 20, 1829–1831. [Google Scholar]
- Bhattacharya, S.; Salamat, S.; Morisette, D.; Banada, P.; Akin, D.; Liu, Y.-S.; Bhunia, A.K.; Ladisch, M.; Bashir, R. PCR-based detection in a micro-fabricated platform. Lab. Chip 2008, 8, 1130–1136. [Google Scholar]
- Yeung, S.-W.; Lee, T.M.-H.; Cai, H.; Hsing, I.-M. A DNA biochip for on-the-spot multiplexed pathogen identification. Nucleic Acids Res. 2006, 34, e118. [Google Scholar]
- Lee, J.-G.; Cheong, K.H.; Huh, N.; Kim, S.; Choi, J.-W.; Ko, C. Microchip-based one step DNA extraction and real-time PCR in one chamber for rapid pathogen identification. Lab. Chip 2006, 6, 886–895. [Google Scholar]
- Cheong, K.H.; Yi, D.K.; Lee, J.-G.; Park, J.-M.; Kim, M.J.; Edel, J.B.; Ko, C. Gold nanoparticles for one step for DNA extraction and real-time PCR of pathogens in a single chamber. Lab. Chip 2008, 8, 810–813. [Google Scholar]
- Di Carlo, D.; Jeong, K.H.; Lee, L.P. Reagentless mechanical cell lysis by nanoscale barbs in microchannels for sample preparation. Lab. Chip 2003, 3, 287–291. [Google Scholar]
- El-Ali, J.; Gaudet, S.; Guenther, A.; Sorger, P.K.; Jensen, K.F. Cell stimulus and lysis in a microfluidic device with segmented gas-liquid flow. Anal. Chem. 2005, 77, 3629–3636. [Google Scholar]
- Li, P.C.H.; Harrison, D.J. Transport, manipulation, and reaction of biological cells on-chip using electrokinetic effects. Anal. Chem. 1997, 69, 1564–1568. [Google Scholar]
- Wolfe, K.A.; Breadmore, M.C.; Ferrance, J.P.; Power, M.E.; Conroy, J.F.; Norris, P.M.; Landers, J.P. Toward a microchip-based solid-phase extraction method for isolation of nucleic acids. Electrophoresis 2002, 23, 727–733. [Google Scholar]
- Cady, N.C.; Stelick, S.; Batt, C.A. Nucleic acid purification using microfabricated silicon structures. Biosens. Bioelectron. 2003, 19, 59–66. [Google Scholar]
- Easley, C.J.; Karlinsey, J.M.; Bienvenue, J.M.; Legendre, L.A.; Roper, M.G.; Feldman, S.H.; Hughes, M.A.; Hewlett, E.L.; Merkel, T.J.; Ferrance, J.P.; Landers, J.P. A fully integrated microfluidic genetic analysis system with sample-in-answer-out capability. Proc. Natl. Acad. Sci. USA 2006, 103, 19272–19277. [Google Scholar]
- Easley, C.J.; Legendre, L.A.; Roper, M.G.; Wavering, T.A.; Ferrance, J.P.; Landers, J.P. Extrinsic Fabry-Perot interferometry for noncontact temperature control of nanoliter-volume enzymatic reactions in glass microchips. Anal. Chem. 2005, 77, 1038–1045. [Google Scholar]
- Waters, L.C.; Jacobson, S.C.; Kroutchinina, N.; Khandurina, J.; Foote, R.S.; Ramsey, J.M. Microchip device for cell lysis, multiplex PCR amplification, and electrophoretic sizing. Anal. Chem. 1998, 70, 158–162. [Google Scholar]
- Koh, C.G.; Tan, W.; Zhao, M.-Q.; Ricco, A.J.; Fan, Z.H. Integrating polymerase chain reaction, valving, and electrophoresis in a plastic device for bacterial detection. Anal. Chem. 2003, 75, 4591–4598. [Google Scholar]
- Liu, R.H.; Yang, J.; Lenigk, R.; Bonanno, J.; Grodzinski, P. Self-contained, fully integrated biochip for sample preparation, polymerase chain reaction amplification, and DNA microarray detection. Anal. Chem. 2004, 76, 1824–1831. [Google Scholar]
- Mao, X.; Yang, L.; Su, X.-L.; Li, Y. A nanoparticle amplification based quartz crystal microbalance DNA sensor for detection of Escherichia coli O157:H7. Biosens. Bioelectron. 2006, 21, 1178–1185. [Google Scholar]
- Dimov, I.K.; Garcia-Cordero, J.L.; O'Grady, J.; Poulsen, C.R.; Viguier, C.; Kent, L.; Daly, P.; Lincoln, B.; Maher, M.; O'Kennedy, R.; Smith, T.J.; Ricco, A.J.; Lee, L.P. Integrated microfluidic tmRNA purification and real-time NASBA device for molecular diagnostics. Lab. Chip 2008, 8, 2071–2078. [Google Scholar]
- Elsholz, B.; Wo, R.; Blohm, L.; Albers, J.; Feucht, H.; Grunwald, T.; Juergen, B.; Schweder, T.; Hintsche, R. Automated detection and quantitation of bacterial RNA by using electrical microarrays. Anal. Chem. 2006, 78, 4794–4802. [Google Scholar]
- Wei, C.-W.; Cheng, J.-W.; Huang, C.-T.; Yen, M.-H.; Young, T.-H. Using a microfluidic device for 1 ml DNA microarray hybridization in 500 s. Nucleic Acids Res. 2005, 33, e78. [Google Scholar]
- Peplies, J.; Lachmund, C.; Gloeckner, F.O.; Manz, W. A DNA microarray platform based on direct detection of rRNA for characterization of freshwater sediment-related prokaryotic communities. Appl. Environ. Microbiol. 2006, 72, 4829–4838. [Google Scholar]
- Cookson, A.L.; Woodward, M.J. The role of intimin in the adherence of enterohaemorrhagic Escherichia coli (EHEC) O157: H7 to HEp-2 tissue culture cells and to bovine gut explant tissues. Int. J. Med. Microbiol. 2003, 292, 547–553. [Google Scholar]
- Blanco, M.; Blanco, J.E.; Dahbi, G.; Alonso, M.P.; Mora, A.; Coira, M.A.; Madrid, C.; Juárez, A.; Bernárdez, M.I.; González, E.A.; Blanco, J. Identification of two new intimin types in atypical enteropathogenic Escherichia coli. Int. Microbiol. 2006, 9, 103–110. [Google Scholar]
- Torres, A.G.; Zhou, X.; Kaper, J.B. Adherence of diarrheagenic Escherichia coli strains to epithelial cells. Infect. Immun. 2005, 73, 18–29. [Google Scholar]
- Basselet, P.; Wegrzyn, G.; Enfors, S.-O.; Gabig-Ciminska, M. Sample processing for DNA chip array-based analysis of enterohemorrhagic Escherichia coli (EHEC). Microb. Cell Fact. 2008, 7. [Google Scholar] [CrossRef]
- Lay, C.; Teo, C.Y.; Zhu, L.; Peh, X.L.; Ji, H.M.; Chew, B.-R.; Murthy, R.; Feng, H.H.; Liu, W.-T. Enhanced microfiltration devices configured with hydrodynamic trapping and a rain drop bypass filtering architecture for microbial cell detection. Lab. Chip 2008, 8, 830–833. [Google Scholar]
- Lantz, A.W.; Bao, Y.; Armstrong, D.W. Single-cell detection: test of microbial contamination using capillary electrophoresis. Anal. Chem. 2007, 79, 1720–1724. [Google Scholar]
- Rodriguez, M.A.; Lantz, A.W.; Armstrong, D.W. Capillary electrophoretic method for the detection of bacterial contamination. Anal. Chem. 2006, 78, 4759–4767. [Google Scholar]
- Law, W.S.; Tay, E.T.T.; Feng, H.; Yu, L.; Zha, J.H.; Li, S.F.Y. Rapid identification of purified enteropathogenic Escherichia coli by microchip electrophoresis. J. Sep. Sci. 2007, 30, 1446–1452. [Google Scholar]
- Zourob, M.; Hawkes, J.J.; Coakley, W.T.; Brown, B.J.T.; Fielden, P.R.; McDonnell, M.B.; Goddard, N.J. Optical leaky waveguide sensor for detection of bacteria with ultrasound attractor force. Anal. Chem. 2005, 77, 6163–6168. [Google Scholar]
- McGovern, J.-P.; Shih, W.Y.; Shih, W.-H. In situ detection of Bacillus anthracis spores using fully submersible, self-exciting, self-sensing PMN-Pt/Sn piezolelectric microcantilevers. Analyst 2007, 132, 777–783. [Google Scholar]
- McGovern, J.-P.; Shih, W.Y.; Rest, R.; Purohit, M.; Pandya, Y.; Shih, W.-H. Label-free flow-enhanced specific detection of Bacillus anthracis using a piezoelectric microcantilever sensor. Analyst 2008, 133, 649–654. [Google Scholar]
- Wu, J.; Islam, N. A simple method to integrate in situ nano-particle focusing with cantilever detection. IEEE Sensors Journal 2007, 7, 957–958. [Google Scholar]
- Elkin, T.; Jiang, X.; Taylor, S.; Lin, Y.; Gu, L.; Yang, H.; Brown, J.; Collins, S.; Sun, Y.-P. Immuno-carbon nanotubes and recognition of pathogens. ChemBioChem 2005, 6, 640–643. [Google Scholar]
- Lin, Y.; Jiang, X.P.; Elkin, T.; Fernando, K.A.S.; Gu, L.; Taylor, S.; Yang, H.; Jones, E.; Wang, W.; Sun, Y.-P. Carbon nanotubes for immunomagnetic separation of Escherichia coli O157 : H7. J. Nanosci. Nanotech. 2006, 6, 868–871. [Google Scholar]
- Gu, L.; Elkin, T.; Jiang, X.; Li, H.; Lin, Y.; Qu, L.; Tzeng, T.R.; Joseph, R.; Sun, Y.-P. Single-walled carbon nanotubes displaying multivalent ligands for capturing pathogens. Chem. Commun. 2005, 874–876. [Google Scholar]
- Gu, L.; Luo, P.G.; Wang, H.; Meziani, M.J.; Lin, Y.; Veca, L.M.; Cao, L.; Lu, F.; Wang, X.; Quinn, R.A.; Wang, W.; Zhang, P.; Lacher, S.; Sun, Y.-P. Single-walled carbon nanotube as a unique scaffold for the multivalent display of sugars. Biomacromolecules 2008, 9, 2408–2418. [Google Scholar]
- Seo, S.; Kim, H.C.; Cheng, M.; Ruan, X.; Ruan, W. Microelectrical noise detector for rapid, specific, and sensitive identification of bacteria. J. Vac. Sci. Technol., B 2006, 24, 3133–3138. [Google Scholar]
- Villamizar, R.A.; Maroto, A.; Rius, F.X.; Inza, I.; Figueras, M.J. Fast detection of Salmonella Infantis with carbon nanotube field effect transistors. Biosens. Bioelectron. 2008, 24, 279–283. [Google Scholar]
- Suehiro, J.; Ikeda, N.; Ohtsubo, A.; Imasaka, K. Fabrication of bio/nano interfaces between biological cells and carbon nanotubes using dielectrophoresis. Microfluid. Nanofluid 2008, 5, 741–747. [Google Scholar]
- de la Rica, R.; Mendoza, E.; Lechuga, L.M.; Matsui, H. Label-free pathogen detection with sensor chips assembled from peptide nanotubes. Angew. Chem. Int. Ed. 2008, 47, 9752–9755. [Google Scholar]
- Pal, S.; Alocilja, E.C.; Downes, F.P. Nanowire labeled direct-charge transfer biosensor for detecting Bacillus species. Biosens. Bioelectron. 2007, 22, 2329–2336. [Google Scholar]
- Mishra, N.N.; Maki, W.C.; Cameron, E.; Nelson, R.; Winterrowd, P.; Rastogi, S.K.; Filanoski, B.; Maki, G.K. Ultrasensitive detection of bacterial toxin with silicon nanowire transistor. Lab. Chip 2008, 8, 868–871. [Google Scholar]
- Tok, J.B.-H.; Chuang, F.Y.S.; Kao, M.C.; Rose, K.A.; Pannu, S.S.; Sha, M.Y.; Chakarova, G.; Penn, S.G.; Dougherty, G.M. Metallic striped nanowires as multiplexed immunoassay platforms for pathogen detection. Angew. Chem. Int. Ed. 2006, 45, 6900–6904. [Google Scholar]
- Hahn, M.A.; Tabb, J.S.; Krauss, T.D. Detection of single bacterial pathogens with semiconductor quantum dots. Anal. Chem. 2005, 77, 4861–4869. [Google Scholar]
- Yang, L.; Li, Y. Simultaneous detection of Esherichia coli O157:H7 and Salmonella Typhimurium using quantum dots as fluorescent labels. Analyst 2006, 131, 394–401. [Google Scholar]
- Liu, Y.; Brandon, R.; Cate, M.; Peng, X.; Stony, R.; Johnson, M. Detection of pathogen using luminescent CdSe/ZnS dendron nanocrystals and a porous membrane immunofilter. Anal. Chem. 2007, 79, 8796–8802. [Google Scholar]
- Huang, C.-C.; Chen, C.-T.; Shiang, Y.-C.; Lin, Z.-H.; Chang, H.-T. Synthesis of fluorescent carbohydrate-protected Au nanodots for detection of Concanavalin A and Escherichia coli. Anal. Chem. 2009, 81, 875–882. [Google Scholar]
- Zhao, X.; Hilliard, L.R.; Mechery, S.J.; Wang, Y.; Bagwe, R.P.; Jin, S.; Tan, W. A rapid bioassay for single bacteria cell quantitation using bioconjugated nanoparticles. Proc. Natl. Acad. Sci. USA 2004, 101, 15027–15032. [Google Scholar]
- Edgar, R.; McKinstry, M.; Hwang, J.; Oppenheim, A.B.; Fekete, R.A.; Giulian, G.; Merril, C.; Nagashima, K.; Adhya, S. High-sensitivity bacterial detection using biotin-tagged phage and quantum-dot nanocomplexes. Proc. Natl. Acad. Sci. USA 2006, 103, 4841–4845. [Google Scholar]
- Naja, G.; Bouvrette, P.; Hrapovich, S.; Liu, Y.; Luong, J.H.T. Detection of bacteria aided by immuno-nanoparticles. J. Raman Spectrosc. 2007, 38, 1383–1389. [Google Scholar]
- Farrel, S.; Halsall, B.; Heineman, W.R. Immunoassay for B. globigii spores as a model for detecting B. anthracis spores in finished water. Analyst 2005, 130, 489–497. [Google Scholar]
- El-Boubbou, K.; Gruden, C.; Huang, X. Magnetic glyco-nanoparticles: a unique tool for rapid pathogen detection, decontamination, and strain differentiation. J. Am. Chem. Soc. 2007, 129, 13392–13393. [Google Scholar]
- Lin, Y.-H.; Chen, S.-H.; Chuang, Y.-C.; Lu, Y.-C.; Shen, T.-Y.; Chang, C.-A.; Lin, C.-S. Disposable amperometric immunosensing strips fabricated by Au nanoparticles-modified screen-printed carbon electrodes for the detection of foodborne pathogen Escherichia coli O157:H7. Biosens. Bioelectron. 2008, 23, 1832–1837. [Google Scholar]

© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Heo, J.; Hua, S.Z. An Overview of Recent Strategies in Pathogen Sensing. Sensors 2009, 9, 4483-4502. https://doi.org/10.3390/s90604483
Heo J, Hua SZ. An Overview of Recent Strategies in Pathogen Sensing. Sensors. 2009; 9(6):4483-4502. https://doi.org/10.3390/s90604483
Chicago/Turabian StyleHeo, Jinseok, and Susan Z Hua. 2009. "An Overview of Recent Strategies in Pathogen Sensing" Sensors 9, no. 6: 4483-4502. https://doi.org/10.3390/s90604483
APA StyleHeo, J., & Hua, S. Z. (2009). An Overview of Recent Strategies in Pathogen Sensing. Sensors, 9(6), 4483-4502. https://doi.org/10.3390/s90604483



