Development of QCM Trimethylamine Sensor Based on Water Soluble Polyaniline
Abstract
:1. Introduction
2. Experiment
2.1. Materials
2.2. Preparation and characterization of water soluble polyaniline
2.3. Preparation and pretreatment of coated sensor
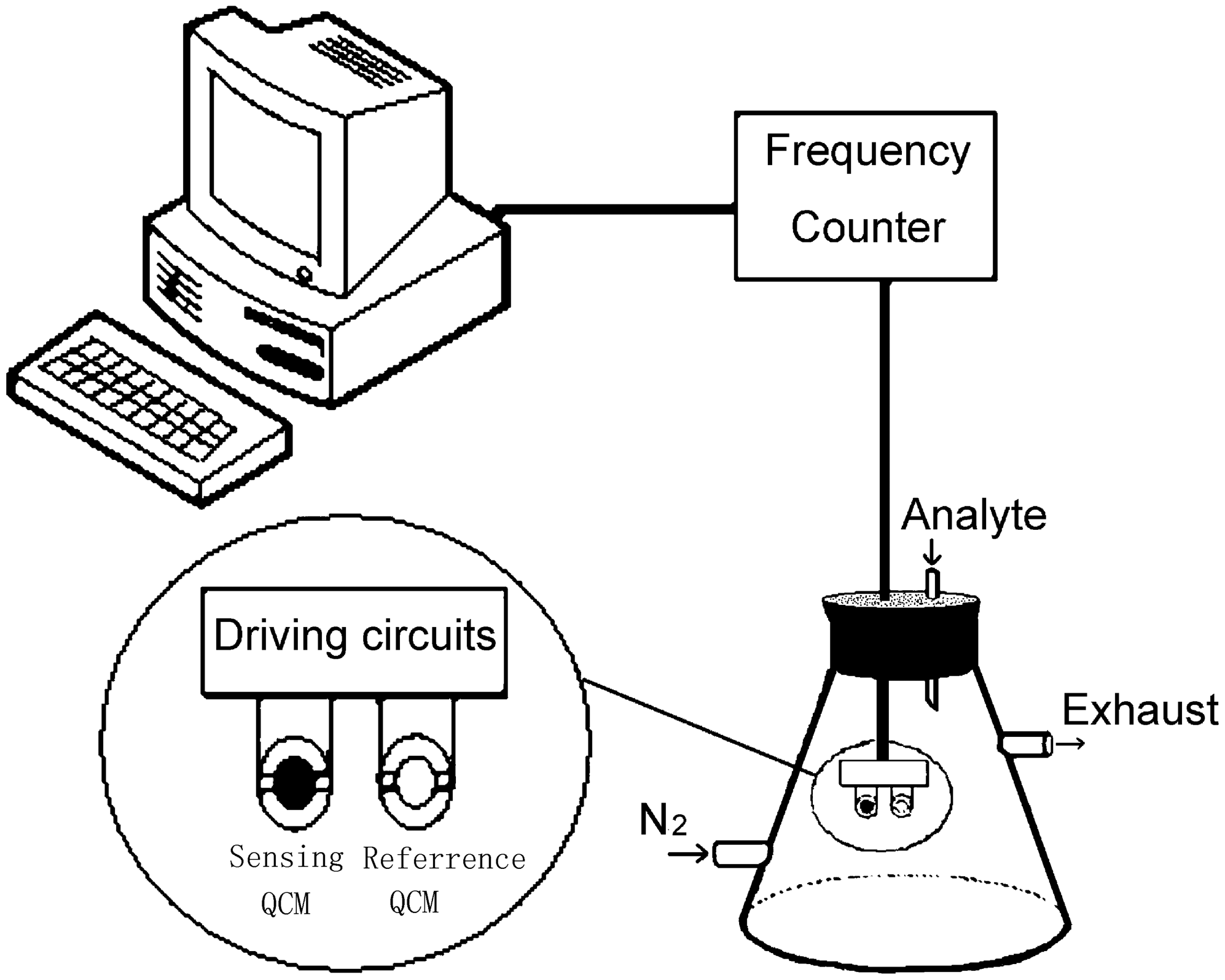
2.4. Gas sensing experiments
3. Results and discussion
3.1. Water soluble polyaniline film characteristics
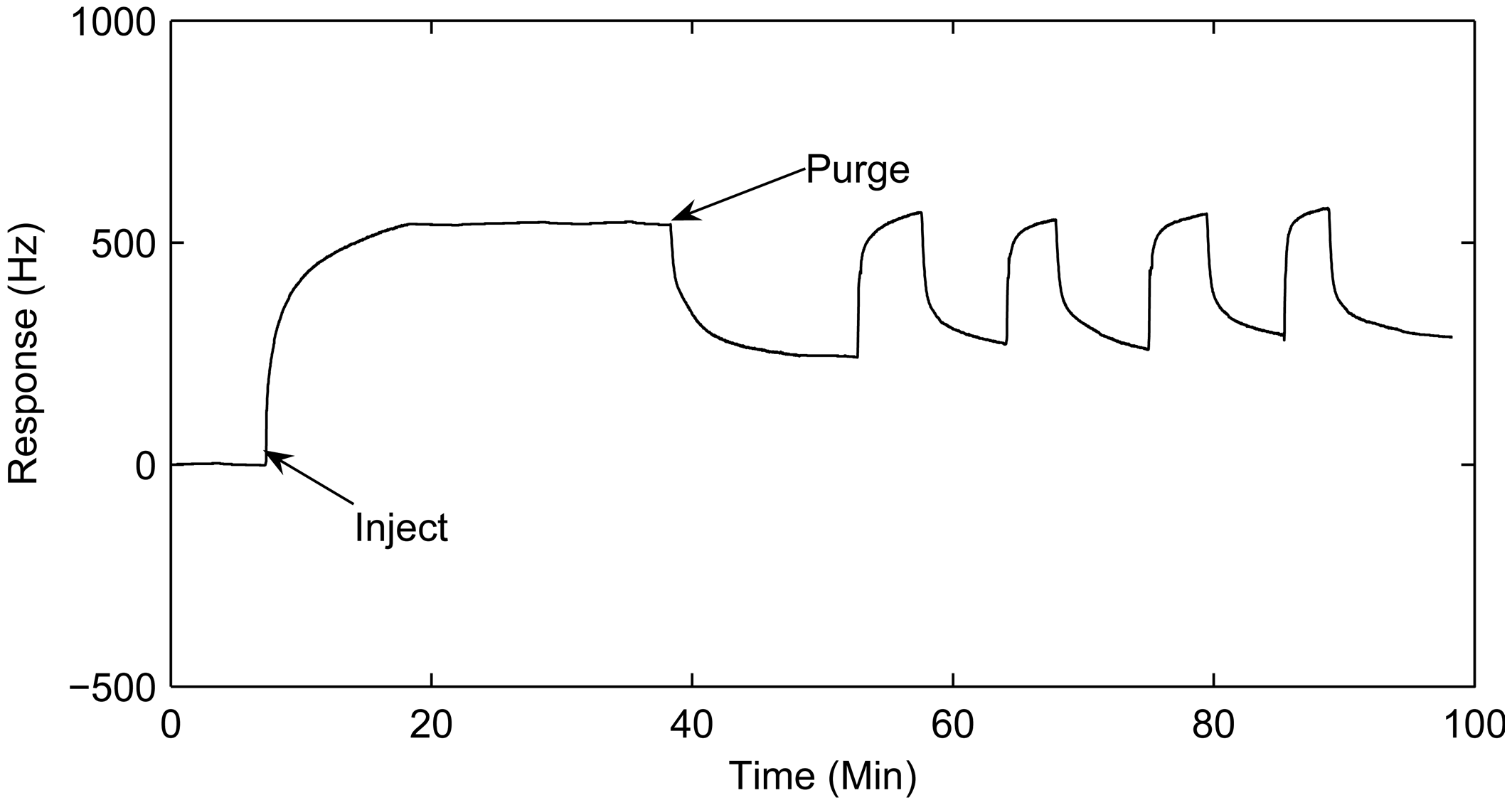
3.2. Pretreatment of the polyaniline-coated sensor
3.3. Repeatability
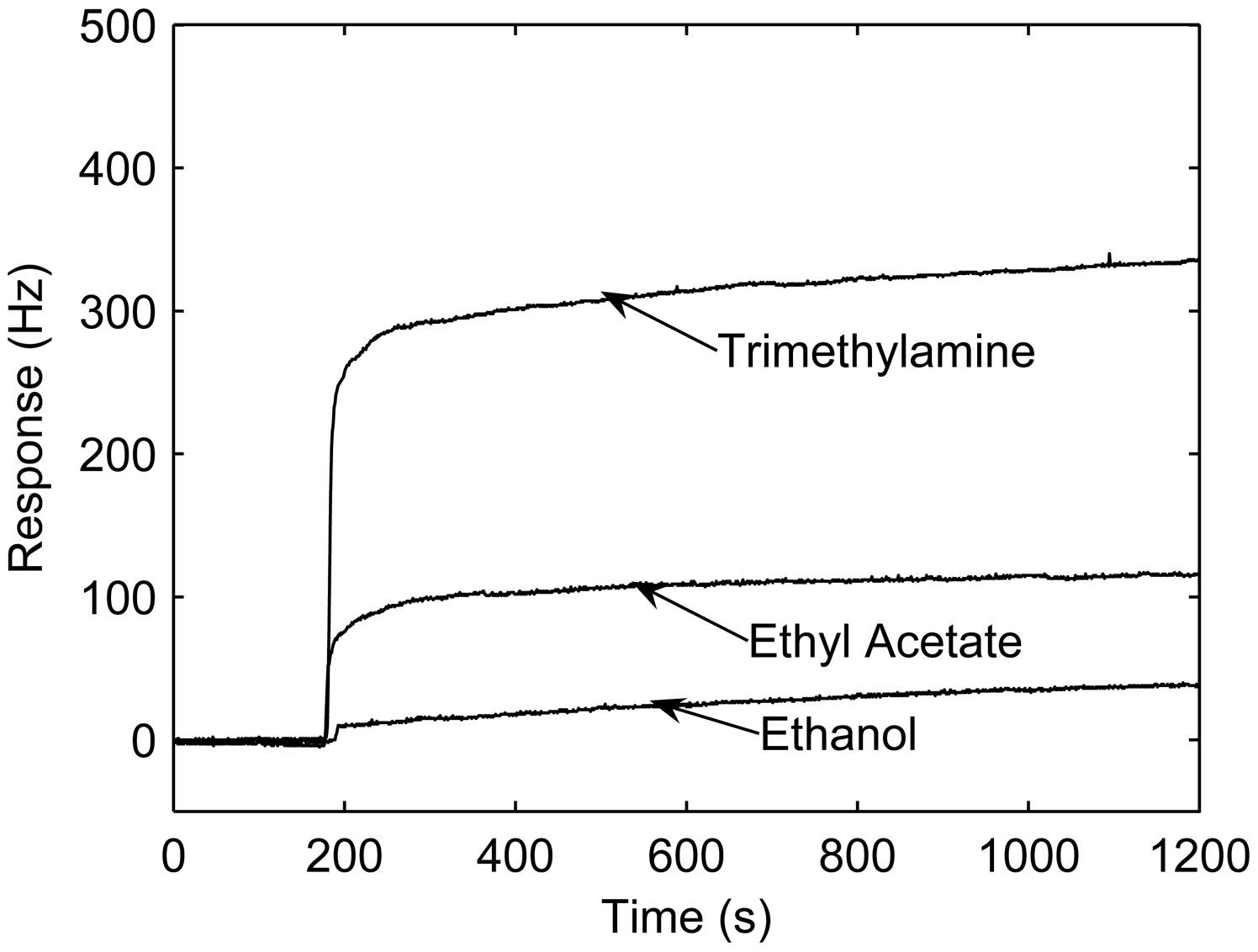
3.4. Responses to different vapors
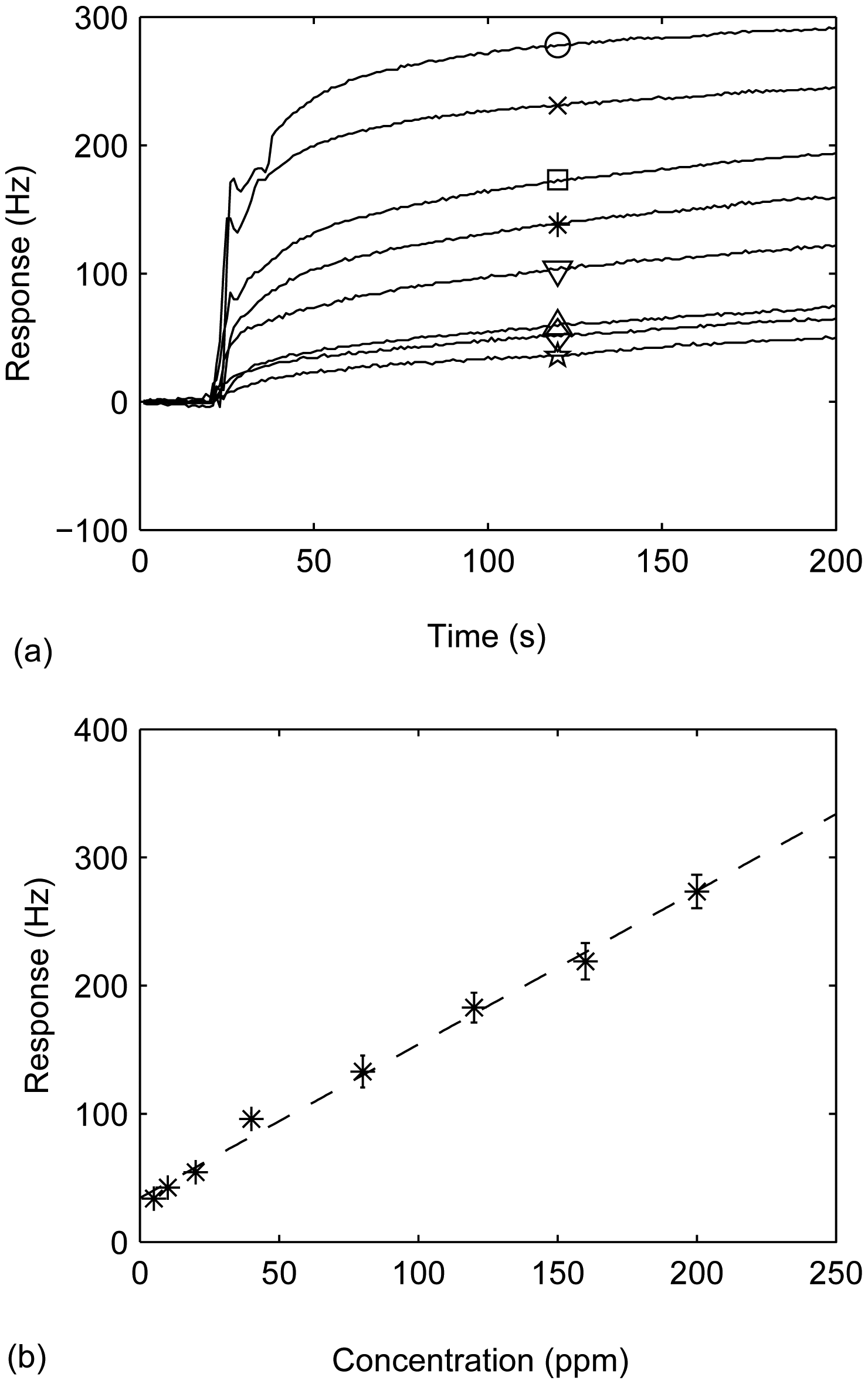
3.5. Sensitivity of the responses
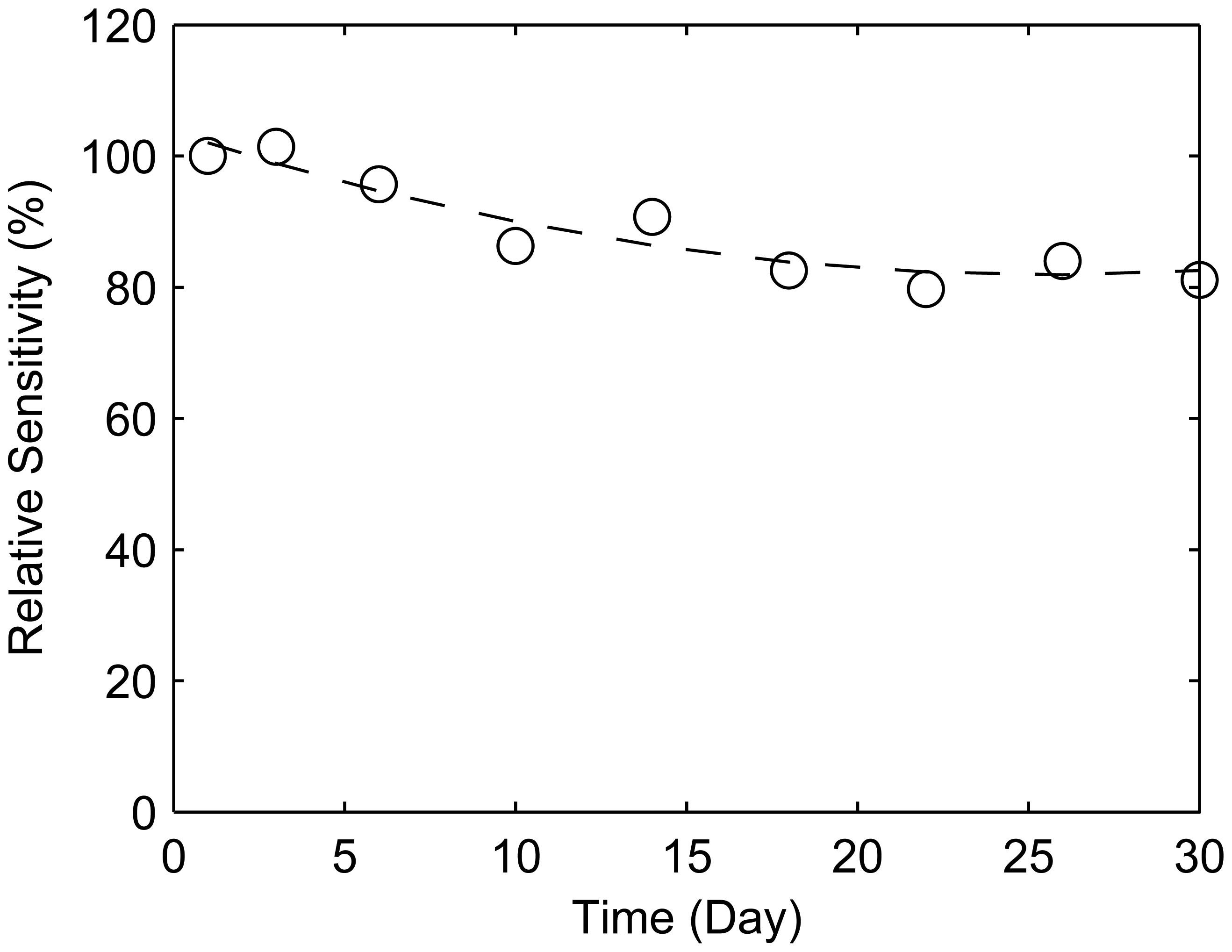
3.6. Durability
4. Conclusion
Acknowledgments
References and Notes
- Bota, G. M.; Harrington, P. B. Direct detection of trimethylamine in meat food products using ion mobility spectrometry. Talanta 2006, 68, 629–635. [Google Scholar]
- Chan, S.T.; Yao, M.W.Y.; Wong, Y.C.; Wong, T.; Mok, C.S.; Sin, D.W.M. Evaluation of chemical indicators for monitoring freshness of food and determination of volatile amines in fish by headspace solid-phase microextraction and gas chromatography-mass spectrometry. Eur. Food Res. Technol. 2006, 224, 67–74. [Google Scholar]
- Periago, M.J.; Rodrigo, J.; Ros, G.; Rodriguez-Jerez, J.J.; Hernandez-Herrero, M. Monitoring volatile and nonvolatile amines in dried and salted roes of tuna (Thunnus thynnus L.) during manufacture and storage. J. Food Prot. 2005, 66, 335–340. [Google Scholar]
- Bain, M.A.; Fornasini, G.; Evans, A.M. Trimethylamine: metabolic, pharmacokinetic and safety aspects. Curr. Drug Metab. 2005, 6, 227–240. [Google Scholar]
- Bain, M.A.; Faull, R.; Fornasini, G.; Milne, R.W.; Evans, A.M. Accumulation of trimethylamine and trimethylamine-N-oxide in end-stage renal disease patients undergoing haemodialysis. Nephrol. Dial. Transplant. 2006, 21, 1300–1304. [Google Scholar]
- Claeson, A.S.; Sandström, M.; Sunesson, A.L. Volatile organic compounds (VOCs) emitted from materials collected from buildings affected by microorganisms. J. Environ. Monit. 2007, 9, 240–245. [Google Scholar]
- Greim, H.; Bury, D.; Klimisch, H.J.; Oeben-Negele, M.; Ziegler-Skylakakis, K. Toxicity of aliphatic amines: Structure-activity relationship. Chemosphere 1998, 36, 271. [Google Scholar]
- Kaniou, I.; Samouris, G.; Mouratidou, T.; Eleftheriadou, A.; Zantopoulos, N. Determination of biogenic amines in fresh unpacked and vacuum-packed beef during storage at 4±°C. Food Chem. 2001, 74, 515–519. [Google Scholar]
- Cháfer-Pericás, C.; Campíns-Falcó, P.; Herráez-Hernández, R. Comparative study of the determination of trimethylamine in water and air by combining liquid chromatography and solid-phase microextraction with on-fiber derivatization. Talanta 2006, 69, 716–723. [Google Scholar]
- Namieśnik, J.; Jastrzębska, A.; Zygmunt, B. Determination of volatile aliphatic amines in air by solid-phase microextraction coupled with gas chromatography with flame ionization detection. J. Chromatogr. A 2003, 1016, 1–9. [Google Scholar]
- Adhoum, N.; Monser, L.; Sadok, S.; El-Abed, A.; Greenway, G.M.; Uglow, R.F. Flow injection potentiometric detection of trimethylamine in seafood using tungsten oxide electrode. Anal. Chim. Acta 2003, 478, 53–58. [Google Scholar]
- Sadek, A.Z.; Wlodarski, W.; Shin, K.; Kaner, R.B.; Kalantar-zadeh, K. A layered surface acoustic wave gas sensor based on a polyaniline/In2O3 nanofibre composite. Nanotechnology 2006, 17, 4488–4492. [Google Scholar]
- Athawale, A.A.; Bhagwat, S.V.; Katre, P.P. Nanocomposite of Pd-polyaniline as a selective methanol sensor. Sens. Actutator B: Chem. 2006, 114, 263–267. [Google Scholar]
- Prasad, G.K.; Radhakrishnan, T.P.; Kumar, D.S.; Ghanashyam Krishna, M. Ammonia sensing characteristics of thin film based on polyelectrolyte templated polyaniline. Sens. Actutator B: Chem. 2005, 106, 626–631. [Google Scholar]
- Lee, Y.S.; Song, K.D.; Huh, J.S.; Chung, W.Y.; Lee, D.D. Fabrication of clinical gas sensor using MEMS process. Sens. Actutator B: Chem. 2005, 108, 292–297. [Google Scholar]
- Dixit, V.; Misra, S.C.K.; Sharma, B.S. Carbon monoxide sensitivity of vacuum deposited polyaniline semiconducting thin films. Sens. Actutator B: Chem. 2005, 104, 90–93. [Google Scholar]
- Bai, H.; Shi, G. Gas sensors based on conducting polymers. Sensors 2007, 7, 267–307. [Google Scholar]
- Ma, X.; Wang, M.; Chen, H.; Li, G. J. Sun; Bai, R. Preparation of water soluble poly(aniline) and its gas-sensitivity. Green Chem. 2005, 7, 507–513. [Google Scholar]
- Sauerbrey, G. The use of quartz oscillators for weighing layers and for micro-weighing. Z. Phys. 1959, 155, 206–222. [Google Scholar]
- Matshuguchi, M.; Kadowaki, Y.; Tanaka, M. A QCM-based NO2 gas detector using morpholine-functional cross-linked copolymer coatings. Sens. Actutator B: Chem. 2005, 108, 572–575. [Google Scholar]
- Koshets, I.A.; Kazantseva, Z.I.; Shirshov, Y.M.; Cherenok, S.A.; Kalchenko, V.I. Calixarene films as sensitive coating for QCM-based sensors. Sens. Actutator B: Chem. 2005, 106, 177–181. [Google Scholar]
- Vilaseca, M.; Yagüe, C.; Coronas, J.; Santamaria, J. Development of QCM sensors modified by AlPO4-18 films. Sens. Actutator B: Chem. 2006, 117, 143–150. [Google Scholar]
- Sasaki, I.; Tshuchiya, H.; Nishioka, M.; Sadakata, M.; Okubo, T. Gas sensing with zeolite-coated quartz crystal microbalances—principal component analysis approach. Sens. Actutator B: Chem. 2002, 86, 26–33. [Google Scholar]
- Mirmohseni, A.; Oladegaragoze, A. Construction of a sensor for determination of ammonia and aliphatic amines using polyvinylpyrrolidone coated quartz crystal microbalance. Sens. Actutator B: Chem. 2003, 89, 164–172. [Google Scholar]
- Stahl, U.; Rapp, M.; Wessa, T. Adhesives: a new class of polymer coating for surface acoustic wave sensors for fast and reliable process control applications. Anal. Chim. Acta 2001, 450, 27–36. [Google Scholar]
- Ayad, M.M.; Shenashin, M.A. Polyaniline film deposition from the oxidative polymerization of aninie using K2Cr2O7. Eur. Polym. J. 2004, 40, 197–202. [Google Scholar]
- Ma, X.; Li, G.; Wang, M.; Bai, R.; Yang, F.; Chen, H. Unusual electrical response of a poly(aniline) composite film on exposure to a basic atmosphere and its application to sensors. Green Chem. 2006, 8, 63–691. [Google Scholar]

© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Li, G.; Zheng, J.; Ma, X.; Sun, Y.; Fu, J.; Wu, G. Development of QCM Trimethylamine Sensor Based on Water Soluble Polyaniline. Sensors 2007, 7, 2378-2388. https://doi.org/10.3390/s7102378
Li G, Zheng J, Ma X, Sun Y, Fu J, Wu G. Development of QCM Trimethylamine Sensor Based on Water Soluble Polyaniline. Sensors. 2007; 7(10):2378-2388. https://doi.org/10.3390/s7102378
Chicago/Turabian StyleLi, Guang, Junbao Zheng, Xingfa Ma, Yu Sun, Jun Fu, and Gang Wu. 2007. "Development of QCM Trimethylamine Sensor Based on Water Soluble Polyaniline" Sensors 7, no. 10: 2378-2388. https://doi.org/10.3390/s7102378
APA StyleLi, G., Zheng, J., Ma, X., Sun, Y., Fu, J., & Wu, G. (2007). Development of QCM Trimethylamine Sensor Based on Water Soluble Polyaniline. Sensors, 7(10), 2378-2388. https://doi.org/10.3390/s7102378



_Ma.jpg)
