Influence of Different Genotypes on Trypsin Inhibitor Levels and Activity in Soybeans
Abstract
:1. Introduction
2. Experimental Section
Materials
Protein extracts
PAGE
Trypsin inhibitor activity (TIA)
Statistical analysis
3. Discussion
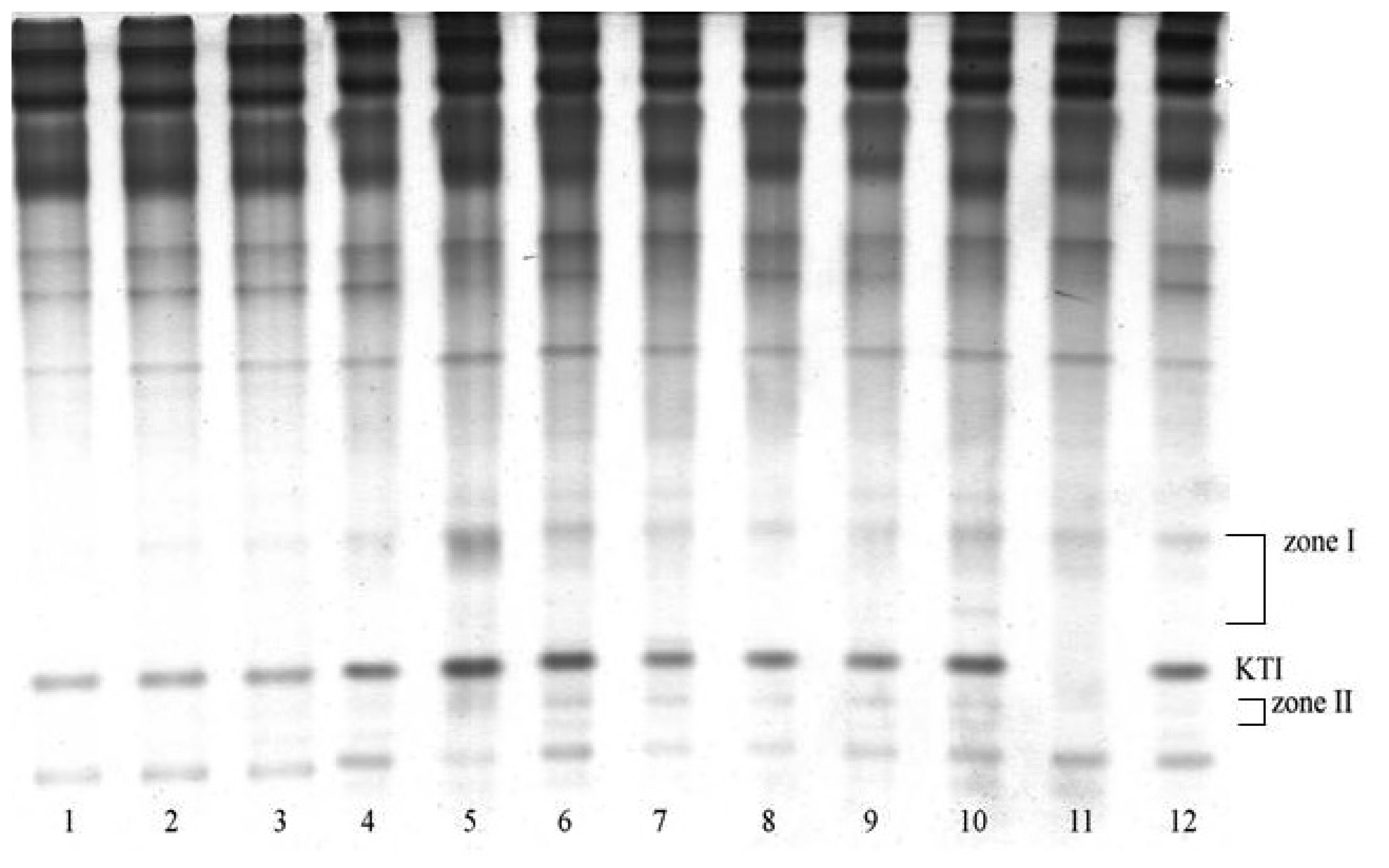
3.1. Electrophoretic analysis
3.2 Correlation analysis
3.3 Trypsin inhibitor activity
Acknowledgments
References
- Wang, C.; Wikson, R. Phytochemicals in Soybeans-Their Potential Health Benefits. Inform 1999, 10, 315–320. [Google Scholar]
- Sessa, D.J.; Ghantous, P.E. Chemical Inactivation of Soybean Trypsin Inhibitors. J. Am. Oil Chem. Soc. 1987, 64, 1682–1687. [Google Scholar]
- DiPietro, C.M.; Liener, I.E. Heat inactivation of the Kunitz and Bowman-Birk soybean protease inhibitors. J. Agric. Food Chem. 1989, 37, 39–44. [Google Scholar]
- Stanojević, S.; Barać, M.; Vucelić -Radović, B.; Pešić, M. The effect of autoclaving on soluble protein composition and trypsin inhibitor activity of cracked soybeans. Acta Period. Technol (APTEFF) 2004, 35, 49–58. [Google Scholar]
- Huang, H.; Liang, H.; Kwok, K-C. Effects of Ultrasound Fields on the Activity and Secondary Structures of Soybean Trypsin Inhibitors. Abstracts of Papers, 226th ACS National Meeting, New York, NY, September 7-11; 2003. [Google Scholar]
- Van der Ven, C.; Matser, A.M.; van den Berg, C. Inactivation of Soybean Trypsin Inhibitors and Lipoxygenase by High-Pressure Processing. J. Agric. Food Chem. 2005, 53, 1087–1092. [Google Scholar]
- Barac, M.; Stanojevic, S. The Effect of Microwave Roasting on Soybean Protein Composition and Components with Trypsin Inhibitor Activity. Acta Aliment. 2005, 34, 23–31. [Google Scholar]
- Kennedy, A.R. Cancer Prevention by Protease Inhibitors. Prevent. Med. 1993, 22, 796–811. [Google Scholar]
- Kennedy, A.R.; Szuhaja, B.F. Method of Making Soybean Bowman-Birk Inhibitor Concentrate and Use of Same as a Human Cancer Preventative and Therapy. US Patent No. 5,217,717, 1993. [Google Scholar]
- Kennedy, A.R.; Szuhaja, B.F. Bowman-Birk Inhibitor Product for Use as an Anticancerogenesis Agent. US Patent No. 5,338,547, 1994. [Google Scholar]
- Sessa, D.J.; Wolf, W.J. Bowman-Birk inhibitors in soybean seed coats. Ind. Crop. Prod. 2001, 14, 73–83. [Google Scholar]
- Esposito, E.; Cortesi, R.; Bortolotti, F.; Menegatti, E.; Nastruzzi, C. Production and Characterization of Biodegradable Microparticles for the controlled delivery of Proteinase Inhibitors. Int. J. Pharmaceut. 1996, 129, 263–273. [Google Scholar]
- Friedman, M.; Brandon, D.L.; Bates, A.H.; Hymowitz, T. Comparison of a Commercial Soybean Cultivar and an Isoline Lacking the Kunitz Trypsin Inhibitors: Composition, Nutritional Value, and Effects of Heating. J. Agric. Food Chem. 1991, 39, 327–335. [Google Scholar]
- Carvalho, W.L.; Almeida Oliveira, M.G.; Barros, E.G.; Moreira, M.A. Lipoxygenases affect protease inhibitor levels in soybean seeds. Plant Phisiol. Biochem. 1999, 37, 497–501. [Google Scholar]
- Mejia, E.G.; Guzman-Maldonado, S.H.; Acosta-Gallegos, J.A.; Reynoso-Camacho, R.; Ramirez-Rodriguez, E.; Pons-Hernandez, J.L.; Gonzalez-Chavira, M.M.; Castellanos, J.Z.; Kelly, J.D. Effect of cultivar and Growing Location on the Trypsin Inhibitors, Tannins, and Lectins of Common Beans (Phaseolus vulgaris L.) Grown in the Semiarid Highlands of Mexico. J. Agric. Food Chem. 2003, 51, 5962–5966. [Google Scholar]
- Rackis, J.J.; Wolf, W.J.; Backer, E.C. Protease Inhibitors on Plant Foods: Content and Inactivation. Adv. Exp. Med. Biol. 1986, 199, 299–347. [Google Scholar]
- Tan-Wilson, A.L.; Wilson, K.A. Relevance of Multiple Soybean Trypsin Inhibitor Forms to Nutritional Quality. Adv. Exp. Med. Biol. 1986, 199, 391–411. [Google Scholar]
- Brandon, D.L.; Friedman, M. Immunoassays of Soy Proteins. J. Agric. Food Chem. 2002, 50, 6635–6642. [Google Scholar]
- Barac, M.B.; Jovanovic, S.T.; Stanojevic, S.P.; Pesic, M.B. Effect of Limited Hydrolysis on Traditional Soy Protein Concentrate. Sensors 2006, 6, 1087–1101. [Google Scholar]
- Pesic, M.B.; Vucelic-Radovic, B.V.; Barac, M.B.; Stanojevic, S.P. The Influence of Genotypic Variation in Protein Composition on Emulsifying Properties of Soy Proteins. J. Am. Oil Chem. Soc. 2005, 82, 667–672. [Google Scholar]
- Vollmann, J.; Grausgruber, H.; Wagentristl, H.; Wohleser, H.; Michele, P. Trypsin inhibitor activity of soybean as affected by genotype and fertilisation. J. Sci. Food Agric. 2003, 83, 1581–1586. [Google Scholar]
- Lowry, O.H.; Rosenbrough, N.J.; Farr, A.L.; Randall, R.J. Protein Measurement with the Folin Phenol Reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar]
- Davis, J. Disc Electrophoresis-II Method and Application to Human Serum Proteins. Ann. N. Y. Acad. Sci 1964, 121, 404–427. [Google Scholar]
- Liu, F.; Markakis, P. An Improved Colorimetric Method for Determining Antitryptic Activity in Soybean Products. Cereal Chem. 1989, 66, 415–422. [Google Scholar]
- Sessa, D.J.; Bietz, J.A. Toasted soybean flour components with trypsin inhibitor activity. J. Am. Oil Chem. Soc. 1986, 63, 784–788. [Google Scholar]
- Foard, D.E.; Gutay, P.A.; Ladin, B.; Beachy, R.N.; Larkins, B.A. In Vitro Synthesis of the Bowman-Birk and Related Soybean Protease Inhibitors. Plant. Mol. Biol. 1982, 1, 227–243. [Google Scholar]
- Mejia, E.G.; Vasconez, M.; Lumen, B.O.; Nelson, R. Lunasin Concentration in Different Genotypes, Commercial Soy Protein and Isoflavone Products. J. Agric. Food Chem. 2004, 52, 5882–5887. [Google Scholar]
- Tolstoguzov, V.B. Functional Properties of Food Proteins. Role of Interactions in Protein Systems. In Food Protein Structure and Functionality; Schwenke, K.D., Mothes, R., Eds.; VHC: Weinheim, 1993; pp. 203–209. [Google Scholar]
| Genotype | KTI2 | BBI2 | Total | KTI/BB I | TIA2 | ||
|---|---|---|---|---|---|---|---|
| polymeric forms | monomeri c forms | BBI | TI2 | ||||
| (%EP3) | (TUI/mg) | ||||||
| Lana | / | 2.02 a | 0.30 a | 2.32 a | 2.32 a | 0 | 60.36 a |
| ZPS-015 | 5.63 a | 2.84 b | 0.46 b | 3.30 b | 8.93 b | 1.71 | 73.65 b |
| Nena | 4.48 b.c | 0.89 c | 0.51 c,d | 0.60 c | 5.88 c | 7.47 | 87.12 c,d,e,h |
| L91-31022 | 4.61d | 0.52 d | 0.08 e | 1.40 d | 5.21 d | 3.29 | 89.38 c |
| L94-1171 | 4.28 e | 0.72 e | 0.19 f | 0.91 e | 5.19 d,e | 4.7 | 85.18 c,d,g |
| Sg1-1 | 6.03 f | 2.0 a,b | 0.47 b,c | 2.47 f | 8.50 f | 2.44 | 98.99 f |
| Krajina | 6.85 g | 5.78 f | 0.54 d | 6.32 g | 13.17g | 1.08 | 100.95 f |
| Vojvodjanka | 4.37 b,e | 0.23 g | 0.01 g | 0.24 h | 4.61 h | 18.21 | 85.78 d,g |
| Novosadjank a | 4.62 c,d | 0.91 c | 0.06 h | 0.97 e,i | 5.59 i | 4.76 | 95.53 e,f |
| Proteinka | 4.37 b,e | 0.95 c | 0.05 h | 1.00 i | 5.37 e,i | 4.37 | 97.32 f |
| Balkan | 4.35 e | 0.47 d | 0.30 a | 0.77 j | 5.12 d | 5.65 | 80.22 g,b,i |
| Ravnica | 4.80 h | 0.84 c | 0.31 a | 1.15 k | 5.95 c | 4.17 | 79.55 h,i |
| Factors | KTI | BBI | Total | ||
|---|---|---|---|---|---|
| polymeric forms | monomeric forms | BBI | TI | ||
| KTI | 0.93 2 | 0.69 2 | 0.94 2 | 0.97 2 | |
| BBIp | 0.93 2 | 0.65 2 | 0.99 2 | 0.99 2 | |
| BBIm | 0.69 2 | 0.65 2 | 0.62 2 | 0.72 2 | |
| total BBI2 | 0.94 | 0.99 2 | 0.62 2 | 0.98 2 | |
| TIA | 0.37 | 0.35 | -0.05 | 0.33 | 0.34 |
© 2007 by MDPI ( http://www.mdpi.org). Reproduction is permitted for noncommercial purposes.
Share and Cite
Pesic, M.B.; Vucelic-Radovic, B.V.; Barac, M.B.; Stanojevic, S.P.; Nedovic, V.A. Influence of Different Genotypes on Trypsin Inhibitor Levels and Activity in Soybeans. Sensors 2007, 7, 67-74. https://doi.org/10.3390/s7010067
Pesic MB, Vucelic-Radovic BV, Barac MB, Stanojevic SP, Nedovic VA. Influence of Different Genotypes on Trypsin Inhibitor Levels and Activity in Soybeans. Sensors. 2007; 7(1):67-74. https://doi.org/10.3390/s7010067
Chicago/Turabian StylePesic, Mirjana B., Biljana V. Vucelic-Radovic, Miroljub B. Barac, Sladjana P. Stanojevic, and Viktor A. Nedovic. 2007. "Influence of Different Genotypes on Trypsin Inhibitor Levels and Activity in Soybeans" Sensors 7, no. 1: 67-74. https://doi.org/10.3390/s7010067
APA StylePesic, M. B., Vucelic-Radovic, B. V., Barac, M. B., Stanojevic, S. P., & Nedovic, V. A. (2007). Influence of Different Genotypes on Trypsin Inhibitor Levels and Activity in Soybeans. Sensors, 7(1), 67-74. https://doi.org/10.3390/s7010067




