Herbicide Bioassay Using a Multi-Well Plate and Plant Spectral Image Analysis
Abstract
1. Introduction
2. Materials and Methods
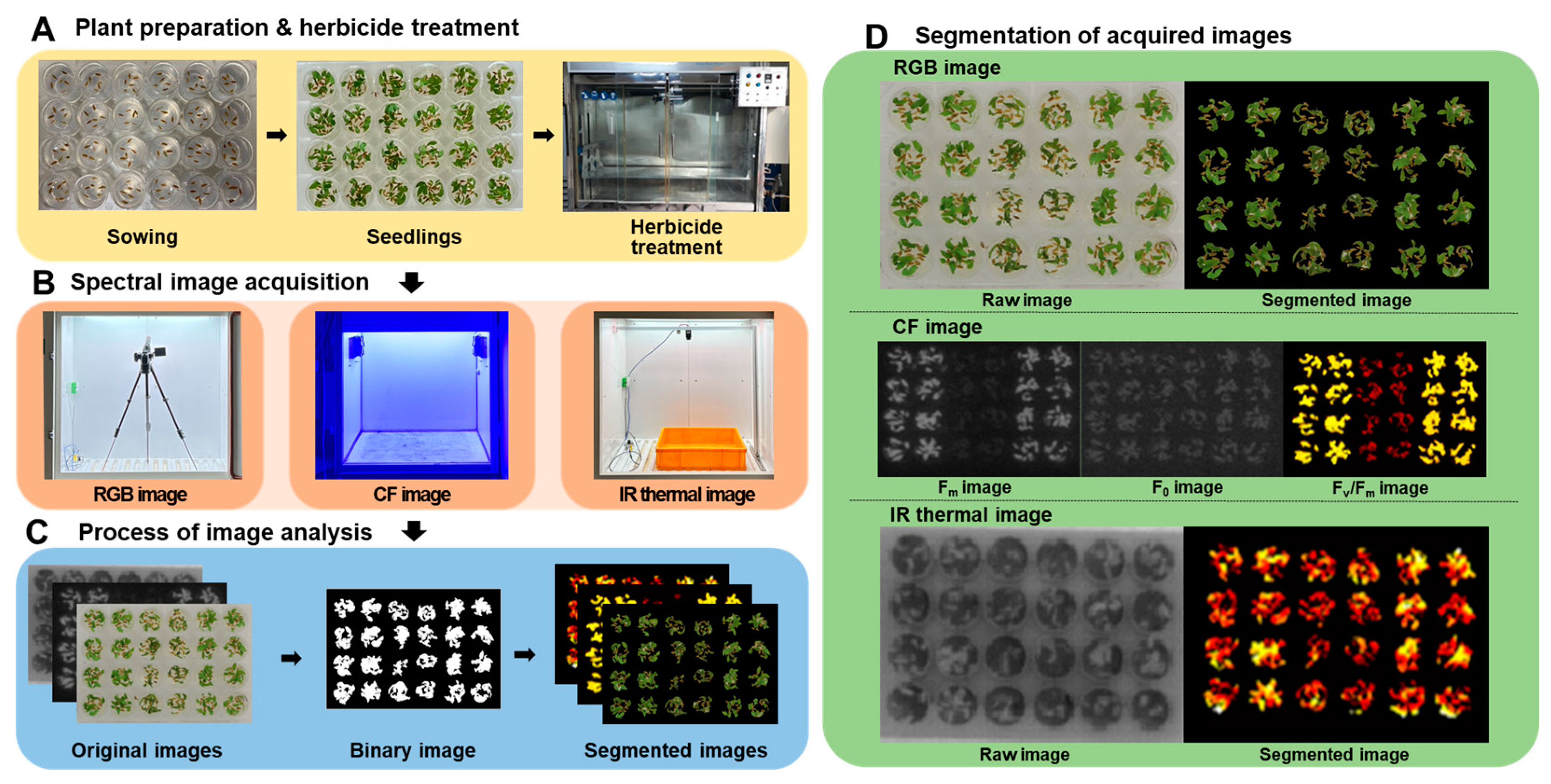
2.1. Plant Materials and Growing the Plant in Multi-Well Plates
2.2. Herbicide Treatment
2.3. Spectral Image Acquisition and Analysis
2.3.1. RGB Images
2.3.2. CF Images
2.3.3. IR Thermal Images
2.4. Statistical Analysis
3. Results
3.1. Optimization of Growing Crabgrass in Multi-Well Plates
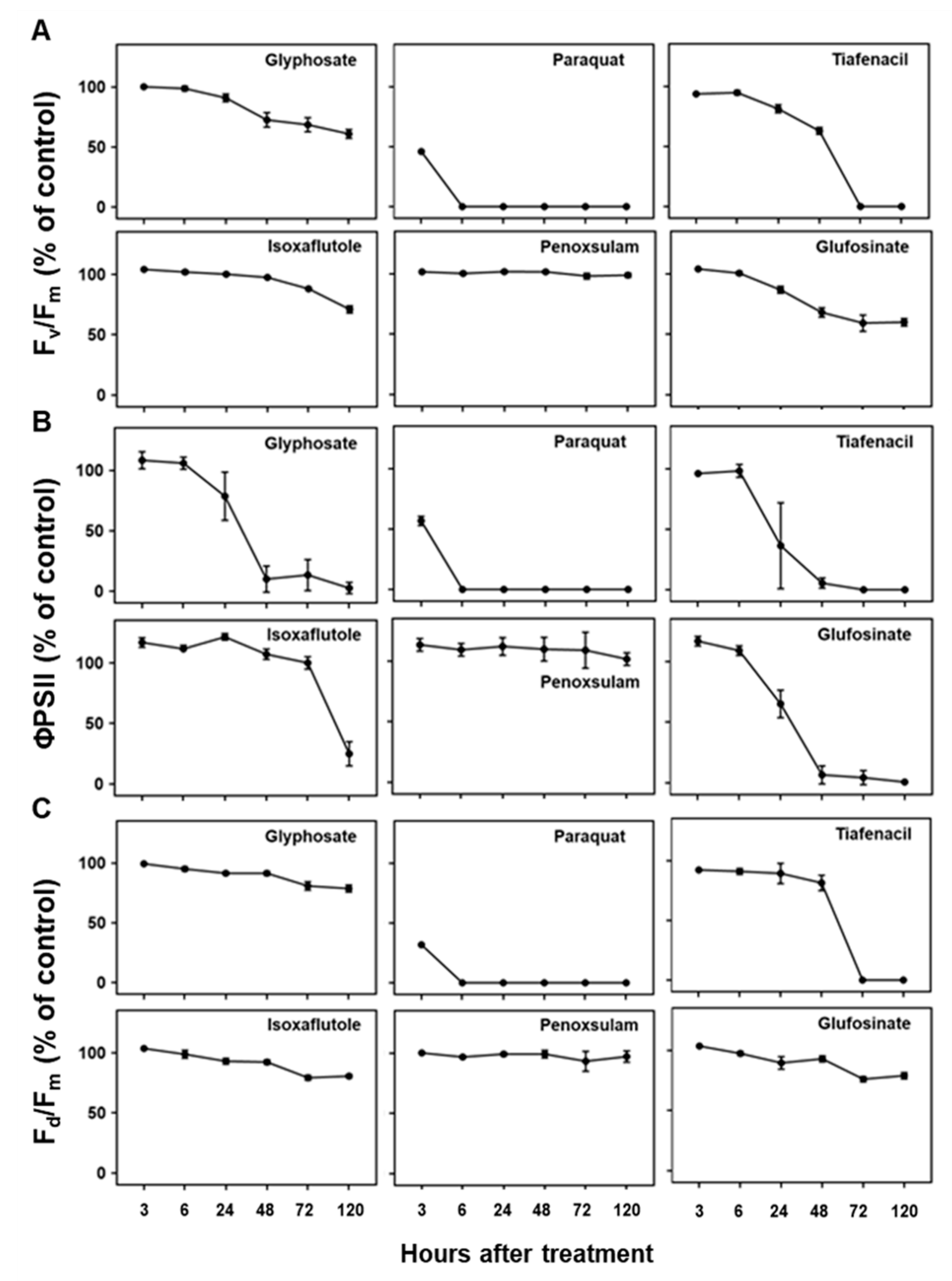
3.2. Changes in Spectral Response of Crabgrass to Herbicides
3.3. Changes in RGB Spectral Responses to Herbicides
3.4. Changes in CF Spectral Responses to Herbicides
3.5. Changes in IR Thermal Spectral Responses to Herbicides
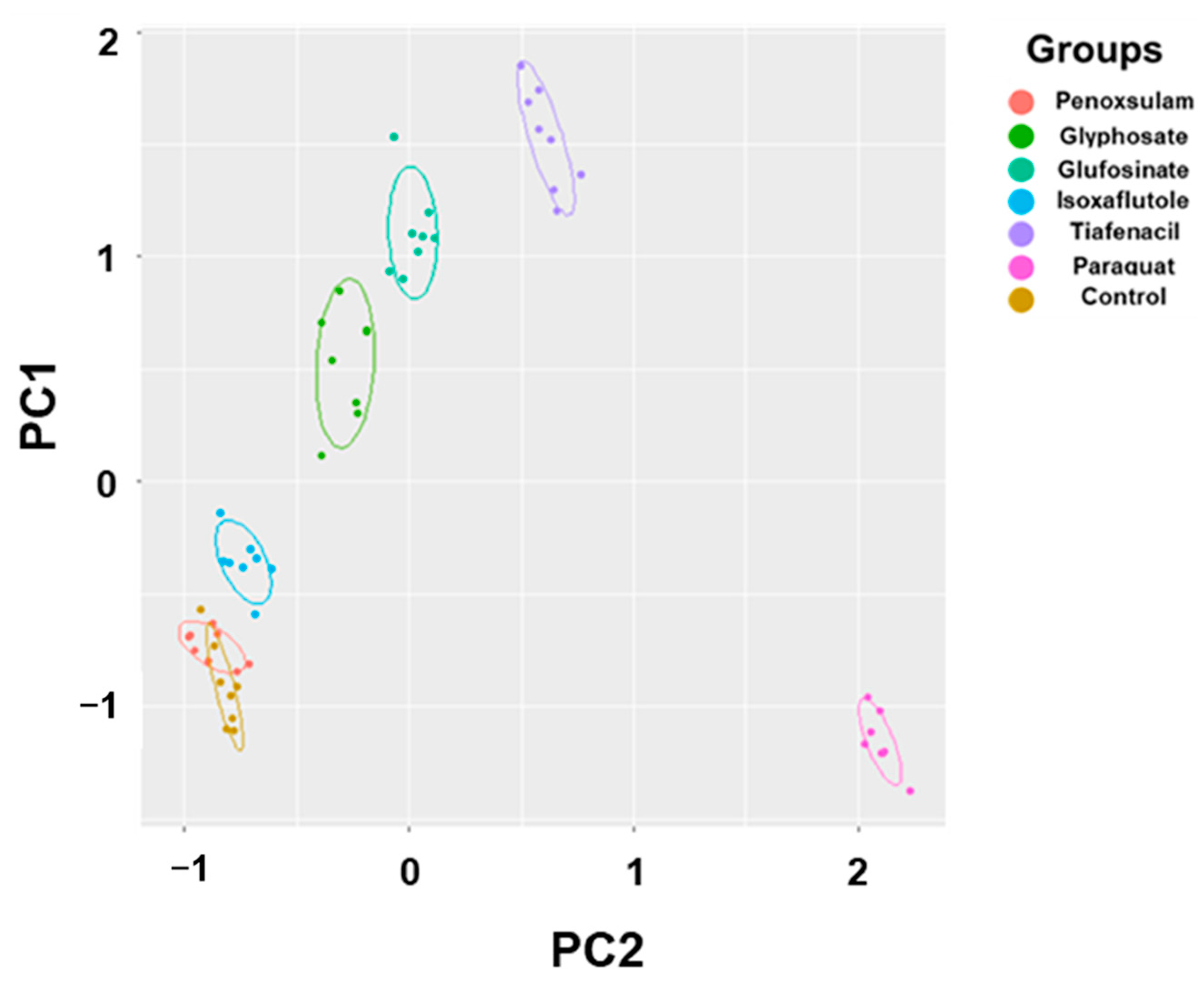
3.6. Principal Component Analysis
4. Discussion
4.1. Spectral Image Responses to Herbicides
4.2. Diagnosis of Herbicide Mode of Action Based on Spectral Response Data
4.3. High-Throughput Screening of Herbicides Using Multi-Well Plates Assay Combined with Plant Spectral Image Analysis
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Olofsdotter, M.; Olesen, A.; Andersen, S.; Streibig, J. A comparison of herbicide bioassays in cell cultures and whole plants. Weed Res. 1994, 34, 387–394. [Google Scholar] [CrossRef]
- Beckie, H.J.; Friesen, L.F.; Nawolsky, K.M.; Morrison, I.N. A rapid bioassay to detect trifluralin-resistant green foxtail (Setaria viridis). Weed Technol. 1990, 4, 505–508. [Google Scholar] [CrossRef]
- Zhang, C.; Lim, S.; Kim, J.; Song, J.; Yook, M.; Nah, G.; Valverde, B.; Kim, D. Quantifying herbicide dose–response and resistance in Echinochloa spp. by measuring root length in growth pouches. Can. J. Plant Sci. 2015, 95, 1181–1192. [Google Scholar] [CrossRef]
- Kim, D.-S.; Caseley, J.C.; Brain, P.; Riches, C.R.; Valverde, B.E. Rapid detection of propanil and fenoxaprop resistance in Echinochloa colona. Weed Sci. 2000, 48, 695–700. [Google Scholar] [CrossRef]
- Koger, C.H.; Shaner, D.L.; Henry, W.B.; Nadler-Hassar, T.; Thomas, W.E.; Wilcut, J.W. Assessment of two nondestructive assays for detecting glyphosate resistance in horseweed (Conyza canadensis). Weed Sci. 2005, 53, 559–566. [Google Scholar] [CrossRef]
- Shaner, D.L.; Nadler-Hassar, T.; Henry, W.B.; Koger, C.H. A rapid in vivo shikimate accumulation assay with excised leaf discs. Weed Sci. 2005, 53, 769–774. [Google Scholar] [CrossRef]
- Wu, C.; Varanasi, V.; Perez-Jones, A. A nondestructive leaf-disk assay for rapid diagnosis of weed resistance to multiple herbicides. Weed Sci. 2021, 69, 274–283. [Google Scholar] [CrossRef]
- Soukupová, J.; Smatanová, S.; Nedbal, L.; Jegorov, A. Plant response to destruxins visualized by imaging of chlorophyll fluorescence. Physiol. Plant. 2003, 118, 399–405. [Google Scholar] [CrossRef]
- Zhang, C.; Lim, S.; Kim, J.; Nah, G.; Fischer, A.; Kim, D. Leaf chlorophyll fluorescence discriminates herbicide resistance in Echinochloa species. Weed Res. 2016, 56, 424–433. [Google Scholar] [CrossRef]
- Chaerle, L.; Leinonen, I.; Jones, H.G.; Van Der Straeten, D. Monitoring and screening plant populations with combined thermal and chlorophyll fluorescence imaging. J. Exp. Bot. 2007, 58, 773–784. [Google Scholar] [CrossRef] [PubMed]
- Jones, H.G. Application of thermal imaging and infrared sensing in plant physiology and ecophysiology. In Advances in Botanical Research; Elsevier: Amsterdam, The Netherlands, 2004; Volume 41, pp. 107–163. [Google Scholar]
- Kim, J.-W.; Lee, T.-Y.; Nah, G.; Kim, D.-S. Potential of thermal image analysis for screening salt stress-tolerant soybean (Glycine max). Plant Genet. Resour. 2014, 12, S134–S136. [Google Scholar] [CrossRef]
- Stoll, M.; Jones, H.G. Thermal imaging as a viable tool for monitoring plant stress. OENO One 2007, 41, 77–84. [Google Scholar] [CrossRef]
- Liu, P.; Tian, Z.; Hao, N.; Bachman, H.; Zhang, P.; Hu, J.; Huang, T.J. Acoustofluidic multi-well plates for enrichment of micro/nano particles and cells. Lab Chip 2020, 20, 3399–3409. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, R.P.; Oxborough, K.; Pallett, K.E.; Baker, N.R. Rapid, noninvasive screening for perturbations of metabolism and plant growth using chlorophyll fluorescence imaging. Plant Physiol. 2003, 132, 485–493. [Google Scholar] [CrossRef] [PubMed]
- De Diego, N.; Fürst, T.; Humplík, J.F.; Ugena, L.; Podlešáková, K.; Spíchal, L. An automated method for high-throughput screening of Arabidopsis rosette growth in multi-well plates and its validation in stress conditions. Front. Plant Sci. 2017, 8, 1702. [Google Scholar] [CrossRef] [PubMed]
- Wilkinson, A.D.; Collier, C.J.; Flores, F.; Mercurio, P.; O’Brien, J.; Ralph, P.J.; Negri, A.P. A miniature bioassay for testing the acute phytotoxicity of photosystem II herbicides on seagrass. PLoS ONE 2015, 10, e0117541. [Google Scholar] [CrossRef] [PubMed]
- Classic Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Noh, T.K.; Kim, D.S. Weed research using plant image science. Weed Turfgrass Sci. 2018, 7, 285–296. [Google Scholar]
- Ali, M.M.; Bachik, N.A.; Muhadi, N.A.; Yusof, T.N.T.; Gomes, C. Non-destructive techniques of detecting plant diseases: A review. Physiol. Mol. Plant Pathol. 2019, 108, 101426. [Google Scholar] [CrossRef]
- Asaari, M.S.M.; Mertens, S.; Verbraeken, L.; Dhondt, S.; Inzé, D.; Bikram, K.; Scheunders, P. Non-destructive analysis of plant physiological traits using hyperspectral imaging: A case study on drought stress. Comput. Electron. Agric. 2022, 195, 106806. [Google Scholar] [CrossRef]
- Patane, P.; Vibhute, A. Chlorophyll and nitrogen estimation techniques: A Review. Int. J. Eng. Res. Rev. 2014, 2, 33–41. [Google Scholar]
- Padmavathi, K.; Thangadurai, K. Implementation of RGB and grayscale images in plant leaves disease detection–comparative study. Indian J. Sci. Technol. 2016, 9, 1–6. [Google Scholar] [CrossRef]
- MacDonald, G.; Netherland, M. Herbicide assays for predicting or determining plant responses in aquatic systems. J. Aquat. Plant Manag. 2018, 56, 67–73. [Google Scholar]
- Blokhina, O.; Virolainen, E.; Fagerstedt, K.V. Antioxidants, oxidative damage and oxygen deprivation stress: A review. Ann. Bot. 2003, 91, 179–194. [Google Scholar] [CrossRef]
- Römer, S.; Fraser, P.D. Recent advances in carotenoid biosynthesis, regulation and manipulation. Planta 2005, 221, 305–308. [Google Scholar] [CrossRef]
- Sumesh, K.; Ninsawat, S.; Som-ard, J. Integration of RGB-based vegetation index, crop surface model and object-based image analysis approach for sugarcane yield estimation using unmanned aerial vehicle. Comput. Electron. Agric. 2021, 180, 105903. [Google Scholar]
- Quan, L.; Chen, K.; Chen, T.; Li, H.; Li, W.; Cheng, T.; Xia, F.; Lou, Z.; Geng, T.; Sun, D. Monitoring weed mechanical and chemical damage stress based on chlorophyll fluorescence imaging. Front. Plant Sci. 2023, 14, 1188981. [Google Scholar] [CrossRef] [PubMed]
- Turgeon, R.; Wolf, S. Phloem transport: Cellular pathways and molecular trafficking. Annu. Rev. Plant Biol. 2009, 60, 207–221. [Google Scholar] [CrossRef]
- Pineda, M.; Barón, M.; Pérez-Bueno, M.-L. Thermal imaging for plant stress detection and phenotyping. Remote Sens. 2020, 13, 68. [Google Scholar] [CrossRef]
- Leinonen, I.; Jones, H.G. Combining thermal and visible imagery for estimating canopy temperature and identifying plant stress. J. Exp. Bot. 2004, 55, 1423–1431. [Google Scholar] [CrossRef] [PubMed]
- Ishimwe, R.; Abutaleb, K.; Ahmed, F. Applications of thermal imaging in agriculture—A review. Adv. Remote Sens. 2014, 3, 128. [Google Scholar] [CrossRef]
- Vítek, P.; Veselá, B.; Klem, K. Spatial and temporal variability of plant leaf responses cascade after PSII inhibition: Raman, chlorophyll fluorescence and infrared thermal imaging. Sensors 2020, 20, 1015. [Google Scholar] [CrossRef] [PubMed]
- Gao, Z.; Luo, Z.; Zhang, W.; Lv, Z.; Xu, Y. Deep learning application in plant stress imaging: A review. AgriEngineering 2020, 2, 29. [Google Scholar] [CrossRef]
- Ghosal, S.; Blystone, D.; Singh, A.K.; Ganapathysubramanian, B.; Singh, A.; Sarkar, S. An explainable deep machine vision framework for plant stress phenotyping. Proc. Natl. Acad. Sci. USA 2018, 115, 4613–4618. [Google Scholar] [CrossRef]




| Herbicide | Mode of Action 1 | Dose (g a.i. ha−1) | Product Name | Formulation 2 | Manufacturer | |
|---|---|---|---|---|---|---|
| Recommended | Tested | |||||
| Paraquat | PSI inhibitor | 500 | 125 | Gramoxone | SL | Farmhannong Ltd., Seoul, Republic of Korea |
| Tiafenacil | PPO inhibitor | 160 | 40 | Terrad’or | ME | Farmhannong Ltd., Seoul, Republic of Korea |
| Penoxsulam | ALS inhibitor | 120 | 30 | Salchodaechup | SC | Hankooksamgong Ltd., Seoul, Republic of Korea |
| Isoxaflutole | HPPD inhibitor | 200 | 50 | Merlin | WG | BASF, Lutwigshafen, Germany |
| Glufosinate | GS inhibitor | 1440 | 360 | Basta | SL | Bayer Crop Science Korea, Seoul, Republic of Korea |
| Glyphosate | EPSPS inhibitor | 3690 | 922.5 | Keunsami | SL | Farmhannong Ltd., Seoul, Republic of Korea |
| Spectral Parameter | Source of Variation | F Value | p Value | p Value Summary | |
|---|---|---|---|---|---|
| RGB | mNDI | Herbicide | 1063.089 | <0.0001 | **** |
| Time | 606.495 | <0.0001 | **** | ||
| Herbicide × Time | 95.708 | <0.0001 | **** | ||
| ExG | Herbicide | 1816.526 | <0.0001 | **** | |
| Time | 1185.414 | <0.0001 | **** | ||
| Herbicide × Time | 166.365 | <0.0001 | **** | ||
| CF | Fv/Fm | Herbicide | 8354.295 | <0.0001 | **** |
| Time | 1894.120 | <0.0001 | **** | ||
| Herbicide × Time | 341.633 | <0.0001 | **** | ||
| ΦPSII | Herbicide | 859.989 | <0.0001 | **** | |
| Time | 653.637 | <0.0001 | **** | ||
| Herbicide × Time | 73.445 | <0.0001 | **** | ||
| Fd/Fm | Herbicide | 5966.657 | <0.0001 | **** | |
| Time | 661.369 | <0.0001 | **** | ||
| Herbicide × Time | 238.078 | <0.0001 | **** | ||
| IR | Temperature difference | Herbicide | 11.923 | <0.0001 | **** |
| Time | 37.174 | <0.0001 | **** | ||
| Herbicide × Time | 15.847 | <0.0001 | **** | ||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jeong, S.-M.; Noh, T.-K.; Kim, D.-S. Herbicide Bioassay Using a Multi-Well Plate and Plant Spectral Image Analysis. Sensors 2024, 24, 919. https://doi.org/10.3390/s24030919
Jeong S-M, Noh T-K, Kim D-S. Herbicide Bioassay Using a Multi-Well Plate and Plant Spectral Image Analysis. Sensors. 2024; 24(3):919. https://doi.org/10.3390/s24030919
Chicago/Turabian StyleJeong, Seung-Min, Tae-Kyeong Noh, and Do-Soon Kim. 2024. "Herbicide Bioassay Using a Multi-Well Plate and Plant Spectral Image Analysis" Sensors 24, no. 3: 919. https://doi.org/10.3390/s24030919
APA StyleJeong, S.-M., Noh, T.-K., & Kim, D.-S. (2024). Herbicide Bioassay Using a Multi-Well Plate and Plant Spectral Image Analysis. Sensors, 24(3), 919. https://doi.org/10.3390/s24030919






