The Role of Biomarkers in Monitoring Chronic Fatigue Among Male Professional Team Athletes: A Systematic Review
Abstract
1. Introduction
2. Materials and Methods
2.1. Design
2.2. Search Strategy
2.3. Inclusion and Exclusion Criteria
2.4. Screening Strategy and Study Selection
2.5. Quality of Studies
3. Results
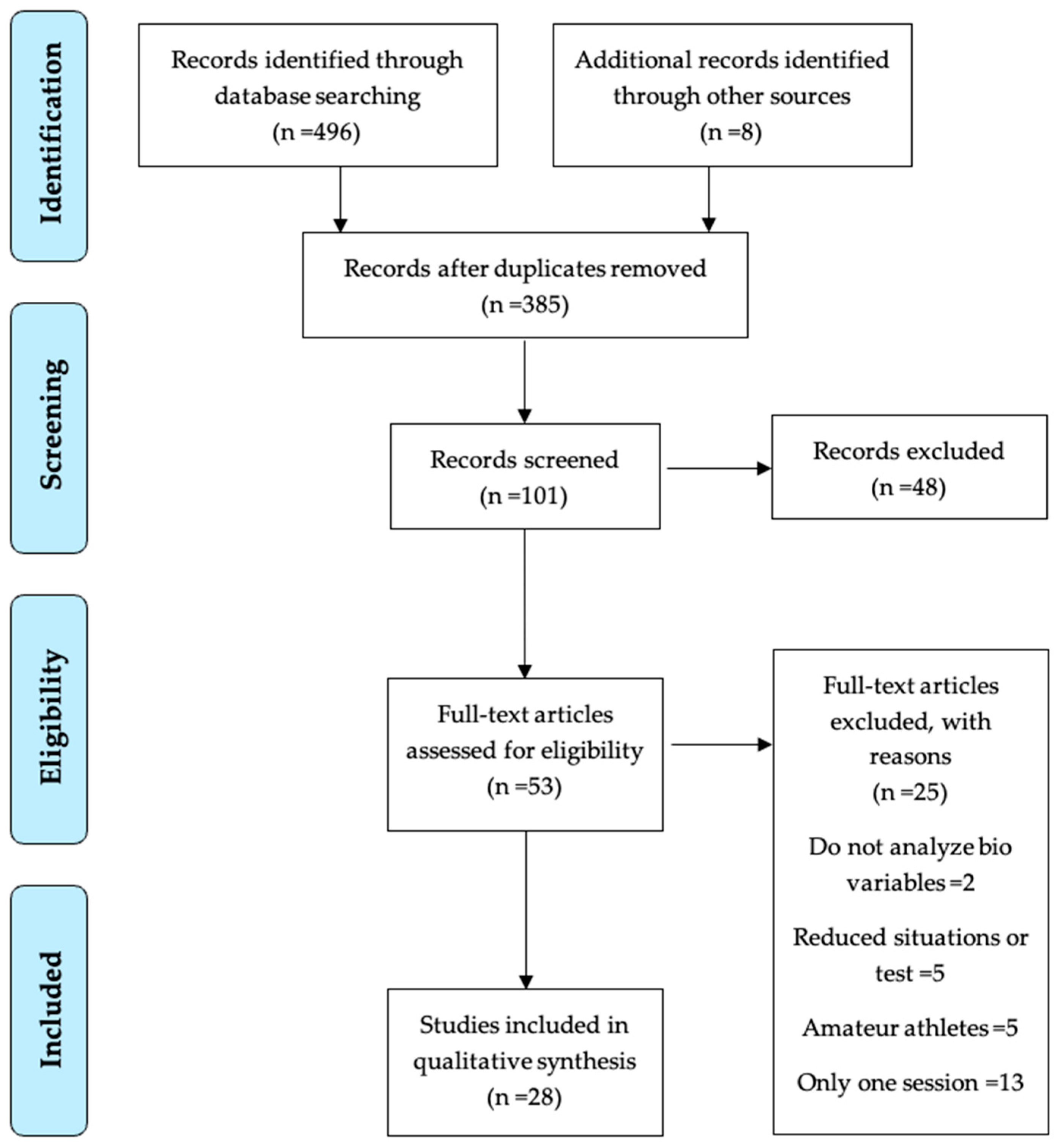
3.1. Identification and Selection of Studies
3.2. Methodological Quality
3.3. Characteristics of the Selected Studies
4. Discussion
4.1. Hormonal Markers
4.2. Muscular Damage Markers
4.3. Immunological Markers
4.4. Inflammatory Markers and Oxidative Stress Markers
4.5. Sex Differences in Chronic Fatigue Monitoring
4.6. Limitations and Future Research Directions
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Borresen, J.; Lambert, M.I. The Quantification of Training Load, the Training Response and the Effect on Performance. Sports Med. 2009, 39, 779–795. [Google Scholar] [CrossRef] [PubMed]
- Halson, S.L. Monitoring Training Load to Understand Fatigue in Athletes. Sports Med. 2014, 44 (Suppl. 2), S139–S147. [Google Scholar] [CrossRef] [PubMed]
- Jaspers, A.; Brink, M.S.; Probst, S.G.M.; Frencken, W.G.P.; Helsen, W.F. Relationships Between Training Load Indicators and Training Outcomes in Professional Soccer. Sports Med. 2017, 47, 533–544. [Google Scholar] [CrossRef] [PubMed]
- Gamble, P. Periodization of Training for Team Sports Athletes. Strength Cond. J. 2006, 28, 56–66. [Google Scholar] [CrossRef]
- Issurin, V.B. New Horizons for the Methodology and Physiology of Training Periodization. Sports Med. 2010, 40, 189–206. [Google Scholar] [CrossRef]
- Impellizzeri, F.M.; Rampinini, E.; Coutts, A.J.; Sassi, A.; Marcora, S.M. Use of RPE-Based Training Load in Soccer. Med. Sci. Sports Exerc. 2004, 36, 1042–1047. [Google Scholar] [CrossRef]
- Mroczek, D.; Januszkiewicz, A.; Kawczyński, A.S.; Borysiuk, Z.; Chmura, J. Analysis of Male Volleyball Players’ Motor Activities during a Top Level Match. J. Strength Cond. Res. 2014, 28, 2297–2305. [Google Scholar] [CrossRef]
- Aubert, A.E.; Seps, B.; Beckers, F. Heart Rate Variability in Athletes. Sports Med. 2003, 33, 889–919. [Google Scholar] [CrossRef]
- Jiménez Morgan, S.; Molina Mora, J.A. Effect of Heart Rate Variability Biofeedback on Sport Performance, a Systematic Review. Appl. Psychophysiol. Biofeedback 2017, 42, 235–245. [Google Scholar] [CrossRef]
- Haddad, M.; Stylianides, G.; Djaoui, L.; Dellal, A.; Chamari, K. Session-RPE Method for Training Load Monitoring: Validity, Ecological Usefulness, and Influencing Factors. Front. Neurosci. 2017, 11, 612. [Google Scholar] [CrossRef]
- García-Ramos, A.; Torrejón, A.; Feriche, B.; Morales-Artacho, A.J.; Pérez-Castilla, A.; Padial, P.; Haff, G.G. Prediction of the Maximum Number of Repetitions and Repetitions in Reserve From Barbell Velocity. Int. J. Sports Physiol. Perform. 2018, 13, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Haff, G.G.; Nimphius, S. Training Principles for Power. Strength Cond. J. 2012, 34, 2. [Google Scholar] [CrossRef]
- Sánchez-Medina, L.; González-Badillo, J.J. Velocity Loss as an Indicator of Neuromuscular Fatigue during Resistance Training. Med. Sci. Sports Exerc. 2011, 43, 1725–1734. [Google Scholar] [CrossRef] [PubMed]
- Ehrmann, F.E.; Duncan, C.S.; Sindhusake, D.; Franzsen, W.N.; Greene, D.A. GPS and Injury Prevention in Professional Soccer. J. Strength Cond. Res. 2016, 30, 360–367. [Google Scholar] [CrossRef]
- Díaz-Soto, F.J.; Rico-González, M.; Palucci Vieira, L.H.; Clemente, F.M.; Nobari, H.; Pino-Ortega, J. A Systematic Review of Velocity and Accelerometer Thresholds in Soccer. Int. J. Sports Sci. Coach. 2023, 18, 1768–1781. [Google Scholar] [CrossRef]
- Rowsell, G.J.; Coutts, A.J.; Reaburn, P.; Hill-Haas, S. Effects of Cold-Water Immersion on Physical Performance between Successive Matches in High-Performance Junior Male Soccer Players. J. Sports Sci. 2009, 27, 565–573. [Google Scholar] [CrossRef]
- Coutts, A.; Reaburn, P.; Piva, T.J.; Murphy, A. Changes in Selected Biochemical, Muscular Strength, Power, and Endurance Measures during Deliberate Overreaching and Tapering in Rugby League Players. Int. J. Sports Med. 2007, 28, 116–124. [Google Scholar] [CrossRef]
- Córdova, A.; Sureda, A.; Tur, J.A.; Pons, A. Immune Response to Exercise in Elite Sportsmen during the Competitive Season. J. Physiol. Biochem. 2010, 66, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Kelly, V.G.; Coutts, A.J. Planning and Monitoring Training Loads during the Competition Phase in Team Sports. Strength Cond. J. 2007, 29, 32–37. [Google Scholar] [CrossRef]
- Miloski, B.; de Freitas, V.H.; Nakamura, F.Y.; de A Nogueira, F.C.; Bara-Filho, M.G. Seasonal Training Load Distribution of Professional Futsal Players: Effects on Physical Fitness, Muscle Damage and Hormonal Status. J. Strength Cond. Res. 2016, 30, 1525–1533. [Google Scholar] [CrossRef]
- Moreira, A.; Freitas, C.G.; Nakamura, F.Y.; Drago, G.; Drago, M.; Aoki, M.S. Effect of Match Importance on Salivary Cortisol and Immunoglobulin A Responses in Elite Young Volleyball Players. J. Strength Cond. Res. 2013, 27, 202–207. [Google Scholar] [CrossRef] [PubMed]
- Palao, J.M.; Manzanares, P.; Valadés, D. Anthropometric, Physical, and Age Differences by the Player Position and the Performance Level in Volleyball. J. Hum. Kinet. 2014, 44, 223–236. [Google Scholar] [CrossRef] [PubMed]
- Freitas, V.H.; Nakamura, F.Y.; Miloski, B.; Samulski, D.; Bara-Filho, M.G. Sensitivity of Physiological and Psychological Markers to Training Load Intensification in Volleyball Players. J. Sports Sci. Med. 2014, 13, 571–579. [Google Scholar] [PubMed]
- Saw, A.E.; Main, L.C.; Gastin, P.B. Monitoring the Athlete Training Response: Subjective Self-Reported Measures Trump Commonly Used Objective Measures: A Systematic Review. Br. J. Sports Med. 2016, 50, 281–291. [Google Scholar] [CrossRef] [PubMed]
- Filaire, E.; Bernain, X.; Sagnol, M.; Lac, G. Preliminary Results on Mood State, Salivary Testosterone: Cortisol Ratio and Team Performance in a Professional Soccer Team. Eur. J. Appl. Physiol. 2001, 86, 179–184. [Google Scholar] [CrossRef]
- Fragala, M.S.; Kraemer, W.J.; Denegar, C.R.; Maresh, C.M.; Mastro, A.M.; Volek, J.S. Neuroendocrine-Immune Interactions and Responses to Exercise. Sports Med. 2011, 41, 621–639. [Google Scholar] [CrossRef]
- Maso, F.; Lac, G.; Filaire, E.; Michaux, O.; Robert, A. Salivary Testosterone and Cortisol in Rugby Players: Correlation with Psychological Overtraining Items. Br. J. Sports Med. 2004, 38, 260–263. [Google Scholar] [CrossRef]
- Papacosta, E.; Nassis, G.P. Saliva as a Tool for Monitoring Steroid, Peptide and Immune Markers in Sport and Exercise Science. J. Sci. Med. Sport 2011, 14, 424–434. [Google Scholar] [CrossRef]
- Owen, A.L.; Wong, D.P.; Dunlop, G.; Groussard, C.; Kebsi, W.; Dellal, A.; Morgans, R.; Zouhal, H. High-Intensity Training and Salivary Immunoglobulin A Responses in Professional Top-Level Soccer Players: Effect of Training Intensity. J. Strength Cond. Res. 2016, 30, 2460–2469. [Google Scholar] [CrossRef]
- McLean, B.D.; Coutts, A.J.; Kelly, V.; McGuigan, M.R.; Cormack, S.J. Neuromuscular, Endocrine, and Perceptual Fatigue Responses During Different Length Between-Match Microcycles in Professional Rugby League Players. Int. J. Sports Physiol. Perform. 2010, 5, 367–383. [Google Scholar] [CrossRef]
- Moreira, A.; Mortatti, A.L.; Arruda, A.F.S.; Freitas, C.G.; de Arruda, M.; Aoki, M.S. Salivary IgA Response and Upper Respiratory Tract Infection Symptoms during a 21-Week Competitive Season in Young Soccer Players. J. Strength Cond. Res. 2014, 28, 467–473. [Google Scholar] [CrossRef] [PubMed]
- Dunbar, J.; Armitage, M.; Jehanli, A.; Browne, A. Mucosal Immunity and Self-Reported Upper Respiratory Symptoms in a Cohort of Premier League Academy Soccer Players. Int. J. Exerc. Sci. Conf. Proc. 2013, 10, 45. [Google Scholar]
- Clemente, F.M.; Mendes, B.; Bredt, S.d.G.T.; Praça, G.M.; Silvério, A.; Carriço, S.; Duarte, E. Perceived Training Load, Muscle Soreness, Stress, Fatigue, and Sleep Quality in Professional Basketball: A Full Season Study. J. Hum. Kinet. 2019, 67, 199–207. [Google Scholar] [CrossRef]
- Hagstrom, A.D.; Shorter, K.A. Creatine Kinase, Neuromuscular Fatigue, and the Contact Codes of Football: A Systematic Review and Meta-Analysis of Pre- and Post-Match Differences. Eur. J. Sport Sci. 2018, 18, 1234–1244. [Google Scholar] [CrossRef] [PubMed]
- Miguel, M.; Oliveira, R.; Loureiro, N.; García-Rubio, J.; Ibáñez, S.J. Load Measures in Training/Match Monitoring in Soccer: A Systematic Review. Int. J. Environ. Res. Public. Health 2021, 18, 2721. [Google Scholar] [CrossRef]
- Teixeira, J.E.; Forte, P.; Ferraz, R.; Leal, M.; Ribeiro, J.; Silva, A.J.; Barbosa, T.M.; Monteiro, A.M. Monitoring Accumulated Training and Match Load in Football: A Systematic Review. Int. J. Environ. Res. Public. Health 2021, 18, 3906. [Google Scholar] [CrossRef]
- Moreno-Villanueva, A.; Rico-González, M.; Pino-Ortega, J. Monitoring of Internal and External Loads to Interpret Acute and Chronic Fatigue in Indoor Team Sports: A Systematic Review. Int. J. Sports Sci. Coach. 2022, 17, 1531–1552. [Google Scholar] [CrossRef]
- Doeven, S.H.; Brink, M.S.; Kosse, S.J.; Lemmink, K.A.P.M. Postmatch Recovery of Physical Performance and Biochemical Markers in Team Ball Sports: A Systematic Review. BMJ Open Sport Exerc. Med. 2018, 4, e000264. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.A.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Healthcare Interventions: Explanation and Elaboration. BMJ 2009, 339, b2700. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 Statement: An Updated Guideline for Reporting Systematic Reviews. BMJ 2021, 372, n71. [Google Scholar] [CrossRef]
- Rico-González, M.; Pino-Ortega, J.; Clemente, F.; Arcos, A.L. Guidelines for Performing Systematic Reviews in Sports Science. Biol. Sport 2021, 39, 463–471. [Google Scholar] [CrossRef] [PubMed]
- Slim, K.; Nini, E.; Forestier, D.; Kwiatkowski, F.; Panis, Y.; Chipponi, J. Methodological Index for Non-Randomized Studies (Minors): Development and Validation of a New Instrument. ANZ J. Surg. 2003, 73, 712–716. [Google Scholar] [CrossRef]
- Kim, S.Y.; Park, J.E.; Lee, Y.J.; Seo, H.-J.; Sheen, S.-S.; Hahn, S.; Jang, B.-H.; Son, H.-J. Testing a Tool for Assessing the Risk of Bias for Nonrandomized Studies Showed Moderate Reliability and Promising Validity. J. Clin. Epidemiol. 2013, 66, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Barcelos, R.P.; Tocchetto, G.L.; Lima, F.D.; Stefanello, S.T.; Rodrigues, H.F.M.; Sangoi, M.B.; Moresco, R.N.; Royes, L.F.F.; Soares, F.A.A.; Bresciani, G. Functional and Biochemical Adaptations of Elite Level Futsal Players from Brazil along a Training Season. Medicina 2017, 53, 285–293. [Google Scholar] [CrossRef]
- Birdsey, L.P.; Weston, M.; Russell, M.; Johnston, M.; Cook, C.J.; Kilduff, L.P. Neuromuscular, Physiological and Perceptual Responses to an Elite Netball Tournament. J. Sports Sci. 2019, 37, 2169–2174. [Google Scholar] [CrossRef]
- Botonis, P.G.; Toubekis, A.G. Intensified Olympic Preparation: Sleep and Training-Related Hormonal and Immune Responses in Water Polo. Int. J. Sports Physiol. Perform. 2023, 18, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Bresciani, G.; Cuevas, M.J.; Garatachea, N.; Molinero, O.; Almar, M.; De Paz, J.A.; Márquez, S.; González-Gallego, J. Monitoring Biological and Psychological Measures throughout an Entire Season in Male Handball Players. Eur. J. Sport Sci. 2010, 10, 377–384. [Google Scholar] [CrossRef]
- Coad, S.; Gray, B.; Wehbe, G.; McLellan, C. Physical Demands and Salivary Immunoglobulin A Responses of Elite Australian Rules Football Athletes to Match Play. Int. J. Sports Physiol. Perform. 2015, 10, 613–617. [Google Scholar] [CrossRef]
- Coad, S.; Gray, B.; McLellan, C. Seasonal Analysis of Mucosal Immunological Function and Physical Demands in Professional Australian Rules Footballers. Int. J. Sports Physiol. Perform. 2016, 11, 574–580. [Google Scholar] [CrossRef]
- Cormack, S.J.; Newton, R.U.; McGuigan, M.R.; Cormie, P. Neuromuscular and Endocrine Responses of Elite Players during an Australian Rules Football Season. Int. J. Sports Physiol. Perform. 2008, 3, 439–453. [Google Scholar] [CrossRef]
- Cunniffe, B.; Griffiths, H.; Proctor, W.; Davies, B.; Baker, J.S.; Jones, K.P. Mucosal Immunity and Illness Incidence in Elite Rugby Union Players across a Season. Med. Sci. Sports Exerc. 2011, 43, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Horta, T.A.; Bara Filho, M.G.; Coimbra, D.R.; Miranda, R.; Werneck, F.Z. Training Load, Physical Performance, Biochemical Markers, and Psychological Stress during a Short Preparatory Period in Brazilian Elite Male Volleyball Players. J. Strength Cond. Res. 2019, 33, 3392–3399. [Google Scholar] [CrossRef] [PubMed]
- Kamarauskas, P.; Lukonaitienė, I.; Kvedaras, M.; Venckūnas, T.; Conte, D. Relationships between Weekly Changes in Salivary Hormonal Responses and Load Measures during the Pre-Season Phase in Professional Male Basketball Players. Biol. Sport 2023, 40, 353–358. [Google Scholar] [CrossRef]
- Lindsay, A.; Lewis, J.G.; Scarrott, C.; Gill, N.; Gieseg, S.P.; Draper, N. Assessing the Effectiveness of Selected Biomarkers in the Acute and Cumulative Physiological Stress Response in Professional Rugby Union through Non-Invasive Assessment. Int. J. Sports Med. 2015, 36, 446–454. [Google Scholar] [CrossRef]
- Marin, D.P.; Bolin, A.P.; Campoio, T.R.; Guerra, B.A.; Otton, R. Oxidative Stress and Antioxidant Status Response of Handball Athletes: Implications for Sport Training Monitoring. Int. Immunopharmacol. 2013, 17, 462–470. [Google Scholar] [CrossRef]
- Martínez, A.C.; Seco Calvo, J.; Tur Marí, J.A.; Abecia Inchaurregui, L.C.; Orella, E.E.; Biescas, A.P. Testosterone and Cortisol Changes in Professional Basketball Players through a Season Competition. J. Strength Cond. Res. 2010, 24, 1102–1108. [Google Scholar] [CrossRef] [PubMed]
- Mohr, M.; Draganidis, D.; Chatzinikolaou, A.; Barbero-Álvarez, J.C.; Castagna, C.; Douroudos, I.; Avloniti, A.; Margeli, A.; Papassotiriou, I.; Flouris, A.D.; et al. Muscle Damage, Inflammatory, Immune and Performance Responses to Three Football Games in 1 Week in Competitive Male Players. Eur. J. Appl. Physiol. 2016, 116, 179–193. [Google Scholar] [CrossRef]
- Moreira, A.; Arsati, F.; de Oliveira Lima Arsati, Y.B.; da Silva, D.A.; de Araújo, V.C. Salivary Cortisol in Top-Level Professional Soccer Players. Eur. J. Appl. Physiol. 2009, 106, 25–30. [Google Scholar] [CrossRef]
- Moreira, A.; de Moura, N.R.; Coutts, A.; Costa, E.C.; Kempton, T.; Aoki, M.S. Monitoring Internal Training Load and Mucosal Immune Responses in Futsal Athletes. J. Strength Cond. Res. 2013, 27, 1253–1259. [Google Scholar] [CrossRef]
- Rowell, A.E.; Aughey, R.J.; Hopkins, W.G.; Esmaeili, A.; Lazarus, B.H.; Cormack, S.J. Effects of Training and Competition Load on Neuromuscular Recovery, Testosterone, Cortisol, and Match Performance During a Season of Professional Football. Front. Physiol. 2018, 9, 668. [Google Scholar] [CrossRef]
- Saidi, K.; Zouhal, H.; Boullosa, D.; Dupont, G.; Hackney, A.C.; Bideau, B.; Granacher, U.; Ben Abderrahman, A. Biochemical Markers and Wellness Status During a Congested Match Play Period in Elite Soccer Players. Int. J. Sports Physiol. Perform. 2022, 17, 605–620. [Google Scholar] [CrossRef] [PubMed]
- Schelling, X.; Calleja-González, J.; Terrados Cepeda, N. Hormonal Analysis in Elite Basketball during a Season. Rev. Psicol. Deporte 2009, 18, 0363–0367. [Google Scholar]
- Schelling, X.; Calleja-González, J.; Torres-Ronda, L.; Terrados, N. Using Testosterone and Cortisol as Biomarker for Training Individualization in Elite Basketball: A 4-Year Follow-up Study. J. Strength Cond. Res. 2015, 29, 368–378. [Google Scholar] [CrossRef] [PubMed]
- Souglis, A.; Bogdanis, G.C.; Giannopoulou, I.; Papadopoulos, C.; Apostolidis, N. Comparison of Inflammatory Responses and Muscle Damage Indices Following a Soccer, Basketball, Volleyball and Handball Game at an Elite Competitive Level. Res. Sports Med. Print 2015, 23, 59–72. [Google Scholar] [CrossRef]
- Spanidis, Y.; Goutzourelas, N.; Stagos, D.; Mpesios, A.; Priftis, A.; Bar-Or, D.; Spandidos, D.A.; Tsatsakis, A.M.; Leon, G.; Kouretas, D. Variations in Oxidative Stress Markers in Elite Basketball Players at the Beginning and End of a Season. Exp. Ther. Med. 2016, 11, 147–153. [Google Scholar] [CrossRef][Green Version]
- Springham, M.; Williams, S.; Waldron, M.; Strudwick, A.J.; Mclellan, C.; Newton, R.U. Salivary Immunoendocrine and Self-Report Monitoring Profiles across an Elite-Level Professional Football Season. Med. Sci. Sports Exerc. 2021, 53, 918–927. [Google Scholar] [CrossRef]
- Talaee, M.; Nazem, F.; Taherabadi, S.J.; Sajadi, S. Effects of Six Weeks Combined Training Program on Hematological Parameters in Elite Basketball Players. Ann. Appl. Sport Sci. 2017, 5, 15–23. [Google Scholar] [CrossRef]
- Tiernan, C.; Lyons, M.; Comyns, T.; Nevill, A.M.; Warrington, G. Salivary IgA as a Predictor of Upper Respiratory Tract Infections and Relationship to Training Load in Elite Rugby Union Players. J. Strength Cond. Res. 2020, 34, 782–790. [Google Scholar] [CrossRef]
- Twist, C.; Waldron, M.; Highton, J.; Burt, D.; Daniels, M. Neuromuscular, Biochemical and Perceptual Post-Match Fatigue in Professional Rugby League Forwards and Backs. J. Sports Sci. 2012, 30, 359–367. [Google Scholar] [CrossRef]
- Cunniffe, B.; Griffiths, H.; Proctor, W.; Jones, K.P.; Baker, J.S.; Davies, B. Illness Monitoring in Team Sports Using a Web-Based Training Diary. Clin. J. Sport Med. 2009, 19, 476. [Google Scholar] [CrossRef]
- Cunniffe, B.; Hore, A.J.; Whitcombe, D.M.; Jones, K.P.; Baker, J.S.; Davies, B. Time Course of Changes in Immuneoendocrine Markers Following an International Rugby Game. Eur. J. Appl. Physiol. 2010, 108, 113–122. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, U.; Mester, J. Training and Overtraining Markers in Selected Sport Events. Med. Sci. Sports Exerc. 2000, 32, 209–215. [Google Scholar] [CrossRef] [PubMed]
- Hoffman, J.R.; Kang, J.I.E.; Ratamess, N.A.; Faigenbaum, A.D. Biochemical and Hormonal Responses during an Intercollegiate Football Season. Med. Sci. Sports Exerc. 2005, 37, 1237–1241. [Google Scholar] [CrossRef] [PubMed]
- Jones, M.R.; West, D.J.; Harrington, B.J.; Cook, C.J.; Bracken, R.M.; Shearer, D.A.; Kilduff, L.P. Match Play Performance Characteristics That Predict Post-Match Creatine Kinase Responses in Professional Rugby Union Players. BMC Sports Sci. Med. Rehabil. 2014, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- McLellan, C.P.; Lovell, D.I.; Gass, G.C. Creatine Kinase and Endocrine Responses of Elite Players Pre, during, and Post Rugby League Match Play. J. Strength Cond. Res. 2010, 24, 2908–2919. [Google Scholar] [CrossRef]
- Roe, G.; Darrall-Jones, J.; Till, K.; Phibbs, P.; Read, D.; Weakley, J.; Jones, B. Between-Days Reliability and Sensitivity of Common Fatigue Measures in Rugby Players. Int. J. Sports Physiol. Perform. 2016, 11, 581–586. [Google Scholar] [CrossRef]
- Twist, C.; Highton, J. Monitoring Fatigue and Recovery in Rugby League Players. Int. J. Sports Physiol. Perform. 2013, 8, 467–474. [Google Scholar] [CrossRef]
- Gutenbrunner, C. Circadian Variations of the Serum Creatine Kinase Level—A Masking Effect? Chronobiol. Int. 2000, 17, 583–590. [Google Scholar] [CrossRef]
- Rico-González, M.; Clemente, F.M.; Oliveira, R.; Bustamante-Hernández, N.; Pino-Ortega, J. Part I: Relationship among Training Load Management, Salivary Immunoglobulin A, and Upper Respiratory Tract Infection in Team Sport: A Systematic Review. Healthcare 2021, 9, 366. [Google Scholar] [CrossRef]
- Keaney, L.C.; Kilding, A.E.; Merien, F.; Dulson, D.K. The Impact of Sport Related Stressors on Immunity and Illness Risk in Team-Sport Athletes. J. Sci. Med. Sport 2018, 21, 1192–1199. [Google Scholar] [CrossRef]
- Gleeson, M.; Bishop, N.; Oliveira, M.; McCauley, T.; Tauler, P.; Muhamad, A.S. Respiratory Infection Risk in Athletes: Association with Antigen-Stimulated IL-10 Production and Salivary IgA Secretion. Scand. J. Med. Sci. Sports 2012, 22, 410–417. [Google Scholar] [CrossRef] [PubMed]
- Mortatti, A.L.; Moreira, A.; Aoki, M.S.; Crewther, B.T.; Castagna, C.; de Arruda, A.F.S.; Filho, J.M. Effect of Competition on Salivary Cortisol, Immunoglobulin A, and Upper Respiratory Tract Infections in Elite Young Soccer Players. J. Strength Cond. Res. 2012, 26, 1396–1401. [Google Scholar] [CrossRef] [PubMed]
- Novas, A.M.P.; Rowbottom, D.G.; Jenkins, D.G. Tennis, Incidence of URTI and Salivary IgA. Int. J. Sports Med. 2003, 24, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Carmichael, M.A.; Thomson, R.L.; Moran, L.J.; Wycherley, T.P. The Impact of Menstrual Cycle Phase on Athletes’ Performance: A Narrative Review. Int. J. Environ. Res. Public. Health 2021, 18, 1667. [Google Scholar] [CrossRef]
- Williams, T.; Walz, E.; Lane, A.; Pebole, M.; Hackney, A. The Effect of Estrogen on Muscle Damage Biomarkers Following Prolonged Aerobic Exercise in Eumenorrheic Women. Biol. Sport 2015, 32, 193–198. [Google Scholar] [CrossRef]
- Dannecker, E.A.; Liu, Y.; Rector, R.S.; Thomas, T.R.; Fillingim, R.B.; Robinson, M.E. Sex Differences in Exercise-Induced Muscle Pain and Muscle Damage. J. Pain Off. J. Am. Pain Soc. 2012, 13, 1242–1249. [Google Scholar] [CrossRef]
- Hunter, S.K. The Relevance of Sex Differences in Performance Fatigability. Med. Sci. Sports Exerc. 2016, 48, 2247–2256. [Google Scholar] [CrossRef]
| Study | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | Score |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Barcelos et al. (2017) [44] | 2 | 2 | 1 | 1 | 0 | 2 | 2 | 0 | 2 | 2 | 1 | 2 | 17/24 |
| Birdsey et al. (2019) [45] | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Botonis et al. (2023) [46] | 1 | 2 | 1 | 1 | 2 | 1 | 2 | 0 | 2 | 1 | 0 | 1 | 14/24 |
| Bresciani et al. (2010) [47] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 2 | 2 | 20/24 |
| Coad et al. (2015) [48] | 1 | 2 | 1 | 1 | 2 | 1 | 2 | 0 | 2 | 1 | 0 | 1 | 14/24 |
| Coad et al. (2016) [49] | 2 | 2 | 2 | 2 | 1 | 2 | 2 | - | - | - | - | 2 | 15/16 |
| Cormack et al. (2008) [50] | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Cunniffe et al. (2011) [51] | 2 | 1 | 2 | 2 | 1 | 2 | 2 | - | - | - | - | 2 | 14/16 |
| Horta et al. (2019) [52] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 1 | 1 | 18/24 |
| Kamarauskas et al. (2023) [53] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 2 | 2 | 20/24 |
| Lindsay et al. (2015) [54] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 1 | 1 | 18/24 |
| Marin et al. (2013) [55] | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 0 | 2 | 1 | 2 | 2 | 20/24 |
| Martínez et al. (2010) [56] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 1 | 1 | 18/24 |
| McLean et al. (2010) [30] | 2 | 2 | 1 | 1 | 0 | 2 | 2 | 0 | 2 | 2 | 1 | 2 | 17/24 |
| Miloski et al. (2016) [20] | 2 | 2 | 2 | 1 | 0 | 2 | 2 | - | - | - | - | 2 | 13/16 |
| Mohr et al. (2016) [57] | 1 | 2 | 2 | 1 | 2 | 1 | 2 | - | - | - | - | 1 | 12/16 |
| Moreira et al. (2009) [58] | 2 | 2 | 2 | 2 | 0 | 2 | 2 | 0 | 2 | 0 | 1 | 1 | 16/24 |
| Moreira et al. (2013) [59] | 1 | 1 | 2 | 2 | 1 | 2 | 2 | 0 | 2 | 0 | 2 | 1 | 16/24 |
| Rowell et al. (2018) [60] | 2 | 2 | 2 | 2 | 1 | 0 | 2 | - | - | - | - | 1 | 12/16 |
| Saidi et al. (2022) [61] | 2 | 2 | 2 | 2 | 1 | 2 | 2 | - | - | - | - | 2 | 15/16 |
| Schelling et al. (2009) [62] | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Schelling et al. (2015) [63] | 2 | 2 | 2 | 2 | 1 | 2 | 2 | - | - | - | - | 2 | 15/16 |
| Souglis et al. (2015) [64] | 2 | 2 | 2 | 2 | 1 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Spanidis et al. (2016) [65] | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Springham et al. (2021) [66] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | - | - | - | - | 1 | 13/16 |
| Talaee et al. (2017) [67] | 2 | 2 | 2 | 2 | 1 | 1 | 2 | 0 | 2 | 2 | 2 | 2 | 20/24 |
| Tiernan et al. (2020) [68] | 2 | 2 | 1 | 2 | 2 | 2 | 2 | - | - | - | - | 1 | 14/16 |
| Twist et al. (2012) [69] | 2 | 2 | 2 | 2 | 0 | 2 | 2 | - | - | - | - | 2 | 14/16 |
| Study | Sample | Period | Type | Test | Frequency of Tests | Results | Conclusions |
|---|---|---|---|---|---|---|---|
| Barcelos et al. (2017) [44] | 8 elite male futsal players (Age: 25.5 ± 5.4 years) | Preseason and season | Matches and training | Muscle damage markers CK and LDH. Oxidative stress markers (IMA and AOPP) | At 3 points in time: End of preseason (T1) Two weeks before Intercontinental Cup (T2) End of season (T3) | Lower values of CK (271–413 vs. 446–777 U/L) and LDH (175–232 vs. 359 441 U/L). Seasonal values of IMA and AOPP. | Biochemical markers can be useful as a means of training monitoring. A one-week fine-tuning period before the main championship (T2) seems to be successful in achieving an optimal state of recovery. |
| Birdsey et al. (2019) [45] | 11 international female netball players (Age: 25 ± 4 years; Mass: 71.8 ± 7.8 kg; Height: 1.8 ± 0.1 m) | During a three-day tournament and the following three days. | Matches | Testosterone, Cortisol, CK | CK measured in the morning of each game day and 62 h post-tournament. Cortisol and testosterone measured at similar intervals. | Cortisol: Observed a small and possibly significant decrease on the second tournament day (0.47 µg/dL ± 0.23). On the third day, a trivial, likely non-significant change (0.65 µg/dL ± 0.29). Three days post-tournament, changes were unclear (0.58 µg/dL ± 0.34). Testosterone: Registered a small and possibly significant decrease both on the second (102.9 pg/mL ± 25.9) and third days (105.4 pg/mL ± 25.3) of the tournament. Three days later, the decrease remained small and possibly significant (95.7 pg/mL ± 27.0). Creatine Kinase (CK): A very large and likely significant increase on the second (217.2 U/L ± 67.4) and third days (283.0 U/L ± 121.3) of the tournament. Three days later, changes in CK levels were unclear (141.9 U/L ± 113.0) | CK buildup suggests muscle damage during the tournament, with recovery after three days. The decrease in testosterone suggests an influence on performance and motivation. Cortisol showed an initial decrease followed by normalization, indicating adaptation to tournament stress. |
| Botonis et al. (2023) [46] | 8 international water polo players (Age: 28.6 ± 3.9 years; Body mass: 98.9 ± 11.0 kg; Stature: 190.4 ± 6.1 cm) | 16 days divided into three phases: PRE-CAMP (3 days before training camp), CAMP (5-day training camp), and POST-CAMP (8 days of congested training and competition). | Training | salivary cortisol, immunoglobulin A | Collection of salivary cortisol, immunoglobulin-A, and subjective wellness measured during PRE-CAMP, CAMP, and POST-CAMP. | In CAMP compared with PRE-CAMP sleep interruptions and salivary cortisol were higher (p < 0.01, d = 1.6, d = 1.9, respectively). In POST-CAMP, reduced workload was followed by increased sleep efficiency, reduced sleep disruptions, and moderately affected salivary cortisol; however, overall well-being remained unchanged. | Significant workload increases during a training camp induce sleep disturbances and salivary cortisol increases, which are reversed in POST-CAMP. This suggests that increased workload alongside inadequate recovery affects sleep patterns and may elevate infection risk. |
| Bresciani et al. (2010) [47] | 14 handball male (Age: 20.1 ± 2.5 years) | Preseason and season | Training | Oxidative stress markers (C-reactive protein, GSSG, GSH, and GSH/GSSG ratio) | At 5 time points: Before preseason End of preseason After 1st competition phase (CP) After 2nd CP 7 weeks post-season | Periods of high load: - ↑ GSSG (21.6–38.6 mmol/L). - ↓ GSH/GSSG ratio (18.8–28.9). Positive correlation of GSSG (r = 0.65), GSH/GSSG ratio (r = 0.63) with s-RPE. | Results show that during high-intensity training periods, handball players exhibit minor inflammation and oxidative stress. This highlights the value of closely monitoring psychological and biological markers related to inflammation, oxidative stress, and training load during the season. |
| Coad et al. (2015) [48] | 11 elite male Australian Football League athletes. (Age: 21.8 ± 2.4 years; Height: 186.9 ± 7.9 cm; Mass: 87.4 ± 7.5 kg) | throughout 3 matches during the preseason that were separated by 7 days. | Matches | salivary immunoglobulin A concentration | Saliva samples were collected across each match 24 h and 1 h pre-match and 1, 12, 36, and 60 h post-match. | Across match 3, sIgA was significantly (p < 0.01) suppressed at 2 post-match measures (12 and 36 h) compared with pre-match measures (24 and 1 h), which coincided with significantly (p < 0.01) elevated player load. | The findings indicated that an increase in player load during the match resulted in compromised post-match mucosal immunological function. |
| Coad et al. (2016) [49] | 18 elite male Australian Football League athletes. (Age: 24 ± 4.2 years; Height: 187.0 ± 7.1 cm; Mass: 87.0 ± 7.6 kg) | 16 consecutive matches in an Australian Football League premiership season. | Matches | salivary immunoglobulin A concentration | A concentration (s-IgA) measured at 36 h postmatch throughout an Australian Football League. | Significant (p < 0.05) effects compared with baseline sIgA. | Matches may delay sIgA recovery beyond 36 h post-match for full recovery and may be at higher risk of illness during the initial 36 h post-match. |
| Cormack et al. (2008) [50] | 15 elite Australian football League players (Age: 24.9 ± 2.4 years; Height: 1.87 ± 0.07 m; Weight: 88.0 ± 7.9 kg). | Before and during the 22-match season. | Matches and training | Cortisol (C) and Testosterone (T) | Initial data collected at rest approximately 36 h before the first match of the season and on 20 occasions throughout the 22-match season | Cortisol was substantially lower (up to −40 ± 14.1%, ES of −2.17 ± 0.56) than Pre in all but one comparison. Testosterone response was varied, whereas T/C increased substantially on 70% of occasions, with increases to 92.7 ± 27.8% (ES 2.03 ± 0.76). | Change in T/C indicates subjects were unlikely to have been in a catabolic state during the season. Increase in Cortisol compared with Pre had a small negative correlation with performance. |
| Cunniffe et al. (2011) [51] | 31 professional rugby union players (Forwards: n = 16, Age: 26.8 ± 0.9 years; Weight: 112 ± 2.6 kg; Height: 188.3 ± 1.7 cm. Backs: n = 14, Age: 25.9 ± 0.9 years; Weight: 91 ± 2.0 kg; Height: 182.6 ± 2.4 cm). | 48-week competitive season. | Training | Upper respiratory illness (URI), salivary immunoglobulin A (s-IgA), salivary lysozyme (s-Lys), and cortisol. | Weekly illness and TL data were collected during the season. Timed resting morning saliva samples were taken (s-IgA n = 11; s-cortisol (n = 7) across the season (n = 48 weeks). | No significant correlation found between absolute s-IgA or s-Lys concentrations and URI incidence. Peaks in URI were preceded by periods of increased training intensity and reduced game activity. Lower s-IgA (p < 0.05) and s-Lys concentrations were consistently observed in backs than forwards, whereas URI incidence also differed for player position (3.4 forwards vs. 4.3 backs). Decreases in absolute s-IgA (December) and s-Lys (November and February) concentrations were associated with a corresponding increase in saliva cortisol (p < 0.05). | Regular monitoring of s-IgA and s-Lys may be useful in assessing exercise stress and URI risk status in elite team sport athletes. Stress-induced increases in cortisol release are likely to contribute to reductions in mucosal immunity, predisposing rugby players to increased illness risk. |
| Horta et al. (2019) [52] | 12 elite male volleyball players (Age: 26.9 ± 4.6 years; Body mass: 94.9 ± 11.6 kg; Height: 194.6 ± 8 cm). | A 6-week Short Preparatory Period | Preparatory training sessions | Creatine Kinase (CK), Testosterone (T), Cortisol (Cr), and T/Cr ratio. | Assessments at baseline, after 2nd, 4th, and 6th weeks. | Significant increases in training load and CK levels, indicating muscle damage (r = 0.32; p = 0.05) Psychological stress increased, as reflected in the Stress Questionnaire for Athletes (RESTQ-Sport) responses. No significant changes in T, Cr, and T/Cr ratio. | A short preparatory period led to increased training load, muscle damage, and psychological stress without a concurrent increase in physical performance. |
| Kamarauskas et al. (2023) [53] | 21 professional male basketball players (age: 26.2 ± 4.9 years; height: 198.7 ± 6.7 cm; body mass: 93.2 ± 10.0 kg) | 5 weeks Pre-season phase | Training and Matches | Testosterone (T), Cortisol (C), and their ratio (T/C) | Saliva samples were collected during an experimental day at the beginning of each week of the preseason phase | No significant (p > 0.05) relationships were evident between weekly changes in T, C, or T/C | These results suggest that internal load measures cannot be used to anticipate weekly hormonal responses during the pre-season phase in professional male basketball players. |
| Lindsay et al. (2015) [54] | 24 professional rugby players (Age: 24.2 ± 2.9 years; Mass: 103.3 ± 11.6 kg; Height: 1.87 ± 0.06 m) | 3 professional rugby games. | Matches | Myoglobin, salivary immunoglobulin A, cortisol, neopterin and total neopterin | Saliva samples were collected ~120 min pre-game and ~30–40 min post-game. | Post-game decrements (p < 0.001), sIgA decreases for game 2 (p = 0.019). Mean sIgA decreases following all games. | Significant decreases in sIgA concentration and secretion were observed for game 2. Post-game secretion rate is affected by pre-game rate and number of impacts. |
| Marin et al. (2013) [55] | 10 professional handball players (Age: 25 ± 4.5 years; Mass: 95.3 ± 9.8 kg; Height: 187 ± 6.6 cm) | Over 6 months of competitive season, with evaluations every six weeks. | Matches and training | Oxidative stress and antioxidant biomarkers, muscle damage, biochemical parameters, antioxidant enzymatic activities, functional parameters of immune cells, production of superoxide anion, nitric oxide, and hydrogen peroxide. | Blood samples were collected four times every six weeks throughout the season. At each blood collection (T1–T4), samples were collected in the morning (10 a.m.) | Plasma TBARS: Increased significantly (4.4-fold post-T3, 3.2-fold post-4). Plasma Thiols: Marked decrease during intense periods. Erythrocyte TBARS: Transient rise, significant reduction by T4. Erythrocyte Antioxidant Enzymes: Dramatic increase (up to 14.7-fold for superoxide dismutase at T4). Creatine Kinase: 94% increase after T4, indicating muscle damage. Lactate Dehydrogenase: Decreased, then normalized. IL-1β: Significant decrease post-T2. IL-6 and TNF-α: Stable levels. Lymphocyte Proliferation & Neutrophil Phagocytic Capacity: Notable fluctuations, 20% decrease in phagocytic capacity. | Oxidative stress and antioxidant biomarkers can change throughout the season in competitive athletes, reflecting the physical stress and muscle damage that occurs as the result of competitive handball training. In addition, these biochemical measurements can be applied in the physiological follow-up of athletes. |
| Martínez et al. (2010) [56] | 12 professional basketball players (Age: 25.3 ± 4.4 years; Height: 1.98 ± 0.10 m; Weight: 96.8 ± 13 kg) | Preseason and season | Matches and training | Cortisol and Testosterone | At 4 time points: Preseason (T1) End of 2nd mesocycle (T2) King’s Cup (T3) End of regular season and Eurocup (T4) | Catabolic/anabolic balance throughout the season: Decrease in cortisol levels at T2 and T4. Increase in T/C ratio at T2 and decrease at T3. | Increase in testosterone and decrease or maintenance of cortisol levels can contribute to effective recovery. Monitoring cortisol, testosterone, and training levels is useful to prevent stress and manage recovery periods during the season. |
| McLean et al. (2010) [30] | 12 professional rugby league players (Age: 24.3 ± 3.6 years; Body mass: 101.9 ± 8.4 kg; Stature: 184.7 ± 6.1 cm) | During three different duration training weeks throughout a 26-week rugby league season. | Matches and training | Testosterone, Cortisol, and Testosterone/Cortisol ratio (T/C) | Saliva samples collected 4 h pre-match and 1, 2, and 4 days post-match in all three experimental weeks. | A significantly higher mean daily load was found in the 7-day (p < 0.05, d = 0.45) and 9-day (p < 0.01, d = 0.59) microcycles compared with the 5-day microcycle. Day 4 cortisol measures in the 9-day and 7-day microcycles were significantly higher than game day (p < 0.01, d = 0.60) and tended to be higher than day 1 measures, approaching significance (p = 0.07, d = 0.69). | The study highlights the complexity of using salivary hormones, especially testosterone and the T/C ratio, as reliable indicators of fatigue or anabolic/catabolic state in professional rugby players. Cortisol showed some correlation with training load and recovery, but its variability also suggests limitations in its use as a sole indicator of physiological stress or fatigue in this sporting context. |
| Miloski et al. (2016) [20] | Twelve male professional futsal players (24.3 ± 4.7 years old; 75.5 ± 7.7 kg; and 173.4 ± 4.5 cm) | Preseason and partially (midway) season | Training | Testosterone, Cortisol, and Testosterone/Cortisol ratio (T/C). CK | Every 2 weeks (preseason) and every 4 weeks (season) | Increase in CK (266 μ/L) and T/C ratio (2.0) at the end of preseason. Increase in cortisol (+3.9 mg/dL) and decrease in T/C ratio (−0.5) from Blood samples 4 to Blood Samples 5 | During the in-season, players kept their CK values stable without any loss in physical performance, suggesting that stable blood CK levels are a physiological feature of active futsal players. Seasonal hormonal data show futsal players effectively handled training and competition stress, as indicated by the stable T/C ratio without linked performance decline. |
| Mohr et al. (2016) [57] | 40 competitive male soccer players (Age: 21.5 ± 0.3 years; Height: 1.77 ± 0.01 m; Weight: 73.4 ± 0.9 kg) | Congested 1-week study over 11 days, including baseline testing, three 90 min games, and 9 days of practice sessions and testing between and after games. | Matches and training | PC, NEFA, Urea, Ammonia, Glycerol, Adhesion molecule concentrations, CK GSH, GSSG, TBARS, CAT, Cortisol, Testosterone, Cytokines, CRP, TAC. | Venous blood samples collected every morning until the 3rd day after the final match. Additional samples before each match and 3–4 min after the end of the first and second half of each match for metabolite measurement. | Elevated levels of CK, CRP, and cortisol were noted 48 h post-games, with more significant increases following the second match. Oxidative stress markers such as TBARS and carbonylated proteins showed substantial increases post-games. The reduced/oxidized glutathione ratio declined during the first 24 h post-games | Inflammatory and oxidative stress responses to consecutive match microcycles indicate increased physiological stress and more pronounced fatigue after the second game, particularly due to the short three-day recovery period. |
| Moreira et al. (2009) [58] | 22 male professional soccer players (Age: 23 ± 4 years; Height: 182 ± 6.8 cm; Body mass: 78.6 ± 8.4 kg). | During the competitive season. | Competitive training soccer match | Salivary cortisol concentrations | Subjects provided resting saliva samples approximately 10 min before the pre-session warm-up (PRE) and post-session saliva samples were col- lected within 10 min after the conclusion of the match (POST). | No significant changes in salivary cortisol concentrations (p > 0.05) were observed between teams or time points. Individual responses varied, showing both increases and decreases in cortisol levels. | The study indicates that a competitive soccer match does not significantly impact salivary cortisol levels in top-level professional soccer players adapted to this type of stress. Highlights the need for individual analysis due to response variability among players. |
| Moreira et al. (2013) [59] | 12 elite Brazilian futsal players (age: 19 ± 1 years; height: 180 ± 4 cm; and body mass: 73 ± 7 kg). | 4 weeks of intensive training during the competitive season with 27 training sessions performed. | Training | salivary immunoglobulin A, cortisol, and upper respiratory tract infection (URTI) | Salivary immunoglobulin A, salivary cortisol, and symptoms of URTIs were assessed weekly. | No significant differences were observed for sIgA during the study (p > 0.05). The relative change in sIgA absolute was associated with the URTI severity during week 4 (r = −0.74; p < 0.05). | Futsal athletes were more susceptible to high URTI symptom severity in periods of higher training intensity and volume. A reduction in training load before competitions is an appropriate strategy to minimize URTI symptoms, ensuring the athlete’s ability to train and compete. |
| Rowell et al. (2018) [60] | 23 elite soccer players (Mean age: 23.3 ± 4.1 years; Height: 180 ± 10.0 cm; Weight: 75.7 ± 4.4 kg) | Competitive season with 34 matches (27 regular league and 7 Asian Champions League matches). | Matches | testosterone, and cortisol | Saliva collection before the last training session prior to match play, between 09:00 and 09:30 a.m., following strict pre-test procedures. | Position-specific responses to training loads. Center defenders showed a reduction in performance ratings with increased load, while strikers and wide midfielders tended to improve with increased load. Wide midfielders also showed increased testosterone levels with increased training load. | Increases in training load significantly affect hormonal levels, especially an increase in cortisol and testosterone in center defenders and changes in the testosterone/cortisol ratio. |
| Saidi et al. (2022) [61] | 14 elite soccer players (Age: 20.9 ± 0.8 years; Height: 177 ± 5 cm; Weight: 72.4 ± 5.2 kg) | 12 weeks (T1–T2: 6 regular weeks; T2–T3: 6 congested weeks) | Matches and training | CRP, CK, Creatinine | Evaluations at T1 (week 1), T2 (week 6), and T3 (week 12) | Significant increase in stress, fatigue, DOMS, and Hopper Index during congested period. Notable correlations between Δ% of CRP, Δ% of CK, Hopper Index, and Δ% of fatigue | Increases in biochemical markers and changes in well-being during congested periods indicate a direct relationship with training load. Monitoring these parameters is vital to prevent overtraining and optimize performance |
| Schelling et al. (2009) [62] | Male professional basketball team (27.8 ± 4.8 years; 97 ± 9.5 kg; 197.2 ± 7.3 cm) | During the competitive season | Matches and training | Testosterone, Cortisol, and Testosterone/Cortisol ratio (T/C). | 4–6 weeks (8 Samples) | Increase in cortisol in preseason (+33%) and maintained throughout the season (0.393–0.516 mmol/L). Increase of testosterone after 3.5 days of rest and decrease at the end of the season (20.6–24.9 vs. 18.0 mmol/L). T/C ratio decreases at the end of the season (48.0–61.7 vs. 35.4). | The T/C ratio and/or testosterone could be used as an indicator of the state of be used as indicators of recovery status and help to optimize individualized individualized training loads to avoid episodes of excessive fatigue. |
| Schelling et al. (2015) [63] | 20 professional male basketball players | Four consecutive seasons | Regular season matches and training | Blood plasma total testosterone (TT) and cortisol (C) levels, testosterone-to-cortisol ratio (TT/C). | Blood samples were collected periodically every 4–6 weeks. Always after a 24- to 36-h break after the last game played. | Hormonal levels in professional basketball players were found to be position-dependent. Power forwards (PF) showed the lowest total testosterone (TT) levels (median 18.1 ± 4.9 nmol/L), while small forwards had the highest cortisol levels (0.55 ± 0.118 mmol/L). Players with 13–25 min of game time per match exhibited the highest TT (22.8 ± 6.9 nmol/L) and TT/C ratios (47.1 ± 21.2). The most stressed hormonal state, characterized by low TT/C and high cortisol levels, was observed in March and April, highlighting the need for tailored management based on playing time and position. | Monitoring plasma TT and cortisol is crucial for managing stress induced by the demands of a professional basketball season. Hormonal status varies according to playing position and game time, impacting training and recovery strategies. |
| Souglis et al. (2015) [64] | 72 elite male players from four team sports (soccer, basketball, volleyball, handball). | Start of the regular season. | Matches. | TNFα, IL-6, CRP, CK, LDH, Urea, Ammonia, Cortisol | Pre, post, 13 h, and 37 h after the match. | Soccer showed the highest increase in inflammatory cytokines and muscle damage markers. Volleyball showed the least increase compared to the other three sports. | Professional soccer matches impose higher metabolic demands and cause greater inflammatory responses and muscle damage compared to handball, basketball, and volleyball. |
| Spanidis et al. (2016) [65] | 14 adult male basketball players (age, 26.8 ± 1.2 years; height, 1.99 ± 0.02 m; weight, 101.6 ± 2.63 kg) | Internal Season | Regular season matches and training. | Markers of oxidative stress (TAC, TBARS, GSH, CARB, and sORP) | Beginning and end of season | Increase in sORP values (200 vs. 220 mV; 9.6%) and TAC (0.8 vs. 1.0 mmol/L; 12.9%) at the end of the season. Decrease in GSH (3.7 vs. 2.5 mmol/g; 35%) at the end of the season. Large inter-individual variation in TBARS, CARB of TBARS, CARB, TAC. High correlation between sORP and CARB (r = 0.798). | The sORP can help to monitor the redox status of a group of the redox status of a group of athletes, with higher greater completeness than TAC. An individualized examination of the redox status through TBARS, CARB, and TAC is required to identify critical recovery periods for each athlete. |
| Springham et al. (2021) [66] | 18 senior professional male soccer players (Age: 24 ± 3.8 years; Height: 181 ± 7.0 cm; Body mass: 72.4 ± 5.2 kg). | A 6-week preseason and a 40-week season, divided into eight 5-week mesocycles. | Regular season matches and training. | Salivary immunoglobulin-A (s-IgA), α-amylase (s-AA), testosterone (s-T), cortisol (s-C), and testosterone/cortisol ratio (s-T/C); Athlete Self-Report Measures (ASRM) including fatigue, sleep quality, and muscle soreness. | Bi-weekly collection following recovery days | The study found small reductions in salivary immunoglobulin-A (p = 0.003), α-amylase (p = 0.047), and cortisol (p = 0.007), with trivial changes in testosterone. The testosterone/cortisol ratio varied inversely with workload. Self-reported fatigue, sleep quality, and muscle soreness improved across the season’s first half (p = 0.030 to p = 0.005, small effect). Hormonal changes correlated with self-reports (R2 = 0.43 to 0.45), with cortisol linked to worse and testosterone/cortisol to better reports. Non-linear relationships were found between some hormones and self-reports, indicating optimal levels for best responses. | Chronic suppression of mucosal immunity was observed. Salivary measures related to self-report measures, indicating the need for reduced workload to improve wellbeing. Monitoring s-IgA, s-T, s-C, and s-T/C can be effective in assessing players’ health and performance status. |
| Talaee et al. (2017) [67] | 15 elite male basketball players (Age: 24 ± 1.5 years; weight: 83 ± 3.3 kg; height: 188 ± 6.1 cm) | 6 weeks | Combined training | RBC, Hb, Hct, MCV, MCH, WBC, PLT, MCHC | Pre-, post-, and 24 h after training | Significant increase in white blood cell and platelet counts at two stages; post-training and 24 h after recovery. Hemoglobin, hematocrit, and red blood cell count significantly decreased after 24 h of recovery. | Combined training plays a significant role in physiological and hematological adaptation processes, enhancing athletic performance. Prescribing this training method should be coupled with regular blood biochemical monitoring to balance exercise stress and recovery strategies. |
| Tiernan et al. (2020) [68] | 19 male elite rugby union players. | over a 10-week training period | Training | Upper respiratory illness (URI), salivary immunoglobulin A (s-IgA) | Saliva samples were collected twice a week, Monday and Friday, within 1 h of the players waking up before training. | No significant differences in weekly sIgA levels were found over the 10-week period. The likelihood of suffering from a URTI increased when sIgA significantly decreased (p = 0.046). | A decrease in >65% of sIgA meant players were at risk within the following 2 weeks of contracting a URTI. |
| Twist et al. (2012) [69] | 23 male rugby league players. Players were categorized as backs (n = 10; age 25.9 ± 5.1 years; stature 1.82 ± 0.08 m; body mass 91.9 ± 11.6 kg) or forwards (n = 13; age 26.0 ± 4.1 years; stature 1.83 ± 0.06 m; body mass 102.0 ± 6.7 kg) | European Rugby League Super League season | Matches | Creatine Kinase (CK) | Assessments conducted pre-match, one day after (day 1), and two days after the match (day 2). | Creatine kinase was higher both 1 and 2 days after than before matches (p < 0.05) in forwards and backs. | Despite the mechanisms of fatigue being different between forwards and backs, our results highlight the multidimensional nature of fatigue after a rugby league match and that markers do not differ between positions. |
| Biomarker Category | Specific Biomarkers | Relevance to Chronic Fatigue | Typical Measurement Method |
|---|---|---|---|
| Muscle anabolic/catabolic hormones | Testosterone, Cortisol, Testosterone/Cortisol ratio | Reflect metabolic strain and stress responses to training/competition loads | Blood or saliva samples; ELISA or radioimmunoassay |
| Muscle damage markers | Creatine Kinase (CK), Lactate Dehydrogenase (LDH) | Quantify extent of exercise-induced muscle damage | Blood samples; Spectrophotometry |
| Immunological markers | Salivary Immunoglobulin A (s-IgA), Immune cell function | Indicate mucosal immunity status and potential vulnerability to upper respiratory tract infections | Saliva samples; ELISA |
| Oxidative stress markers | Reactive Oxygen Species, Antioxidant Capacity, TBARS, Protein Carbonyls | Assess cellular stress and redox balance | Blood samples; Spectrophotometry, ELISA |
| Inflammatory markers | C-Reactive Protein (CRP), Cytokines (e.g., IL-6, TNF-α) | Indicate systemic inflammatory responses to prolonged intense training | Blood samples; ELISA, Flow cytometry |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Soler-López, A.; Moreno-Villanueva, A.; Gómez-Carmona, C.D.; Pino-Ortega, J. The Role of Biomarkers in Monitoring Chronic Fatigue Among Male Professional Team Athletes: A Systematic Review. Sensors 2024, 24, 6862. https://doi.org/10.3390/s24216862
Soler-López A, Moreno-Villanueva A, Gómez-Carmona CD, Pino-Ortega J. The Role of Biomarkers in Monitoring Chronic Fatigue Among Male Professional Team Athletes: A Systematic Review. Sensors. 2024; 24(21):6862. https://doi.org/10.3390/s24216862
Chicago/Turabian StyleSoler-López, Alejandro, Adrián Moreno-Villanueva, Carlos D. Gómez-Carmona, and José Pino-Ortega. 2024. "The Role of Biomarkers in Monitoring Chronic Fatigue Among Male Professional Team Athletes: A Systematic Review" Sensors 24, no. 21: 6862. https://doi.org/10.3390/s24216862
APA StyleSoler-López, A., Moreno-Villanueva, A., Gómez-Carmona, C. D., & Pino-Ortega, J. (2024). The Role of Biomarkers in Monitoring Chronic Fatigue Among Male Professional Team Athletes: A Systematic Review. Sensors, 24(21), 6862. https://doi.org/10.3390/s24216862









