Near-Infrared Spectroscopy for Rapid Differentiation of Fresh and Frozen–Thawed Common Carp (Cyprinus carpio)
Abstract
1. Introduction
2. Materials and Methods
2.1. Samples and Experimental Design
2.2. NIR Measurements
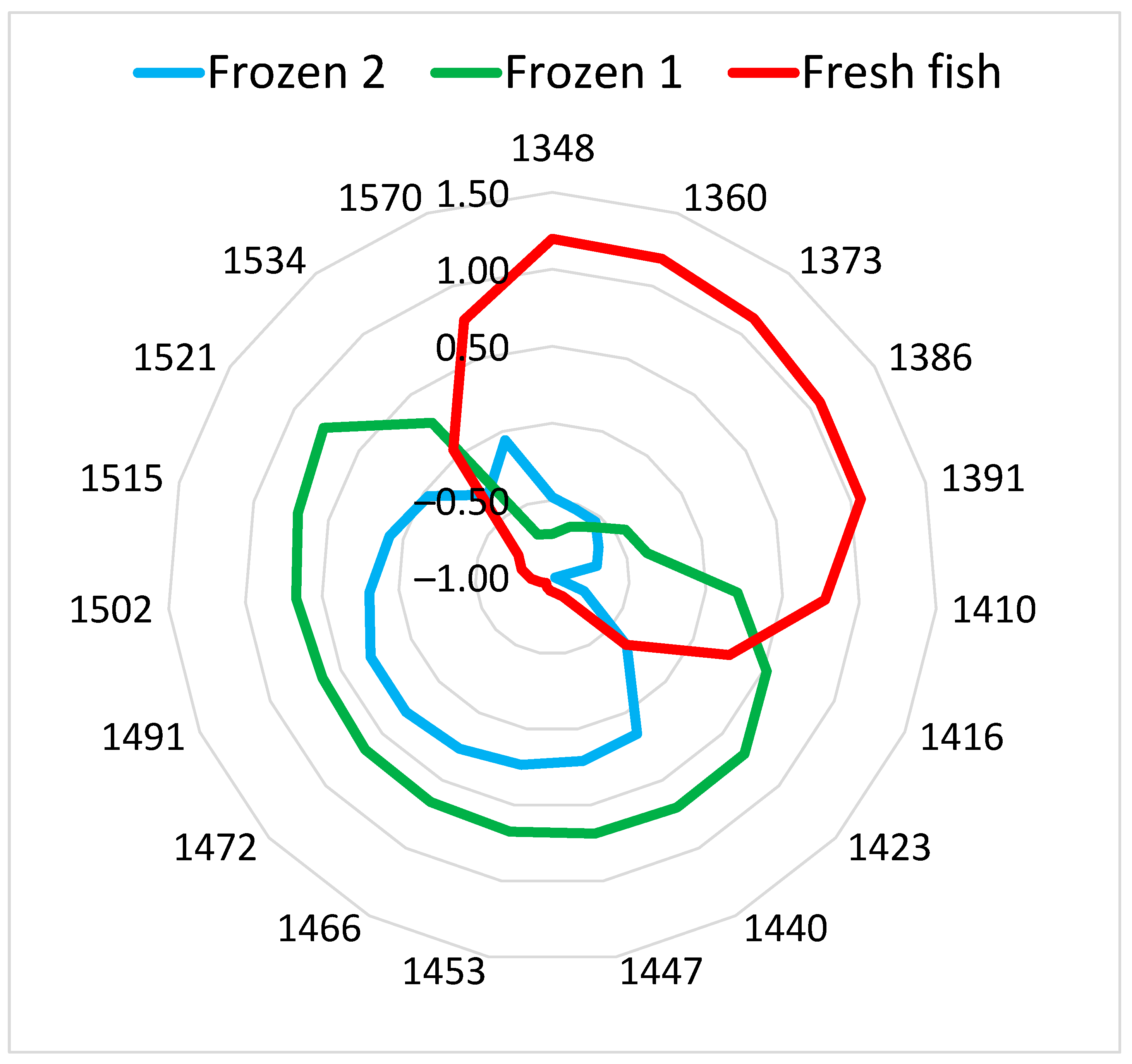
2.3. Data Analysis, Classification Models, and Aquagrams
3. Results and Discussion
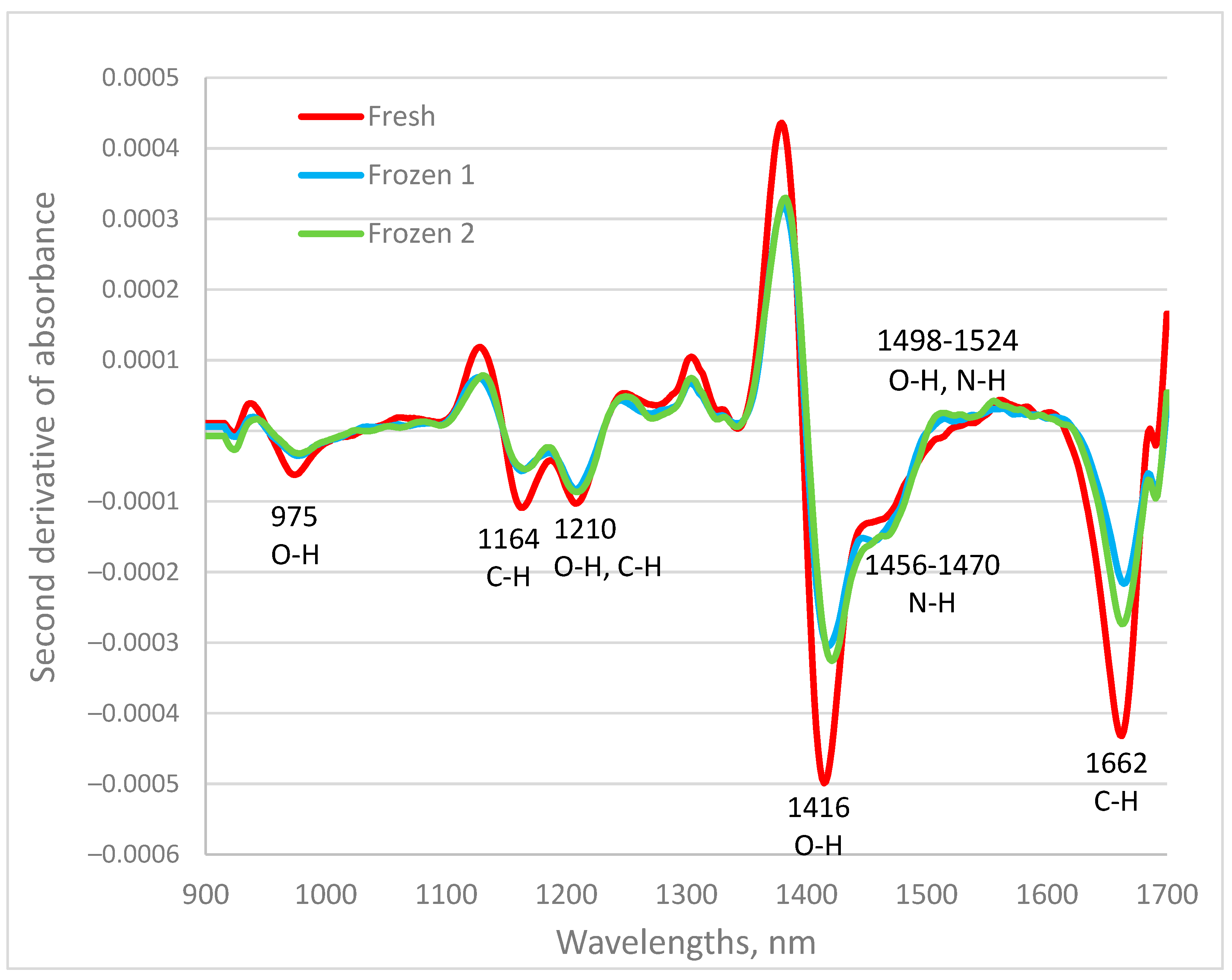
3.1. NIR Spectra of Fish Samples
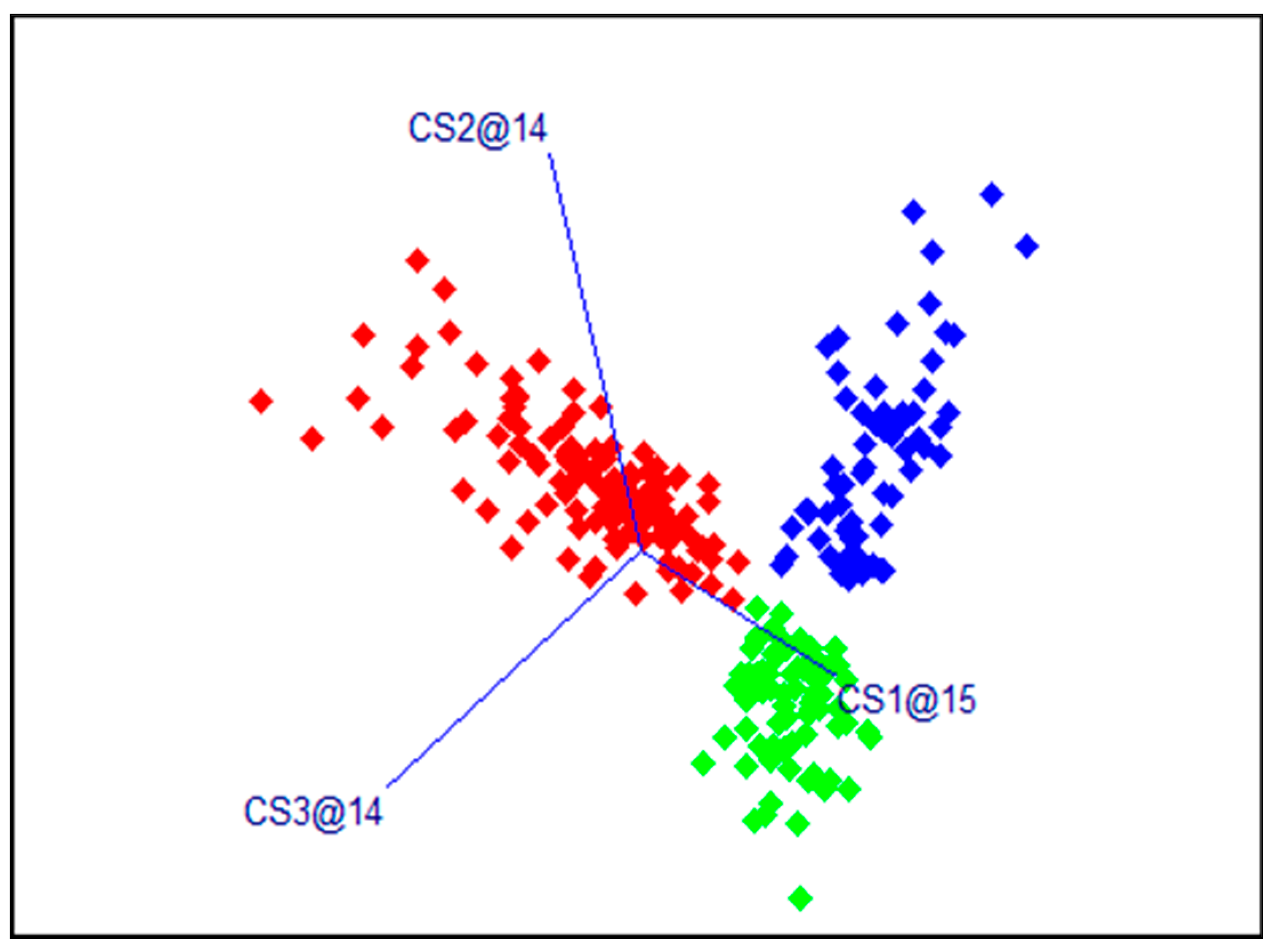
3.2. SIMCA Classification of Fresh and Frozen Fish Samples
3.3. PLS-DA Classification of Fresh and Frozen Fish Samples
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kaliniak-Dziura, A.; Skałecki, P.; Florek, M.; Kędzierska-Matysek, M.; Sobczak, P. Chemical Composition and Elements Concentration of Fillet, Spine and Bones of Common Carp (Cyprinus carpio) in Relation to Nutrient Requirements for Minerals. Animals 2024, 14, 1311. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chiesa, L.M.; Pavlovic, R.; Nobile, M.; Di Cesare, F.; Malandra, R.; Pessina, D.; Panseri, S. Discrimination between Fresh and Frozen-Thawed Fish Involved in Food Safety and Fraud Protection. Foods 2020, 12, 1896. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pezzolato, M.; Baioni, E.; Maurella, C.; Varello, K.; Meistro, S.; Balsano, A.; Bozzetta, E. Distinguishing between Fresh and Frozen-Thawed Smoked Salmon: Histology to Detect Food Adulteration in High-Value Products. J. Food Prot. 2020, 83, 52–53. [Google Scholar] [CrossRef] [PubMed]
- Maritano, V.; Barge, P.; Biglia, A.; Comba, L.; Ricauda Aimonino, D.; Tortia, C.; Gay, P. Anticounterfeiting and Fraud Mitigation Solutions for High-value Food Products. J. Food Prot. 2024, 87, 100251. [Google Scholar] [CrossRef] [PubMed]
- Rahman, M.M. Role of common carp (Cyprinus carpio) in aquaculture production systems. Front. Life Sci. 2015, 8, 399–410. [Google Scholar] [CrossRef]
- Zander, K.; Feucht, Y. How to increase demand for carp? Consumer attitudes and preferences in Germany and Poland. Br. Food J. 2020, 122, 3267–3282. [Google Scholar] [CrossRef]
- Vanhonacker, F.; Pieniak, Z.; Verbeke, W. European consumer perceptions and barriers for fresh, frozen, preserved and ready-meal fish products. Br. Food J. 2013, 115, 508–525. [Google Scholar] [CrossRef]
- Nakazawa, N.; Okazaki, E. Recent research on factors influencing the quality of frozen seafood. Fish. Sci. 2020, 86, 231–244. [Google Scholar] [CrossRef]
- Tolstorebrov, I.; Eikevik, T.M.; Bantle, M. Effect of low and ultra-low temperature applications during freezing and frozen storage on quality parameters for fish. Int. J. Refr. 2016, 63, 37–47. [Google Scholar] [CrossRef]
- Hong, H.; Luo, Y.; Zhou, Z.; Bao, Y.; Lu, H.; Shen, H. Effects of different freezing treatments on the biogenic amine and quality changes of bighead carp (Aristichthys nobilis) heads during ice storage. Food Chem. 2013, 138, 1476–1482. [Google Scholar] [CrossRef]
- European Parliament. Regulation No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers. O. J. Eur. Union 2011, 50, 18–63. [Google Scholar]
- European Parliament. Regulation (EU) No 1379/2013 of the European Parliament and of the Council of 11 December 2013 on the common organisation of the markets in fishery and aquaculture products, amending Council Regulations (EC) No 1184/2006 and (EC) No 1224. O. J. Eur. Union 2013, 354, 1–21. [Google Scholar]
- Bozzetta, E.; Pezzolato, M.; Cencetti, E.; Varello, K.; Abramo, F.; Mutinelli, F.; Ingravalle, F.; Teneggi, E. Histology as a valid and reliable tool to differentiate fresh from frozen-thawed fish. J. Food Prot. 2012, 75, 1536–1541. [Google Scholar] [CrossRef]
- Tinacci, L.; Armani, A.; Guidi, A.; Nucera, D.; Shvartzman, D.; Miragliotta, V.; Coli, A.; Giannessi, E.; Stornelli, M.R.; Fronte, B.; et al. Histological discrimination of fresh and frozen/thawed fish meat: European hake (Merluccius merluccius) as a possible model for white meat fish species. Food Contr. 2018, 92, 154–161. [Google Scholar] [CrossRef]
- Ethuin, P.; Marlard, S.; Delosière, M.; Carapito, C.; Delalande, F.; Van Dorsselaer, A.; Dehaut, A.; Lencel, V.; Duflos, G.; Grard, T. Differentiation between fresh and frozen-thawed sea bass (Dicentrarchus labrax) fillets using two-dimensional gel electrophoresis. Food Chem. 2015, 176, 294–301. [Google Scholar] [CrossRef]
- Diop, M.; Watier, D.; Masson, P.-Y.; Diouf, A.; Amara, R.; Grard, T.; Lencel, P. Assessment of freshness and freeze-thawing of sea bream fillets (Sparus aurata) by a cytosolic enzyme: Lactate dehydrogenase. Food Chem. 2016, 210, 428–434. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Zhang, L.; Lu, H.; Shen, H.; Yu, X.; Luo, Y. Protein and lipid changes of mud shrimp (Solenocera melantho) during frozen storage: Chemical properties and their prediction. Int. J. Food Prop. 2017, 20, 2043–2056. [Google Scholar] [CrossRef]
- Fuentes, A.; Masot, R.; Fernández-Segovia, I.; Ruiz-Rico, M.; Alcañiz, M.; Barat, J.M. Diferentiation between fresh and frozen-thawed sea bream (Sparus aurata) using impedance spectroscopy techniques. Innov. Food Sci. Emerg. Technol. 2013, 19, 210–217. [Google Scholar] [CrossRef]
- Zhang, L.; Shen, H.; Luo, Y. Study on the electric conduction properties of fresh and frozen-thawed grass carp (Ctenopharyngodon idellus) and tilapia (Oreochromis niloticus). Int. J. Food Sci. Technol. 2010, 45, 2560–2564. [Google Scholar] [CrossRef]
- Kaltenbach, K.; Kuballa, T.; Schroder, U.; Fritsche, J.; Bunzel, M.; Haase, M. Evaluation of NMR-based strategies to differentiate fresh from frozen-thawed fish supported by multivariate data analysis. Eur. Food Res. Technol. 2024, 250, 239–251. [Google Scholar] [CrossRef]
- Hassoun, A.; Shumilina, E.; Di Donato, F.; Foschi, M.; Simal-Gandara, J.; Biancolillo, A. Emerging techniques for differentiation of fresh and frozen-thawed seafoods: Highlighting the potential of spectroscopic techniques. Molecules 2020, 25, 4472. [Google Scholar] [CrossRef] [PubMed]
- Reis, M.M.; Martínez, E.; Saitua, E.; Rodríguez, R.; Pérez, I.; Olabarrieta, I. Non-invasive differentiation between fresh and frozen/thawed tuna fillets using near infrared spectroscopy (Vis-NIRS). LWT 2017, 78, 129–137. [Google Scholar] [CrossRef]
- Ottavian, M.; Fasolato, L.; Serva, L.; Facco, P.; Barolo, M. Data fusion for food authentication: Fresh/frozen–thawed discrimination in west African goatfish (Pseudupeneus prayensis) fillets. Food Bioprocess. Technol. 2014, 7, 1025–1036. [Google Scholar] [CrossRef]
- Fasolato, L.; Balzan, S.; Riovanto, R.; Berzaghi, P.; Mirisola, M.; Ferlito, J.C.; Serva, L.; Benozzo, F.; Passera, R.; Tepedino, V.; et al. Comparison of Visible and Near-Infrared Reflectance Spectroscopy to Authenticate Fresh and Frozen-Thawed Swordfish (Xiphias gladius L). J. Aquat. Food Prod. Tech. 2012, 21, 493–507. [Google Scholar] [CrossRef]
- Goncalves, D.B.; Santos, C.S.P.; Pinho, T.; Queiro´s, R.; Vaz, P.D.; Bloore, M.; Satta, P.; Kovacs, Z.; Casal, S.; Hoffmann, I. Near Infrared Reflectance Spectroscopy Coupled to Chemometrics as a Cost-Effective, Rapid, and Non-Destructive Tool for Fish Fraud Control: Monitoring Source, Condition, and Nutritional Value of Five Common Whitefish Species. J. AOAC Int. 2021, 104, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Nieto-Ortega, S.; Melado-Herreros, Á.; Foti, G.; Olabarrieta, I.; Ramilo-Fernández, G.; Gonzalez Sotelo, C.; Teixeira, B.; Velasco, A.; Mendes, R. Rapid Differentiation of Unfrozen and Frozen-Thawed Tuna with Non-Destructive Methods and Classification Models: Bioelectrical Impedance Analysis (BIA), Near-Infrared Spectroscopy (NIR) and Time Domain Reflectometry (TDR). Foods 2022, 11, 55. [Google Scholar] [CrossRef] [PubMed]
- Nimbkar, S.; Auddy, M.; Manoj, I.; Shanmugasundaram, S. Novel Techniques for Quality Evaluation of Fish: A Review. Food Rev. Int. 2021, 39, 639–662. [Google Scholar] [CrossRef]
- Nieto-Ortega, S.; Lara, R.; Foti, G.; Melado-Herreros, Á.; Olabarrieta, I. Applications of Near-Infrared Spectroscopy (NIRS) in Fish Value Chain. In Infrared Spectroscopy–Perspectives and Applications; El-Azazy, M., Al-Saad, K., El-Shafie, A.S., Eds.; IntechOpen: London, UK, 2022. [Google Scholar] [CrossRef]
- Tsenkova, R. Aquaphotomics: Dynamic Spectroscopy of Aqueous and Biological Systems Describes Peculiarities of Water. J. Near Inf. Spec. 2009, 17, 303–314. [Google Scholar] [CrossRef]
- Blazhekovikj-Dimovska, D.; Ahmed, S. Comparative indicators of the chemical composition of farmed and wild common carp (Cyprinus carpio L.). Croat. J. Food Sci. Technol. 2022, 14, 9. [Google Scholar]
- Bao, Y.; Ertbjerg, P.; Estévez, M.; Yuan, L.; Gao, R. Freezing of meat and aquatic food: Underlying mechanisms and implications on protein oxidation. Compr. Rev. Food Sci. Food Saf. 2021, 20, 5548–5569. [Google Scholar] [CrossRef]
- Bao, Y.; Wang, K.; Yang, H.; Regenstein, J.M.; Ertbjerg, P.; Zhou, P. Protein degradation of black carp (Mylopharyngodon piceus) muscle during cold storage. Food Chem. 2020, 308, 125576. [Google Scholar] [CrossRef] [PubMed]
- Gökoğlu, N.; Yerlikaya, P. Freezing and frozen storage of fish. In Seafood Chilling, Refrigeration and Freezing: Science and Technology; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2015. [Google Scholar] [CrossRef]
- Hu, C.; Xie, J. The Effect of Multiple Freeze-Thaw Cycles on the Microstructure and Quality of Trachurus murphyi. Foods 2021, 10, 1350. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Bai, X.; Xia, X.; Chen, H. Effect of sodium alginate ice glazing on the quality of the freeze-thawed fish balls. Int. J. Biol. Macromol. 2024, 254, 128097. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.; Feng, J.; Wang, H.; Du, X.; Wang, B.; Yu, G.; Xia, X. Influence of repeated freeze-thaw treatments on the oxidation and degradation of muscle proteins from mirror carp (Cyprinus carpio L.), based on myofibrillar protein structural changes. Int. J. Biol. Macromol. 2023, 226, 454–462. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Yu, Q.; Liu, Y.; Sun, X.; Li, Q.; Fan, H.; Benjakul, S.; Tan, Y.; Luo, Y.; Hong, H. Dual cryoprotective and antioxidant effects of young apple polyphenols on myofibrillar protein degradation and gelation properties of bighead carp mince during frozen storage. J. Food Sci. 2023, 88, 4560–4573. [Google Scholar] [CrossRef] [PubMed]
- Gao, S.; Liu, Y.; Fu, Z.; Zhang, H.; Zhang, L.; Li, B.; Tan, Y.; Hong, H.; Luo, Y. Uncovering quality changes of salted bighead carp fillets during frozen storage: The potential role of time-dependent protein denaturation and oxidation. Food Chem. 2023, 15, 135714. [Google Scholar] [CrossRef] [PubMed]
- Méndez, L.; Zhang, B.; Aubourg, S.P. Enhancement of Lipid Stability of Frozen Fish by Octopus-Waste Glazing. Foods 2023, 12, 2298. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Du, X.; Li, H.; Dong, C.; Ren, Y.; Pan, N.; Kong, B.; Liu, H.; Xia, X. Effect of ice structuring protein on the microstructure and myofibrillar protein structure of mirror carp (Cyprinus carpio L.) induced by freeze-thaw processes. LWT 2021, 139, 110570. [Google Scholar] [CrossRef]
- Strateva, M.; Penchev, G.; Stratev, D. Histological, Physicochemical and Microbiological Changes in the Carp (Cyprinus carpio) Muscles after Freezing. J. Aquat. Food Prod. Tech. 2021, 30, 323–337. [Google Scholar] [CrossRef]
- Giró-Candanedo, M.; Cruz, J.J.; Comaposada, C.; Barnés-Calle, P.; Gou, E. Fulladosa. Differentiation between fresh and frozen-thawed mackerel fish using low-cost portable near infrared spectrometry devices. J. Food Eng. 2024, 379, 112131. [Google Scholar] [CrossRef]
- Tsenkova, R.; Muncan, J.; Pollner, B.; Kovacs, Z. Essentials of Aquaphotomics and Its Chemometrics Approaches. Front. Chem. 2018, 6, 363. [Google Scholar] [CrossRef] [PubMed]
- Popelka, P.; Nagy, J.; Pipová, M.; Marcinčák, S.; Lenhardt, Ľ. Comparison of chemical, microbiological and histological changes in fresh, frozen and double frozen rainbow trout (Oncorhynchus mykiss). Acta Vet. Brno. 2014, 83, 157–161. [Google Scholar] [CrossRef]


| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | F1 Score, % | |
|---|---|---|---|---|
| Fresh fish | 130 | 4 | 0 | 98.5 |
| After a single freezing | 0 | 84 | 0 | 97.1 |
| After a second freezing | 0 | 1 | 64 | 99.2 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 51 | 2 | 0 | 0 | 95.3 |
| After a single freezing | 3 | 35 | 1 | 1 | 84.4 |
| After a second freezing | 0 | 4 | 29 | 0 | 90.6 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | F1 Score, % | |
|---|---|---|---|---|
| Fresh fish | 133 | 1 | 0 | 99.6 |
| After a single freezing | 0 | 83 | 0 | 98.8 |
| After a second freezing | 0 | 1 | 62 | 99.2 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 48 | 0 | 0 | 5 | 86.5 |
| After a single freezing | 4 | 30 | 4 | 1 | 76.0 |
| After a second freezing | 1 | 2 | 28 | 2 | 82.4 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 128 | 0 | 0 | 6 | 97.7 |
| After a single freezing | 0 | 84 | 0 | 0 | 99.7 |
| After a second freezing | 0 | 1 | 64 | 0 | 99.2 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 41 | 9 | 2 | 1 | 85.4 |
| After a single freezing | 2 | 33 | 1 | 4 | 78.6 |
| After a second freezing | 0 | 2 | 28 | 3 | 87.5 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 129 | 0 | 0 | 4 | 98.5 |
| After a single freezing | 0 | 80 | 0 | 1 | 99.4 |
| After a second freezing | 0 | 0 | 66 | 0 | 100.0 |
| Determined as Fresh | Determined as Single-Frozen | Determined as Double-Frozen | No Match | F1 Score, % | |
|---|---|---|---|---|---|
| Fresh fish | 43 | 3 | 1 | 6 | 89.6 |
| After a single freezing | 0 | 31 | 4 | 4 | 82.7 |
| After a second freezing | 0 | 2 | 30 | 1 | 88.2 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Atanassova, S.; Yorgov, D.; Stratev, D.; Veleva, P.; Stoyanchev, T. Near-Infrared Spectroscopy for Rapid Differentiation of Fresh and Frozen–Thawed Common Carp (Cyprinus carpio). Sensors 2024, 24, 3620. https://doi.org/10.3390/s24113620
Atanassova S, Yorgov D, Stratev D, Veleva P, Stoyanchev T. Near-Infrared Spectroscopy for Rapid Differentiation of Fresh and Frozen–Thawed Common Carp (Cyprinus carpio). Sensors. 2024; 24(11):3620. https://doi.org/10.3390/s24113620
Chicago/Turabian StyleAtanassova, Stefka, Dimitar Yorgov, Deyan Stratev, Petya Veleva, and Todor Stoyanchev. 2024. "Near-Infrared Spectroscopy for Rapid Differentiation of Fresh and Frozen–Thawed Common Carp (Cyprinus carpio)" Sensors 24, no. 11: 3620. https://doi.org/10.3390/s24113620
APA StyleAtanassova, S., Yorgov, D., Stratev, D., Veleva, P., & Stoyanchev, T. (2024). Near-Infrared Spectroscopy for Rapid Differentiation of Fresh and Frozen–Thawed Common Carp (Cyprinus carpio). Sensors, 24(11), 3620. https://doi.org/10.3390/s24113620







