Discriminative ‘Turn-on’ Detection of Al3+ and Ga3+ Ions as Well as Aspartic Acid by Two Fluorescent Chemosensors
Abstract
:1. Introduction
2. Materials and Methods
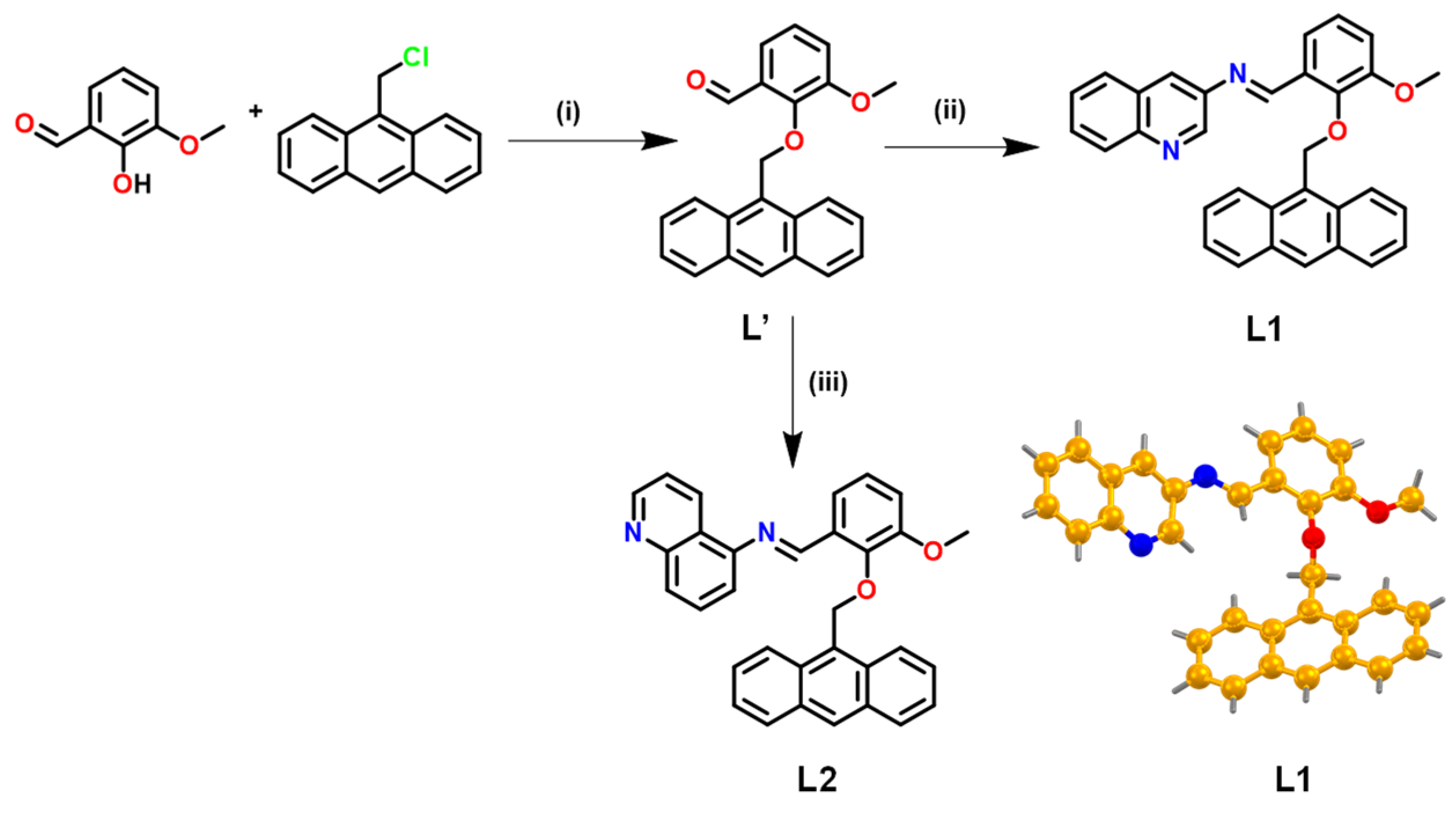
2.1. Synthesis and Characterization
2.1.1. 2-(Anthracen-9-ylmethoxy)-3-methoxybenzaldehyde (L’)
2.1.2. (E)-1-(2-(Anthracen-9-ylmethoxy)-3-methoxyphenyl)-N-(quinolin-3-yl)methanimine (L1)
2.1.3. (E)-1-(2-(Anthracen-9-ylmethoxy)-3-methoxyphenyl)-N-(quinolin-5-yl)methanimine (L2)
2.2. General Information and Methods
2.3. Physical Measurements
2.4. Crystallography
2.5. DFT Studies
2.6. Determination of Binding Constant (Kb)
2.7. Determination of Detection Limit
2.8. Determination of Quantum Yield
2.9. Colorimetric Analysis
2.10. Fabrication of Filter Paper Strips
2.11. Fabrication of Polystyrene Films
3. Results and Discussion
3.1. Synthesis and Characterization of Chemosensors L1 and L2
3.2. Photophysical Properties of L1 and L2
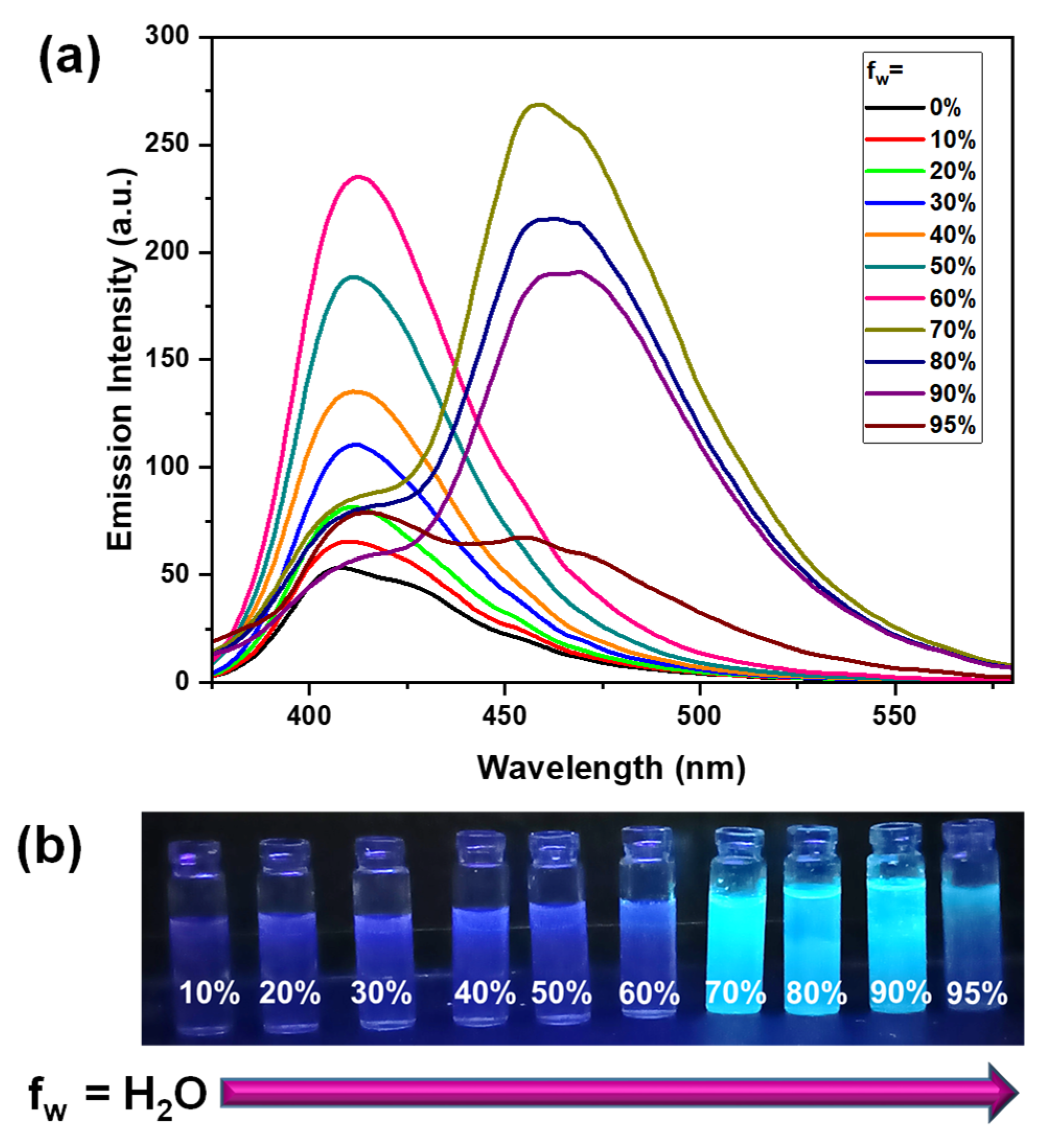
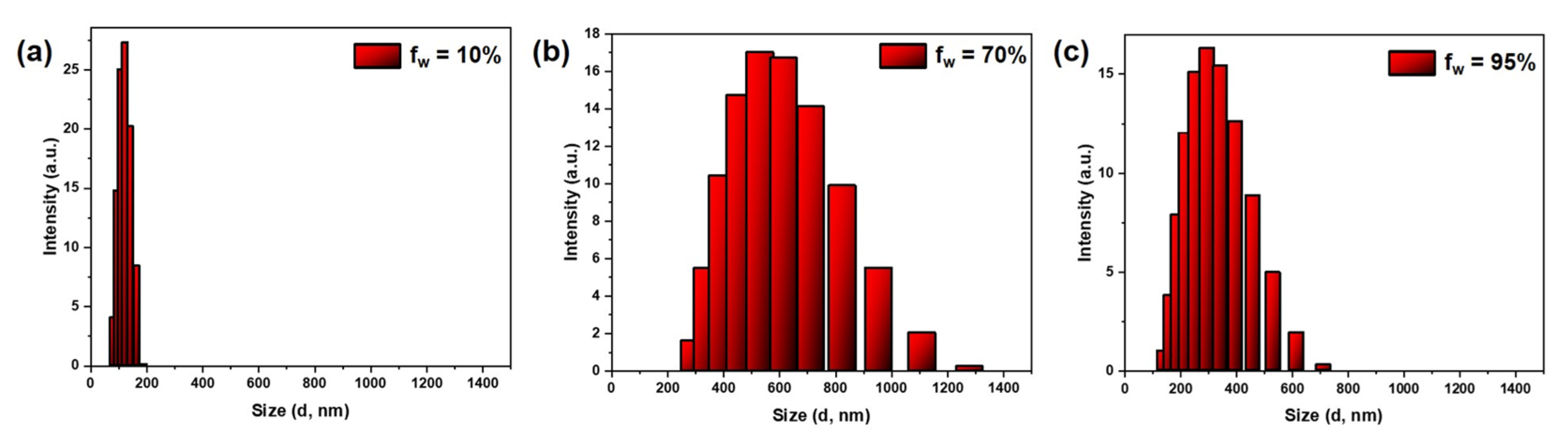
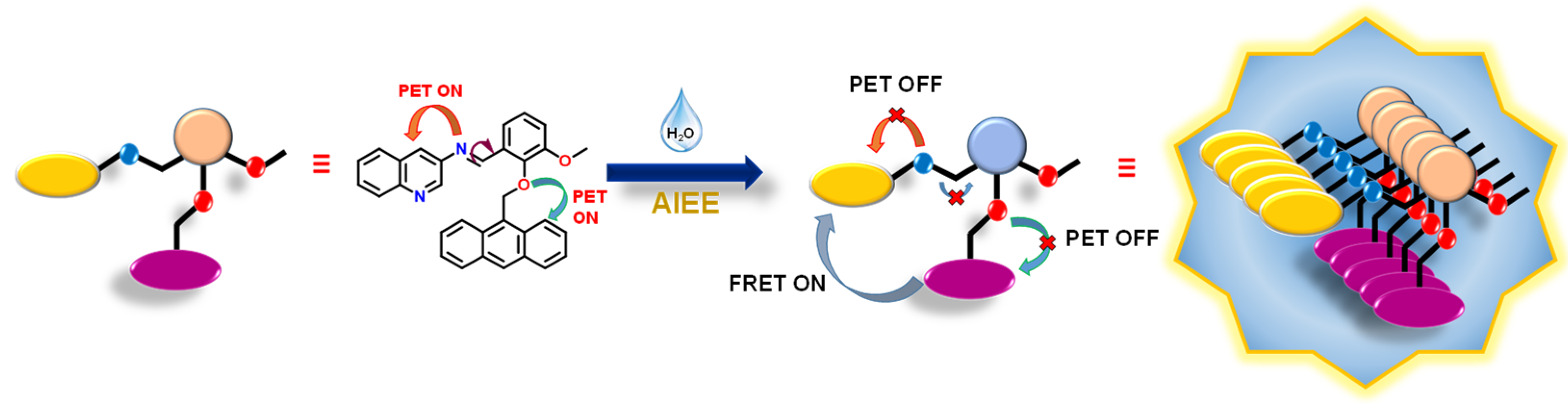
3.3. AIEE Studies
3.4. Detection of Al3+ and Ga3+ Ions by UV-Vis Spectral Studies
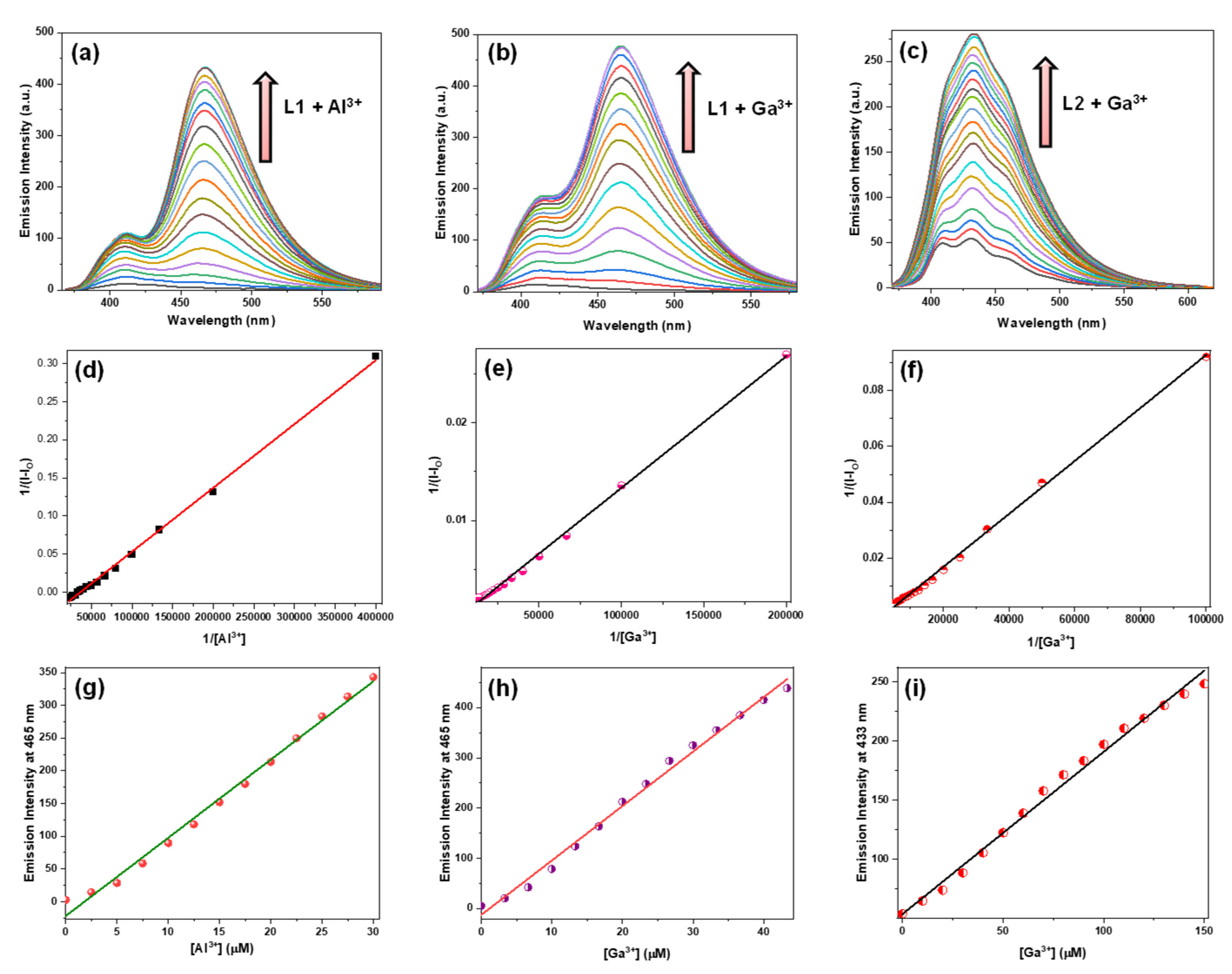
3.5. Detection of Al3+ and Ga3+ Ions by Fluorescence Spectral Studies
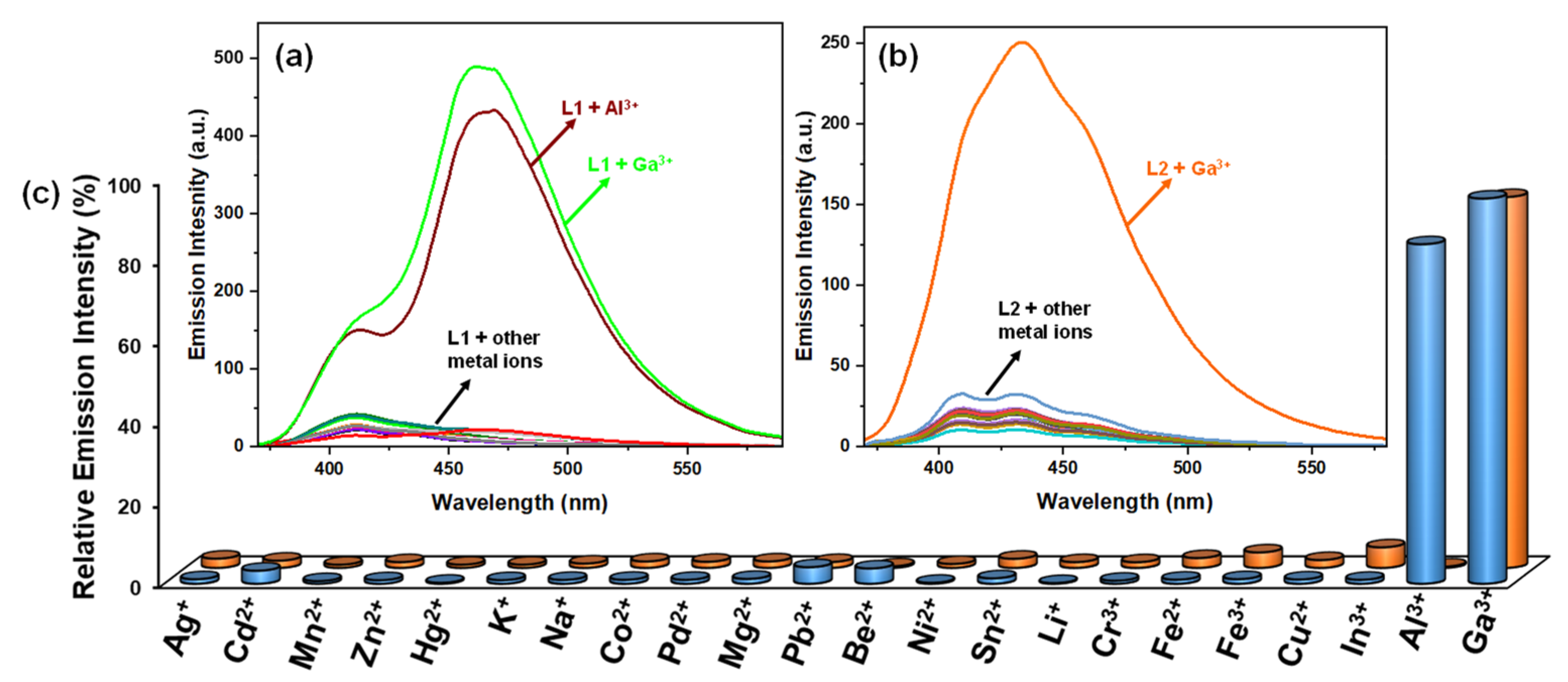
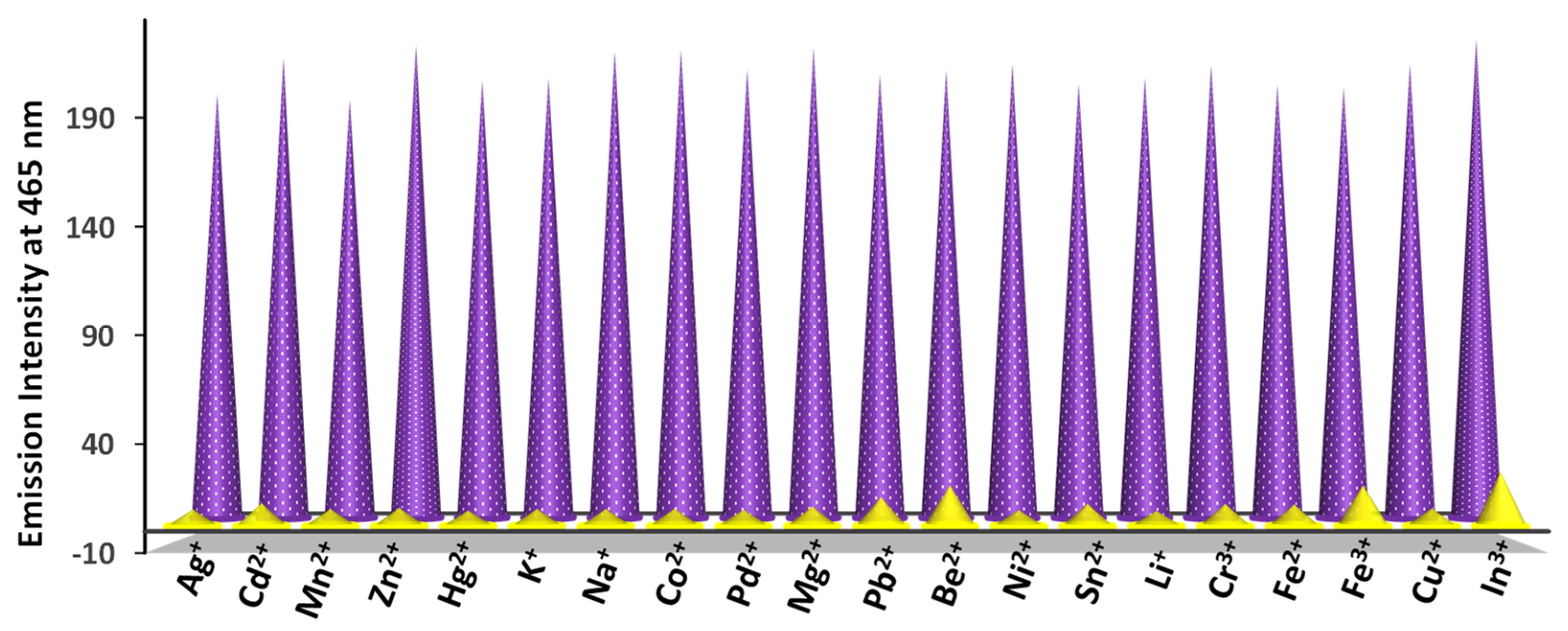
3.6. Selectivity, Lifetime, and Quantum Yield Measurements
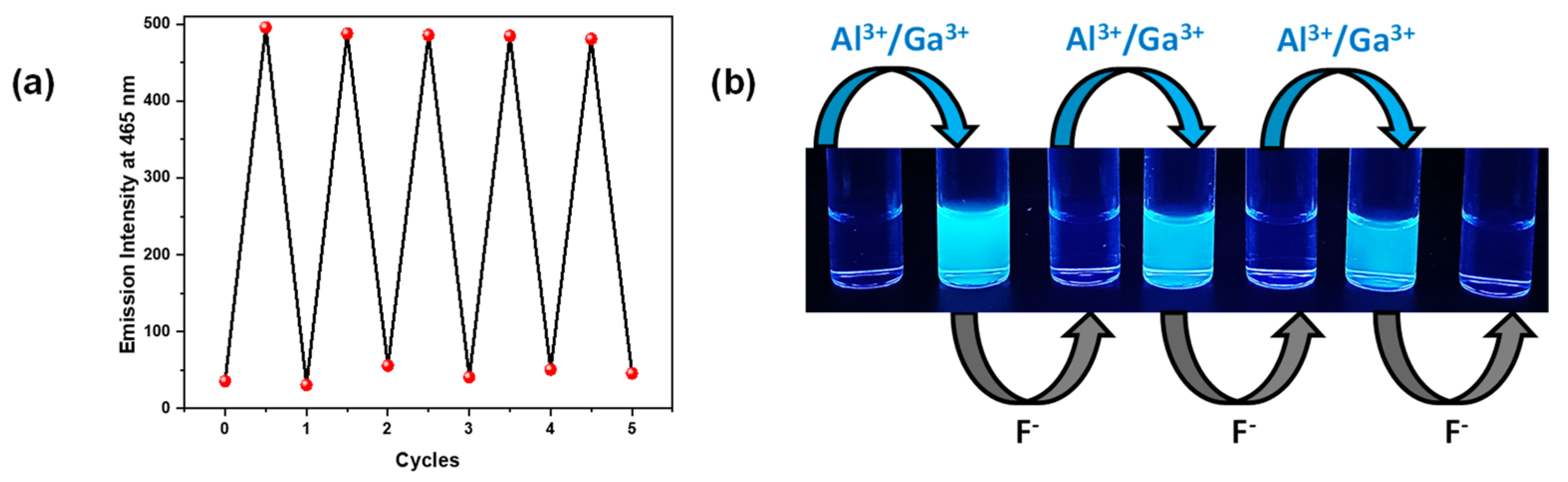
3.7. Reversibility Studies
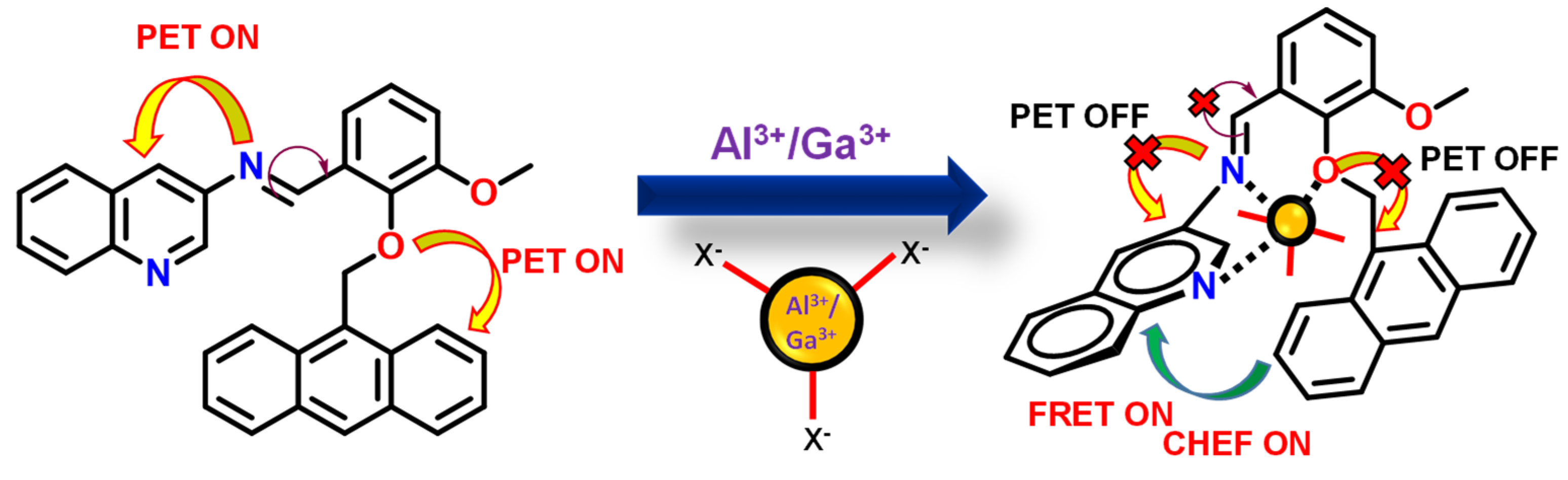
3.8. Mode of Binding
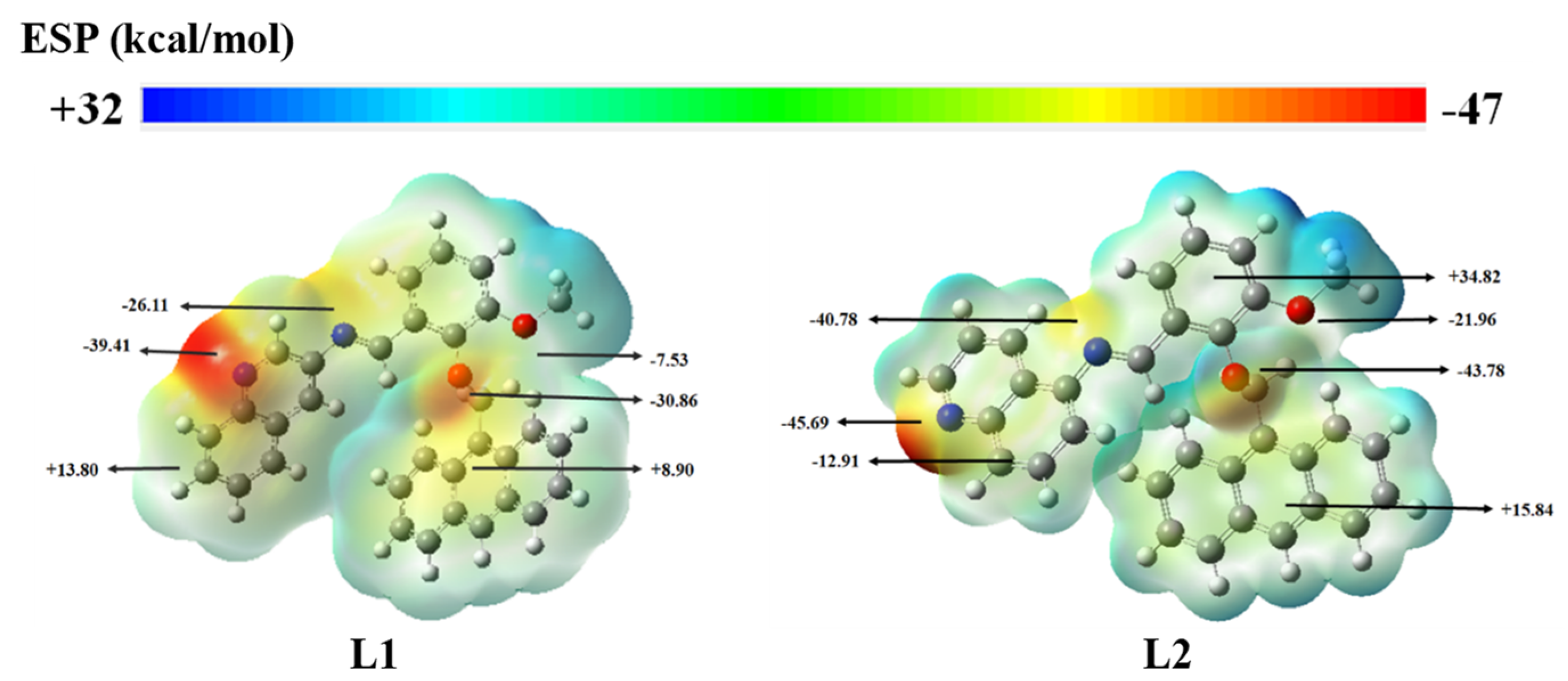
3.9. DFT Studies
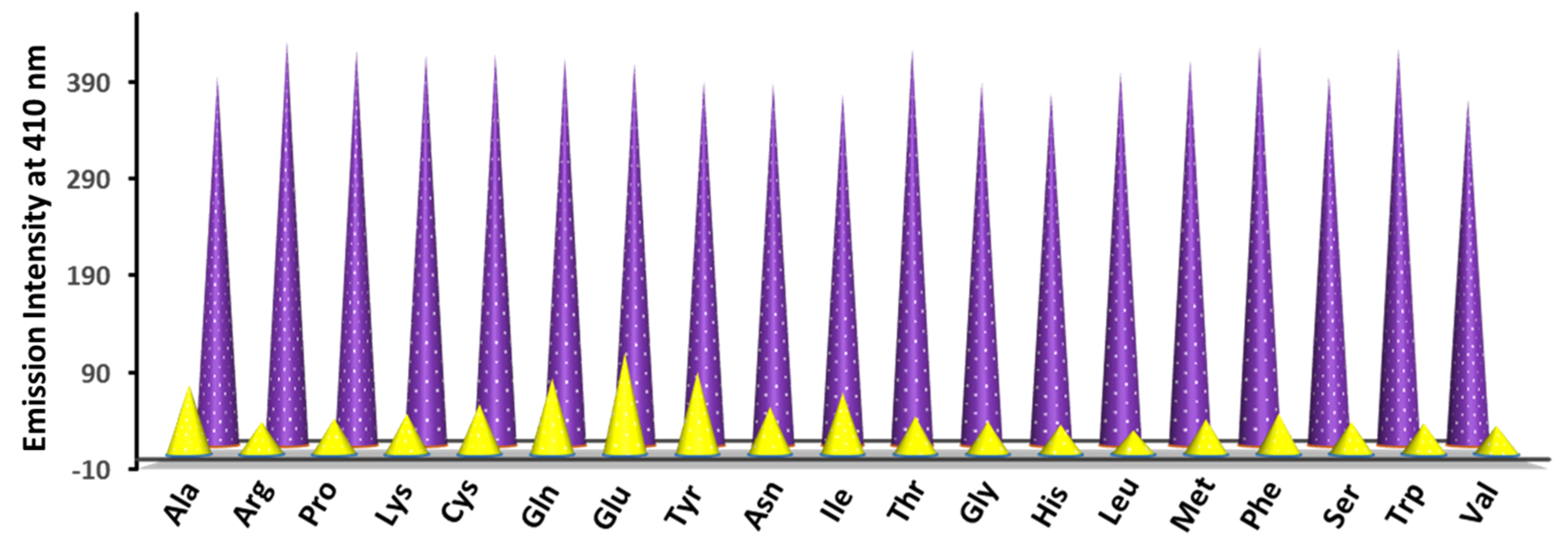
3.10. Detection of Aspartic Acid
3.11. Low-Cost Detection Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balamurugan, G.; Velmathi, S.; Thirumalaivasan, N.; Wu, S.P. New Phenazine Based AIE Probes for Selective Detection of Aluminium(III) Ions in Presence of Other Trivalent Metal Ions in Living Cells. Analyst 2017, 142, 4721–4726. [Google Scholar] [CrossRef] [PubMed]
- Manna, A.; Sain, D.; Guchhait, N.; Goswami, S. FRET Based Selective and Ratiometric Detection of Al(III) with Live-Cell Imaging. New J. Chem. 2017, 41, 14266–14271. [Google Scholar] [CrossRef]
- Wang, H.; Xu, X.; Yin, J.; Zhang, Z.; Xue, L. A Highly Selective “Turn-On” Fluorescent Sensor for Aluminum Ion Detection in Aqueous Solution Based on Imidazo[2,1-b]Thiazole Schiff Base. ChemistrySelect 2021, 6, 6454–6459. [Google Scholar] [CrossRef]
- Kumar, V.; Kumar, P.; Kumar, S.; Singhal, D.; Gupta, R. Turn-On Fluorescent Sensors for the Selective Detection of Al3+ (and Ga3+) and PPi Ions. Inorg. Chem. 2019, 58, 10364–10376. [Google Scholar] [CrossRef]
- Purwanti, I.F.; Kurniawan, S.B.; Ismail, N.I.; Imron, M.F.; Abdullah, S.R.S. Aluminium Removal and Recovery from Wastewater and Soil Using Isolated Indigenous Bacteria. J. Environ. Manage. 2019, 249, 109412. [Google Scholar] [CrossRef]
- Roy, A.; Dey, S.; Roy, P. A Ratiometric Chemosensor for Al3+ Based on Naphthalene-Quinoline Conjugate with the Resultant Complex as Secondary Sensor for F−: Interpretation of Molecular Logic Gates. Sens. Actuators B Chem. 2016, 237, 628–642. [Google Scholar] [CrossRef]
- Cai, D.-G.; Qiu, C.-Q.; Zhu, Z.-H.; Zheng, T.-F.; Wei, W.-J.; Chen, J.-L.; Liu, S.-J.; Wen, H.-R. Fabrication and DFT Calculation of Amine-Functionalized Metal–Organic Framework as a Turn-On Fluorescence Sensor for Fe3+ and Al3+ Ions. Inorg. Chem. 2022, 61, 14770–14777. [Google Scholar] [CrossRef]
- Chai, B.-L.; Yao, S.-L.; Xie, X.; Xu, H.; Zheng, T.-F.; Li, J.-Y.; Chen, J.-L.; Liu, S.-J.; Wen, H.-R. Luminescent Metal–Organic Framework-Based Fluorescence Turn-On and Red-Shift Sensor toward Al3+ and Ga3+: Experimental Study and DFT Calculation. Cryst. Growth Des. 2022, 22, 277–284. [Google Scholar] [CrossRef]
- Liu, Q.; Liu, Y.; Xing, Z.; Huang, Y.; Ling, L.; Mo, X. A Novel Dual-Function Probe for Fluorescent Turn-on Recognition and Differentiation of Al3+ and Ga3+ and Its Application. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 287, 122076. [Google Scholar] [CrossRef]
- Gupta, A.; Kumar, N. A Review of Mechanisms for Fluorescent “Turn-on” Probes to Detect Al3+ Ions. RSC Adv. 2016, 6, 106413–106434. [Google Scholar] [CrossRef]
- Dudev, T.; Cheshmedzhieva, D.; Doudeva, L. Competition between Abiogenic Al3+ and Native Mg2+, Fe2+ and Zn2+ Ions in Protein Binding Sites: Implications for Aluminum Toxicity. J. Mol. Model. 2018, 24, 55. [Google Scholar] [CrossRef] [PubMed]
- Hazra, A.; Roy, A.; Mukherjee, A.; Maiti, G.P.; Roy, P. Remarkable Difference in Al3+ and Zn2+ Sensing Properties of Quinoline Based Isomers. Dalton Trans. 2018, 47, 13972–13989. [Google Scholar] [CrossRef]
- Tian, J.; Yan, X.; Yang, H.; Tian, F. A Novel Turn-on Schiff-Base Fluorescent Sensor for Aluminum(III) Ions in Living Cells. RSC Adv. 2015, 5, 107012–107019. [Google Scholar] [CrossRef]
- Roy, P. Recent Advances in the Development of Fluorescent Chemosensors for Al3+. Dalton Trans. 2021, 50, 7156–7165. [Google Scholar] [CrossRef]
- Banerjee, S.; Brandão, P.; Saha, A. A Robust Fluorescent Chemosensor for Aluminium Ion Detection Based on a Schiff Base Ligand with an Azo Arm and Application in a Molecular Logic Gate. RSC Adv. 2016, 6, 101924–101936. [Google Scholar] [CrossRef]
- Garg, M.; Sud, D. Tracking the Concentration of Al3+ in the Aqueous System up to the Nanomolar Range Using a Modified Biopolymer Chitosan Based Fluorophore. New J. Chem. 2022, 46, 6946–6955. [Google Scholar] [CrossRef]
- Ghorai, P.; Pal, K.; Karmakar, P.; Saha, A. The Development of Two Fluorescent Chemosensors for the Selective Detection of Zn2+ and Al3+ Ions in a Quinoline Platform by Tuning the Substituents in the Receptor Part: Elucidation of the Structures of the Metal-Bound Chemosensors and Biological Studies. Dalton Trans. 2020, 49, 4758–4773. [Google Scholar] [CrossRef]
- Mishra, S.; Mamidi, P.; Chattopadhyay, S.; Singh, A.K. Economically Viable Multi-Responsive Probes for Fluorimetric Detection of Trace Levels of Ga3+, Al3+ and PPi in near Aqueous Medium. J. Photochem. Photobiol. Chem. 2023, 434, 114225. [Google Scholar] [CrossRef]
- Xiang, H.; Wang, T.; Tang, S.; Wang, Y.; Xiao, N. A Novel Hydrazone-Based Fluorescent “off-on-off” Probe for Relay Sensing of Ga3+ and PPi Ions. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2022, 267, 120510. [Google Scholar] [CrossRef]
- Yang, L.; Li, M.; Wang, Y.; Zhang, Y.; Liu, Z.; Ruan, S.; Wang, Z.; Wang, S. An Isocamphanyl-Based Fluorescent “Turn-on” Probe for Highly Sensitive and Selective Detection of Ga3+ and Application In Vivo and In Vitro. Analyst 2021, 146, 7294–7305. [Google Scholar] [CrossRef]
- Zhu, Y.-Y.; Wu, X.-D.; Gu, S.-X.; Pu, L. Free Amino Acid Recognition: A Bisbinaphthyl-Based Fluorescent Probe with High Enantioselectivity. J. Am. Chem. Soc. 2019, 141, 175–181. [Google Scholar] [CrossRef]
- Wang, B.; Han, J.; Bojanowski, N.M.; Bender, M.; Ma, C.; Seehafer, K.; Herrmann, A.; Bunz, U.H.F. An Optimized Sensor Array Identifies All Natural Amino Acids. ACS Sens. 2018, 3, 1562–1568. [Google Scholar] [CrossRef]
- Yang, A.-F.; Hou, S.-L.; Shi, Y.; Yang, G.-L.; Qin, D.-B.; Zhao, B. Stable Lanthanide–Organic Framework as a Luminescent Probe To Detect Both Histidine and Aspartic Acid in Water. Inorg. Chem. 2019, 58, 6356–6362. [Google Scholar] [CrossRef] [PubMed]
- Guria, S.; Ghosh, A.; Manna, K.; Pal, A.; Adhikary, A.; Adhikari, S. Rapid Detection of Aspartic Acid and Glutamic Acid in Water by BODIPY-Based Fluorescent Probe: Live-Cell Imaging and DFT Studies. Dye. Pigment. 2019, 168, 111–122. [Google Scholar] [CrossRef]
- Ji, G.; Zheng, T.; Gao, X.; Liu, Z. A Highly Selective Turn-on Luminescent Logic Gates Probe Based on Post-Synthetic MOF for Aspartic Acid Detection. Sens. Actuators B Chem. 2019, 284, 91–95. [Google Scholar] [CrossRef]
- Zhu, Y.; Cui, M.; Ma, J.; Zhang, Q. Fluorescence Detection of D-Aspartic Acid Based on Thiol-Ene Cross-Linked Molecularly Imprinted Optical Fiber Probe. Sens. Actuators B Chem. 2020, 305, 127323. [Google Scholar] [CrossRef]
- Elamathi, C.; Butcher, R.J.; Mohankumar, A.; Sundararaj, P.; Elango, K.P.; Kalaivani, P.; Prabhakaran, R. Dual Sensing of Methionine and Aspartic Acid in Aqueous Medium by a Quinoline-Based Fluorescent Probe. Dalton Trans. 2021, 50, 8820–8830. [Google Scholar] [CrossRef]
- Nasomphan, W.; Tangboriboonrat, P.; Tanapongpipat, S.; Smanmoo, S. Selective Fluorescent Detection of Aspartic Acid and Glutamic Acid Employing Dansyl Hydrazine Dextran Conjugate. J. Fluoresc. 2014, 24, 7–11. [Google Scholar] [CrossRef]
- Ranjani, M.; Kalaivani, P.; Dallemer, F.; Selvakumar, S.; Kalpana, T.; Prabhakaran, R. Fluorescent Cu(II) Complex as Chemosensor for the Detection of l-Aspartic Acid with High Selectivity and Sensitivity. Inorg. Chim. Acta 2022, 530, 120683. [Google Scholar] [CrossRef]
- Tian, J.; Wang, Y.; Chen, Y.; Zhao, F.; Jiang, Y.; Yu, S.; Yu, X.; Pu, L. Chemoselective and Enantioselective Fluorescent Recognition of Glutamic and Aspartic Acids. Chem. Commun. 2020, 56, 15012–15015. [Google Scholar] [CrossRef]
- Ren, T.-L.; Zhang, X.-N.; Hu, J.-J.; Wen, H.-R.; Liu, S.-J.; Peng, Y. Stable Terbium Metal–Organic Framework with Turn-on and Blue-Shift Fluorescence Sensing for Acidic Amino Acids (L-Aspartate and L-Glutamine) and Cations (Al3+ and Ga3+). Dalton Trans. 2022, 51, 14858–14864. [Google Scholar] [CrossRef] [PubMed]
- Goyal, H.; Kumar, V.; Saini, A.K.; Kedawat, G.; Gupta, B.K.; Gupta, R. A Multifunctional Schiff Base with Aggregation-Induced Enhanced Emission, Gelation, and Mechanochromic Properties for Anti-Counterfeiting Applications. Mater. Today Chem. 2023, 27, 101306. [Google Scholar] [CrossRef]
- Khanra, S.; Ta, S.; Ghosh, M.; Chatterjee, S.; Mukherjee, P.; Das, D. Al3+ Triggered Aggregation Induced Emission of an Anthracence Based Azine Derivative in SDS Medium. New J. Chem. 2020, 44, 8477–8485. [Google Scholar] [CrossRef]
- Mao, L.; Liu, Y.; Yang, S.; Li, Y.; Zhang, X.; Wei, Y. Recent Advances and Progress of Fluorescent Bio-/Chemosensors Based on Aggregation-Induced Emission Molecules. Dye. Pigment. 2019, 162, 611–623. [Google Scholar] [CrossRef]
- R, S.G.; Pandey, M.; Chakravarthy, A.S.J. Review on New Horizons of Aggregation Induced Emission: From Design to Development. Mater. Chem. Front. 2021, 5, 1541–1584. [Google Scholar] [CrossRef]
- Würthner, F. Aggregation-Induced Emission (AIE): A Historical Perspective. Angew. Chem. Int. Ed. 2020, 59, 14192–14196. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Meng, Q.; Yang, Y.; Zhong, S.; Zhang, R.; Fang, Y.; Gao, Y.; Cui, X. Schiff Base Aggregation-Induced Emission Luminogens for Sensing Applications: A Review. ACS Sens. 2022, 7, 2521–2536. [Google Scholar] [CrossRef] [PubMed]
- Munir, F.; Waseem, M.T.; Khan, Z.A.; Majeed, S.; Farooq, U.; Shahzad, S.A. Synthesis of AIEE Active Triazine Based New Fluorescent and Colorimetric Probes: A Reversible Mechanochromism and Sequential Detection of Picric Acid and Ciprofloxacin. J. Photochem. Photobiol. Chem. 2022, 429, 113921. [Google Scholar] [CrossRef]
- Parambil, A.R.U.; P, K.; Silswal, A.; Koner, A.L. Water-Soluble Optical Sensors: Keys to Detect Aluminium in Biological Environment. RSC Adv. 2022, 12, 13950–13970. [Google Scholar] [CrossRef]
- Xu, Z.; Liu, Y.; Wang, R.; Zhang, J.; Cheng, J.; Chen, S.; Miao, M.; Zhang, D. AIEE Based “Turn-on” Fluorescent Sensor for Al3+ Ions and Induced Tetraphenylethene Self-Assemblies. Org. Electron. 2020, 85, 105820. [Google Scholar] [CrossRef]
- Nguyen, H.L.; Kumar, N.; Audibert, J.-F.; Ghasemi, R.; Lefevre, J.-P.; Ha-Thi, M.-H.; Mongin, C.; Leray, I. Water-Soluble Aluminium Fluorescent Sensor Based on Aggregation-Induced Emission Enhancement. New J. Chem. 2019, 43, 15302–15310. [Google Scholar] [CrossRef]
- Armarego, W.L.F. Purification of Laboratory Chemicals; Butterworth-Heinemann: Oxford, UK, 2017; ISBN 978-0-12-805456-7. [Google Scholar]
- CrysAlisPro; Version 1.171.33.49b; Oxford Diffraction Ltd.: Abingdon, UK, 2009.
- Altomare, A.; Burla, M.C.; Camalli, M.; Cascarano, G.L.; Giacovazzo, C.; Guagliardi, A.; Moliterni, A.G.G.; Polidori, G.; Spagna, R. SIR97: A New Tool for Crystal Structure Determination and Refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. A Short History of SHELX. Acta Crystallogr. A 2008, 64, 112–122. [Google Scholar] [CrossRef]
- Farrugia, L.J. WinGX, v. 1.70, An Integrated System of Windows Programs for the Solution, Refinement and Analysis of Single-Crystal X-ray Diffraction Data; Department of Chemistry, University of Glasgow: Glasgow, Scotland, 2003. [Google Scholar]
- Hohenberg, P.; Kohn, W. Inhomogeneous Electron Gas. Phys. Rev. 1964, 136, B864–B871. [Google Scholar] [CrossRef]
- Frisch, M.J.; Trucks, G.W.; Schlegel, H.B.; Scuseria, G.E.; Robb, M.A.; Cheeseman, J.R.; Scalmani, G.; Barone, V.; Petersson, G.A.; Nakatsuji, H.; et al. Gaussian 09 Rev. A.01; Gaussian, Inc.: Wallingford, CT, USA, 2009. [Google Scholar]
- Chiodo, S.; Russo, N.; Sicilia, E. LANL2DZ Basis Sets Recontracted in the Framework of Density Functional Theory. J. Chem. Phys. 2006, 125, 104107. [Google Scholar] [CrossRef] [PubMed]
- Blaudeau, J.-P.; McGrath, M.P.; Curtiss, L.A.; Radom, L. Extension of Gaussian-2 (G2) Theory to Molecules Containing Third-Row Atoms K and Ca. J. Chem. Phys. 1997, 107, 5016–5021. [Google Scholar] [CrossRef]
- Tomasi, J.; Mennucci, B.; Cammi, R. Quantum Mechanical Continuum Solvation Models. Chem. Rev. 2005, 105, 2999–3094. [Google Scholar] [CrossRef]
- Dennington, R.; Keith, T.A.; Millam, J.M. GaussView, Version 6; Shawnee Mission, K.; Semichem Inc.: Shawnee, KS, USA, 2016. [Google Scholar]
- Hanwell, M.D.; Curtis, D.E.; Lonie, D.C.; Vandermeersch, T.; Zurek, E.; Hutchison, G.R. Avogadro: An Advanced Semantic Chemical Editor, Visualization, and Analysis Platform. J. Cheminformatics 2012, 4, 17. [Google Scholar] [CrossRef] [PubMed]
- Benesi, H.A.; Hildebrand, J.H. A Spectrophotometric Investigation of the Interaction of Iodine with Aromatic Hydrocarbons. J. Am. Chem. Soc. 1949, 71, 2703–2707. [Google Scholar] [CrossRef]
- Kumar, V.; Singh, D.; Kumar, P.; Chaudhary, G.; Singh, A.P.; Gupta, R. Turn-on Fluorescent Detection of Nickel and Zinc Ions by Two Related Chemosensors Containing Naphthalimide Ring(s). J. Mol. Struct. 2022, 1261, 132901. [Google Scholar] [CrossRef]
- Singh, D.; Tomar, S.; Singh, S.; Chaudhary, G.; Singh, A.P.; Gupta, R. A Fluorescent PH Switch Probe for the ‘Turn-on’ Dual-Channel Discriminative Detection of Magnesium and Zinc Ions. J. Photochem. Photobiol. Chem. 2023, 435, 114334. [Google Scholar] [CrossRef]
- Prabha, D.; Singh, D.; Kumar, P.; Gupta, R. Selective Detection of Picric Acid and Pyrosulfate Ion by Nickel Complexes Offering a Hydrogen-Bonding-Based Cavity. Inorg. Chem. 2021, 60, 17889–17899. [Google Scholar] [CrossRef]
- Singh, D.; Ibrahim, A.; Gupta, R. Coumarin-Based Reversible Fluorescent Chemosensors for the Sequential Detection of Copper and Citrate Ions. ChemistrySelect 2022, 7, e202203326. [Google Scholar] [CrossRef]
- Singh, D.; Ibrahim, A.; Kumar, P.; Gupta, R. Methylene Spacer Mediated Detection Switch Between Copper and Zinc Ions by Two Coumarin-Pyrene Based Chemosensors. ChemistrySelect 2022, 7, e202202574. [Google Scholar] [CrossRef]
- Di Bella, S.; Fragalà, I.; Ledoux, I.; Diaz-Garcia, M.A.; Marks, T.J. Synthesis, Characterization, Optical Spectroscopic, Electronic Structure, and Second-Order Nonlinear Optical (NLO) Properties of a Novel Class of Donor−Acceptor Bis(Salicylaldiminato)Nickel(II) Schiff Base NLO Chromophores. J. Am. Chem. Soc. 1997, 119, 9550–9557. [Google Scholar] [CrossRef]
- Cheng, J.; Wei, K.; Ma, X.; Zhou, X.; Xiang, H. Synthesis and Photophysical Properties of Colorful Salen-Type Schiff Bases. J. Phys. Chem. C 2013, 117, 16552–16563. [Google Scholar] [CrossRef]
- Luo, H.; Li, N.; Liu, L.; Wang, H.; He, F. Synthesis of New AIEE-Active Chalcones for Imaging of Mitochondria in Living Cells and Zebrafish In Vivo. Int. J. Mol. Sci. 2021, 22, 8949. [Google Scholar] [CrossRef] [PubMed]
- Islam, A.S.M.; Sasmal, M.; Maiti, D.; Dutta, A.; Show, B.; Ali, M. Design of a Pyrene Scaffold Multifunctional Material: Real-Time Turn-On Chemosensor for Nitric Oxide, AIEE Behavior, and Detection of TNP Explosive. ACS Omega 2018, 3, 10306–10316. [Google Scholar] [CrossRef] [PubMed]
- Goyal, H.; Pachisia, S.; Gupta, R. Systematic Design of a Low-Molecular-Weight Gelator and Its Application in the Sensing and Retention of Residual Antibiotics. Cryst. Growth Des. 2020, 20, 6117–6128. [Google Scholar] [CrossRef]
- Shyamal, M.; Mazumdar, P.; Maity, S.; Sahoo, G.P.; Salgado-Morán, G.; Misra, A. Pyrene Scaffold as Real-Time Fluorescent Turn-on Chemosensor for Selective Detection of Trace-Level Al(III) and Its Aggregation-Induced Emission Enhancement. J. Phys. Chem. A 2016, 120, 210–220. [Google Scholar] [CrossRef]
- Kathiravan, A.; Sundaravel, K.; Jaccob, M.; Dhinagaran, G.; Rameshkumar, A.; Arul Ananth, D.; Sivasudha, T. Pyrene Schiff Base: Photophysics, Aggregation Induced Emission, and Antimicrobial Properties. J. Phys. Chem. B 2014, 118, 13573–13581. [Google Scholar] [CrossRef] [PubMed]
- Jinbo, D.; Imato, K.; Ooyama, Y. Fluorescent Sensor for Water Based on Photo-Induced Electron Transfer and Förster Resonance Energy Transfer: Anthracene-(Aminomethyl)Phenylboronic Acid Ester-BODIPY Structure. RSC Adv. 2019, 9, 15335–15340. [Google Scholar] [CrossRef]
- Kumar, S.; Kishan, R.; Kumar, P.; Pachisia, S.; Gupta, R. Size-Selective Detection of Picric Acid by Fluorescent Palladium Macrocycles. Inorg. Chem. 2018, 57, 1693–1697. [Google Scholar] [CrossRef]
- Sudheer; Kumar, V.; Kumar, P.; Gupta, R. Detection of Al3+ and Fe3+ Ions by Nitrobenzoxadiazole Bearing Pyridine-2,6-Dicarboxamide Based Chemosensors: Effect of Solvents on Detection. New J. Chem. 2020, 44, 13285–13294. [Google Scholar] [CrossRef]
- Kumar, P.; Kumar, V.; Pandey, S.; Gupta, R. Detection of Sulfide Ion and Gaseous H2S Using a Series of Pyridine-2,6-Dicarboxamide Based Scaffolds. Dalton Trans. 2018, 47, 9536–9545. [Google Scholar] [CrossRef] [PubMed]
- Kumar, P.; Kumar, V.; Gupta, R. Arene-Based Fluorescent Probes for the Selective Detection of Iron. RSC Adv. 2015, 5, 97874–97882. [Google Scholar] [CrossRef]
- Kumar Goshisht, M.; Kumar Patra, G.; Tripathi, N. Fluorescent Schiff Base Sensors as a Versatile Tool for Metal Ion Detection: Strategies, Mechanistic Insights, and Applications. Mater. Adv. 2022, 3, 2612–2669. [Google Scholar] [CrossRef]
- Gupta, A.S.; Paul, K.; Luxami, V. A New ‘Turn-on’ PET-CHEF Based Fluorescent Sensor for Al3+ and CN− Ions: Applications in Real Samples. Anal. Methods 2018, 10, 983–990. [Google Scholar] [CrossRef]
- Wang, P.; Meng, F.; Su, H.; Liu, L.; Khan, M.A.; Li, H. A Highly Selective “Turn-on” Water-Soluble Fluorescent Sensor for Gallium Ion Detection. RSC Adv. 2021, 11, 19747–19754. [Google Scholar] [CrossRef]
- Noh, J.Y.; Kim, S.; Hwang, I.H.; Lee, G.Y.; Kang, J.; Kim, S.H.; Min, J.; Park, S.; Kim, C.; Kim, J. Solvent-Dependent Selective Fluorescence Assay of Aluminum and Gallium Ions Using Julolidine-Based Probe. Dye. Pigment. 2013, 99, 1016–1021. [Google Scholar] [CrossRef]
- Tabaraki, R.; Abdi, O. Microwave Assisted Synthesis of N-Doped Carbon Dots: An Easy, Fast and Cheap Sensor for Determination of Aspartic Acid in Sport Supplements. J. Fluoresc. 2019, 29, 751–756. [Google Scholar] [CrossRef]
- Zhou, Y.; Yoon, J. Recent Progress in Fluorescent and Colorimetric Chemosensors for Detection of Amino Acids. Chem. Soc. Rev. 2012, 41, 52–67. [Google Scholar] [CrossRef] [PubMed]
- Goyal, H.; Gupta, R. Sensing and Formation of a Stable Gel in the Presence of Picric Acid by a Low-Molecular-Weight-Gelator. J. Indian Chem. Soc. 2022, 99, 100521. [Google Scholar] [CrossRef]
- Kumar, G.; Pachisia, S.; Kumar, P.; Kumar, V.; Gupta, R. Zn- and Cd-Based Coordination Polymers Offering H-Bonding Cavities: Highly Selective Sensing of S2O72− and Fe3+ Ions. Chem.—Asian J. 2019, 14, 4594–4600. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wei, T.; Zhang, Z.; Chen, T.; Li, J.; Qiang, J.; Lv, J.; Wang, F.; Chen, X. A Benzothiazole-Based Fluorescent Probe for Ratiometric Detection of Al3+ in Aqueous Medium and Living Cells. Ind. Eng. Chem. Res. 2017, 56, 12267–12275. [Google Scholar] [CrossRef]
- Guo, A.; Zhu, R.; Zhang, Z.; Ren, Y.; Dong, J.; Feng, L. A “Turn-on” Fluorescent Chemosensor for Aluminum Ion and Cell Imaging Application. Spectrochim. Ind. Eng. Chem. Res. 2016, 153, 530–534. [Google Scholar] [CrossRef]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Goyal, H.; Annan, I.; Ahluwalia, D.; Bag, A.; Gupta, R. Discriminative ‘Turn-on’ Detection of Al3+ and Ga3+ Ions as Well as Aspartic Acid by Two Fluorescent Chemosensors. Sensors 2023, 23, 1798. https://doi.org/10.3390/s23041798
Goyal H, Annan I, Ahluwalia D, Bag A, Gupta R. Discriminative ‘Turn-on’ Detection of Al3+ and Ga3+ Ions as Well as Aspartic Acid by Two Fluorescent Chemosensors. Sensors. 2023; 23(4):1798. https://doi.org/10.3390/s23041798
Chicago/Turabian StyleGoyal, Hina, Ibrahim Annan, Deepali Ahluwalia, Arijit Bag, and Rajeev Gupta. 2023. "Discriminative ‘Turn-on’ Detection of Al3+ and Ga3+ Ions as Well as Aspartic Acid by Two Fluorescent Chemosensors" Sensors 23, no. 4: 1798. https://doi.org/10.3390/s23041798
APA StyleGoyal, H., Annan, I., Ahluwalia, D., Bag, A., & Gupta, R. (2023). Discriminative ‘Turn-on’ Detection of Al3+ and Ga3+ Ions as Well as Aspartic Acid by Two Fluorescent Chemosensors. Sensors, 23(4), 1798. https://doi.org/10.3390/s23041798






