Interactive Cardio System for Healthcare Improvement
Abstract
1. Introduction
- -
- Linear methods: including time domain, frequency domain, and time-frequency domain methods.
- -
- Nonlinear methods: including fractal methods, Hurst exponent determination, Detrended Fluctuation Analysis, Poincaré plot, and others.
1.1. Background
1.2. The Purpose of This Article
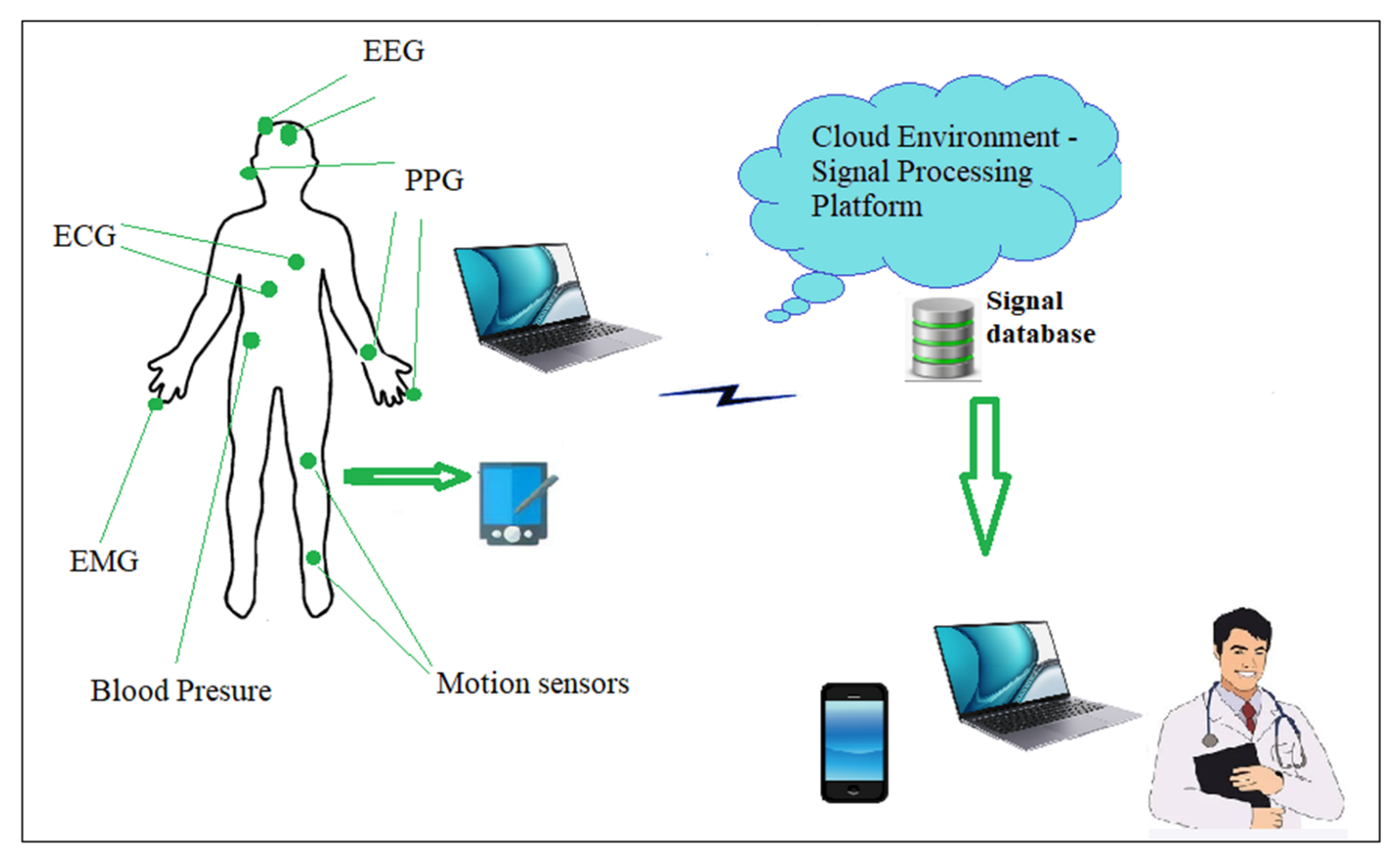
2. Materials and Methods
2.1. Database and Preprocessing
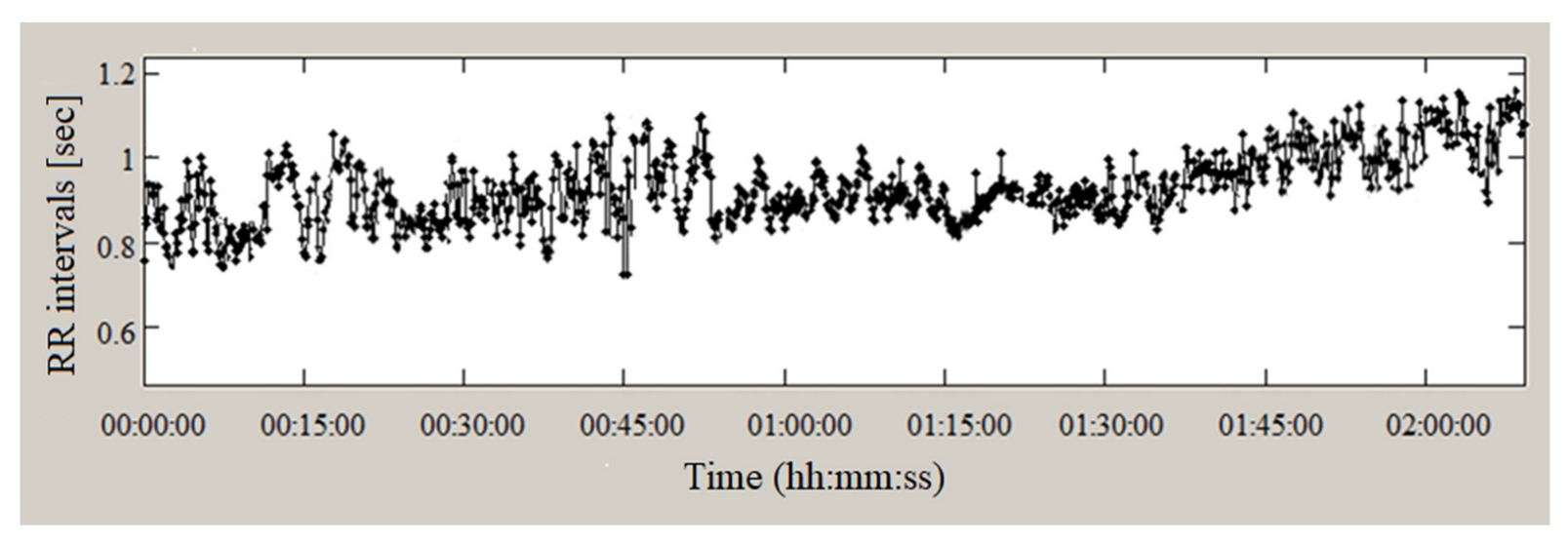
2.2. PPG Sensors to Heart Rate Record
2.3. Mathematical Methods of Cardio Analysis
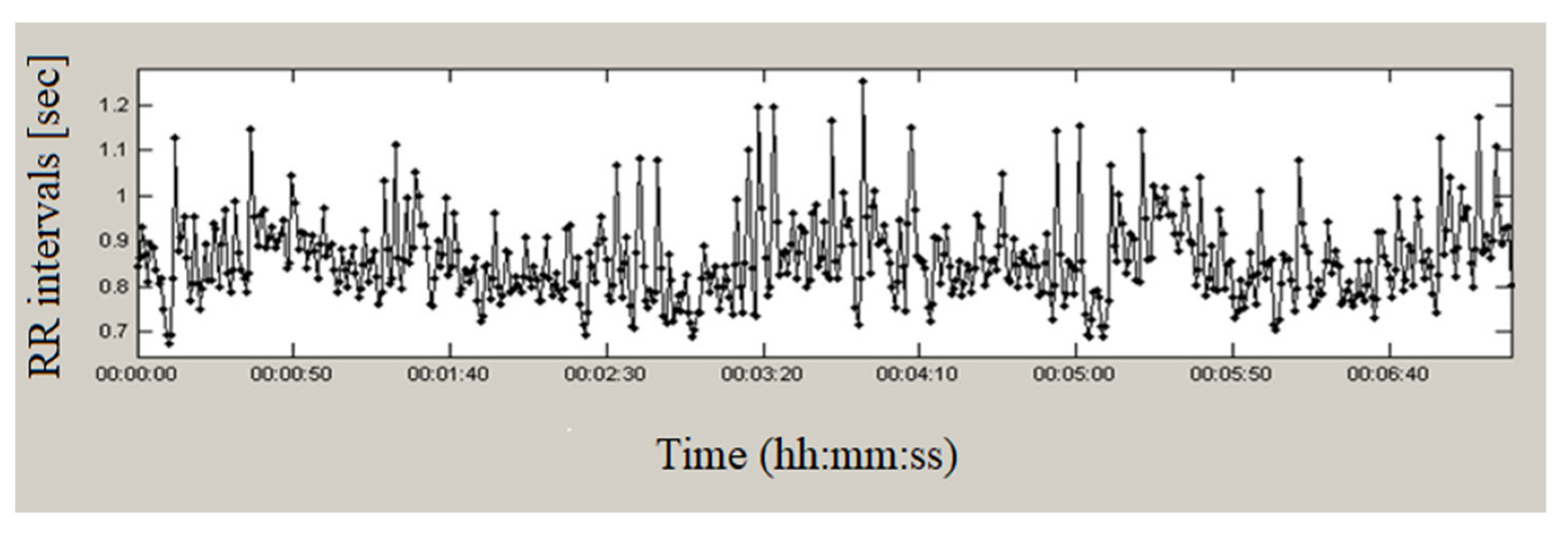
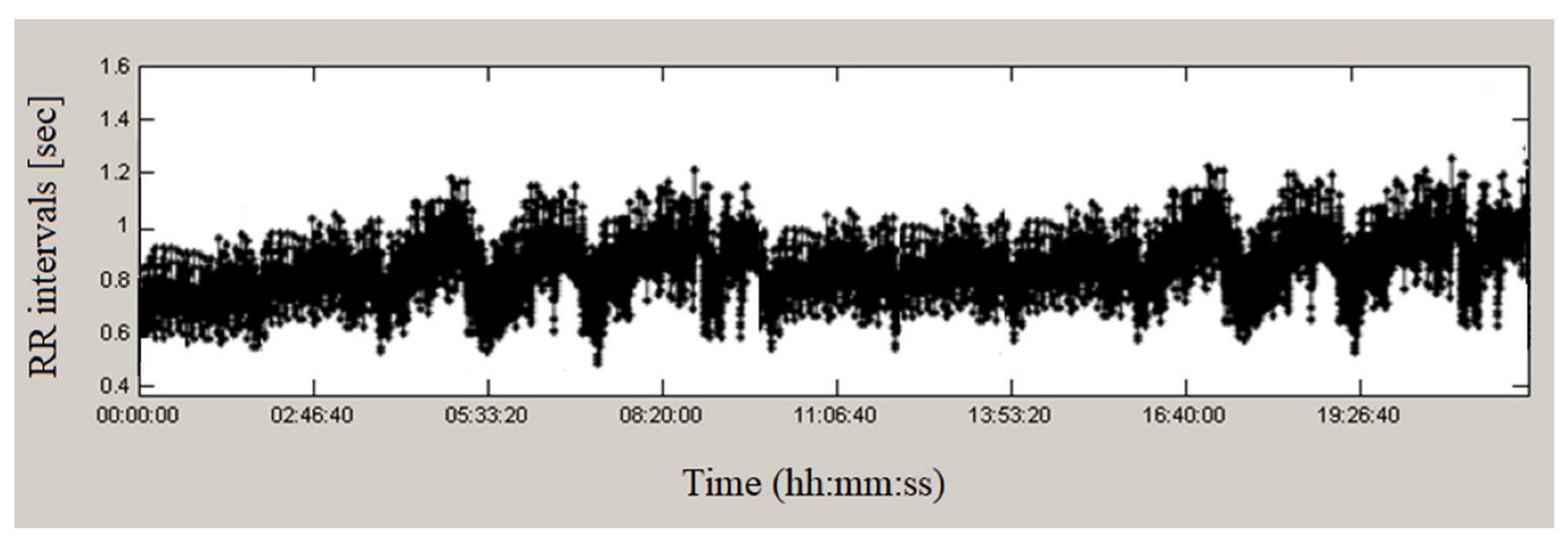
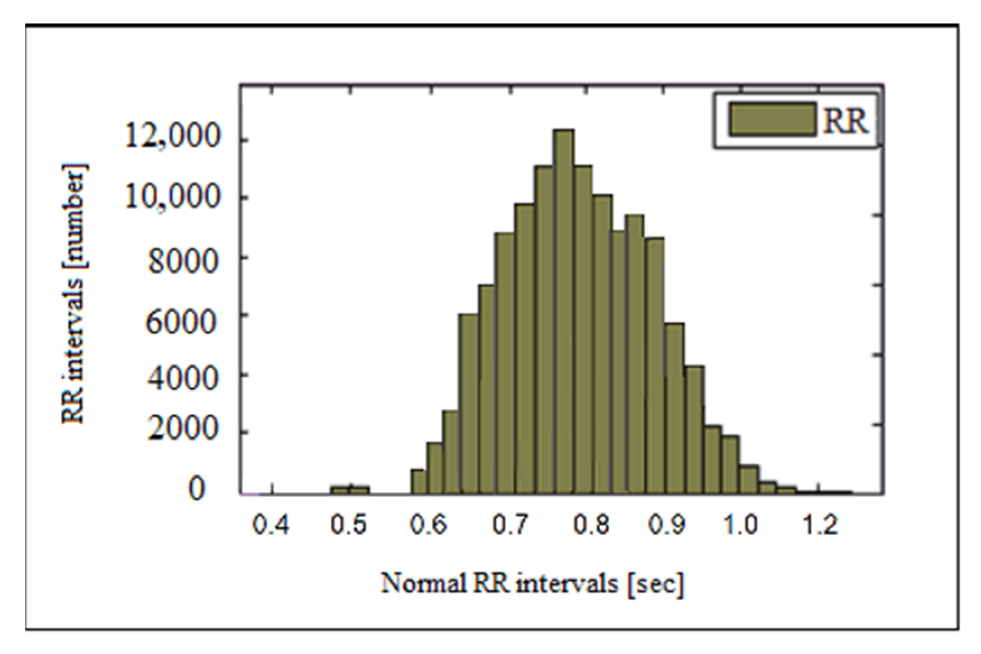
2.3.1. Analysis in the Time Domain
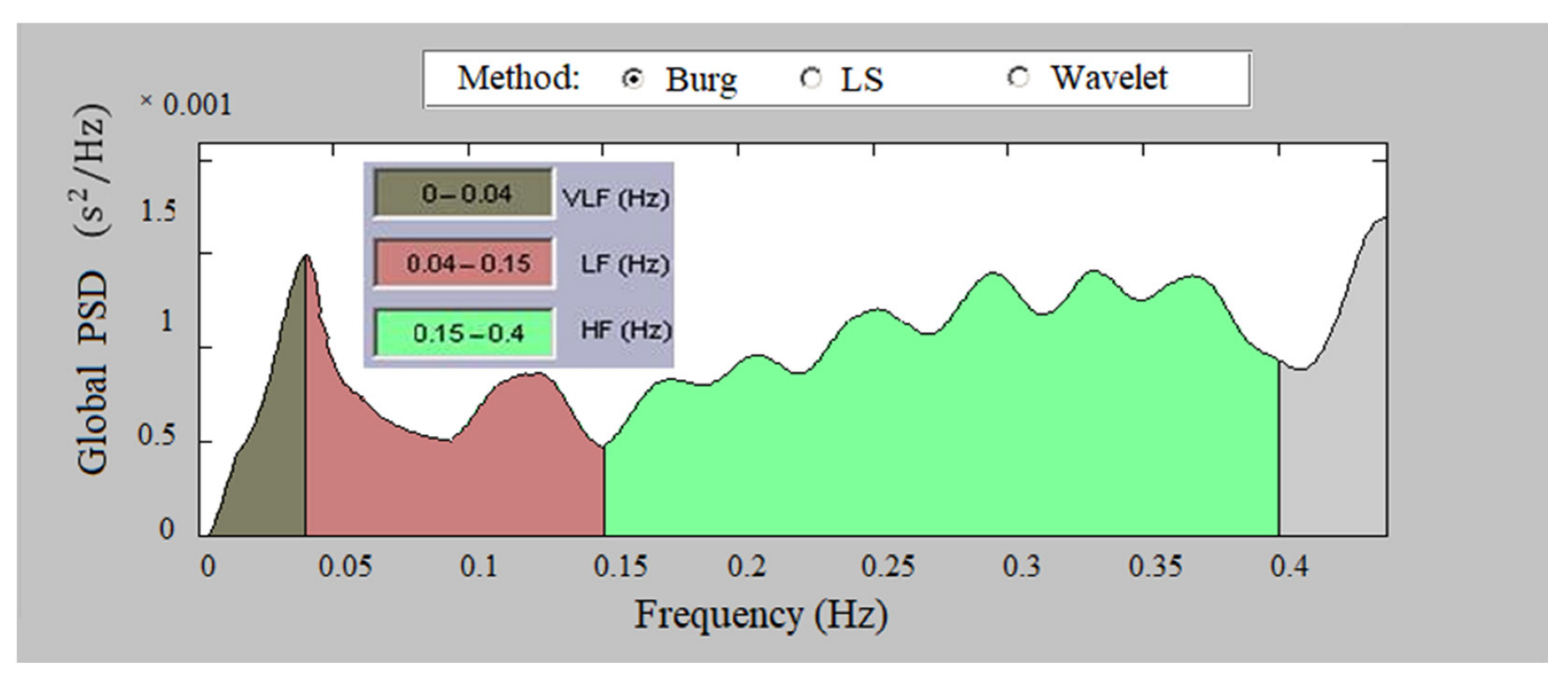
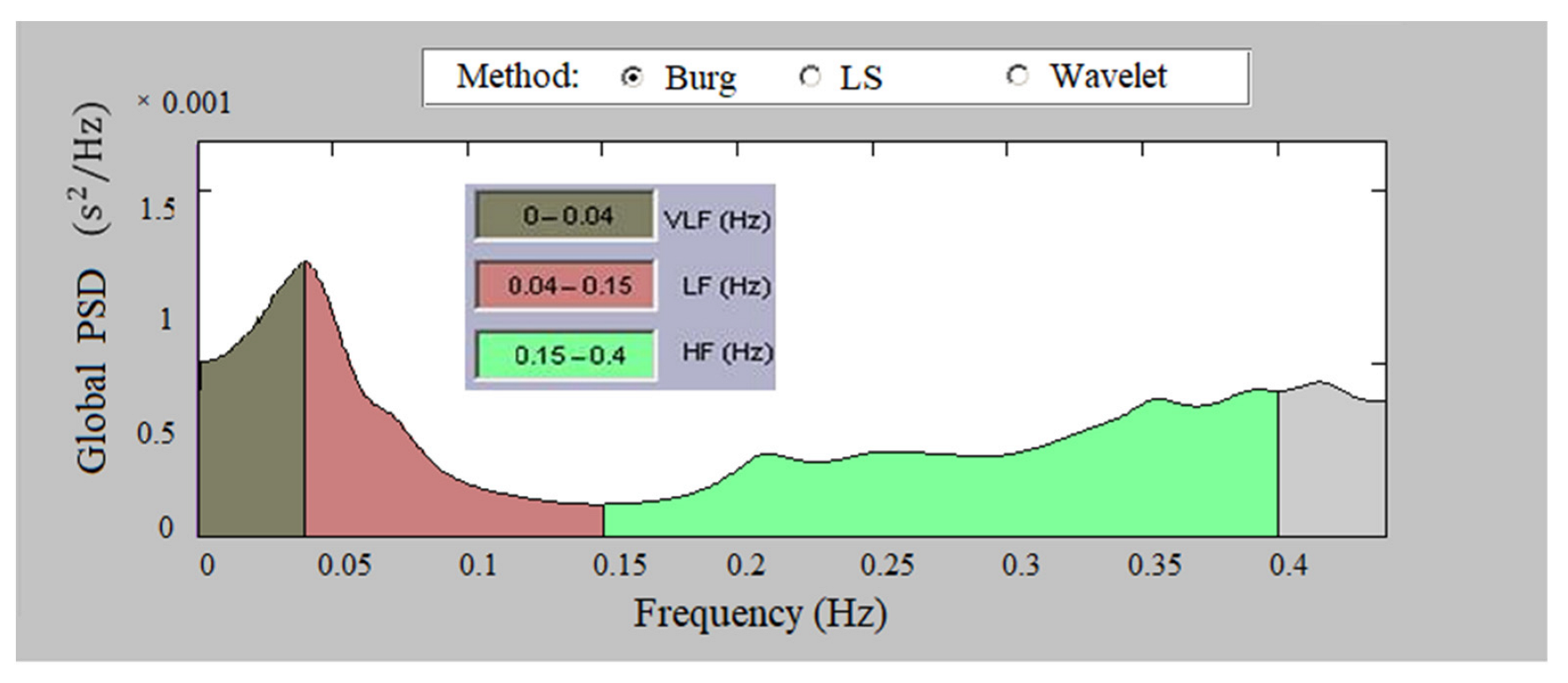
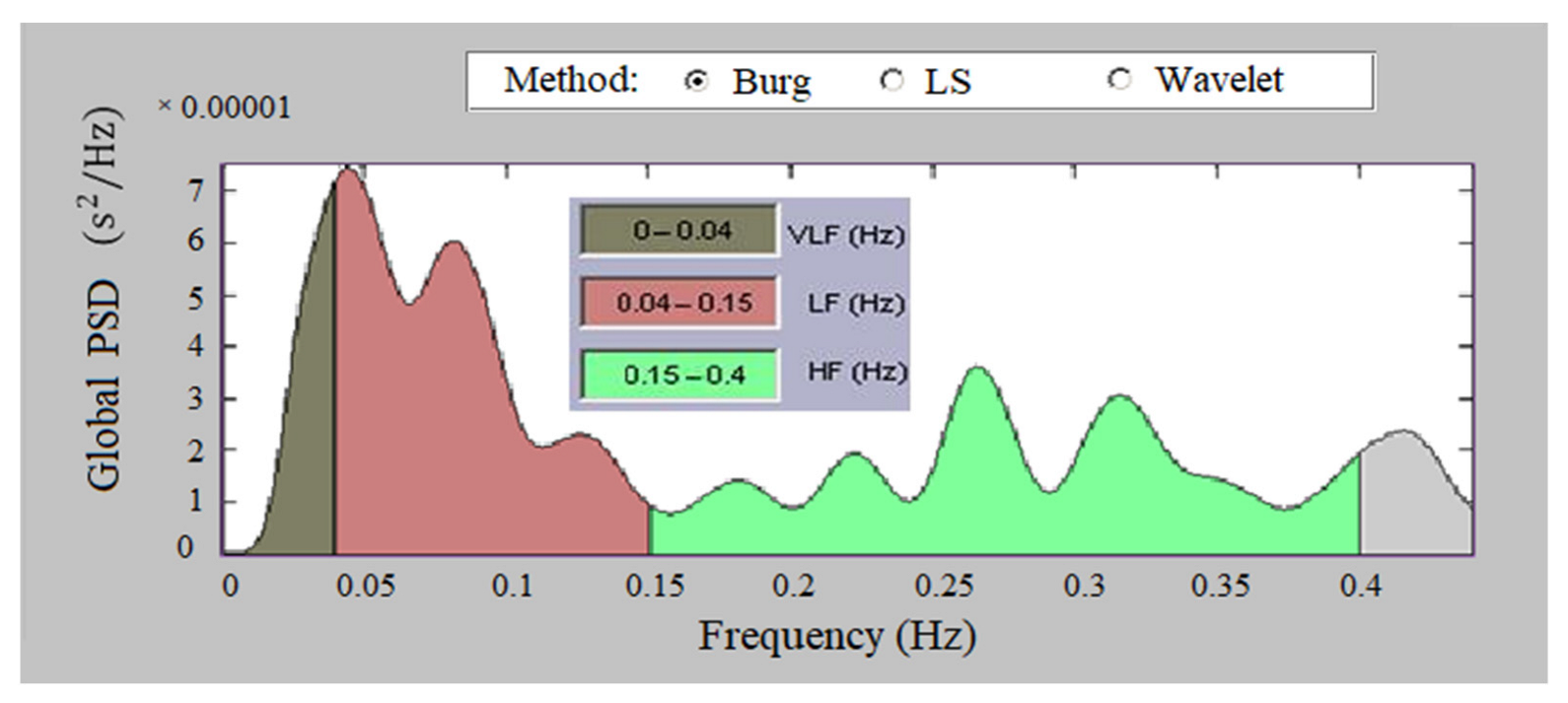
2.3.2. Analysis in the Frequency Domain
- Ultra Low Frequency, ULF (0–0.003 Hz)—reflects the change of day and night;
- Very Low Frequency, VLF (0.003–0.04 Hz)—affects the sympathetic nervous system;
- Low Frequency, LF (0.04–0.15 Hz)—affects the sympathetic and parasympathetic nervous system;
- High Frequency, HF (0.15–0.4 Hz)—influences the parasympathetic nervous system and respiratory sinus arrhythmia;
- Total Power—reflects the influence of the two lobes of the nervous system and the overall nervous regulation of cardiac activity.
2.3.3. Analysis in the Time-Frequency Domain
2.3.4. Surface Method
2.3.5. Analysis with Nonlinear Methods
- Determination of the Hurst exponent (H) performed via the Rescaled adjusted range Statistics plot (R/S). Studies conducted on cardiac signals show that they have a fractal structure characterized by self-similarity. The degree of self-similarity can be determined by the Hurst exponent—at 0.5 < H < 1, the studied process is fractal. It was found that there is a difference in the values of this index in healthy and sick individuals. At values of H close to 1, chronic and pathological diseases are observed.
- Detrended Fluctuation Analysis (DFA). With this method, three parameters (alpha, alpha 1, and alpha 2) are determined using information obtained on the fractal correlations in the studied time series. If there is no correlation in the time series, then an alpha of less than 0.5 is obtained. At alpha > 0.5, there is a correlation dependence in the studied data. Several authors [64,65,66] have declared a difference in the values of alpha parameters in healthy and unhealthy people.
2.3.6. Protection of Research Data
2.4. Statistical Analysis
3. Results
3.1. Time Domain Methods
3.2. Frequency Domain Methods
3.3. Surface Method
3.4. Nonlinear Methods
3.5. Examination of PPG, ECG, and Holter Signals for Health Assessment
4. Discussion
4.1. Limitations
4.2. Future Directions
5. Conclusions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Evans, S.; Seidman, L.C.; Tsao, J.C.; Lung, K.C.; Zeltzer, L.K.; Nalibo, B.D. Heart rate variability as a biomarker for autonomic nervous system response differences between children with chronic pain and healthy control children. J. Pain Res. 2013, 6, 449–457. [Google Scholar] [PubMed]
- Moorman, J.R.; Carlo, W.A.; Kattwinkel, J.; Schelonka, R.L.; Porcelli, P.J.; Navarrete, C.T.; Bancalari, E.; Aschner, J.L.; Walker, M.W.; Perez, J.A.; et al. Mortality reduction by heart rate characteristic monitoring in very low birth weight neonates: A randomized trial. J. Pediatr. 2011, 159, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Draghici, A.E.; Taylor, J.A. The physiological basis and measurement of heart rate variability in humans. J. Physiol Anthr. 2016, 35, 22. [Google Scholar] [CrossRef] [PubMed]
- Ernst, G. Hidden signals—The history and methods of heart rate variability. Front. Public Health 2017, 5, 265. [Google Scholar] [CrossRef]
- Lee, S.; Hwang, H.B.; Park, S.; Kim, S.; Ha, J.H.; Jang, Y.; Hwang, S.; Park, H.-K.; Lee, J.; Kim, I.Y. Mental Stress Assessment Using Ultra Short Term HRV Analysis Based on Non-Linear Method. Biosensors 2022, 12, 465. [Google Scholar] [CrossRef]
- Shen, W.; Gill, R.M.; Zhang, J.P.; Jones, B.D.; Corbly, A.K.; Steinberg, M.I. Sodium channel enhancer restores baroreflex sensitivity in conscious dogs with heart failure. Am. J. Physiol. Heart Circ. Physiol. 2005, 288, H1508–H1514. [Google Scholar] [CrossRef]
- Berger, R.D.; Akselrod, S.; Gordon, D.; Cohen, R.J. An efficient algorithm for spectral analysis of heart rate variability. IEEE Trans. Biomed. Eng. 1986, 33, 900–904. [Google Scholar] [CrossRef]
- Malik, M. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Heart rate variability: Standards of measurement, physiological interpretation, and clinical use. Circulation 1996, 93, 1043–1065. [Google Scholar] [CrossRef]
- Becerra-Luna, B.; Martínez-Memije, R.; Cartas-Rosado, R.; Infante-Vázquez, O.; Sánchez-García, J.C.; Sánchez-Pérez, G. Optimal Window for the Estimation of Very Low Frequency Content in Heart Rate Variability Analysis. In IFMBE Proceedings, Proceedings of the World Congress on Medical Physics and Biomedical Engineering 2018, Prague, the Czech Republic, 3–8 June 2018; Lhotska, L., Sukupova, L., Lacković, I., Ibbott, G., Eds.; Springer: Singapore, 2019; Volume 68, pp. 682331–682334. [Google Scholar] [CrossRef]
- Dong, S.Y.; Lee, M.; Park, H.; Youn, I. Stress resilience measurement with heart-rate variability during mental and physical stress. In Proceedings of the 2018 40th Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Honolulu, HI, USA, 18–21 July 2018; pp. 5290–5293. [Google Scholar]
- Tarvainen, M.; Niskanen, J.; Lipponen, J.; Rantaaho, P.; Karjalainen, P. Kubios HRV-heart rate variability analysis software. Comput. Methods Programs Biomed. 2014, 113, 210–220. [Google Scholar] [CrossRef]
- Mourot, L. CODESNA_HRV, a new tool to assess the activity of the autonomic nervous system from heart rate variability. Phys. Med. Rehabil. Res. 2018, 3, 2–6. [Google Scholar] [CrossRef]
- Lee, R.-G.; Chou, I.-C.; Lai, C.-C.; Liu, M.-H.; Chiu, M.-J. A Novel QRS detection algorithm to the analysis for heart rate variability of patients with sleep apnea. Biomed. Eng. Appl. Basis Comm. 2005, 17, 258–262. [Google Scholar] [CrossRef]
- Billman, G.E.; Huikuri, H.V.; Sacha, J.; Trimmel, K. An introduction to heart rate variability: Methodological considerations and clinical applications. Front. Physiol. 2015, 6, 55. [Google Scholar] [CrossRef] [PubMed]
- Burger, A.J.; Charlamb, M.; Sherman, H.B. Circadian patterns of heart rate variability in normals, chronic stable angina and diabetes mellitus. Int. J. Cardiol. 1999, 71, 41–48. [Google Scholar] [CrossRef] [PubMed]
- Malia, B.; Zuljb, S.; Magjarevic, R.; Miklavcic, D.; Jarm, T. Matlab-based tool for ECG and HRV analysis. Biomed. Signal Process. Control. 2014, 10, 108–116. [Google Scholar] [CrossRef]
- Selvaraj, J.; Murugappan, M.; Wan, K.; Yaacob, S. Classification of emotional states from electrocardiogram signals: A non-linear approach based on Hurst. BioMedical Eng. OnLine 2013, 12, 12–44. [Google Scholar] [CrossRef]
- Park, Y.H.; Koo, C.-H.; Kim, J.-T.; Kim, H.-S.; Byon, H.-J. Differences of Heart Rate Variability during Sevoflurane Anesthesia in Children by Age. Open J. Anesthesiol. 2012, 2, 74–78. [Google Scholar] [CrossRef]
- Reed, M.J.; Robertson, C.E.; Addison, P.S. Heart rate variability measurements and the prediction of ventricular arrhythmias. QJM Int. J. Med. 2005, 98, 87–95. [Google Scholar] [CrossRef]
- Wolf, M.M.; Varigos, G.A.; Hunt, D.; Sloman, J.G. Sinus arrhythmia in acute myocardial infarction. Med. J. Aust. 1978, 2, 52–53. [Google Scholar] [CrossRef]
- Kleiger, R.; Miller, J.; Bigger, J.; Moss, A.J.; The Multicenter Post-Infarction Research Group. Decreased Heart Rate Variability and Its Association with Increased Mortality After Acute Myocardial Infarction. Am. J. Cardiol. 1987, 59, 256–262. [Google Scholar] [CrossRef]
- Naar, J.; Mlcek, M.; Kruger, A.; Vondrakova, D.; Janotka, M.; Popkova, M.; Kittnar, O.; Neuzil, P.; Ostadal, P. Acute Severe Heart Failure Reduces Heart Rate Variability: An Experimental Study in a Porcine Model. Int. J. Mol. Sci. 2023, 24, 493. [Google Scholar] [CrossRef]
- Neshitov, A.; Tyapochkin, K.; Smorodnikova, E.; Pravdin, P. Wavelet Analysis and Self-Similarity of Photoplethysmography Signals for HRV Estimation and Quality Assessment. Sensors 2021, 21, 6798. [Google Scholar] [CrossRef] [PubMed]
- Paniccia, M.; Paniccia, D.; Thomas, S.; Taha, T.; Reed, N. Clinical and non-clinical depression and anxiety in young people: A scoping review on heart rate variability. Auton. Neurosci. 2017, 208, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Conci, F.; Rienzo, D.; Castiglioni, P. Blood pressure and heart rate variability and baroreflex sensitivity before and after brain death. J. Neurol. Neurosurg. Psychiatry 2001, 71, 621–631. [Google Scholar] [CrossRef] [PubMed]
- Vaisakhi, V.S.; Kumar, K.B.; Sivakami, K. Statistical Analysis of HRV Signals. Int. J. Sci. Eng. Res. 2017, 8, 169–177. [Google Scholar]
- Rich, M.W.; Saini, J.S.; Kleiger, R.E.; Carney, R.M.; teVelde, A.; Freedland, K.E. Correlation of heart rate variability with clinical and angiographic variables and late mortality after coronary angiography. Am. J. Cardiol. 1988, 62 Pt 1, 714–717. [Google Scholar] [CrossRef] [PubMed]
- Alqaraawi, A.; Alwosheel, A.; Alasaade, A. Heart rate variability estimation in photoplethysmography signals using Bayesian learning approach. Healthc. Tech. Lett. 2016, 3, 136–142. [Google Scholar] [CrossRef]
- Buccelletti, F.; Gilardi, E.; Scaini, E.; Galiuto, L.; Persiani, R.; Biondi, A.; Basile, F.; Silveri, N. Heart rate variability and myocardial infarction: Systematic literature review and metanalysis. Eur. Rev. Med. Pharmacol. Sci. 2009, 13, 299–307. [Google Scholar]
- Elghozi, J.-L.; Girard, A.; Laude, D. Effects of drugs on the autonomic control of short-term heart rate variability. Auton. Neurosci. Basic E-Clin. 2001, 90, 116–121. [Google Scholar] [CrossRef]
- Cygankiewicz, I.; Zareba, W. Heart rate variability. Handb. Clin. Neurol. 2013, 117, 379–393. [Google Scholar]
- Abboud, S.; Barnea, O. Errors Due to Sampling Frequency of Electrocardiogram in Spectral Analysis of HR Signals with Low Variability. Comp. Cardiol. 1995, 22, 461–463. [Google Scholar]
- Lombardi, F.; Stein, P.K. Origin of heart rate variability and turbulence: An appraisal of autonomic modulation of cardiovascular function. Front. Physiol. 2011, 2, 95. [Google Scholar] [CrossRef]
- Botman, S.; Borchevkin, D.; Petrov, V.; Bogdanov, E.; Patrushev, M.; Shusharina, N. Photoplethysmography-Based Device Designing for Cardiovascular System Diagnostics. Int. J. Biomed. Biol. Eng. 2015, 9, 689–693. [Google Scholar]
- Aimie-Salleh, N.; Ghani, N.A.A.; Hasanudin, N.; Shafie, S.N.S. Heart Rate Variability Recording System Using Photoplethysmography Sensor. In Autonomic Nervous System Monitoring; IntechOpen: London, UK, 2019. [Google Scholar] [CrossRef]
- Zhang, S.P.; Wu, W. Chapter 12: HRV-based biometric privacy-preserving and security mechanism for wireless body sensor networks. In Wearable Sensors. Applications, Design, and Implementation; IOP Publishing: Bristol, UK, 2017; pp. 12–27. [Google Scholar] [CrossRef]
- Islam, S.M.R.; Kwak, D.; Kabir, M.H.; Hossain, M.; Kwak, K.S. The Internet of Things for Health Care: A Comprehensive Survey. IEEE Access 2015, 3, 678–708. [Google Scholar] [CrossRef]
- Ullah, F.; Islam, I.; Abdullah, H.; Khan, A. Future of Big Data and Deep Learning for Wireless Body Area Networks; Springer: Singapore, 2019; pp. 53–77. [Google Scholar]
- Hassan, M.M.; Huda, M.S.; Uddin, M.Z.; Almogren, A.; AlRubaian, M.A. Human Activity Recognition from Body Sensor Data using Deep Learning. J. Med. Syst. 2018, 42, 99. [Google Scholar] [CrossRef] [PubMed]
- Di, M.; Joo, E.M. A survey of machine learning in Wireless Sensor networks From networking and application perspectives. In Proceedings of the 2007 6th International Conference on Information, Communications Signal Processing, Singapore, 10–13 December 2007; pp. 1–5. [Google Scholar]
- Bilal, M.; Shaikh, F.K.; Arif, M.; Wyne, M.F. A revised framework of machine learning application for optimal activity recognition. Clust. Comput. 2017, 22, 7257–7273. [Google Scholar] [CrossRef]
- Pace, P.; Fortino, G.; Zhang, Y.; Liotta, A. Intelligence at the Edge of Complex Networks: The Case of Cognitive Transmission Power Control. IEEE Wirel. Commun. 2019, 26, 97–103. [Google Scholar] [CrossRef]
- Fortino, G.; Giannantonio, R.; Gravina, R.; Kuryloski, P.; Jafari, R. Enabling Effective Programming and Flexible Management of Efficient Body Sensor Network Applications. IEEE Trans. Hum.-Mach. Syst. 2013, 43, 115–133. [Google Scholar] [CrossRef]
- Gravina, R.; Alinia, P.; Ghasemzadeh, H.; Fortino, G. Multi-sensor fusion in body sensor networks: State-of-the-art and research challenges. Inf. Fusion 2017, 35, 68–80. [Google Scholar] [CrossRef]
- Fortino, G.; Galzarano, S.; Gravina, R.; Li, W. A framework for collaborative computing and multi-sensor data fusion in body sensor networks. Inf. Fusion 2015, 22, 50–70. [Google Scholar] [CrossRef]
- Pandian, P.; Srinivasa, M. A ZigBee-wireless wearable remote physiological monitoring system. In Proceedings of the 2016 International Conference on Signal Processing and Communication (ICSC), Noida, India, 26–28 December 2016; pp. 407–412. [Google Scholar]
- Liu, M.; Jiang, F.; Jiang, H.; Ye, S.; Chen, H. Low-power, noninvasive measurement system for wearable ballistocardiography in sitting and standing positions. Comput. Ind. 2017, 91, 24–32. [Google Scholar] [CrossRef]
- Fortino, G.; Parisi, D.; Pirrone, V.; Di Fatta, G. BodyCloud: A SaaS approach for community body sensor networks. Future Gener. Comput. Syst. 2014, 35, 62–79. [Google Scholar] [CrossRef]
- Hussein, A.F.; Kumar, A.; Burbano-Fernandez, M.; Ramirez-Gonzalez, G.; Abdulhay, E.; de Albuquerque, V.H.C. An Automated Remote Cloud-Based Heart Rate Variability Monitoring System. IEEE Access 2018, 6, 77055–77064. [Google Scholar] [CrossRef]
- Batumalay, M.; Ming, H.S.; Arrova Dewi, D. Cloud based Heartbeat Rate Monitoring System with Location Tracking. INTI J. 2020, 2020. Available online: http://eprints.intimal.edu.my/1415/1/vol.2020_008.pdf (accessed on 5 January 2023).
- Scientific Project “Research on the Application of New Mathematical Methods for the Analysis of Cardiac Data” Financed by the Bulgarian National Science Fund. Available online: https://www.cardiomath.org/ (accessed on 5 January 2023).
- Available online: http://hrvdata.vtlab.eu/ (accessed on 5 January 2023).
- Georgieva-Tsaneva, G. Wavelet Based Interval Varying Algorithm for Optimal Non-Stationary Signal Denoising. In Proceedings of the 20th International Conference on Computer Systems and Technologies, Ruse, Bulgaria, 21–22 June 2019; pp. 200–206. [Google Scholar] [CrossRef]
- Georgieva-Tsaneva, G. QRS detection algorithm for long-term Holter records. In Proceedings of the 14th International Conference on Computer Systems and Technologies, Ruse, Bulgaria, 28–29 June 2013; pp. 112–119. [Google Scholar] [CrossRef]
- Nellcor™ Reusable SpO2 Sensors with OxiMax™ Technology. Available online: http://www.medtronic.com/covidien/en-us/products/pulseoximetry/nellcor-reusable-spo2-sensors.html (accessed on 5 January 2023).
- Gospodinov, M.; Cheshmedziev, K. Three-Sensor Portable Information System for Physiological Data Registration. In Proceedings of the 20th International Conference on Computer Systems and Technologies, Ruse, Bulgaria, 21–22 June 2019; pp. 36–41. [Google Scholar] [CrossRef]
- Cheshmedzhiev, K.; Gospodinova, E.; Gospodinov, M.; Lebamovski, P. Electronic sensor system for registering ECG and PPG signals. In Proceedings of the 22nd International Conference on Computer Systems and Technologies, Ruse, Bulgaria, 18–19 June 2021; pp. 141–144. [Google Scholar] [CrossRef]
- AFE4490 Integrated Analog Front End (AFE) for Pulse Oximeters. Available online: http://www.ti.com/product/AFE4490 (accessed on 5 January 2023).
- MAX30102 High-Sensitivity Pulse Oximeter and Heart-Rate Sensor for Wearable Health. Available online: https://www.maximintegrated.com/en/products/interface/sensor-interface/MAX30102.html (accessed on 5 January 2023).
- Georgieva-Tsaneva, G.; Gospodinova, E.; Gospodinov, M.; Cheshmedzhiev, K. Portable Sensor System for Registration, Processing and Mathematical Analysis of PPG Signals. Appl. Sci. 2020, 10, 1051. [Google Scholar] [CrossRef]
- Akar, S.A.; Kara, S.; Latifoglu, F.; Biggic, V. Spectral analysis of photoplethysmographic signals: The importance of preprocessing. Biomed. Signal Process. Control. 2013, 8, 16–22. [Google Scholar] [CrossRef]
- Boardman, A.; Schlindwein, F.S.; Rocha, A.P.; Leite, A. A study on the optimum order of autoregressive models for heart rate variability. Physiol. Meas. 2002, 23, 325–336. [Google Scholar] [CrossRef]
- Gospodinova, E.; Gospodinov, M.; Dey, N.; Domuschiev, I.; Ashou, A.S.; Balas, S.V.; Olariu, T. Specialized Software System for Heart Rate Variability Analysis: An Implementation of Nonlinear Graphical Methods. In Soft Computing Applications, Proceedings of the SOFA 2016 Advances in Intelligent Systems and Computing, Arad, Romania, 24–26 August 2016; Balas, V., Jain, L., Balas, M., Eds.; Springer: Cham, Switzerland, 2016; Volume 633, p. 633. [Google Scholar] [CrossRef]
- Acharya, U.R.; Min, L.C.; Joseph, P. HRV analysis using correlation dimension and DFA. Innov. Tech. Biol. Med. 2002, 23, 333–339. [Google Scholar]
- Barquero-Perez, O.; Marques de Sa, J.; Rojo-Alvarez, J.L.; Goya-Esteban, R. Changes in detrended fluctuation indices with aging in healthy and congestive heart failure subjects. Comput. Cardiol. 2008, 35, 45–48. [Google Scholar]
- Ernst, G. Heart Rate Variability; Springer: London, UK, 2014; p. 336. [Google Scholar]
- Georgieva-Tsaneva, G.; Bogdanova, G.; Gospodinova, E. Mathematically Based Assessment of the Accuracy of Protection of Cardiac Data Realized with the Help of Cryptography and Steganography. Mathematics 2022, 10, 390. [Google Scholar] [CrossRef]
- Lebamovski, P. Analysis of 3D technologies for stereo visualization. In Proceedings of the International Conference Automatics and Informatics (ICAI), Varna, Bulgaria, 30 September–2 October 2021; pp. 206–209. [Google Scholar] [CrossRef]






| Parameters | Healthy (Mean ± SD) |
|---|---|
| Statistical parameters | |
| HRmin {bpm} | >50 |
| HRmax {bpm} | <120 |
| MeanHR {bpm} | >50, <120 |
| MeanRR {ms} | - |
| SDNN {ms} | 141 ± 39 (102–180) |
| SDANN {ms} | 127 ± 35 (92–162) |
| RMSSD {ms} | 27 ± 12 (15–39) |
| NN50 | - |
| pNN50 {%} | - |
| SDindex {ms} | - |
| Geometrical parameters | |
| HRVti {numb} | 37 ± 15 (22–52) |
| TINN {ms} | - |
| Parameters | Frequency Range {Hz} | Healthy (Mean ± SD) |
|---|---|---|
| TP {ms2} | ≤0.4 | 3466 ± 1018 |
| VLF {ms2} | ≤0.04 | - |
| LF {ms2} | 0.04–0.15 | 1170 ± 416 |
| HF {ms2} | 0.15–0.4 | 975 ± 203 |
| LFnu {n.e.} | - | 54 ± 4 |
| HFnu {n.e.} | - | 29 ± 3 |
| LF/HF {-} | - | 1.5–2.0 |
| Parameter | Heart Failure n = 14 | Tachycardia n = 12 | Healthy n = 12 | p-Value |
|---|---|---|---|---|
| Men {%} | 57.14 | 58.33 | 42.67 | NS |
| Age {years ± sd} | 62.43 ± 23.08 | 52.28 ± 13.26 | 51.62 ± 20.36 | NS |
| Parameters | Heart Failure (Mean ± SD) | Tachycardia (Mean ± SD) | Healthy (Mean ± SD) | p Value (Mean ± SD) |
|---|---|---|---|---|
| Statistical parameters | ||||
| HRmin {bpm} | 51 ± 13 | 61 ± 29 | 56 ± 14 | NS |
| Hrmax {bpm} | 112 ± 27 | 140 ± 38 | 103 ± 16 | <0.05 |
| MeanHR {bpm} | 94.79 ± 22 | 103 ± 26 | 72 ± 26 | NS |
| MeanRR {ms} | 633.64 ± 123.86 | 580.56 ± 231.95 | 849.35 ± 321.32 | NS |
| SDNN {ms} | 82.44 ± 19.04 | 101.34 ± 23.63 | 141.82 ± 22.08 | <0.001 |
| SDANN {ms} | 61.73 ± 12.92 | 91 ± 13.43 | 130.64 ± 1.5 | <0.001 |
| RMSSD {ms} | 18.52 ± 2.86 | 8.37 ± 4.03 | 26.85 ± 2.3 | <0.0001 |
| NN50 | 640.3 ± 20.41 | 862.11 ± 6.06 | 1347.04 ± 87.36 | <0.001 |
| pNN50 {%} | 14.21 ± 2.65 | 23.43 ± 8.15 | 34.92 ± 46.1 | <0.001 |
| Sdindex {ms} | 56.42 ± 16.32 | 52.32 ± 12.03 | 63.04 ± 23.06 | NS |
| Geometrical parameters | ||||
| HRVti {numb} | 11.53 ± 4.02 | 28.43 ± 7.32 | 42.61 ± 14.2 | <0.001 |
| TINN {ms} | 481.62 ± 61.73 | 420.42 ± 21.31 | 498.22 ± 48.09 | NS |
| Parameters | Heart Failure (Mean ± SD) | Tachycardia (Mean ± SD) | Healthy (Mean ± SD) | p-Value (Mean ± SD) |
|---|---|---|---|---|
| Statistical Parameters | ||||
| Total Power } | 12,803.92 ± 969.65 | 11,870.26 ± 863.14 | 13,921.02 ± 691.08 | NS |
| Power VLF } | 11,939.57 ± 489.73 | 10,453.88 ± 23.75 | 11,620.22 ± 348.41 | NS |
| Power LF } | 482.53 ± 113.06 | 693.71 ± 103.82 | 1428.31 ± 241.84 | <0.05 |
| Power HF } | 381.65 ± 98.55 | 724.85 ± 111.62 | 873.02 ± 183.32 | <0.05 |
| Power LF {nu} | 55.85 ± 7.98 | 48.92 ± 10.54 | 62.42 ± 6.24 | NS |
| Power HF {nu} | 44.37 ± 8.71 | 51.11 ± 11.43 | 37.53 ± 4.06 | NS |
| LF/HF (ratio) | 1.26 ± 0.27 | 0.96 ± 0.16 | 1.64 ± 0.02 | <0.001 |
| Parameters | Heart Failure (Mean ± SD) | Tachycardia (Mean ± SD) | Healthy (Mean ± SD) | p-Value (Mean ± SD) |
|---|---|---|---|---|
| Statistical Parameters | ||||
| Alpha (DFA) | 0.91 ± 0.36 | 0.83 ± 0.34 | 1.05 ± 0.74 | <0.001 |
| Alpha1 (DFA) | 0.94 ± 0.12 | 0.89 ± 0.72 | 1.21 ± 0.83 | <0.001 |
| Alpha2 (DFA) | 0.82 ± 0.03 | 0.64 ± 0.71 | 0.99 ± 0.31 | <0.001 |
| Hurst (R/S method) | 0.91 ± 0.18 | 0.88 ± 0.11 | 0.76 ± 0.04 | <0.001 |
| Parameters | Group 1 ECG (Mean ± SD) | Group 2 Holter (Mean ± SD) | Group 3 PPG (Mean ± SD) | |
|---|---|---|---|---|
| Time domain | Mean RR (PP) {ms} | 684.22 ± 214.68 | 661.33 ± 189.13 | 692.11 ± 223.83 |
| SDNN {ms} | 84.08 ± 16.88 | 82.77 ± 24.32 | 88.66 ± 32.09 | |
| SDANN {ms} | 72.56 ± 34.21 | 76.01 ± 35.43 | 74.67 ± 31.08 | |
| RMSSD {ms} | 13.18 ± 8.65 | 12.35 ± 14.15 | 11.06 ±18.98 | |
| SDindex {ms} | 61.33 ± 26.11 | 64.07 ± 22.18 | 63.88 ± 26.44 | |
| Frequency domain | Power VLF {ms2} | 3098.51 ± 654.22 | 3127.06 ± 487.34 | 2995.78 ± 586.39 |
| Power LF {ms2} | 688.22 ± 183.06 | 691.89 ± 243.99 | 704.05 ± 433.01 | |
| Power HF {ms2} | 586.23 ± 204.55 | 582.99 ± 244.13 | 602.33 ± 212.03 | |
| Power LF {nu} | 0.54 ± 0.19 | 0.54 ± 0.16 | 0.53 ± 0.87 | |
| Power HF {nu} | 0.46 ± 0.23 | 0.46 ± 0.43 | 0.47 ± 0.68 | |
| LF/HF {-} | 1.17 ± 0.78 | 1.19 ± 0.81 | 1.17 ± 0.93 | |
| Parameters | ||||
|---|---|---|---|---|
| Time domain | MeanRR(PP) {ms} | 1.34 | 3.31 | 0.6 |
| SDNN {ms} | 0.64 | 1.47 | 0.88 | |
| SDANN {ms} | 1.49 | 0.69 | 0.83 | |
| RMSSD {ms} | 2.27 | 3.13 | 5.27 | |
| SDindex {ms} | 4.01 | 3.18 | 3.58 | |
| Frequency domain | Power VLF {ms2} | 2.96 | 4.92 | 5.93 |
| Power LF {ms2} | 3.04 | 4.07 | 1.69 | |
| Power HF {ms2} | 4.33 | 6.71 | 2.78 | |
| Power LF {nu} | 0.04 | 1.65 | 1.97 | |
| Power HF {н.e} | 0.1 | 1.37 | 2.06 | |
| LF/HF {-} | 0.49 | 0.08 | 1.02 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Georgieva-Tsaneva, G. Interactive Cardio System for Healthcare Improvement. Sensors 2023, 23, 1186. https://doi.org/10.3390/s23031186
Georgieva-Tsaneva G. Interactive Cardio System for Healthcare Improvement. Sensors. 2023; 23(3):1186. https://doi.org/10.3390/s23031186
Chicago/Turabian StyleGeorgieva-Tsaneva, Galya. 2023. "Interactive Cardio System for Healthcare Improvement" Sensors 23, no. 3: 1186. https://doi.org/10.3390/s23031186
APA StyleGeorgieva-Tsaneva, G. (2023). Interactive Cardio System for Healthcare Improvement. Sensors, 23(3), 1186. https://doi.org/10.3390/s23031186






