Photoactivated Processes on the Surface of Metal Oxides and Gas Sensitivity to Oxygen
Abstract
1. Introduction
2. Materials and Methods
2.1. Synthesis on Nanocrystalline Oxides
2.2. Characterization
2.3. Gas-Sensing Measurements
2.4. In Situ Ambient Pressure UV-Assisted Mass Spectrometry
3. Results and Discussion
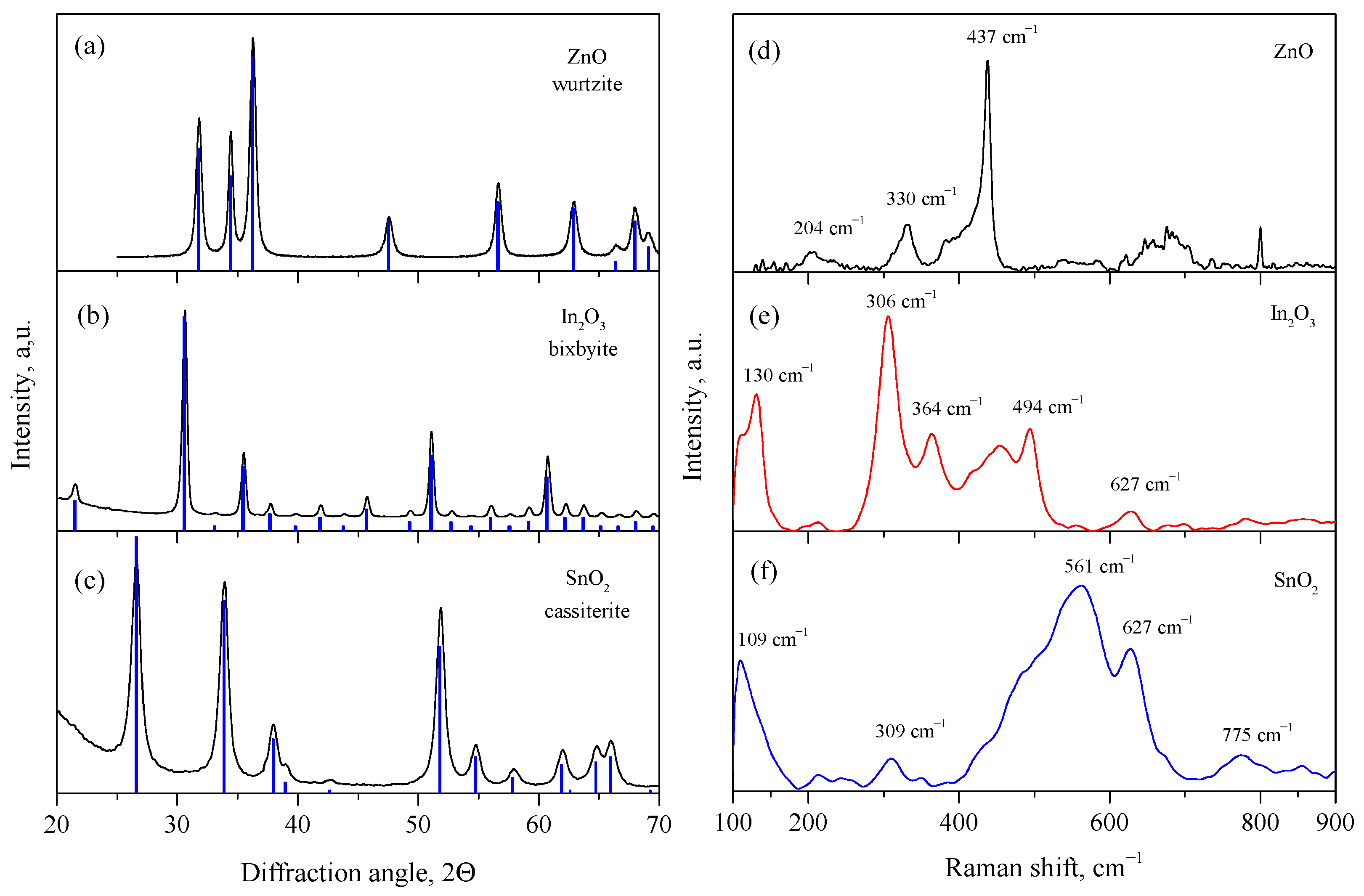
3.1. Structure and Morphology of Synthesized Metal Oxides
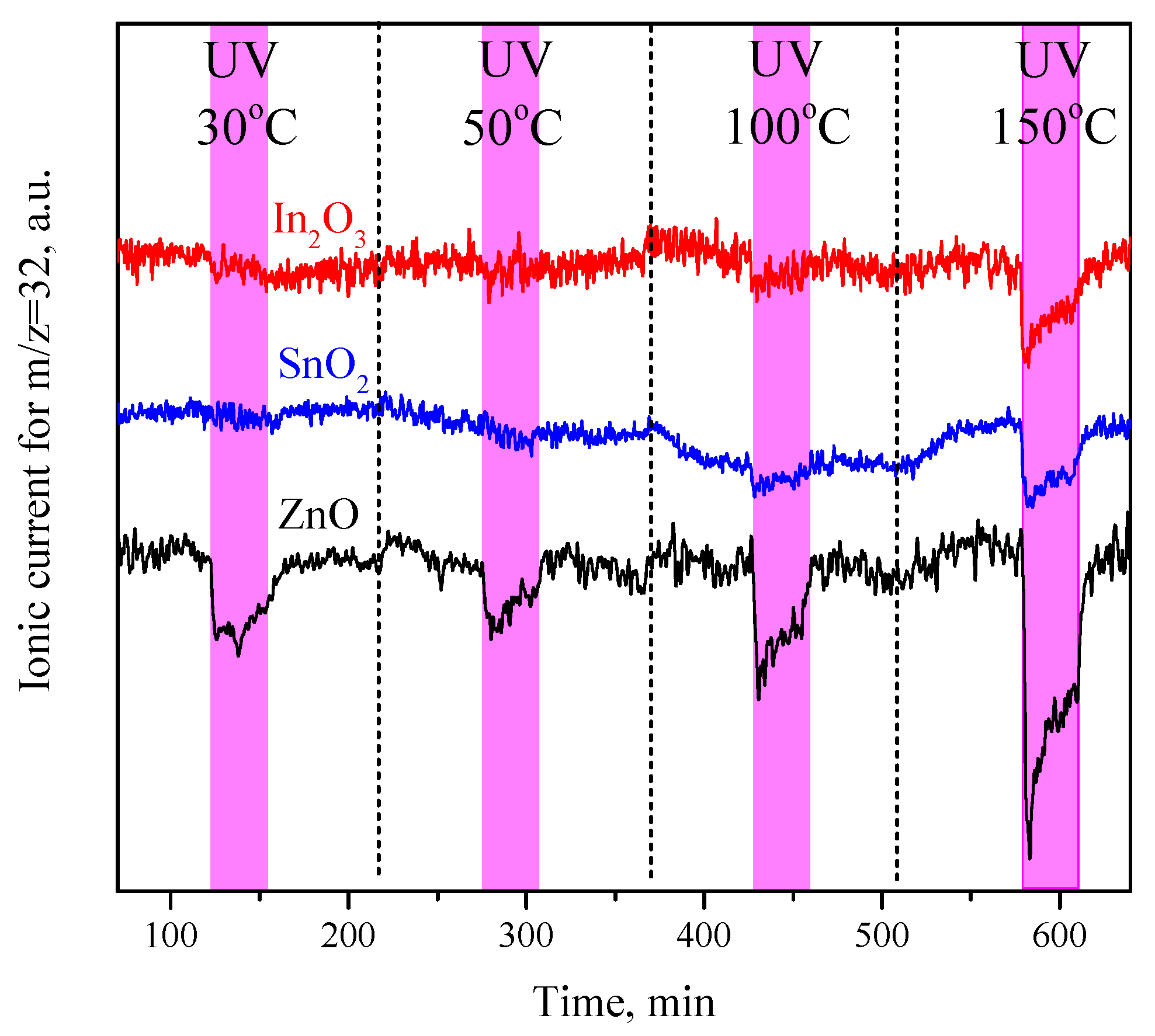
3.2. Oxygen Photoadsorption and UV-Activated Oxygen Isotopic Exchange
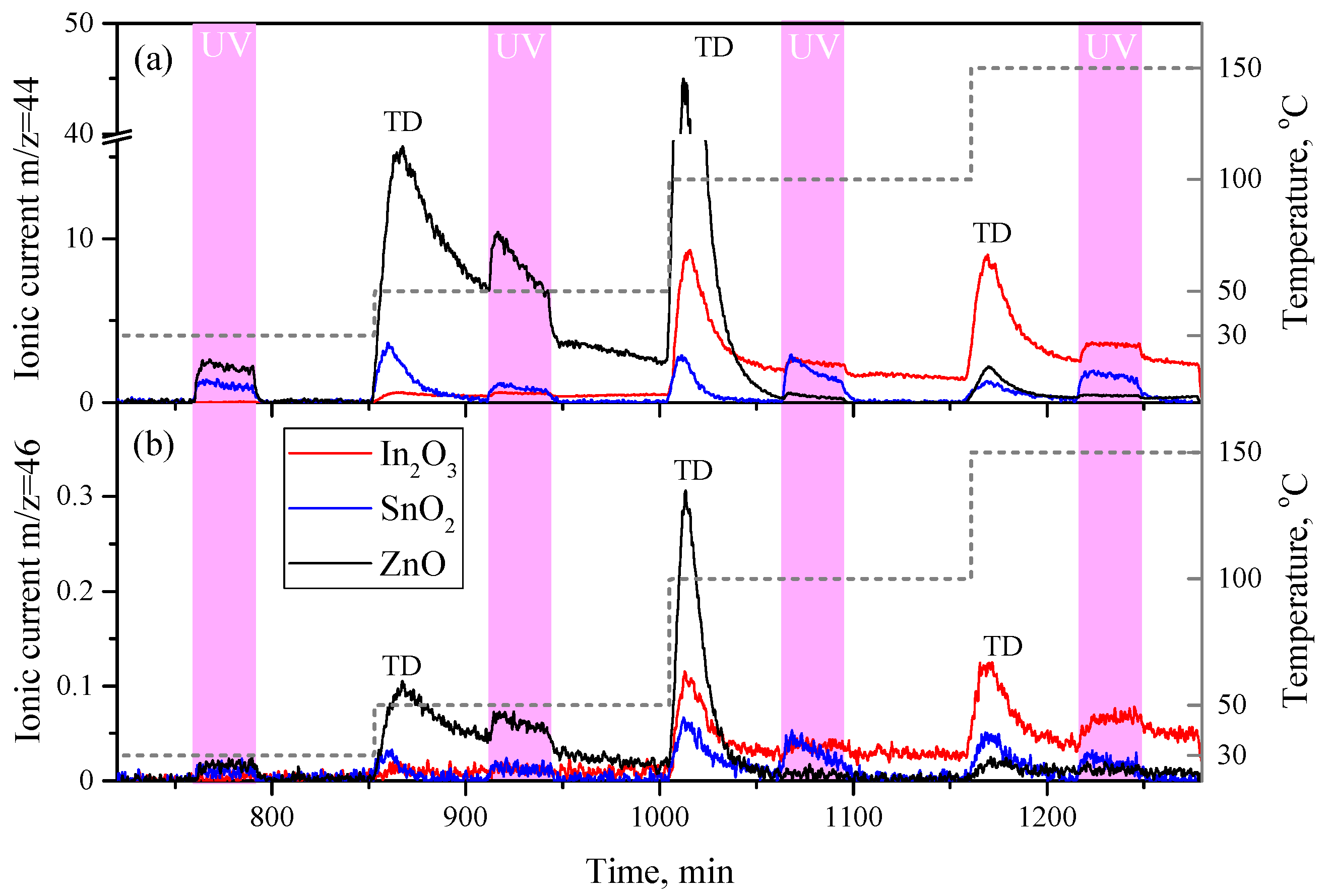
3.3. CO2 Photodesorption
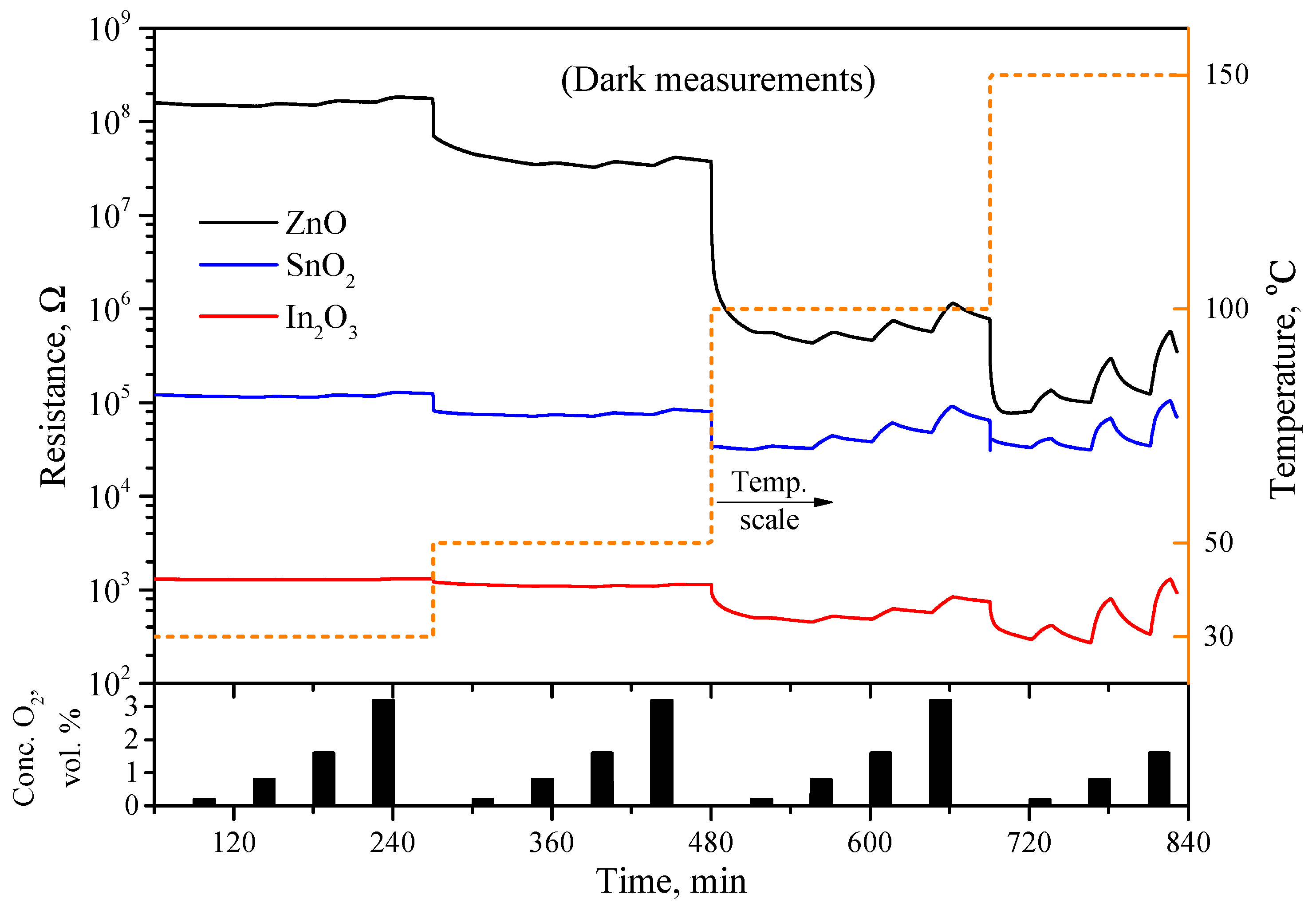
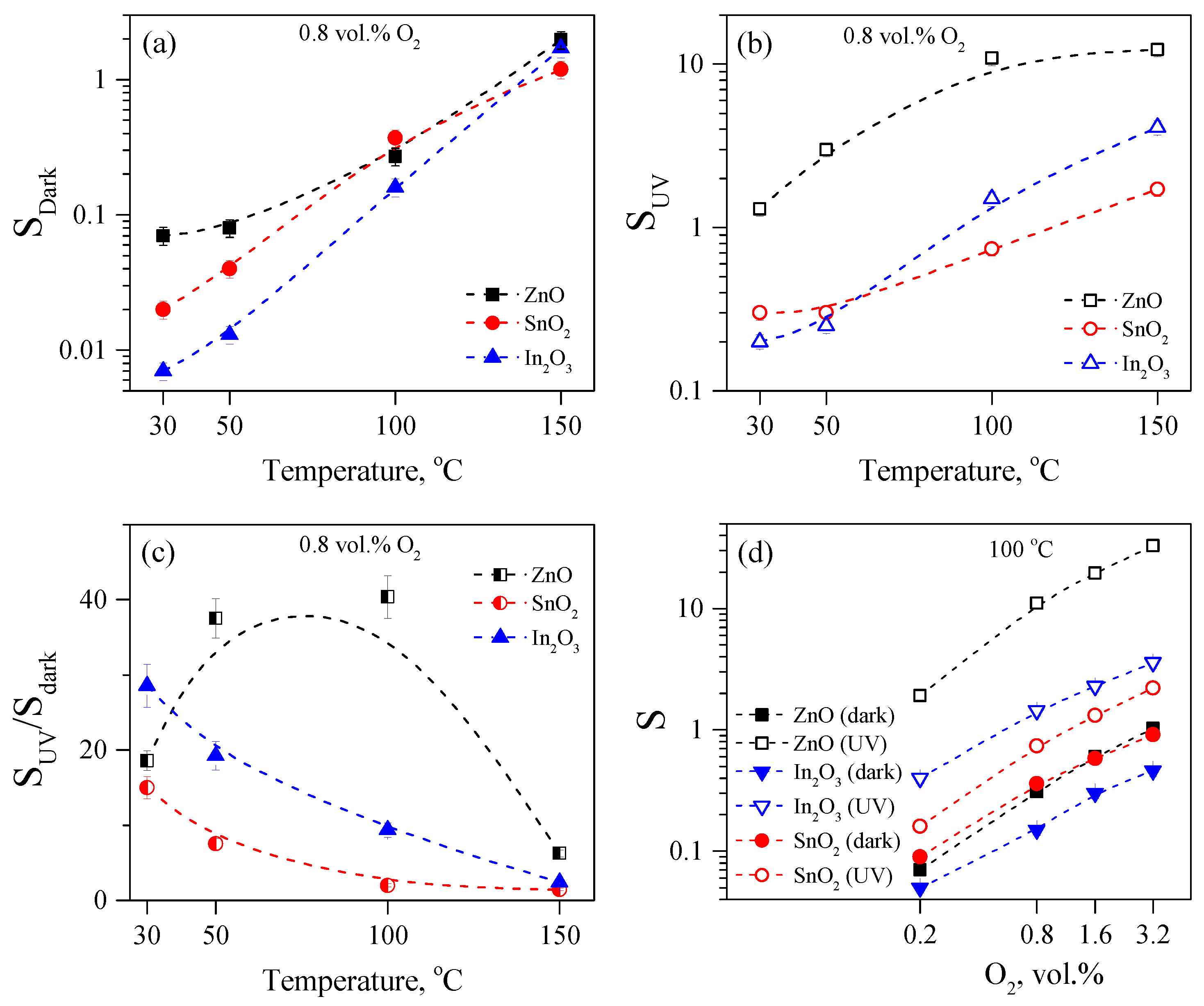
3.4. Gas Sensitivity to Oxygen
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Li, Q.; Zeng, W.; Li, Y. Metal oxide gas sensors for detecting NO2 in industrial exhaust gas: Recent developments. Sens. Actuators B Chem. 2022, 359, 131579. [Google Scholar] [CrossRef]
- Das, S.; Mojumder, S.; Saha, D.; Pal, M. Influence of major parameters on the sensing mechanism of semiconductor metal oxide based chemiresistive gas sensors: A review focused on personalized healthcare. Sens. Actuators B Chem. 2022, 352, 131066. [Google Scholar] [CrossRef]
- Isaac, N.; Pikaar, I.; Biskos, G. Metal oxide semiconducting nanomaterials for air quality gas sensors: Operating principles, performance, and synthesis techniques. Microchim. Acta 2022, 189, 196. [Google Scholar] [CrossRef] [PubMed]
- Landini, N.; Malagù, C.; Guidi, V. Review—Nanostructured chemoresistive sensors as detectors of volatile biomarkers for medical screening purposes of mundane and oncological human pathologies. Sen. Actuators B Chem. 2022, 371, 132493. [Google Scholar] [CrossRef]
- Yun, J.; Cho, M.; Lee, K.; Kang, M.; Park, I. A review of nanostructure-based gas sensors in a power consumption perspective. Sen. Actuators B Chem. 2022, 372, 132612. [Google Scholar] [CrossRef]
- Chai, H.; Zheng, Z.; Liu, K.; Xu, J.; Wu, K.; Luo, Y.; Liao, H.; Debliquy, M.; Zhang, C. Stability of Metal Oxide Semiconductor Gas Sensors: A Review. IEEE Sens. J. 2022, 22, 5470–5481. [Google Scholar] [CrossRef]
- Xu, F.; HO, H.P. Light-Activated Metal Oxide Gas Sensors: A Review. Micromachines 2017, 8, 333. [Google Scholar] [CrossRef]
- Wang, J.; Shen, H.; Xia, Y.; Komarneni, S. Light-activated room-temperature gas sensors based on metal oxide nanostructures: A review on recent advances. Ceram. Int. 2021, 47, 7353–7368. [Google Scholar] [CrossRef]
- Šetka, M.; Claros, M.; Chmela, O.; Vallejos, S. Photoactivated materials and sensors for NO2 monitoring. J. Mater. Chem. C 2021, 9, 16804–16827. [Google Scholar] [CrossRef]
- Staerz, A.; Weimar, U.; Barsan, N. Current state of knowledge on the metal oxide based gas sensing mechanism. Sen. Actuators B Chem. 2022, 358, 131531. [Google Scholar] [CrossRef]
- Barsan, N.; Weimar, U. Conduction model of metal oxide gas sensors. J. Electroceramics 2001, 7, 143–167. [Google Scholar] [CrossRef]
- Gurlo, A. Interplay between O2 and SnO2: Oxygen Ionosorption and Spectroscopic Evidence for Adsorbed Oxygen. ChemPhysChem 2006, 7, 2041–2052. [Google Scholar] [CrossRef] [PubMed]
- Blackman, C. Do We Need “Ionosorbed” Oxygen Species? (Or, “A Surface Conductivity Model of Gas Sensitivity in Metal Oxides Based on Variable Surface Oxygen Vacancy Concentration”). ACS Sens. 2021, 6, 3509–3516. [Google Scholar] [CrossRef]
- Carpenter, M.A.; Mathur, S.; Kolmakov, A. Metal Oxide Nanomaterials for Chemical Sensors; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2012. [Google Scholar]
- Chizhov, A.; Rumyantseva, M.; Gaskov, A. Light Activation of Nanocrystalline Metal Oxides for Gas Sensing: Principles, Achievements, Challenges. Nanomaterials 2021, 11, 892. [Google Scholar] [CrossRef]
- Tiburcio-Silver, A.; Sánchez-Juárez, A. SnO2:Ga thin films as oxygen gas sensor. Mater. Sci. Eng. B 2004, 110, 268–271. [Google Scholar] [CrossRef]
- Chaabouni, F.; Abaab, M.; Rezig, B. Metrological characteristics of ZnO oxygen sensor at room temperature. Sen. Actuators B Chem. 2004, 100, 200–204. [Google Scholar] [CrossRef]
- Neri, G.; Bonavita, A.; Micali, G.; Rizzo, G.; Pinna, N.; Niederberger, M. In2O3 and Pt-In2O3 nanopowders for low temperature oxygen sensors. Sens. Actuators B Chem. 2007, 127, 455–462. [Google Scholar] [CrossRef]
- Lu, C.Y.; Chang, S.P.; Chang, S.J.; Hsueh, T.J.; Hsu, C.L.; Chiou, Y.Z.; Chen, I.C. ZnO Nanowire-Based Oxygen Gas Sensor. IEEE Sens. J. 2009, 9, 485–489. [Google Scholar] [CrossRef]
- Sharma, A.P.; Dhakal, P.; Pradhan, D.K.; Behera, M.K.; Xiao, B.; Bahoura, M. Fabrication and characterization of SnO2 nanorods for room temperature gas sensors. AIP Adv. 2018, 8, 095219. [Google Scholar] [CrossRef]
- Xiong, Y.; Li, H.; Guo, T.; Xue, Q. A strategy to improve O2 gas response of La-SnO2 nanofibers based sensor through temperature modulation. Mater. Sci. Forum 2018, 944, 657–665. [Google Scholar] [CrossRef]
- Chang, X.; Li, S.; Chu, D. Sensing of oxygen partial pressure in air with ZnO nanoparticles. Sensors 2020, 20, 562. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Lu, W.; Ding, D.; Zhu, L.; Li, X.; Ling, C.; Xue, Q. Enhanced Room Temperature Oxygen Sensing Properties of LaOCl-SnO2 Hollow Spheres by UV Light Illumination. ACS Sens. 2017, 2, 679–686. [Google Scholar] [CrossRef] [PubMed]
- Lavanya, N.; Sekar, C.; Donato, N.; Leonardi, S.G.; Neri, G. Development of a high performance oxygen sensor operating at room temperature. In Proceedings of the 2018 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Rome, Italy, 11–13 June 2018; IEEE: Piscataway, NJ, USA, 2018; pp. 1–6. [Google Scholar] [CrossRef]
- Jyothilal, H.; Shukla, G.; Walia, S.; Bharath, S.P.; Angappane, S. UV assisted room temperature oxygen sensors using titanium dioxide nanostructures. Mater. Res. Bull. 2021, 140, 111324. [Google Scholar] [CrossRef]
- Chhetri, T.P.; Kerr, L.; Masmali, N.; Jaeger, H.; Eid, K.F. Oxygen sensing with individual ZnO: Sb micro-wires: Effects of temperature and light exposure on the sensitivity and stability. R. Soc. Open Sci. 2022, 9, 211243. [Google Scholar] [CrossRef]
- Naliwajko, P.; Peppel, T.; Strunk, J. Thermal and light induced infrared blackening of ZnO revisited: Rediscovery of fundamental scientific knowledge. React. Kinet. Mech. Catal. 2022, 135, 2291–2305. [Google Scholar] [CrossRef]
- Barry, T.; Stone, F. The reactions of oxygen at dark and irradiated zinc oxide surfaces. Proc. R. Soc. London. Ser. A Math. Phys. Sci. 1960, 255, 124–144. [Google Scholar] [CrossRef]
- Titov, V.V.; Lisachenko, A.A.; Labzovskaya, M.E.; Akopyan, I.K.; Novikov, B.V. Exciton Channel of Photoactivation for Redox Reactions on the Surface of 2D ZnO Nanostructures. J. Phys. Chem. C 2019, 123, 27399–27405. [Google Scholar] [CrossRef]
- Cunningham, J.; Finn, E.; Samman, N. Photo-assisted surface reactions studied by dynamic mass spectrometry. Faraday Discuss. Chem. Soc. 1974, 58, 160–174. [Google Scholar] [CrossRef]
- Melnick, D.A. Zinc Oxide Photoconduction, an Oxygen Adsorption Process. J. Chem. Phys. 1957, 26, 1136–1146. [Google Scholar] [CrossRef]
- Li, Q.; Gao, T.; Wang, Y.; Wang, T. Adsorption and desorption of oxygen probed from ZnO nanowire films by photocurrent measurements. Appl. Phys. Lett. 2005, 86, 123117. [Google Scholar] [CrossRef]
- Yin, Z.G.; Zhang, X.W.; Fu, Z.; Yang, X.L.; Wu, J.L.; Wu, G.S.; Gong, L.; Chu, P.K. Persistent photoconductivity in ZnO nanostructures induced by surface oxygen vacancy. Phys. Status Solidi (RRL)–Rapid Res. Lett. 2012, 6, 117–119. [Google Scholar] [CrossRef]
- Lin, Y.; Wang, D.; Zhao, Q.; Li, Z.; Ma, Y.; Yang, M. Influence of adsorbed oxygen on the surface photovoltage and photoluminescence of ZnO nanorods. Nanotechnology 2006, 17, 2110. [Google Scholar] [CrossRef]
- Titov, V.; Lisachenko, A.; Akopyan, I.; Labzowskaya, M.; Novikov, B. On the nature of the effect of adsorbed oxygen on the excitonic photoluminescence of ZnO. J. Lumin. 2018, 195, 153–158. [Google Scholar] [CrossRef]
- Cerrato, E.; Paganini, M.C.; Giamello, E. Photoactivity under visible light of defective ZnO investigated by EPR spectroscopy and photoluminescence. J. Photochem. Photobiol. A Chem. 2020, 397, 112531. [Google Scholar] [CrossRef]
- Klimovskii, A.; Lisachenko, A. Determination of the kinetic-parameters for the photoadsorption and photodesorption of oxygen on zinc-oxide. Kinet. Catal. 1991, 32, 373–377. [Google Scholar]
- Kuz’min, G.; Purevdorzh, D.; Shenderovich, I. Spectral and temperature dependences of the quantum yield of the photoadsorption of simple gases on dispersed zinc oxide. Kinet. Catal. 1995, 36, 728–732. [Google Scholar]
- Zakharenko, V.; Cherkashin, A. Sensitization of the photoadsorption of O2 and NO on SnO2. React. Kinet. Catal. Lett. 1983, 23, 131–135. [Google Scholar] [CrossRef]
- Volodin, A.; Zakharenko, V.; Cherkashin, A. ESR studies of spectral dependences and kinetics of O2 photoadsorption on SnO2. React. Kinet. Catal. Lett. 1981, 18, 321–324. [Google Scholar] [CrossRef]
- Sato, S. Photocatalytic activities of indium oxide powder prepared from indium hydroxide. J. Photochem. Photobiol. A Chem. 1988, 45, 361–367. [Google Scholar] [CrossRef]
- He, K.; Chen, N.; Wang, C.; Wei, L.; Chen, J. Method for Determining Crystal Grain Size by X-ray Diffraction. Cryst. Res. Technol. 2018, 53, 1700157. [Google Scholar] [CrossRef]
- Chizhov, A.; Kutukov, P.; Gulin, A.; Astafiev, A.; Rumyantseva, M. UV-Activated NO2 Gas Sensing by Nanocrystalline ZnO: Mechanistic Insights from Mass Spectrometry Investigations. Chemosensors 2022, 10, 147. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, J.; Liu, J.; Nasir, M.S.; Zhu, J.; Li, S.; Liang, J.; Yan, W. Smart formaldehyde detection enabled by metal organic framework-derived doped electrospun hollow nanofibers. Sen. Actuators B Chem. 2021, 326, 128819. [Google Scholar] [CrossRef]
- Šćepanović, M.; Grujić-Brojčin, M.; Vojisavljević, K.; Bernik, S.; Srećković, T. Raman study of structural disorder in ZnO nanopowders. J. Raman Spectrosc. 2010, 41, 914–921. [Google Scholar] [CrossRef]
- Gan, J.; Lu, X.; Wu, J.; Xie, S.; Zhai, T.; Yu, M.; Zhang, Z.; Mao, Y.; Wang, S.C.I.; Shen, Y.; et al. Oxygen vacancies promoting photoelectrochemical performance of In2O3 nanocubes. Sci. Rep. 2013, 3, 1021. [Google Scholar] [CrossRef] [PubMed]
- Kranert, C.; Schmidt-Grund, R.; Grundmann, M. Raman active phonon modes of cubic In2O3. Phys. Status Solidi (RRL)–Rapid Res. Lett. 2014, 8, 554–559. [Google Scholar] [CrossRef]
- Rumyantseva, M.N.; Gaskov, A.M.; Rosman, N.; Pagnier, T.; Morante, J.R. Raman Surface Vibration Modes in Nanocrystalline SnO2: Correlation with Gas Sensor Performances. Chem. Mater. 2005, 17, 893–901. [Google Scholar] [CrossRef]
- Liu, L.; Wu, X.; Gao, F.; Shen, J.; Li, T.; Chu, P.K. Determination of surface oxygen vacancy position in SnO2 nanocrystals by Raman spectroscopy. Solid State Commun. 2011, 151, 811–814. [Google Scholar] [CrossRef]
- Mikhaylov, R.V.; Lisachenko, A.A.; Titov, V.V. Investigation of Photostimulated Oxygen Isotope Exchange on TiO2 Degussa P25 Surface upon UV–Vis Irradiation. J. Phys. Chem. C 2012, 116, 23332–23341. [Google Scholar] [CrossRef]
- Gurylev, V.; Perng, T.P. Defect engineering of ZnO: Review on oxygen and zinc vacancies. J. Eur. Ceram. Soc. 2021, 41, 4977–4996. [Google Scholar] [CrossRef]
- Lv, Y.; Pan, C.; Ma, X.; Zong, R.; Bai, X.; Zhu, Y. Production of visible activity and UV performance enhancement of ZnO photocatalyst via vacuum deoxidation. Appl. Catal. B Environ. 2013, 138–139, 26–32. [Google Scholar] [CrossRef]
- Li, G.; Zhang, H.; Meng, L.; Sun, Z.; Chen, Z.; Huang, X.; Qin, Y. Adjustment of oxygen vacancy states in ZnO and its application in ppb-level NO2 gas sensor. Sci. Bull. 2020, 65, 1650–1658. [Google Scholar] [CrossRef]
- Xu, H.; Zhang, R.Q.; Tong, S.Y. Interaction of O2, H2O, N2, and O3 with stoichiometric and reduced ZnO() surface. Phys. Rev. B 2010, 82, 155326. [Google Scholar] [CrossRef]
- Hirschwald, W.H. Zinc oxide: An outstanding example of a binary compound semiconductor. Acc. Chem. Res. 1985, 18, 228–234. [Google Scholar] [CrossRef]
- Agrawal, J.; Dixit, T.; Palani, I.; Singh, V. Systematic investigations on the effect of prolong UV illumination on optoelectronic properties of ZnO honeycomb nanostructures. Scr. Mater. 2019, 163, 1–4. [Google Scholar] [CrossRef]
- Asakuma, N.; Fukui, T.; Toki, M.; Awazu, K.; Imai, H. Photoinduced hydroxylation at ZnO surface. Thin Solid Film. 2003, 445, 284–287. [Google Scholar] [CrossRef]
- Asakuma, N.; Hirashima, H.; Imai, H.; Fukui, T.; Toki, M. Crystallization and reduction of sol-gel-derived zinc oxide films by irradiation with ultraviolet lamp. J. Sol-Gel Sci. Technol. 2003, 26, 181–184. [Google Scholar] [CrossRef]
- Drouilly, C.; Krafft, J.M.; Averseng, F.; Casale, S.; Bazer-Bachi, D.; Chizallet, C.; Lecocq, V.; Vezin, H.; Lauron-Pernot, H.; Costentin, G. ZnO Oxygen Vacancies Formation and Filling Followed by in Situ Photoluminescence and in Situ EPR. J. Phys. Chem. C 2012, 116, 21297–21307. [Google Scholar] [CrossRef]
- Yan, Y.; Al-Jassim, M.M.; Wei, S.H. Oxygen-vacancy mediated adsorption and reactions of molecular oxygen on the ZnO() surface. Phys. Rev. B 2005, 72, 161307. [Google Scholar] [CrossRef]
- Saputro, A.G.; Akbar, F.T.; Setyagar, N.P.P.; Agusta, M.K.; Pramudya, A.D.; Dipojono, H.K. Effect of surface defects on the interaction of the oxygen molecule with the ZnO(100) surface. New J. Chem. 2020, 44, 7376–7385. [Google Scholar] [CrossRef]
- Wang, X.; Lu, B.; Li, L.; Qiu, H. Exploring the Interactions of Oxygen with Defective ZnO. ChemistryOpen 2018, 7, 491–494. [Google Scholar] [CrossRef]
- Imai, H.; Tominaga, A.; Hirashima, H.; Toki, M.; Asakuma, N. Ultraviolet-reduced reduction and crystallization of indium oxide films. J. Appl. Phys. 1999, 85, 203–207. [Google Scholar] [CrossRef]
- Bender, M.; Katsarakis, N.; Gagaoudakis, E.; Hourdakis, E.; Douloufakis, E.; Cimalla, V.; Kiriakidis, G. Dependence of the photoreduction and oxidation behavior of indium oxide films on substrate temperature and film thickness. J. Appl. Phys. 2001, 90, 5382–5387. [Google Scholar] [CrossRef]
- Kurmangaleev, K.; Mikhailova, T.Y.; Trakhtenberg, L. Oxygen chemisorption on the surface of an In2O3 (011) nanocrystal. Inorg. Mater. 2020, 56, 1138–1146. [Google Scholar] [CrossRef]
- Sensato, F.R.; Custódio, R.; Calatayud, M.; Beltrán, A.; Andrés, J.; Sambrano, J.R.; Longo, E. Periodic study on the structural and electronic properties of bulk, oxidized and reduced SnO2(110) surfaces and the interaction with O2. Surf. Sci. 2002, 511, 408–420. [Google Scholar] [CrossRef]
- Sopiha, K.V.; Malyi, O.I.; Persson, C.; Wu, P. Chemistry of Oxygen Ionosorption on SnO2 Surfaces. ACS Appl. Mater. Interfaces 2021, 13, 33664–33676. [Google Scholar] [CrossRef]
- Oviedo, J.; Gillan, M. First-principles study of the interaction of oxygen with the SnO2(110) surface. Surf. Sci. 2001, 490, 221–236. [Google Scholar] [CrossRef]
- Marikutsa, A.; Rumyantseva, M.; Konstantinova, E.A.; Gaskov, A. The Key Role of Active Sites in the Development of Selective Metal Oxide Sensor Materials. Sensors 2021, 21, 2554. [Google Scholar] [CrossRef]
- Gurwitz, R.; Cohen, R.; Shalish, I. Interaction of light with the ZnO surface: Photon induced oxygen “breathing,” oxygen vacancies, persistent photoconductivity, and persistent photovoltage. J. Appl. Phys. 2014, 115, 033701. [Google Scholar] [CrossRef]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chizhov, A.; Kutukov, P.; Astafiev, A.; Rumyantseva, M. Photoactivated Processes on the Surface of Metal Oxides and Gas Sensitivity to Oxygen. Sensors 2023, 23, 1055. https://doi.org/10.3390/s23031055
Chizhov A, Kutukov P, Astafiev A, Rumyantseva M. Photoactivated Processes on the Surface of Metal Oxides and Gas Sensitivity to Oxygen. Sensors. 2023; 23(3):1055. https://doi.org/10.3390/s23031055
Chicago/Turabian StyleChizhov, Artem, Pavel Kutukov, Artyom Astafiev, and Marina Rumyantseva. 2023. "Photoactivated Processes on the Surface of Metal Oxides and Gas Sensitivity to Oxygen" Sensors 23, no. 3: 1055. https://doi.org/10.3390/s23031055
APA StyleChizhov, A., Kutukov, P., Astafiev, A., & Rumyantseva, M. (2023). Photoactivated Processes on the Surface of Metal Oxides and Gas Sensitivity to Oxygen. Sensors, 23(3), 1055. https://doi.org/10.3390/s23031055






