Design and Fabrication of a Thin and Micro-Optical Sensor for Rapid Prototyping
Abstract
:1. Introduction
2. Materials and Methods
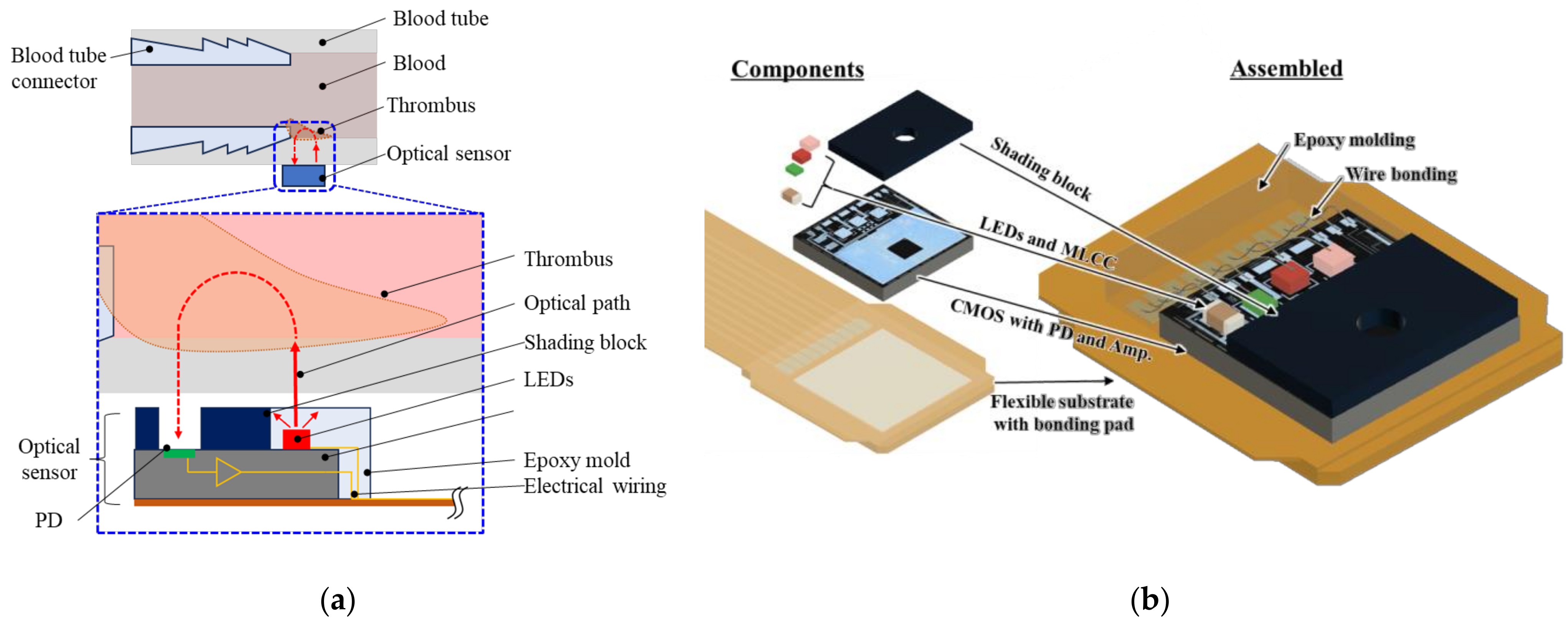
2.1. Basic Design of the Optical Sensor
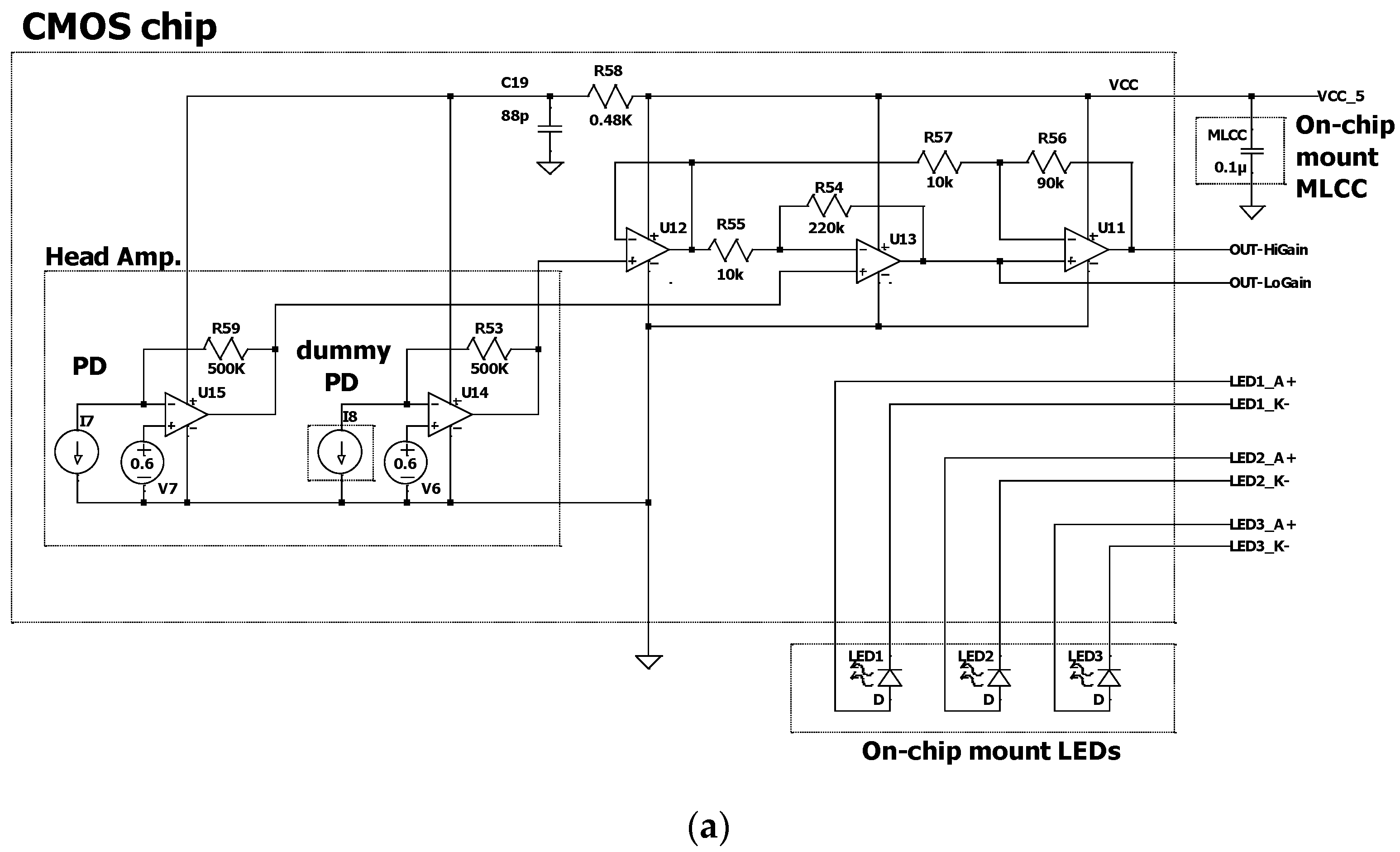
2.2. CMOS Chip
2.3. Shading Block
2.4. Assembly
2.5. Evaluation of Light Detection Performance
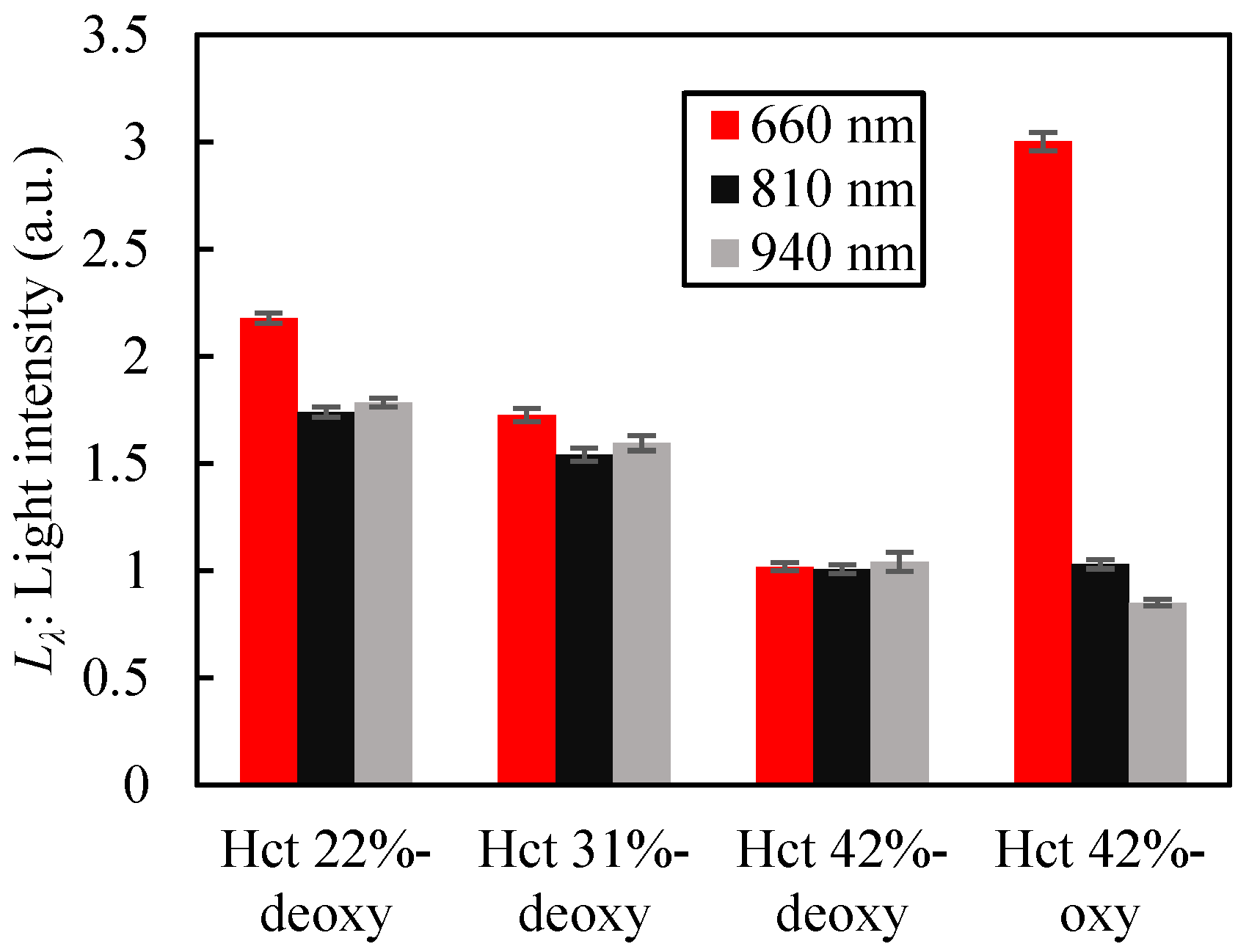
2.6. Optical Measurement Test of Blood
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Balas, C. Review of biomedical optical imaging—A powerful, non-invasive, non-ionizing technology for improving in vivo diagnosis. Meas. Sci. Technol. 2009, 20, 104020. [Google Scholar] [CrossRef]
- Song, G.; Chu, K.K.; Kim, S.; Crose, M.; Cox, B.; Jelly, E.T.; Ulrich, J.N.; Wax, A. First Clinical Application of Low-Cost OCT. Transl. Vis. Sci. Technol. 2019, 8, 61. [Google Scholar] [CrossRef]
- Shin, Y.-H.; Teresa Gutierrez-Wing, M.; Choi, J.-W. Review—Recent Progress in Portable Fluorescence Sensors. J. Electrochem. Soc. 2021, 168, 017502. [Google Scholar] [CrossRef]
- Heikenfeld, J.; Jajack, A.; Rogers, J.; Gutruf, P.; Tian, L.; Pan, T.; Li, R.; Khine, M.; Kim, J.; Wang, J.; et al. Wearable sensors: Modalities, challenges, and prospects. Lab Chip 2018, 18, 217–248. [Google Scholar] [CrossRef]
- Morita, N.; Nogami, H.; Higurashi, E.; Sawada, R. Grasping Force Control for a Robotic Hand by Slip Detection Using Developed Micro Laser Doppler Velocimeter. Sensors 2018, 18, 326. [Google Scholar] [CrossRef]
- Sugie, K.; Sasagawa, K.; Guinto, M.; Haruta, M.; Tokuda, T.; Ohta, J. Implantable CMOS image sensor with incident-angle-selective pixels. Electron. Lett. 2019, 55, 729–731. [Google Scholar] [CrossRef]
- Liao, Z.; Zhang, Y.; Li, Y.; Miao, Y.; Gao, S.; Lin, F.; Deng, Y.; Geng, L. Microfluidic chip coupled with optical biosensors for simultaneous detection of multiple analytes: A review. Biosens. Bioelectron. 2019, 126, 697–706. [Google Scholar] [CrossRef]
- Chen, Y.-T.; Lee, Y.-C.; Lai, Y.-H.; Lim, J.-C.; Huang, N.-T.; Lin, C.-T.; Huang, J.-J. Review of Integrated Optical Biosensors for Point-of-Care Applications. Biosensors 2020, 10, 209. [Google Scholar] [CrossRef]
- Umapathi, R.; Park, B.; Sonwal, S.; Rani, G.M.; Cho, Y.; Huh, Y.S. Advances in optical-sensing strategies for the on-site detection of pesticides in agricultural foods. Trends Food Sci. Technol. 2022, 119, 69–89. [Google Scholar] [CrossRef]
- Koydemir, H.C.; Ozcan, A. Wearable and Implantable Sensors for Biomedical Applications. Annu. Rev. Anal. Chem. 2018, 11, 127–146. [Google Scholar] [CrossRef]
- ADPD144RI PPG Optical Sensor Module. Available online: https://www.analog.com/jp/products/adpd144ri.html (accessed on 27 May 2023).
- Nissinbo-Microdevices, NJL5510R Datasheet. Available online: https://www.nisshinbo-microdevices.co.jp/ja/products/bio-monitoring-sensor/spec/?product=njl5510r (accessed on 27 May 2023).
- Zeibi Shirejini, S.; Carberry, J.; McQuilten, Z.K.; Burrell, A.J.C.; Gregory, S.D.; Hagemeyer, C.E. Current and future strategies to monitor and manage coagulation in ECMO patients. Thromb. J. 2023, 21, 11. [Google Scholar] [CrossRef]
- Hastings, S.M.; Ku, D.N.; Wagoner, S.; Maher, K.O.; Deshpande, S. Sources of Circuit Thrombosis in Pediatric Extracorporeal Membrane Oxygenation. ASAIO J. 2017, 63, 86–92. [Google Scholar] [CrossRef]
- Morita, N.; Sakota, D.; Oota-Ishigaki, A.; Kosaka, R.; Maruyama, O.; Nishida, M.; Kondo, K.; Takeshita, T.; Iwasaki, W. Real-time, non-invasive thrombus detection in an extracorporeal circuit using micro-optical thrombus sensors. Int. J. Artif. Organs 2021, 44, 565–573. [Google Scholar] [CrossRef]
- Sakota, D.; Murashige, T.; Kosaka, R.; Fujiwara, T.; Ouchi, K.; Nishida, M.; Maruyama, O. Noninvasive optical imaging of thrombus formation in mechanical circulatory support devices. J. Biorheol. 2016, 30, 6–12. [Google Scholar] [CrossRef]
- Mitzner, K. Complete PCB Design Using OrCAD Capture and PCB Editor; Newnes: Burlington, MA, USA, 2009. [Google Scholar]
- Reed, T.B. Induction-coupled plasma torch. J. Appl. Phys. 1961, 32, 821–824. [Google Scholar] [CrossRef]
- Boulos, M.I. The inductively coupled RF (radio frequency) plasma. Pure Appl. Chem. 1985, 57, 1321–1352. [Google Scholar] [CrossRef]
- Laerme, F.; Schilp, A.; Funk, K.; Offenberg, M. Bosch Deep Silicon Etching: Improving Uniformity and Etch Rate for Advanced MEMS Applications. In Proceedings of the Twelfth IEEE International Conference on Micro Electro Mechanical Systems (Cat. No. 99CH36291), Orlando, FL, USA, 21 January 1999; pp. 211–216. [Google Scholar]
- Tanaka, H.; Khumpuang, S.; Hara, S. Small Plasma Space with a Small Plasma Source and Its Advantage in Minimal Fab. J. Photopolym. Sci. Technol. 2019, 32, 747–752. [Google Scholar] [CrossRef]
- Khumpuang, S.; Tanaka, H.; Kara, S. Effective Performance of a Tiny-Chamber Plasma Etcher in Scallop Reduction. In Proceedings of the 2017 IEEE Electron Devices Technology and Manufacturing Conference (EDTM), Toyama, Japan, 28 February–2 March 2017; pp. 150–151. [Google Scholar]
- Roggan, A.; Friebel, M.; DöRschel, K.; Hahn, A.; MüLler, G. Optical Properties of Circulating Human Blood in the Wavelength Range 400–2500 nm. J. Biomed. Opt. 1999, 4, 36. [Google Scholar] [CrossRef]
- Sakota, D.; Kosaka, R.; Nishida, M.; Maruyama, O. Optical aggregometry of red blood cells associated with the blood-clotting reaction in extracorporeal circulation support. J. Artif. Organs 2016, 19, 241–248. [Google Scholar] [CrossRef]
- Sultanova, N.G.; Kasarova, S.N.; Nikolov, I.D. Characterization of optical properties of optical polymers. Opt. Quantum Electron. 2013, 45, 221–232. [Google Scholar] [CrossRef]
- Walker, H.K.; Hall, W.D.; Hurst, J.W. Clinical Methods: The History, Physical, and Laboratory Examinations; Butterworths: Boston, MA, USA, 1990. [Google Scholar]
- EPIGAP OSA LED-Chips. Available online: https://www.epigap-osa.com/products/led-chips/ (accessed on 17 July 2023).





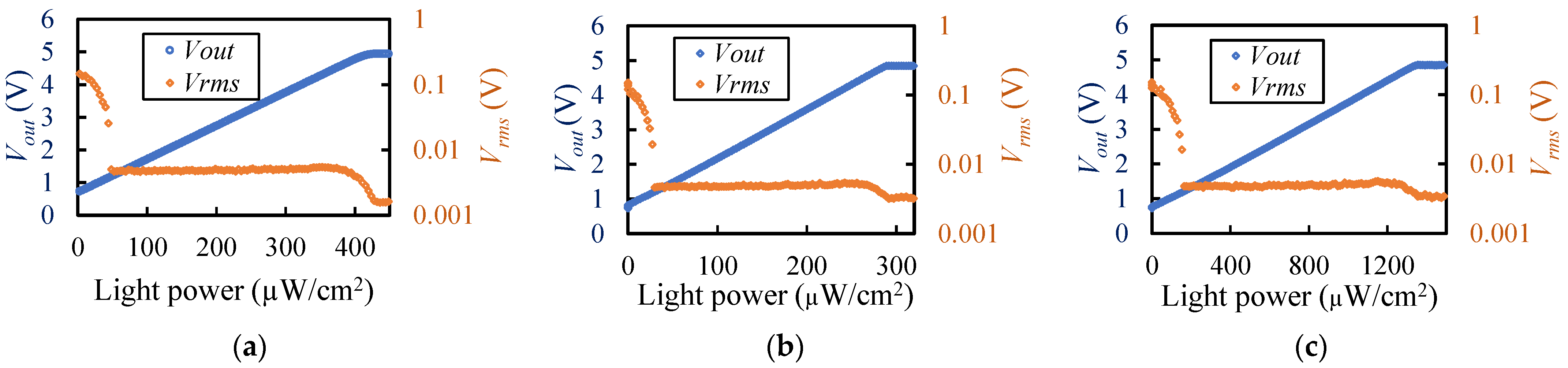
| Wavelength (nm) | Min. Detection Limit (µW/cm2) | Max. Detection Limit (µW/cm2) | Vrms (V) | Vrms in Optical Power (µW/cm2) |
|---|---|---|---|---|
| 660 | 30 | 271 | 0.00493 | 0.350 |
| 810 | 48.3 | 386.1 | 0.00496 | 0.487 |
| 940 | 164.6 | 1271 | 0.00496 | 1.605 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morita, N.; Iwasaki, W. Design and Fabrication of a Thin and Micro-Optical Sensor for Rapid Prototyping. Sensors 2023, 23, 7658. https://doi.org/10.3390/s23177658
Morita N, Iwasaki W. Design and Fabrication of a Thin and Micro-Optical Sensor for Rapid Prototyping. Sensors. 2023; 23(17):7658. https://doi.org/10.3390/s23177658
Chicago/Turabian StyleMorita, Nobutomo, and Wataru Iwasaki. 2023. "Design and Fabrication of a Thin and Micro-Optical Sensor for Rapid Prototyping" Sensors 23, no. 17: 7658. https://doi.org/10.3390/s23177658
APA StyleMorita, N., & Iwasaki, W. (2023). Design and Fabrication of a Thin and Micro-Optical Sensor for Rapid Prototyping. Sensors, 23(17), 7658. https://doi.org/10.3390/s23177658






