A Neural Network Approach for Inertial Measurement Unit-Based Estimation of Three-Dimensional Spinal Curvature
Abstract
1. Introduction
2. Materials and Methods
2.1. Sensor Attachment
2.2. Data collection
2.2.1. Participants
2.2.2. Instrumentation
2.2.3. Motion Capture Experiment
2.3. Data Processing
2.3.1. Motion Capture Data Processing
2.3.2. Sensor Strip Data Processing
2.4. Neural Network
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
| Marker | Error in x-Axis (cm) | Error in y-Axis (cm) | Error in z-Axis (cm) |
|---|---|---|---|
| Mean ± SD | |||
| Marker 2 | −0.039246 ± 0.654629 | 0.009011 ± 0.407979 | 0.009011 ± 0.407979 |
| Marker 3 | −0.082154 ± 1.458481 | 0.101323 ± 0.535569 | −0.018093 ± 1.402238 |
| Marker 4 | −0.144836 ± 1.789642 | 0.046761 ± 0.779287 | −0.166073 ± 1.779907 |
| Marker 5 | −0.291647 ± 2.414006 | 0.099789 ± 0.898368 | −0.245607 ± 2.405230 |
| Marker 6 | −0.469228 ± 3.085831 | 0.176705 ± 1.103621 | −0.440173 ± 3.037856 |
| Marker 7 | −0.632811 ± 3.805284 | 0.213977 ± 1.293863 | −0.593567 ± 3.683613 |
| Marker 8 | −0.821146 ± 4.575898 | 0.298431 ± 1.531894 | −0.721798 ± 4.285308 |
| Marker 9 | −0.995856 ± 5.349810 | 0.353473 ± 1.820733 | −0.879281 ± 4.881974 |
| Marker 10 | −1.187598 ± 6.222460 | 0.343759 ± 2.205439 | −1.053195 ± 5.482679 |
Appendix B
| Item | Subject #1 | Subject #2 |
|---|---|---|
| IMU 1—Acceleration X | −0.04517 | 0.04333 |
| IMU 1—Acceleration Y | 0.61755 | 0.89661 |
| IMU 1—Acceleration Z | −0.67151 | −0.44287 |
| IMU 2—Acceleration X | −0.05713 | 0.00903 |
| IMU 2—Acceleration Y | 0.98938 | 0.99658 |
| IMU 2—Acceleration Z | −0.00684 | 0.03027 |
| IMU 3—Acceleration X | 0.01709 | −0.02991 |
| IMU 3—Acceleration Y | 0.97375 | 0.98401 |
| IMU 3—Acceleration Z | 0.04199 | −0.17786 |
| Marker 2—X | −2.4248275756835938 | −2.441648244857788 |
| Marker 2—Y | 27.026416778564453 | 34.43146133422852 |
| Marker 2—Z | −16.348039627075195 | −8.949712753295898 |
| Marker 3—X | 2.98522424697876 | 1.0537102222442627 |
| Marker 3—Y | 81.97868347167969 | 88.9962158203125 |
| Marker 3—Z | −26.029380798339844 | −15.354696273803713 |
| Marker 4—X | 5.769716739654541 | 2.9824023246765137 |
| Marker 4—Y | 110.39649963378906 | 124.36925506591795 |
| Marker 4—Z | −23.595386505126957 | −6.963764190673828 |
| Marker 5—X | 8.40865707397461 | 5.85613489151001 |
| Marker 5—Y | 160.68186950683594 | 177.68435668945312 |
| Marker 5—Z | −10.85873794555664 | 6.670775413513184 |
| Marker 6—X | 8.916449546813965 | 6.876500129699707 |
| Marker 6—Y | 210.945068359375 | 230.6820373535156 |
| Marker 6—Z | −8.876883506774902 | 12.186483383178713 |
| Marker 7—X | 8.91054630279541 | 7.606303215026855 |
| Marker 7—Y | 258.7609558105469 | 281.3783569335937 |
| Marker 7—Z | −10.991703033447266 | 12.565881729125977 |
| Marker 8—X | 11.808712005615234 | 11.296859741210938 |
| Marker 8—Y | 310.0574035644531 | 334.2227478027344 |
| Marker 8—Z | −14.924169540405272 | 9.177355766296388 |
| Marker 9—X | 13.446818351745604 | 13.42614459991455 |
| Marker 9—Y | 358.4640197753906 | 384.51568603515625 |
| Marker 9—Z | −24.18433952331543 | 1.3085908889770508 |
| Marker 10—X | 12.886392593383787 | 13.651782035827637 |
| Marker 10—Y | 403.5655212402344 | 431.4049072265625 |
| Marker 10—Z | −47.075775146484375 | −20.73443984985352 |
| Cobb Angle | 10.957183742351745 | 13.13111123849918 |
References
- Frost, B.A.; Camarero-Espinosa, S.; Foster, E.J. Materials for the Spine: Anatomy, Problems, and Solutions. Materials 2019, 12, 253. [Google Scholar] [CrossRef]
- Kostuik, J.P.; Bentivoglio, J. The Incidence of Low-Back Pain in Adult Scoliosis. Spine 1981, 6, 268–273. [Google Scholar] [CrossRef]
- Coenen, P.; Smith, A.; Paananen, M.; Osullivan, P.; Beales, D.; Straker, L. Trajectories of Low Back Pain from Adolescence to Young Adulthood. Arthritis Care Res. 2017, 69, 403–412. [Google Scholar] [CrossRef]
- Alshami, A.M. Prevalence of Spinal Disorders and Their Relationships with Age and Gender. Saudi Med. J. 2015, 36, 725–730. [Google Scholar] [CrossRef]
- Borzı`, F.; Szychlinska, M.; Rosa, M.D.; Musumeci, G. A Short Overview of the Effects of Kinesio Taping for Postural Spine Curvature Disorders. J. Funct. Morphol. Kinesiol. 2018, 3, 59. [Google Scholar] [CrossRef]
- Raciborski, F.; Gasik, R.; Klak, A. Disorders of the spine. A major health and social problem. Reumatol./Rheumatol. 2016, 4, 196–200. [Google Scholar] [CrossRef]
- Teixeira da Silva, L.E.C.; de Barros, A.G.C.; de Azevedo, G.B.L. Management of severe and rigid idiopathic scoliosis. Eur. J. Orthop. Surg. Traumatol. Orthop. Traumatol. 2015, 25 (Suppl. S1), S7–S12. [Google Scholar] [CrossRef]
- Kwok, G.; Yip, J.; Yick, K.L.; Cheung, M.C.; Tse, C.Y.; Ng, S.P.; Luximon, A. Postural Screening for Adolescent Idiopathic Scoliosis with Infrared Thermography. Sci. Rep. 2017, 7, 14431. [Google Scholar] [CrossRef]
- Akazawa, T.; Torii, Y.; Ueno, J.; Saito, A.; Niki, H. Mobile Application for Scoliosis Screening Using a Standard 2D Digital Camera. Cureus 2021, 13, e13944. [Google Scholar] [CrossRef]
- Oakley, P.A.; Ehsani, N.N.; Harrison, D.E. The Scoliosis Quandary: Are Radiation Exposures from Repeated X-Rays Harmful? Dose-Response 2019, 17, 155932581985281. [Google Scholar] [CrossRef]
- Tambe, A.D.; Panikkar, S.J.; Millner, P.A.; Tsirikos, A.I. Current concepts in the surgical management of adolescent idiopathic scoliosis. Bone Jt. J. 2018, 100-B, 415–424. [Google Scholar] [CrossRef]
- Fairbank, J. Historical Perspective. Spine 2004, 29, 1953–1955. [Google Scholar] [CrossRef]
- Plaszewski, M.; Nowobilski, R.; Kowalski, P.; Cieslinski, M. Screening for scoliosis. Int. J. Rehabil. Res. 2012, 35, 13–19. [Google Scholar] [CrossRef]
- Senkoylu, A.; Ilhan, M.N.; Altun, N.; Samartzis, D.; Luk, K.D.K. A simple method for assessing rotational flexibility in adolescent idiopathic scoliosis: Modified Adam’s forward bending test. Spine Deform. 2020, 9, 333–339. [Google Scholar] [CrossRef]
- Willner, S. Moiré Topography for the Diagnosis and Documentation of Scoliosis. Acta Orthop. Scand. 1979, 50, 295–302. [Google Scholar] [CrossRef]
- Drerup, B.; Hierholzer, E. Automatic localization of anatomical landmarks on the back surface and construction of a body-fixed coordinate system. J. Biomech. 1987, 20, 961–970. [Google Scholar] [CrossRef]
- Bassani, T.; Stucovitz, E.; Galbusera, F.; Brayda-Bruno, M. Is Rasterstereography a Valid Noninvasive Method for the Screening of Juvenile and Adolescent Idiopathic Scoliosis? Eur. Spine J. 2019, 28, 526–535. [Google Scholar] [CrossRef]
- Severijns, P.; Overbergh, T.; Thauvoye, A.; Baudewijns, J.; Monari, D.; Moke, L.; Desloovere, K.; Scheys, L. A subject-specific method to measure dynamic spinal alignment in adult spinal deformity. Spine J. 2020, 20, 934–946. [Google Scholar] [CrossRef]
- Michaud, F.; Lugr’ıs, U.; Cuadrado, J. Determination of the 3D Human Spine Posture from Wearable Inertial Sensors and a Multibody Model of the Spine. Sensors 2022, 22, 4796. [Google Scholar] [CrossRef]
- Janssen, M.; Nabih, A.; Moussa, W.; Kawchuk, G.N.; Carey, J.P. Evaluation of diagnosis techniques used for spinal injury related back pain. Pain Res. Treat. 2011, 2011, 478798. [Google Scholar] [CrossRef]
- Faria, R.; McKenna, C.; Wade, R.; Yang, H.; Woolacott, N.; Sculpher, M. The EOS 2D/3D X-ray imaging system: A cost-effectiveness analysis quantifying the health benefits from reduced radiation exposure. Eur. J. Radiol. 2013, 82, e342–e349. [Google Scholar] [CrossRef]
- Kumar, V.; Cole, A.; Breakwell, L.; Michael, A.L.R. Comparison of the DIERS Formetric 4D Scanner and Plain Radiographs in Terms of Accuracy in Idiopathic Scoliosis Patients. Glob. Spine J. 2016, 6 (Suppl. 1), s-0036. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, K.; Fan, H.; Huang, Z.; Xiang, Y.; Yang, J.; He, L.; Zhang, L.; Yang, Y.; Li, R.; et al. Development and validation of deep learning algorithms for scoliosis screening using back images. Commun. Biol. 2019, 2, 390. [Google Scholar] [CrossRef]
- Moke, L.; Severijns, P.; Schelfaut, S.; Van de Loock, K.; Hermans, L.; Molenaers, G.; Jonkers, I.; Scheys, L. Performance on Balance Evaluation Systems Test (BESTest) Impacts Health-Related Quality of Life in Adult Spinal Deformity Patients. Spine 2018, 43, 637–646. [Google Scholar] [CrossRef]
- Brage, S.; Brage, N.; Franks, P.W.; Ekelund, U.; Wareham, N.J. Reliability and validity of the combined heart rate and movement sensor Actiheart. Eur. J. Clin. Nutr. 2005, 59, 561–570. [Google Scholar] [CrossRef]
- Irawan, Y.; Fernando, Y.; Wahyuni, R. Detecting Heart Rate Using Pulse Sensor As Alternative Knowing Heart Condition. J. Appl. Eng. Technol. Sci. 2019, 1, 30–42. [Google Scholar] [CrossRef]
- Maity, K.; Mondal, A.; Saha, M.C. Cellulose Nanocrystal-Based All-3D-Printed Pyro-Piezoelectric Nanogenerator for Hybrid Energy Harvesting and Self-Powered Cardiorespiratory Monitoring toward the Human–Machine Interface. ACS Appl. Mater. Interfaces 2023, 15, 13956–13970. [Google Scholar] [CrossRef]
- Aly, A.H.; Zaky, Z.A.; Shalaby, A.S.; Ahmed, A.M.; Vigneswaran, D. Theoretical study of hybrid multifunctional one-dimensional photonic crystal as a flexible blood sugar sensor. Phys. Scr. 2020, 95, 035510. [Google Scholar] [CrossRef]
- Wijaya, N.H.; Oktavihandani, Z.; Kunal, K.; Thelmy, E.; Nguyen, P.T. The Design of Tympani Thermometer Using Passive Infrared Sensor. J. Robot. Control 2020, 1, 27–30. [Google Scholar] [CrossRef]
- Szumilas, M.; Władziński, M.; Wildner, K. A Coupled Piezoelectric Sensor for MMG-Based Human-Machine Interfaces. Sensors 2021, 21, 8380. [Google Scholar] [CrossRef]
- Connolly, J.; Condell, J.; Oflynn, B.; Sanchez, J.T.; Gardiner, P. IMU Sensor-based Electronic Goniometric Glove (iSEG-Glove) for clinical finger movement analysis. IEEE Sens. J. 2017, 18, 1273–1281. [Google Scholar] [CrossRef]
- Kim, M.; Cho, J.; Lee, S.; Jung, Y. IMU Sensor-Based Hand Gesture Recognition for Human-Machine Interfaces. Sensors 2019, 19, 3827. [Google Scholar] [CrossRef]
- Brabandere, A.D.; Emmerzaal, J.; Timmermans, A.; Jonkers, I.; Vanwanseele, B.; Davis, J. A Machine Learning Approach to Estimate Hip and Knee Joint Loading Using a Mobile Phone-Embedded IMU. Front. Bioeng. Biotechnol. 2020, 8, 320. [Google Scholar] [CrossRef]
- Beange KHe Chan ADc Beaudette, S.M.; Graham, R.B. Concurrent validity of a wearable IMU for objective assessments of functional movement quality and control of the lumbar spine. J. Biomech. 2019, 97, 109356. [Google Scholar] [CrossRef]
- Lim, K.; Park, S. Prediction of Lower Limb Kinetics and Kinematics during Walking by a Single IMU on the Lower Back Using Machine Learning. Sensors 2019, 20, 130. [Google Scholar] [CrossRef]
- Kim, M.; Park, S. Golf Swing Segmentation from a Single IMU Using Machine Learning. Sensors 2020, 20, 4466. [Google Scholar] [CrossRef]
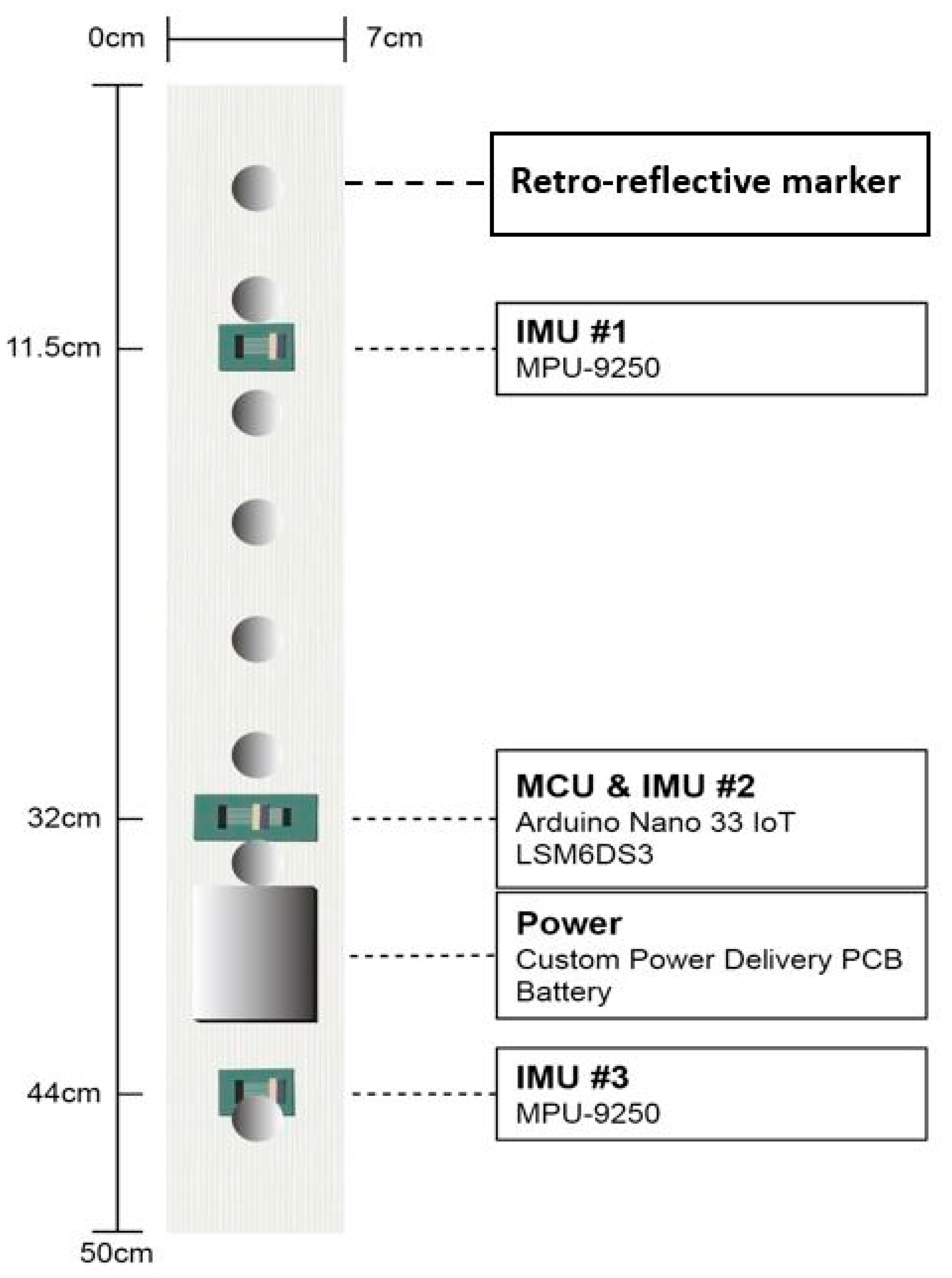
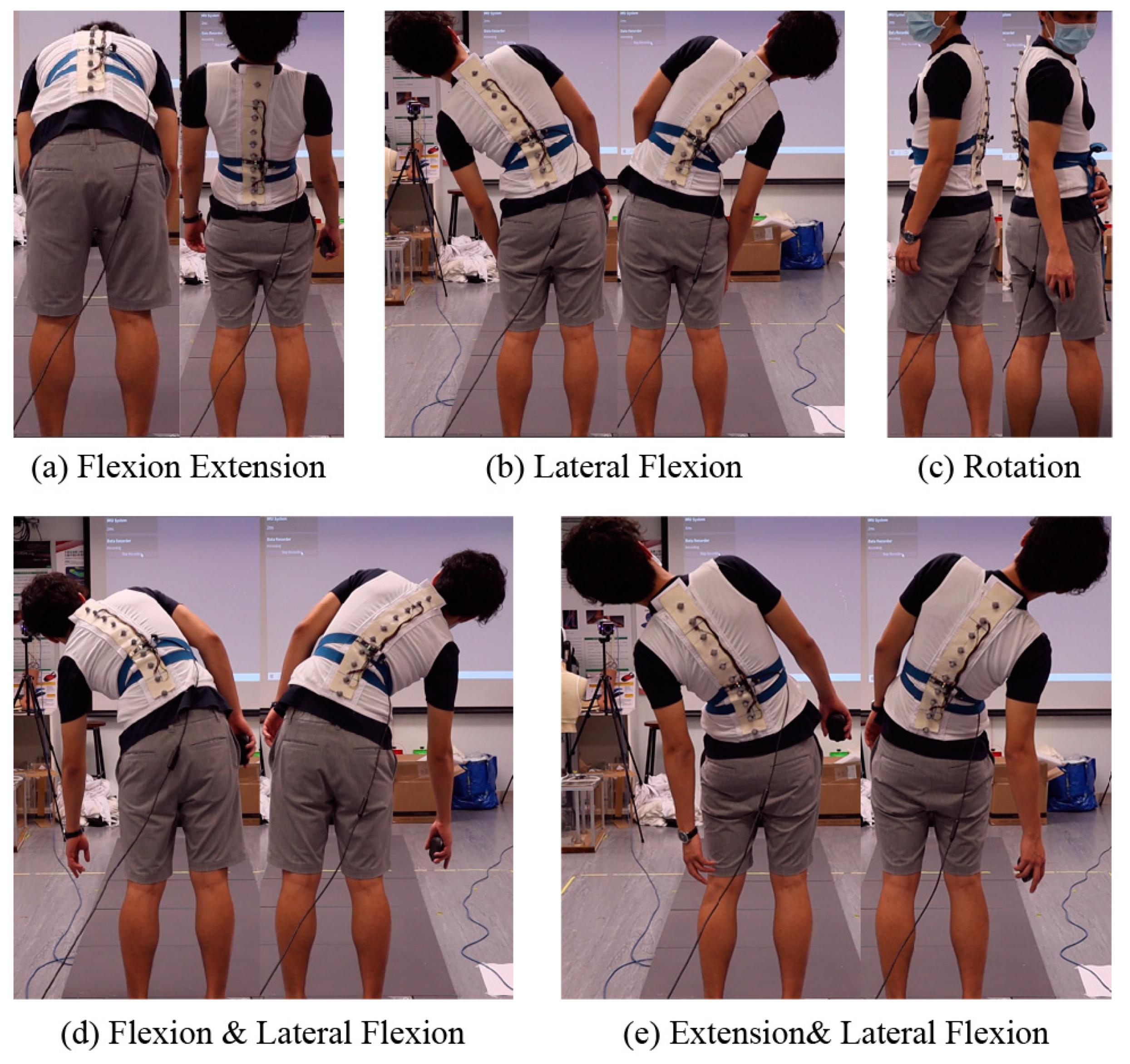
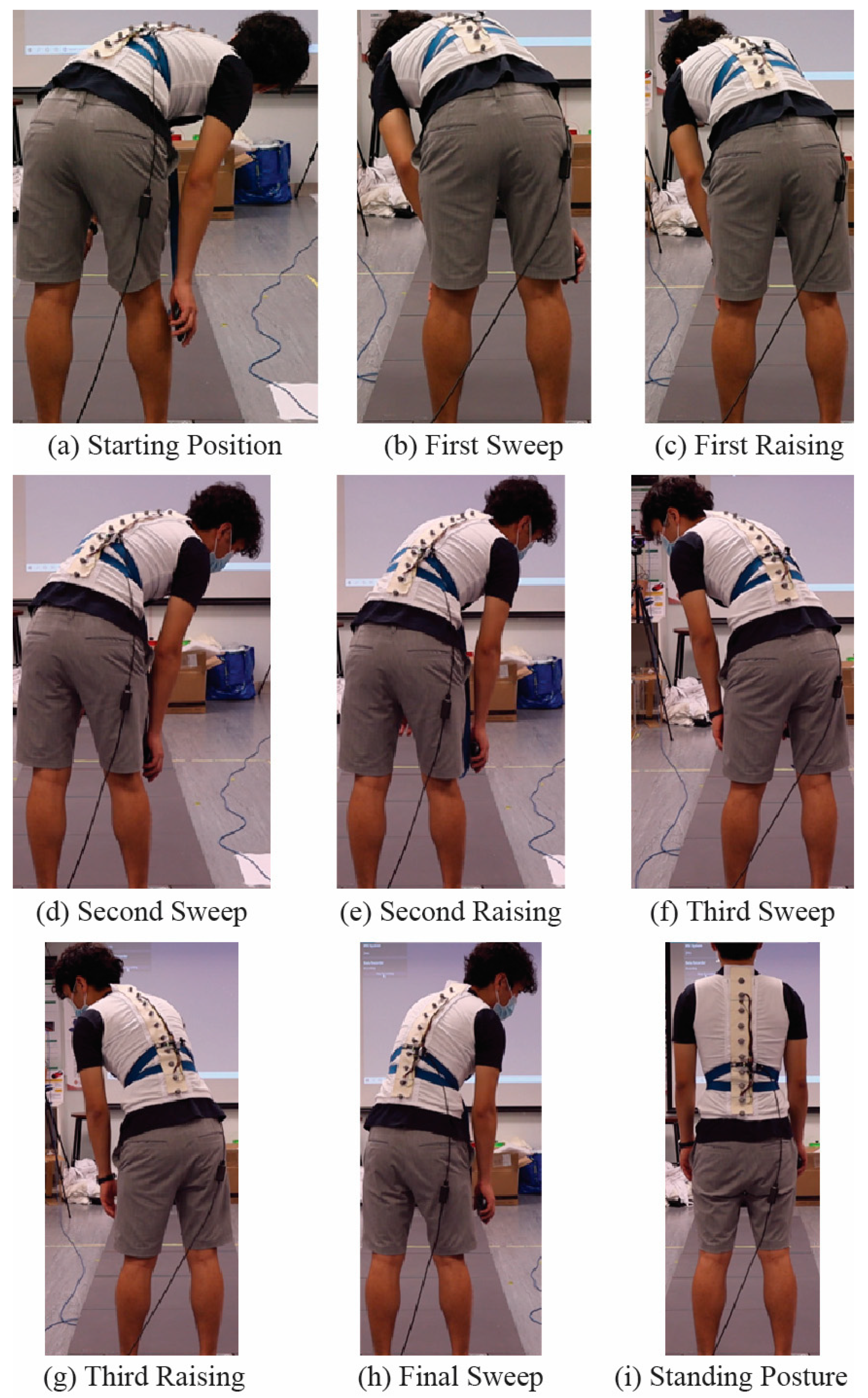


| Characteristics | Value |
|---|---|
| Number of participants | 15 |
| Number of male/female participants | 4/11 |
| Height (cm) (Mean ± SD) | 163.53 ± 7.74 |
| Length of spine (cm) (C7 to S2) (Mean ± SD) | 50.33 ± 3.96 |
| Age (Mean ± SD) | 25.40 ± 3.85 |
| Learning Rate | Rho | Momentum | Epochs | Batch Size |
|---|---|---|---|---|
| 0.0001 | 0.9 | 0 | 250 | 512 |
| No. of Participants | Gender | Age (Years Old) | Height (cm) | Spine Length (cm) |
|---|---|---|---|---|
| 5 | Male | 26 | 174 | 54 |
| 13 | Female | 24 | 160 | 47 |
| Subject Code | Measured Cobb Angle | |
|---|---|---|
| X-Ray Image (°) | Estimated Spinal Curve (°) | |
| 1 | 11.0 | 10.957184 |
| 2 | 13.1 | 13.131111 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mak, T.H.A.; Liang, R.; Chim, T.W.; Yip, J. A Neural Network Approach for Inertial Measurement Unit-Based Estimation of Three-Dimensional Spinal Curvature. Sensors 2023, 23, 6122. https://doi.org/10.3390/s23136122
Mak THA, Liang R, Chim TW, Yip J. A Neural Network Approach for Inertial Measurement Unit-Based Estimation of Three-Dimensional Spinal Curvature. Sensors. 2023; 23(13):6122. https://doi.org/10.3390/s23136122
Chicago/Turabian StyleMak, T. H. Alex, Ruixin Liang, T. W. Chim, and Joanne Yip. 2023. "A Neural Network Approach for Inertial Measurement Unit-Based Estimation of Three-Dimensional Spinal Curvature" Sensors 23, no. 13: 6122. https://doi.org/10.3390/s23136122
APA StyleMak, T. H. A., Liang, R., Chim, T. W., & Yip, J. (2023). A Neural Network Approach for Inertial Measurement Unit-Based Estimation of Three-Dimensional Spinal Curvature. Sensors, 23(13), 6122. https://doi.org/10.3390/s23136122







