Versatile Confocal Raman Imaging Microscope Built from Off-the-Shelf Opto-Mechanical Components
Abstract
1. Introduction
2. Experimental Setup
- -
- Segment A. Laser light coupling into the CRM via a single-mode (SLM) optical fiber; (optional) monitoring of the laser power; and guidance of laser and Raman light through the microscope objective. Note the subtle differences in Figure 1 and Figure 2; the schematic sketch in Figure 1 shows the setup of our second CRM, in which the laser coupling was switched to the opposite side as in the original CRM (photo in Figure 2), making the system more compact.
- -
- Segment B. Wide-field imaging arm to record images of the sample using a 2D CMOS camera.
- -
- Segment C. Confocal detection arm to image the laser excitation region on the sample onto the confocal pinhole and onto a fiber bundle carrying the Raman light to the spectrometer.
2.1. CRM Construction
2.2. Motorized Sample Motion
- -
- a three-axis combination of stepper-motorized translation stages Standa 8MT173-20-MEn1 with optical encoders (with full-step resolution = 1.25 μm and nominal micro-step resolution = 0.156 μm ≡ 1/8 step); and
- -
- a combination of piezo-inertia translators; comprising two Thorlabs PD1/M linear stages (with nominal step resolution = 1 μm) for the lateral xy-motion, and a Thorlabs LX20/M translation stage with PIA25 actuator (with nominal step resolution = 20 nm) for the axial z-translation.
3. Alignment and Characterization
3.1. Alignment of Confocal Raman Light Path
3.2. Alignment and Use of the Wide Field CMOS Camera Path
3.3. Axial Focussing onto the Sample Surface
4. Results of Test Measurements
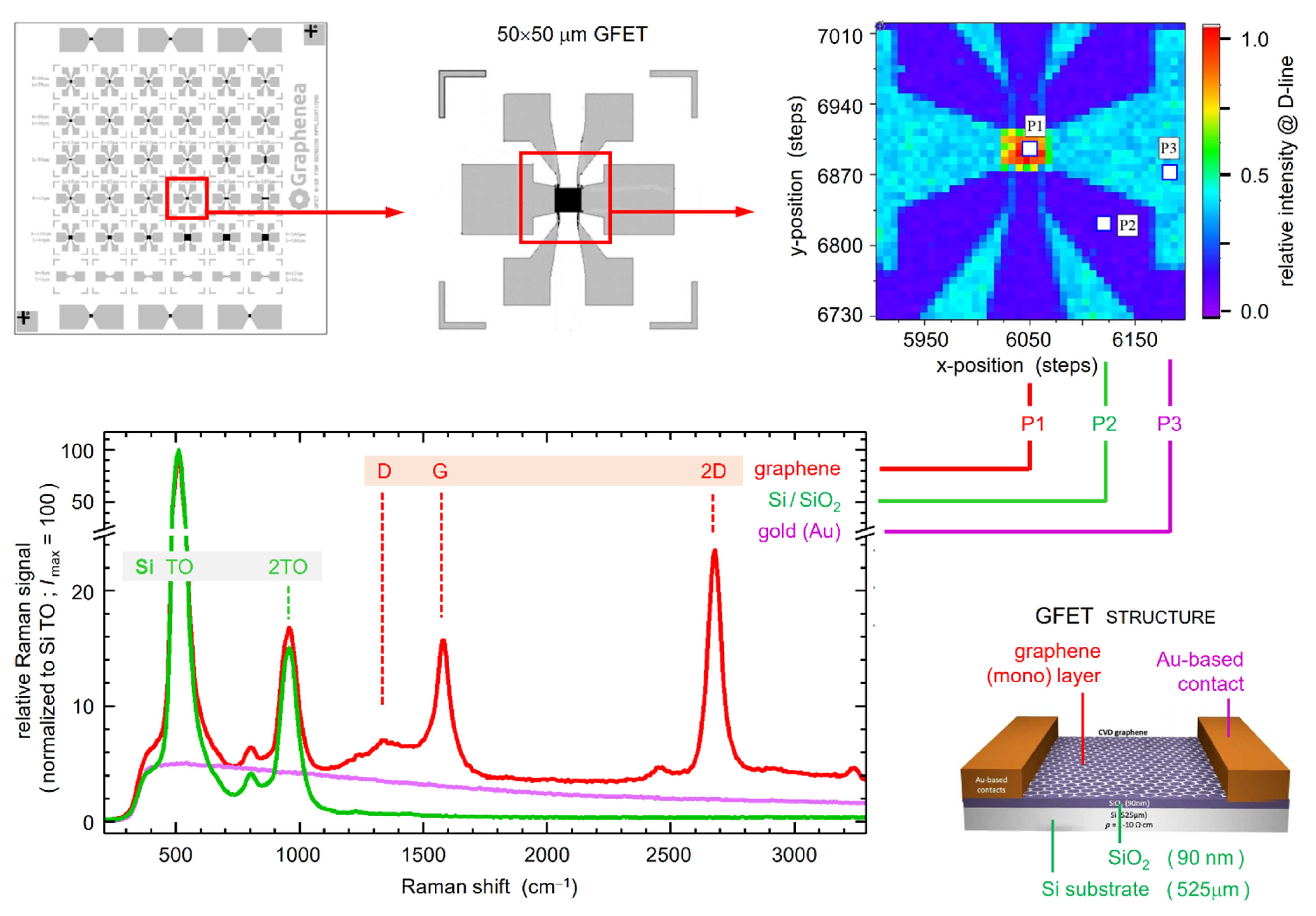
4.1. Concepts of Raster Scans and Raman Signal Analysis
- -
- P1: the spectral slice incorporates the D-line signal of graphene (plus residual background from the substrate material);
- -
- P2: there is almost no signal contribution from the SiO2 substrate within the slice;
- -
- P3: fluorescence from the gold contact contributes to the spectral slice.
4.2. Determination of the Laser Focal Beam Diameter
4.3. The Use of Different Pin Holes and Objectives
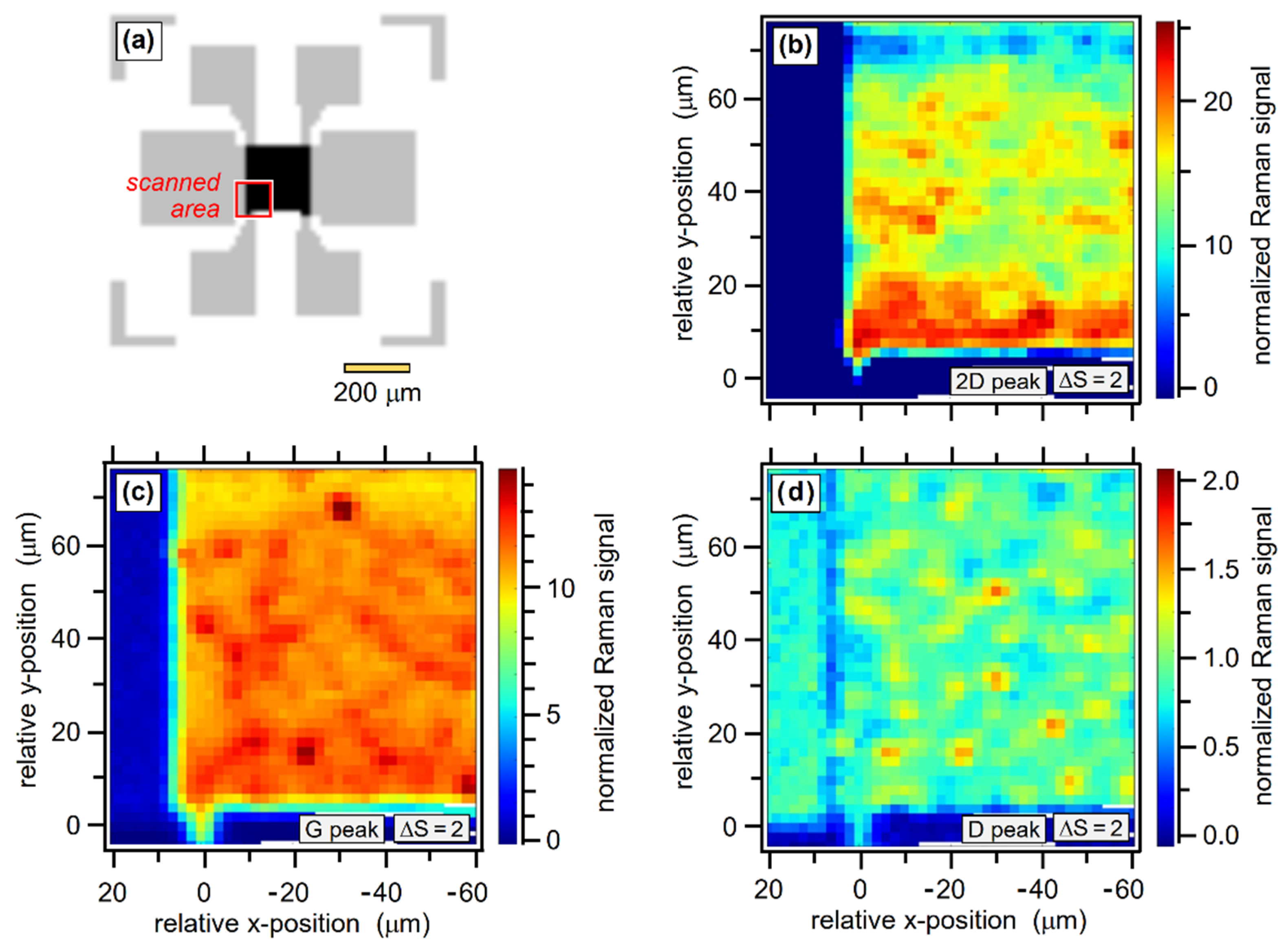
4.4. Raster Scans of Graphene Samples
4.5. Comparison of Raman Raster Maps with Inages Obtained by Complementary Techniques
5. Conclusions and Outlook
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Stewart, S.; Priore, R.J.; Nelson, M.P.; Treado, P.J. Raman Imaging. Annu. Rev. Anal. Chem. 2012, 5, 337–360. [Google Scholar] [CrossRef] [PubMed]
- Opilik, L.; Schmid, T.; Zenobi, R. Modern Raman imaging: Vibrational spectroscopy on the micrometer and nanometer scales. Annu. Rev. Anal. Chem. 2013, 6, 379–398. [Google Scholar] [CrossRef] [PubMed]
- Ni, Z.; Wang, Y.; Yu, T.; Shen, Z. Raman Spectroscopy and Imaging of Graphene. Nano Res. 2008, 1, 273–291. [Google Scholar] [CrossRef]
- Toporski, J.; Dieing, T.; Hollricher, O. (Eds.) Confocal Raman Microscopy, 2nd ed.; Springer International Publishing AG: Cham, Switzerland, 2018. [Google Scholar]
- Rzhevskii, A. Modern Raman Microscopy: Technique and Practice; Cambridge Scholars Publishing: Newcastle-upon-Tyne, UK, 2021. [Google Scholar]
- Beltran-Parrazal, L.; Morgado-Valle, C.; Serrano, R.E.; Manzo, J.; Vergara, J.L. Design and construction of a modular low-cost epifluorescence upright microscope for neuron visualized recording and fluorescence detection. J. Neurosci. Methods 2014, 225, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Rosenegger, D.G.; Tran, C.H.T.; LeDue, J.; Zhou, N.; Gordon, G.R. A high performance, cost-effective, open-source microscope for scanning two-photon microscopy that is modular and readily adaptable. PLoS ONE 2014, 9, e110475. [Google Scholar] [CrossRef]
- Ye, X.; McCluskey, M.D. Modular scanning confocal microscope with digital image processing. PLoS ONE 2016, 11, e0166212. [Google Scholar] [CrossRef]
- AlShehab, M.; Mousavi, S.S.; Amyot-Bourgeois, M.; Walia, J.; Olivieri, A.; Ghamsari, B.; Berini, P. Design and construction of a Raman microscope and characterization of plasmon-enhanced Raman scattering in graphene. J. Opt. Soc. Am. B 2019, 36, F49–F59. [Google Scholar] [CrossRef]
- Gu, M.; Sheppard, C.J.R.; Gan, X. Image formation in a fiber-optical confocal scanning microscope. J. Opt. Soc. Am. A 1991, 8, 1755–1761. [Google Scholar] [CrossRef]
- Dabbs, T.; Glass, M. Single-mode fibers used as confocal microscope pinholes. Appl. Opt. 1992, 31, 705–706. [Google Scholar] [CrossRef]
- Aspnes, D.E.; Studna, A.A. Dielectric functions and optical parameters of Si, Ge, GaP, GaAs, GaSb, InP, InAs, and InSb from 1.5 to 6.0 eV. Phys. Rev. B 1983, 27, 985–1009. [Google Scholar] [CrossRef]
- Green, M.A.; Keevers, M. Optical properties of intrinsic silicon at 300 K. Prog. Photovolt. 1995, 3, 189–192. Online access to numerical Si-reflectivity data: https://www.pveducation.org/pvcdrom/materials/optical-properties-of-silicon#footnote1_ciy5rpk (accessed on 10 December 2022). [CrossRef]
- Deschaines, T.; Hodkiewicz, J.; Henson, P. Characterization of Amorphous and Microcrystalline Silicon Using Raman Spectroscopy; Application Note 51735; Thermo Fisher Scientific: Madison, WI, USA, 2009; Available online: https://assets.thermofisher.com/TFS-Assets/CAD/Application-Notes/D16998~.pdf (accessed on 10 December 2022).
- Yazdanfar, S.; Kenny, K.B.; Tasimi, K.; Corwin, A.D.; Dixon, E.L.; Filkins, R.J. Simple and robust image-based autofocusing for digital microscopy. Opt. Express 2008, 16, 8670–8677. [Google Scholar] [CrossRef] [PubMed]
- James, T.M.; Schlösser, M.; Lewis, R.J.; Fischer, S.; Bornschein, B.; Telle, H.H. Automated quantitative spectroscopic analysis combining background subtraction, cosmic ray removal, and peak fitting. Appl. Spectrosc. 2013, 67, 949–959. [Google Scholar] [CrossRef] [PubMed]
- Keshava, N.; Mustard, J.F. Spectral unmixing. IEEE Signal Proc. Mag. 2002, 19, 44–57. [Google Scholar] [CrossRef]
- Telle, H.H.; Ureña, A.G. Laser Spectroscopy and Laser Imaging—An Introduction; CRC Press, Taylor & Francis Group: Boca Raton, FL, USA, 2018. [Google Scholar]
- Schmidt, R.W.; Woutersen, S.; Ariese, F. RamanLIGHT—A graphical user-friendly tool for pre-processing and unmixing hyperspectral Raman spectroscopy images. J. Opt. 2022, 24, 064011. [Google Scholar] [CrossRef]
- He, H.; Lin, C.; Zong, C.; Xu, M.; Zheng, P.; Ye, R.; Wang, L.; Ren, B. Automated weak signal extraction of hyperspectral Raman imaging data by adaptive low-rank matrix approximation. J. Raman Spectrosc. 2020, 51, 2552–2561. [Google Scholar] [CrossRef]
- Smith, J.P.; Liu, M.; Lauro, M.L.; Balasubramanian, M.; Forstater, J.H.; Grosser, S.T.; Dance, Z.E.X.; Rhodes, T.A.; Bu, X.; Booksh, K.S. Raman hyperspectral imaging with multivariate analysis for investigating enzyme immobilization. Analyst 2020, 145, 7571–7581. [Google Scholar] [CrossRef]
- Skokan, L.; Ruediger, A.; Muehlethaler, C. Hyperspectral Raman imaging and multivariate statistical analysis for the reconstruction of obliterated serial numbers in polymers. J. Raman Spectrosc. 2022, 53, 1415–1427. [Google Scholar] [CrossRef]
- Wu, J.B.; Lin, M.L.; Cong, X.; Liu, H.N.; Tan, P.H. Raman spectroscopy of graphene-based materials and its applications in related devices. Chem. Soc. Rev. 2018, 47, 1822–1873. [Google Scholar] [CrossRef]
- Melios, C.; Huang, N.; Callegaro, L.; Centeno, A.; Cultrera, A.; Cordon, A.; Panchal, V.; Arnedo, I.; Redo-Sanchez, A.; Etayo, D.; et al. Towards standardisation of contact and contactless electrical measurements of CVD graphene at the macro-, micro- and nano-scale. Sci. Rep. 2020, 10, 3223. [Google Scholar] [CrossRef]
- Khosrofian, J.M.; Garetz, B.A. Measurement of a Gaussian laser beam diameter through the direct inversion of knife-edge data. Appl. Opt. 1983, 22, 3406–3410. [Google Scholar] [CrossRef] [PubMed]
- de Araújo, M.A.C.; Silva, R.; de Lima, E.; Pereira, D.P.; de Oliveira, P.C. Measurement of Gaussian laser beam radius using the knife-edge technique: Improvement on data analysis. Appl. Opt. 2009, 48, 393–396. [Google Scholar] [CrossRef] [PubMed]
- Wilson, T. Resolution and optical sectioning in the confocal microscope. J. Microsc. 2011, 244 Pt 2, 113–121. [Google Scholar] [CrossRef]
- Borlinghaus, R.T. Super-Resolution: On a Heuristic Point of View about the Resolution of a Light Microscope. Technological Readings, Leica Microsystems CMS GmbH, Mannheim Germany, December 2014. Available online: https://downloads.leica-microsystems.com/Leica%20TCS%20SP8%20MP/Dedicated%20Articles/Super-Resolution_Technological%20Readings_Dec2014.pdf (accessed on 10 December 2022).
- Borlinghaus, R.T. Pinhole Effect in Confocal Microscopes. Science Lab—The Knowledge Portal of Leica Microsystems CMS GmbH, Mannheim, Germany, 26 April 2017. Available online: https://www.leica-microsystems.com/science-lab/pinhole-effect-in-confocal-microscopes/ (accessed on 10 December 2022).
- Zhang, X.; Chen, S.; Ling, Z.; Zhou, X.; Ding, D.Y.; Kim, Y.S.; Xu, F. Method for removing spectral contaminants to improve analysis of Raman imaging data. Sci. Rep. 2017, 7, 39891. [Google Scholar] [CrossRef] [PubMed]
- Mitsutake, H.; Poppi, R.J.; Breitkreitz, M.C. Raman imaging spectroscopy: History, fundamentals and current scenario of the technique. J. Braz. Chem. Soc. 2019, 30, 2243–2258. [Google Scholar] [CrossRef]
- Ferrari, A.C.; Basko, D.M. Raman spectroscopy as a versatile tool for studying the properties of graphene. Nat. Nanotechnol. 2013, 8, 235–246. [Google Scholar] [CrossRef]
- Lancelot, E. Graphene Studies Using Raman Spectroscopy. Application Note—Nanotechnology RA50. Horiba Jobin Yvon, Palaiseau, France. 2015. Available online: https://static.horiba.com/fileadmin/Horiba/Application/Materials/Material_Research/Graphene/Graphene-studies-using_Raman_Spectroscopy.pdf (accessed on 10 December 2022).
- The KATRIN collaboration; Aker, M.; Altenmüller, K.; Amsbaugh, J.; Arenz, M.; Babutzka, M.; Bast, J.; Bauer, S.; Bechtler, H.; Beck, M.; et al. The design, construction, and commissioning of the KATRIN experiment. J. Instrum. 2021, 16, T08015. [Google Scholar] [CrossRef]
- Trägårdh, J.; MacRae, K.; Travis, C.; Amor, R.; Norris, G.; Wilson, S.H.; Oppo, G.L.; McConnell, G. A simple but precise method for quantitative measurement of the quality of the laser focus in a scanning optical microscope. J. Microscopy 2015, 259, 66–73. [Google Scholar] [CrossRef]
- Itoh, N.; Hanari, N. Reliable evaluation of the lateral resolution of a confocal Raman microscope by using the tungsten-dot array certified reference material. Anal. Sci. 2020, 36, 1009–1013. [Google Scholar] [CrossRef]
- Sacco, A.; Portesi, C.; Giovannozzi, A.M.; Rossi, A.M. Graphene edge method for three-dimensional probing of Raman microscopes focal volumes. J. Raman Spectrosc. 2021, 52, 1671–1684. [Google Scholar] [CrossRef]
- Bian, Z.; Guo, C.; Jiang, S.; Zhu, J.; Wang, R.; Song, P.; Zhang, Z.; Hoshino, K.; Zheng, G. Autofocusing technologies for whole slide imaging and automated microscopy. J. Biophotonics 2020, 13, e202000227. [Google Scholar] [CrossRef] [PubMed]
- Everall, N.J. Confocal Raman microscopy: Performance, pitfalls, and best practice. Appl. Spectrosc. 2009, 63, 245A–262A. [Google Scholar] [CrossRef] [PubMed]
- Everall, N.J. Confocal Raman microscopy: Common errors and artefacts. Analyst 2010, 135, 2512–2522. [Google Scholar] [CrossRef] [PubMed]
- Vallee, O.; Soares, M.; Soares, M. Airy functions and applications to physics, 2nd ed.; Imperial College Press: London, UK, 2010. [Google Scholar]
- Braat, J.J.M.; Dirksen, P.; van Haver, S.; Janssen, A.J.E.M. Extended Nijboer-Zernike (ENZ) Analysis and Aberration Retrieval (Last Update: 25 February 2022). Available online: https://nijboerzernike.nl/ (accessed on 10 December 2022).
- Garcia-Lechuga, M.; Grojo, D. Simple and robust method for determination of laser fluence thresholds for material modifications: An extension of Liu’s approach to imperfect beams. Open Res. Europe 2021, 1, 7. [Google Scholar] [CrossRef]
- Hunstig, M. Piezoelectric Inertia Motors—A critical review of history, concepts, design, applications, and perspectives. Actuators 2017, 6, 7. [Google Scholar] [CrossRef]
- GFET-S10 for Sensing Applications. Graphenea, San Sebastian, Spain. Available online: https://www.graphenea.com/products/gfet-s10-for-sensing-applications-10-mm-x-10-mm (accessed on 10 December 2022).
- Monolayer graphene film on quartz—Product Datasheet. Graphenea, San Sebastian, Spain. Available online: https://cdn.shopify.com/s/files/1/0191/2296/files/Graphenea_Monolayer_Graphene_on_Quartz_Datasheet_02-16-2022.pdf?v=1645025384 (accessed on 10 December 2022).
- Whitener, K.E., Jr. Review Article—Hydrogenated graphene: A user’s guide. J. Vac. Sci. Technol. A 2018, 36, 05G401. [Google Scholar] [CrossRef]
- Fabriciu, A.; Catanzaro, A.; Cultrera, A. (Eds.) The 16NRM01 EMPIR GRACE Consortium. Good Practice Guide on the Electrical Characterisation of Graphene Using Contact Methods. 2020. Available online: http://empir.npl.co.uk/grace/wp-content/uploads/sites/32/2020/04/20200428_GRACE_GPG_1_v1.pdf (accessed on 10 December 2022).
- Jiang, L.; Fu, W.; Birdja, Y.Y.; Koper, M.T.M.; Schneider, G.F. Quantum and electrochemical interplays in hydrogenated graphene. Nature Commun. 2018, 9, 793. [Google Scholar] [CrossRef]
- Scardaci, V.; Compagnini, G. Raman spectroscopy investigation of graphene oxide reduction by laser scribing. Carbon 2021, 7, 48. [Google Scholar] [CrossRef]
- Guillemette, J. Electronic transport in hydrogenated graphene. Ph.D. Thesis, McGill University, Montréal, QC, Canada, 2014. [Google Scholar]
- Lee, E.; Adar, F.; Whitle, A. Multivariate data processing of spectral images: The ugly, the bad, and the true. Spectroscopy 2007, 9, 1–5. [Google Scholar]



| Component | Part Number | Remarks |
|---|---|---|
| Fixed-focus collimator | F260FC-A, Thorlabs | FC-coupled fiber collimator (1) |
| Laser beam steering | KC1-S/M, Thorlabs | kinematic mount |
| Laser line filter | LL01-532-12.5, Semrock | Laser clean-up filter |
| Laser beam separator | Di02-R532-25×36, Semrock | Single-edge dichroic beam splitter |
| Microscope objective | LIO-10X, Newport/MKS | 10× infinity-corrected objective (2) |
| Laser power monitor | SM1PD1A, Thorlabs | SM1-mounted Si photodiode |
| Beam splitter cube | BS025, Thorlabs | Non-polarizing, ratio 10:90 (R:T) |
| Focusing lens | AC254-150-A-ML, Thorlabs | AR-coated achromat, f = 150 mm |
| Color camera | CS165CU/M, Thorlabs | Color CMOS camera, USB2 |
| Raman edge filter | LP03-532RU-25, Semrock | Long-pass 532 nm edge filter (3) |
| Focusing lens | AC254-150-A-ML, Thorlabs | AR-coated achromat, f = 150 mm |
| Confocal pinhole | PxxK, Thorlabs | SS-foil pinhole, xx = ∅ in μm (4) |
| Pinhole xy-adjustment | ST1XY-S/M, Thorlabs | xy-translator (micrometer drive) |
| Spectrometer fiber bundle | Custom-made, CeramOptec | 48-fiber bundle, circular-to-slit (5) |
| Parameter | 8MT173-20-MEn1 | PD1/M | PIA25 (+LX20/M) |
|---|---|---|---|
| Travel range | 20 mm | 20 mm | 25 mm |
| Drive | Stepper motor | Piezo inertia | Piezo inertia |
| Position encoder | Yes | No | No |
| Resolution, full-step | 1.25 μm | 1 μm (3) | 20 nm (3) |
| Resolution, micro-step | 0.156 μm (1/8 step) (1) | NA | NA |
| Backlash | ~2 steps (2) | None | None |
| Speed, continuous stepping | 5 mm/s | 3 mm/s | 2 mm/min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Diaz Barrero, D.; Zeller, G.; Schlösser, M.; Bornschein, B.; Telle, H.H. Versatile Confocal Raman Imaging Microscope Built from Off-the-Shelf Opto-Mechanical Components. Sensors 2022, 22, 10013. https://doi.org/10.3390/s222410013
Diaz Barrero D, Zeller G, Schlösser M, Bornschein B, Telle HH. Versatile Confocal Raman Imaging Microscope Built from Off-the-Shelf Opto-Mechanical Components. Sensors. 2022; 22(24):10013. https://doi.org/10.3390/s222410013
Chicago/Turabian StyleDiaz Barrero, Deseada, Genrich Zeller, Magnus Schlösser, Beate Bornschein, and Helmut H. Telle. 2022. "Versatile Confocal Raman Imaging Microscope Built from Off-the-Shelf Opto-Mechanical Components" Sensors 22, no. 24: 10013. https://doi.org/10.3390/s222410013
APA StyleDiaz Barrero, D., Zeller, G., Schlösser, M., Bornschein, B., & Telle, H. H. (2022). Versatile Confocal Raman Imaging Microscope Built from Off-the-Shelf Opto-Mechanical Components. Sensors, 22(24), 10013. https://doi.org/10.3390/s222410013







