Advantage of Vital Sign Monitoring Using a Wireless Wearable Device for Predicting Septic Shock in Febrile Patients in the Emergency Department: A Machine Learning-Based Analysis
Abstract
1. Introduction
2. Materials and Methods
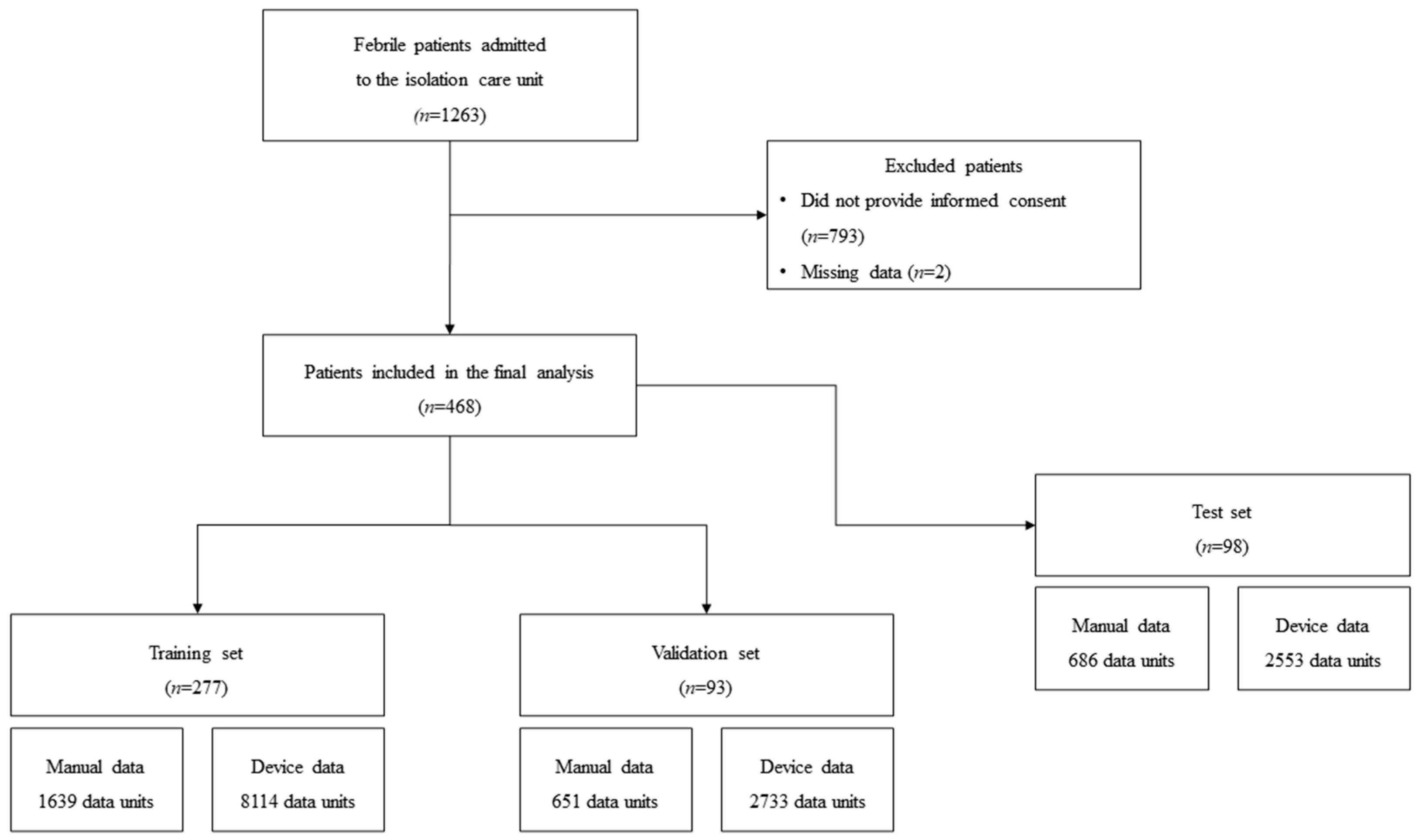
2.1. Study Design and Population
2.2. Description of the Wireless Wearable Device
2.3. Data Acquisition
2.4. Signal Data Filtering Process
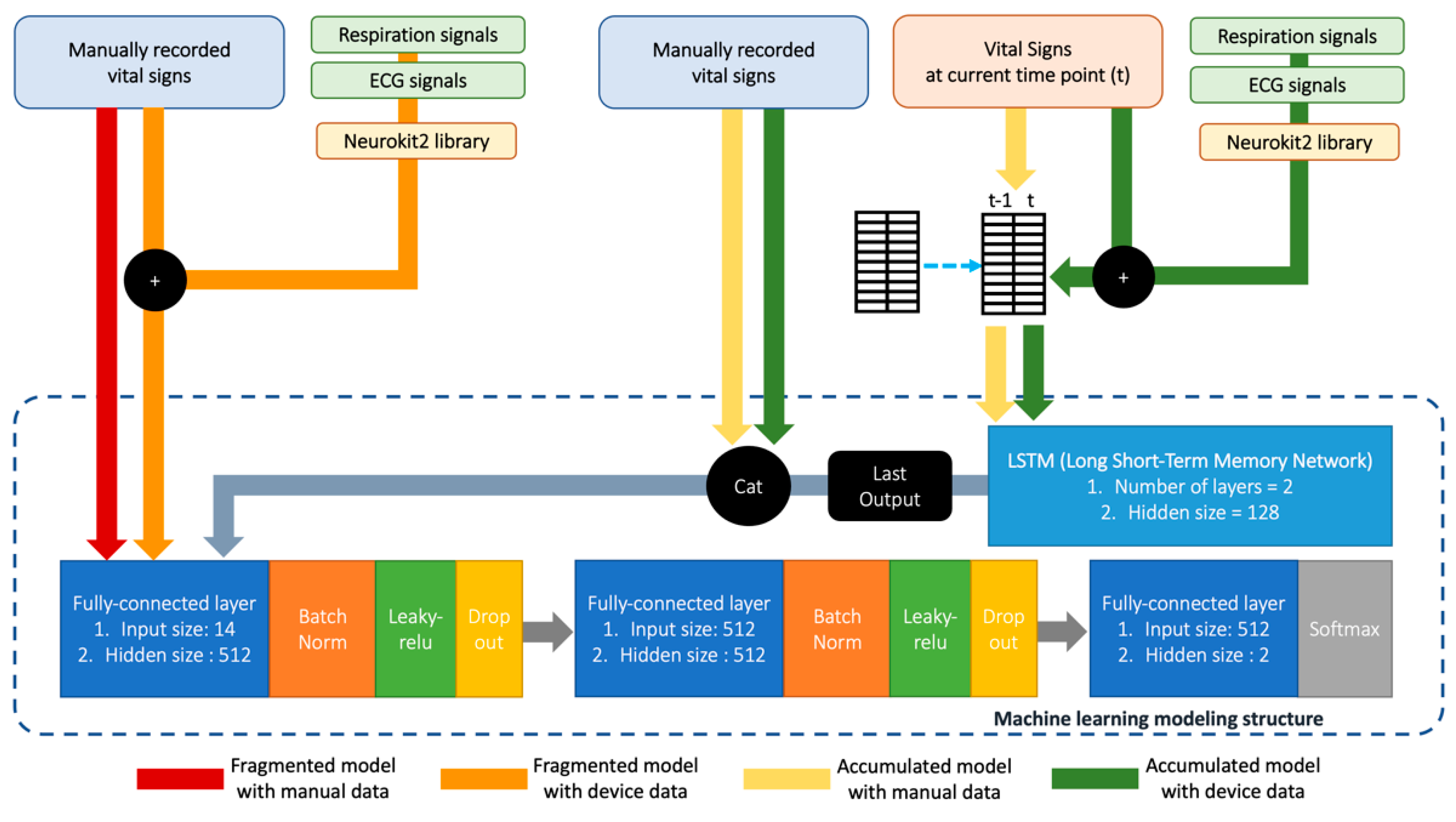
2.5. Development of the Prediction Model
2.6. Outcomes
2.7. Statistical Analysis
3. Results
3.1. Baseline Characteristics
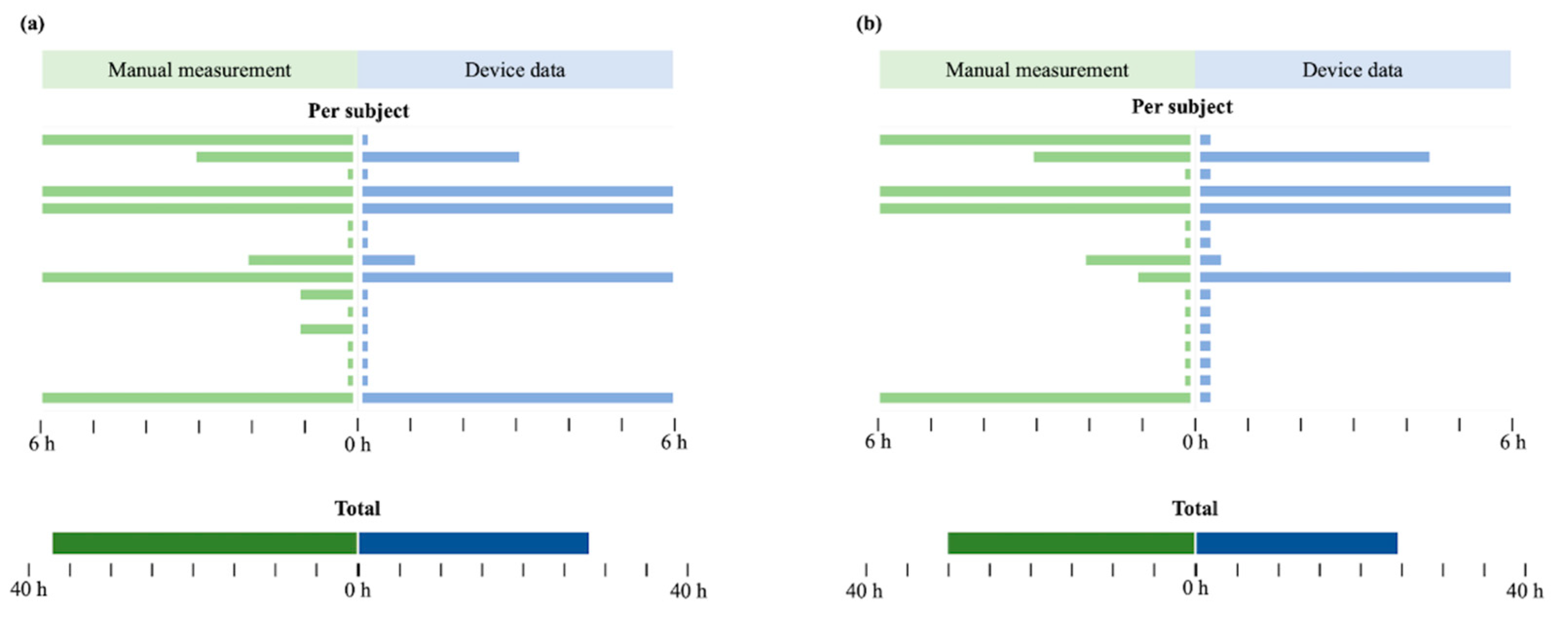
3.2. Reliability of the Wireless Monitoring Device
3.3. Predictive Performance
3.4. Time to Predict Deterioration
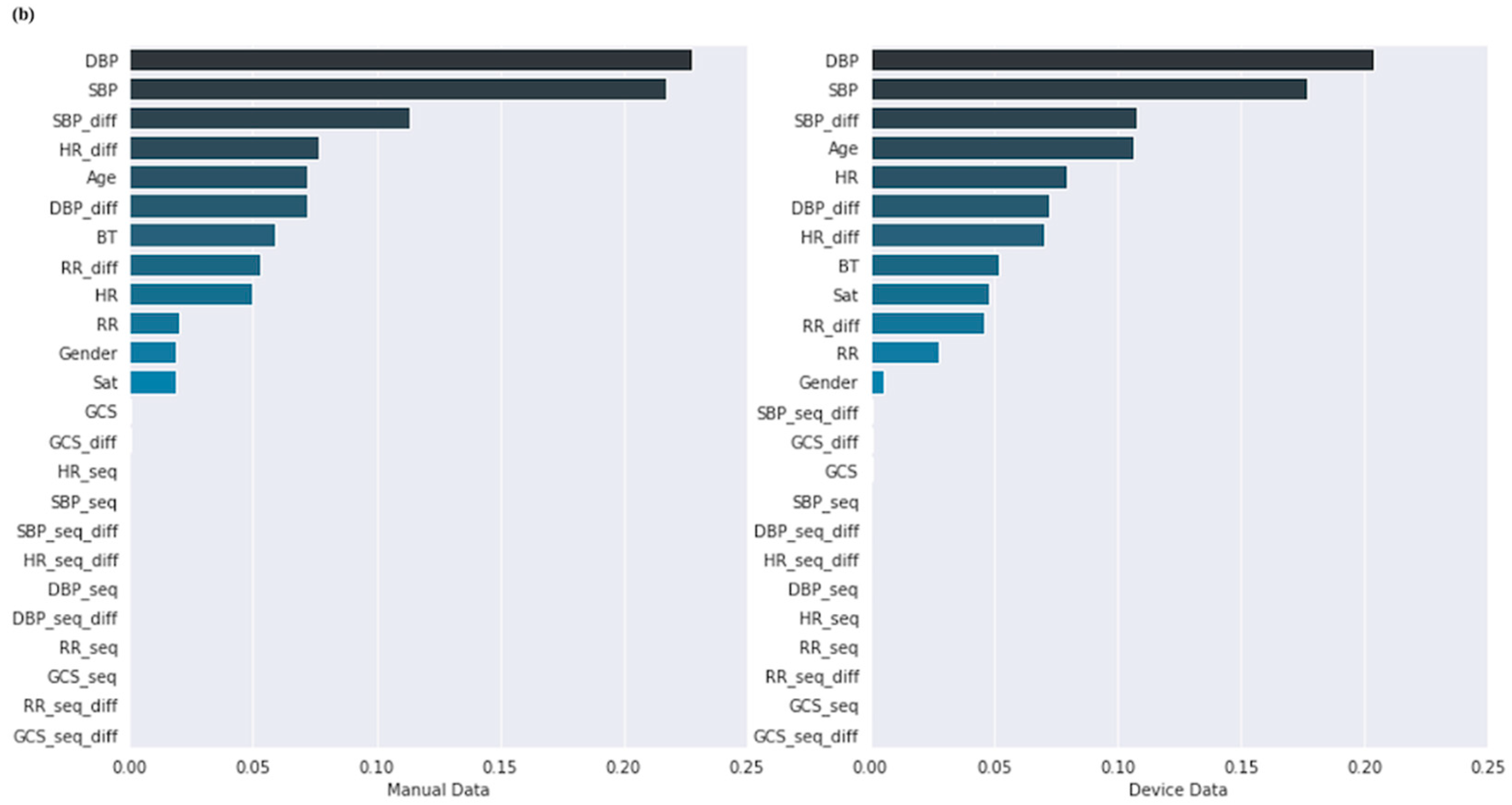
3.5. Feature Importance Scores of the Models
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lee, S.J.; Choi, A.; Ryoo, H.W.; Pak, Y.S.; Kim, H.C.; Kim, J.H. Changes in Clinical Characteristics among Febrile Patients Visiting the Emergency Department before and after the COVID-19 Outbreak. Yonsei Med. J. 2021, 62, 1136–1144. [Google Scholar] [CrossRef] [PubMed]
- Yang, M.; Liu, C.; Wang, X.; Li, Y.; Gao, H.; Liu, X.; Li, J. An Explainable Artificial Intelligence Predictor for Early Detection of Sepsis. Crit. Care Med. 2020, 48, e1091–e1096. [Google Scholar] [CrossRef] [PubMed]
- Cho, K.J.; Kwon, O.; Kwon, J.M.; Lee, Y.; Park, H.; Jeon, K.H.; Kim, K.H.; Park, J.; Oh, B.H. Detecting Patient Deterioration Using Artificial Intelligence in a Rapid Response System. Crit. Care Med. 2020, 48, e285–e289. [Google Scholar] [CrossRef] [PubMed]
- Nemati, S.; Holder, A.; Razmi, F.; Stanley, M.D.; Clifford, G.D.; Buchman, T.G. An Interpretable Machine Learning Model for Accurate Prediction of Sepsis in the ICU. Crit. Care Med. 2018, 46, 547–553. [Google Scholar] [CrossRef] [PubMed]
- Buist, M.; Bernard, S.; Nguyen, T.V.; Moore, G.; Anderson, J. Association between clinically abnormal observations and subsequent in-hospital mortality: A prospective study. Resuscitation 2004, 62, 137–141. [Google Scholar] [CrossRef] [PubMed]
- Cuthbertson, B.H.; Boroujerdi, M.; McKie, L.; Aucott, L.; Prescott, G. Can physiological variables and early warning scoring systems allow early recognition of the deteriorating surgical patient? Crit. Care Med. 2007, 35, 402–409. [Google Scholar] [CrossRef]
- Desautels, T.; Calvert, J.; Hoffman, J.; Jay, M.; Kerem, Y.; Shieh, L.; Shimabukuro, D.; Chettipally, U.; Feldman, M.D.; Barton, C.; et al. Prediction of Sepsis in the Intensive Care Unit With Minimal Electronic Health Record Data: A Machine Learning Approach. JMIR Med. Inform. 2016, 4, e28. [Google Scholar] [CrossRef]
- Mayaud, L.; Lai, P.S.; Clifford, G.D.; Tarassenko, L.; Celi, L.A.; Annane, D. Dynamic data during hypotensive episode improves mortality predictions among patients with sepsis and hypotension. Crit. Care Med. 2013, 41, 954–962. [Google Scholar] [CrossRef]
- Beckett, D.; Gordon, C.; Paterson, R.; Chalkley, S.; Macleod, D.; Bell, D. Assessment of clinical risk in the out of hours hospital prior to the introduction of Hospital at Night. Acute Med. 2009, 8, 33–38. [Google Scholar] [CrossRef]
- Fuhrmann, L.; Lippert, A.; Perner, A.; Ostergaard, D. Incidence, staff awareness and mortality of patients at risk on general wards. Resuscitation 2008, 77, 325–330. [Google Scholar] [CrossRef]
- Calzavacca, P.; Licari, E.; Tee, A.; Egi, M.; Downey, A.; Quach, J.; Haase-Fielitz, A.; Haase, M.; Bellomo, R. The impact of Rapid Response System on delayed emergency team activation patient characteristics and outcomes—A follow-up study. Resuscitation 2010, 81, 31–35. [Google Scholar] [CrossRef] [PubMed]
- Taenzer, A.H.; Pyke, J.B.; McGrath, S.P. A review of current and emerging approaches to address failure-to-rescue. Anesthesiology 2011, 115, 421–431. [Google Scholar] [CrossRef] [PubMed]
- DeVita, M.A.; Smith, G.B.; Adam, S.K.; Adams-Pizarro, I.; Buist, M.; Bellomo, R.; Bonello, R.; Cerchiari, E.; Farlow, B.; Goldsmith, D.; et al. “Identifying the hospitalised patient in crisis”—A consensus conference on the afferent limb of rapid response systems. Resuscitation 2010, 81, 375–382. [Google Scholar] [CrossRef] [PubMed]
- Hammer, J.A.; Jones, T.L.; Brown, S.A. Rapid response teams and failure to rescue: One community’s experience. J. Nurs. Care Qual. 2012, 27, 352–358. [Google Scholar] [CrossRef]
- Weenk, M.; Koeneman, M.; van de Belt, T.H.; Engelen, L.; van Goor, H.; Bredie, S.J.H. Wireless and continuous monitoring of vital signs in patients at the general ward. Resuscitation 2019, 136, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zong, L.; Zhang, J.; Sun, H.; Harold Walline, J.; Sun, P.; Xu, S.; Li, Y.; Wang, C.; Liu, J.; et al. Identifying the effects of an upgraded ‘fever clinic’ on COVID-19 control and the workload of emergency department: Retrospective study in a tertiary hospital in China. BMJ Open 2020, 10, e039177. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.; Kim, H.Y.; Cho, A.; Noh, J.; Park, I.; Chung, H.S. Efficacy of a four-tier infection response system in the emergency department during the coronavirus disease-2019 outbreak. PLoS ONE 2021, 16, e0256116. [Google Scholar] [CrossRef]
- Breteler, M.J.M.; KleinJan, E.; Numan, L.; Ruurda, J.P.; Van Hillegersberg, R.; Leenen, L.P.H.; Hermans, M.; Kalkman, C.J.; Blokhuis, T.J. Are current wireless monitoring systems capable of detecting adverse events in high-risk surgical patients? A descriptive study. Injury 2020, 51 (Suppl. 2), S97–S105. [Google Scholar] [CrossRef]
- Downey, C.; Randell, R.; Brown, J.; Jayne, D.G. Continuous Versus Intermittent Vital Signs Monitoring Using a Wearable, Wireless Patch in Patients Admitted to Surgical Wards: Pilot Cluster Randomized Controlled Trial. J. Med. Internet Res. 2018, 20, e10802. [Google Scholar] [CrossRef]
- Sun, L.; Joshi, M.; Khan, S.N.; Ashrafian, H.; Darzi, A. Clinical impact of multi-parameter continuous non-invasive monitoring in hospital wards: A systematic review and meta-analysis. J. R. Soc. Med. 2020, 113, 217–224. [Google Scholar] [CrossRef]
- Khanna, A.K.; Hoppe, P.; Saugel, B. Automated continuous noninvasive ward monitoring: Future directions and challenges. Crit. Care 2019, 23, 194. [Google Scholar] [CrossRef] [PubMed]
- Breteler, M.J.M.; KleinJan, E.J.; Dohmen, D.A.J.; Leenen, L.P.H.; van Hillegersberg, R.; Ruurda, J.P.; van Loon, K.; Blokhuis, T.J.; Kalkman, C.J. Vital Signs Monitoring with Wearable Sensors in High-risk Surgical Patients: A Clinical Validation Study. Anesthesiology 2020, 132, 424–439. [Google Scholar] [CrossRef] [PubMed]
- Youssef Ali Amer, A.; Wouters, F.; Vranken, J.; de Korte-de Boer, D.; Smit-Fun, V.; Duflot, P.; Beaupain, M.H.; Vandervoort, P.; Luca, S.; Aerts, J.M.; et al. Vital Signs Prediction and Early Warning Score Calculation Based on Continuous Monitoring of Hospitalised Patients Using Wearable Technology. Sensors 2020, 20, 6593. [Google Scholar] [CrossRef] [PubMed]
- Leenen, J.P.L.; Leerentveld, C.; van Dijk, J.D.; van Westreenen, H.L.; Schoonhoven, L.; Patijn, G.A. Current Evidence for Continuous Vital Signs Monitoring by Wearable Wireless Devices in Hospitalized Adults: Systematic Review. J. Med. Internet Res. 2020, 22, e18636. [Google Scholar] [CrossRef]
- Weenk, M.; Bredie, S.J.; Koeneman, M.; Hesselink, G.; van Goor, H.; van de Belt, T.H. Continuous Monitoring of Vital Signs in the General Ward Using Wearable Devices: Randomized Controlled Trial. J. Med. Internet Res. 2020, 22, e15471. [Google Scholar] [CrossRef]
- Jordan, M.I.; Mitchell, T.M. Machine learning: Trends, perspectives, and prospects. Science 2015, 349, 255–260. [Google Scholar] [CrossRef]
- Muralitharan, S.; Nelson, W.; Di, S.; McGillion, M.; Devereaux, P.J.; Barr, N.G.; Petch, J. Machine Learning-Based Early Warning Systems for Clinical Deterioration: Systematic Scoping Review. J. Med. Internet Res. 2021, 23, e25187. [Google Scholar] [CrossRef]
- Mendo, I.R.; Marques, G.; de la Torre Diez, I.; Lopez-Coronado, M.; Martin-Rodriguez, F. Machine Learning in Medical Emergencies: A Systematic Review and Analysis. J. Med. Syst. 2021, 45, 88. [Google Scholar] [CrossRef]
- Brekke, I.J.; Puntervoll, L.H.; Pedersen, P.B.; Kellett, J.; Brabrand, M. The value of vital sign trends in predicting and monitoring clinical deterioration: A systematic review. PLoS ONE 2019, 14, e0210875. [Google Scholar] [CrossRef]
- Shafaf, N.; Malek, H. Applications of Machine Learning Approaches in Emergency Medicine; a Review Article. Arch. Acad. Emerg. Med. 2019, 7, 34. [Google Scholar]
- Stewart, J.; Sprivulis, P.; Dwivedi, G. Artificial intelligence and machine learning in emergency medicine. Emerg. Med. Australas. 2018, 30, 870–874. [Google Scholar] [CrossRef] [PubMed]
- U.S. Food and Drug Administration Center for Devices and Radiological Health. Content of Premarket Submissions for Management of Cybersecurity in Medical Devices; U.S. Food and Drug Administration Center for Devices and Radiological Health: Silver Spring, MD, USA, 2014. Available online: https://www.fda.gov/regulatory-information/search-fda-guidance-documents/content-premarket-submissions-management-cybersecurity-medical-devices (accessed on 19 February 2020).
- International Medical Device Regulators Forum. Principles and Practices for Medical Device Cybersecurity; International Medical Device Regulators Forum: Silver Spring, MD, USA, 2020; Available online: https://www.imdrf.org/sites/default/files/docs/imdrf/final/technical/imdrf-tech-200318-pp-mdc-n60.pdf (accessed on 19 February 2020).
- Song, M.H.; Cho, S.P.; Kim, W.; Lee, K.J. New real-time heartbeat detection method using the angle of a single-lead electrocardiogram. Comput. Biol. Med. 2015, 59, 73–79. [Google Scholar] [CrossRef] [PubMed]
- An Introductory Resource Guide for Implementing the HIPAA Security Rule; US Department of Commerce: Gaithersburg, MD, USA, 2008. Available online: https://csrc.nist.gov/publications/detail/sp/800-66/rev-1/final (accessed on 1 October 2008).
- Khodadad, D.; Nordebo, S.; Muller, B.; Waldmann, A.; Yerworth, R.; Becher, T.; Frerichs, I.; Sophocleous, L.; van Kaam, A.; Miedema, M.; et al. Optimized breath detection algorithm in electrical impedance tomography. Physiol. Meas. 2018, 39, 094001. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, R.; Nishio, M.; Do, R.K.G.; Togashi, K. Convolutional neural networks: An overview and application in radiology. Insights Imaging 2018, 9, 611–629. [Google Scholar] [CrossRef]
- Muraki, R.; Teramoto, A.; Sugimoto, K.; Sugimoto, K.; Yamada, A.; Watanabe, E. Automated detection scheme for acute myocardial infarction using convolutional neural network and long short-term memory. PLoS ONE 2022, 17, e0264002. [Google Scholar] [CrossRef]
- Singer, M.; Deutschman, C.S.; Seymour, C.W.; Shankar-Hari, M.; Annane, D.; Bauer, M.; Bellomo, R.; Bernard, G.R.; Chiche, J.D.; Coopersmith, C.M.; et al. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA 2016, 315, 801–810. [Google Scholar] [CrossRef]
- Darwish, A.; Hassanien, A.E. Wearable and implantable wireless sensor network solutions for healthcare monitoring. Sensors 2011, 11, 5561–5595. [Google Scholar] [CrossRef]
- Majumder, S.; Mondal, T.; Deen, M.J. Wearable Sensors for Remote Health Monitoring. Sensors 2017, 17, 130. [Google Scholar] [CrossRef]
- Michard, F.; Gan, T.J.; Kehlet, H. Digital innovations and emerging technologies for enhanced recovery programmes. Br. J. Anaesth. 2017, 119, 31–39. [Google Scholar] [CrossRef]
- Fung, E.; Jarvelin, M.R.; Doshi, R.N.; Shinbane, J.S.; Carlson, S.K.; Grazette, L.P.; Chang, P.M.; Sangha, R.S.; Huikuri, H.V.; Peters, N.S. Electrocardiographic patch devices and contemporary wireless cardiac monitoring. Front. Physiol. 2015, 6, 149. [Google Scholar] [CrossRef]
- Diomidous, M.; Dalamaga, M.; Nikolopoulos, M.; Tzortzis, E.; Stratigou, T.; Antonakos, G.; Karampela, I. Wireless Monitoring Through Wearable Devices in the ICU: Are We Close? Stud. Health Technol. Inform. 2020, 272, 175–178. [Google Scholar] [CrossRef]
- Downey, C.L.; Tahir, W.; Randell, R.; Brown, J.M.; Jayne, D.G. Strengths and limitations of early warning scores: A systematic review and narrative synthesis. Int. J. Nurs. Stud. 2017, 76, 106–119. [Google Scholar] [CrossRef] [PubMed]
- Clifton, L.; Clifton, D.A.; Pimentel, M.A.; Watkinson, P.J.; Tarassenko, L. Predictive monitoring of mobile patients by combining clinical observations with data from wearable sensors. IEEE J. Biomed. Health Inform. 2014, 18, 722–730. [Google Scholar] [CrossRef]
- Appelboom, G.; Camacho, E.; Abraham, M.E.; Bruce, S.S.; Dumont, E.L.; Zacharia, B.E.; D’Amico, R.; Slomian, J.; Reginster, J.Y.; Bruyere, O.; et al. Smart wearable body sensors for patient self-assessment and monitoring. Arch. Public Health 2014, 72, 28. [Google Scholar] [CrossRef] [PubMed]
- Credland, N.; Dyson, J.; Johnson, M.J. What are the patterns of compliance with Early Warning Track and Trigger Tools: A narrative review. Appl. Nurs. Res. 2018, 44, 39–47. [Google Scholar] [CrossRef] [PubMed]
- Sahandi, R.; Noroozi, S.; Roushan, G.; Heaslip, V.; Liu, Y. Wireless technology in the evolution of patient monitoring on general hospital wards. J. Med. Eng. Technol. 2010, 34, 51–63. [Google Scholar] [CrossRef]
- Kim, J.; Chang, H.; Kim, D.; Jang, D.H.; Park, I.; Kim, K. Machine learning for prediction of septic shock at initial triage in emergency department. J. Crit. Care 2020, 55, 163–170. [Google Scholar] [CrossRef]
- Hong, S.; Lee, S.; Lee, J.; Cha, W.C.; Kim, K. Prediction of Cardiac Arrest in the Emergency Department Based on Machine Learning and Sequential Characteristics: Model Development and Retrospective Clinical Validation Study. JMIR Med. Inform. 2020, 8, e15932. [Google Scholar] [CrossRef]
- Kim, J.H.; Han, S.G.; Cho, A.; Shin, H.J.; Baek, S.E. Effect of deep learning-based assistive technology use on chest radiograph interpretation by emergency department physicians: A prospective interventional simulation-based study. BMC Med. Inform. Decis. Mak. 2021, 21, 311. [Google Scholar] [CrossRef]
- Padilla, R.M.; Mayo, A.M. Clinical deterioration: A concept analysis. J. Clin. Nurs. 2018, 27, 1360–1368. [Google Scholar] [CrossRef]
- Churpek, M.M.; Yuen, T.C.; Edelson, D.P. Predicting clinical deterioration in the hospital: The impact of outcome selection. Resuscitation 2013, 84, 564–568. [Google Scholar] [CrossRef] [PubMed]


| Characteristics | Value |
|---|---|
| sex (male) | 202 (44.20) |
| age (years) | 56.87 ± 18.80 |
| duration of device use (min) | 300.27 ± 100.03 |
| vital signs on arrival | |
| systolic blood pressure (mmHg) | 127.36 ± 22.06 |
| diastolic blood pressure (mmHg) | 75.72 ± 11.46 |
| heart rate (bpm) | 108.59 ± 18.52 |
| respiratory rate (bpm) | 18.16 ± 2.71 |
| body temperature (°C) | 38.64 ± 0.57 |
| oxygen saturation (%) | 96.77 ± 2.47 |
| Glasgow Coma Scale score | 14.98 ± 0.23 |
| use of vasopressors | 85 (18.60) |
| Model | Data | AUROC (95% CI) | AUPRC (95% CI) | Sensitivity (95% CI) | Specificity (95% CI) |
|---|---|---|---|---|---|
| fragmented model | manual data | 0.841 (0.789–0.893) | 0.699 (0.598–0.783) | 0.731 (0.633–0.811) | 0.836 (0.796–0.870) |
| device data | 0.858 (0.809–0.908) | 0.761 (0.664–0.837) | 0.710 (0.611–0.792) | 0.936 (0.907–0.956) | |
| accumulated model | manual data | 0.853 (0.803–0.903) | 0.679 (0.578–0.766) | 0.710 (0.611–0.792) | 0.841 (0.802–0.874) |
| device data | 0.861 (0.811–0.910) | 0.689 (0.588–0.775) | 0.699 (0.599–0.783) | 0.880 (0.844–0.908) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Choi, A.; Chung, K.; Chung, S.P.; Lee, K.; Hyun, H.; Kim, J.H. Advantage of Vital Sign Monitoring Using a Wireless Wearable Device for Predicting Septic Shock in Febrile Patients in the Emergency Department: A Machine Learning-Based Analysis. Sensors 2022, 22, 7054. https://doi.org/10.3390/s22187054
Choi A, Chung K, Chung SP, Lee K, Hyun H, Kim JH. Advantage of Vital Sign Monitoring Using a Wireless Wearable Device for Predicting Septic Shock in Febrile Patients in the Emergency Department: A Machine Learning-Based Analysis. Sensors. 2022; 22(18):7054. https://doi.org/10.3390/s22187054
Chicago/Turabian StyleChoi, Arom, Kyungsoo Chung, Sung Phil Chung, Kwanhyung Lee, Heejung Hyun, and Ji Hoon Kim. 2022. "Advantage of Vital Sign Monitoring Using a Wireless Wearable Device for Predicting Septic Shock in Febrile Patients in the Emergency Department: A Machine Learning-Based Analysis" Sensors 22, no. 18: 7054. https://doi.org/10.3390/s22187054
APA StyleChoi, A., Chung, K., Chung, S. P., Lee, K., Hyun, H., & Kim, J. H. (2022). Advantage of Vital Sign Monitoring Using a Wireless Wearable Device for Predicting Septic Shock in Febrile Patients in the Emergency Department: A Machine Learning-Based Analysis. Sensors, 22(18), 7054. https://doi.org/10.3390/s22187054







