HoloLens 1 vs. HoloLens 2: Improvements in the New Model for Orthopedic Oncological Interventions
Abstract
:1. Introduction
2. Materials and Methods
2.1. Clinical Data
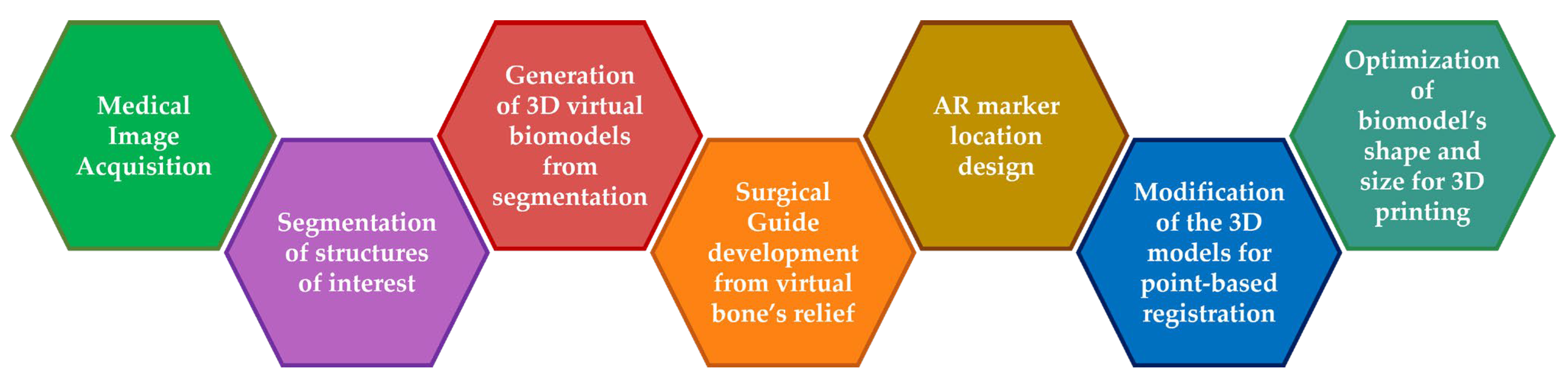
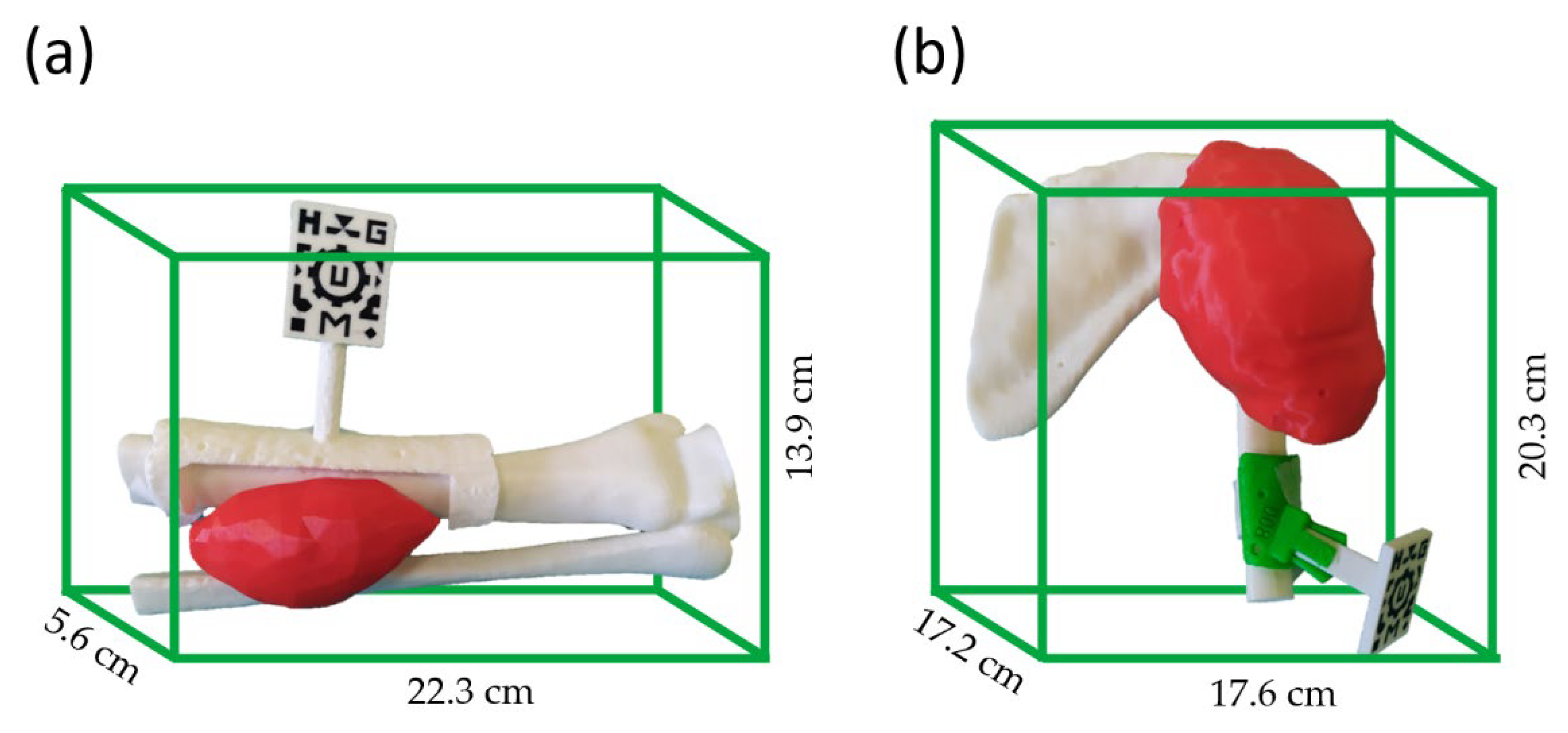
2.2. Phantom Development
2.3. Augmented Reality Applications
2.4. System Performance
2.4.1. AR Projection Error
2.4.2. Technical Differences and Survey
2.4.3. Surgical Experience
3. Results
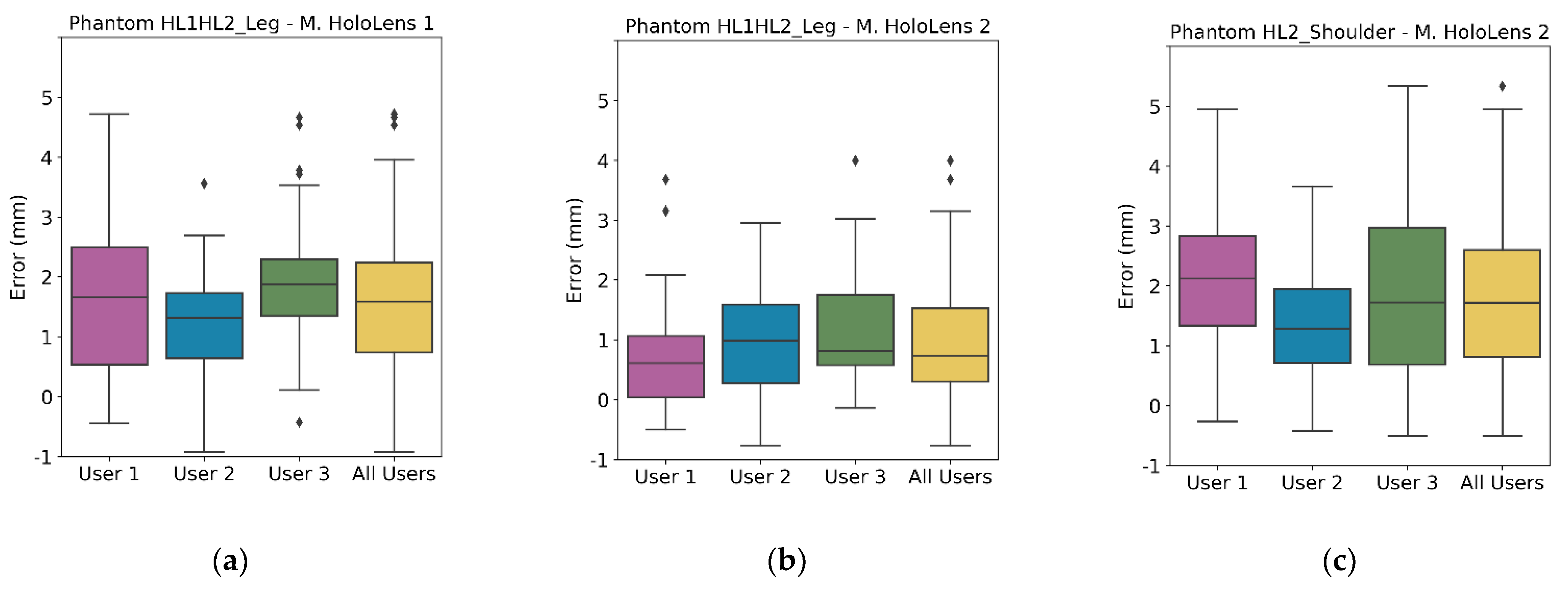
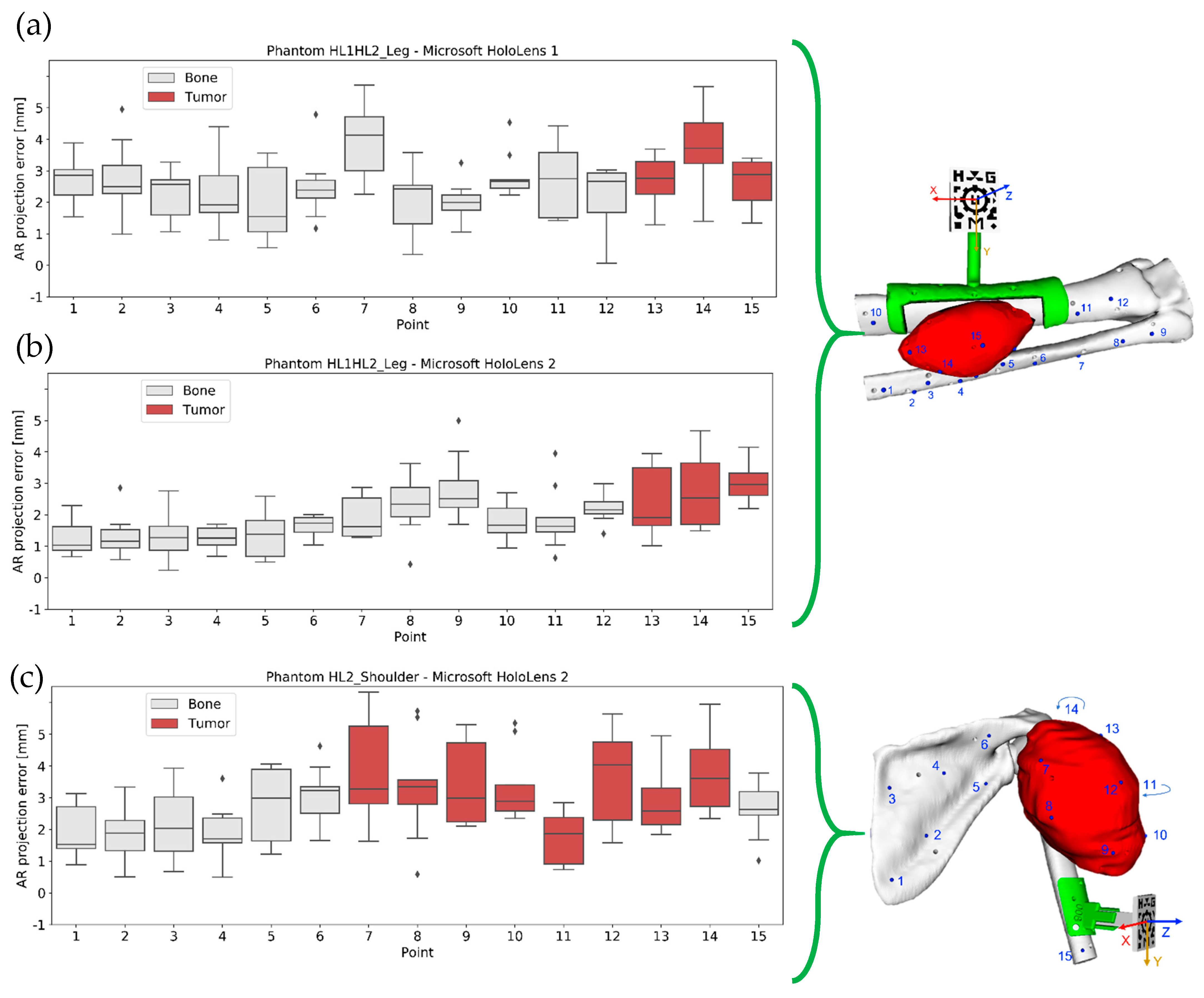
3.1. AR Projection Error
3.2. Technical Differences and Survey
3.3. Surgical Experience
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- El-Hariri, H.; Pandey, P.; Hodgson, A.J.; Garbi, R. Augmented reality visualisation for orthopaedic surgical guidance with pre- and intra-operative multimodal image data fusion. Healthc. Technol. Lett. 2018, 5, 189–193. [Google Scholar] [CrossRef]
- Carl, B.; Bopp, M.; Saß, B.; Pojskic, M.; Nimsky, C. Augmented reality in intradural spinal tumor surgery. Acta Neurochir. 2019, 161, 2181–2193. [Google Scholar] [CrossRef]
- Farshad, M.; Fürnstahl, P.; Spirig, J.M. First in man in-situ augmented reality pedicle screw navigation. N. Am. Spine Soc. J. 2021, 6, 100065. [Google Scholar] [CrossRef]
- Amiras, D.; Hurkxkens, T.J.; Figueroa, D.; Pratt, P.J.; Pitrola, B.; Watura, C.; Rostampour, S.; Shimshon, G.J.; Hamady, M. Augmented reality simulator for CT-guided interventions. Eur. Radiol. 2021, 31, 8897–8902. [Google Scholar] [CrossRef]
- Baum, Z.M.C.; Lasso, A.; Ryan, S.; Ungi, T.; Rae, E.; Zevin, B.; Levy, R.; Fichtinger, G. Augmented Reality Training Platform for Neurosurgical Burr Hole Localization. J. Med. Robot. Res. 2020, 4, 1942001. [Google Scholar] [CrossRef]
- Uppot, R.N.; Laguna, B.; McCarthy, C.J.; De Novi, G.; Phelps, A.; Siegel, E.; Courtier, J. Implementing virtual and augmented reality tools for radiology education and training, communication, and clinical care. Radiology 2019, 291, 570–580. [Google Scholar] [CrossRef]
- Vávra, P.; Roman, J.; Zonča, P.; Ihnát, P.; Němec, M.; Kumar, J.; Habib, N.; El-Gendi, A. Recent Development of Augmented Reality in Surgery: A Review. J. Healthc. Eng. 2017, 2017, 4574172. [Google Scholar] [CrossRef]
- Pratt, P.; Ives, M.; Lawton, G.; Simmons, J.; Radev, N.; Spyropoulou, L.; Amiras, D. Through the HoloLensTM looking glass: Augmented reality for extremity reconstruction surgery using 3D vascular models with perforating vessels. Eur. Radiol. Exp. 2018, 2, 2. [Google Scholar] [CrossRef]
- Yoon, J.; Chen, R.; Kim, E.; Akinduro, O.; Kerezoudis, P.; Han, P.; Si, P.; Freeman, W.; Diaz, R.; Komotar, R.; et al. Augmented reality for the surgeon: Systematic review. Int. J. Med. Robot. 2018, 14, e1914. [Google Scholar] [CrossRef]
- Pan, Z.; David Cheok, A.; Yang, H.; Zhu, J.; Shi, J. Virtual reality and mixed reality for virtual learning environments. Comput. Graph. 2006, 30, 20–28. [Google Scholar] [CrossRef]
- Gerup, J.; Soerensen, C.B.; Dieckmann, P. Augmented reality and mixed reality for healthcare education beyond surgery: An integrative review. Int. J. Med. Educ. 2020, 11, 1–18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jud, L.; Fotouhi, J.; Andronic, O.; Aichmair, A.; Osgood, G.; Navab, N.; Farshad, M. Applicability of augmented reality in orthopedic surgery—A systematic review. BMC Musculoskelet. Disord. 2020, 21, 103. [Google Scholar] [CrossRef] [PubMed]
- Wayne, M.; Ryan, J.; Stephen, S. Virtual and Augmented Reality Applications in Medicine and Surgery The Fantastic Voyage is here. Anat. Physiol. Curr. Res. 2019, 9, 313. [Google Scholar] [CrossRef]
- Azimi, E.; Qian, L.; Navab, N.; Kazanzides, P. Alignment of the Virtual Scene to the Tracking Space of a Mixed Reality Head-Mounted Display. arXiv 2017, arXiv:1703.05834. [Google Scholar]
- de Oliveira, M.E.; Debarba, H.G.; Lädermann, A.; Chagué, S.; Charbonnier, C. A hand-eye calibration method for augmented reality applied to computer-assisted orthopedic surgery. Int. J. Med. Robot. Comput. Assist. Surg. 2019, 15, e1969. [Google Scholar] [CrossRef]
- Hajek, J.; Unberath, M.; Fotouhi, J.; Bier, B.; Lee, S.C.; Osgood, G.; Maier, A.; Armand, M.; Navab, N. Closing the Calibration Loop: An Inside-Out-Tracking Paradigm for Augmented Reality in Orthopedic Surgery. In Proceedings of the Lecture Notes in Computer Science; Including Subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics; Springer: Cham, Switzerland, 2018; Volume 11073 LNCS, pp. 299–306. [Google Scholar]
- Tang, R.; Ma, L.; Xiang, C.; Wang, W.; Li, A.; Liao, H.; Dong, J. Augmented reality navigation in open surgery for hilar cholangiocarcinoma resection with hemihepatectomy using video-based in situ three-dimensional anatomical modeling: A case report. Medicine 2017, 96, e8083. [Google Scholar] [CrossRef]
- Meulstee, J.W.; Nijsink, J.; Schreurs, R.; Verhamme, L.M.; Xi, T.; Delye, H.H.K.; Borstlap, W.A.; Maal, T.J.J. Toward Holographic-Guided Surgery. Surg. Innov. 2019, 26, 86–94. [Google Scholar] [CrossRef] [Green Version]
- Kuhlemann, I.; Kleemann, M.; Jauer, P.; Schweikard, A.; Ernst, F. Towards X-ray free endovascular interventions—Using HoloLens for on-line holographic visualisation. Healthc. Technol. Lett. 2017, 4, 184–187. [Google Scholar] [CrossRef]
- Frantz, T.; Jansen, B.; Duerinck, J.; Vandemeulebroucke, J. Augmenting microsoft’s HoloLens with vuforia tracking for neuronavigation. Healthc. Technol. Lett. 2018, 5, 221–225. [Google Scholar] [CrossRef]
- Moreta-Martinez, R.; Pose-Díez-de-la-Lastra, A.; Calvo-Haro, J.A.; Mediavilla-Santos, L.; Pérez-Mañanes, R.; Pascau, J. Combining Augmented Reality and 3D Printing to Improve Surgical Workflows in Orthopedic Oncology: Smartphone Application and Clinical Evaluation. Sensors 2021, 21, 1370. [Google Scholar] [CrossRef]
- Solbiati, M.; Passera, K.M.; Rotilio, A.; Oliva, F.; Marre, I.; Goldberg, S.N.; Ierace, T.; Solbiati, L. Augmented reality for interventional oncology: Proof-of-concept study of a novel high-end guidance system platform. Eur. Radiol. Exp. 2018, 2, 18. [Google Scholar] [CrossRef] [PubMed]
- Microsoft About HoloLens 2. Available online: https://docs.microsoft.com/en-us/hololens/hololens2-hardware (accessed on 3 March 2022).
- García-sevilla, M.; Moreta-martinez, R.; García-mato, D.; Pose-diez-de-la-lastra, A.; Pérez-mañanes, R.; Calvo-haro, J.A.; Pascau, J. Augmented reality as a tool to guide psi placement in pelvic tumor resections. Sensors 2021, 21, 7824. [Google Scholar] [CrossRef] [PubMed]
- Moreta-Martinez, R.; García-Mato, D.; García-Sevilla, M.; Pérez-Mañanes, R.; Calvo-Haro, J.; Pascau, J. Augmented reality in computer-assisted interventions based on patient-specific 3D printed reference. Healthc. Technol. Lett. 2018, 5, 162–166. [Google Scholar] [CrossRef] [PubMed]
- United States Pharmacopeia. United States Pharmacopeial Convention; United States Pharmacopeia: Rockville, MD, USA, 2012; Volume 1, Available online: https://www.usp.org/ (accessed on 11 October 2021).
- Zislis, T.; Martin, S.A.; Cerbas, E.; Heath, J.R.; Mansfield, J.L.; Hollinger, J.O. A scanning electron microscopic study of in vitro toxicity of ethylene-oxide-sterilized bone repair materials. J. Oral Implantol. 1989, 15, 41–46. [Google Scholar]
- Sharma, N.; Cao, S.; Msallem, B.; Kunz, C.; Brantner, P.; Honigmann, P.; Thieringer, F.M. Effects of Steam Sterilization on 3D Printed Biocompatible Resin Materials for Surgical Guides-An Accuracy Assessment Study. J. Clin. Med. 2020, 9, 1506. [Google Scholar] [CrossRef]
- Microsoft Choosing a Unity Version and XR Plugin. Available online: https://docs.microsoft.com/en-us/windows/mixed-reality/develop/unity/choosing-unity-version (accessed on 27 May 2022).
- Hübner, P.; Clintworth, K.; Liu, Q.; Weinmann, M.; Wursthorn, S. Evaluation of HoloLens Tracking and Depth Sensing for Indoor Mapping Applications. Sensors 2020, 20, 1021. [Google Scholar] [CrossRef] [Green Version]
- Moreta-Martinez, R.; García-Mato, D.; García-Sevilla, M.; Pérez-Mañanes, R.; Calvo-Haro, J.A.; Pascau, J. Combining augmented reality and 3d printing to display patient models on a smartphone. J. Vis. Exp. 2020, 155, e60618. [Google Scholar] [CrossRef] [Green Version]
- Tokuda, J.; Fischer, G.S.; Papademetris, X.; Yaniv, Z.; Ibanez, L.; Cheng, P.; Liu, H.; Blevins, J.; Arata, J.; Golby, A.J.; et al. OpenIGTLink: An open network protocol for image-guided therapy environment. Int. J. Med. Robot. Comput. Assist. Surg. 2009, 5, 423–434. [Google Scholar] [CrossRef]
- Li, L.; Yang, J.; Chu, Y.; Wu, W.; Xue, J.; Liang, P.; Chen, L. A novel augmented reality navigation system for endoscopic sinus and skull base surgery: A feasibility study. PLoS ONE 2016, 11, e0146996. [Google Scholar] [CrossRef]
- Al-Bishri, A.; Barghash, Z.; Rosenquist, J.; Sunzel, B. Neurosensory disturbance after sagittal split and intraoral vertical ramus osteotomy: As reported in questionnaires and patients’ records. Int. J. Oral Maxillofac. Surg. 2005, 34, 247–251. [Google Scholar] [CrossRef]
- Microsoft HoloLens (1st Gen) Hardware. Available online: https://docs.microsoft.com/en-us/hololens/hololens1-hardware (accessed on 3 March 2022).
- Fitzpatrick, J.M.; West, J.B.; Maurer, C.R. Predicting Error in Rigid-body, Point-based Registration. IEEE Trans. Med. Imaging 1998, 17, 694–702. [Google Scholar] [CrossRef] [PubMed]



| Patient ID | Gender/Age | Diagnosis | Tumor Location | CT Resolution (mm) |
|---|---|---|---|---|
| HL1HL2_Leg | F/17 | Extraosseous Ewing’s sarcoma | Distal leg | 0.98 × 0.98 × 2.50 |
| HL2_Shoulder | M/50 | Undifferentiated pleomorphic sarcoma | Shoulder | 1.22 × 1.22 × 3.00 |
| Device | Patient ID | Root Mean Square Error (RMSE) (mm) | Median/Interquartile Error (IQR) (mm) |
|---|---|---|---|
| Microsoft HoloLens 1 | HL1HL2_Leg | 2.833 | 2.587/1.501 |
| Microsoft HoloLens 2 | 2.165 | 1.734/1.224 | |
| HL2_Shoulder | 3.108 | 2.717/1.787 |
| Phantom ID | Median Distance to Origin/IQR [mm] | Median Distance to Origin in X Axis/IQR [mm] | Median Distance to Origin in Y Axis/IQR [mm] | Median Distance to Origin in Z Axis/IQR [mm] |
|---|---|---|---|---|
| HL1HL2_Leg | 108.011/19.667 | 39.537/42.119 | 96.310/19.872 | 11.901/8.888 |
| HL2_Shoulder | 144.308/84.185 | 47.505/60.248 | 96.944/49.271 | 49.430/102.559 |
| Device | Phantom ID | Median Error/IQR in X-Axis | Median Error/IQR in Y-Axis | Median Error/IQR in Z-Axis |
|---|---|---|---|---|
| Microsoft HoloLens 1 | HL1HL2_Leg | 1.713/1.611 | 1.187/1.345 | 0.587/0.952 |
| Microsoft HoloLens 2 | 1.172/1.054 | 0.574/1.056 | 0.643/0.719 | |
| HL2_Shoulder | 1.343/1.525 | 1.168/1.120 | 1.061/1.522 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pose-Díez-de-la-Lastra, A.; Moreta-Martinez, R.; García-Sevilla, M.; García-Mato, D.; Calvo-Haro, J.A.; Mediavilla-Santos, L.; Pérez-Mañanes, R.; von Haxthausen, F.; Pascau, J. HoloLens 1 vs. HoloLens 2: Improvements in the New Model for Orthopedic Oncological Interventions. Sensors 2022, 22, 4915. https://doi.org/10.3390/s22134915
Pose-Díez-de-la-Lastra A, Moreta-Martinez R, García-Sevilla M, García-Mato D, Calvo-Haro JA, Mediavilla-Santos L, Pérez-Mañanes R, von Haxthausen F, Pascau J. HoloLens 1 vs. HoloLens 2: Improvements in the New Model for Orthopedic Oncological Interventions. Sensors. 2022; 22(13):4915. https://doi.org/10.3390/s22134915
Chicago/Turabian StylePose-Díez-de-la-Lastra, Alicia, Rafael Moreta-Martinez, Mónica García-Sevilla, David García-Mato, José Antonio Calvo-Haro, Lydia Mediavilla-Santos, Rubén Pérez-Mañanes, Felix von Haxthausen, and Javier Pascau. 2022. "HoloLens 1 vs. HoloLens 2: Improvements in the New Model for Orthopedic Oncological Interventions" Sensors 22, no. 13: 4915. https://doi.org/10.3390/s22134915
APA StylePose-Díez-de-la-Lastra, A., Moreta-Martinez, R., García-Sevilla, M., García-Mato, D., Calvo-Haro, J. A., Mediavilla-Santos, L., Pérez-Mañanes, R., von Haxthausen, F., & Pascau, J. (2022). HoloLens 1 vs. HoloLens 2: Improvements in the New Model for Orthopedic Oncological Interventions. Sensors, 22(13), 4915. https://doi.org/10.3390/s22134915












