Development of a New Integrated System for Vital Sign Monitoring in Small Animals
Abstract
:1. Introduction
2. Materials and Methods
2.1. Imaging System—easyPET.3D
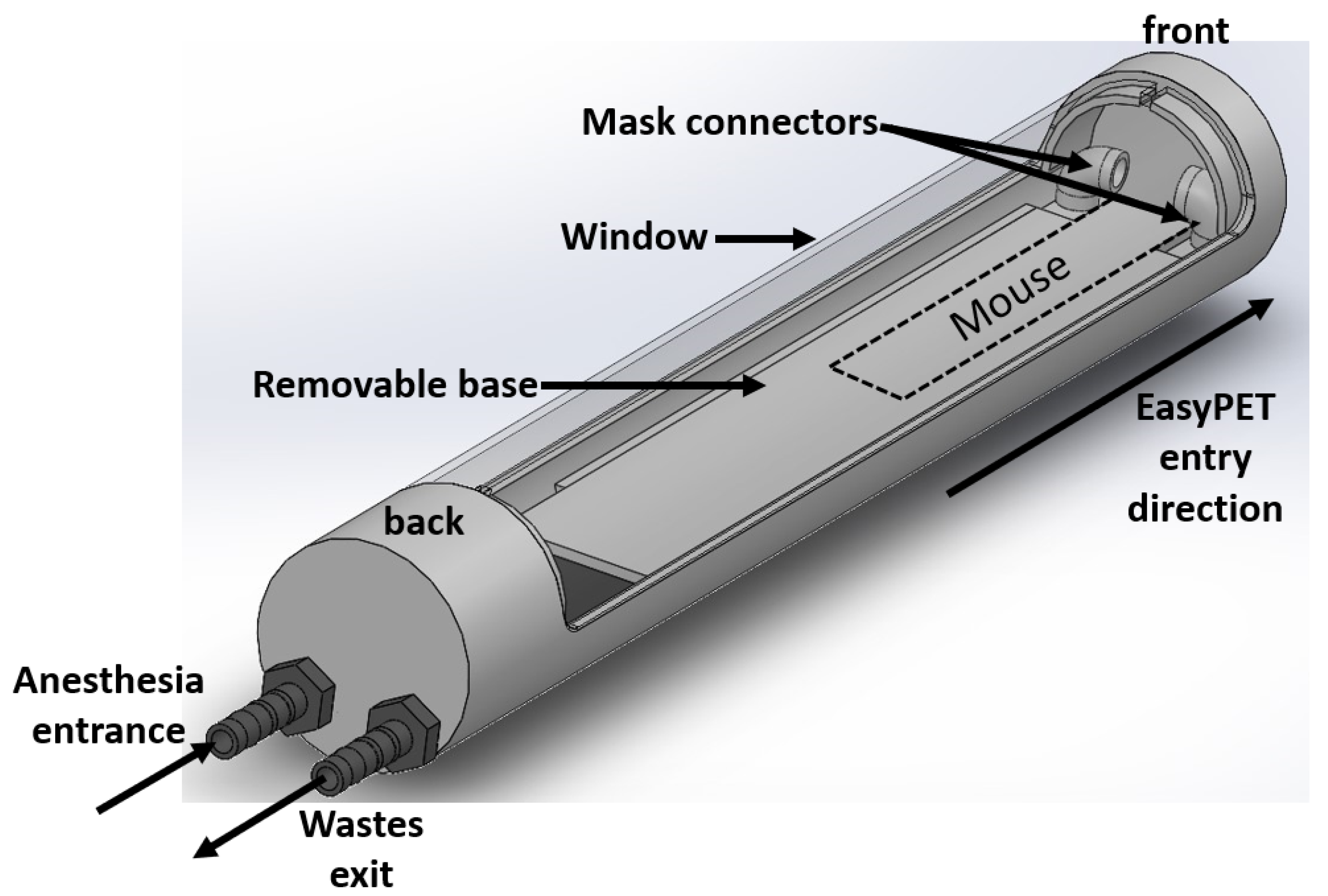
2.2. easyPET.3d Mouse Bed
2.3. Animal Model—Mice
2.4. Anesthesia Protocols
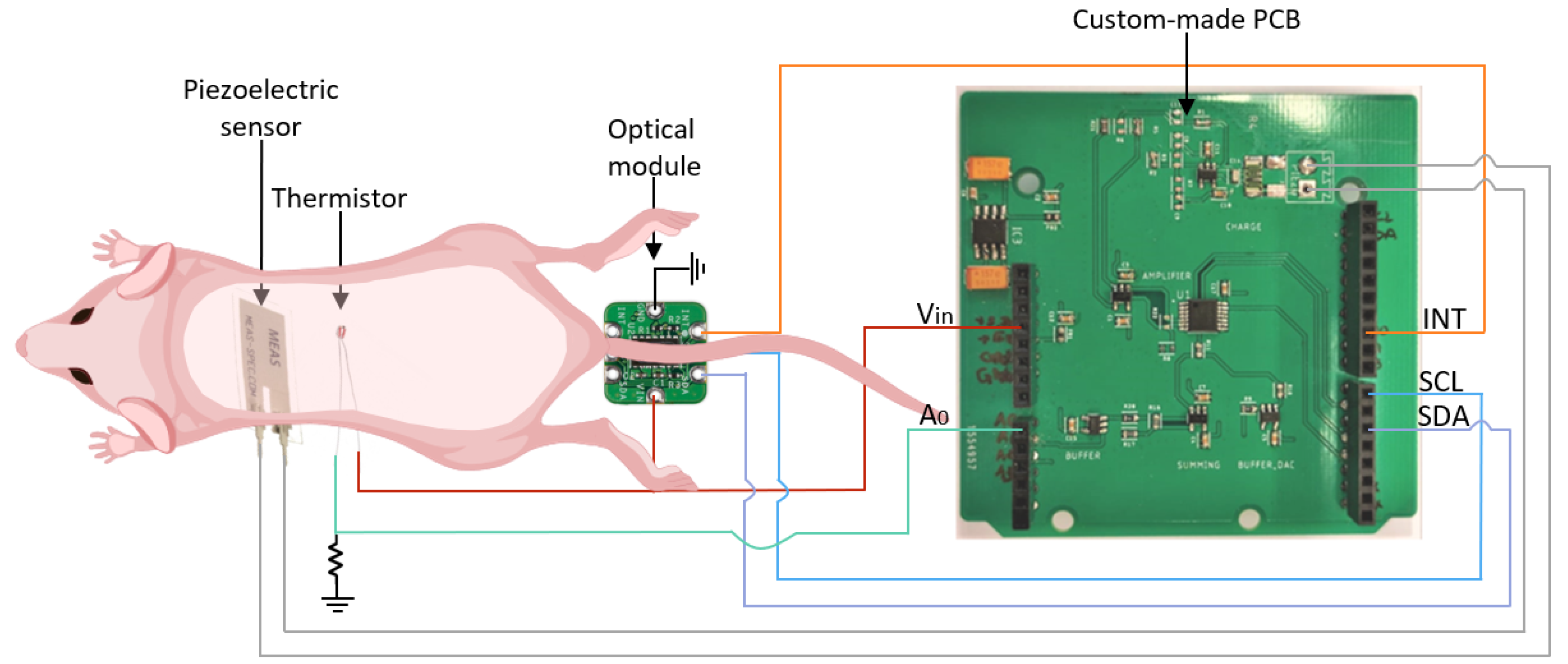
2.5. Vital Sign Measuring Devices
2.5.1. Optical Module
2.5.2. Piezoelectric Sensor
2.5.3. Thermistor
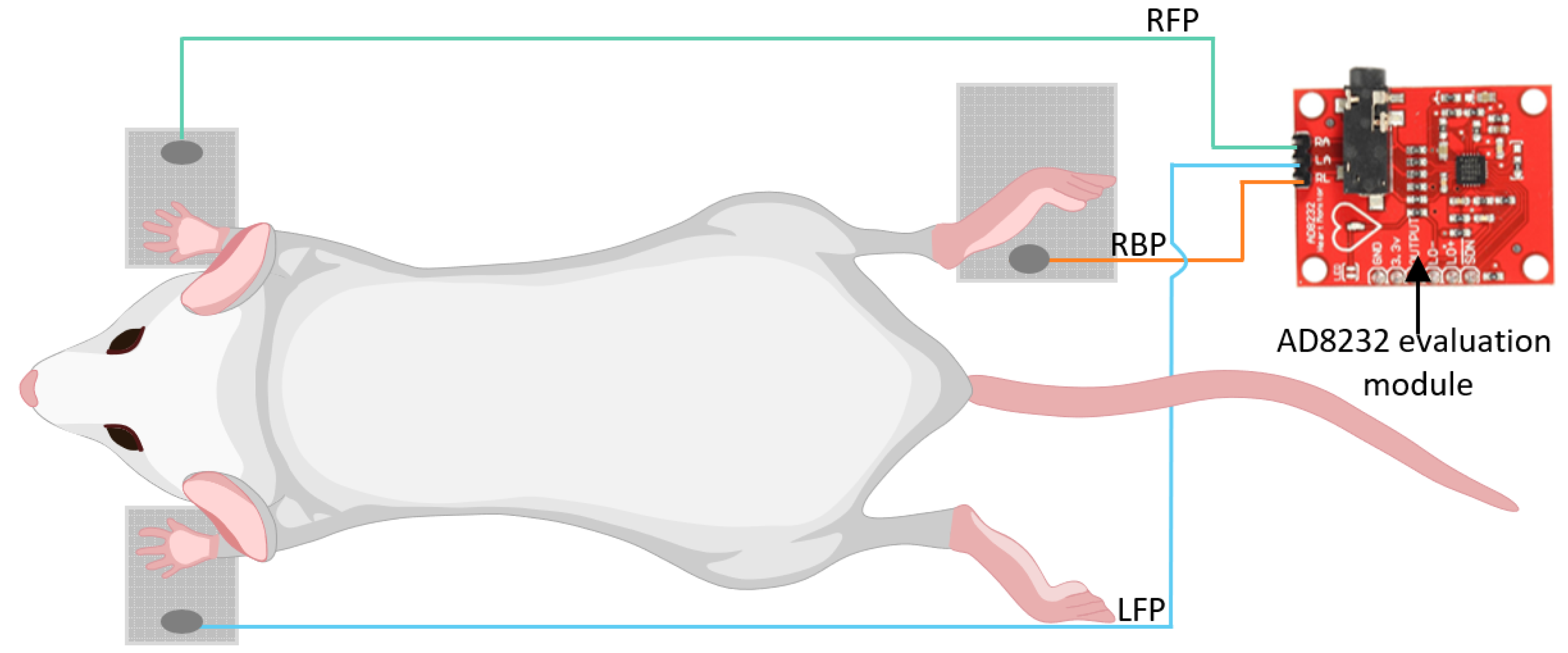
2.5.4. Non-Invasive ECG
2.6. Signal Acquisition
2.6.1. Hardware
- The first stage is a charge amplifier responsible for converting the charge generated from the piezoelectric material into a usable voltage, which can be further amplified in the next stages [17];
- Following the charge amplifier, a gain stage was implemented with an inverting amplifier setup, where the gain value is adjustable by firmware using a digital potentiometer;
- The next stage is necessary to control the offset voltage of the signal by summing an external voltage defined by software to the signal from the previous stage, ensuring that it is between 0 and 5 V, which is the microcontroller ADC’s (Analog to Digital Converter) input range;
- Before entering the assigned ADC (A1), the signal passes through a buffer stage, which truncates the signal between 0 and 5 V, to be readable by this port.
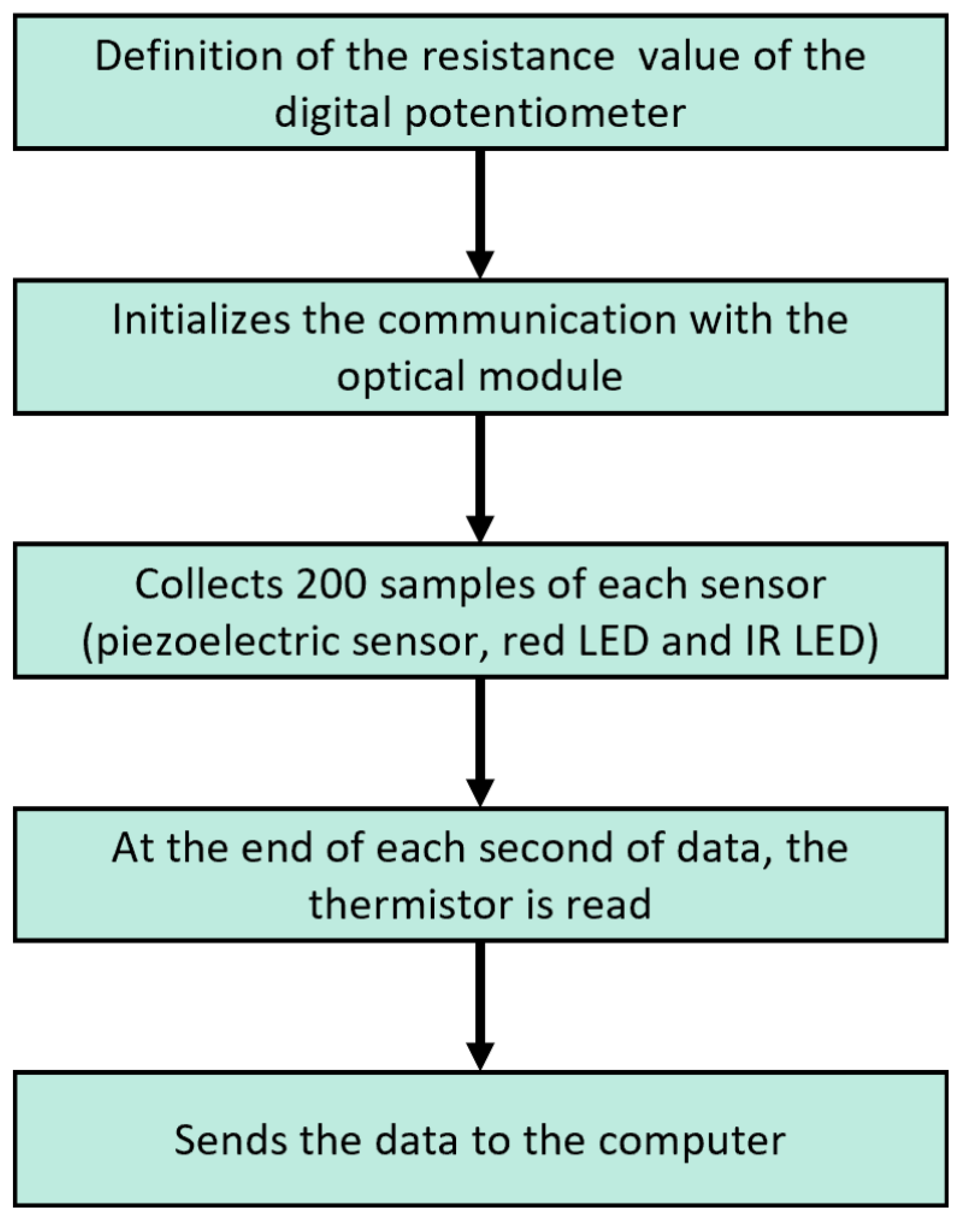
2.6.2. Software
3. Analysis and Discussion of Results
3.1. Respiratory Rate
3.2. Heart Rate
3.3.
3.4. Body Temperature
3.5. Non-Invasive ECG
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tremoleda, J.L.; Kerton, A.; Gsell, W. Anaesthesia and physiological monitoring during in vivo imaging of laboratory rodents: Considerations on experimental outcomes and animal welfare. Lab. Mouse 2012, 2, 1–23. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lister, D.; Leopold, W.R.; McConville, P. Imaging the Laboratory Mouse in vivo. In The Laboratory Mouse; Hedrich, H., Ed.; Academic Press: Cambridge, MA, USA, 2012; pp. 761–780. [Google Scholar]
- Tsukamoto, A.; Serizawa, K.; Sato, R.; Yamazaki, J. Vital signs monitoring during injectable and inhalant anesthesia in mice. Exp. Anim. 2015, 64, 57–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Flecknell, P. Basic Principles of Anaesthesia. In Laboratory Animal Anaesthesia; Flecknell, P., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 77–108. [Google Scholar]
- Flecknell, P. Managing and Monitoring Anaesthesia. In Laboratory Animal Anaesthesia; Flecknell, P., Ed.; Academic Press: Cambridge, MA, USA, 2016; pp. 1–75. [Google Scholar]
- Pereira, C.; Kunczik, J.; Zieglowski, L.; Tolba, R.; Abdelrahman, A.; Zechner, D.; Vollmar, B.; Janssen, H.; Thum, T.; Czaplik, M. Remote Welfare Monitoring of Rodents Using Thermal Imaging. Sensors 2018, 18, 3653. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meleppat, R.; Zhang, P.; Ju, M.; Manna, S.; Jian, Y.; Pugh, E.; Zawadzki, R. Directional optical coherence tomography reveals melanin concentration-dependent scattering properties of retinal pigment epithelium. J. Biomed. Opt. 2019, 24, 066011. [Google Scholar] [CrossRef] [PubMed]
- Müller, J.; Schürer, M.; Neubert, C.; Tillner, F.; Beyreuther, E.; Suckert, T.; Peters, N.; von Neubeck, C.; Lühr, A.; Krause, M.; et al. Multi-modality bedding platform for combined imaging and irradiation of mice. Biomed. Phys. Eng. Express 2020, 6, 037003. [Google Scholar] [CrossRef] [PubMed]
- Arosio, V.; Caccia, M.; Castro, I.F.; Correia, P.M.M.; Mattone, C.; Moutinho, L.M.; Santoro, R.; Silva, A.L.M.; Veloso, J.F.C.A. easyPET: A novel concept for an affordable tomographic system. Nucl. Instruments Methods Phys. Res. Sect. A Accel. Spectrometers Detect. Assoc. Equip. 2016, 845, 644–647. [Google Scholar] [CrossRef]
- Gargiulo, S.; Greco, A.; Gramanzini, M.; Esposito, S.; Affuso, A.; Brunetti, A.; Vesce, G. Mice Anesthesia, Analgesia, and Care, Part I: Anesthetic Considerations in Preclinical Research. ILAR J. 2012, 53, E55–E69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maxim Integrated. Maxrefdes117#: Heart-Rate and Pulse-Oximetry Monitor. Available online: https://www.maximintegrated.com/en/design/reference-design-center/system-board/6300.html (accessed on 7 October 2021).
- Maxim Integrated. MAX30102—High-Sensitivity Pulse Oximeter and Heart-Rate Sensor for Wearable Health. Available online: https://www.maximintegrated.com/en/products/sensors/MAX30102.html (accessed on 7 October 2021).
- Measurement Specialties Inc. LDT with Crimps Vibration Sensor-Switch. Available online: https://cdn.sparkfun.com/datasheets/Sensors/ForceFlex/LDT_Series.pdf (accessed on 8 October 2021).
- Filament2print. Thermistor NTC 100k. Available online: https://filament2print.com/pt/reposicao-extras/926-termistor-ntc-100k.html (accessed on 3 October 2021).
- Analog Devices. AD8232 Single-Lead ECG. Available online: www.analog.com/AD8232 (accessed on 9 October 2021).
- Wang, T.-W.W.; Lin, S.-F. Non-contact Capacitive Sensing for ECG Recording in small Animals. Meas. Sci. Technol. 2020, 31, 12. [Google Scholar] [CrossRef]
- Keim, R. Understanding and Implementing Charge Amplifiers for Piezoelectric Sensors Systems. All About Circuits. Available online: https://www.allaboutcircuits.com/technical-articles/understanding-and-implementing-charge-amplifiers-for-piezoelectric-sensor-s/ (accessed on 30 August 2021).
- University of British Columbia. UBC ACC Guideline on Imaging of Rodents. Available online: https://animalcare.ubc.ca/sites/default/files/documents/Guideline%20-%20Rodent%20Imaging%20%282014%29.pdf (accessed on 12 August 2021).
- Python Software Foundation. Python. Available online: https://www.python.org/ (accessed on 8 October 2021).
- Scipy. scipy.signal.find_peaks. Available online: https://docs.scipy.org/doc/scipy/reference/generated/scipy.signal.find_peaks.html (accessed on 8 October 2021).
- Cesarovic, N.; Nicholls, F.; Rettich, A.; Kronen, P.; Hassig, M.; Jirkof, P.; Arras, M. Isoflurane and sevoflurane provide equally effective anaesthesia in laboratory mice. Lab. Anim. 2010, 44, 329–336. [Google Scholar] [CrossRef] [PubMed]
- Navarro, K.L.; Huss, M.; Smith, J.C.; Sharp, P.; Marx, J.O.; Pacharinsak, C. Mouse Anesthesia: The Art and Science. ILAR J. 2021, 1084–2020. [Google Scholar] [CrossRef] [PubMed]
- Mahling, M.; Fuchs, K.; Thaiss, W.M.; Maier, F.C.; Feger, M.; Bukala, D.; Harant, M.; Eichner, M.; Reutershan, J.; Lang, F.; et al. A comparative pO2 probe and [18F]-fluoro-azomycinarabino-furanoside ([18F]FAZA) PET study reveals anesthesia-induced impairment of oxygenation and perfusion in tumor and muscle. PLoS ONE 2015, 10, e0124665. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seeley, R.R.; Stephens, T.D.; Tate, P. Respiratory System. In Anatomy & Physiology, 6th ed.; McGraw-Hill Higher Education: New York, NY, USA, 2003; pp. 813–858. [Google Scholar]




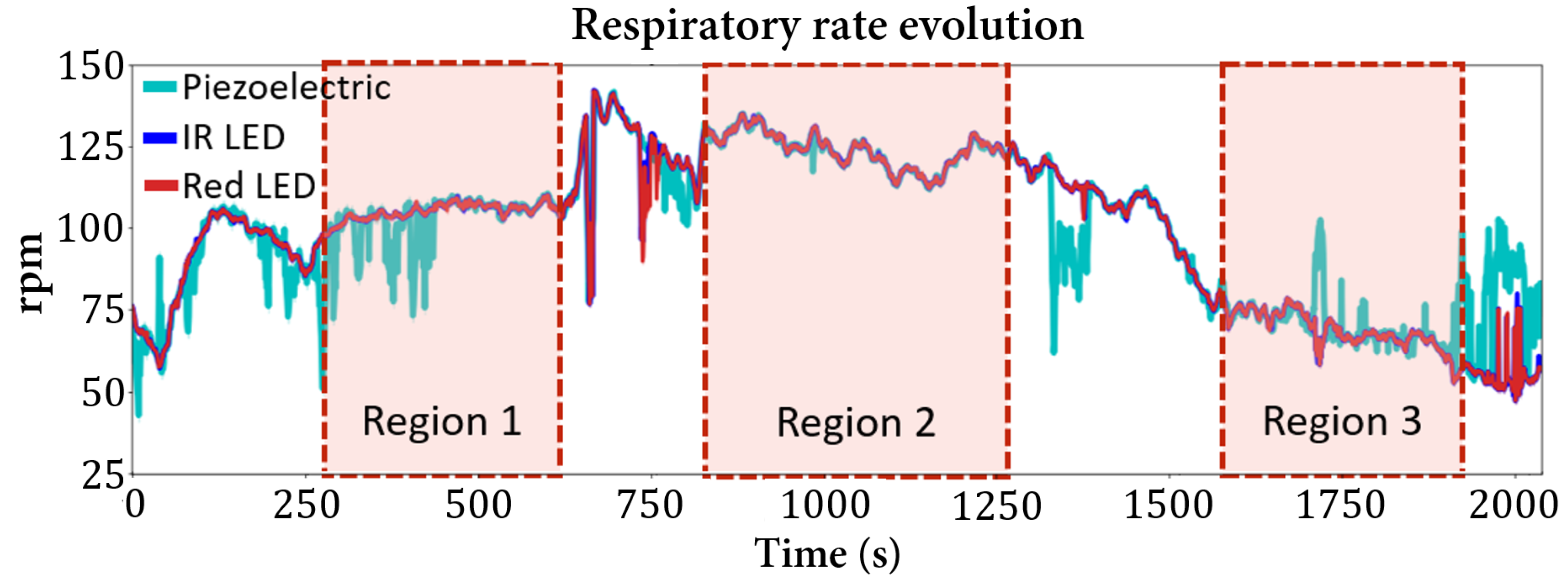
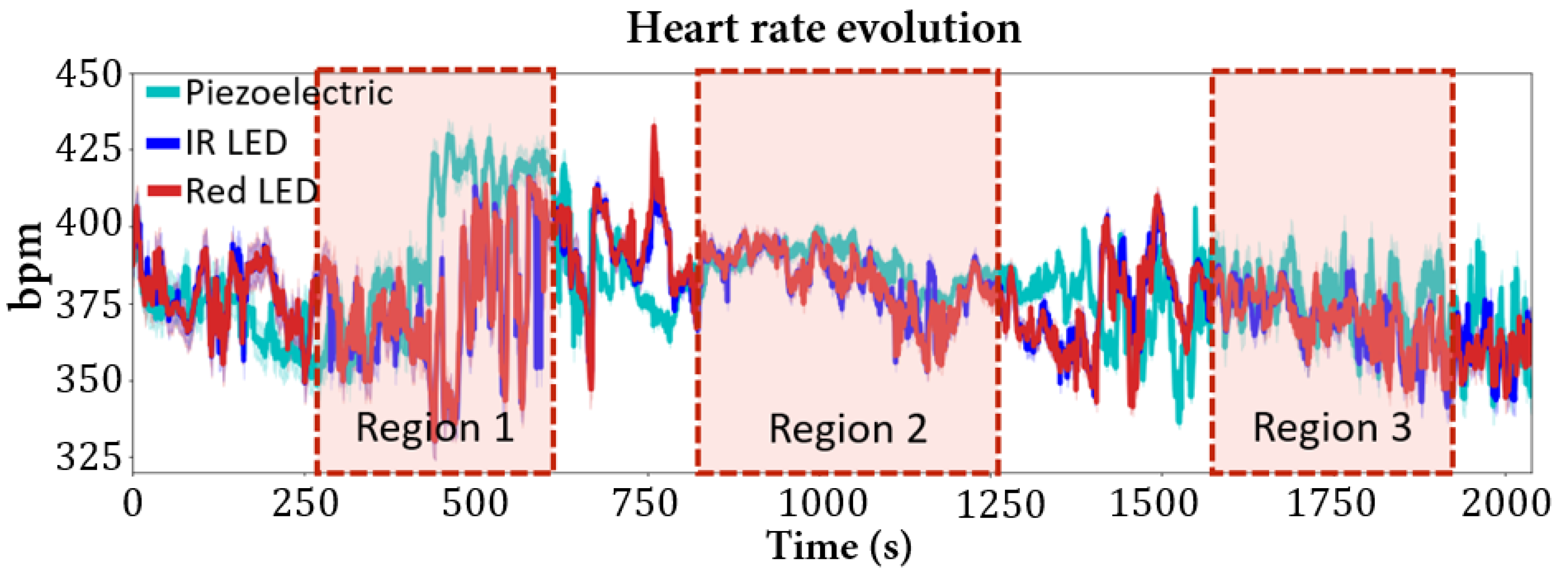
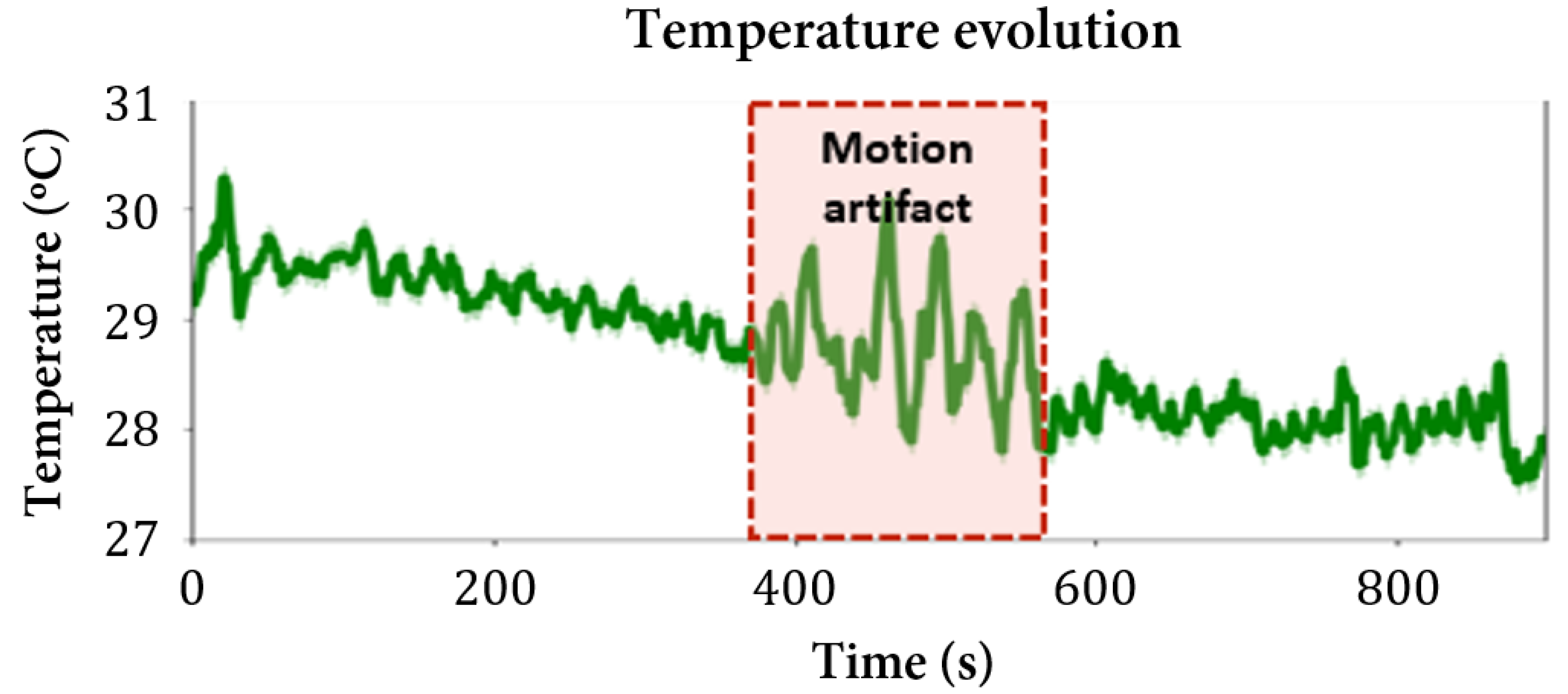
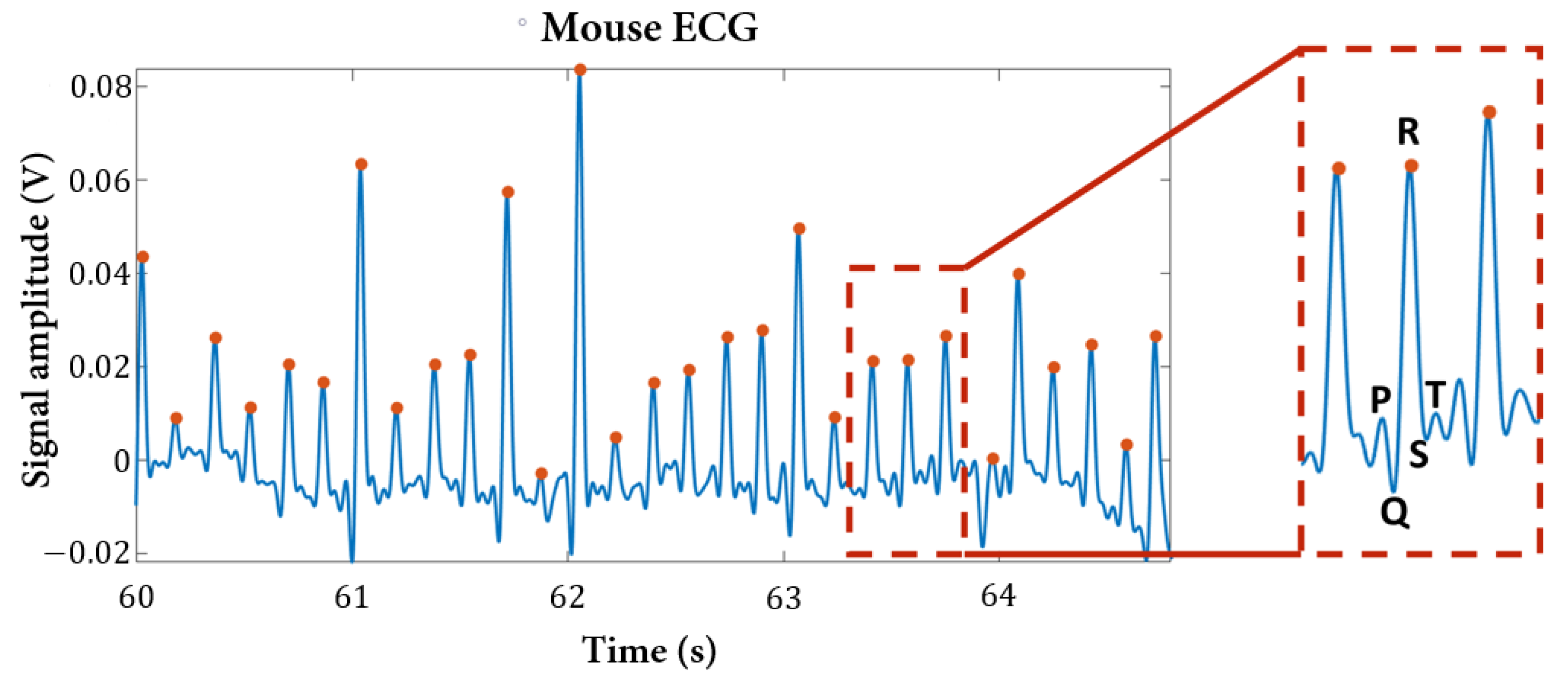
| Mouse | Sevoflurane (%) | (rpm) |
|---|---|---|
| 1 | 1.5 | P: 106 ± 2 IR: 105 ± 1 Red: 105 ± 1 |
| 2 | P: 124 ± 1 IR: 124 ± 1 Red: 124 ± 1 | |
| 4 | P: 70 ± 1 IR: 71 ± 1 Red: 71 ± 1 | |
| 2 | 2.5 | P: 84 ± 1 IR: 84 ± 1 Red: 84 ± 1 |
| 5 | P: 39 ± 1 IR: 39 ± 1 Red: 39 ± 1 |
| Mouse | Sevoflurane (%) | (bpm) |
|---|---|---|
| 1 | 1.5 | P: 387 ± 2 IR: 388 ± 3 Red: 388 ± 3 |
| 2 | P: 402 ± 6 IR: 392 ± 6 Red: 393 ± 7 | |
| 4 | P: 374 ± 6 IR: 368 ± 5 Red: 369 ± 5 | |
| 2 | 2.5 | P: 407 ± 5 IR: 398 ± 3 Red: 405 ± 4 |
| 5 | P: 404 ± 3 IR: 401 ± 2 Red: 401 ± 3 |
| Mouse | Sevoflurane (%) | (%) |
|---|---|---|
| 1 | 1.5 | 88.9 ± 0.6 |
| 2 | 87.6 ± 0.6 | |
| 4 | 92.0 ± 0.3 | |
| 2 | 2.5 | 89.5 ± 0.6 |
| 5 | 88.7 ± 0.7 |
| Mouse | (C) |
|---|---|
| 1 | 24.3–30.7 |
| 2 | 27.5–30.3 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oliveira, R.G.; Correia, P.M.M.; Silva, A.L.M.; Encarnação, P.M.C.C.; Ribeiro, F.M.; Castro, I.F.; Veloso, J.F.C.A. Development of a New Integrated System for Vital Sign Monitoring in Small Animals. Sensors 2022, 22, 4264. https://doi.org/10.3390/s22114264
Oliveira RG, Correia PMM, Silva ALM, Encarnação PMCC, Ribeiro FM, Castro IF, Veloso JFCA. Development of a New Integrated System for Vital Sign Monitoring in Small Animals. Sensors. 2022; 22(11):4264. https://doi.org/10.3390/s22114264
Chicago/Turabian StyleOliveira, Regina G., Pedro M. M. Correia, Ana L. M. Silva, Pedro M. C. C. Encarnação, Fabiana M. Ribeiro, Ismael F. Castro, and João F. C. A. Veloso. 2022. "Development of a New Integrated System for Vital Sign Monitoring in Small Animals" Sensors 22, no. 11: 4264. https://doi.org/10.3390/s22114264
APA StyleOliveira, R. G., Correia, P. M. M., Silva, A. L. M., Encarnação, P. M. C. C., Ribeiro, F. M., Castro, I. F., & Veloso, J. F. C. A. (2022). Development of a New Integrated System for Vital Sign Monitoring in Small Animals. Sensors, 22(11), 4264. https://doi.org/10.3390/s22114264







