A Non-Invasive IR Sensor Technique to Differentiate Parkinson’s Disease from Other Neurological Disorders Using Autonomic Dysfunction as Diagnostic Criterion
Abstract
1. Introduction
Pathophysiology of PD
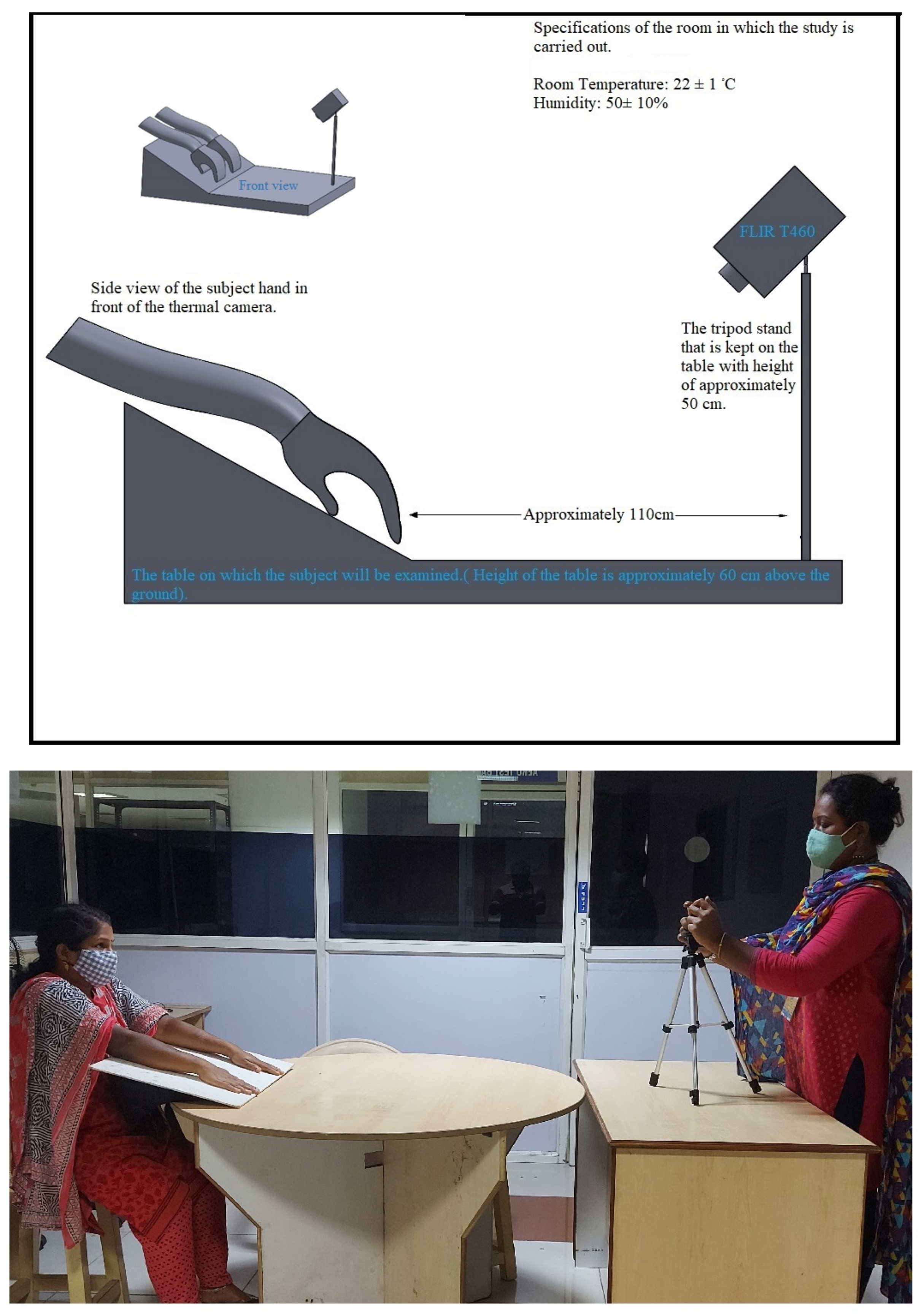
2. Materials and Methods
2.1. Experimental Setup
2.2. Data Collection and Statistical Analysis
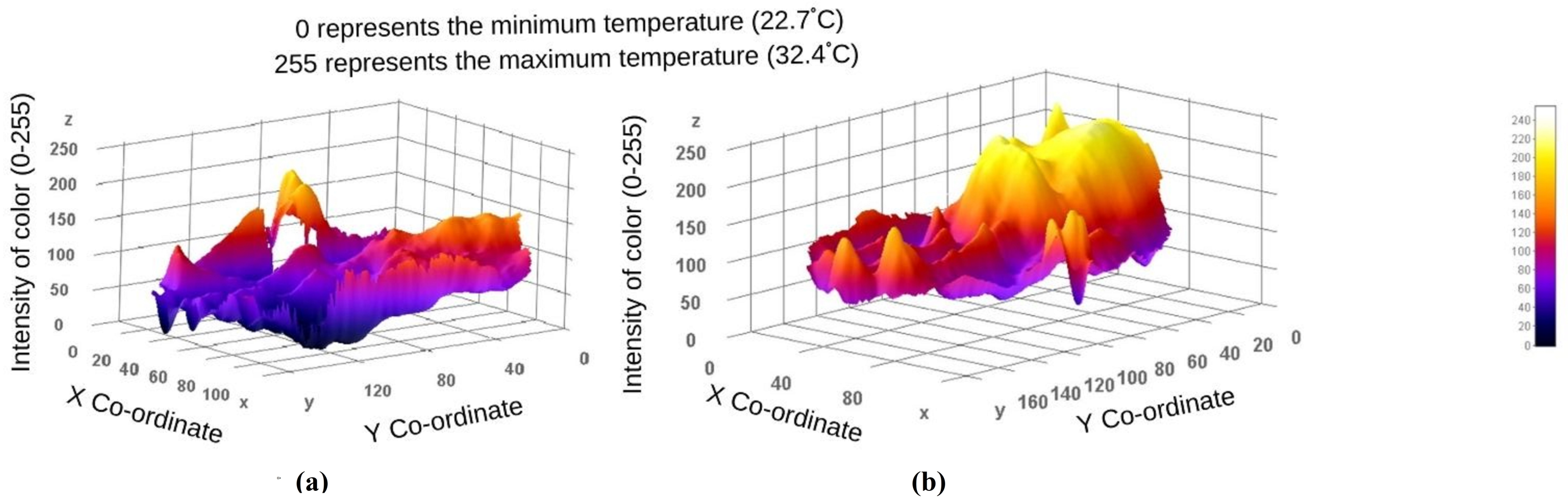
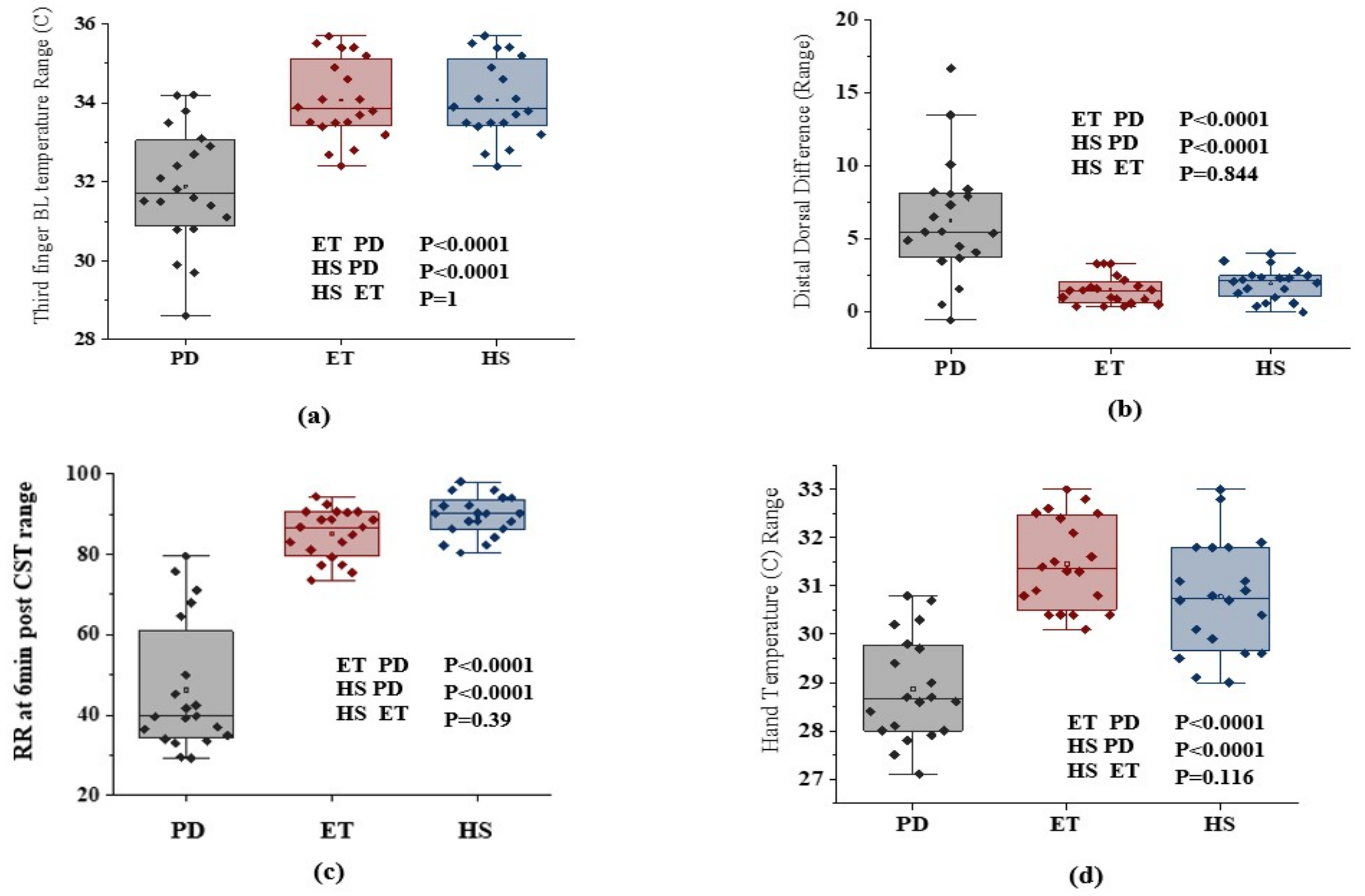
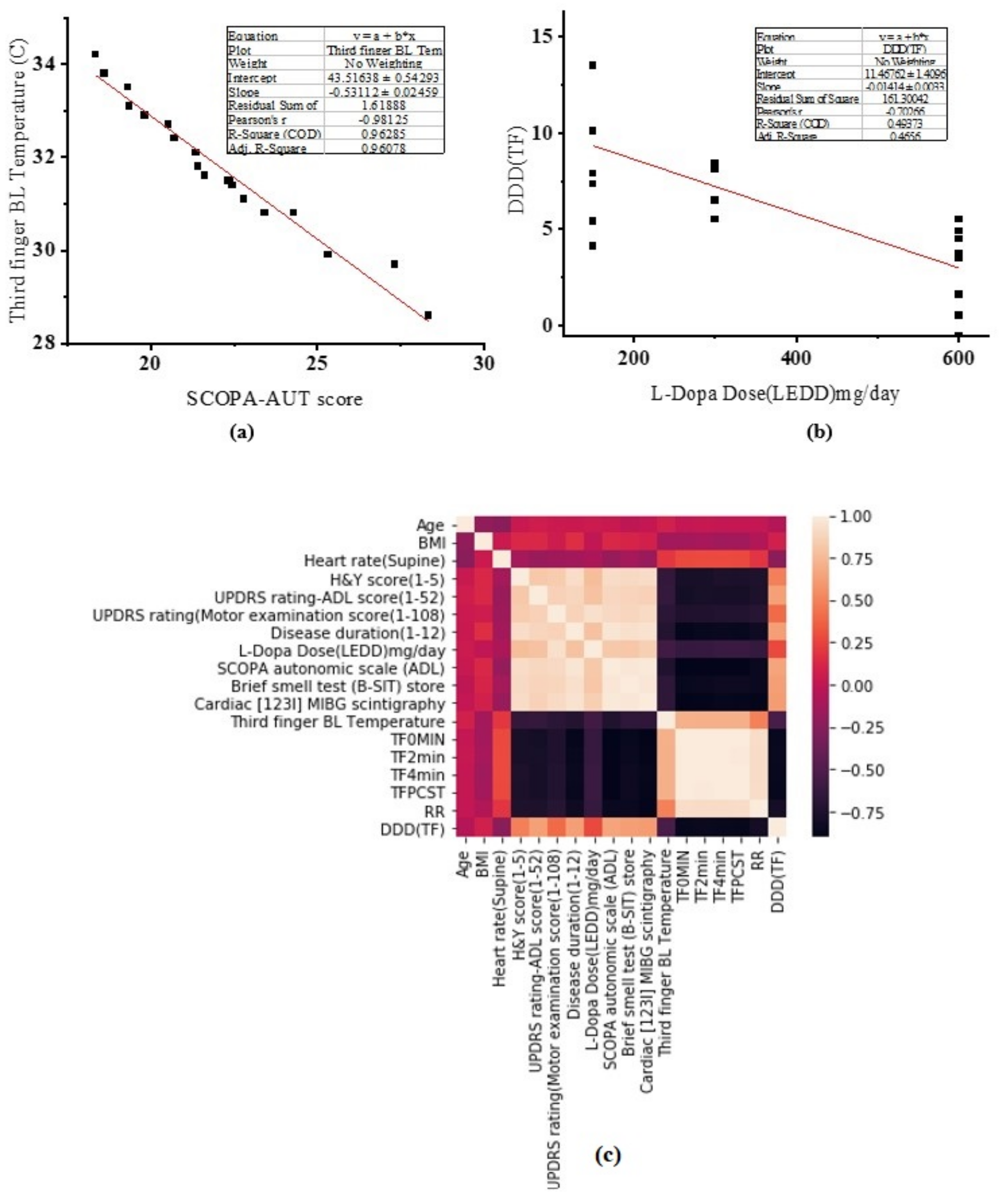
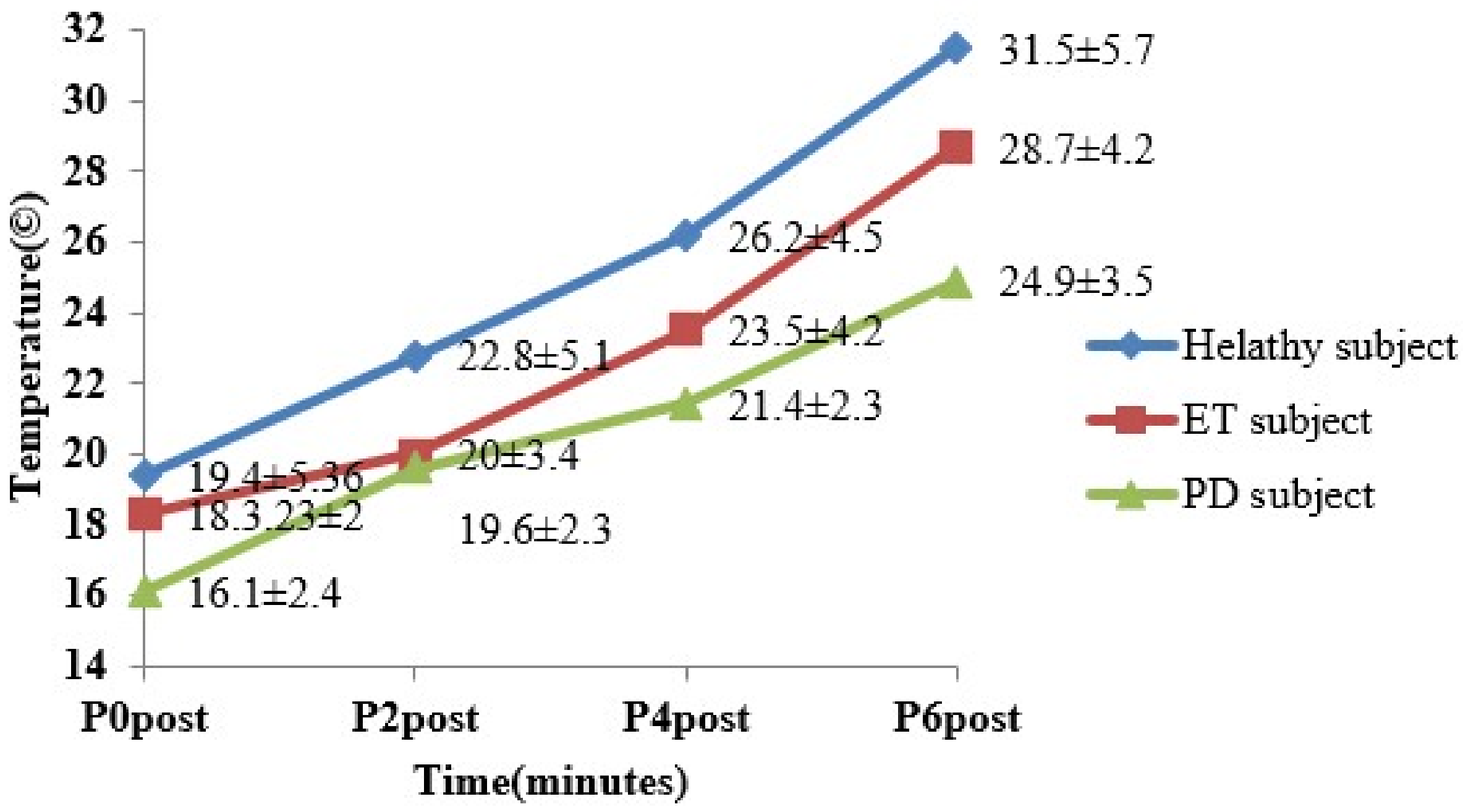
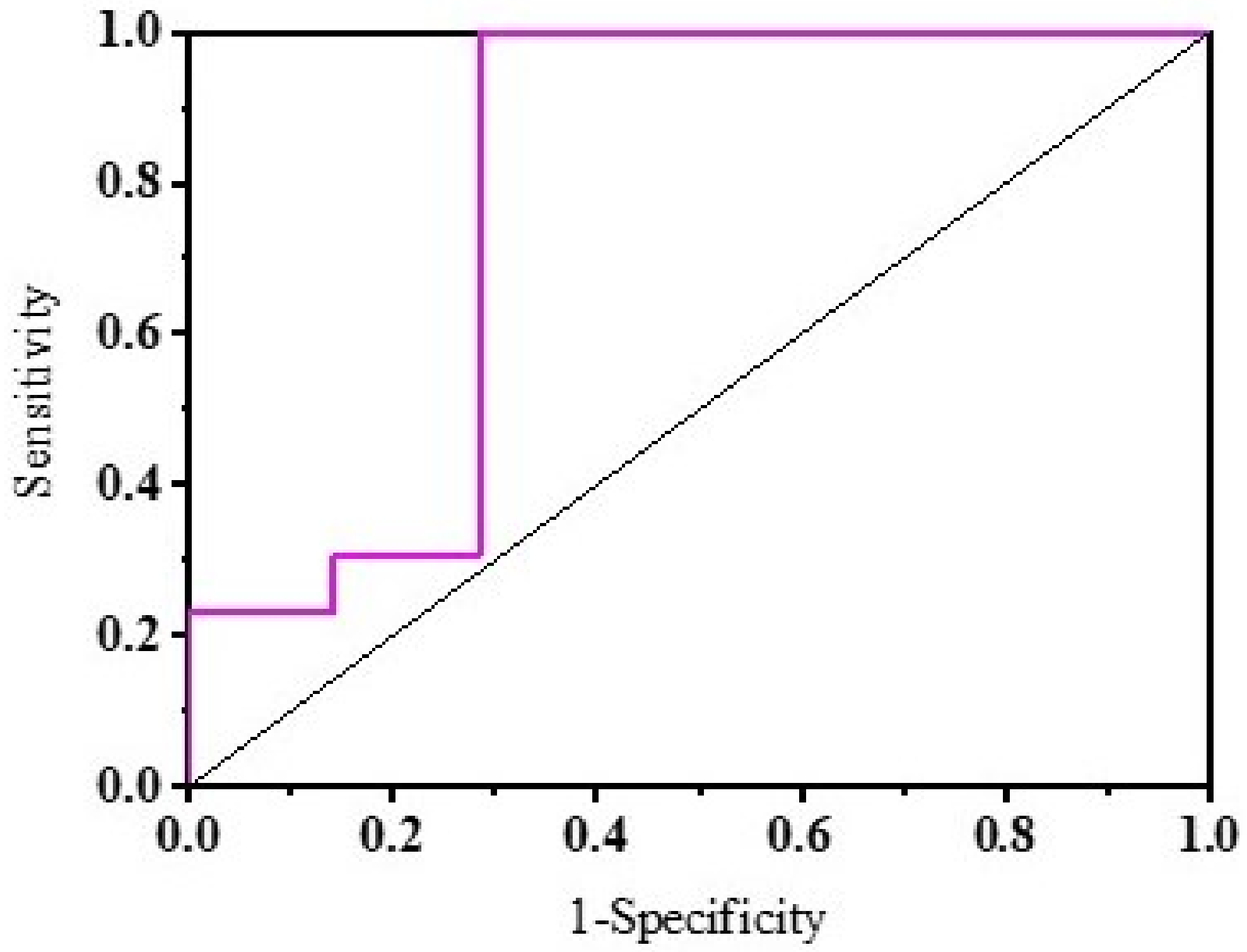
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ball, N.; Teo, W.P.; Chandra, S.; Chapman, J. Parkinson’s disease and the environment. Front. Neurol. 2019, 10, 218. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.L.; Gilmour, H.L.; Ramage-Morin, P.L. Parkinson’s Disease: Prevalence, Diagnosis and Impact; Statistics: Ottawa, ON, Canada, 2014. [Google Scholar]
- Zesiewicz, T.A.; Chari, A.; Jahan, I.; Miller, A.M.; Sullivan, K.L. Overview of essential tremor. Neuropsychiatr. Dis. Treat. 2010, 6, 401. [Google Scholar] [CrossRef]
- Elble, R.J. The essential tremor rating assessment scale. J. Neurol. Neuromed. 2016, 1, 34–38. [Google Scholar]
- Tinelli, M.; Kanavos, P.; Grimaccia, F. The Value of Early Diagnosis and Treatment in Parkinson’s Disease: A Literature Review of the Potential Clinical and Socioeconomic Impact of Targeting Unmet Needs in Parkinson’s Disease; The London School of Economics and Political Science: London, UK, 2016. [Google Scholar]
- Recchia, A.; Rota, D.; Debetto, P.; Peroni, D.; Guidolin, D.; Negro, A.; Skaper, S.D.; Giusti, P. Generation of a α-synuclein-based rat model of Parkinson’s disease. Neurobiol. Dis. 2008, 30, 8–18. [Google Scholar] [CrossRef]
- Lee, V.M.Y.; Trojanowski, J.Q. Mechanisms of Parkinson’s disease linked to pathological α-synuclein: New targets for drug discovery. Neuron 2006, 52, 33–38. [Google Scholar] [CrossRef]
- Tanner, C. Occupational and environmental causes of parkinsonism. Occup. Med. 1992, 7, 503–513. [Google Scholar]
- Chinta, S.J.; Andersen, J.K. Dopaminergic neurons. Int. J. Biochem. Cell Biol. 2005, 37, 942–946. [Google Scholar] [CrossRef] [PubMed]
- Dawson, T.M.; Dawson, V.L. Molecular pathways of neurodegeneration in Parkinson’s disease. Science 2003, 302, 819–822. [Google Scholar] [CrossRef]
- Nolano, M.; Provitera, V.; Estraneo, A.; Selim, M.M.; Caporaso, G.; Stancanelli, A.; Saltalamacchia, A.M.; Lanzillo, B.; Santoro, L. Sensory deficit in Parkinson’s disease: Evidence of a cutaneous denervation. Brain 2008, 131, 1903–1911. [Google Scholar] [CrossRef] [PubMed]
- Volpicelli-Daley, L.A.; Luk, K.C.; Patel, T.P.; Tanik, S.A.; Riddle, D.M.; Stieber, A.; Meaney, D.F.; Trojanowski, J.Q.; Lee, V.M.Y. Exogenous α-synuclein fibrils induce Lewy body pathology leading to synaptic dysfunction and neuron death. Neuron 2011, 72, 57–71. [Google Scholar] [CrossRef]
- Thenganatt, M.A.; Louis, E.D. Distinguishing essential tremor from Parkinson’s disease: Bedside tests and laboratory evaluations. Expert Rev. Neurother. 2012, 12, 687–696. [Google Scholar] [CrossRef]
- Berg, D.; Postuma, R.B.; Adler, C.H.; Bloem, B.R.; Chan, P.; Dubois, B.; Gasser, T.; Goetz, C.G.; Halliday, G.; Joseph, L.; et al. MDS research criteria for prodromal Parkinson’s disease. Mov. Disord. 2015, 30, 1600–1611. [Google Scholar] [CrossRef]
- Habipoglu, Y.; Alpua, M.; Bilkay, C.; Turkel, Y.; Dag, E. Autonomic dysfunction in patients with essential tremor. Neurol. Sci. 2017, 38, 265–269. [Google Scholar] [CrossRef] [PubMed]
- Pauling, J.D.; Flower, V.; Shipley, J.; Harris, N.D.; McHugh, N.J. Influence of the cold challenge on the discriminatory capacity of the digital distal–dorsal difference in the thermographic assessment of Raynaud’s phenomenon. Microvasc. Res. 2011, 82, 364–368. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Wigley, F.M.; Flavahan, N.A. Raynaud’s phenomenon. Rheum. Dis. Clin. N. Am. 1996, 22, 765–781. [Google Scholar] [CrossRef]
- Goldstein, D.S.; Sewell, L.; Sharabi, Y. Autonomic dysfunction in PD: A window to early detection? J. Neurol. Sci. 2011, 310, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Antonio-Rubio, I.; Madrid-Navarro, C.; Salazar-López, E.; Pérez-Navarro, M.; Sáez-Zea, C.; Gómez-Milán, E.; Mínguez-Castellanos, A.; Escamilla-Sevilla, F. Abnormal thermography in Parkinson’s disease. Park. Relat. Disord. 2015, 21, 852–857. [Google Scholar] [CrossRef] [PubMed]
- Ring, E.; Ammer, K. Infrared thermal imaging in medicine. Physiol. Meas. 2012, 33, R33. [Google Scholar] [CrossRef] [PubMed]
- Greenstein, D.; Gupta, N.; Martin, P.; Walker, D.; Kester, R. Impaired thermoregulation in Raynaud’s phenomenon. Angiology 1995, 46, 603–611. [Google Scholar] [CrossRef]
- Akaogi, Y.; Asahina, M.; Yamanaka, Y.; Koyama, Y.; Hattori, T. Sudomotor, skin vasomotor, and cardiovascular reflexes in 3 clinical forms of Lewy body disease. Neurology 2009, 73, 59–65. [Google Scholar] [CrossRef] [PubMed]
- Louis, E.D.; Faust, P.L.; Vonsattel, J.P.G.; Honig, L.S.; Rajput, A.; Robinson, C.A.; Rajput, A.; Pahwa, R.; Lyons, K.E.; Ross, G.W.; et al. Neuropathological changes in essential tremor: 33 cases compared with 21 controls. Brain 2007, 130, 3297–3307. [Google Scholar] [CrossRef] [PubMed]
- Chandran, V.; Pal, P.K. Essential tremor: Beyond the motor features. Park. Relat. Disord. 2012, 18, 407–413. [Google Scholar] [CrossRef]
- Benito-León, J.; Bermejo-Pareja, F.; Louis, E.D. Incidence of essential tremor in three elderly populations of central Spain. Neurology 2005, 64, 1721–1725. [Google Scholar] [CrossRef]
- Benito-Leon, J. Essential tremor: A neurodegenerative disease? Tremor Other Hyperkinetic Movements 2014, 4, 252. [Google Scholar] [CrossRef]
- Lee, S.M.; Kim, M.; Lee, H.M.; Kwon, K.Y.; Koh, S.B. Nonmotor symptoms in essential tremor: Comparison with Parkinson’s disease and normal control. J. Neurol. Sci. 2015, 349, 168–173. [Google Scholar] [CrossRef]
- Damian, A.; Adler, C.H.; Hentz, J.G.; Shill, H.A.; Caviness, J.N.; Sabbagh, M.N.; Evidente, V.G.; Beach, T.G.; Driver-Dunckley, E. Autonomic function, as self-reported on the SCOPA-autonomic questionnaire, is normal in essential tremor but not in Parkinson’s disease. Park. Relat. Disord. 2012, 18, 1089–1093. [Google Scholar] [CrossRef] [PubMed]
- Purup, M.M.; Knudsen, K.; Karlsson, P.; Terkelsen, A.J.; Borghammer, P. Skin Temperature in Parkinson’s Disease Measured by Infrared Thermography. Parkinson Dis. 2020, 2020, 2349469. [Google Scholar] [CrossRef] [PubMed]



| Stages | Symptoms |
|---|---|
| Stage 0 | Anxiety, mood disorders, dysautonomia, REM sleep disorders, sensory dysfunction. |
| Stage 1 | Resting tremor symptoms on one side of the body, mild postural change, difference in walking patterns and difference in facial expressions occur. |
| Stage 2 | Signs and symptoms in stage 1 affect both sides of the body; inability to perform tasks like dressing, bathing, eating, etc.; walking and gait problems. |
| Stage 3 | Loss of balance, slowness of movements, frequent falls common. |
| Stage 4 | Symptoms become severe and limiting. PD patients need assistance to stand, e.g., a walker to move. |
| Stage 5 | Stiff legs, thus difficulty standing or walking. Mostly use walker. |
| PD Subjects (n = 20) | ET Subjects (n = 20) | Healthy Subjects (n = 20) | p Value | |
|---|---|---|---|---|
| Age (years): mean ± SD (range) | 61.6 ± 6.82 | 62.35 ± 6.34 | 60.15 ± 7.28 | p = 0.585 |
| Sex: (M/F) | 13/7 | 12/8 | 12/8 | p = 0.307 |
| BMI (kg/m ): mean ± SD (range) | 25.46 ± 2.78 | 23.81 ± 2.38 | 25.8 ± 2.76 | p = 0.470 |
| Heart rate: mean ± SD (range) | 66.7 ± 4.76 | 69.15 ± 5.86 | 68.15 ± 5.15 | p = 0.343 |
| Arterial hypertension (yes/no) | 3/17 | 5/15 | 8/12 | p < 0.0001 |
| Subjective hyposmia (yes/no) | 9/11 | 6/14 | 2/18 | p < 0.0001 |
| Diabetes mellitus (yes/no) | 9/11 | 10/10 | 11/9 | p = 0.159 |
| Orthostatic hypotension (yes/no) | 6/14 | 4/16 | 4/16 | p = 0.211 |
| B-SIT: mean ± SD (range) | 6.98 ± 1.23 | - | - | - |
| Cardiac (I) MIBG scintigraphy: mean ± SD | 1.29 ± 0.12 | - | - | - |
| SCOPA-AUT score: mean ± SD (range) | 21.91 ± 2.8 | 5.02 ± 11.8 | - | - |
| Disease duration: mean ± SD (range) | 7.7 ± 1.95 | 7.3 ± 1.84 | - | - |
| HY score: median (range) | (1–3) 2 | - | - | - |
| UPDRS-ADL score (1–52): median (range) | (11–42) 21.5 | - | - | - |
| UPDRS-Motor examination score (1–108): median (range) | (11–83) 54 | - | - | - |
| Parkinsonian phenotype tremoric/akinetic-rigid/mixed | 12/3/5 | - | - | - |
| L-Dopa dose (LEDD) mg/day: median (range) | 300 (150–600) | - | - | - |
| TETRAS rating score (0–4): median (range) | - | 2 (1–3) | - | - |
| Propranolol with any other necessary drugs (mg): median (range) | - | 350 (120–600) | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Anbalagan, B.; Karnam Anantha, S.; Arjunan, S.P.; Balasubramanian, V.; Murugesan, M.; R, K. A Non-Invasive IR Sensor Technique to Differentiate Parkinson’s Disease from Other Neurological Disorders Using Autonomic Dysfunction as Diagnostic Criterion. Sensors 2022, 22, 266. https://doi.org/10.3390/s22010266
Anbalagan B, Karnam Anantha S, Arjunan SP, Balasubramanian V, Murugesan M, R K. A Non-Invasive IR Sensor Technique to Differentiate Parkinson’s Disease from Other Neurological Disorders Using Autonomic Dysfunction as Diagnostic Criterion. Sensors. 2022; 22(1):266. https://doi.org/10.3390/s22010266
Chicago/Turabian StyleAnbalagan, Brindha, Sunitha Karnam Anantha, Sridhar P. Arjunan, Venkatraman Balasubramanian, Menaka Murugesan, and Kalpana R. 2022. "A Non-Invasive IR Sensor Technique to Differentiate Parkinson’s Disease from Other Neurological Disorders Using Autonomic Dysfunction as Diagnostic Criterion" Sensors 22, no. 1: 266. https://doi.org/10.3390/s22010266
APA StyleAnbalagan, B., Karnam Anantha, S., Arjunan, S. P., Balasubramanian, V., Murugesan, M., & R, K. (2022). A Non-Invasive IR Sensor Technique to Differentiate Parkinson’s Disease from Other Neurological Disorders Using Autonomic Dysfunction as Diagnostic Criterion. Sensors, 22(1), 266. https://doi.org/10.3390/s22010266







