In this section’s first part we detail the production and characterization of tissue mimicking phantoms, that still provide a gold standard for evaluation and comparison of digital signal processing methods to estimate various tissue properties. These phantoms allow for small systematic errors, a good reproducibility and small variances of mimicked tissue parameters.
We continue on with a discussion of the physics on ultrasonic attenuation governed by absorption and scattering properties of the isonified medium, be it a phantom or human tissue. We then continue with a discussion and the well-founded selection of a highly sophisticated ultrasound simulation environment (
k-Wave, ref. [
13]), which allows to simulate the full-blown, non-linear wave equation for an absorbing and structured medium, and report on results obtained and their numerical validity even for highly absorbing media as typically seen in malignant processes of human tissue.
The subsequent section details the mathematical basis for two estimators and we report on results obtained for a non-structured but scattering and absorbing numerical phantom and show that both estimators are able to provide low variance estimates of tissue attenuation.
2.1. Tissue-Mimicking Phantoms
With tissue-mimicking materials it is possible to produce phantoms which mimic human breast tissue in all acoustic aspects [
14,
15]. Therefore we focussed on finding a connection between a material composition and the resulting acoustic properties, providing the opportunity to later test the estimation algorithms.
The essential ingredients for the production of tissue-mimicking phantoms are water, agar for the solidification and silicon carbide for the scattering effect, which leads to the grainy pattern, the so-called speckle exhibited in ultrasound images obtained by clinical equipment. To prevent the emergence of microbubbles that would lead to the distortion of the measurements of the phantoms’ acoustic properties, it is important to use distilled and degassed water. There are additional components to the mixture, which have different effects on the phantom’s acoustic properties, glycerol for instance increases the speed of sound, aluminium oxide of different grain sizes on the other hand increases the attenuation coefficient [
14].
As we aim to estimate attenuation coefficients, we used the list of composition of the International Electrotechnical Commission (IEC) TMM [
16], see
Table 2, and varied the concentration of aluminium oxide (grain sizes 0.3 and 3 μm) to determine the effect on the acoustic properties of the phantoms.
For measurements an US setup as shown in
Figure 1 is used in transmission mode. After a pulse generator (here: 5072PR, Olympus Corporation, Shinjuku, Tokyo, Japan) excites the US transducer and a needle hydrophone measures the sound pressure level with and without the phantom in place, the attenuation for a given thickness can then be directly determined using the spectral-log-difference technique (SLD), which will be discussed in more detail below [
9]. A short pulse in the time domain is desired as it results in a high spatial resolution in imaging applications. The transducer was driven with a narrow pulse exciting its
MHz center frequency, large relative bandwidth, resulting in a single cycle pressure wave emitted into the volume. According to the SLD method, the attenuation coefficient is calculated from the log-difference between the two spectra,
where
d is the thickness of the phantom and
and
are the magnitude spectra of the phantom data and the reference data. The reference data were acquired with the same settings as were used without the phantom in place.
The setup (
Figure 1) was used to estimate the attenuation coefficient
for different aluminium oxide concentrations in tissue mimicking phantoms.
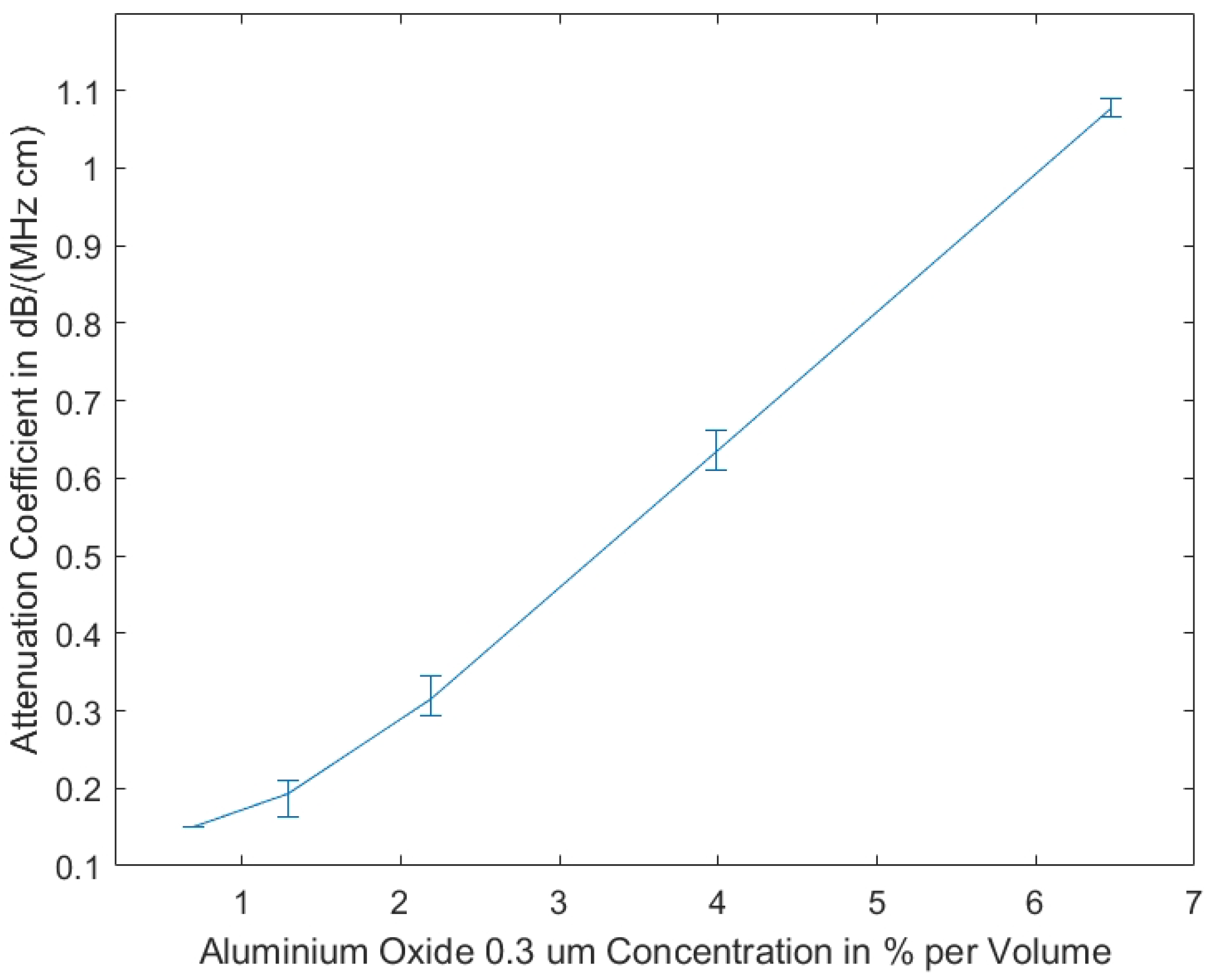
Our studies show that the measurements of the attenuation coefficients of the tissue-mimicking phantoms produced using 0.3 μm aluminium oxide show less variance, due to homogeneity resulting from smaller grain size, as compared to the measurements of the phantoms including aluminium oxide 3 μm. In
Figure 2, the measurement results are displayed, showing that the attenuation coefficient is almost linearly dependent on the volume concentration of aluminium oxide.
2.2. Ultrasound Attenuation
When an ultrasound wave propagates, it generally loses some acoustic energy to random thermal motion resulting in acoustic absorption. Scattering loss, as a second component of attenuation, is caused by the partial interception by the receive aperture of the angular distribution of the backscattered signal of sub-wavelength sized scatterers. Scattering can be classified depending on the relative size of the scattering inhomogenities of the tissue relative to the interrogating wavelength
, with
c, being the speed of sound, and
f, the excitation frequency. Living cells are sub-wavelength scatterers with concentrations of more than 25 per resolution cell ([
4], Chapter 9) and act diffusive. Larger inhomogeneities of length scales on the order of
with concentrations lower than one per resolution cell are independent and distinguishable by their characteristics.
Absorption and scattering combined effects are summarized as attenuation and can be modeled as a sound-absorbing fluid in which the absorption follows a frequency power law of the form
where,
, is the absorption coefficient in coherent units of Np/m,
, is the power law prefactor in Np (rad/s)
m
,
y is the power law exponent,
, is the angular frequency and the resulting attenuation is measured in the coherent units Np (rad/s)
m
.
In medical terms, the attenuation will be referred to the frequency
f in Hz, and
be given in dB/cm as opposed to Np/m, thus further on we will be using
in dB/(MHz
cm), and
in dB/cm. The power law exponent,
y, for human tissue is close to unity, as can be seen from
Table 1 [
4], so the SLD algorithm will assume
, whereas the also discussed method-of-moments (MoM) will consider
y as variable to also be estimated from the B-mode scan lines. In ultrasonic imaging of human tissue, the propagating wave cannot be observed directly but its amplitude needs to be estimated from observations of the backscattered signals as seen at the location of the ultrasound transducer’s receiving aperture. The estimation of the spatially varying attenuation coefficient
is thus an ill-posed inverse problem that needs to be solved.
The backscattering occurs due to the cell-based structure of all biological tissues (except maybe fluid filled cysts that appear to be almost anechoic and thus image dark in B-mode scans). These scattering centers are very small sized as compared to the US wavelength , thus scatter very weakly but are present at a high scatterer number density randomly distributed in space. So the overall action of these scatterers can be modeled in a first order approximation as a spatial white noise process, which, assuming an almost constant speed of sound c, gives an impulse response-like behavior to the imaging process. How strongly valid this simplifying assumption is, needs to be thoroughly analyzed, but this is out of scope of this contribution.
In this paper, we concentrate on the estimation of the attenuation parameter
(in units of dB/cm). US absorption, as characterized by Equation (
2), for soft biological tissues ranges from
dB/(MHz cm) for water up to a maximum
dB/(MHz cm) for certain lesions. Some authors have directed their interest into estimating the backscatter-coefficient, too, in order to then be able to estimate the attenuation coefficient much more accurately [
17].
Testing of algorithms for estimating absorption parameters from acquired B-mode images, an example of which is shown in Figure 5, right, requires a well characterized set of training data that can be obtained by either tissue mimicking phantoms (see
Section 2.1), exhibiting fairly homogeneous properties that can both be directly measured using the transmission mode or the backscatter mode as in imaging US, respectively.
Another less involved way to obtain calibration data for testing
–estimators is using simulation software to solve the US wave equation directly for known properties of the propagation medium, including source terms, absorption and dispersion relations and potentially also the nonlinear pressure-density relation. An open source time-domain simulation software package developed for this purpose is
k-Wave [
8,
13] which will be discussed next.
2.3. Discussion of Simulation Software
There are several open source software packages available that allow the time-domain simulation of propagating acoustic waves including various aspects of an ultrasonic imaging system. A good overview is given in [
18]. We evaluated two software tools, Field II [
19] and
k-Wave [
8,
13] for their suitability to the problem of estimating tissue properties (attenuation coefficient in particular) besides simulating the imaging capabilities of an ultrasonic scanner. Field II being orders of magnitude faster in execution than the alternative and finally chosen
k-Wave software, soon turned out to be unsuitable, since it relies on the spatial impulse response method [
20] that only allows one to simulate linear wave propagation, a restriction we could not accept since the simulation results should, in as many aspects as possible, mimic the real and physical propagation of sound waves in a medium (breast tissue).
The much more powerful k-Wave simulation environment, on the other hand, is able to select all possible parameters of the propagation medium on a voxel by voxel basis and simulates linear and non-linear wave propagation for an arbitrary distribution of heterogeneous material parameters and power law acoustic absorption although at the cost of a dramatic increase in necessary processing power.
If a reasonably sized, structured volume is to be simulated, e.g., mm at a spatial resolution of 30 μm as demanded for an MHz excitation frequency, a data array size of 333 Mvoxels results and needs to be simulated at time increments of a few ns up over a total simulation time, allowing the wave to propagate back and forth along the longest geometrical path in the volume. This results for the example in about 10,000 time increments @ 5 ns to simulate a single B-mode line. This size can only be handled utilizing either a supercomputing environment or employing state of the art graphics processing units (GPU) with several thousand GPU cores programmed to operate concurrently.
For a numerically stable solution, of the non-linear partial differential equations governing the wave propagation at least 10 grid points per acoustic wavelength are generally required. The problem is confounded further by the requirement for small time steps to keep the simulation numerically stable and to minimize unwanted numerical errors to accumulate.
We tested the suitability of an NVIDIA Titan RTX 24 GB GPU (Nvidia, Santa Clara, CA, USA) that is marginally able to simulate such a huge volume array in an acceptable simulation time frame of a few days (for 201 B-mode scan lines).
Obtaining numerical stability and keeping the round-off errors small, an IEEE-854 double precision number format should be selected. GPUs, however, typically are restricted to single-precision arithmetic and, what is not widely known, do not realize the full IEEE-754 single precision format, that would provide 23 bits for the mantissa (=
· 10
in relative resolution resulting in a dynamic range for the sound pressure of approximately 130 dB) but specify for some basic operations just 21 or even only 20 significant bits [
21].
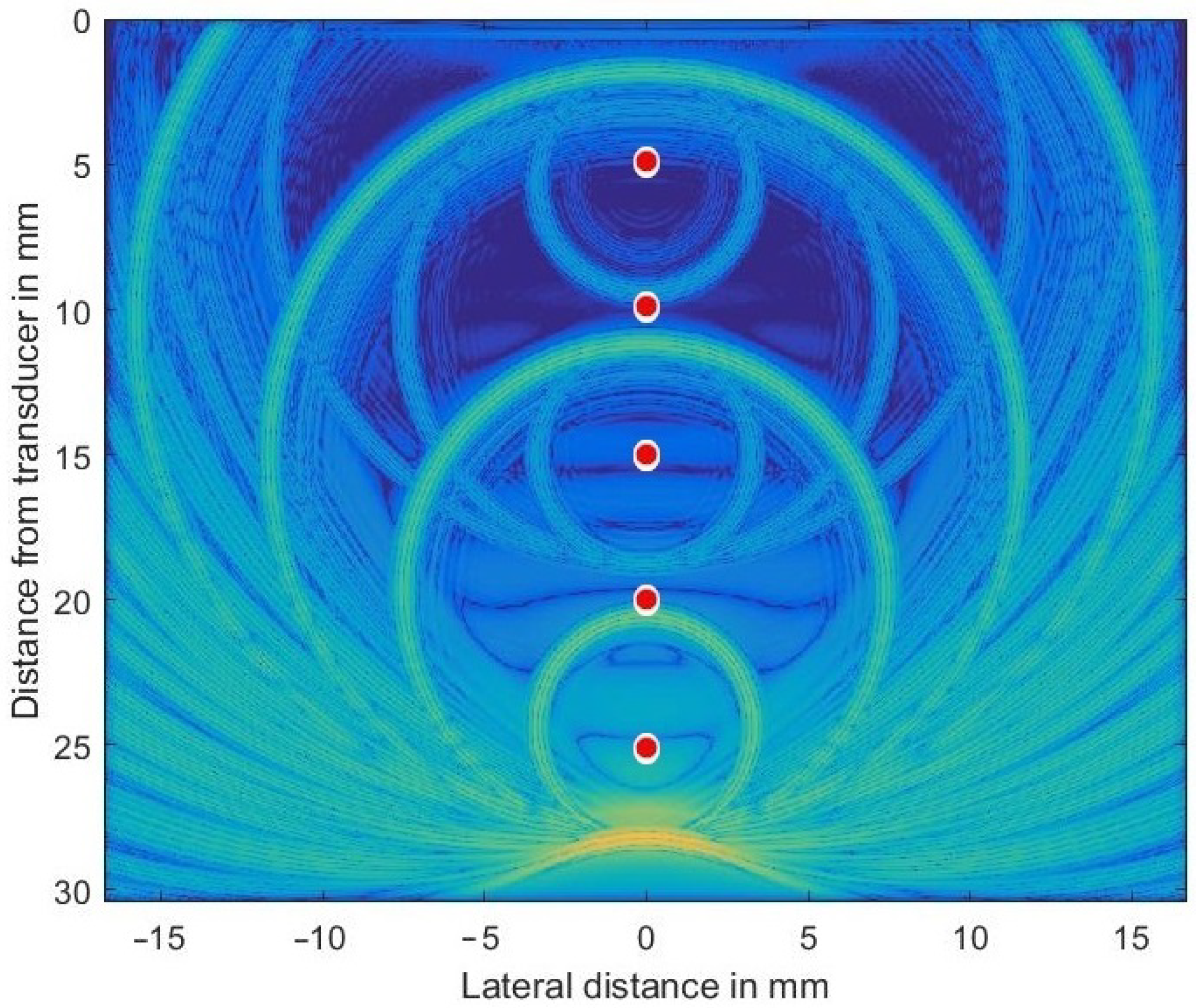
In order to check the validity of the simulation in
k-Wave, a homogeneous and isotropic medium was simulated that included five discretely placed scatterers (at depths of
mm) and
was set at 2.3 dB/(MHz cm) throughout the volume to generate both large and very small sound pressure levels propagating concurrently through the volume. The transducer (center frequency
MHz) was focused both in azimuth and elevation at 35 mm in depth. For such a simple problem an analytic solution can be given and so allows the validity of the numerical results to be checked.
Figure 3 shows the complex envelope of the propagating wave some 17.5 μs after simulation started. The main pulse located at 28 mm depth had excited all 5 scatterers while propagating downwards.
The secondary waves emitted from scatterers located at 10, 15, 20, and 25 mm depths are still clearly visible (the secondary wave from scatterer at 5 mm has already left the simulated volume), as are all re-scattered waves. Very low spatial frequency waves, as seen in the very center, are an indication of numerical problems encountered if the very large dynamic range necessary cannot be met by the GPU arithmetic.
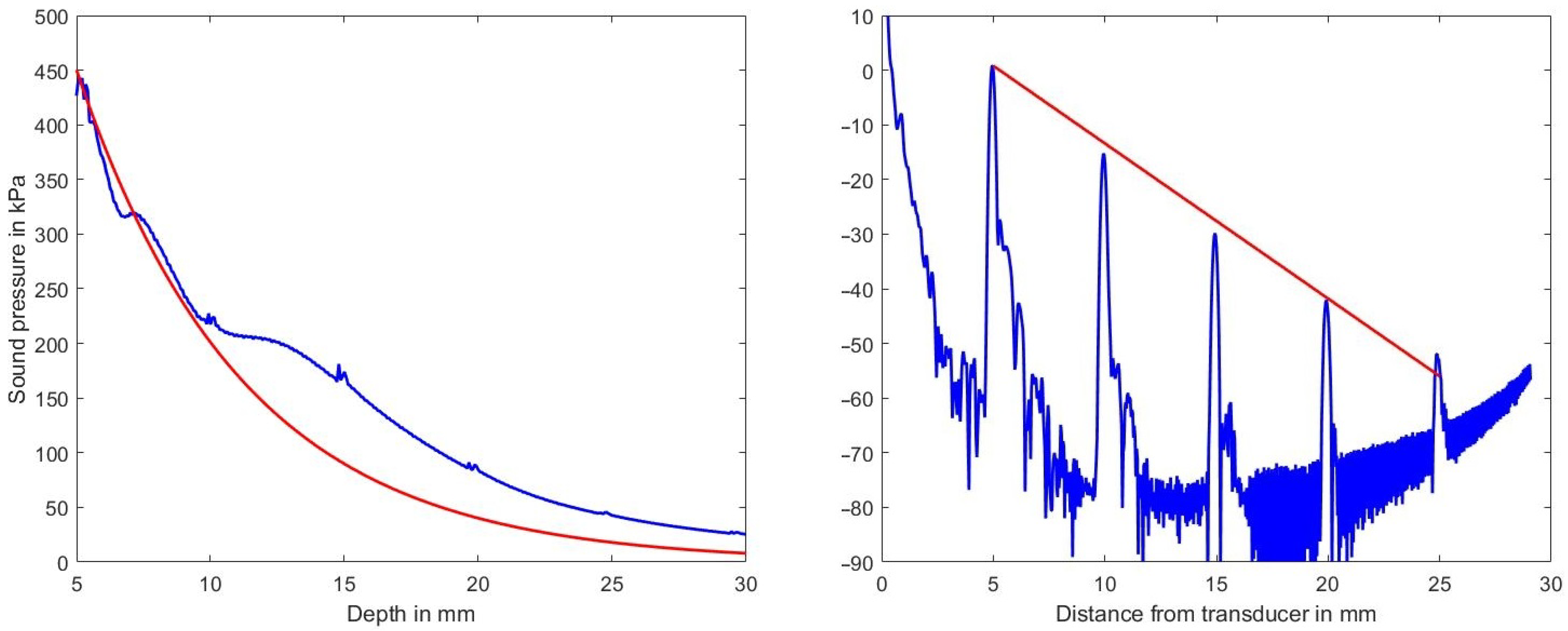
Figure 4, left, shows the sound pressure amplitude vs. depth in blue on a linear scale and the expected decrease in amplitude (according to the selected medium parameters of 2.3 dB/(MHz cm)) in red. The deviation between the two graphs is attributed to the focusing action of the phased-array transducer which is focusing at a depth of 35 mm.
Clearly visible also is the effect of the scatterers at
mm.
Figure 4, right, on the other hand, shows on a logarithmic scale in blue the sound pressure level (in dB ref. 100 kPa) and in red the expected decrease due to the chosen attenuation coefficient as derived from a single B-mode line. Simulation artifacts at low sound pressure values increase the noise floor beyond 17 mm in depth and those artifacts raise the noise floor at greater depths.
Concluding this section, k-Wave run on a GPU gives numerical stable results for attenuation coefficients up to and including 2.5 dB/(MHz cm) for a typical volume size, which is sufficient for simulating most breast tissue types.
2.4. Estimators of Tissue Attenuation
Of all fluid properties, quantities that describe loss are among the most challenging to estimate. In part, this is due to superimposed dissipative mechanisms, as described above, and diffraction effects from sub-wavelength scatterers. Inherent to these phenomena is a specific frequency dependence as given by Equation (
2). According to
Table 1 the power law exponent
y varies with the respective absorption mechanism. Liquid-filled cysts behave similar to like pure water and thus have an exponent close to
, combined with an attenuation of
dB/(MHz cm), whereas solid masses have an exponent closer to
combined with
of 2.50 dB/(MHz cm). In order to improve the diagnostic accuracy, both parameters
y and
are of interest and shall be estimated.
Several authors have elaborated on the estimation of
, or
, respectively, and
y but conclude that it is an ill-posed mathematical problem [
4,
9]. This difficulty arises in part from the demand to spatially map these parameters in order to detect small to very small lesions. The severe tradeoff between estimator’s variance and spatial resolution impacts both methods analyzed in this work.
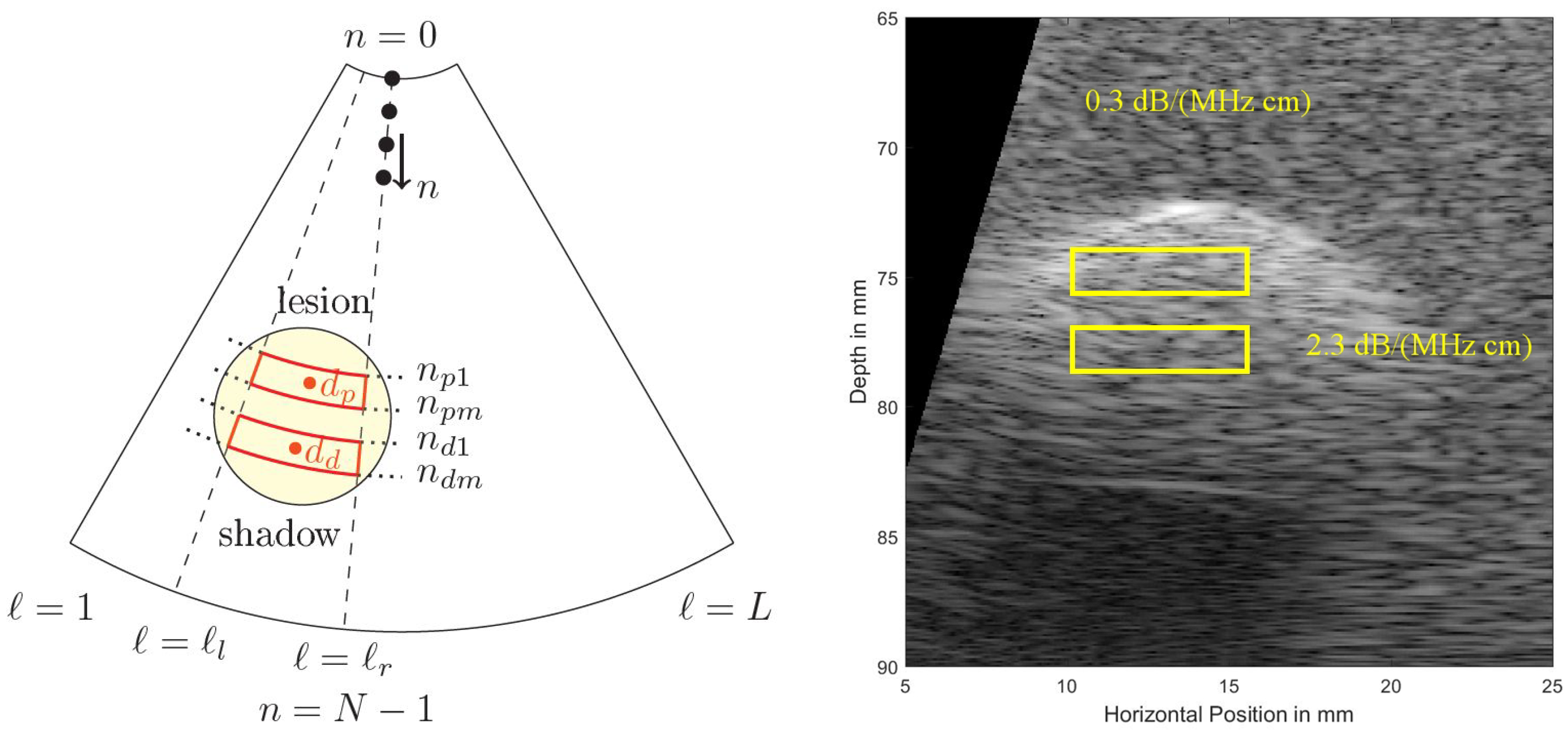
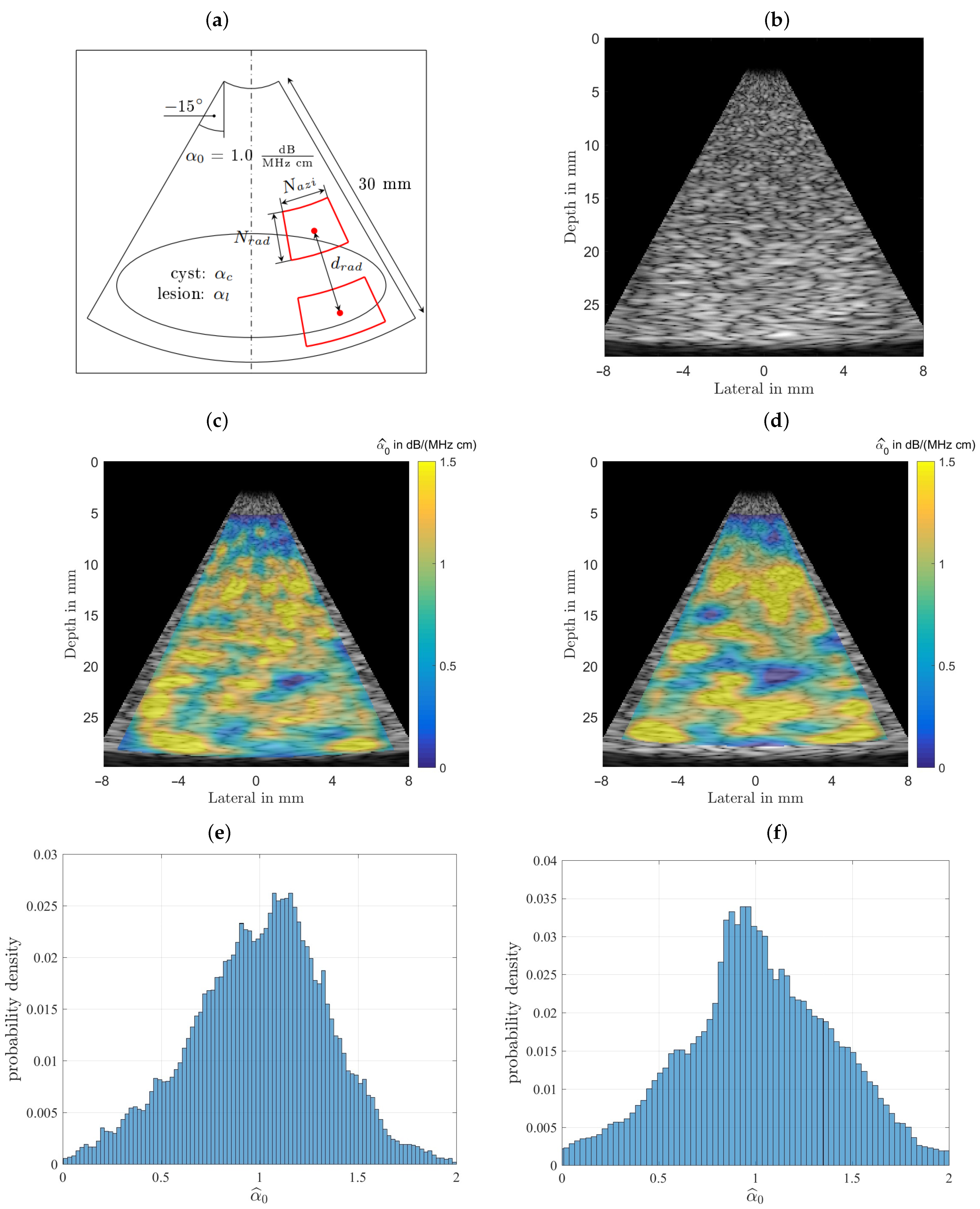
Figure 5 shows a cropped B-mode scan exhibiting the typical US speckles that are detrimental in the estimation process. Furthermore, the distal and proximal data windows over which both estimation algorithms operate are indicated. Note that here a phased array transducer was used with a steering angle of
. The sketch in
Figure 5, left, defines all indices used in both algorithms. Index
is indicating the lateral sector scan line (typically going up to and beyond
), and index
is indicating the depth dimension (typically being sampled at and beyond an
MHz sampling rate). Given that the speed of sound,
m/s, on the order of
samples are acquired for each scan line for a typical
cm depth range.
In our work, we compare results obtained by the spectral-log-difference (SLD) method [
3,
9,
22] and the statistical moments (MoM) based approach [
10,
11] for three numerical phantoms.
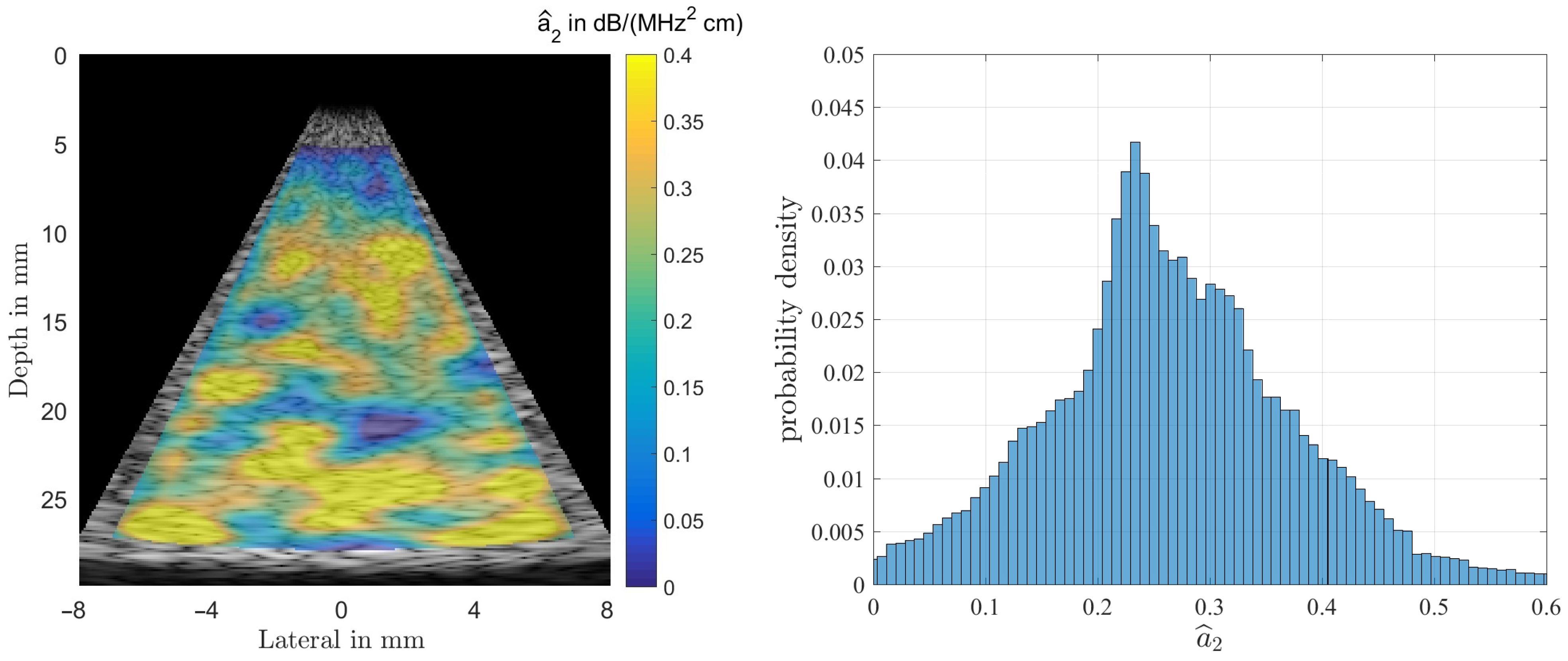
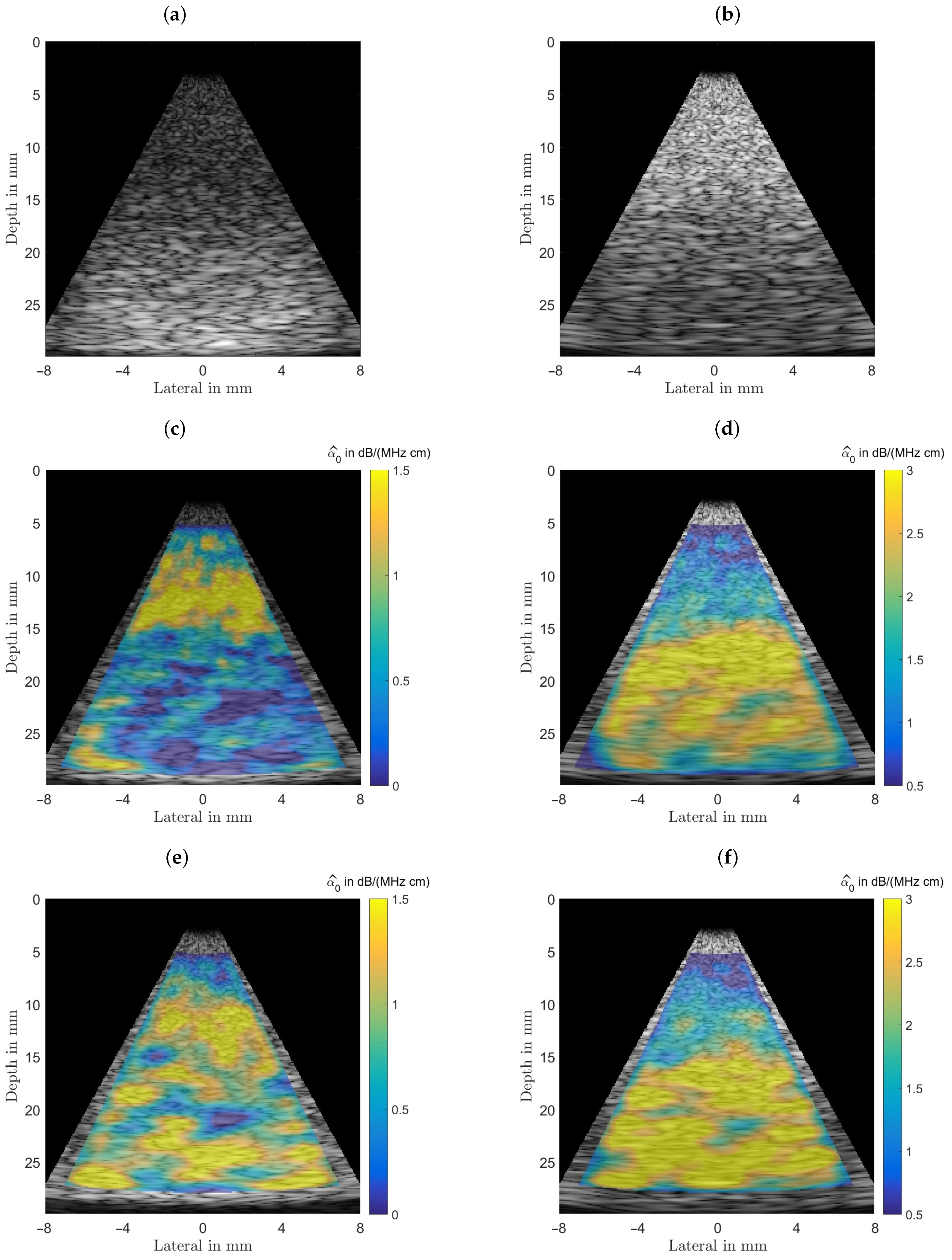
The first phantom is a homogeneous medium with dB/(MHz cm) used to obtain statistical properties of the estimators. The second and third phantoms mimic a liquid filled cyst with lower than average attenuation but a power-law exponent of , and a dense lesion with dB/(MHz cm) embedded in fat as medium. The SLD allows to estimate the attenuation coefficient only, the MoM also allows to estimate higher order terms of the frequency dependency of that might help to differentiate between normal tissue (), and fluid filled cysts ().
2.4.1. Spectral-Log-Difference Method
In order to estimate
over a region of interest (ROI) set by range-gated proximal and distal windows within a B-mode scan of a breast, the same time-gain-compensation (TGC), and the same power settings are used to obtain backscattered signals from different depths over a lateral range of B-mode scan lines
(see
Figure 5) to then be averaged. The authors in [
23] report results using block sizes of
axially by
laterally so considering an
MHz transducer this results in a block size of
mm
.
We define
, the power spectral density of the appropriately time-windowed (subscript
) sequence
evaluated at frequencies
, with
k, the frequency index,
the sampling frequency in Hz, and
, the lengths of the time windowed sequences. Please note, this window or tapering function used to suppress spectral side lobes is to be distinguished from the above mentioned depth windows that allow to select spectra from different depths.
Now define the transducer’s output signal representing the TGC-compensated B-mode image with ℓ being the scan line index of the sector scan, and n being the time index representing the depth dimension. Depth (assuming an approximately constant over depth speed of sound c).
Both windows are limited laterally by , thus in total scan lines are averaged over (denoted by ) in the following spectral estimation process. The proximal window extends between , and the distal window between samples.
Using the above definitions, the power spectral density of a depth-windowed region (mean depth for the proximal window
, and for the distal window
) in a statistically homogeneous tissue, varying only in the attenuation coefficient, and averaged over all
selected scan lines is given by (subscripts
p and
d indicate the proximal and distal ROIs).
for the proximal window, and
for the distal window, where
represents the combined effect of the transmit pulse and the sensitivity (electro-acoustic and acousto-electric transfer functions) of the transducer,
denotes the effects of focusing and diffraction that are related to the transducer geometry and its numerical aperture NA at average depth
,
is the cumulative attenuation along the propagation path from the surface of the transducer to depth
d of the respective depth window which corresponds to the centers
and
, respectively, of the ROIs,
is the attenuation coefficient within the considered ROIs,
is a result of the scattering properties of the tissue within the ROIs, namely the effective scatterer size, the scatterer number density, and the mean square variation in acoustic impedance between the scatterers and the background.
Dividing the power spectra obtained from different average depths
and
and assuming that the tissue within these depth ranges is homogeneous and isotropic, i.e., the scattering term
does not vary with depth, yields (for depths parameters in cm):
Taking the logarithm (to the base 10 to express the spectra in dB/Hz, and thus the attenuation coefficient in dB/(MHz cm)) yields:
In Equation (
3) [
24] the factor 4 stems from the fact that the attenuation ratio as measured in Np is referring to the attenuation of field quantities (as opposed to dB which gauges power quantities) combined with the round-trip propagation that effectively doubles the associated path lengths. The attenuation coefficient of the sample (in dB/(MHz cm)) can be estimated at each frequency component
(see Equation (
3)) by calculating
, the slope of the straight line that fits the log-ratio of the two spectra
, i.e., the slope of the straight line that fits Equation (
3) versus depth. If the attenuation as measured in dB is assumed to increase linearly with frequency, then the attenuation coefficient’s slope is used as a measure for the attenuation in the tissue of interest. The attenuation coefficient can then be simplified to
where the parameter
is the attenuation coefficient’s slope in dB/(MHz cm) that can be estimated by finding the slope that fits Equation (
3).
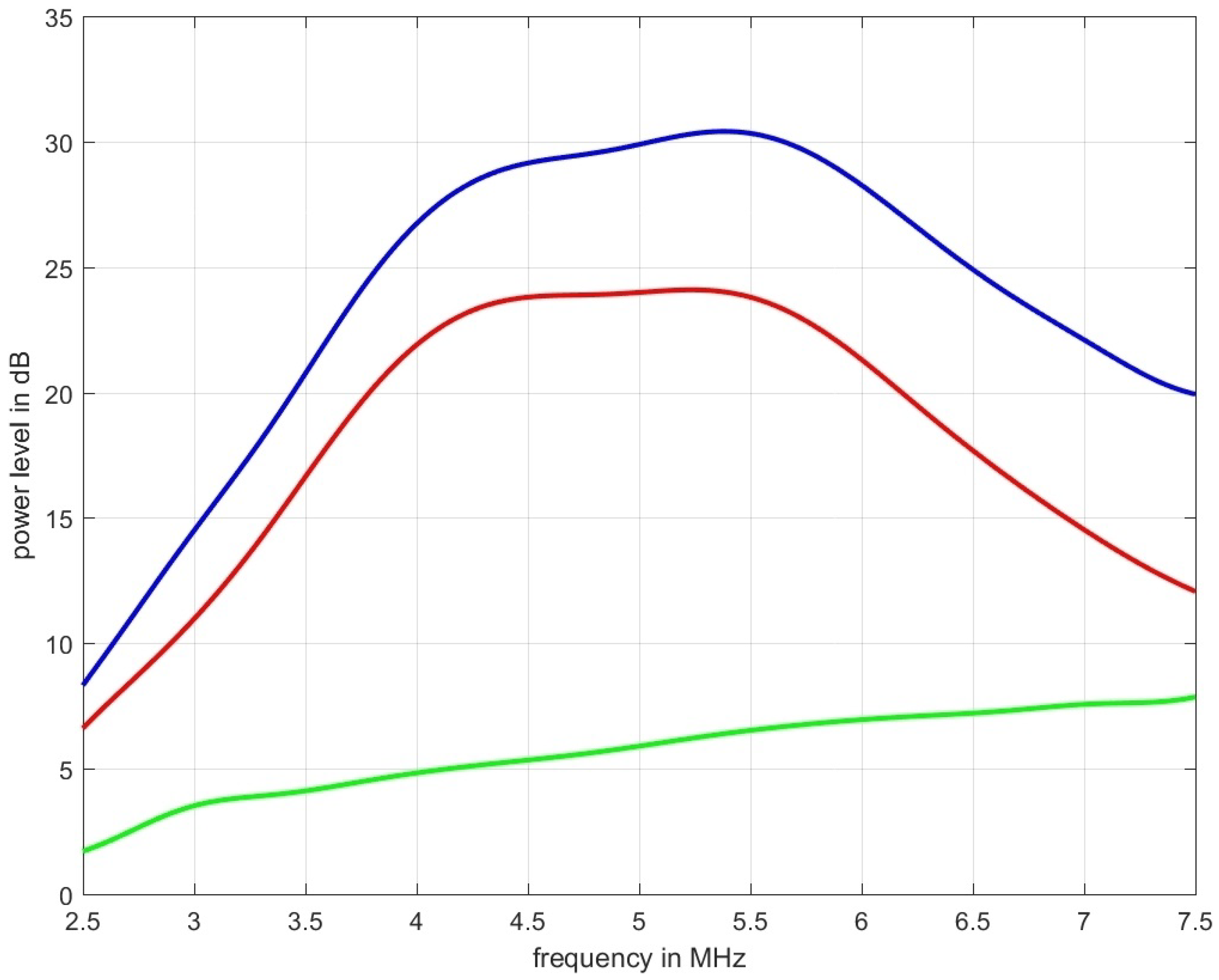
Figure 6 shows measurement data acquired for the tissue mimicking phantom shown in
Figure 1. The blue graph represents the power spectral density for the ultrasound propagation in pure water, the red line is the measured spectrum with the phantom inserted, and the green line is the spectral-log-difference to which a straight line could be fitted to obtain
(in this case
dB/(MHz cm)).
2.4.2. Method-of-Moments (MoM)
Another method for estimating the attenuation coefficient as discussed in the literature [
10,
11] is again based on the power spectral densities
and
averaged over two spatially limited regions of interest (cf.
Figure 5) from
to
laterally and from
to
for the distal, and from
to
for the proximal window, respectively, radially.
By taking their zeroth
to second order
moments according to
i being the moment order, and expanding
into a power series:
and noting that the derivative
can be written as:
and expressed via the
th moment of the spectrum
.
The parameter
, which is equivalent to
, and
, which is equivalent to
y of Equation (
2) can then be expressed as:
As a note: in estimating the ultrasound’s attenuation the method-of-moments not only utilizes the decline in amplitude with increasing depth d but in addition takes into account that the frequency dependent attenuation also reduces the center frequency of the propagating wave. It thus should allow for smaller variances of the estimators. Our analysis, however, does not seem to support that assumption, as can be seen by observing the estimation results shown in the following section.