Evaluation of Erythema Severity in Dermatoscopic Images of Canine Skin: Erythema Index Assessment and Image Sampling Reliability
Abstract
1. Introduction
2. Materials and Methods
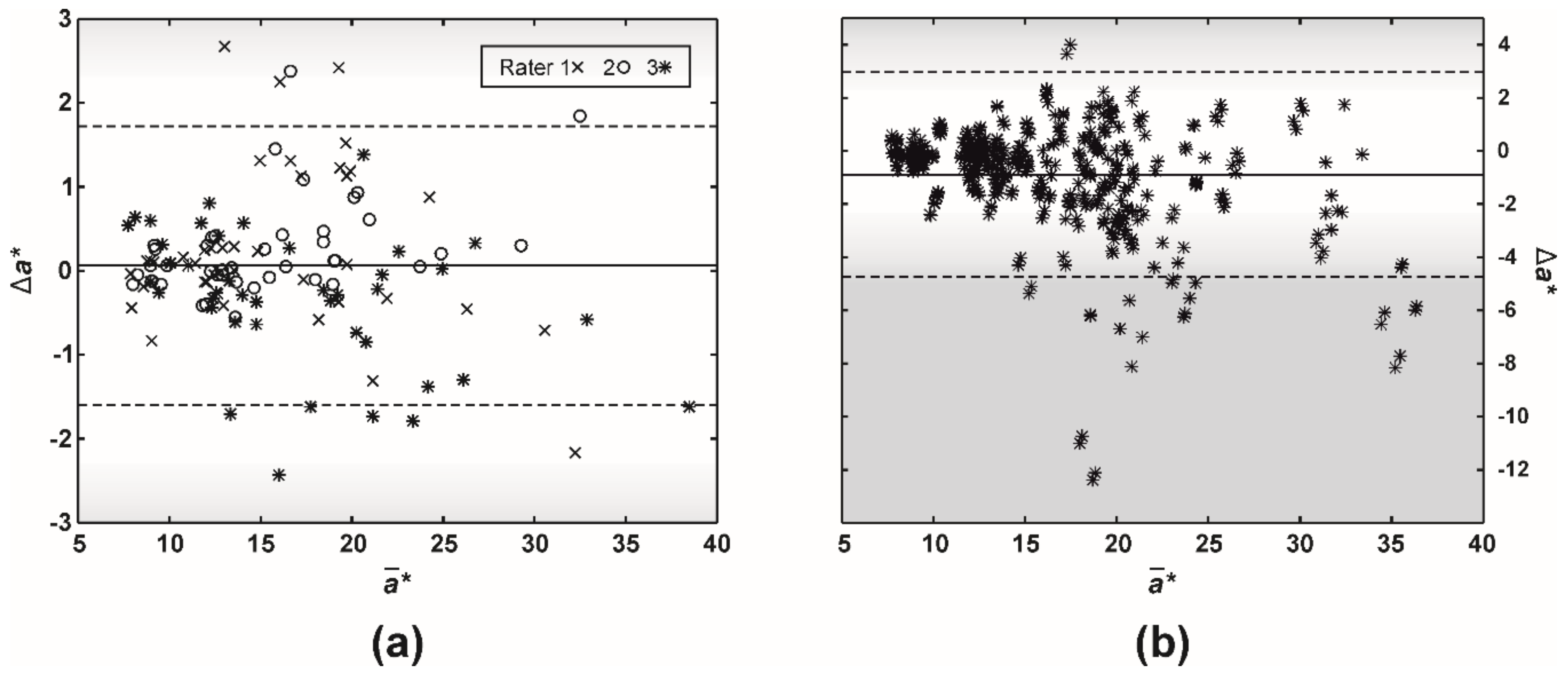
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Olivry, T.; Saridomichelakis, M.; Nuttall, T.; Bensignor, E.; Griffin, C.E.; Hill, P.B. Validation of the Canine Atopic Dermatitis Extent and Severity Index (CADESI)-4, a simplified severity scale for assessing skin lesions of atopic dermatitis in dogs. Vet. Dermatol. 2014, 25, 77. [Google Scholar] [CrossRef] [PubMed]
- Saridomichelakis, M.N.; Olivry, T. An update on the treatment of canine atopic dermatitis. Vet. J. 2016, 207, 29–37. [Google Scholar] [CrossRef]
- Olivry, T.; DeBoer, D.J.; Favrot, C.; Jackson, H.A.; Mueller, R.S.; Nuttall, T.; Prélaud, P. Treatment of canine atopic dermatitis: 2015 updated guidelines from the International Committee on Allergic Diseases of Animals (ICADA). BMC Vet. Res. 2015, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Rybníĉek, J.; Harvey, R.; Hill, P.B.; Lau-Gillard, P.J. Further validation of a pruritus severity scale for use in dogs. Vet. Dermatol. 2009, 20, 115–122. [Google Scholar] [CrossRef]
- Olivry, T. Could we use the erythema grading of the CADESI4 as a simple instrument for future short-duration clinical trials of dogs with atopic dermatitis? Vet. Dermatol. 2018, 30, 80–81. [Google Scholar] [CrossRef]
- Frew, J.; Penzi, L.; Suárez-Fariñas, M.; Garcet, S.; Brunner, P.M.; Czarnowicki, T.; Kim, J.; Bottomley, C.; Finney, R.; Cueto, I.; et al. The erythema Q-score, an imaging biomarker for redness in skin inflammation. Exp. Dermatol. 2020, 1–7. [Google Scholar] [CrossRef]
- High, E.J.; Olivry, T. Development and validation of a graphic 2D investigator’s global assessment instrument for grading the overall severity of atopic dermatitis in dogs. Vet. Dermatol. 2020, 31, 207. [Google Scholar] [CrossRef] [PubMed]
- Tan, J.; Liu, H.; Leyden, J.J.; Leoni, M.J. Reliability of Clinician Erythema Assessment grading scale. J. Am. Acad. Dermatol. 2014, 71, 760–763. [Google Scholar] [CrossRef]
- Raina, A.; Hennessy, R.J.; Rains, M.; Allred, J.; Hirshburg, J.M.; Diven, D.G.; Markey, M.K. Objective measurement of erythema in psoriasis using digital color photography with color calibration. Ski. Res. Technol. 2016, 22, 375–380. [Google Scholar] [CrossRef]
- Bożek, A.; Reich, A. Assessment of Intra- and Inter-Rater Reliability of Three Methods for Measuring Atopic Dermatitis Severity: EASI, Objective SCORAD, and IGA. Dermatology 2017, 233, 16–22. [Google Scholar] [CrossRef]
- Riordan, B.; Sprigle, S.; Linden, M. Testing the validity of erythema detection algorithms. J. Rehabil. Res. Dev. 2001, 38, 13–22. [Google Scholar]
- Zhao, C.; Wijayanti, A.; Doria, M.; Harris, A.G.; Jain, S.; Legaspi, K.; Dlova, N.; Law, M.; Murrell, D.F. The reliability and validity of outcome measures for atopic dermatitis in patients with pigmented skin: A grey area. Int. J. Womens Dermatol. 2015, 1, 150–154. [Google Scholar] [CrossRef]
- Dall’Oglio, F.; Verzì, A.E.; Lacarrubba, F.; Giuffrida, G.; Milani, M.; Micali, G. Inter-observer evaluation of erythema-directed photography for the assessment of erythema and telangiectasias in rosacea. Ski. Res. Technol. 2020, 1–5. [Google Scholar] [CrossRef]
- Clarys, P.; Alewaeters, K.; Lambrecht, R.; Barel, A.O. Skin color measurements: Comparison between three instruments: The Chromameter® the DermaSpectrometer® and the Mexameter®. Ski. Res. Technol. 2000, 6, 230–238. [Google Scholar] [CrossRef]
- Baquié, M.; Kasraee, B. Discrimination between cutaneous pigmentation and erythema: Comparison of the skin colorimeters Dermacatch and Mexameter. Ski. Res. Technol. 2013, 20, 218–227. [Google Scholar] [CrossRef]
- Abdlaty, R.; Doerwald-Munoz, L.; Madooei, A.; Sahli, S.; Yeh, S.-C.A.; Zerubia, J.; Wong, R.K.W.; Hayward, J.E.; Farrell, T.J.; Fang, Q. Hyperspectral Imaging and Classification for Grading Skin Erythema. Front. Phys. 2018, 6, 72. [Google Scholar] [CrossRef]
- Jung, B.; Kim, C.-S.; Choi, B.; Kelly, K.M.; Nelson, J.S. Use of erythema index imaging for systematic analysis of port wine stain skin response to laser therapy. Lasers Surg. Med. 2005, 37, 186–191. [Google Scholar] [CrossRef]
- Saknite, I.; Zavorins, A.; Jakovels, D.; Spigulis, J.; Kisis, J. Comparison of single-spot technique and RGB imaging for erythema index estimation. Physiol. Meas. 2016, 37, 333–346. [Google Scholar] [CrossRef] [PubMed]
- Cugmas, B.; Olivry, T. Evaluation of skin erythema severity by dermatoscopy in dogs with atopic dermatitis. Vet. Dermatol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Uthoff, R.D.; Song, B.; Maarouf, M.; Shi, V.Y.; Liang, R. Point-of-care, multispectral, smartphone-based dermascopes for dermal lesion screening and erythema monitoring. J. Biomed. Opt. 2020, 25, 066004. [Google Scholar] [CrossRef]
- Cugmas, B.; Olivry, T.; Olivrī, A.; Spigulis, J. Skimager for the objective erythema estimation in atopic dogs. Photonics Dermatol. Plast. Surg. 2020, 11211, 1121110. [Google Scholar] [CrossRef]
- Bodekaer, M.; Philipsen, P.A.; Karlsmark, T.; Wulf, H.C.; Bodekær, M. Good agreement between minimal erythema dose test reactions and objective measurements: An in vivo study of human skin. Photodermatol. Photoimmunol. Photomed. 2013, 29, 190–195. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, T.; Takiwaki, H.; Arase, S.; Ohshima, H. Derivation and clinical application of special imaging by means of digital cameras and Image J freeware for quantification of erythema and pigmentation. Ski. Res. Technol. 2007, 14, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Ohshima, H.; Takiwaki, H.; Washizaki, K.; Ishiko, A.; Itoh, M.; Kanto, H. Quantitative evaluation of patch test reactions: A comparison between visual grading and erythema index image analysis. Ski. Res. Technol. 2011, 17, 220–225. [Google Scholar] [CrossRef]
- Logger, J.G.M.; De Jong, E.M.G.J.; Driessen, R.J.B.; Van Erp, P.E.J. Evaluation of a simple image-based tool to quantify facial erythema in rosacea during treatment. Ski. Res. Technol. 2020, 26, 804–812. [Google Scholar] [CrossRef] [PubMed]
- Wagner, J.K.; Jovel, C.; Norton, H.L.; Parra, E.J.; Shriver, M.D. Comparing Quantitative Measures of Erythema, Pigmentation and Skin Response using Reflectometry. Pigment. Cell Res. 2002, 15, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Hua, W.; Li, A.; He, H.; Xie, L.; Li, L. Analysis of facial redness by comparing VISIA® from Canfield and CSKIN® from Yanyun Technology. Ski. Res. Technol. 2020, 26, 696–701. [Google Scholar] [CrossRef]
- Cho, M.; Lee, D.-H.; Doh, E.J.; Kim, Y.; Chung, J.H.; Kim, H.C.; Kim, S. Development and clinical validation of a novel photography-based skin erythema evaluation system: A comparison with the calculated consensus of dermatologists. Int. J. Cosmet. Sci. 2017, 39, 426–434. [Google Scholar] [CrossRef] [PubMed]
- Cugmas, B.; Štruc, E. Accuracy of an Affordable Smartphone-Based Teledermoscopy System for Color Measurements in Canine Skin. Sensors 2020, 20, 6234. [Google Scholar] [CrossRef]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr. Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Boysen, L.; Sørensen, P.; Larsen, M.; Serup, J.; Kristensen, F. Evaluation of skin erythema by use of chromametry and image analysis of digital photographs after intradermal administration of histamine in dogs. Am. J. Vet. Res. 2002, 63, 565–569. [Google Scholar] [CrossRef] [PubMed]
- Nicholson, M.; Willis, C.M. The influence of patch test size and design on the distribution of erythema induced by sodium lauryl sulfate. Contact Dermat. 1999, 41, 264–267. [Google Scholar] [CrossRef] [PubMed]
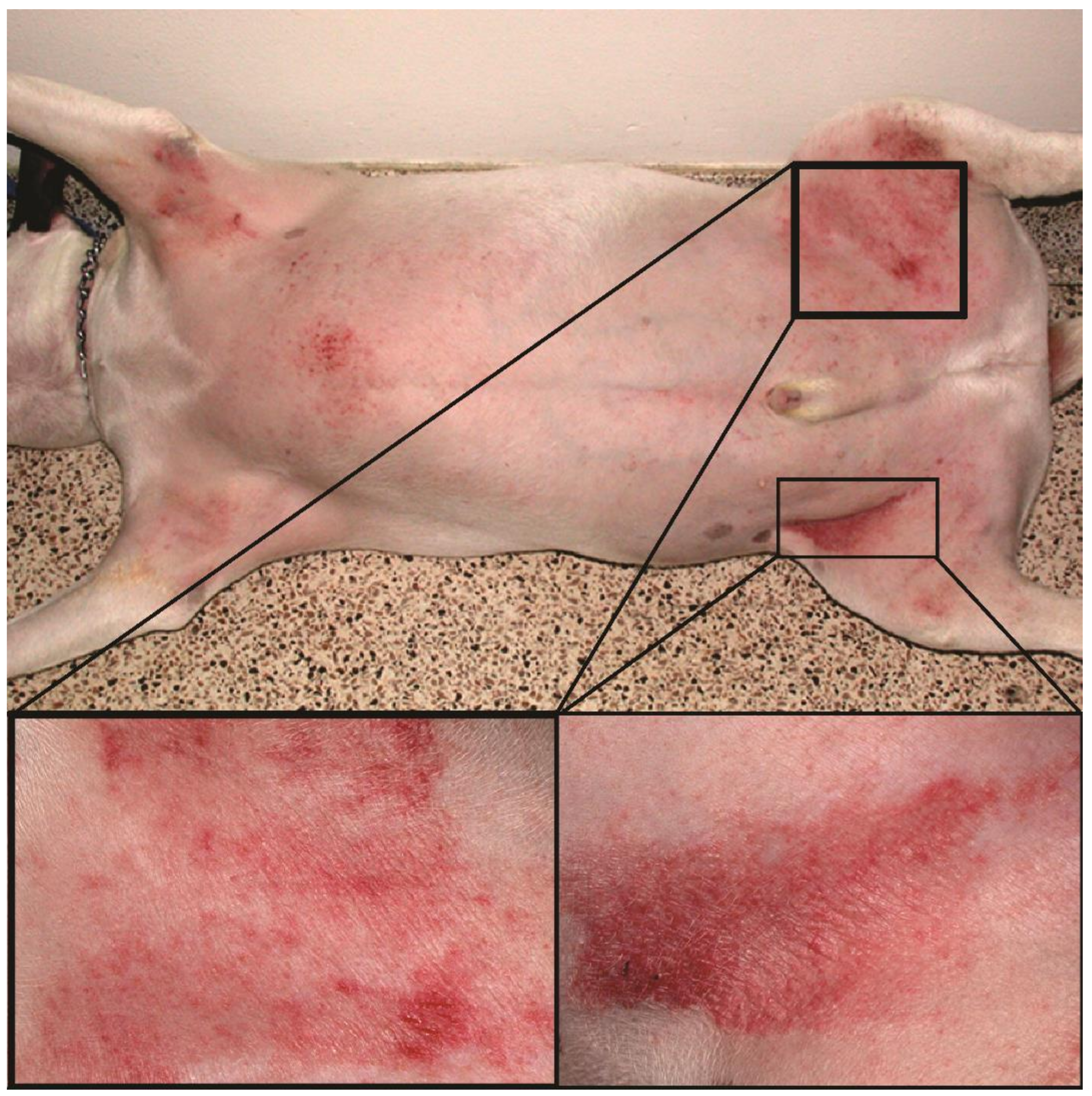

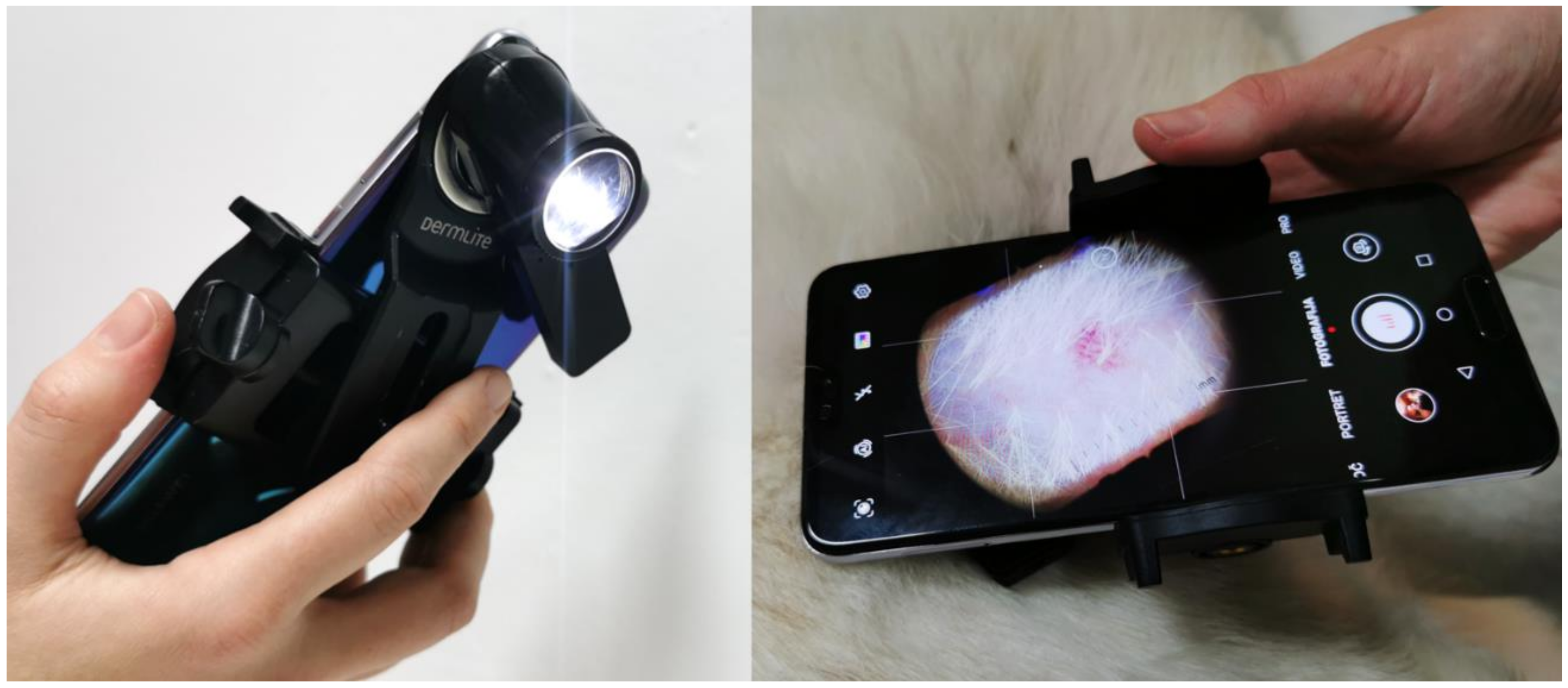
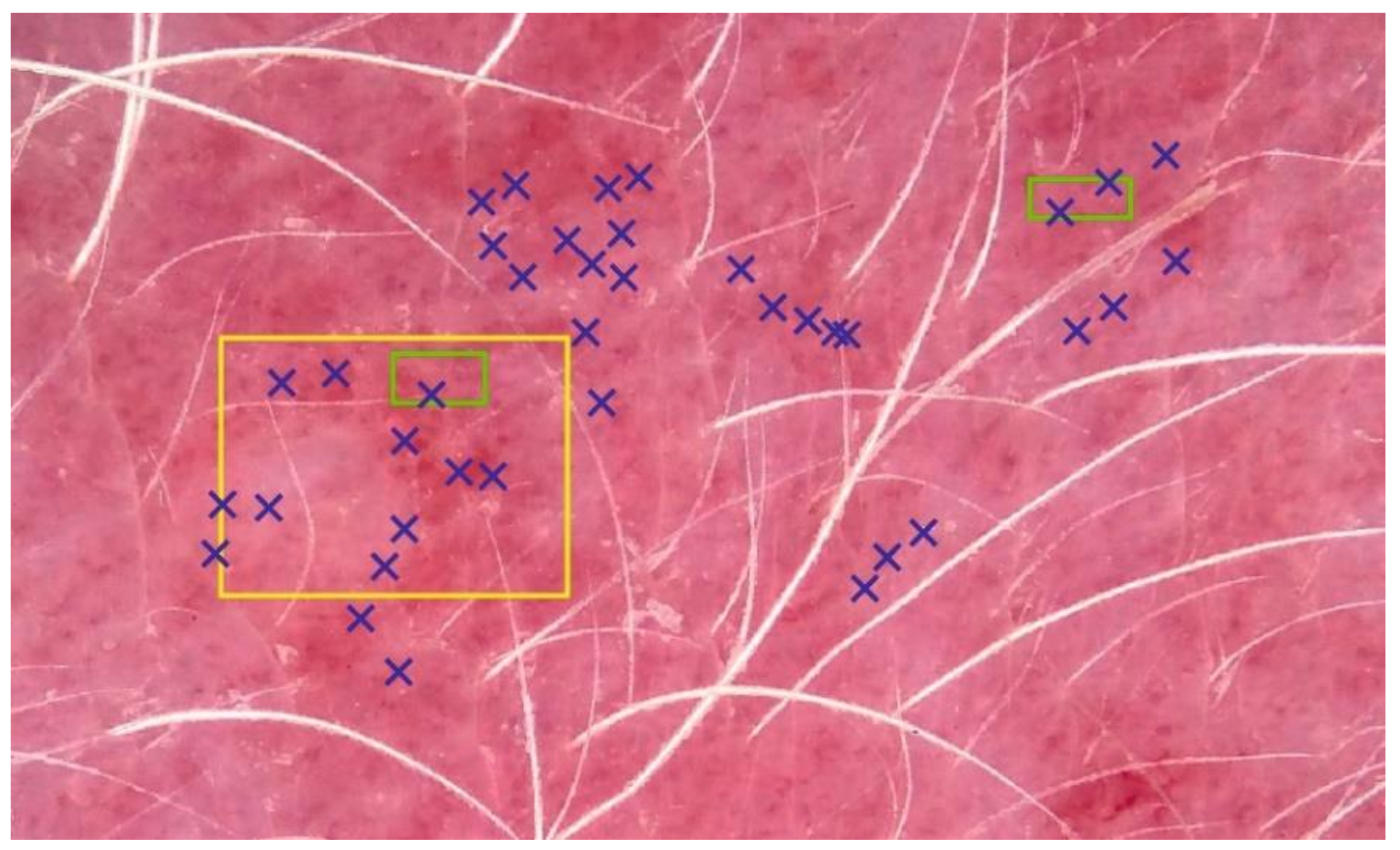
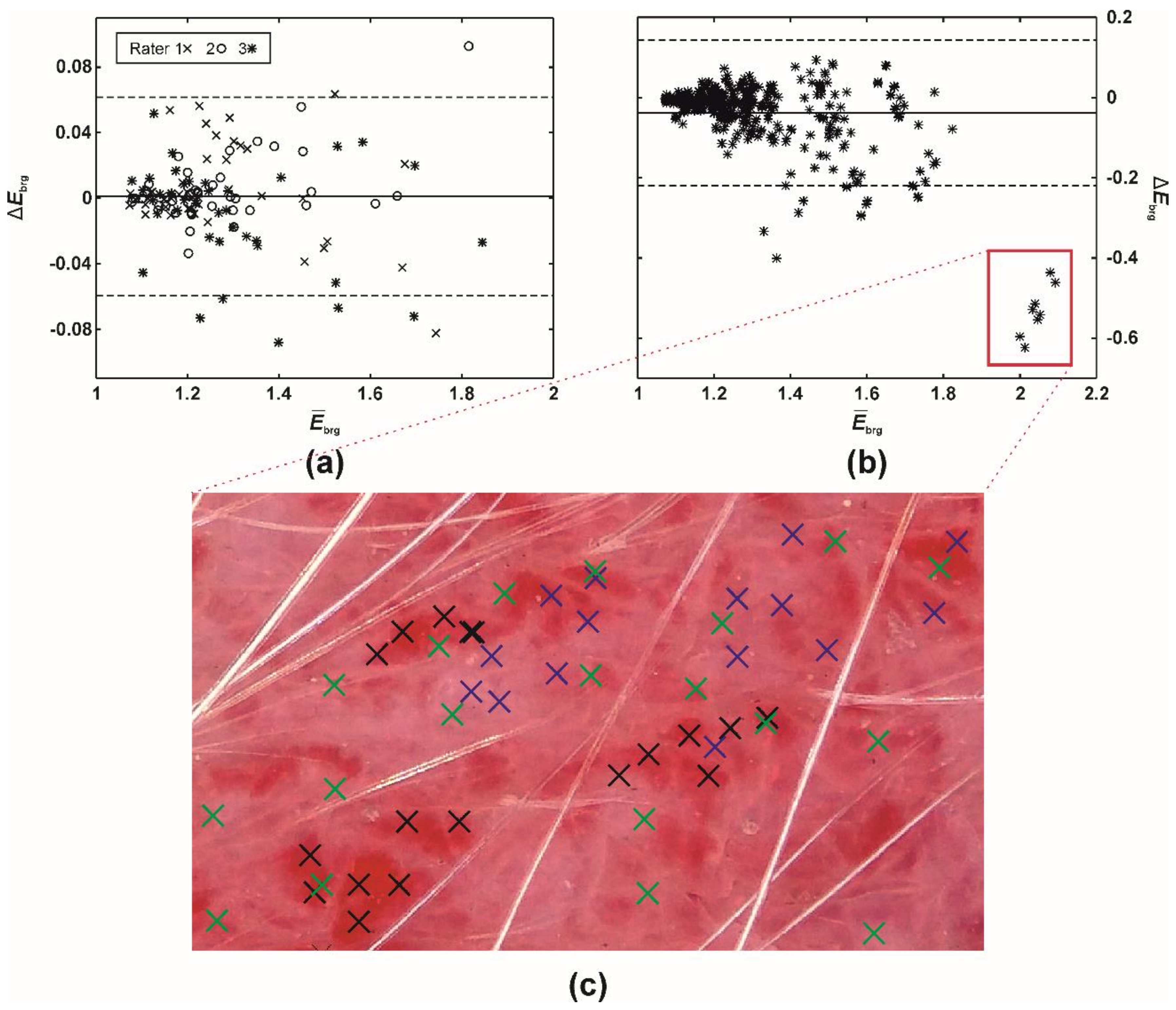
| ISM\EI | RG | GR | BRG | BG | a* | Mean |
|---|---|---|---|---|---|---|
| PT | 0.73 ± 0.04 | −0.73 ± 0.04 | 0.75 ± 0.05 | 0.66 ± 0.05 | 0.70 ± 0.03 | 0.71 ± 0.05 |
| SQ2 | 0.72 ± 0.05 | −0.72 ± 0.05 | 0.75 ± 0.05 | 0.67 ± 0.05 | 0.70 ± 0.05 | 0.71 ± 0.05 |
| SQ1 | 0.70 ± 0.05 | −0.70 ± 0.05 | 0.73 ± 0.05 | 0.61 ± 0.06 | 0.69 ± 0.04 | 0.68 ± 0.06 |
| Mean | 0.71 ± 0.05 | −0.72 ± 0.05 | 0.74 ± 0.05 | 0.65 ± 0.06 | 0.70 ± 0.04 |
| ISM\EI | RG | GR | BRG | BG | a* | Mean |
|---|---|---|---|---|---|---|
| PT | 11.7 ± 0.7 | 11.7 ± 0.6 | 11.6 ± 0.5 | 12.7 ± 0.6 | 12.1 ± 0.3 | 12.0 ± 0.7 |
| SQ2 | 11.9 ± 1.0 | 11.9 ± 0.8 | 11.6 ± 0.8 | 12.6 ± 0.8 | 12.3 ± 0.5 | 12.1 ± 0.8 |
| SQ1 | 12.2 ± 0.7 | 12.3 ± 0.6 | 12.0 ± 0.5 | 12.9 ± 0.9 | 12.4 ± 0.3 | 12.4 ± 0.7 |
| Mean | 11.9 ± 0.8 | 12.0 ± 0.6 | 11.7 ± 0.6 | 12.7 ± 0.7 | 12.3 ± 0.4 |
| Rater | Spearman | ICC (2, k) | αk | ΔEIBRG | Δa* |
|---|---|---|---|---|---|
| Intra- | 0.97 ± 0.03 | 0.99 ± 0.01 | 0.98 ± 0.02 | 0.02 ± 0.02 | 0.6 ± 0.6 |
| Inter- | 0.94 ± 0.03 | 0.95 ± 0.04 | 0.90 ± 0.07 | 0.05 ± 0.09 | 1.3 ± 1.7 |
| Rater | PT | SQ2 | SQ1 |
|---|---|---|---|
| Intra- | 0.99 ± 0.01 | 0.98 ± 0.01 | 0.97 ± 0.02 |
| Inter- | 0.88 ± 0.08 | 0.88 ± 0.07 | 0.93 ± 0.04 |
| Rater | 0 (None) | 1 (Mild) | 2 (Moderate) | 3 (Severe) |
|---|---|---|---|---|
| Intra- | 0.01 [0.00–0.07] | 0.01 [0.00–0.06] | 0.02 [0.00–0.06] | 0.03 [0.00–0.14] |
| Inter- | 0.01 [0.00–0.33] | 0.01 [0.00–0.22] | 0.05 [0.00–0.22] | 0.05 [0.00–0.60] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cugmas, B.; Viškere, D.; Štruc, E.; Olivry, T. Evaluation of Erythema Severity in Dermatoscopic Images of Canine Skin: Erythema Index Assessment and Image Sampling Reliability. Sensors 2021, 21, 1285. https://doi.org/10.3390/s21041285
Cugmas B, Viškere D, Štruc E, Olivry T. Evaluation of Erythema Severity in Dermatoscopic Images of Canine Skin: Erythema Index Assessment and Image Sampling Reliability. Sensors. 2021; 21(4):1285. https://doi.org/10.3390/s21041285
Chicago/Turabian StyleCugmas, Blaž, Daira Viškere, Eva Štruc, and Thierry Olivry. 2021. "Evaluation of Erythema Severity in Dermatoscopic Images of Canine Skin: Erythema Index Assessment and Image Sampling Reliability" Sensors 21, no. 4: 1285. https://doi.org/10.3390/s21041285
APA StyleCugmas, B., Viškere, D., Štruc, E., & Olivry, T. (2021). Evaluation of Erythema Severity in Dermatoscopic Images of Canine Skin: Erythema Index Assessment and Image Sampling Reliability. Sensors, 21(4), 1285. https://doi.org/10.3390/s21041285







