Textile-Based Sensors for Biosignal Detection and Monitoring
Abstract
:1. Introduction
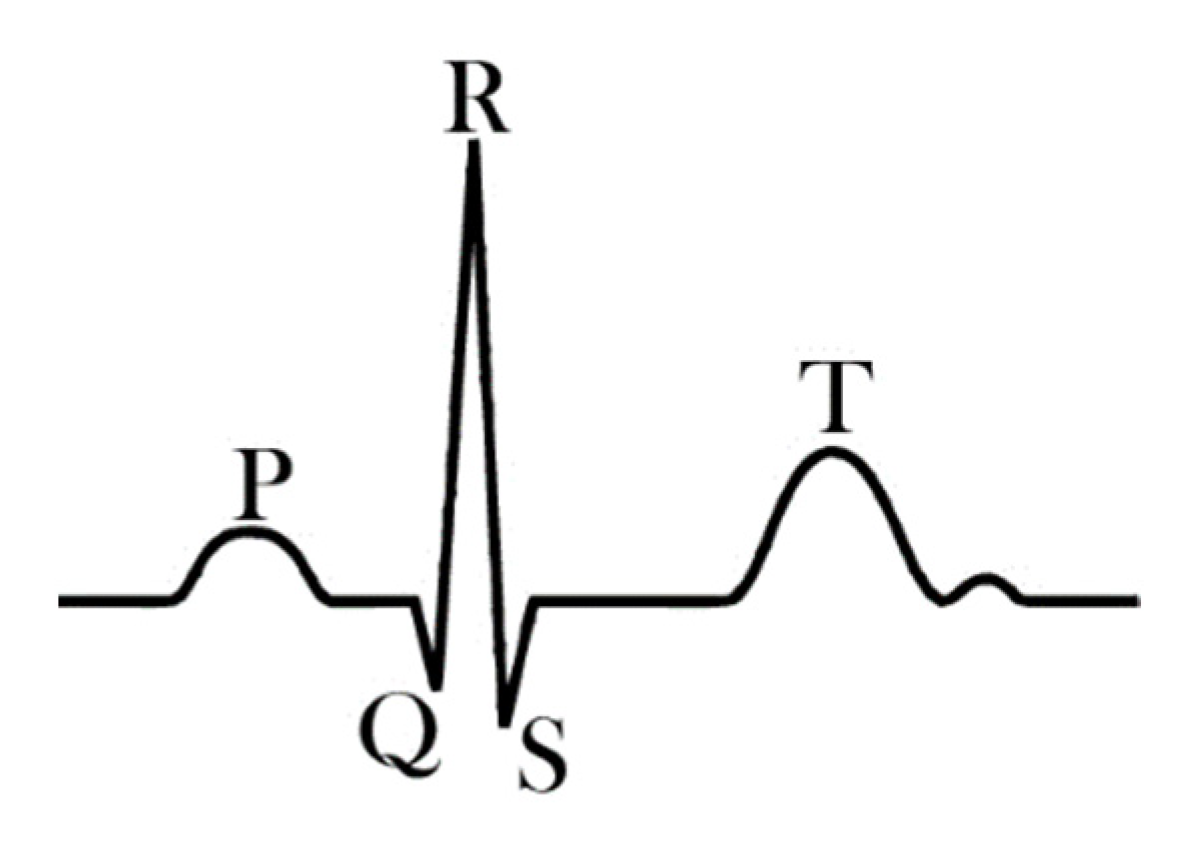
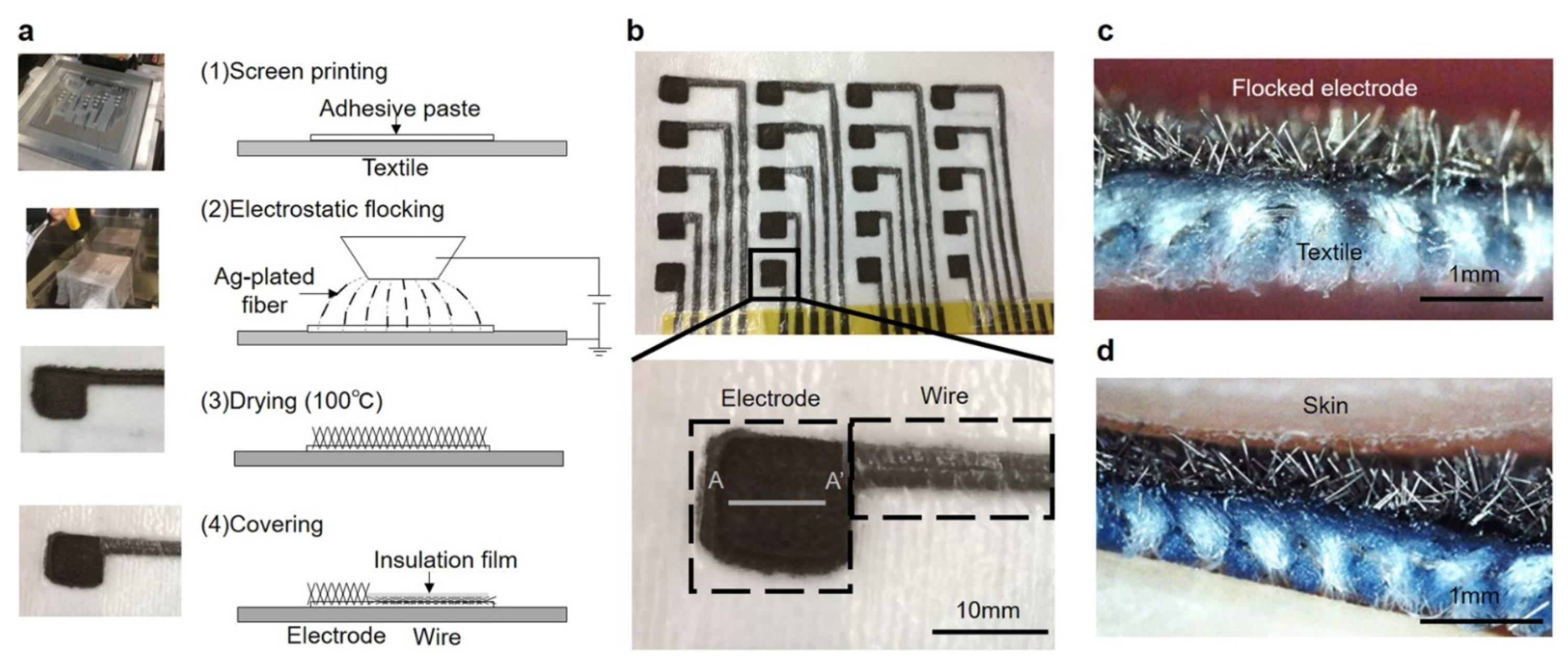
2. ECG Measurement
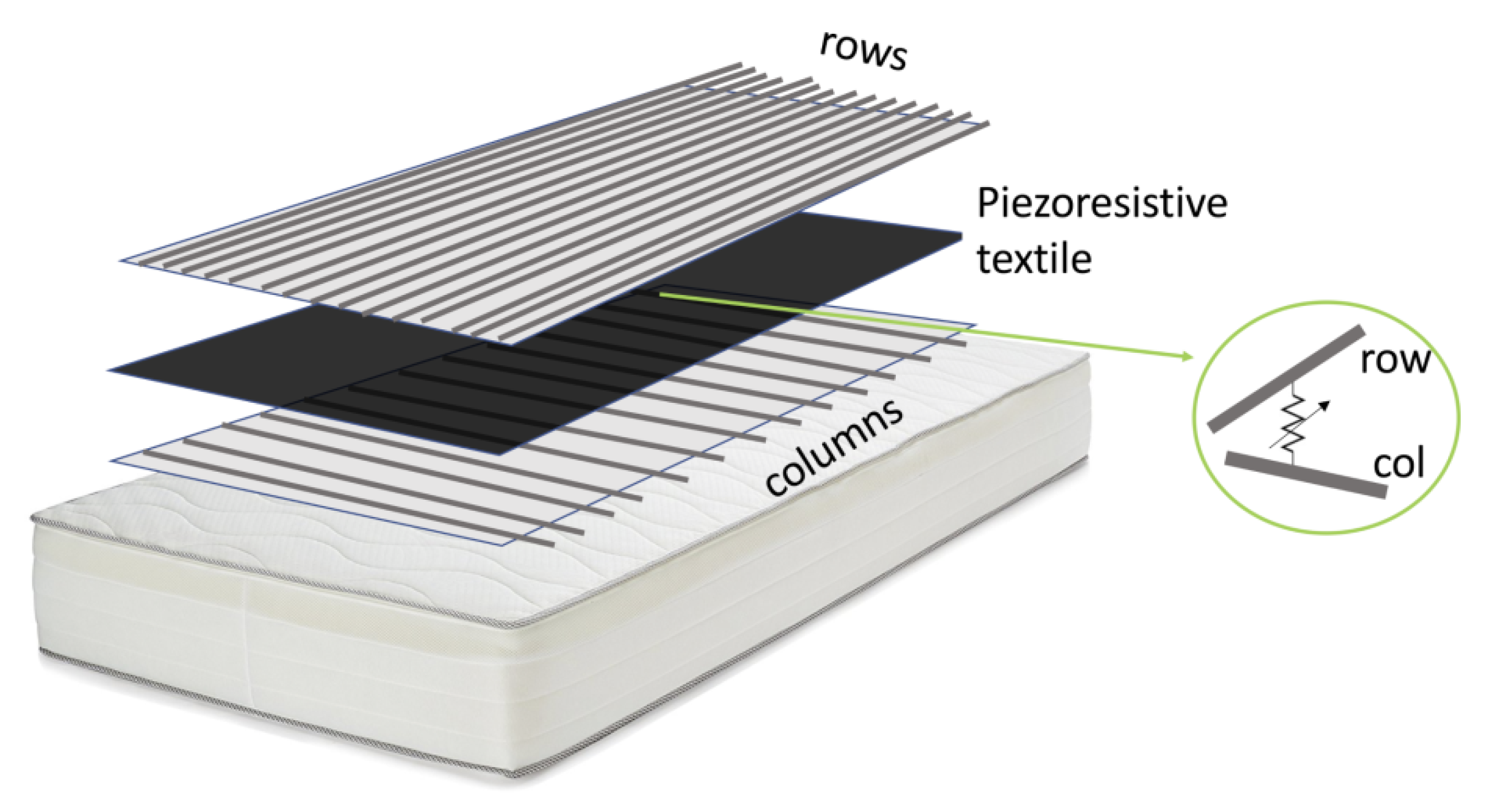
3. Breathing Measurement
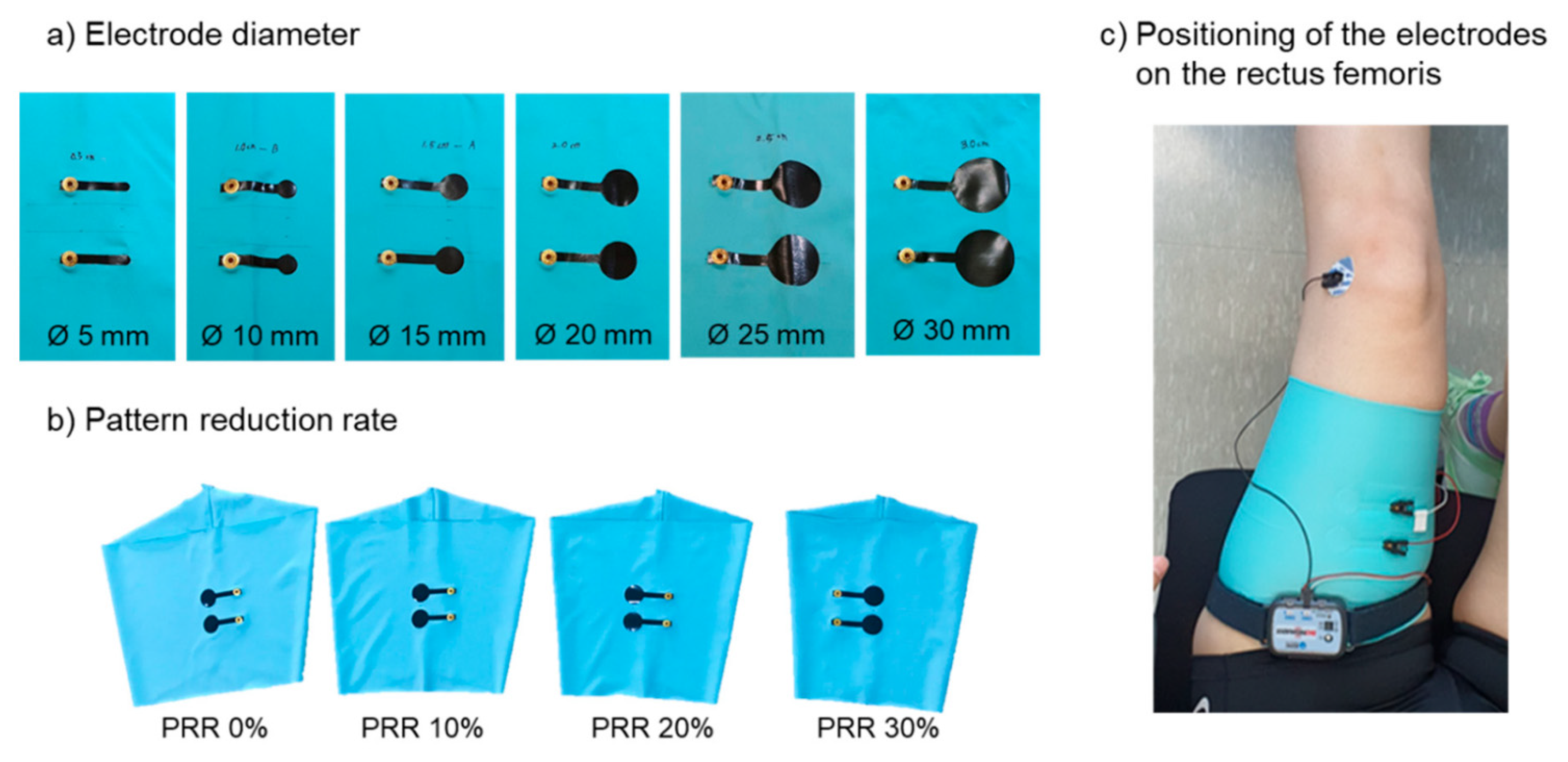
4. EMG Measurement
5. EEG Measurement
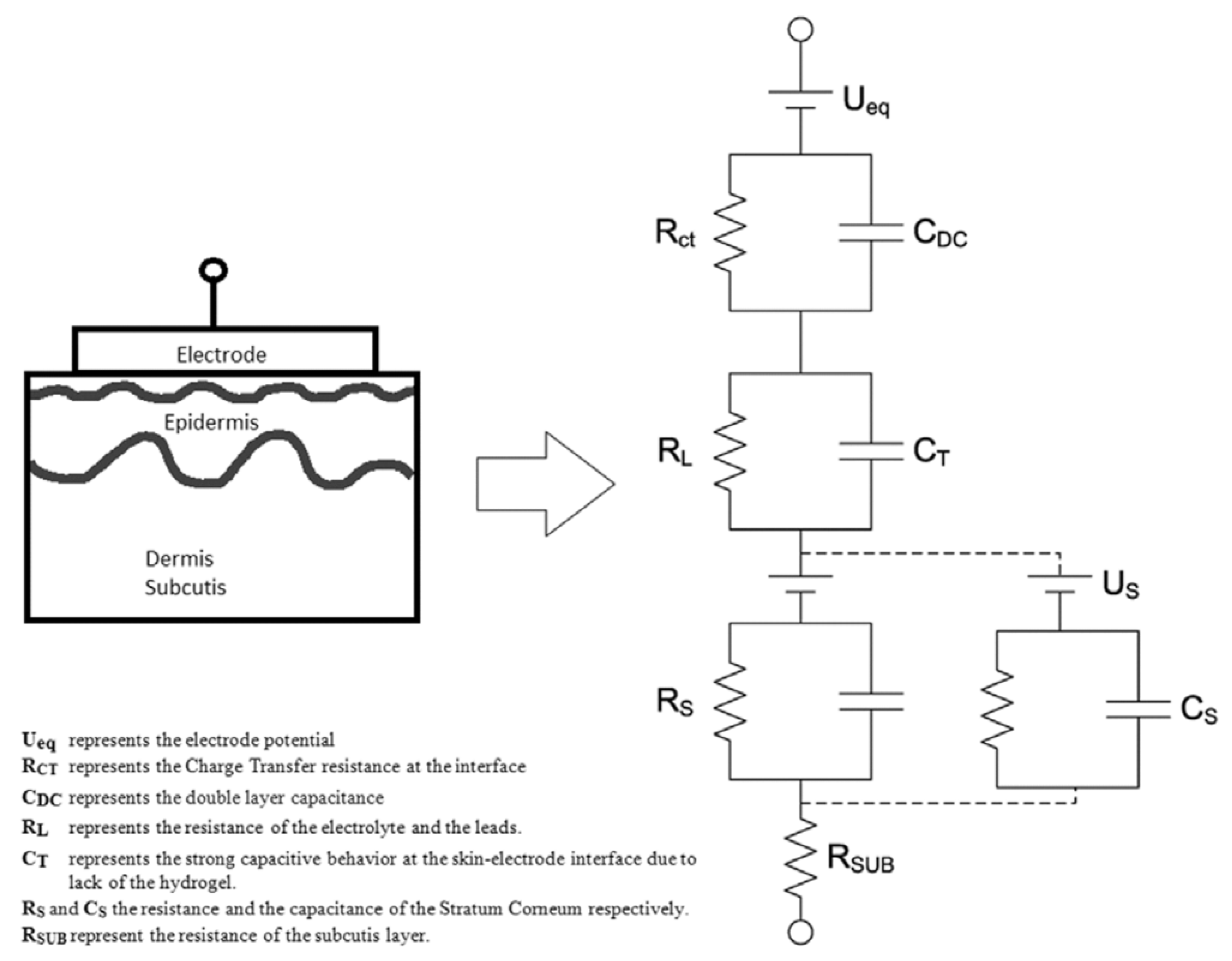
6. Bioimpedance Measurement
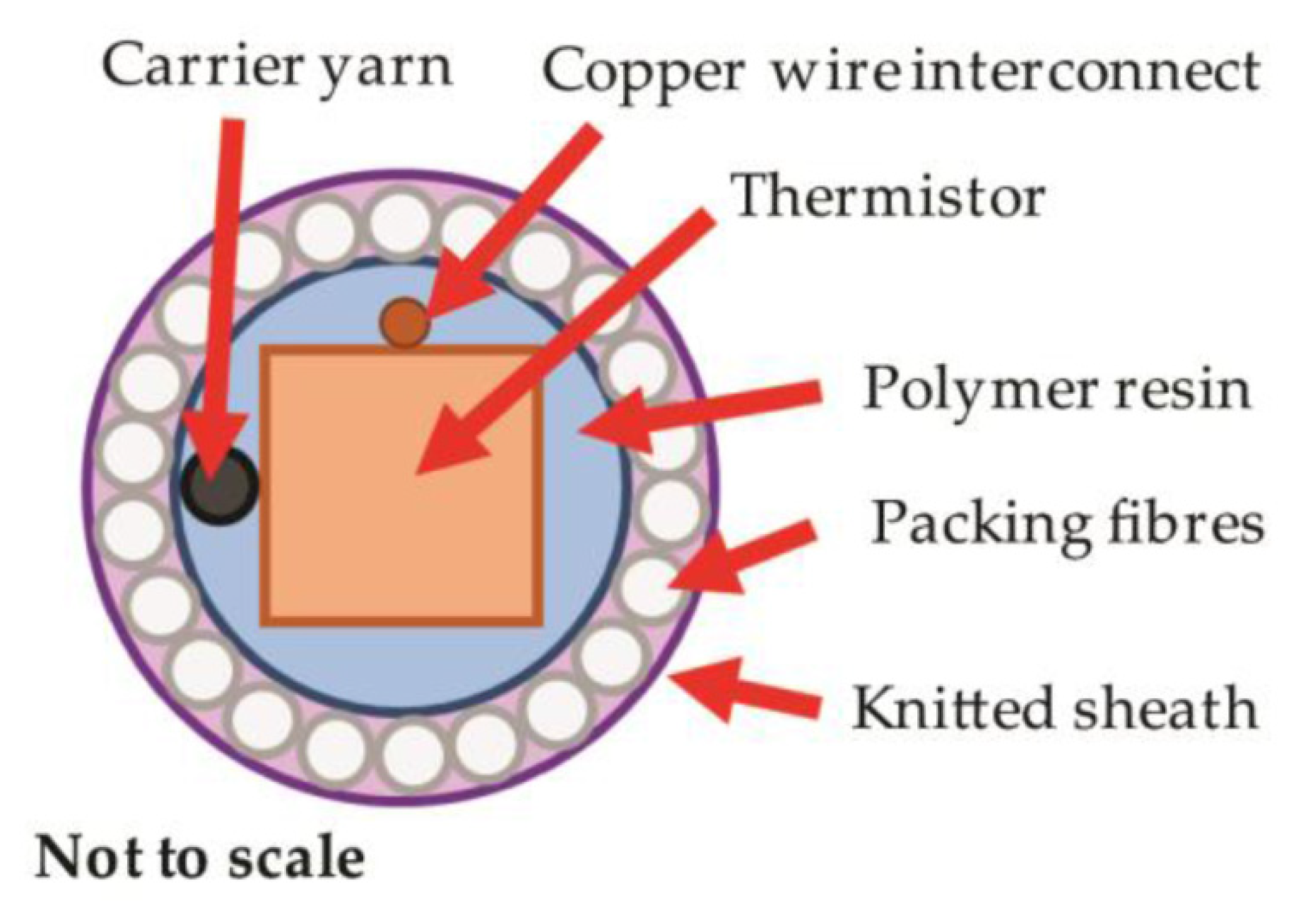
7. Skin Temperature Measurement
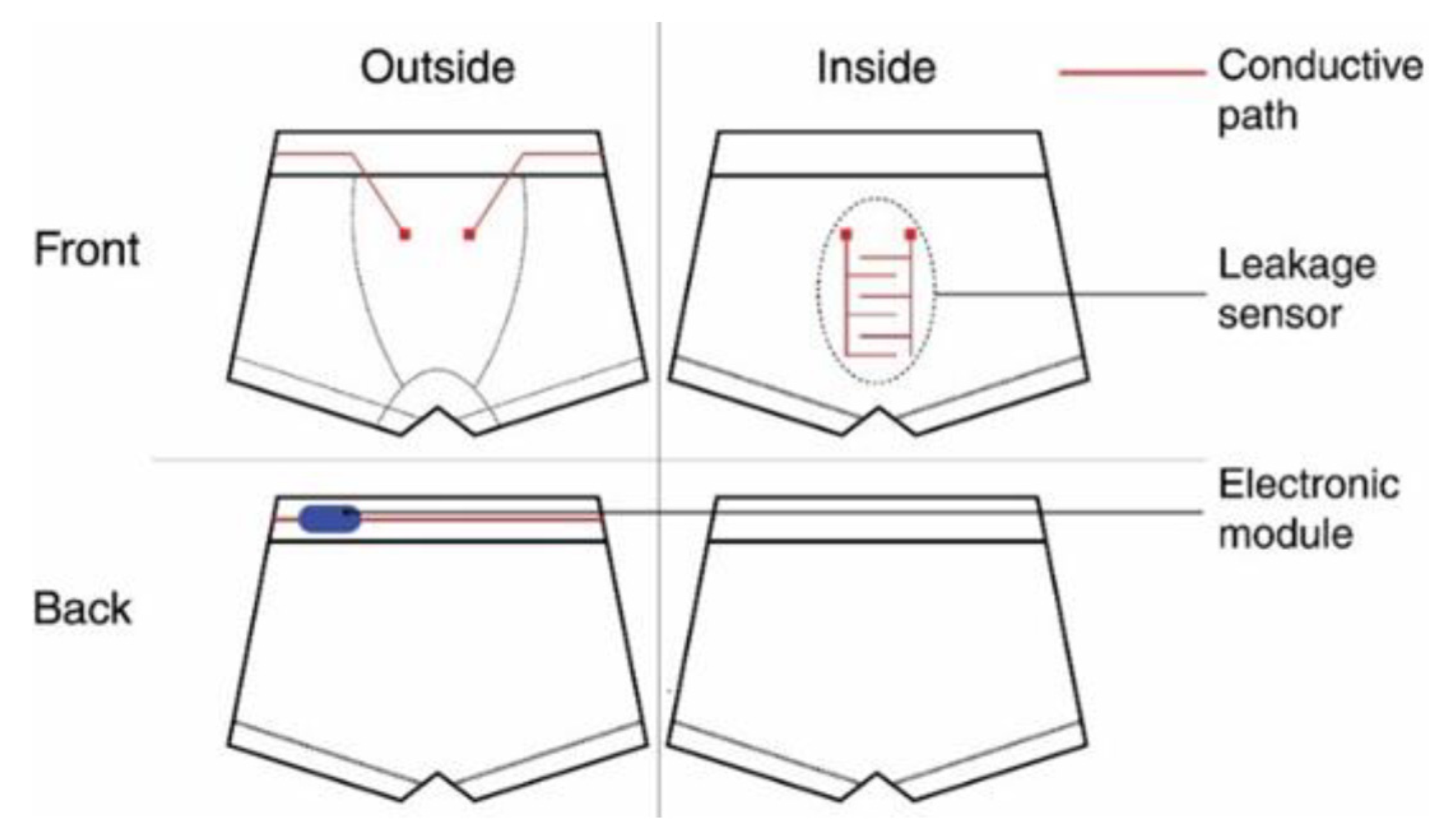
8. Moisture Detection
9. Sweat Examination
10. Advantages and Challenges of Textile-Based Biosensors
11. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Pantelopoulos, A.; Bourbakis, N. A survey on wearable biosensor systems for health monitoring. In Proceedings of the 2008 30th Annual Conference of the IEEE Engineering in Medicine and Biology Society, Vancouver, BC, Canada, 20–25 August 2008; pp. 4887–4890. [Google Scholar]
- Schwarz, A.; van Langenhove, L.; Guermonprez, P.; Deguillemont, D. A roadmap on smart textiles. Text. Progress 2010, 42, 99–180. [Google Scholar] [CrossRef]
- Stoppa, M.; Chiolerio, A. Wearable electronics and smart textiles: A critical review. Sensors 2014, 14, 11957–11992. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Omura, Y. Sensor Technology for Monitoring of Health-Related Conditions. In Comprehensive Materials Processing; Sensor Materials, Technologies and Applications; Hashmi, S., Ed.; Elsevier: Amsterdam, The Netherlands, 2014; Volume 13. [Google Scholar]
- Koncar, V. (Ed.) Introduction to smart textile and their applications. In Smart Textiles and their Applications; Woodhead Publishing Series in Textiles: Cambridge, UK, 2016; pp. 1–8. [Google Scholar]
- Ehrmann, G.; Ehrmann, A. Electronic textiles. Encyclopedia 2021, 1, 115–130. [Google Scholar] [CrossRef]
- Hughes-Riley, T.; Dias, T.; Cork, C. A historical review of the development of electronic textiles. Fibers 2018, 6, 34. [Google Scholar] [CrossRef] [Green Version]
- Wagner, S.; Bonderover, E.; Jordan, W.B.; Sturm, J.C. Electrotextiles: Concepts and challenges. Int. J. High Speed Electron. Syst. 2002, 12, 391–399. [Google Scholar] [CrossRef]
- Ehrmann, G.; Ehrmann, A. Suitability of common single circuit boards for sensing and actuating in smart textiles. Commun. Dev. Assem. Text. Prod. 2020, 1, 170–179. [Google Scholar]
- Paiva, A.; Ferreira, F.; Catarino, A.; Carvalho, M.; Carvalho, H. Design of smart garments for sports and rehabilitation. IOP Conf. Ser. Mater. Sci. Eng. 2018, 459, 012083. [Google Scholar] [CrossRef]
- Khundaqji, H.; Hing, W.; Furness, J.; Climstein, M. Smart shirts for monitoring physiological parameters: Scoping review. JMIR Mhealth Uhealth 2020, 8, e18092. [Google Scholar] [CrossRef]
- Scataglini, S.; Moorhead, A.P.; Feletti, F. A systematic review of smart clothing in sports: Possible applications to extreme sports. Muscles Ligaments Tendons J. 2020, 10, 333–342. [Google Scholar] [CrossRef]
- Bartlett, P.N.; Archer, P.B.M.; Ling-Chung, S.K. Conducting polymer gas sensors part I: Fabrication and characterization. Sens. Actuators 1989, 19, 125–140. [Google Scholar] [CrossRef]
- Han, J.-W.; Kim, B.S.; Li, J.; Meyyappan, M. A carbon nanotube based ammonia sensor on cotton textile. Appl. Phys. Lett. 2013, 102, 193104. [Google Scholar] [CrossRef]
- He, W.; Wang, C.; Wang, H.M.; Jian, M.Q.; Lu, W.D.; Liang, X.P.; Zhang, X.; Yang, F.C.; Zhang, Y.Y. Integrated textile sensor patch for real-time and multiplex sweat analysis. Sci. Adv. 2019, 5, eaax0649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Angelucci, A.; Cavicchioli, M.; Cintorrino, I.A.; Lauricella, G.; Rossi, C.; Strati, S.; Aliverti, A. Smart textiles and sensorized garments for physiological monitoring: A review of available solutions and techniques. Sensors 2021, 21, 814. [Google Scholar] [CrossRef] [PubMed]
- Kamarudin, S.F.; Mustapha, M.; Kim, J.-K. Green strategies to printed sensors for healthcare applications. Polymer Rev. 2021, 61, 116–156. [Google Scholar] [CrossRef]
- Meghrazi, M.A.; Tian, Y.; Mahnam, A.; Bhattachan, P.; Eskandarian, L.; Kakhki, S.T.; Popovic, M.R.; Lankarany, M. Multichannel ECG recording from waist using textile sensors. BioMed. Eng. OnLine 2020, 19, 48. [Google Scholar] [CrossRef]
- Silva, M.; Catarino, A.; Carvalho, H.; Rocha, A.; Monteiro, J.; Montagna, G. Textile sensors for ECG and respiratory frequency on swimsuits. In Proceedings of the Intelligent Textiles and Mass Customisation International Conference, Casablanca, Morocco, 12–14 November 2009; pp. 301–310. [Google Scholar]
- An, X.; Stylios, G.K. A hybrid textile electrode for electrocardiogram (ECG) measurement and motion tracking. Materials 2018, 11, 1887. [Google Scholar] [CrossRef] [Green Version]
- Arquilla, K.; Webb, A.K.; Anderson, A.P. Textile electrocardiogram (ECG) electrodes for wearable health monitoring. Sensors 2020, 20, 1013. [Google Scholar] [CrossRef] [Green Version]
- Lee, E.; Cho, G.S. PU nanoweb-based textile electrode treated with single-walled carbon nanotube/silver nanowire and its application to ECG monitoring. Smart Mater. Struct. 2019, 28, 045004. [Google Scholar] [CrossRef]
- Jourand, P.; Clercq, H.D.; Corthout, R.; Puers, R. Textile Integrated Breathing and ECG Monitoring System. Procedia Chem. 2009, 1, 722–725. [Google Scholar] [CrossRef]
- Cho, G.; Jeong, K.; Paik, M.J.; Kwun, Y.; Sung, M. Performance evaluation of textile-based electrodes and motion sensors for smart clothing. IEEE Sens. J. 2011, 11, 3183–3193. [Google Scholar] [CrossRef]
- Rai, P.; Kumar, P.S.; Oh, S.C.; Kwon, H.J.; Mathur, G.N.; Varadan, V.K.; Agarwal, M.P. Smart healthcare textile sensor system for unhindered-pervasive health monitoring. Proc. SPIE 2012, 8344, 83440E. [Google Scholar]
- Bouwstra, S.; Chen, W.; Oetomo, S.B.; Feijs, L.M.G.; Gluitmans, P.J.M. Designing for reliable textile neonatal ECG monitoring using multi-sensor recordings. In Proceedings of the 2011 Annual International Conference of the IEEE Engineering in Medicine and Biology Society, Boston, MA, USA, 30 August–3 September 2011; pp. 2488–2491. [Google Scholar]
- Wiklund, U.; Karlsson, M.; Östlund, N.; Berlin, L.; Lindecrantz, K.; Karlsson, S.; Sandsjö, L. Adaptive spatio-temporal filtering of disturbed ECGs: A multi-channel approach to heartbeat detection in smart clothing. Med. Biol. Eng. Comput. 2007, 45, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Trummer, S.; Ehrmann, A.; Büsgen, A. Development of underwear with integrated 12 channel ECG for men and women. AUTEX Res. J. 2017, 17, 344–349. [Google Scholar] [CrossRef]
- Takeshita, T.; Yoshida, M.; Takei, Y.; Ouchi, A.; Hinoki, A.; Uchida, H.; Kobayashi, T. Relationship between contact pressure and motion artifacts in ECG measurements with electrostatic flocked electrodes fabricated on textile. Sci. Rep. 2019, 9, 5897. [Google Scholar] [CrossRef]
- Lidón-Roger, J.V.; Prats-Boluda, G.; Ye-Lin, Y.Y.; Garcia-Casado, J.; Garcia-Breijo, E. Textile concentric ring electrodes for ECG recording based on screen-printing technique. Sensors 2018, 18, 300. [Google Scholar] [CrossRef] [Green Version]
- Ankhili, A.; Tao, X.Y.; Cochrane, C.; Koncar, V.; Coulon, D.; Tarlet, J.-M. Ambulatory evaluation of ECG signals obtained using washable textile-based electrodes made with chemically modified PEDOT:PSS. Sensors 2019, 19, 416. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ankhili, A.; Tao, X.Y.; Cochrane, C.; Koncar, V.; Coulon, D.; Tarlet, J.-M. Comparative study on conductive knitted fabric electrodes for long-term electrocardiography monitoring: Silver-plated and PEDOT:PSS coated fabrics. Sensors 2018, 18, 3890. [Google Scholar] [CrossRef] [Green Version]
- Wattal, S.S.; Spear, S.K.; Imtiaz, M.H.; Sazonov, E. A polypyrrole-coated textile electrode and connector for wearable ECG monitoring. In Proceedings of the 2018 IEEE International Conference on Wearable and Implantable Body Sensor Networks (BSN), Las Vegas, NV, USA, 4–7 March 2018; pp. 54–57. [Google Scholar]
- Akter Shathi, M.; Chen, M.Z.; Khoso, N.A.; Rahman, M.T.; Bhattacharjee, B. Graphene coated textile based highly flexible and washable sports bra for human health monitoring. Mater. Des. 2020, 193, 108792. [Google Scholar] [CrossRef]
- Yapici, M.K.; Aklhidir, T.; Samad, Y.A.; Liao, K. Graphene-clad textile electrodes for electrocardiogram monitoring. Sens. Actuators A Chem. 2015, 221, 1469–1474. [Google Scholar] [CrossRef]
- Xu, X.W.; Luo, M.; He, P.; Guo, X.J.; Yang, J.L. Screen printed graphene electrodes on textile for wearable electrocardiogram monitoring. Appl. Phys. A 2019, 125, 714. [Google Scholar] [CrossRef]
- Aumann, S.; Trummer, S.; Brücken, A.; Ehrmann, A.; Büsgen, A. Conceptual design of a sensory shirt for fire-fighters. Text. Res. J. 2014, 84, 1661–1665. [Google Scholar] [CrossRef]
- Schäl, P.; Junger, I.J.; Grimmelsmann, N.; Ehrmann, A. Development of graphite-based conductive textile coatings. J. Coat. Technol. Res. 2018, 15, 875–883. [Google Scholar] [CrossRef]
- Weder, M.; Hegemann, D.; Amberg, M.; Hess, M.; Boesel, L.F.; Abächerli, R.; Meyer, V.R.; Rossi, R.M. Embroidered electrode with silver/titanium coating for long-term ECG monitoring. Sensors 2015, 15, 1750–1759. [Google Scholar] [CrossRef] [Green Version]
- Nigusse, A.B.; Malengier, B.; Mengistie, D.A.; van Langenhove, L. Evaluation of silver-coated textile electrodes for ECG recording. In Proceedings of the 2021 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), Manchester, UK, 20–23 June 2021; pp. 1–4. [Google Scholar]
- Fong, E.-M.; Chung, W.-Y. A hygroscopic sensor electrode for fast stabilized non-contact ECG signal acquisition. Sensors 2015, 15, 19237–19250. [Google Scholar] [CrossRef] [Green Version]
- Majumder, S.; Chen, L.; Marinov, O.; Chen, C.-H.; Mondal, T.; Deen, M.J. Noncontact wearable wireless ECG systems for long-term monitoring. IEEE Rev. Biomed. Eng. 2018, 11, 306–321. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.-W.; Zhang, H.; Lin, S.-F. Influence of capacitive coupling on high-fidelity non-contact ECG measurement. IEEE Sens. J. 2020, 20, 9265–9273. [Google Scholar] [CrossRef]
- Mitchell, E.; Coyle, S.; O’Connor, N.; Diamond, D.; Ward, T. Breathing feedback system with wearable textile sensors. In Proceedings of the 2010 IEEE International Conference on Body Sensor Networks, Singapore, 7–9 June 2010; pp. 56–61. [Google Scholar]
- Guo, L.; Berglin, L.; Li, Y.J.; Mattila, H.; Kalantar Mehrjerdi, A.; Skrifvars, M. ‘Disappearing sensor’-textile based sensor for monitoring breathing. In Proceedings of the 2011 International Conference on Control, Automation and Systems Engineering (CASE), Singapore, 30–31 July 2011; pp. 1–4. [Google Scholar]
- Ramos-Garcia, R.I.; da Silva, F.; Kondi, Y.; Sazonov, E.; Dunne, L.E. Analysis of a coverstitched stretch sensor for monitoring of breathing. In Proceedings of the 2016 10th International Conference on Sensing Technology (ICST), Nanjing, China, 11–13 November 2016; pp. 1–6. [Google Scholar]
- Rovira, C.; Coyle, S.; Corcoran, B.; Diamond, D.; Stroiescu, F.; Daly, K. Integration of textile-based sensors and Shimmer for breathing rate and volume measurement. In Proceedings of the 2011 5th International Conference on Pervasive Computing Technologies for Healthcare (PervasiveHealth) and Workshops, Dublin, Ireland, 23–26 May 2011; pp. 238–241. [Google Scholar]
- Yang, C.-M.; Huang, W.-T.; Yang, T.-L.; Hsieh, M.-C.; Liu, C.-T. Textile digital sensors for detecting breathing frequency. In Proceedings of the 2008 5th International Summer School and Symposium on Medical Devices and Biosensors, Hong Kong, China, 1–3 June 2008; pp. 276–279. [Google Scholar]
- Zięba, J.; Frydrysiak, M. Textronics–electrical and electronic textiles. Sensors for breathing frequency measurement. Fibres Text. East. Eur. 2006, 14, 43–48. [Google Scholar]
- Zięba, J.; Frydrysiak, M.; Gniotek, K. Textronics system for breathing measurement. Fibres Text. East. Eur. 2007, 15, 105–108. [Google Scholar]
- Zięba, J.; Frydrysiak, M.; Błaszczyk, J. Textronic clothing with resistance textile sensor to monitoring frequency of human breathing. In Proceedings of the 2012 IEEE International Symposium on Medical Measurements and Applications, Budapest, Hungary, 18–19 May 2012; pp. 1–6. [Google Scholar]
- Carvalho, H.; Catarino, A.P.; Rocha, A.; Postolache, O. Health monitoring using textile sensor and electrodes: An overview and integration of technologies. In Proceedings of the 2014 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Lisboa, Portugal, 11–12 June 2014; pp. 1–6. [Google Scholar]
- Ehrmann, A.; Heimlich, F.; Brücken, A.; Weber, M.O.; Haug, R. Suitability of knitted fabrics as elongation sensors subject to structure, stitch dimension and elongation direction. Text. Res. J. 2014, 84, 2006–2012. [Google Scholar] [CrossRef]
- Furtak, N.T.; Skrzetuska, E.; Krucinska, I. Development of screen-printed breathing rate sensor. Fibres Text. East. Eur. 2013, 21, 84–88. [Google Scholar]
- Krucińska, I.; Skrzetuska, E.; Urbaniak-Domagala, W. Prototypes of carbon nanotube-based textile sensors manufactured by the screen printing method. Fibres Text. East. Eur. 2012, 20, 85–89. [Google Scholar]
- Guo, L.; Berglin, L.; Wiklund, U.; Mattila, H. Design of a garment-based sensing system for breathing monitoring. Text. Res. J. 2012, 83, 499–509. [Google Scholar] [CrossRef]
- Zeagler, C.; Gilliland, S.; Audy, S.; Starner, T. Can I wash it? The effect of washing conductive materials used in making textile based wearable electronic interfaces. In Proceedings of the 2013 International Symposium on Wearable Computers, Zurich, Switzerland, 9–12 September 2013; pp. 143–144. [Google Scholar]
- Berglund, M.E.; Coughlin, J.; Gioberto, G.; Dunne, L.E. Washability of E-Textile Stretch Sensors and Sensor Insulation. In Proceedings of the 2014 ACM International Symposium on Wearable Computers; ACM: New York, NY, USA, 2014; pp. 127–128. [Google Scholar]
- Kang, T.-H.; Merritt, C.; Karaguzel, B.; Wilson, J.; Franzon, P.; Pourdeyhimi, B.; Grant, E.; Nagle, T. Sensors on textile substrates for home-based healthcare monitoring. In Proceedings of the 1st Transdisciplinary conference on Distributed Diagnosis and Home Healthcare, Arlington, VA, USA, 2–4 April 2006; pp. 5–7. [Google Scholar]
- Min, S.D.; Yun, Y.Y.; Shin, H.S. Simplified structural textile respiration sensor based on capacitive pressure sensing method. IEEE Sens. J. 2014, 14, 3245–3251. [Google Scholar]
- Teichmann, D.; Foussier, J.; Buscher, M.; Walter, M.; Leonhardt, S. Textile integration of a magnetic induction sensor for monitoring of cardiorespiratory activity. In Proceedings of the World Congress on Medical Physics and Biomedical Engineering, Beijing, China, 26–31 May 2012. [Google Scholar]
- Yang, X.F.; Chen, Z.H.; Ming Elvin, C.S.; Ying Janice, L.H.; Ng, S.H.; Teo, J.T.; Wu, R.F. Textile fiber optic microbend sensor used for heartbeat and respiration monitoring. IEEE Sens. J. 2015, 15, 757–761. [Google Scholar] [CrossRef]
- Yang, C.M.; Yang, T.L.; Wu, C.C.; Hung, S.H.; Liao, M.H.; Su, M.J.; Hsieh, H.C. Textile-based capacitive sensor for a wireless wearable breath monitoring system. In Proceedings of the 2014 IEEE International Conference on Consumer Electronics (ICCE), Las Vegas, NV, USA, 10–13 January 2014; pp. 232–233. [Google Scholar]
- Agcayazi, T.; Yokus, M.A.; Gordon, M.; Ghosh, T.; Bozkurt, A. A stitched textile-based capacitive respiration sensor. In Proceedings of the 2017 IEEE Sensors, Glasgow, UK, 29 October–1 November 2017; pp. 1–3. [Google Scholar]
- Raiano, L.; di Tocco, J.; Massaroni, C.; di Pino, G.; Schena, E.; Formica, D. Clean-breathing: A novel sensor fusion algorithm based on ICA to remove motion artifacts from breathing signal. In Proceedings of the 2020 IEEE International Workshop on Metrology for Industry 4.0 & IoT, Roma, Italy, 3–5 June 2020; pp. 734–739. [Google Scholar]
- Carbonaro, N.; Laurino, M.; Arcarisi, L.; Menicuddi, D.; Gemignani, A.; Tognetti, A. Textile-based pressure sensing matrix for in-bed monitoring of subject sleeping posture and breathing activity. Appl. Sci. 2021, 11, 2552. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, M.J.; Hu, X.L.; Yu, L.M.; Fan, X.H.; Huang, C.S.; Li, Y.L. Graphdiyne-based flexible respiration sensors for monitoring human health. NanoToday 2021, 39, 101214. [Google Scholar] [CrossRef]
- Atalay, O.; Kennon, W.R.; Demirok, E. Weft-knitted strain sensor for monitoring respiratory rate and its electro-mechanical modeling. IEEE Sens. J. 2015, 15, 110–122. [Google Scholar] [CrossRef]
- Bahadir, S.K. Identification and modeling of sensing capability of force sensing resistor integrated to e-textile structure. IEEE Sens. J. 2018, 18, 9770–9780. [Google Scholar] [CrossRef]
- Ejupi, A.; Ferrone, A.; Menon, C. Quantification of textile-based stretch sensors using machine learning: An exploratory study. In Proceedings of the 2018 7th IEEE International conference on Biomedical Robotics and Biomechatronics (Biorob), Enschede, The Netherlands, 26–29 August 2018; pp. 254–259. [Google Scholar]
- Zhang, H.S.; Zhang, L.Q.; Li, G.L. Using textile electrode EMG for prosthetic movement identification in transradial amputees. In Proceedings of the 2013 IEEE International Conference on Body Sensor Networks, Cambridge, MA, USA, 6–9 May 2013; pp. 1–5. [Google Scholar]
- Niijima, A.; Isezaki, T.; Aoki, R.; Watanabe, T.; Yamada, T. hitoeCap: Wearable EMG sensor for monitoring masticatory muscles with PEDOT-PSS textile electrodes. In Proceedings of the 2017 ACM International Symposium on Wearable Computers, Maui Hawaii, HI, USA, 11–15 September 2017; pp. 215–220. [Google Scholar]
- Di Giminiani, R.; Lancia, S.; Ferrari, M.; Quaresima, V.; Tilma Vistisen, H.; Kliltgaard, A.; Arbjerg Heick, R.; Oestergard, K.; Yndgaard Soerensen, K.; Cardinale, M. A wearable integrated textile EMG and muscle oximetry system for monitoring exercise-induced effects: A feasibility study. In Proceedings of the 2018 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Rome, Italy, 11–13 June 2018; pp. 1–5. [Google Scholar]
- Paul, G.M.; Cao, F.; Torah, R.; Yang, K.; Beeby, S.; Tudor, J. A smart textile based facial EMG and EOG computer interface. IEEE Sens. J. 2014, 14, 393–400. [Google Scholar] [CrossRef]
- Linz, T.; Gourmelon, L.; Langereis, G. Contactless EMG sensors embroidered onto textile. In 4th International Workshop on Wearable and Implantable Body Sensor Networks (BSN 2007); Leonhardt, S., Falck, T., Mähönen, P., Eds.; IFMBE Proceedings; Springer: Berlin/Heidelberg, Germany, 2007; Volume 13, pp. 29–34. [Google Scholar]
- Taelman, J.; Adriaensen, T.; van Der Horst, C.; Linz, T.; Spaepen, A. Textile integrated contactless EMG sensing for stress analysis. In Proceedings of the 2007 29th Annual International conference of the IEEE Engineering in Medicine and Biology Society, Lyon, France, 22–26 August 2007; pp. 3966–3969. [Google Scholar]
- Pani, D.; Achilli, A.; Spanu, A.; Bonfiglio, A.; Gazzoni, M.; Botter, A. Validation of polymer-based screen-printed textile electrodes for surface EMG detection. IEEE Trans. Neural Syst. Rehabil. Eng. 2019, 27, 1370–1377. [Google Scholar] [CrossRef]
- Finni, T.; Hu, M.; Kettunen, P.; Vilavuo, T.; Cheng, S. Measurement of EMG activity with textile electrodes embedded into clothing. Physiol. Meas. 2007, 28, 1405–1419. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Lee, S.J.; Jeong, W.Y. EMG measurement with textile-based electrodes in different electrode sizes and clothing pressures for smart clothing design optimization. Polymers 2020, 12, 2406. [Google Scholar] [CrossRef]
- Lorussi, F.; Carbonaro, N.; de Rossi, D.; Paradiso, R.; Veltink, P.; Tognetti, A. Wearable textile platform for assessing stroke patient treatment in daily life conditions. Front. Bioeng. Biotechnol. 2016, 4, 28. [Google Scholar] [CrossRef] [PubMed]
- Di Giminiani, R.; Cardinale, M.; Ferrari, M.; Quaresima, V. Validation of fabric-based thigh-wearable EMG sensors and oximetry for monitoring quadriceps activity during strength and endurance exercises. Sensors 2020, 20, 4664. [Google Scholar] [CrossRef]
- Löfhede, J.; Seoane, F.; Thordstein, M. Soft textile electrodes for EEG monitoring. In Proceedings of the 10th IEEE International Conference on Information Technology and Applications in Biomedicine, Corfu, Greece, 3–5 November 2010; pp. 1–4. [Google Scholar]
- Löfhede, J.; Seoane, F.; Thordstein, M. Textile electrodes for EEG recording–a pilot study. Sensors 2012, 12, 16907–16919. [Google Scholar] [CrossRef] [PubMed]
- Reis Carneiro, M.; de Almeida, A.T.; Tavakoli, M. Wearable and comfortable e-textile headband for long-term acquisition of forehead EEG signals. IEEE Sens. J. 2020, 20, 15107–15116. [Google Scholar] [CrossRef]
- Shu, L.; Xu, T.Y.; Xu, X.M. Mulilayer sweat-absorbable textile electrode for EEG measurement in forehead site. IEEE Sens. J. 2019, 19, 5995–6005. [Google Scholar] [CrossRef]
- Sahi, A.; Rai, P.; Oh, S.C.; Ramasamy, M.; Harbauth, R.E.; Varadan, V.K. Neural activity based biofeedback therapy for Autism spectrum disorder through wearable wireless textile EEG monitoring system. Proc. SPIE 2014, 9060, 90600D. [Google Scholar]
- Nano-bio-textile sensors with mobile wireless platform for wearable health monitoring of neurological and cardiovascular disorders. J. Electrochem. Soc. 2013, 161, B3116.
- Asl, S.N.; Ludwig, F.; Schilling, M. Nose properties of textile, capacitive EEG electrodes. Curr. Dir. Biomed. Eng. 2015, 1, 34–37. [Google Scholar] [CrossRef]
- Fleury, A.; Alizadeh, M.; Stefan, G.; Chau, T. Toward fabric-based EEG access technologies: Seamless knit electrodes for a portable brain-computer interface. In Proceedings of the 2017 IEEE Life Sciences Conference, Sydney, NSW, Australia, 13–15 December 2017; pp. 35–38. [Google Scholar]
- Tseghai, G.B.; Malengier, B.; Fante, K.A.; van Langenhove, L. A dry EEG textrode from a PEDOT:PSS/PDMS-coated cotton fabric for brain activity monitoring. In Proceedings of the 2021 IEEE International Conference on Flexible and Printable Sensors and Systems (FLEPS), Manchester, UK, 20–23 June 2021; pp. 1–4. [Google Scholar]
- Tseghai, G.B.; Malengier, B.; Fante, K.A.; van Langenhove, L. A long-lasting textile-based anatomically realistic head phantom for validation of EEG electrodes. Sensors 2021, 21, 4658. [Google Scholar] [CrossRef]
- Tseghai, G.B.; Malengier, B.; Fante, K.A.; van Langenhove, L. The status of textile-based dry EEG electrodes. AUTEX Res. J. 2021, 21, 63–70. [Google Scholar] [CrossRef]
- Medrano, G.; Ubl, A.; Zimmermann, N.; Gries, T.; Leonhardt, S. Skin electrode impedance of textile electrodes for bioimpedance spectroscopy. In 13th International Conference on Electrical Bioimpedance and the 8th Conference on Electrical Impedance Tomography; Scharfetter, H., Merwa, R., Eds.; IFMBE Proceedings; Springer: Berlin/Heidelberg, Germany, 2007; Volume 17, pp. 260–263. [Google Scholar]
- Márquez, J.C.; Seoane, F.; Välimäki, E.; Lindecrantz, K. Comparison of dry-textile electrodes for electrical bioimpedance spectroscopy measurements. J. Phys. Conf. Ser. 2010, 224, 012140. [Google Scholar] [CrossRef] [Green Version]
- Márquez, J.C.; Seoane, F.; Välimäki, E.; Lindecrantz, K. Textrode-enabled transthoracic electrical bioimpedance measurements –towards wearable applications of impedance cardiography. J. Electr. Bioimpedance 2013, 4, 45–50. [Google Scholar] [CrossRef] [Green Version]
- Márquez, J.C.; Seoane, F.; Lindecrantz, K. Textrode functional straps for bioimpedance measurements–experimental results for body composition analysis. Eur. J. Clin. Nutr. 2013, 67, S22–S27. [Google Scholar] [CrossRef]
- Ferreira, J.; Pau, I.; Lindecrantz, K.; Seoane, F. A handheld and textile-enabled bioimpedance system for ubiquitous body composition analysis. An initial functional validation. IEEE J. Biomed. Health Inform. 2017, 21, 1224–1232. [Google Scholar] [CrossRef] [PubMed]
- Meding, J.T.; Tuvshinbayar, K.; Döpke, C.; Tamoue, F. Textile electrodes for bioimpedance measuring. Commun. Dev. Assem. Text. Prod. 2021, 2, 49–60. [Google Scholar]
- Jose, M.; Lemmens, M.; Bormans, S.; Thoelen, R.; Deferme, W. Fully printed, stretchable und wearable bioimpedance sensor on textiles for tomography. Flex. Print. Electron. 2021, 6, 015010. [Google Scholar] [CrossRef]
- Medrano, G.; Beckmann, L.; Zimmermann, N.; Grundmann, T.; Gries, T.; Leonhardt, S. Bioimpedance spectroscopy with textile electrodes for a continous monitoring application. In 4th International Workshop on Wearable and Implantable Body Sensor Networks (BSN 2007); Leonhardt, S., Falck, T., Mähönen, P., Eds.; IFMBE Proceedings; Springer: Berlin/Heidelberg, Germany, 2007; Volume 13, pp. 23–28. [Google Scholar]
- Posada-Quintero, H.F.; Reljin, N.; Eaton-Robb, C.; Noh, Y.S.; Riistama, J.; Chon, K.H. Analysis of consistency of transthoracic bioimpedance measurements acquired with dry carbon black PDMS electrodes, adhesive electrodes, and wet textile electrodes. Sensors 2018, 18, 1719. [Google Scholar] [CrossRef] [Green Version]
- Corchia, L.; Monti, G.; Raheli, F.; Candelieri, G.; Tarricone, L. Dry textile electrodes for wearable bio-impedance analyzers. IEEE Sens. J. 2020, 20, 6139–6147. [Google Scholar] [CrossRef]
- Ulbrich, M.; Lüken, M.; Mühlsteff, J.; Leonhardt, S. Wearable bioimpedance systems for home-care monitoring using BSNs. In Wearable Sensors, 2nd ed.; Academic Press: Cambridge, MA, USA, 2021; pp. 519–540. [Google Scholar]
- Pavlin, M.; Novak, F.; Papa, G. Low power contactless bioimpedance sensor for monitoring breathing activity. Sensors 2021, 21, 2081. [Google Scholar] [CrossRef]
- Hughes-Riley, T.; Lugoda, P.; Dias, T.; Trabi, C.L.; Morris, R.H. A study of thermistor performance within a textile structure. Sensors 2017, 17, 1804. [Google Scholar] [CrossRef] [Green Version]
- Lugoda, P.; Hughes-Riley, T.; Oliveira, C.; Morris, R.; Dias, T. Developing novel temperature sensing garments for health monitoring applications. Fibers 2018, 6, 46. [Google Scholar] [CrossRef] [Green Version]
- Lugoda, P.; Costa, J.C.; Oliveira, C.; Garcia-Garcia, L.A.; Wickramasinghe, S.D.; Pouryazdan, A.; Roggen, D.; Dias, T.; Münzenrieder, N. Flexible temperature sensor integration into e-textiles using different industrial yarn fabrication processes. Sensors 2020, 20, 73. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, Q.; Chen, H.; Ran, Z.-Y.; Zhang, L.-N.; Xiang, R.-F.; Wang, X.; Tao, X.-M.; Ding, X. Full fabric sensing network with large deformation for continuous detection of skin temperature. Smart Mater. Struct. 2018, 27, 105017. [Google Scholar] [CrossRef]
- Wicaksoni, I.; Tucker, C.I.; Sun, T.; Guerrero, C.A.; Liu, C.; Wo, W.M.; Pence, E.J.; Dagdeviren, C. A tailored, electronic textile conformable suit for large-scale spatiotemporal physiological sensing in vivo. Npj Flex. Electron. 2020, 4, 5. [Google Scholar] [CrossRef] [Green Version]
- Trung, T.Q.; Dang, T.M.L.; Ramasundaram, S.; Toi, P.T.; Park, S.Y.; Lee, N.-E. A stretchable strain-insensitive temperature sensor based on free-standing elastomeric composite fibers for on-body monitoring of skin temperature. ACS Appl. Mater. Interfaces 2019, 11, 2317–2327. [Google Scholar] [CrossRef] [PubMed]
- Lugoda, P.; Hughes-Riley, T.; Morris, R.; Dias, T. A wearable textile thermograph. Sensors 2018, 18, 2369. [Google Scholar] [CrossRef] [Green Version]
- Trung, T.Q.; Le, H.S.; Dang, T.M.L.; Ju, S.H.; Park, S.Y.; Lee, N.-E. Freestanding, fiber-based, wearable temperature sensor with tunable thermal index for healthcare monitoring. Adv. Healthc. Mater. 2018, 7, 1800074. [Google Scholar] [CrossRef]
- Wu, R.H.; Ma, L.Y.; Hou, C.; Meng, Z.H.; Guo, W.X.; Yu, W.D.; Yu, R.; Hu, F.; Liu, X.Y. Silk composite electronic textile sensor for high space precision 2D combo temperature-pressure sensing. Small 2019, 15, 1901558. [Google Scholar] [CrossRef]
- Hughes-Riley, T.; Jobling, P.; Dias, T.; Faulkner, S.H. An investigation of temperature-sensing textiles for temperature monitoring during sub-maximal cycling trials. Text. Res. J. 2021, 91, 624–645. [Google Scholar] [CrossRef]
- Wang, Y.B.; Zhu, M.M.; Wie, X.D.; Yu, J.Y.; Li, Z.L.; Ding, B. A dual-mode electronic skin textile for pressure and temperature sensing. Chem. Eng. J. 2021, 425, 130599. [Google Scholar] [CrossRef]
- Chen, X.C.; He, H.; Khan, Z.; Sydanheimo, L.; Ukkonen, L.; Virkki, J. Textile-based batteryless moisture sensor. IEEE Antennas Wirel. Propag. Lett. 2020, 19, 198–202. [Google Scholar] [CrossRef]
- Chen, X.C.; He, H.; Khan, Z.; Sydänheimo, L.; Ukkonen, L.; Virkki, J. Design, fabrication and wireless evaluation of a passive 3D-printed moisture sensor on a textile substrate. In Proceedings of the 2019 Photonics & Electromagnetics Research Symposium–Spring (PIERS-Spring), Rome, Italy, 17–20 June 2019; pp. 1027–1030. [Google Scholar]
- Grethe, T.; Borczyk, S.; Plenkmann, K.; Normann, M.; Rabe, M.; Schwarz-Pfeiffer, A. Textile humidity sensor. In Proceedings of the 2018 Symposium on Design, Test, Integration & Packaging of MEMS and MOEMS (DTIP), Rome, Italy, 22–25 May 2018; pp. 1–3. [Google Scholar]
- Gaubert, V.; Gidik, H.; Koncar, V. Boxer underwear incorporating textile moisture sensor to prevent nocturnal enuresis. Sensors 2020, 20, 3546. [Google Scholar] [CrossRef]
- Tanaka, M.; Utsunomiya, F.; Douseki, T. Wearable self-powered diaper-shaped urinary-incontinence sensor suppressing response-time variation with 0.3 V start-up converter. IEEE Sens. J. 2016, 16, 3472–3479. [Google Scholar] [CrossRef]
- Sudo, M.; Utsunomiya, F.; Tanaka, M.; Douseki, T. Temperature-robuts 0.48-V FD-SOI intermittent startup circuit with 300-nA quiescent current for batteryless wireless sensor capable of 1-µA energy harvesting sources. IEICE Trans. 2021, E104-A, 506–515. [Google Scholar] [CrossRef]
- Pereira, T.; Silva, P.; Carvalho, H.; Carvalho, M. Textile moisture sensor matrix for monitoring of disabled and bed-rest patients. In Proceedings of the 2011 IEEE Eurocon–International Conference on Computer as a Tool, Lisbon, Portugal, 27–29 April 2011; pp. 1–4. [Google Scholar]
- Devaux, E.; Aubry, C.; Campagne, C.; Rochery, M. PLA/carbon nanotubes multifilament yarns for relative humidity textile sensor. J. Eng. Fibers Fabr. 2011, 6, 13–24. [Google Scholar] [CrossRef] [Green Version]
- Mecnika, V.; Hoerr, M.; Jockenhoefel, S.; Gries, T.; Krievins, I.; Schwarz-Pfeiffer, A. Preliminary study on textile humidity sensors. In Proceedings of the Smart SysTech 2015–European Conference on Smart Objects, Systems and Technologies, Aachen, Germany, 16–17 July 2015; pp. 1–9. [Google Scholar]
- Wendler, J.; Maraite, D.; Nocke, A.; Cherif, C. Novel textile moisture sensors based on multi-layered braiding constructions. Text. Res. J. 2020, 90, 469–477. [Google Scholar] [CrossRef]
- Banitaba, S.N.; Ehrmann, A. Application of electrospun nanofibers for fabrication of versatile and highly efficient electrochemical devices: A review. Polymers 2021, 13, 1741. [Google Scholar] [CrossRef] [PubMed]
- Coyle, S.; Morris, D.; Lau, K.-T.; Diamond, D.; Taccini, N.; Costanzo, D.; Salvo, P.; di Francesco, F.; Trivella, M.G.; Porchet, J.-A.; et al. Textile sensors to measure sweat pH and sweat-rate during exercise. In Proceedings of the 2009 3rd International Conference on Pervasive Computing Technologies for Healthcare, London, UK, 1–3 April 2009; pp. 1–6. [Google Scholar]
- Possanzini, L.; Decataldo, F.; Mariani, F.; Gualandi, I.; Tessarolo, M.; Scavetta, E.; Fraboni, B. Textile sensors platform for the selective and simultaneous detection of chloride ion and pH in sweat. Sci. Rep. 2020, 10, 17180. [Google Scholar] [CrossRef]
- Promphet, N.; Rattanawaleedirojn, P.; Siralertmukul, K.; Soatthiyanon, N.; Potiyaraj, P.; Thanawattano, C.; Hinestroza, J.P.; Rodthongkum, N. Non-invasive textile based colorimetric sensor for the simultaneous detection of sweat pH and lactate. Talanta 2019, 192, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Caldara, M.; Colleoni, C.; Guido, E.; Re, V.; Rosace, G. Optical monitoring of sweat pH by a textile fabric wearable sensor based on covalently bonded litmus- 3-glycidoxypropyltrimethoxysilane coating. Sens. Actuators B Chem. 2016, 222, 213–220. [Google Scholar] [CrossRef]
- Morris, D.; Coyle, S.; Wu, Y.Z.; Lau, K.T.; Wallace, G.; Diamond, D. Bio-sensing textile based patch with integrated optical detection system for sweat monitoring. Sens. Actuators B Chem. 2009, 139, 231–236. [Google Scholar] [CrossRef] [Green Version]
- He, J.; Xiao, G.; Chen, X.D.; Qiao, Y.; Xu, D.; Lu, Z.S. A thermoresponsive microfluidic system integrating a shape memory-modified textile and a paper-based colorimetric sensor for the detection of glucose in human sweat. RSC Adv. 2019, 9, 23957–23963. [Google Scholar] [CrossRef] [Green Version]
- Siripongpreda, T.; Somchob, B.; Rodthongkum, N.; Hoven, V.P. Bacterial cellulose-based re-swellable hydrogel: Facile preparation and it potential application as colorimetric sensor of sweat pH and glucose. Carbohydr. Polym. 2021, 256, 117506. [Google Scholar] [CrossRef]
- Zhou, Y.; Han, T.; Naw, N.P.P.; Lammy, A.V.; Goh, C.H.; Boujday, S.; Steele, T.W.J. Real-time colorimetric hydration sensor for sport activities. Mater. Des. 2016, 90, 1181–1185. [Google Scholar] [CrossRef]
- Venkatesan, M.; Veeramuthu, L.; Liang, F.-C.; Chen, W.-C.; Cho, C.-J.; Chen, C.-W.; Chen, J.-Y.; Yan, Y.; Chang, S.-H.; Kuo, C.-C. Evolution of electrospun nanofibers fluorescent and colorimetric sensors for environmental toxicants, pH, temperature, and cancer cells–a review with insights on applications. Chem. Eng. J. 2020, 397, 125431. [Google Scholar] [CrossRef]
- Gualandi, I.; Tessarolo, M.; Mariani, F.; Possanzini, L.; Scavetta, E.; Fraboni, B. Textile chemical sensors based on conductive polymers for the analysis of sweat. Polymers 2021, 13, 894. [Google Scholar] [CrossRef]
- Terse-Thakoor, T.; Punjiya, M.; Matharu, Z.; Lyu, B.; Ahmad, M.; Giles, G.E.; Owyeung, R.; Alaimo, F.; Baghini, M.S.; Brunyé, T.T.; et al. Thread-based multiplexed sensor patch for real-time sweat monitoring. Npj Flex. Electron. 2020, 4, 18. [Google Scholar] [CrossRef]
- Coppedè, N.; Giannetto, M.; Villani, M.; Lucchini, V.; Battista, E.; Careri, M.; Zappettini, A. Ion selective textile organic electrochemical transistor for wearable sweat monitoring. Org. Electron. 2020, 78, 105579. [Google Scholar] [CrossRef]
- Gualandi, I.; Marzocchi, M.; Achilli, A.; Cavedale, D.; Bonfiglio, A.; Fraboni, B. Textile organic electrochemical transistors as a platform for wearable biosensors. Sci. Rep. 2016, 6, 33637. [Google Scholar] [CrossRef] [PubMed]
- Jose, M.; Oudebrouckx, G.; Bormans, S.; Veske, P.; Thoelen, R.; Deferme, W. Monitoring body fluids in textiles: Combining impedance and thermal principles in a printed, wearable, and washable sensor. ACS Sens. 2021, 6, 896–907. [Google Scholar] [CrossRef] [PubMed]
- Hexoskin. Available online: https://www.hexoskin.com/ (accessed on 15 August 2021).
- Ambiotex. Available online: https://www.ambiotex.com/ (accessed on 15 August 2021).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Blachowicz, T.; Ehrmann, G.; Ehrmann, A. Textile-Based Sensors for Biosignal Detection and Monitoring. Sensors 2021, 21, 6042. https://doi.org/10.3390/s21186042
Blachowicz T, Ehrmann G, Ehrmann A. Textile-Based Sensors for Biosignal Detection and Monitoring. Sensors. 2021; 21(18):6042. https://doi.org/10.3390/s21186042
Chicago/Turabian StyleBlachowicz, Tomasz, Guido Ehrmann, and Andrea Ehrmann. 2021. "Textile-Based Sensors for Biosignal Detection and Monitoring" Sensors 21, no. 18: 6042. https://doi.org/10.3390/s21186042
APA StyleBlachowicz, T., Ehrmann, G., & Ehrmann, A. (2021). Textile-Based Sensors for Biosignal Detection and Monitoring. Sensors, 21(18), 6042. https://doi.org/10.3390/s21186042







