Lung Auscultation Using the Smartphone—Feasibility Study in Real-World Clinical Practice
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design
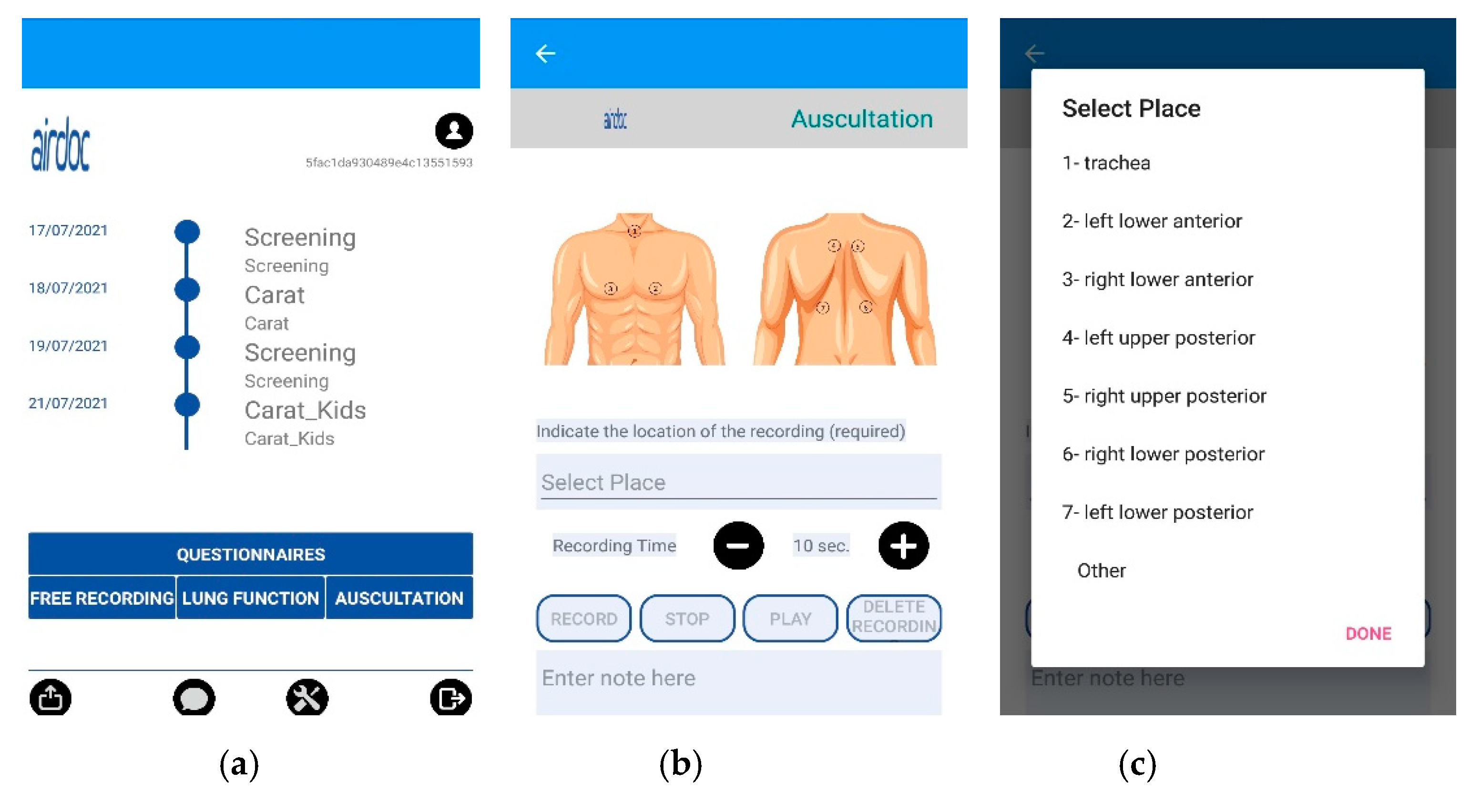
2.2. AIRDOC Mobile Application (App)
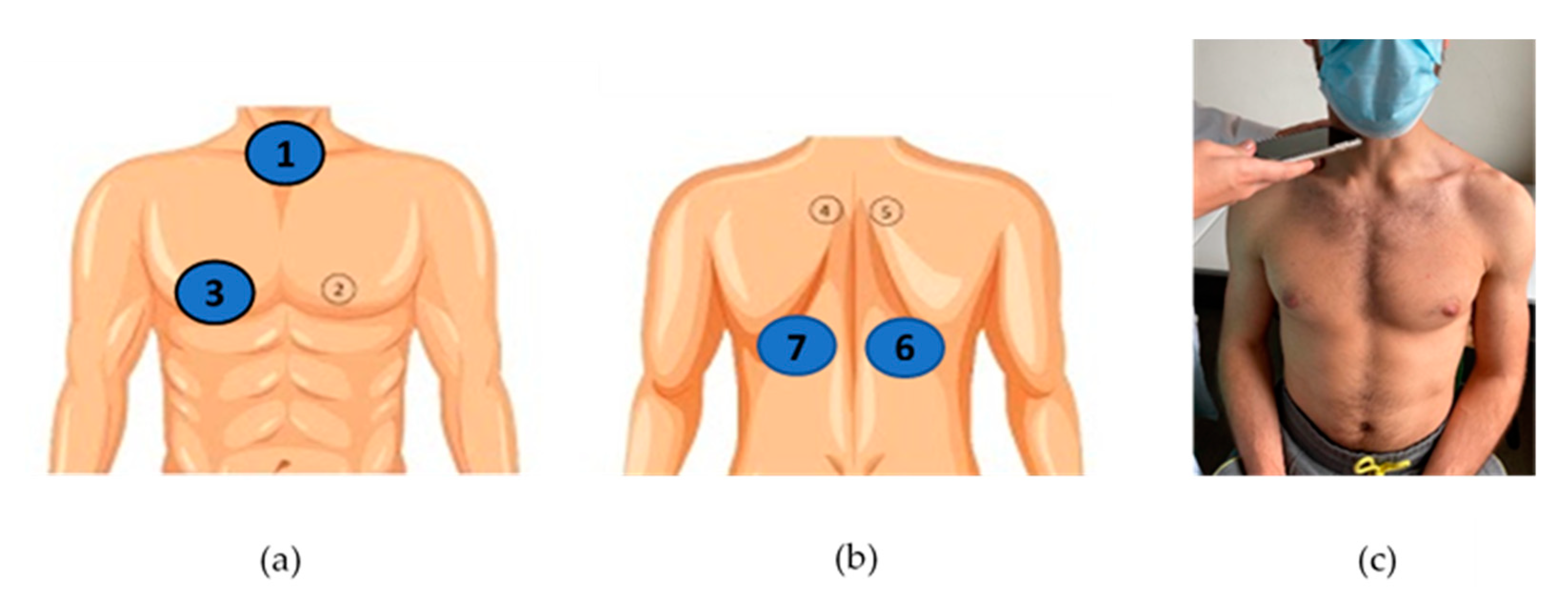
2.3. Data Collection
2.4. Lung Sound Recording Classification
2.5. Data Analysis
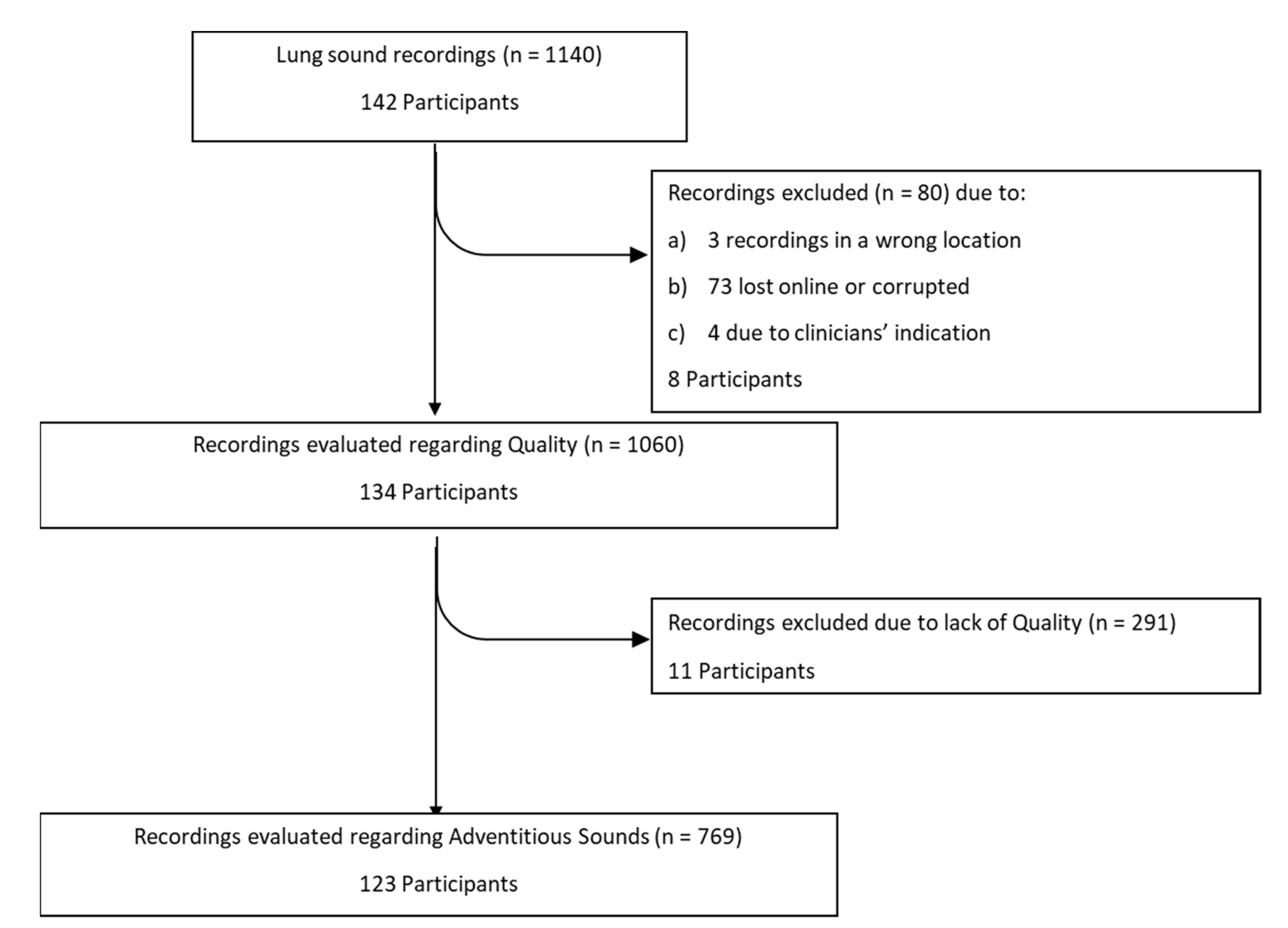
3. Results
3.1. Participants’ Characteristics
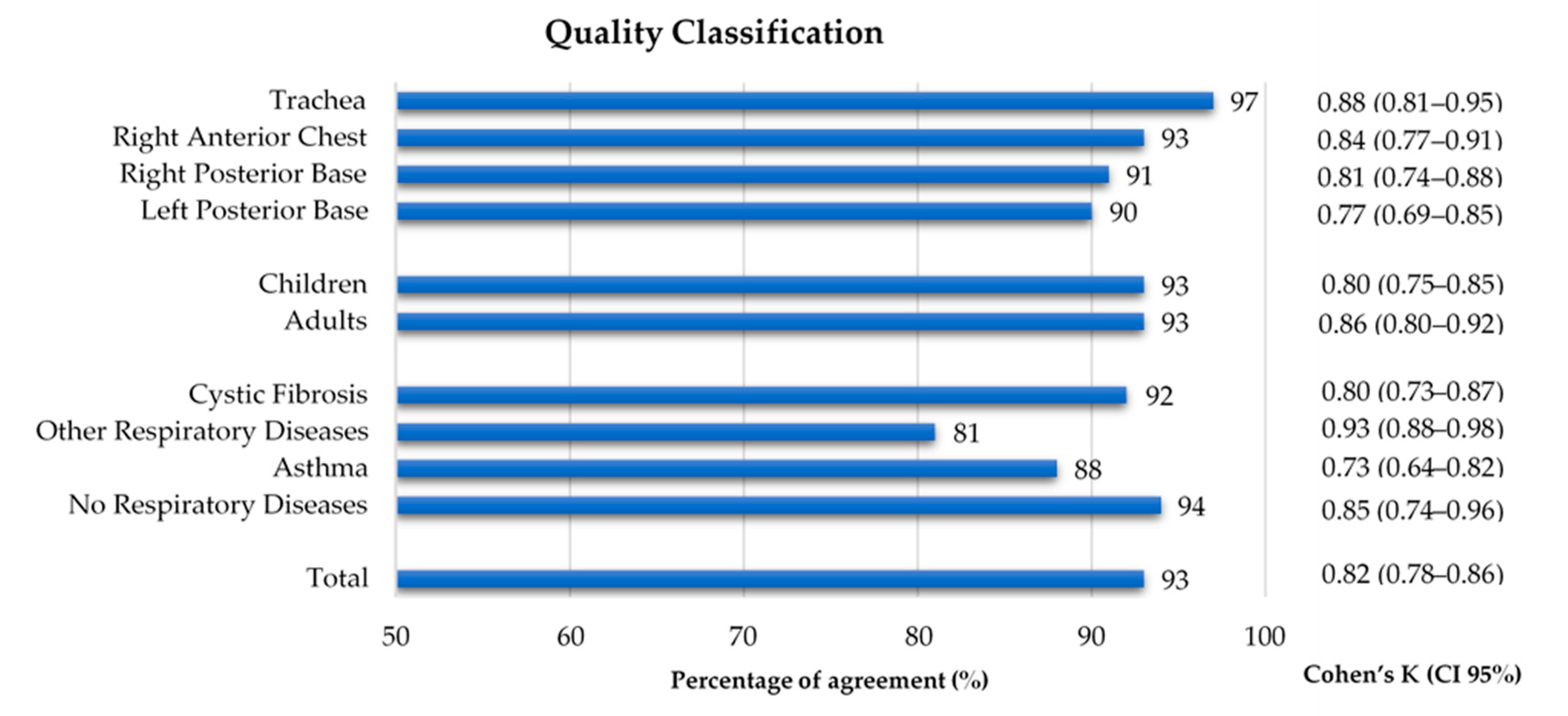
3.2. Quality Classification Agreement
3.3. Proportion of Quality
3.4. Adventitious Sounds Classification Agreement
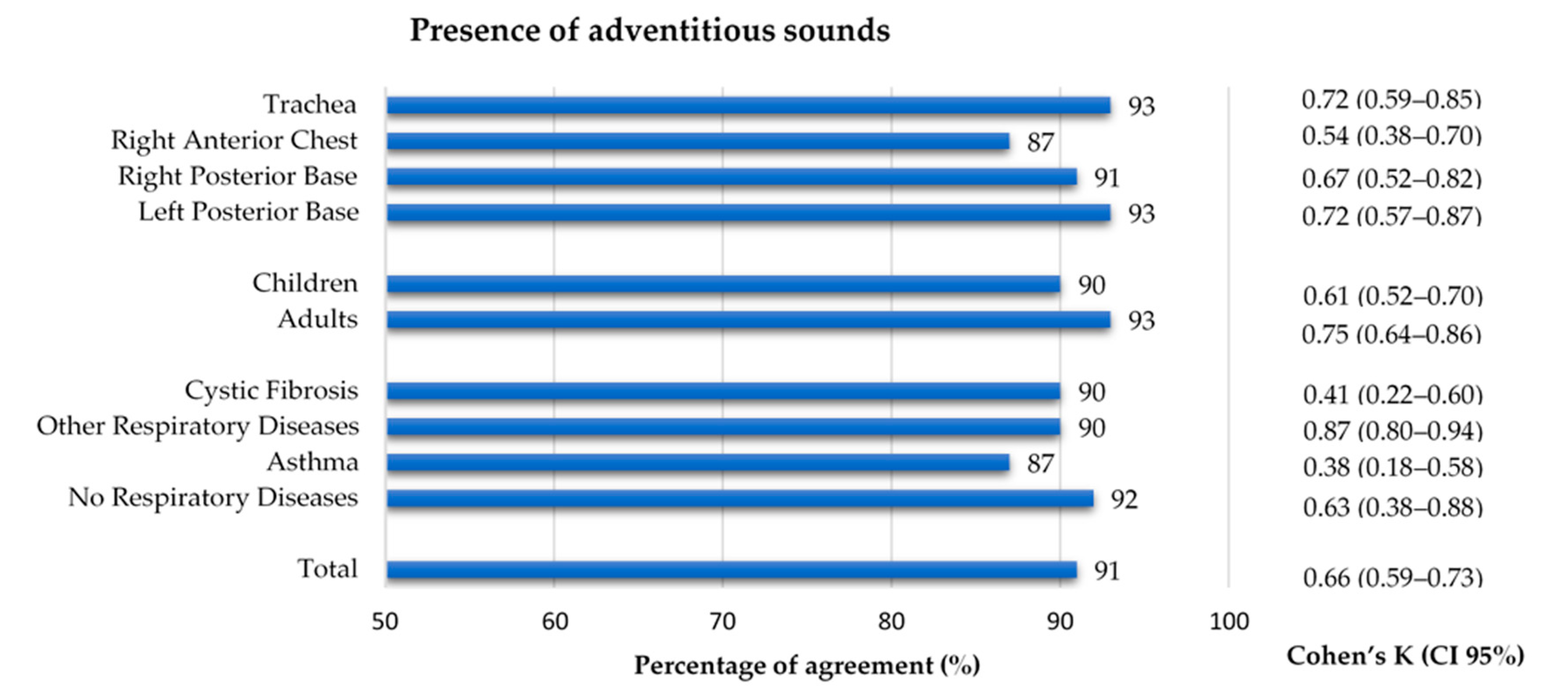
3.5. Proportion of Adventitious Sounds
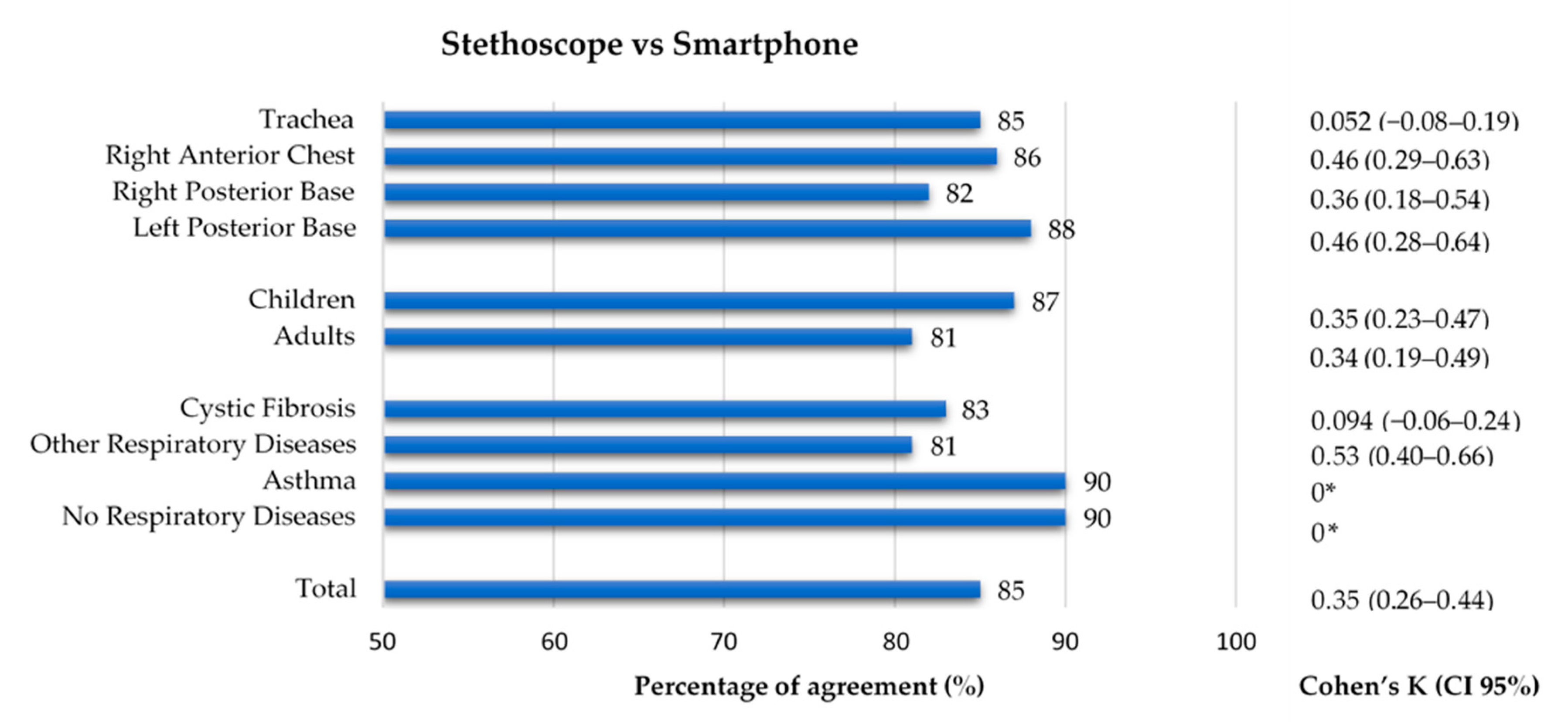
3.6. Conventional and Smartphone Lung Auscultation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- European Respiratory Society. 2017 Forum of International Respiratory Societies the Global Impact of Respiratory Disease. 2017, pp. 1–43. Available online: https://www.who.int/gard/publications/The_Global_Impact_of_Respiratory_Disease.pdf (accessed on 1 March 2021).
- Gibson, G.J.; Loddenkemper, R.; Lundbäck, B.; Sibille, Y. Respiratory health and disease in Europe: The new European Lung White Book. Eur. Respir. J. 2013, 42, 559–563. [Google Scholar] [CrossRef]
- Soriano, J.B.; Kendrick, P.J.; Paulson, K.R.; Gupta, V.; Abrams, E.M.; Adedoyin, R.A.; Adhikari, T.B.; Advani, S.M.; Agrawal, A.; Ahmadian, E.; et al. Prevalence and attributable health burden of chronic respiratory diseases, 1990–2017: A systematic analysis for the Global Burden of Disease Study 2017. Lancet Respir. Med. 2020, 8, 585–596. [Google Scholar] [CrossRef]
- WHO. Global Asthma Network the Global Asthma Report; WHO: Geneva, Switzerland, 2018; ISBN 9780473465230. Available online: http://globalasthmareport.org/ (accessed on 1 March 2021).
- Ferreira de Magalhães, M.; Amaral, R.; Pereira, A.M.; Sá-Sousa, A.; Azevedo, I.; Azevedo, L.F.; Fonseca, J.A. Cost of asthma in children: A nationwide, population-based, cost-of-illness study. Pediatr. Allergy Immunol. 2017, 28, 683–691. [Google Scholar] [CrossRef]
- Barbosa, J.P.; Ferreira-Magalhães, M.; Sá-Sousa, A.; Azevedo, L.F.; Fonseca, J.A. Cost of asthma in Portuguese adults: A population-based, cost-of-illness study. Rev. Port. Pneumol. (Engl. Ed.) 2017, 23, 323–330. [Google Scholar] [CrossRef]
- Scotet, V.; L’hostis, C.; Férec, C. The changing epidemiology of cystic fibrosis: Incidence, survival and impact of the CFTRGene discovery. Genes 2020, 11, 589. [Google Scholar] [CrossRef]
- Kelly, J. Environmental scan of cystic fibrosis research worldwide. J. Cyst. Fibros. 2017, 16, 367–370. [Google Scholar] [CrossRef]
- Schwarz, C.; Staab, D. Zystische Fibrose und ihre Komplikationen. Der Internist 2015, 56, 263–274. [Google Scholar] [CrossRef]
- Van Gool, K.; Norman, R.; Delatycki, M.B.; Hall, J.; Massie, J. Understanding the costs of care for cystic fibrosis: An analysis by age and health state. Value Health 2013, 16, 345–355. [Google Scholar] [CrossRef]
- Bilton, D.; Canny, G.; Conway, S.; Dumcius, S.; Hjelte, L.; Proesmans, M.; Tümmler, B.; Vavrova, V.; De Boeck, K. Pulmonary exacerbation: Towards a definition for use in clinical trials. Report from the EuroCareCF Working Group on outcome parameters in clinical trials. J. Cyst. Fibros. 2011, 10. [Google Scholar] [CrossRef]
- Bohadana, A.; Izbicki, G.; Kraman, S.S. Fundamentals of lung auscultation. N. Engl. J. Med. 2014, 370, 744–751. [Google Scholar] [CrossRef] [PubMed]
- Arkar, M.; Madabhavi, I.; Niranjan, N.; Dogra, M. Auscultation of the respiratory system. Ann. Thorac. Med. 2015, 10, 158–168. [Google Scholar] [CrossRef]
- Tabatabaei, S.A.H.; Fischer, P.; Schneider, H.; Koehler, U.; Gross, V.; Sohrabi, K. Methods for Adventitious Respiratory Sound Analyzing Applications Based on Smartphones: A Survey. IEEE Rev. Biomed. Eng. 2020, 14, 98–115. [Google Scholar] [CrossRef] [PubMed]
- Global Initiative for Asthma Global Strategy for Asthma Management and Prevention Updated 2021. Available online: www.ginasthma.org (accessed on 1 March 2021).
- Regelmann, W.E.; Schechter, M.S.; Wagener, J.S.; Morgan, W.J.; Pasta, D.J.; Elkin, E.P.; Konstan, M.W. Pulmonary exacerbations in cystic fibrosis: Young children with characteristic signs and symptoms. Pediatr. Pulmonol. 2013, 48, 649–657. [Google Scholar] [CrossRef]
- Brooks, D.; Thomas, J. Interrater reliability of auscultation of breath sounds among physical therapists. Phys. Ther. 1995, 75, 1082–1088. [Google Scholar] [CrossRef] [PubMed]
- Piirila, P.; Sovijarvi, A.R.A. Crackles: Recording, analysis and clinical significance. Eur. Respir. J. 1995, 8, 2139–2148. [Google Scholar] [CrossRef]
- Waller, M.; Stotler, C. Telemedicine: A Primer. Curr. Allergy Asthma Rep. 2018, 18, 54. [Google Scholar] [CrossRef] [PubMed]
- Mann, D.M.; Chen, J.; Chunara, R.; Testa, P.A.; Nov, O. COVID-19 transforms health care through telemedicine: Evidence from the field. J. Am. Med. Inform. Assoc. 2020, 27, 1132–1135. [Google Scholar] [CrossRef]
- Markwick, L.; McConnochie, K.; Wood, N. Expanding telemedicine to include primary care for the urban adult. J. Health Care Poor Underserved 2015, 26, 771–776. [Google Scholar] [CrossRef]
- McConnochie, K.M.; Wood, N.E.; Herendeen, N.E.; Ng, P.K.; Noyes, K.; Wang, H.; Roghmann, K.J. Acute illness care patterns change with use of telemedicine. Pediatrics 2009, 123. [Google Scholar] [CrossRef]
- Gupta, G. Are medical apps the future of medicine? Med. J. Armed Forces India 2013, 69, 105–106. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Ramos-Lacuey, B.; Herranz Aguirre, M.; Calderón Gallego, C.; Ilundain López de Munain, A.; Gembero Esarte, E.; Moreno-Galarraga, L. ECIEN-2020 study: The effect of COVID-19 on admissions for non-COVID-19 diseases. World J. Pediatr. 2021, 17. [Google Scholar] [CrossRef] [PubMed]
- Faezipour, M.; Abuzneid, A. Smartphone-based self-testing of COVID-19 using breathing sounds. Telemed. e-Health 2020, 26, 1202–1205. [Google Scholar] [CrossRef]
- Beaney, T.; Salman, D.; Samee, T.; Mak, V. Assessment and management of adults with asthma during the COVID-19 pandemic. BMJ 2020, 369. [Google Scholar] [CrossRef]
- Charbonneau, G.; Ademovic, E.; Cheetham, B.M.G.; Malmberg, L.P.; Vanderschoot, J.; Sovijärvi, A.R.A. Basic techniques for respiratory sound analysis. Eur. Respir. Rev. 2000, 10, 625–635. [Google Scholar]
- Rossi, M.; Sovijarvi, A.R.A.; Piirila, P.; Vannuccini, L.; Dalmasso, F.; Vanderschoot, J. Environmental and subject conditions and breathing manoeuvres for respiratory sound recordings. Eur. Respir. Rev. 2000, 10, 611–615. [Google Scholar]
- Sovijärvi, A.R.A.; Dalmasso, F.; Vanderschoot, J.; Malmberg, L.P.; Righini, G.; Stoneman, S.A.T. Definition of terms for applications of respiratory sounds. Eur. Respir. Rev. 2000, 10, 597–610. [Google Scholar]
- Fino, E.; Mazzetti, M. Monitoring healthy and disturbed sleep through smartphone applications: A review of experimental evidence. Sleep Breath. 2019, 23, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Sánchez Morillo, D.; Astorga Moreno, S.; Fernández Granero, M.Á.; León Jiménez, A. Computerized analysis of respiratory sounds during COPD exacerbations. Comput. Biol. Med. 2013, 43, 914–921. [Google Scholar] [CrossRef]
- Jácome, C.; Oliveira, A.; Marques, A. Computerized respiratory sounds: A comparison between patients with stable and exacerbated COPD. Clin. Respir. J. 2017, 11, 612–620. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, T.; Obase, Y.; Nagasaka, Y.; Nakano, H.; Kishikawa, R.; Iwanaga, T. Lung Sound Analysis and Airway Inflammation in Bronchial Asthma. J. Allergy Clin. Immunol. Pract. 2016, 4, 505–511. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, T.; Obase, Y.; Nagasaka, Y.; Nakano, H.; Ishimatsu, A.; Kishikawa, R.; Iwanaga, T. Lung sound analysis helps localize airway inflammation in patients with bronchial asthma. J. Asthma Allergy 2017, 10, 99–108. [Google Scholar] [CrossRef] [PubMed]
- Shimoda, T.; Obase, Y.; Nagasaka, Y.; Nakano, H.; Kishikawa, R.; Iwanaga, T. Lung sound analysis can be an index of the control of bronchial asthma. Allergol. Int. 2017, 66, 64–69. [Google Scholar] [CrossRef][Green Version]
- Kevat, A.C.; Kalirajah, A.; Roseby, R. Digital stethoscopes compared to standard auscultation for detecting abnormal paediatric breath sounds. Eur. J. Pediatr. 2017, 176, 989–992. [Google Scholar] [CrossRef] [PubMed]
- Almeida, R.; Jácome, C.; Martinho, D.; Vieira-Marques, P.; Jacinto, T.; Ferreira, A.; Almeida, A.; Martins, C.; Pereira, M.; Pereira, A.; et al. AIRDOC: Smart mobile application for individualized support and monitoring of respiratory function and sounds of patients with chronic obstructive disease. In Proceedings of the 12th IADIS International Conference e-Health 2020, EH 2020—Part of the 14th Multi Conference on Computer Science and Information Systems, MCCSIS 2020, Online, 21–25 July 2020; pp. 78–88. [Google Scholar]
- Reyes, B.A.; Reljin, N.; Chon, K.H. Tracheal sounds acquisition using smartphones. Sensors 2014, 14, 13830–13850. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. UroToday Int. J. 2009, 2. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, A.; Almeida, R.; Muchagata, J. How Secure Is Your Mobile Health? In Proceedings of the IFMBE Proceedings, Coimbra, Portugal, 26–28 September 2020; Volume 76, pp. 1377–1384. [Google Scholar]
- Adachi, S.; Nakano, H.; Odajima, H.; Motomura, C.; Yoshioka, Y. Lung sounds in children before and after respiratory physical therapy for right middle lobe atelectasis. PLoS ONE 2016, 11, e0162538. [Google Scholar] [CrossRef][Green Version]
- Pasterkamp, H.; Brand, P.L.P.; Everard, M.; Garcia-Marcos, L.; Melbye, H.; Priftis, K.N. Towards the standardisation of lung sound nomenclature. Eur. Respir. J. 2016, 47, 724–732. [Google Scholar] [CrossRef] [PubMed]
- Landis, J.R.; Koch, G.G. The Measurement of Observer Agreement for Categorical Data. Biometrics 1977, 33, 159. [Google Scholar] [CrossRef] [PubMed]
- Pramono, R.X.A.; Bowyer, S.; Rodriguez-Villegas, E. Automatic adventitious respiratory sound analysis: A systematic review. PLoS ONE 2017, 12, e0177926. [Google Scholar] [CrossRef]
- Melbye, H.; Garcia-Marcos, L.; Brand, P.; Everard, M.; Priftis, K.; Pasterkamp, H. Wheezes, crackles and rhonchi: Simplifying description of lung sounds increases the agreement on their classification: A study of 12 physicians’ classification of lung sounds from video recordings. BMJ Open Respir. Res. 2016, 3, e000136. [Google Scholar] [CrossRef] [PubMed]
- Earis, J.E.; Cheetham, B.M.G. Future perspectives for respiratory sound research. Eur. Respir. Rev. 2000, 10, 641–646. [Google Scholar]
- Aviles-Solis, J.C.; Jácome, C.; Davidsen, A.; Einarsen, R.; Vanbelle, S.; Pasterkamp, H.; Melbye, H. Prevalence and clinical associations of wheezes and crackles in the general population: The Tromsø study. BMC Pulm. Med. 2019, 19. [Google Scholar] [CrossRef] [PubMed]
- Murphy, R.L.H. In defense of the stethoscope. Respir. Care 2008, 53, 355–369. [Google Scholar] [CrossRef] [PubMed]
- Fainardi, V.; Longo, F.; Chetta, A.; Esposito, S.; Pisi, G. SARS-CoV-2 infection in patients with cystic fibrosi. An overwiew. Acta Biomed. 2020, 91, 1–5. [Google Scholar] [CrossRef]
- Taquechel, K.; Diwadkar, A.R.; Sayed, S.; Dudley, J.W.; Grundmeier, R.W.; Kenyon, C.C.; Henrickson, S.E.; Himes, B.E.; Hill, D.A. Pediatric Asthma Health Care Utilization, Viral Testing, and Air Pollution Changes During the COVID-19 Pandemic. J. Allergy Clin. Immunol. Pract. 2020, 8, 3378–3387.e11. [Google Scholar] [CrossRef]
- Murphy, R.L.H.; Vyshedskiy, A.; Power-Charnitsky, V.A.; Bana, D.S.; Marinelli, P.M.; Wong-Tse, A.; Paciej, R. Automated lung sound analysis in patients with pneumonia. Respir. Care 2004, 49, 1490–1497. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bettencourt, P.E.; Del Bono, E.A.; Spiegelman, D.; Hertzmark, E.; Murphy, R.L.H. Clinical utility of chest auscultation in common pulmonary diseases. Am. J. Respir. Crit. Care Med. 1994, 150, 1291–1297. [Google Scholar] [CrossRef] [PubMed]
- Spiteri, M.A.; Cook, D.G.; Clarke, S.W. Reliability of eliciting physical signs in examination of the chest. Lancet 1988, 331, 873–875. [Google Scholar] [CrossRef]
- Holleman, D.R.; Simel, D.L.; Goldberg, J.S. Diagnosis of obstructive airways disease from the clinical examination. J. Gen. Intern. Med. 1993, 8, 63–68. [Google Scholar] [CrossRef]
- Prodhan, P.; Dela Rosa, R.S.; Shubina, M.; Haver, K.E.; Matthews, B.D.; Buck, S.; Kacmarek, R.M.; Noviski, N.N. Wheeze detection in the pediatric intensive care unit: Comparison among physician, nurses, respiratory therapists, and a computerized respiratory sound monitor. Respir. Care 2008, 53, 1304–1309. [Google Scholar]
- Guggenmoos-Holzmann, I. The meaning of kappa: Probabilistic concepts of reliability and validity revisited. J. Clin. Epidemiol. 1996, 49, 775–782. [Google Scholar] [CrossRef]
| Characteristics | Total | Cystic Fibrosis | Other Respiratory Diseases | Asthma | No Respiratory Diseases |
|---|---|---|---|---|---|
| Subjects, n | 134 | 42 | 39 | 37 | 16 |
| Children, n (%) | 92 (69) | 16 (38) | 26 (67) | 34 (92) | 16 (100) |
| Male, n (%) | 72 (54) | 19 (45) | 20 (51) | 22 (60) | 11 (69) |
| Age, median [Q1–Q3] y | 16 [11–22.25] | 20 [12.75–30.50] | 16 [12–52] | 12 [9.5–16] | 14 [11.25–16.75] |
| Height, median [Q1–Q3] m | 1.59 [1.47–1.65] | 1.60 [1.53–1.68] | 1.57 [1.42–1.63] | 1.53 [1.39–1.62] | 1.63 [1.50–1.71] |
| With Quality | No Quality | Proportion (%) | ||
|---|---|---|---|---|
| Location | Trachea (n = 272) | 223 | 49 | 82 |
| Right Anterior Chest (n = 262) | 189 | 73 | 72 | |
| Right Posterior Base (n = 267) | 173 | 94 | 65 | |
| Left Posterior Base (n = 259) | 184 | 75 | 71 | |
| Age group | Children (n = 710) | 531 | 139 | 75 |
| Adults (n = 350) | 238 | 112 | 68 | |
| Diagnostic group | Cystic Fibrosis (n = 354) | 273 | 81 | 77 |
| Other Respiratory Diseases (n = 309) | 221 | 88 | 72 | |
| Asthma (n = 272) | 183 | 89 | 67 | |
| No Respiratory Diseases (n = 125) | 92 | 33 | 74 | |
| Total (n = 1060) | 769 | 291 | 73 |
| Adventitious Sounds | Present | Absent | Proportion (%) | |
|---|---|---|---|---|
| Location | Trachea (n = 223) | 30 | 193 | 13 |
| Right Anterior Chest (n = 189) | 31 | 158 | 16 | |
| Right Posterior Base (n = 173) | 27 | 146 | 18 | |
| Left Posterior Base (n = 184) | 20 | 164 | 11 | |
| Age group | Children (n = 531) | 69 | 462 | 13 |
| Adults (n = 238) | 39 | 199 | 16 | |
| Diagnostic group | Cystic Fibrosis (n = 273) | 18 | 255 | 7 |
| Other Respiratory Diseases (n = 231) | 63 | 158 | 29 | |
| Asthma (n = 183) | 18 | 165 | 10 | |
| No Respiratory Diseases (n = 92) | 9 | 83 | 10 | |
| Total (n = 769) | 108 | 661 | 14 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferreira-Cardoso, H.; Jácome, C.; Silva, S.; Amorim, A.; Redondo, M.T.; Fontoura-Matias, J.; Vicente-Ferreira, M.; Vieira-Marques, P.; Valente, J.; Almeida, R.; et al. Lung Auscultation Using the Smartphone—Feasibility Study in Real-World Clinical Practice. Sensors 2021, 21, 4931. https://doi.org/10.3390/s21144931
Ferreira-Cardoso H, Jácome C, Silva S, Amorim A, Redondo MT, Fontoura-Matias J, Vicente-Ferreira M, Vieira-Marques P, Valente J, Almeida R, et al. Lung Auscultation Using the Smartphone—Feasibility Study in Real-World Clinical Practice. Sensors. 2021; 21(14):4931. https://doi.org/10.3390/s21144931
Chicago/Turabian StyleFerreira-Cardoso, Henrique, Cristina Jácome, Sónia Silva, Adelina Amorim, Margarida T. Redondo, José Fontoura-Matias, Margarida Vicente-Ferreira, Pedro Vieira-Marques, José Valente, Rute Almeida, and et al. 2021. "Lung Auscultation Using the Smartphone—Feasibility Study in Real-World Clinical Practice" Sensors 21, no. 14: 4931. https://doi.org/10.3390/s21144931
APA StyleFerreira-Cardoso, H., Jácome, C., Silva, S., Amorim, A., Redondo, M. T., Fontoura-Matias, J., Vicente-Ferreira, M., Vieira-Marques, P., Valente, J., Almeida, R., Fonseca, J. A., & Azevedo, I. (2021). Lung Auscultation Using the Smartphone—Feasibility Study in Real-World Clinical Practice. Sensors, 21(14), 4931. https://doi.org/10.3390/s21144931








